Abstract
Immunoelectron microscopy using chromogranin A-specific antibodies has been proposed as an efficient technique for identification of secretory granules (SGs) in tumor cells with evidence of apparent neuroendocrine differentiation. Using an antigen retrieval (AR) method, we succeeded in immunolabeling SGs with antibodies in ultrathin sections of routinely processed epoxy-embedded blocks of tissue. Samples of an insulinoma were fixed in 2% glutaraldehyde, postfixed in 1% OsO4, and embedded in epoxy resin. Ultrathin sections were immunostained with chromogranin A-specific antibodies and gold-conjugated second antibodies. There was no significant labeling in the absence of AR. Neither etching with sodium metaperiodate nor microwave irradiation of ultrathin sections in citrate buffer (pH 6.0) or in EDTA buffer (pH 8.0) was effective in improving the efficiency of immunolabeling. However, ultrathin epoxy-embedded sections that were microwaved in alkaline solution (pH 10) were adequately labeled (5.2 ± 0.34 particles per SG). Moreover, considerably improved efficiency of immunostaining was achieved by microwaving sections in alkaline solution (pH 10) with subsequent immunostaining at 60C (12.2 ± 0.51 particles per SG). This method can also be applied to epoxy-embedded sections obtained from formalin-fixed, paraffin-embedded blocks of tissue and was even valid for an old epoxy-embedded block of tissue prepared 15 years previously.
Keywords
I
The goal of our study was to establish a method for the immunostaining of CGA on glutaraldehyde-fixed, epoxy-embedded ultrathin sections for identification of SGs. We examined etching and antigen retrieval (AR) techniques that involved microwaving epoxy-embedded sections in several kinds of buffer and compared the results with those for sections prepared in LR White resin. We tested techniques using epoxy-embedded sections of formalin-fixed, paraffin-embedded samples of tissue and even an old block of epoxy-embedded tissue that had been prepared 15 years previously to examine the validity of the methodology.
Materials and Methods
Tissue Samples
A small sample of a fresh pancreatic insulinoma, which had been obtained surgically, was cut into two pieces. One piece was immersed in 2% glutaraldehyde in 0.2 M phosphate buffer, pH 7.4, and the other was immersed in a mixture of 2% paraformaldehyde, 0.5% glutaraldehyde, 0.2 mM CaCl2, and 0.1 mM MgCl2 in 0.1 M sodium cacodylate buffer, pH 7.4, which was supplemented, just before use, with tannic acid at a final concentration of 0.1%. Both pieces were fixed in the respective fixative at 4C overnight. The former piece was postfixed in 1% OsO4 for 2 hr, dehydrated in an ascending graded series of ethanol and propanol, infiltrated with epoxy resin (Epok812; Okenshoji, Tokyo, Japan) in propanol, and finally embedded in Epok812 with DMP-30 (TAAB Laboratories; Berkshire, UK), which was polymerized by heating at 40C for 12 hr and then at 60C for 24 hr. The latter piece was dehydrated in an ascending graded ethanol series and infiltrated with LR White resin (London Resin Company; Berkshire, UK), which was polymerized by heating at 55C for 24 hr.
A tissue sample of surgically resected adrenal pheochromocytoma was fixed in formalin and embedded in paraffin. A small piece of the tumor was cut with a knife from the paraffin block, deparaffinized in xylene, rehydrated in ethanol, postfixed in 2% glutaraldehyde, processed as described above, and finally embedded in epoxy resin. A block of epoxy-embedded, glutaraldehyde-fixed, osmium-postfixed pancreatic islets, prepared 15 years previously, was also used in this study.
Ultrathin sections were cut with an ultramicrotome (Leica Instruments; Nussloch, Germany) at a thickness of 90 nm and mounted on nickel grids that had been coated with carbon by a vacuum evaporator.
Antigen Retrieval
Nickel grids with sections of LR White-embedded blocks were immersed in 10 mM citrate buffer, pH 6.0, and microwaved for 15 min in a domestic microwave oven (600 W). Before immunohistochemical staining, the grids in the citrate buffer were allowed to cool to room temperature (RT) for 30 min.
