Abstract
ABC-type transport proteins, such as P-glycoprotein (P-gp), modify intracellular concentrations of many substrate compounds. They serve as functional barriers against entry of xenobiotics (e.g., in the gut or the blood-brain barrier) or contribute to drug excretion. Expression of transport proteins in the heart could be an important factor modifying cardiac concentrations of drugs known to be transported by P-gp (e.g., β-blockers, cardiac glycosides, doxorubicin). We therefore investigated the expression and localization of P-gp in human heart. Samples from 15 human hearts (left ventricle; five non-failing, five dilated cardiomyopathy, and five ischemic cardiomyopathy) were analyzed for expression of P-gp using real-time RT-PCR, immunohistochemistry, and in situ hybridization. Immunohistochemistry revealed expression of P-gp in endothelium of both arterioles and capillaries of all heart samples. Although P-gp mRNA was detected in all samples, its expression level was significantly reduced in patients with dilated cardiomyopathy. We describe variable expression of P-gp in human heart and its localization in the endothelial wall. Thus, intracardiac concentrations of various compounds may be modified, depending on the individual P-gp level.
I
Expression of P-gp in humans reveals a wide inter-individual variability. Both genetic and environmental factors have been identified that contribute to this variability. For example, Hoffmeyer and co-workers (2000) identified a mutation in the P-gp encoding gene MDR1, which affected the bioavailability of the P-gp substrate digoxin. The antibiotic rifampin is an effective inducer of P-gp and other ABC transporters (e.g., MRP2; Greiner et al. 1999; Fromm et al. 2000), and this effect is mediated by a PXR-binding DR4 motif in an upstream enhancer of MDR1 (Geick et al. 2001).
Substrates for P-gp exhibit a wide structural diversity and cover a wide range of therapeutic indications. Notably, many cardiovascular active compounds are subject to drug transport by P-gp. Digoxin has been unequivocally identified as a P-gp substrate by experiments in cell lines, in animals, and in humans (de Lannoy and Silverman 1992). The disposition of several β-blocking drugs (e.g., talinolol, pafenolol) is modulated by P-gp (Westphal et al. 2000). Moreover, cardiotoxic drugs such as anthracyclines (e.g., doxorubicin) are P-gp substrates (Fardel et al. 1997) and accumulate in cardiac tissue of mdr–/– mice (van Asperen et al. 1999). Therefore, expression of P-gp in human heart may alter the intracardiac concentrations and hence the effects of therapeutic agents and cardiotoxic drugs.
We therefore investigated the expression, localization, and genotype of P-gp in human heart using immunohistochemistry (IHC), in situ hybridization (ISH), real-time RT-PCR, and RFLP. Here we describe variable expression of P-gp in the endothelial wall of cardiac blood vessels. Our data point to reduced expression of P-gp in patients with dilated cardiomyopathy.
Materials and Methods
Tissue Samples
After approval from the local ethics committee, heart tissue samples were taken from excised heart left ventricle during orthotopic heart transplantation due to end-stage heart failure and were immediately frozen in liquid nitrogen or fixed in 4% paraformaldehyde. Of the 15 subjects, five suffered from ischemic cardiomyopathy (ICM) and five from dilated non-ischemic cardiomyopathy (DCM). Medical therapy of patients suffering from DCM and ICM consisted of digitalis, diuretics, nitrates, and angiotensin-converting enzyme inhibitors. Tissue samples from five non-failing hearts (NF), which were not transplanted for surgical reasons or blood group incompatibility, served as controls.
The frozen tissue was homogenized using a vibration grinding mill (Mikro-Dismembrator S; B. Braun Biotech, Melsungen, Germany) for standard RNA and DNA preparation protocols. Fixed tissue was embedded in paraffin for IHC or ISH.
