Abstract
Summary
The inhibitory metabotropic glutamate receptor 3 (mGluR3) plays diverse and complex roles in brain function, including synaptic plasticity and neurotransmission. We recently found that mGluR3 is downregulated in the lateral septum (LS) of postpartum females using microarray and qPCR analysis. In this study, we used double fluorescence immunohistochemical approaches to characterize mGluR3 changes in LS of the postpartum brain. The number of mGluR3-immunoractive cells was significantly reduced in the dorsal (LSD) and intermediate (LSI) but not ventral (LSV) parts of the LS in postpartum versus virgin females. mGluR3 immunoreactivity in the LS was found predominantly in neurons (~70%), with a smaller portion (~20%–30%) in astrocytes. Colocalization analysis revealed a reduced mGluR3 expression in neurons but an increased astrocytic localization in postpartum LSI. This change in the pattern of expression suggests that mGluR3 expression is shifted from neurons to astrocytes in postpartum LS, and the decrease in mGluR3 is neuron-specific. Because mGluR3 is inhibitory and negatively regulates glutamate and GABA release, decreases in neuronal expression would increase glutamate and GABA signaling. Given our recent finding that ~90% of LS neurons are GABAergic, the present data suggest that decreases in mGluR3 are a mechanism for elevated GABA in LS in the postpartum state.
Introduction
Extensive behavioral, physiological and molecular adaptations occur in female mammals during the postpartum period to support maternal care (Neumann 2001; Russell et al. 2001; Kinsley 2008). Changes in neurotransmitter signaling (e.g., glutamate, GABA, oxytocin) are thought to contribute to these physiological events (Zhao et al. 2012a; Lonstein et al. 2014; Sabihi et al. 2014). Glutamate, the major neurotransmitter in the CNS that mediates excitatory synaptic activity, achieves its functional effects via binding and activating specific glutamate receptors. However, group II metabotropic glutamate receptors (mGluR2/3, mGlu2/3, or Grm2/3) represent a subtype of G-protein-coupled glutamate receptors that negatively modulate neurotransmission through presynaptic, postsynaptic and glial mechanisms (Cartmell and Schoepp 2000; Schoepp 2001; Tamaru et al. 2001). Recent progress in the field of mGluR2/3 research has attracted great interest in mGluR3 because of its promising therapeutic applications for the treatment of many psychiatric and neurological disorders, including anxiety, stress-related diseases and schizophrenia (Schoepp et al. 2003; Patil et al. 2007; Gray et al. 2012).
mGluR3 (also known as Grm3) is enriched in the mammalian brain, and highly abundant in the cerebral cortex, lateral septum (LS), dentate gyrus of the hippocampus, and basolateral amygdaloid nucleus (Ohishi et al. 1993; Tamaru et al. 2001). Differing from the exclusive localization of mGluR2 in neurons, mGluR3 is expressed in both neurons (Ohishi et al. 1993; Petralia et al. 1996; Shigemoto et al. 1997; Tamaru et al. 2001; Ferraguti and Shigemoto 2006) and glial cells (Condorelli et al. 1997; Wroblewska et al. 1998; Schools and Kimelberg 1999; Mudo et al. 2007). The dual localization of mGluR3 in neuronal and glial cell populations makes it a good modulator of glial-neuronal signaling (Winder and Conn 1996; Bruno et al. 1997).
Earlier studies show glutamate transmission is altered in the maternal brain. For example, glutamate transmission-related genes and their encoded proteins, including glutamate, glutamine, and glutamine synthetase, were found to be increased in the LS and cingulate cortex of postpartum females (Salmaso et al. 2011; Zhao et al. 2012b; Zhao and Gammie 2014). More recent data obtained from our laboratory indicate that mGluR3 mRNA is significantly downregulated in the medial prefrontal cortex (Eisinger et al. 2014), the nucleus accumbens (Zhao et al. 2014) and the LS (Eisinger et al. 2013) in the maternal brain. These initial results led us to question whether altered mGluR3 mRNA reflects protein level changes and how mGluR3 is expressed among different cell types in the maternal brain.
Although mGluR3 has been intensively studied, there is little information regarding its expression and roles in the postpartum brain. In this study, in order to understand how mGluR3 immunoreactivity changes in the postpartum brain and to characterize the phenotype of mGluR3-expressing cells, we performed double fluorescence immunohistochemistry along with neuronal and glial markers. We focused on mGluR3 in LS because LS is a key brain structure critically involved in behavioral, emotional, and stress responses (Sheehan et al. 2004; Lee and Gammie 2009; Singewald et al. 2011).
Materials & Methods
Subjects
Age-matched nulliparous adult female mice (~70 days of age) from outbred hsd:ICR strain (Mus domesticus) (Harlan; Madison, WI) were used. After a 4–5 day acclimation period in our colony, female mice were housed individually with a sexually experienced breeder male of the same strain for 2 weeks to ensure pregnancy. In the meantime, virgin females were pair-housed to provide similar timing of housing and isolation. After mating, all females (pregnant and virgin) were housed individually and provided precut nesting material until brain dissection. The subjects were maintained under a 12:12 hr light/dark cycle (lights on at 06:00 AM CST) and at a controlled temperature (~22°C) with ad libitum access to breeder chow (Harlan) and tap water. All procedures followed the guidelines of the National Institutes of Health Guide for the Care and Use of Laboratory Animals and were approved by the Animal Care and Use Committee of the University of Wisconsin.