For epoxy-embedded ultrathin sections, we tested four methods of AR: etching; microwave irradiation in 10 mM citrate buffer, pH 6.0; in 1 mM EDTA buffer, pH 8.0; in an alkaline solution, pH 10 [Target Retrieval Solution (TRS); DAKO, Carpinteria, CA], which was diluted according to the manufacturer's instructions. The nickel grids for etching were placed on a droplet of 8% sodium metaperiodate to expose sections to the solution and then incubated for 1 hr at RT. The other grids were microwaved in citrate buffer, EDTA buffer, and TRS, respectively, as described above for ultrathin sections in LR White resin.
Immunostaining
After AR or without this step, all grids were immersed in blocking reagent (DAKO) for 10 min to prevent nonspecific binding of antibodies. Then CGA-specific rabbit antibodies (DAKO), diluted 1:100 in Tris-HCl buffer, pH 7.6, plus 0.8% NaCl and 0.1% bovine serum albumin (TBS-B), were applied to the sections with incubation for 30 min at RT. After washing in TBS-B with 0.1% Tween-20, sections were incubated with 15-nm gold particles that had been conjugated with goat antibodies against rabbit IgG (Amersham; Little Chalfont, Buckinghamshire, UK), diluted 1:10 in TBS-B, for 30 min at RT. After washing in TBS-B with 0.1% Tween-20, all sections were counterstained with uranyl acetate and lead citrate in the usual manner. The control sections that were stained with normal rabbit IgG, with a comparable protein concentration instead of the primary antibodies, showed negative results.
One grid each of LR White-embedded and epoxy-embedded ultrathin sections without AR and one grid each of epoxy-embedded ultrathin sections after AR were immunostained at 60C. Grids were placed in the gap between two ChemMate Capillary Gap microscope slides (DAKO), which had been prepared face-to-face in a slide holder as shown in Figure 1, and they were set in the heating unit of a Manual Capillary Action System (Microprobe; Fisher Scientific, Pittsburgh, PA). This system is designed for rapid