MDR1 Genotype
Genomic DNA was prepared from 30 mg tissue using standard phenol/chloroform extraction. The genotype of each individual at the MDR1 exon 26 C3435T locus was determined using a polymerase chain reaction (PCR)-based restriction fragment length polymorphism (RFLP) assay according to Cascorbi et al. (2001). Briefly, after amplification of a 197-bp DNA fragment, the variant in exon 26 C3435T was discriminated in a subsequent restriction enzyme digest using Sau3AI. The PCR reactions were carried out in a Perkin-Elmer 9700 thermocycler. DNA fragments generated after restriction enzyme digestion were separated on a 3.5% agarose gel (low-temperature melting agarose:standard agarose 3:1). Restriction fragments were visualized after ethidium bromide staining of the agarose gel using a UV transilluminator (Kodak Digital Science Image Station 440CF; Eastman Kodak, Rochester, NY).
RT-PCR Analysis of MDR1
Total RNA was isolated from 50 mg frozen tissue homogenate using a guanidinium isothiocyanate extraction kit (PeqLab; Erlangen, Germany), and subsequent DNase treatment, followed by spin column purification (Strataprep Total RNA Miniprep Kit; Stratagene, Amsterdam, The Netherlands).
For real-time RT-PCR, 200 ng of total RNA was reverse-transcribed using random hexamers and the TaqMan Reverse Transcription Reagents (Applied Biosystems; Weiterstadt, Germany). RT-PCR of MDR1 and 18S rRNA was performed using the primers MDR1F 5′-TTCGCAACCCCAAGATCCTC-3′, MDR1R 5′-ACAATGGTGGTCCGACCTTT-3′, and the TaqMan probe 5′-6FAM-ATCCAGAGCCACCTGAACCACTGCT XTp, as well as TaqMan Ribosomal RNA Control Reagents (Applied Biosystems) for 18S rRNA and the TaqMan universal PCR mastermix (Applied Biosystems). PCR products were amplified (50C, 2 min; 95C, 10 min; followed by 40 cycles of 95C, 15 sec and 60C, 1 min) and analyzed on a real-time RT-PCR cycler (ABI Prism 7700; Applied Biosystems).
For relative quantification, fluorescence intensities were plotted against PCR cycle numbers. The amplification cycle displaying the first significant increase of the fluorescence signal was defined as threshold cycle (CT). The CT value of each sample was compared to the CT values of the standardization series, which consisted of cDNA from P-gp-overexpressing L-MDR1 cells (kindly supplied by Dr. A. Schinkel; The Netherlands Cancer Institute, Amsterdam). The ratio of relative copy numbers of MDR1 divided by those of 18S rRNA thus represents the expression level of P-gp coding MDR1 mRNA.
Conventional RT-PCR was performed as described previously (Westphal et al. 2000). PCR products for MDR1 mRNA (157 bp) and for glyceraldehyde 3-phosphate dehydrogenase (GAPDH; 110 bp) were separated on a 8% polyacrylamide gel, stained with VistraGreen (Amersham Pharmacia Biotech; Freiburg, Germany) and band intensities were analyzed using the Storm 840 Imager (Molecular Dynamics; Krefeld, Germany). The relative amount of MDR1 mRNA was expressed as the ratio of MDR1 signal to GAPDH signal.
ISH of MDR1
Non-radioactive ISH was performed in paraffin sections (7 μm) that had been fixed in 4% paraformaldehyde. Sections were rehydrated and permeabilized by pepsin digestion (750 μg/ml pepsin in 0.2 M HCl, 37C, 30 min) and then post-fixed (paraformaldehyde 4%, 20 min, 4C) and acetylated using 0.25% acetic anhydride in triethanolamine (0.1 M, pH 8.0, 15 min). After dehydration in ethanol (70, 95, 100%), sections were hybridized for 16 hr (56C) in a solution containing 25% formamide, 0.3 M NaCl, 10% dextran sulfate, 20 mM Tris-HCl, pH 7.5, 1 mM EDTA, pH 8.0, 1 × Denhardt's, 0.1 ng/ml herring sperm DNA, 0.5 mg/ml tRNA, 0.1 mg/ml polyuridylic acid, and 125 ng digoxigenin (DIG)-labeled MDR1 cRNA probe (position 471–763 of the human MDR1 cDNA). The corresponding sense cRNA probe served as negative control.