Western Blotting
Western blot analysis was carried out to verify the specificity of mGluR3 antibody based on our published protocol, with a slight modification (Zhao et al. 2012a). Microdissected tissue from a virgin female was homogenized with ice-cold lysis buffer (1.0 ml per 100 mg of tissue) in pre-chilled centrifuge tubes. Following tissue homogenization, samples were centrifuged to sediment unbroken cells and nuclei. The supernatant fraction was collected, and protein concentration determined using BCA Protein Assay (Pierce Chemical Co.; Rockford, IL). Twenty µg of total protein was subjected to gel electrophoresis using a 10% Mini-PROTEAN TGX precast gel (Bio-Rad; Hercules, CA) and transferred to a PVDF membrane. Membranes were washed briefly in 0.1 M Tris-buffered saline containing 0.05% Tween 20 (TBST) and blocked for 1 hr in 0.1 M TBST containing 5% nonfat dry milk. The membrane was incubated with the primary antibody mGluR3 (SP4399P, diluted 1:1000; Acris Antibodies, Herford, Germany) in TBST containing 5% nonfat dry milk overnight at 4°C with agitation, and washed 3×5 min in TBST. Following washes, the membrane was incubated in an HRP-linked secondary antibody (goat anti-rabbit IgG, diluted 1:5000; Cell Signaling Technology, Beverly, MA) for 1 hr at RT, and washed in TBST. Immunoreactive bands were detected using a chemiluminescence kit (Amersham ECL Prime Western Blotting detection Reagent, GE Healthcare UK Ltd.; Buckinghamshire, UK), and the images were captured using C-Digit Blot Scanner (LI-COR Biosciences; Lincoln, NE).
Preparation of Brain Tissue Slices
The perfusion protocol used for double fluorescence immunohistochemistry has been described in our recent published work (Zhao et al. 2013). In brief, on the day of perfusion between 10:00 AM and 12:00 PM, postpartum (day 6 postpartum) and age-matched virgin mice were lightly anaesthetized with isoflurane, further deeply anesthetized with 0.15 ml of sodium pentobarbital, and then transcardially perfused with 4% paraformaldehyde in 0.1 M phosphate buffer (PB; pH 7.4). Brains were post-fixed overnight in the same fixative and then cryoprotected with 0.1 M PB containing 30% sucrose at 4°C for two days. Brains were snap-frozen and 30-µm coronal sections were sliced on a cryostat (CM1850; Leica Microsystems, Wetzlar, Germany) and stored in cryoprotectant solution at -20°C until processing. LS sections including the dorsal (LSD), intermediate (LSI) and ventral (LSV) subdivisions were collected from Bregma levels approximately from 1.045 to 0.02 mm according to The Allen Mouse Brain Atlas (reference atlas version 1, 2008) (Zhao et al. 2013). Immediately prior to perfusion, virgin females were examined for stage of estrus cycle and only diestrus females were included in this study. Diestrus virgin females served as controls, as most commonly used in previous similar studies (Chen et al. 1999; Mann and Bridges 2002), because the diestrus stage maintains a relatively low and consistent level of sex steroids (when estrogen starts to rise and progesterone is low) and avoids a proestrus peak of circulating estrogen and progesterone levels.
Double Fluorescence Immunohistochemistry with Tyramide Signal Amplification (TSA)
Double fluorescence labeling was carried out at room temperature unless otherwise indicated. Brain sections were washed 5×5 min in 0.1 M TBS to remove the cryoprotectant, and then incubated with 3% H2O2 in TBS for 30 min to inhibit endogenous peroxidase activity. Sections were rinsed with wash buffer (TBST) and incubated for 1 hr with blocking buffer (2% blocking reagent in TBS; Roche Applied Science, Indianapolis, IN). Sections were then incubated overnight with a mixture of two primary antibodies in blocking buffer: rabbit anti-mGluR3 (SP4399P, diluted 1:500; Acris Antibodies) and mouse anti-NeuN (MAB377, diluted 1:1000; Millipore, Bilerica, MA), or rabbit anti-mGluR3 and mouse anti-glial fibrillary acidic protein (GFAP; MAB360, diluted 1:1000; Millipore). After primary antibody incubation, sections were washed and incubated for 1 hr with HRP-conjugated goat anti-rabbit antiserum (Cell Signaling Technology; diluted 1:100 in TBST), washed 3×10 min with wash buffer, then incubated for 10 min in Cy3-conjugated tyramide (TSATM Plus Cyanine 3 kit, PerkinElmer, Waltham, MA; red for mGluR3 labeling) by diluting TSA stock solution 1:50 in 1× Amplification Diluent. After washing 3×10 min with wash buffer, sections were again incubated for 30 min with 3% H2O2 in TBS to quench peroxidase activity from the initial TSA reaction, and then incubated for 1 hr with HRP-conjugated horse anti-mouse antiserum (Cell Signaling Technology; diluted 1:100 in TBST). Sections were then washed 3×10 min with wash buffer and then incubated for 30 min in Alexa Fluor 488-conjugated tyramide (Molecular Probes, Eugene, OR; green for NeuN or GFAP labeling) by diluting TSA stock solution 1:100 in 1×Amplification reagent. Following washing 3×10 min with wash buffer, sections were mounted onto slides using DePeX mounting medium (Serva, Heidelberg, Germany), air-dried and stored in the dark at 4°C. As controls, quenching of HRP activity prior to incubation with Cy3- or Alexa Fluor 488-conjugated tyramide, or omission of the primary antibodies or HRP-conjugated secondary antibodies completely abolished corresponding fluorescence signals.