immunostaining and in situ hybridization of samples for light microscopy. The blocking reagent, primary antibodies, second antibodies and buffer were absorbed at and eluted from the edge of the slides. Grids were incubated with CGA-specific antibodies, and second antibodies in this system for 30 min at 60C.
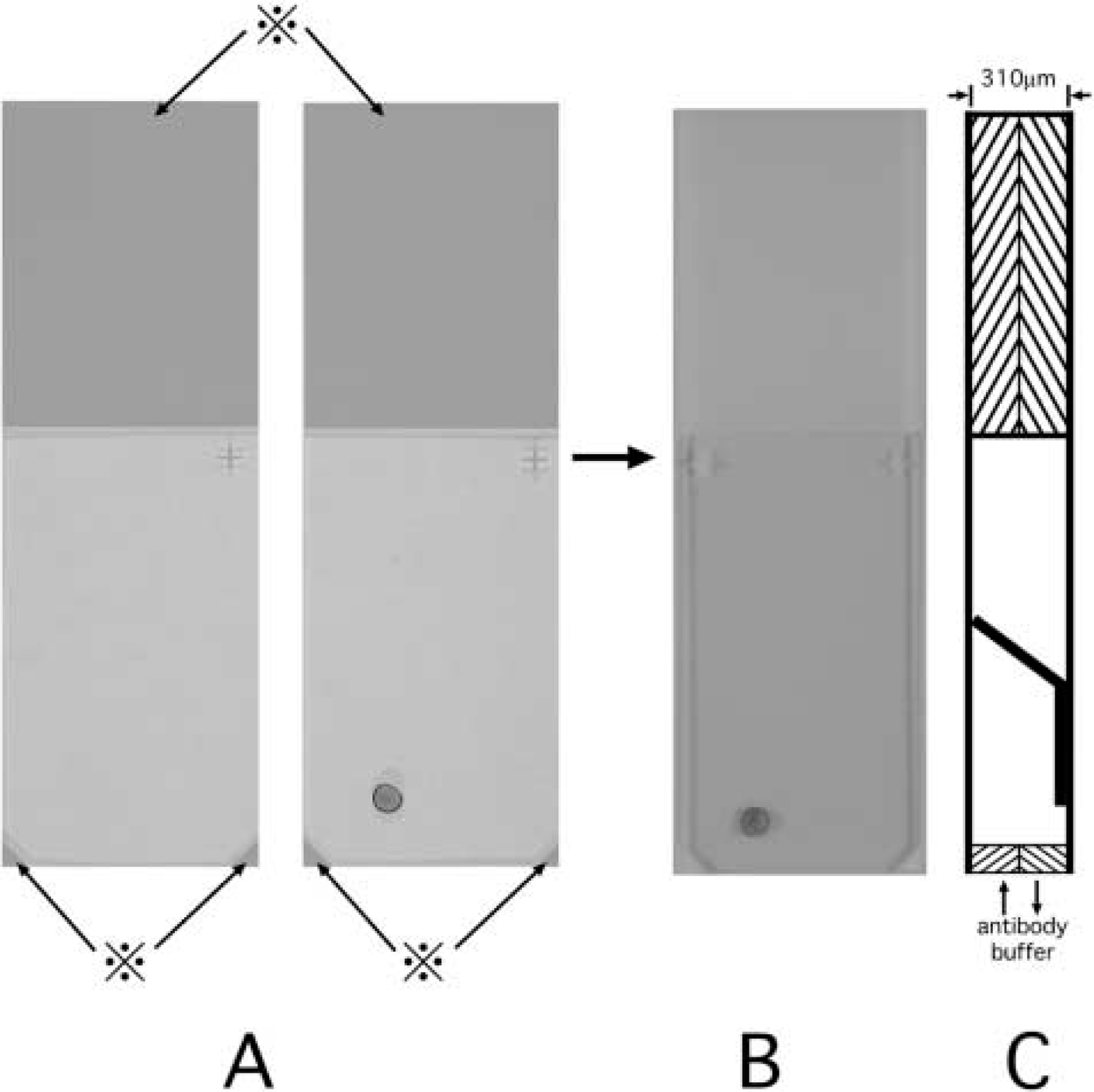
Manual capillary action system. A nickel grid was placed on a ChemMate Capillary Gap Microscope Slide (DAKO) with painted areas (⋇) of 155 μm in thickness (
The efficiency of immunolabeling was estimated in each case by counting the gold particles on 200 SGs. Statistical differences were evaluated by the Student's t-test (p<0.01).
Results
Effects of AR and High-temperature Incubation with Antibodies
The SGs in LR White-embedded sections were specifically labeled with 7.1 ± 0.02 particles per SG (n=200) without microwaving (Figure 2A). The number of particles on SGs was increased by microwaving in citrate buffer with incubation at RT, and by incubation with antibodies at 60C without microwaving (Figure 2B) to 11.4 ± 0.02 and 9.8 ± 0.02 particles per SG (n=200), respectively.
Results for AR techniques and epoxy-embedded ultrathin sections are summarized in Table 1. Sections without pretreatment and with microwave irradiation in citrate buffer (pH 6.0) or EDTA (pH 8.0) showed no significant labeling of SGs irrespective of whether antibodies had been allowed to reach at RT or at 60C (Figures 2C and 2D). Etching did not enhance the efficiency of immunolabeling, and only a few sporadic particles were seen on SGs after incubation at 60C. With TRS (pH 10) and microwaving, significant labeling of SGs with gold particles was evident even at RT (Figure 2E). The number of particles was increased considerably by incubation with antibodies at 60C (Figure 2F), as in the case of LR White-embedded sections that were microwaved in citrate buffer. The immunoreaction was specific, and nonspecific labeling of nuclei and organelles other than SGs was rarely observed.