After washing with 50% formamide in 75 mM NaCl, 7.5 mM sodium citrate, pH 7.0, sections were incubated with RNase A followed by additional washing steps and incubation with blocking reagent (Roche; Mannheim, Germany). Bound riboprobe was visualized by incubation with alkaline phosphatase-conjugated anti-DIG antibody (Roche) and subsequent substrate reaction using 5-bromo-4-chloro-3-indolyl phosphate/nitroblue tetrazolium chloride.
IHC of MDR1
From each heart tissue sample, paraffin sections of 2.0 μm were prepared by standard methods. Staining was performed using the Ventana's NexES IHC Staining System (Ventana Medical Systems; Frankfurt, Germany). For immunostaining, the monoclonal anti-P-gp antibody JSB-1 (mouse, dilution 1:20; Alexis Biochemicals, Grünberg, Germany) was used. The secondary antibody was rabbit anti-mouse diluted 1:100 included in the ABC Detection Kit (Ventana Medical Systems). Renal tissue samples were used as positive controls.
For semi-quantitative evaluation the numbers of specifically stained capillaries in relation to the total numbers of capillaries were determined in DCM, ICM, and NF heart tissue sections.
Statistical Analysis
The amounts of specific mRNA for human P-gp were compared using the Mann-Whitney U-test;
Results
P-glycoprotein was detected by IHC in all heart tissue samples tested. Immunostained heart tissue sections revealed P-gp localization predominantly in endothelial cells of capillaries and arterioles (Figure 1A). Staining intensity showed wide inter-individual variability. Tissue samples from hearts with dilated cardiomyopathy exhibited less staining compared to ischemic cardiomyopathic or non-failing hearts (Figure 1B;
Real-time RT-PCR detected expression of MDR1-specific mRNA in all heart samples. As calculated by comparison with standard curves (MDR1, y = −2.1303x + 38.275; 18S rRNA, y = −2.434x + 28.009) generated from LMDR1 total cellular RNA, ratios of MDR1 to 18S rRNA copy numbers were 0.84 ± 0.22 (NF), 1.15 ± 0.62 (ICM), and 0.41 ± 0.13 (DCM). Patients with DCM had significantly lower MDR1 mRNA expression levels compared to non-failing hearts (
MDR1 C3435T genotype prevalence of the samples did not differ significantly from the known MDR1 frequency in whites (Cascorbi et al. 2001). One DCM subject was an MDR1 3435CC genotype carrier and two were 3435CT. Among ICM samples, one was CC, three were CT, and one was TT. Three of the NF samples had the CC and two the CT genotype. MDR1 genotype at the MDR1 exon 26 C3435T locus did not significantly affect P-gp expression or MDR1 mRNA levels, although a tendency to lower expression of P-gp in samples homozygous for the TT genotype was observed [CC 0.79 ± 0.32,
Discussion
Here we describe expression and localization of the ABC transporter P-gp in human heart. P-gp was detected at the mRNA and protein levels in all 15 left ventricular samples. Both IHC and ISH localize P-gp expression to cardiac arterioles and capillaries. We observed a wide interindividual variability of P-gp. Various factors may be responsible for this phenomenon. Several mutations were described in the P-gp encoding MDR1 gene, some of which had functional consequences (Hoffmeyer et al. 2000; Cascorbi et al. 2001). For example, the synonymous mutation C3435T in exon 26 resulted in reduced intestinal expression of P-gp which, in turn, increased the bioavailability of digoxin. In our study we did not detect an association of genotype and cardiac P-gp expression. Although a reduced expression in samples containing the TT genotype of the C3435T mutation was observed, this effect was not statistically significant, comparable to the work by Hoffmeyer and co-workers (2000). This observation is supported by recent results in placenta trophoblast, showing decreased P-gp expression in cases carrying G22677A/T which is, in turn, known to be linked to C3435T (Tanabe et al. 2001).