Quantitation of Double Fluorescence Labeling
All confocal fluorescence images were captured sequentially using an inverted Zeiss LSM 510 Meta laser scanning confocal microscope (Zeiss; Oberkochen, Germany). All images in each sample area were acquired with a screen resolution of 1024×1024 pixels using a 40× or 63× objective lens. For quantitative analysis of colocalization, cell counting was carried out using 40× magnification photomicrographs in three subdivisions (LSD, LSI and LSV) throughout the rostrocaudal extent of the LS (Zhao et al. 2013). The number of single-labeled cells identified by clearly stained somata was counted unilaterally in every third section within a 230.3 µm×230.3 µm unit area. Simultaneously, double-labeled cells were also enumerated based on the coincidence of green and red labeling in cell bodies (yellow), showing colocalized immunoreactivity (mGluR3 and specific cell markers NeuN or GFAP). The counting was performed manually by investigators blind to the labeling conditions. All confocal images were transferred to Adobe Photoshop 6.0 (Adobe Systems; San Jose, CA), with adjustments of brightness and contrast. Total data were collected from five animals per group for double labeling of mGluR3 and NeuN or GFAP.
Statistical Analysis
Statistical analyses were performed using SPSS 22.0 software (SPSS Inc., Chicago, IL). The number of mGluR3-immunoreactive cells and colocalization ratio were expressed as mean ± SEM and analyzed using an independent samples t-test and one-way ANOVA followed by Tukey tests post hoc comparisons. Overall level of statistical significance was set at p<0.05.
Results
Validation of mGluR3 Antibody
Affinity-purified polyclonal antibody to mGluR3 was raised in rabbit against the N-terminal extracellular domain of human mGluR3. The immunizing peptide showed 88% identity (94% similarity) with mouse and 94% identity (94% similarity) with rat according to the manufacturer’s specifications. The specificity of this antibody was tested using immunohistochemistry in cell lines without mGluR3, and was shown to be specific for mGluR3 (data provided by the vendor). In the present study, we performed additional validation of the anti-mGluR3 antibody using western blot analysis of mouse LS extract. First, the western blot assay revealed that the antibody recognized a single band of ~120 kDa (Fig. 1), which is in good agreement with the size of an mGluR3 antibody that has been demonstrated to be specific to mGluR3 and detect a band at (~116 kDa) (Tamaru et al. 2001). However, this size is slightly higher than the predicted molecular weight of mGluR3 protein (~100 kDa), probably reflecting posttranslational glycosylation of the receptor (Tanabe et al. 1992; Ohishi et al. 1994). Second, the distribution and immunostaining pattern in the mouse brain (data not shown) was very similar to that depicted in previous reports (Tamaru et al. 2001). Further, as controls, the specific labeling of cells was completely abolished in the absence of primary antibody and/or secondary antibody (data not shown), suggesting that the labeled immunoreactivity was specific to the combination of mGluR3 primary and secondary antibodies. All the above observations validate that the anti-mGluR3 antibody employed here binds specifically to the mGluR3 and does not cross-react with other glutamate receptor family members.
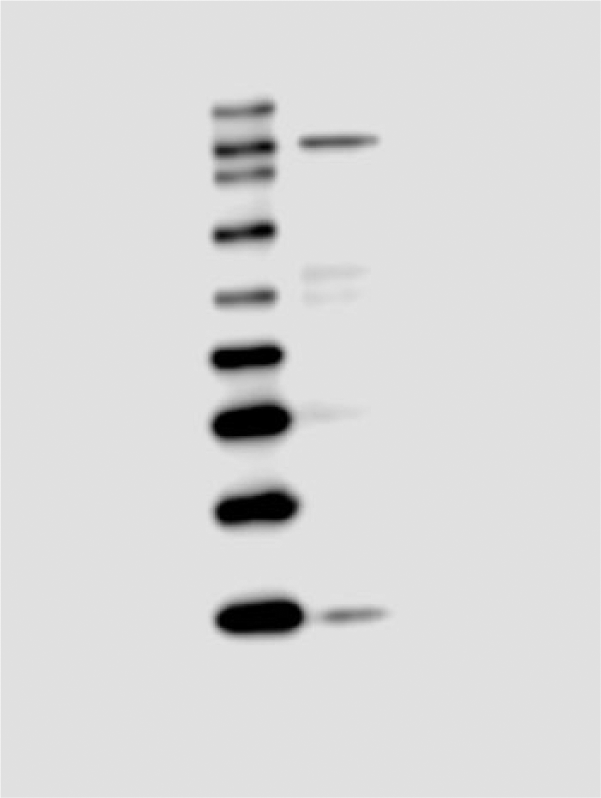
Western blot analysis showing the specificity of the mGluR3 (Grm3) antibody. The N-terminal antibody recognized a single, specific band at a molecular weight of ~120 kDa in protein extracts prepared from the microdissected lateral septum tissue of a virgin female mouse. Positions of the protein molecular weight standards in kDa are indicated on the left side.