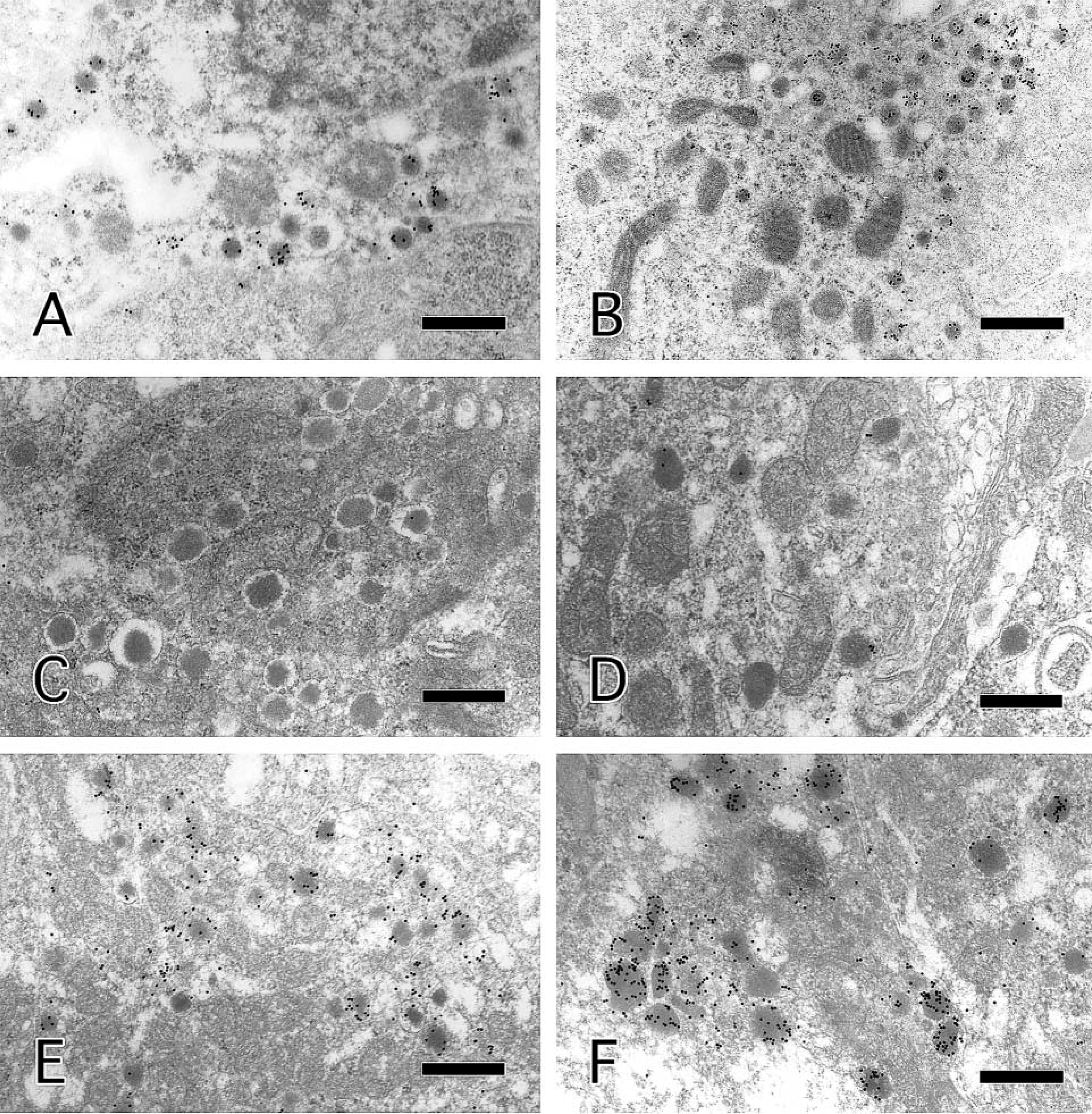
Results for the insulinoma of immunostaining and electron microscopy. Secretory granules in LR White-embedded sections were immunolabeled at RT without microwaving (
Results of immunostaining of LR-White-embedded and epoxy-embedded ultrathin sections
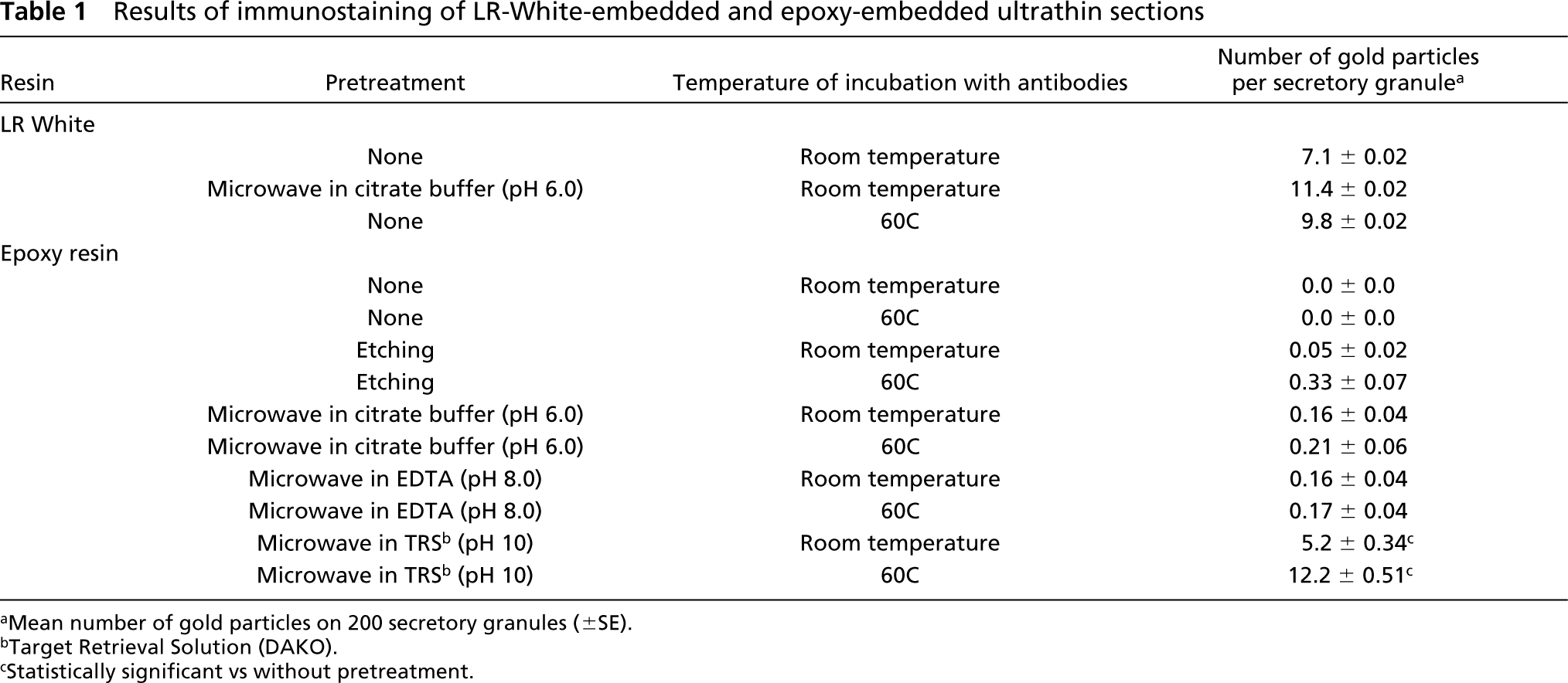
aMean number of gold particles on 200 secretory granules (±SE).
bTarget Retrieval Solution (DAKO).
cStatistically significant vs without pretreatment.
Immunoelectron Microscopy of Paraffin Sections and Old Epoxy Sections
An epoxy-embedded ultrathin section obtained from a block of formalin-fixed, paraffin-embedded pheochromocytoma was examined by the most efficient procedure identified above. The section was microwaved in TRS for 15 min and then immunostained at 60C. Subsequent electron microscopy revealed that many SGs had been labeled with more than a few gold particles (Figure 3A).
We obtained two epoxy-embedded blocks of pancreatic islets prepared 15 years previously. One had been detached from the plastic capsule and trimmed to expose the surface of the tissue at that time, and the other had been left, after polymerization, in a plastic capsule for 15 years. We prepared ultrathin sections of both blocks and immunostained them in the same way as the pheochromocytoma. The first block, which had already been trimmed, did not show significant labeling of SGs (Figure 3B). However, the SGs in the latter block, which had been kept in its plastic capsule, were effectively immunolabeled with gold particles (Figure 3C).
Discussion
There have been several studies aimed at identifying SGs by electron microscopy after immunostaining for CGA. In the case of epoxy-embedded samples, etching with sodium metaperiodate or hydrogen peroxide has been applied to ultrathin sections (Dardick et al. 1993; Herrera et al. 1993; Silver et al. 1993). Silver et al. (1993) showed that immunolabeling of epoxy-embedded sections after etching was less specific than that of Lowicryl-embedded sections. We confirmed that etching with sodium metaperiodate gave unsatisfactory results in immunostaining for CGA.