Moreover, the process of disease may affect individual expression of P-gp. In our study we observed lower expression in patients with DCM compared to ICM and NF. This result was consistent at both the protein and mRNA levels and cannot be attributed to drug therapy because there were no differences in drug therapy between DCM and ICM patients. Two different PCR approaches (conventional vs real-time) employing different housekeeping genes confirmed the observation of reduced P-gp in patients with DCM. Heart failure caused by DCM is believed to be mediated by persistent viral infection or autoimmunity, and a variety of antibodies directed against structural components of human heart have been identified. For example anti-β1-autoantibody treatment of isolated cardiomyocytes leads to decreased expression of β-adrenergic receptor protein and mRNA, similar to patients with DCM (Podlowski et al. 1998). Therefore, it appears possible that anti-P-gp antibodies may lead to reduced expression in patients with DCM.
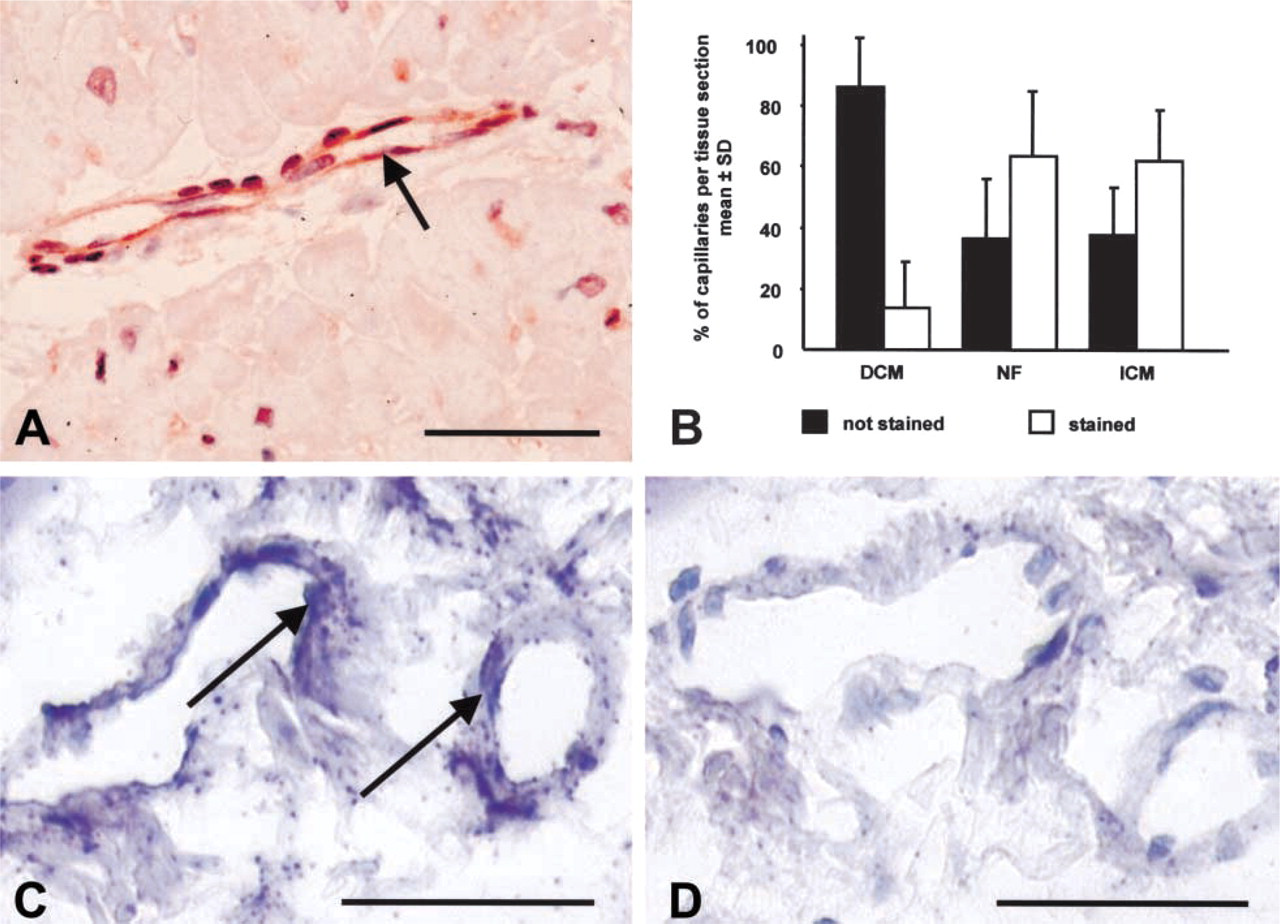