Expression of mGluR3-immunoreactivity in LS
Lightly or intensely labeled mGluR3-immunoreactive cells were observed among multiple subregions of the LS (Figs. 2A, 2D, 3A, 3D). These cells were small-sized and round, with clearly stained cell bodies but no detectable cell processes. This staining pattern is somewhat different from a punctate immunoreactivity in neuropils with no apparently labeled cell bodies in a previous study (Tamaru et al. 2001), probably due to the dramatically enhanced intensity of signals by using the TSA plus fluorescence systems technology (Zhao et al. 2013). Notably, the morphology of the mGluR3-immunoractive cells in LS is largely different from that in the cerebral cortex, where medium- and large-sized cells with unambiguously labeled somal and dendritic profiles were observed (data not shown). Quantitative analysis showed that mGluR3-immunoreactive cells were unevenly distributed throughout the LS in virgin females. One-way ANOVA [F (2, 27) = 49.14, p<0.001] revealed a significant subregional difference in the density of mGluR3-immunoreactive cells. As illustrated in Figure 4A, the number of mGluR3-immunoractive cells in the LSV was lower than that in the LSD (p<0.001) and the LSI (p<0.001) in virgin mice, whereas no difference was observed between the LSD and LSI. By contrast, the number of mGluR3-immunoreactive cells did not differ among the three subregions of the LS in postpartum females (Fig. 4A). In parallel with the changes at the mRNA level (Eisinger et al. 2013), the number of mGluR3-immunoractive cells was significantly reduced in the LSD (Fig. 2A, 2D; p<0.001) and LSI (p<0.001) but not in LSV of postpartum relative to virgin females (Fig. 4A).
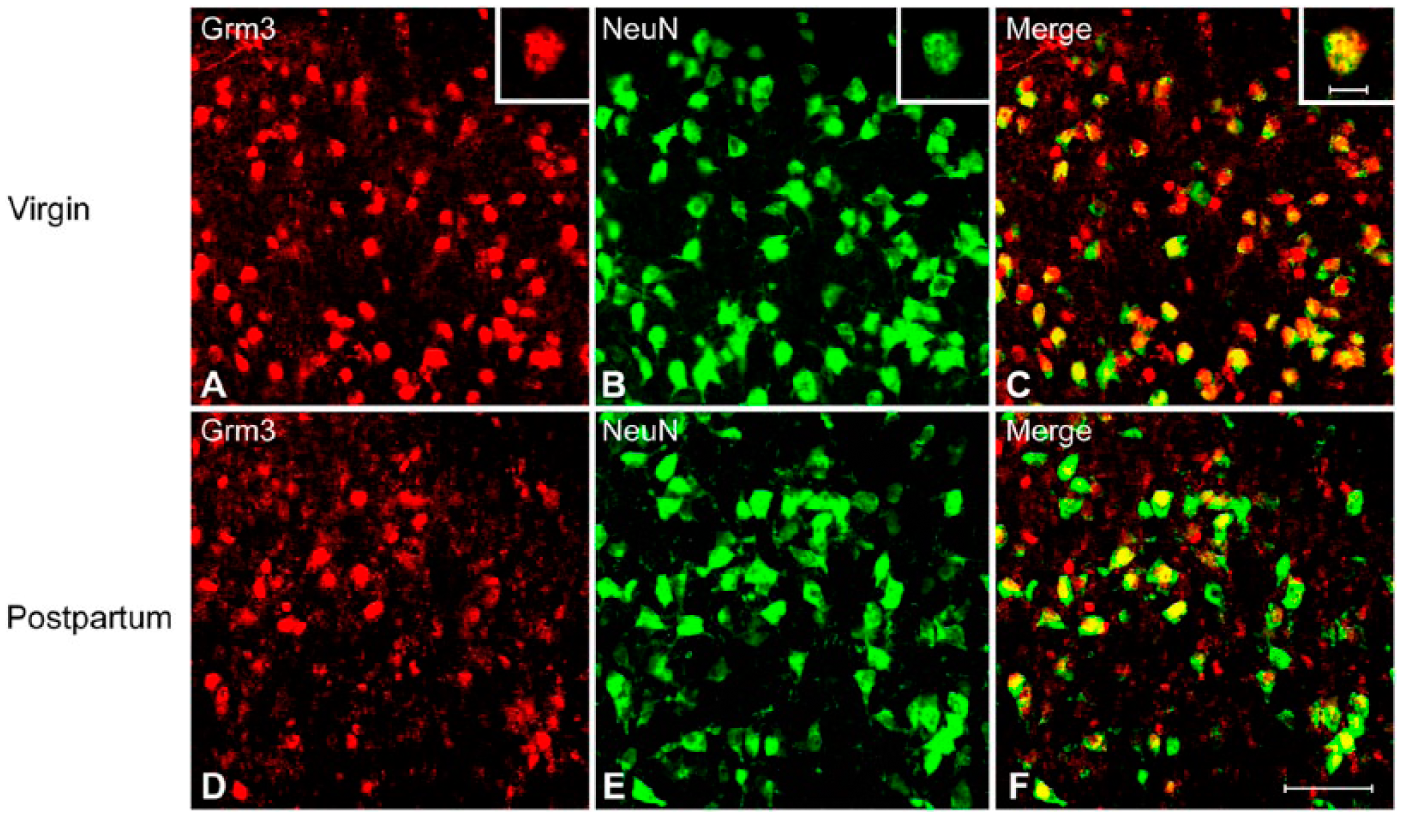
Representative photo-micrographs of double fluorescence immunohistochemistry labeling of cells expressing immunoreactivity for mGluR3 (Grm3) and NeuN in the dorsal lateral septum (LSD) of virgin (A–C) and postpartum (D–F) female mice. A typical example of double-labeled cell (inserts in A–C) is indicated. Scale (A–F) 50 µm; inserts, 10 µm.