Antigen retrieval with microwaving is known to be effective for improving the efficiency of immunostaining at the light microscopic level. Sormunen and Leong (1998) applied this technique to LR White-embedded sections using citrate buffer (pH 6.0) and succeeded in improving the immunolabeling of cytokeratin, vimentin, type IV collagen, and β-catenin. Lowicryl-embedded sections were also examined by this method, and it was clear that citrate buffer and EDTA solution (pH 8.0) were useful for AR of cytokeratin in hepatocytes (Xiao et al. 1996). Röcken and Roessner (1999) examined the effects of etching and of heating in distilled water, citrate buffer, or EDTA solution at 91C for 30 min on conventionally fixed and epoxy-embedded amyloid deposits. They found that heating in EDTA solution gave the best enhancement of immunolabeling with AA amyloid-specific and κ light chain-specific antibodies. In our study, neither citrate buffer nor EDTA solution was effective in enhancing the immnostaining of CGA in epoxy-embedded ultrathin sections. However, microwaving in alkaline solution (pH 10) markedly enhanced the immunostaining of CGA. The alkaline solution, TRS, contains some detergent and was developed for immunostaining and light microscopic analysis of antigens that cannot be satisfactorily retrieved in citrate buffer. Antigen retrieval at high pH, around pH 9.0, was proved to be optimal for many antigens in section for light microscopic studies (Shi et al. 1995; Evers and Uylings 1997). Alkaline solution (citrate buffer with pH 9.0 and pH 12.0) for AR on epoxy sections was reported earlier by Brorson (2002), but this report does not include osmium-fixed tissue or incubation with immunoreagents at 60C. The combination of alkaline solution for AR, osmium fixation, and antibody incubation at 60C, as used in our study, is, as far as we know, original and unique.
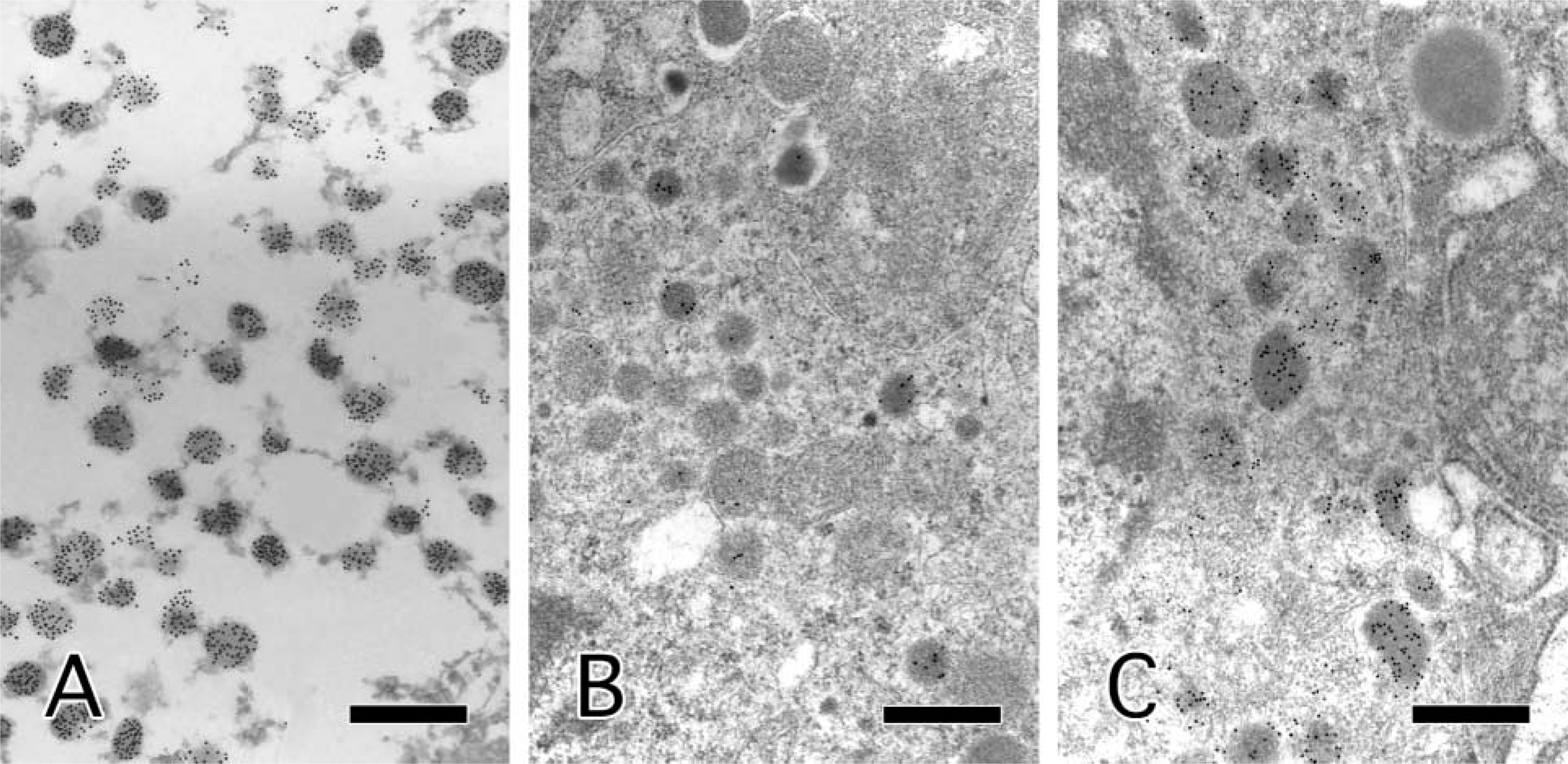
Results for the pheochromocytoma and pancreatic islets of immunostaining and electron microscopy. The formalin-fixed, paraffin-embedded pheochromocytoma was effectively immunolabeled (
Stirling and Graff (1995) showed that a combination of etching by sodium metaperiodate and heating in citrate buffer was more effective than heating in citrate buffer alone or etching alone. Groos et al. (2001) also reported that a combination of etching for 10 sec in 50% sodium ethoxide and subsequent heating in citrate buffer at 95C for 10 min provided reliable results on epoxy-embedded sections. In our preliminary study, we tested the combination of etching and heating in a microwave oven. However, most of our ultrathin sections became too fragile as a result of this procedure. Heating in TRS, citrate buffer, or EDTA solution for 15 min, as tested in the present study, did not damage the ultrathin sections, and ultrastructural morphology was well-preserved in all sections.