Immunohistochemistry and ISH of P-gp in paraffin sections from NF. (
Expression of P-gp in cells of human heart vessels is similar to that of P-gp in brain (Vogelgesang et al. 2001). The functional consequences of P-gp expression in the blood-brain barrier have been addressed in detail [restricted drug entry into the CNS (Schinkel et al. 1996)]. Therefore, we expect P-gp to serve as a functional barrier between blood and cardiac myocytes. High cardiac expression of P-gp can reduce uptake of drugs that are P-gp substrates into heart tissue and may therefore act as an important modulator of drug effects. Therefore, expression of P-gp in human heart can have important therapeutic implications. The cardiac glycoside digoxin has been unequivocally identified as a substrate for P-gp. The positive inotropic effects of digoxin exhibit a substantial inter-individual variability (Dobbs et al. 1987), and cardiac P-gp may be one factor contributing to this effect. Moreover, several β-blocking drugs have been described as P-gp substrates (e.g., talinolol, celiprolol; Karlsson et al. 1993; Westphal et al. 2000). Administration of β-blockers in patients with heart failure has been established as standard therapy and, again, therapeutic effects show a wide inter-individual variability, which may in part be explained by expression of drug transporters in human heart.
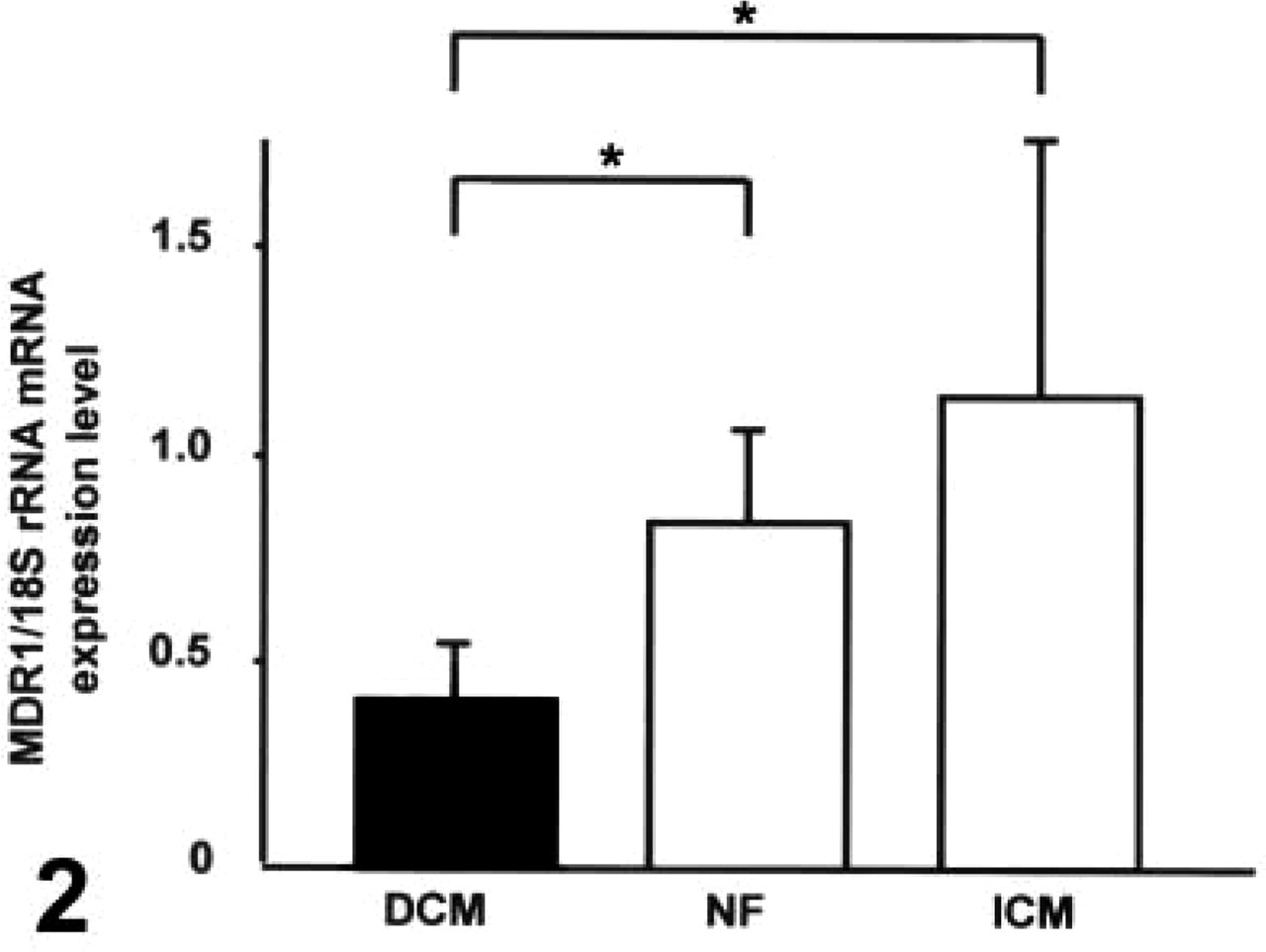
Analysis of P-gp mRNA expression in human heart tissue by real-time RT-PCR. Relative amount of P-gp mRNA determined as ratio P-gp:18S rRNA.
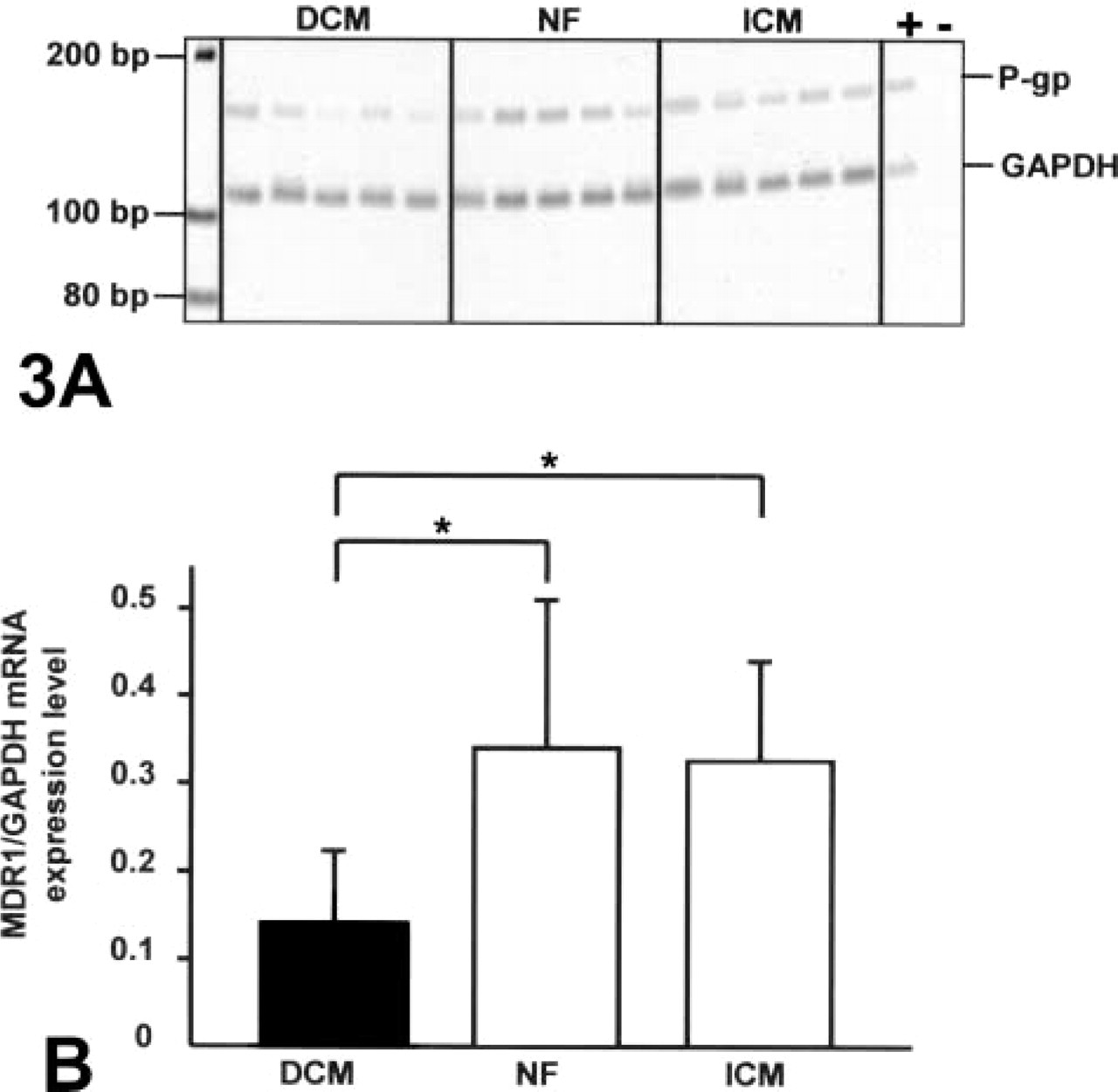
Analysis of P-gp mRNA expression in human heart tissue by conventional RT-PCR. (
Expression of P-gp may also protect heart tissue against cardiac toxicity of certain drugs. The anthracycline doxorubicin is a P-gp substrate [expression of P-gp in cancer cells results in chemoresistance to anthracyclines (Salmon et al. 1989)]. Doxorubicin is known to produce dose-dependent heart failure in patients treated for neoplastic disease (Goorin et al. 1990; Lipshultz et al. 1991) and is known to accumulate in hearts of mice lacking MDR1 (van Asperen et al. 1999). A recent study in children described a reduced ejection fraction after doxorubicin therapy, even with low cumulative doses (Agarwala et al. 2000). The authors observed substantial interindividual variability in cardiac toxicity of doxorubicin, a fact readily explainable by variable expression of P-gp.
In summary, we describe expression of the ABC-transporter P-glycoprotein in human heart. Our findings may have implications for drug therapy (alteration of both effects and toxicity). Moreover, our data point to reduced expression in patients with DCM. In view of recent data indicating cardiac expression of various cytochrome P450 enzymes (Thum and Borlak 2000) the contribution of the human heart to drug disposition should be evaluated.
Footnotes
Acknowledgments
Supported by research grants from the German Cardiac Society (Düsseldorf, Germany) to KM and from Apogepha GmbH (Dresden, Germany) and the Robert-Bosch-Foundation (Stuttgart, Germany) to IC.