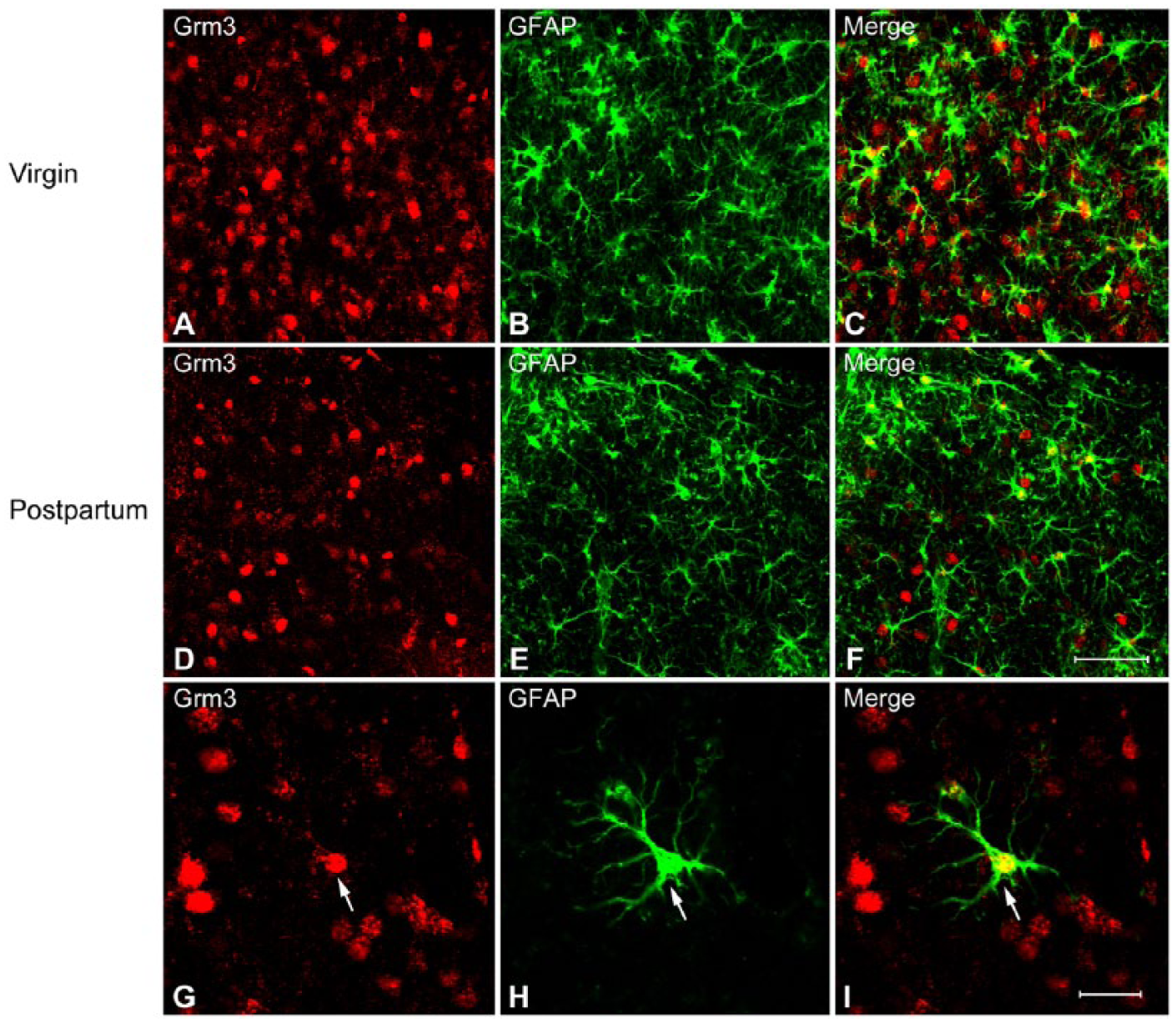
Representative photomicrographs of double fluorescence immunohistochemistry labeling of cells expressing immunoreactivity for mGluR3 (Grm3) and glial fibrillary acidic protein (GFAP) in the dorsal lateral septum (LSD) of virgin (A–C) and postpartum (D–F) female mice. A typical example of double-labeled cell (arrows) is indicated (G–I). Scale (A–F) 50 µm; (G–I) 20 µm.