The temperature of incubation with antibodies is also important in immunolabeling that involves AR. In fact, a high temperature of incubation (60C) with antibodies of LR White-embedded sections gave almost identical results to microwaving in citrate buffer. The SGs of epoxy-embedded sections that had been treated with TRS and incubated with antibodies at RT were specifically immunolabeled but somewhat less effectively than LR White-embedded sections without AR. Then we chose 60C as the temperature for incubation with antibodies of TRS-treated epoxy-embedded sections and we achieved strong immunolabeling, as good as that of LR White-embedded sections, after microwaving in citrate buffer. Nonspecific binding of antibodies to other organelles, such as nuclei and mitochondria, was rare. Therefore, AR in TRS and subsequent immunostaining at 60C was effective for the detection of SGs in conventional epoxy-embedded sections and the specificity of the method was adequate for practical application.
Occasionally paraffin-embedded tissues are utilized for electron microscopy when properly fixed samples for electron microscopy are unavailable. The fixation of formalin-fixed, paraffin-embedded tissues is usually insufficient to preserve the details of ultrastructure, as shown in Figure 3A. Immunostaining for CGA and electron microscopy allows identification of SGs even when these “membrane-bound dense-core granules” are hard to visualize because of poor fixation.
We examined an epoxy-embedded block that had been prepared and trimmed to expose the surface 15 years previously, but its immunoreactivity had decreased during storage. However, an epoxy-embedded block of the same sample, prepared at same time but kept in a plastic capsule after polymerization, still immunoreacted with the CGA-specific antibodies. If blocks have been well preserved, it is possible to use such old epoxy-embedded tissue for immunostaining and electron microscopy.
The manual capillary action system (Microprobe) used in this study was developed for immunostaining with light microscopy and in situ hybridization, and it has the advantage of a stable incubation temperature. For immunostaining of epoxy-embedded ultrathin sections, the humid chamber used for immunohistochemical staining and a conventional incubator could substitute for the manual capillary action system. The system has, however, other advantages, including shortening washing steps and facilitating the handling of grids. Before placing the grids between the two slide glasses, it helps to bend the edges of the grids so that they are well secured in this system.
In the present study, we confirmed that microwaving in TRS and immunostaining at 60C allows detection of CGA in glutaraldehyde-fixed and epoxy-embedded ultrathin sections. This method is useful for identification of SGs even in formalin-fixed and paraffin-embedded tissues and in old epoxy-embedded tissues. The application of this method to other antigens, such as peptide hormones and intermediate filaments, remains to be clarified in further studies.