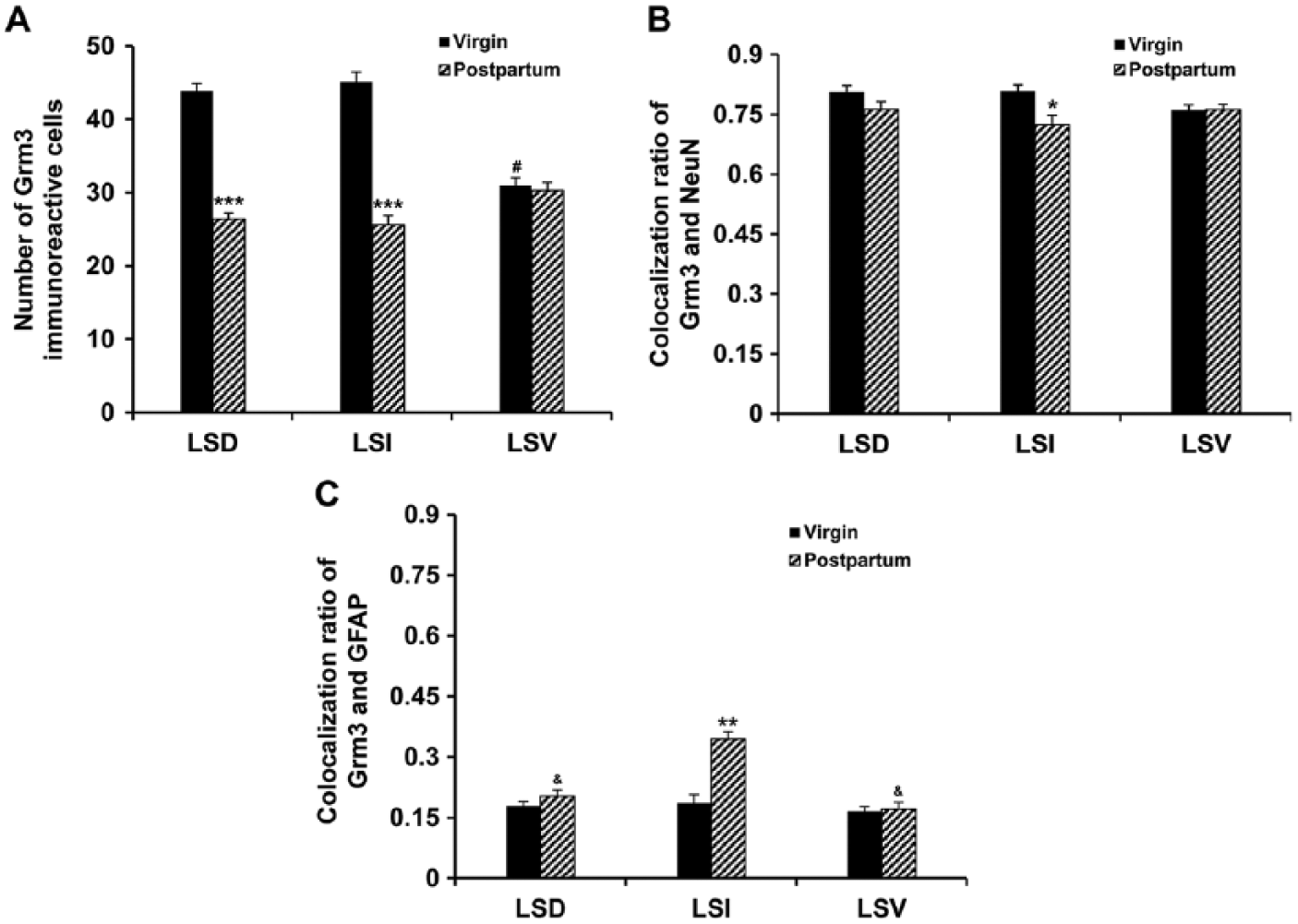
Expression and colocalization of cells expressing immunoreactivity for mGluR3 (Grm3) and NeuN or glial fibrillary acidic protein (GFAP) in the lateral septum. The number of mGluR3-immunoreactive cells (A) and colocalization ratio with NeuN (B) or GFAP (C) are presented. Each bar represents the mean obtained from five mice. Error bars are the SEM. The colocalization ratio was calculated by dividing the number of cells expressing both mGluR3 and NeuN or GFAP immunoreactivity (colocalization) by the total number of cells expressing mGluR3 immunoreactivity, and multiplying by 100. *p<0.05; **p<0.01; ***p<0.001 postpartum relative to virgin mice; #p<0.001 LSV relative to LSD and LSI; &p<0.05 relative to LSI.
Colocalization of mGluR3-immunoreactive Cells with NeuN in LS
To investigate the extent to which the mGluR3-immunoractive cells are neuronal, we performed double fluorescence immunohistochemistry with antibodies that recognize the mGluR3 and neuronal marker, NeuN, respectively. Double labeling revealed that approximately 70% of mGluR3-immunoreactive cells in each subregion within the LS of virgin mice were colocalized with NeuN (Figs. 2A–2C, 4B), suggesting that they are predominantly neurons. Further, the colocalization ratio of mGluR3 and NeuN (mGluR3/NeuN) was reduced in the LSI (p=0.016), whereas no changes occurred in LSD and LSV between the postpartum and virgin females (Fig. 4B).
Colocalization of mGluR3-immunoreactive Cells with GFAP in LS
In order to investigate the extent to which the mGluR3-immunoractive cells are glial cells, we used antibodies that recognize the mGluR3 and astrocytic marker, GFAP, respectively. In marked contrast to the NeuN, double labeling revealed only a small portion of mGluR3-immunoreactive cells (approximately 20%) within the LS of virgin mice with GFAP-immunoreactivity (Figs. 3A–3C, 4C), indicating that these mGluR3 cells are astrocytes. Unlike the virgin females, in postpartum mice more mGluR3 cells co-expressed GFAP in LSI (approximately 30%) than in LSD and LSV (Fig. 4C, p<0.05). An independent-samples t-test confirmed a significantly increased percentage of colocalization of mGluR3 with GFAP (mGluR3/GFAP) in the LSI (p=0.001) but not in the LSD and LSV of the postpartum mice as compared with virgin female mice (Fig. 4C).
Discussion
Building upon our recent work, the present study investigated mGluR3 immunoreactivity in the postpartum LS, and further characterized the phenotype of mGluR3-expressing cells with specific cellular markers. We found that the number of mGluR3-immunoractive cells was significantly reduced in the LSD and LSI but not LSV of postpartum versus virgin female mice. Further, we demonstrated a neuronal and astrocytic localization of mGluR3 in the LS of both the postpartum and virgin brains. Double labeling revealed that mGluR3-immunoreactive cells within the LS are predominantly neurons (~70%), whereas only a small portion (~20%–30%) are astrocytes. An analysis of the colocalization ratio showed a reduced neuronal expression coupled with an increased astrocytic expression in the LSI of the postpartum mice as compared with the virgin females. This change in expression pattern indicates that mGluR3 shifts from neurons to astrocytes in the maternal brain, and that reduced mGluR3 expression primarily occurs in neurons.
Identifying the regional brain distribution and phenotype of mGluR3 using immunohistochemistry has been problematic due to the antibodies used to recognize both mGluR2 and mGluR3 (Ohishi et al. 1994; Petralia et al. 1996). To circumvent the limitation of antibody specificity with immunohistochemistry, in situ hybridization for mGluR3 mRNA in combination with immunohistochemistry for specific cell type markers has been used to reliably identify mGluR3 in both neurons (NeuN-labeled) and glial cells, including astrocytes (GFAP-labeled) and oligodendrocytes (CNPase-labeled) (Mudo et al. 2007). Consistent with previous findings (Tamaru et al. 2001; Mudo et al. 2007), the present immunohistochemical assay with an mGluR3-specific antibody defined the presence of clearly labeled immunoreactivity of mGluR3 in both neurons (identified by NeuN) and astrocytes (identified by GFAP) of the LS. The expression of mGluR3 in LS is also supported by the finding that LY341495 (an antagonist of mGluR2/3)-evoked neuronal activity in LS was significantly reduced in mGluR3 knockout mice (Hetzenauer et al. 2008). Additionally, a downregulation in mGluR3 protein in postpartum LS agrees well with a reduced mRNA level (Eisinger et al. 2013).
mGluR3s are expressed presynaptically and postsynaptically. Presynaptic mGluR3 appears to primarily function to suppress the release of glutamate, providing a negative feedback to prevent excessive glutamate accumulation in the synaptic cleft, whereas postsynaptic mGluR3 may predominantly function to negatively modulate neuronal excitability and synaptic plasticity of glutamate neurons (Anwyl 1999; Poschel et al. 2005). Electrophysiological and biochemical studies have shown that neuronal mGluR3 also negatively regulates GABA signaling, as activation of the receptor inhibits GABA release from neurons (Anwyl 1999; Cartmell and Schoepp 2000; Zhao et al. 2001) and increases the expression of GABA receptor subunits (Ghose et al. 1997). For example, local infusion of a non-selective mGluR2/3 agonist LY379268 into the reticular nucleus of the thalamus, a neural site that is enriched with mGluR3 but not mGluR2, reduced GABA release (Gu et al. 2008). The GABAergic neurons are considerably abundant in the LS (Castaneda et al. 2005; Hrabovszky et al. 2012), and we found that ~90% of LS neurons are GABAergic (Zhao et al. 2013). Further, the vast portion of LS neurons are non-glutamatergic, as identified by a specific marker, vesicular glutamate transporter 2 (VGLUT2) for glutamatergic neurons in the septum-hypothalamus (Lin et al. 2003; Hrabovszky et al. 2012). As VGLUT2 is present in presynaptic axon terminals (Lin et al. 2003; Gritti et al. 2006), the low level of VGLUT2 indicates that mGluR3 appears to have little effect on the release of glutamate in LS. Together with the present observations that the majority of mGluR3-expressing cells within the LS are neurons (~70%), it is likely that mGluR3 is mainly localized in GABAergic neurons of the LS. This is further supported by our recent work showing that mGluR3 is largely colocalized with GABA in LS (Supplemental Fig. S1). Based on the aforementioned findings, it is expected that a downregulation of mGluR3 would primarily increase the extracellular level of GABA, and consequently contribute to elevating GABA signaling in the postpartum LS, as GABA signaling is upregulated in LS during the postpartum period (Zhao et al. 2012a; Zhao and Gammie 2014). We cannot rule out the possibility that downregulated mGluR3 may also increase glutamate release. Future study using pharmacological tools is needed to address how mGluR3 regulates glutamate and GABA signaling in the postpartum LS. It appears that the extent to which mGluR3 is expressed in neurons or glia can vary greatly mainly depending on distinct brain regions examined (Ohishi et al. 1993).
A large body of evidence has emerged in support of the involvement of glial mGluR3 in multiple physiological processes. Astrocytic mGluR3 regulates the expression of the glial glutamate transporter, solute carrier family 1, member 2 (Slc1a2, EAAT2, GLT-1) (Aronica et al. 2003; Lyon et al. 2008). As part of the glutamate/GABA-glutamine cycle, the uptake and release of glutamate from astrocytes through glial glutamate transporters regulate extracellular levels of glutamate and thus control glutamate neurotransmission (Rothstein et al. 1996; Hertz 2013; Schousboe et al. 2013). Activation of mGluR3 in astrocytes stimulates the production and release of neurotrophic factors, including transforming growth factor-β (TGF- β), nerve growth factor (NGF), brain-derived neurotrophic factor (BDNF), and glial-derived neurotrophic factor (GDNF) (Bruno et al. 1998; Ciccarelli et al. 1999; Battaglia et al. 2009; Di Liberto et al. 2010), which is involved in mGluR3-mediated neuroprotective activity (Bruno et al. 1998; Corti et al. 2007; Durand et al. 2008). The above observations favor a concept that the mGluR3-mediated neuronal effect involves an intimate crosstalk between neurons and glial cells (Winder and Conn 1996; Bruno et al. 1997). mGluR3 also participates in gliogenesis, as activation of mGluR3 favors the undifferentiated state and prevents astroglial differentiation (Ciccarelli et al. 1997; Ciceroni et al. 2010).
One striking finding of this study is the reduced neuronal expression of mGluR3 along with an increased astrocytic expression in the postpartum LS. This change in expression pattern indicates that mGluR3 expression is shifted from neurons to astrocytes in the maternal brain, and that the decrease of mGluR3 is neuron-specific. The lateral septal nucleus can be subdivided into three nuclei: the dorsal, intermediate and ventral parts, and this is done on the basis of chemoarchitecture and neuronal connections (Swanson and Cowan 1979; Alonso and Frotscher 1989). The intermediate part (LSI) is the largest and most heterogeneous subdivision of the LS. The LSI has close neural network connectivity with brain regions known to be involved in reproductive-, aggressive-, and mood-related behaviors, including regions of the medial preoptic area, the bed nucleus of the stria terminalis (BNST), the lateral hypothalamus (LH), the periaqueductal gray, the amydgala, the premammillary nucleus, and the paraventricular nucleus (PVN) (Risold and Swanson 1997; Sheehan et al. 2004). The selective shift in mGluR3 expression from neurons to astrocytes in postpartum LSI may participate in these behavioral processes during the postpartum period. To date, it is not clear what functional role such a shift will have, but one possibility is that elevated mGluR3s in glial cells supports the significant glial changes in the postpartum state, including morphology, structure and function (Theodosis and Poulain 1984; Featherstone et al. 2000; Salmaso et al. 2005; Salmaso et al. 2011).
Group II metabotropic glutamate receptors (mGluR2/3) represent promising therapeutic targets for the treatment of psychiatric and neurological disorders, including anxiety, stress-related illnesses, and schizophrenia, since activation of mGluR2/3 leads to anxiolytic effects, reduced stress responses and significant improvements in both positive and negative symptoms of schizophrenia (Schoepp et al. 2003; Patil et al. 2007). The balance of glutamatergic excitation/GABAergic inhibition within the brain has been shown to be altered in pathological states, including depression, schizophrenia and Alzheimer’s Disease (Sanacora et al. 2004; Bak et al. 2006; Rose et al. 2013). A negative relationship between mGluR3 and glutamate has been observed, as the pharmacological activation of mGluR3 reduces the presynaptic and glial release of glutamate, while blockade of mGluR3 increases extracellular glutamate levels (Battaglia et al. 1997; Moghaddam and Adams 1998; Cartmell and Schoepp 2000; Xi et al. 2002a; Xi et al. 2002b). We recently found a synchronously enhanced glutamate and GABA signaling in postpartum LS (Zhao et al. 2012a; Zhao and Gammie 2014); thus, it is logical to speculate that the elevated GABA transmission mediated by mGluR3 may be involved in sustaining the balance of glutamate/GABA that is properly coordinated in the maternal brain (Zhao and Gammie 2014).
A technical limitation in this study is that neurons and glial cells were counted without using unbiased stereological methods. Since neurons are usually larger than glial cells, and thus likely extending farther in the z-axis, these cells might be over-sampled as compared with glia in single sections. Thus, the proportion of double-labeled neurons might be inflated relative to the number of double-labeled glia. Similarly, as pregnancy, parturition and lactation cause changes in size and shape of neurons or glia (Keyser-Marcus et al. 2001; Theodosis and Poulain 2001; Kinsley et al. 2006), these possible changes might affect the proportion of double-labeled cell profiles that were observed.
Our observations demonstrate that, in the LS of both postpartum and virgin female mice, mGluR3-expressing cells are predominantly neurons rather than astrocytes. Furthermore, we found that the postpartum-associated decrease in mGluR3 expression is specific to neurons. We recently demonstrated that almost all neurons (>90%) in LS contain GABA (Zhao et al. 2013), and mGluR3 is present in LS GABAergic neurons (Supplemental Fig. S1); therefore, the selective decrease in mGluR3 in these GABA neurons would primarily lead to an increase in GABA signaling. Thus, the decrease in mGluR3 in neurons could be an important mechanism by which GABA signaling is elevated in postpartum LS. In future studies, it is worth determining the behavioral consequence of the downregulated mGluR3 in the postpartum LS.
Footnotes
Acknowledgements
The authors wish to thank Sarah Swanson from The Newcomb Imaging Center, Terri Driessen and Sharon Stevenson for excellent technical and administrative support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this research was provided by National Institutes of Health Grant R01MH085642 to Stephen Gammie.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
