Abstract
Immunotherapy has considerable potential in the treatment of cancer. Here we report on the uptake of an antibody raised against the CCK-B/Gastrin receptor (CCK-BR) by liver embryonic and liver tumor cell lines. In all five cell lines studied, expression of CCK-BR and uptake of labeled anti-CCK-BR antibody was observed. The labeled anti-CCK-BR antibody was localized in both the cytoplasm and nucleus of cells. In addition, we found a coincidence between the uptake of the labeled antibody by cells and the occurrence of apoptosis (cell death). The results suggest that antibodies directed against CCK-BR have potential for targeting and possibly destroying tumor cells bearing the receptor.
T
Adoptive immunotherapy has been shown to lengthen the time between resection and recurrence in patients with HCC (Takayama et al. 2000). Identifying potential cellular targets is important in developing strategies for treating cancer with this technique.
Gastrin is a peptide hormone which, in addition to stimulating gastric acid secretion, is also now believed to play a role in tumor growth. In addition to normal gut mucosa and brain, the receptor for gastrin (CCK-B/gastrin) is expressed in tumor cells including hepatic, gastrointestinal, and pancreatic cancers (Reubi et al. 1997; Watson et al. 1998; Caplin et al. 1999b; Dockray 2000). These cancers also express gastrin and its precursor forms (Watson et al. 1998; Caplin et al. 1999b; Dockray 2000), suggesting that gastrin may act in an autocrine fashion to stimulate cell growth (Dockray 2000). We have previously shown that the CCK-B/gastrin receptor is present in HCC and have demonstrated the expression of precursor forms of gastrin suggesting the existence of an autocrine pathway in this condition (Caplin et al. 1999b). Gastrin and its precursor forms also promote growth of hepatocyte cell lines (Yao et al. 1995; Caplin et al. 2001). The specific CCK-B/gastrin receptor antagonist PD 135 caused inhibition of growth even in the absence of exogenous gastrin, supporting the idea of an autocrine loop (Caplin et al. 2001).
We have previously demonstrated that exposure of AR42J (pancreatic acinar carcinoma) cells to a labeled antibody (anti-CCK-BR) raised against the aminoterminus of the CCK-B/gastrin receptor results in translocation of the antibody, first to the cytoplasm and then to the nucleus (Savage et al. 1999). Nuclear localization of the CCK-B/gastrin receptor has been detected by immunoelectron microscopy (Caplin et al. 1999a). Administration of anti-CCK-BR antibody increases apoptosis and necrosis in human colon tumors growing in nude mice (Watson et al. 2000).
The aims of this study were to detect the CCK-B/gastrin receptor in hepatoma cells and to determine whether uptake of an anti-CCK-BR antibody was able to induce apoptosis in these cells.
Materials and Methods
Cell lines
HepG2 (human HCC), WRL68 (human liver embryonic), HTC (rat hepatocellular carcinoma), PLC/PRF/5 (human HCC), and MCA RH 7777 (rat HCC) cells were obtained from the European Collection of Cell Cultures (ECACC; Porton Down, UK). NIH3T3 cells were a gift from Professor T. Matsui (Kobe University; Kobe, Japan).
Anti-CCK-BR Antibody
The anti-CCK-BR antibody (Aphton Corporation; Woodland, CA) was raised as previously described (Caplin et al. 1999a). Briefly, the sequence of the immunizing peptide was KLNRSVQGTGPGPGASLSSPPPPC, comprising residues 5-21 of the CCK-B/gastrin receptor plus a seven-residue spacer. This was coupled to diphtheria toxin and injected into New Zealand White rabbits. Blood was collected after the third injection and the antibody was obtained from the serum by affinity purification.
Immunoblotting
Lysate corresponding to 10 μg protein was loaded onto 10% polyacrylamide minigels for SDS-PAGE. In addition to the liver cell lines, normal mouse fibroblast NIH3T3 cells were included as a negative control. After electrotransfer and overnight blocking, the membranes were incubated with 3 μg/ml anti-CCK-BR antibody. The antibody was used either untreated or had been preabsorbed for 48 hr with a molar excess of the immunizing peptide. After incubation with goat anti-rabbit/mouse biotin and streptavidin/biotin-peroxidase complex, labeling was revealed using diaminobenzidine (DAB) reagent.
Uptake and Apoptosis Studies
Anti-CCK-BR antibody was labeled with Alexa Fluor 488 fluorescent dye (Molecular Probes; Eugene, OR). Cells were cultured on chamber well slides and exposed to the labeled antibody (20 μg/ml) for 1 hr at 37C and fixed with buffered formaldehyde solution. Cells were then counterstained with a nuclear stain (propidium iodide) or for apoptosis using the TUNEL method.
For propidium iodide staining, slides were incubated with 100 μg/ml RNase for 90 min at 37C followed by 3 μg/ml propidium iodide (both in 2 × SSC) for 7 min.
Apoptosis staining used the TUNEL method (ApopTag Red in situ apoptosis detection kit; Intergen, Purchase, NY) which labels the 3′-OH ends of DNA fragments generated by apoptosis with chemically modified nucleotides. The latter are then detected using a secondary antibody conjugated to the fluorescent dye rhodamine. Images were obtained using either a confocal or fluorescence microscope with appropriate filters for the Alexa Fluor 488 and rhodamine/propidium iodide fluorescence.
Results
In all hepatocyte cell lines except negative control NIH3T3 (normal mouse fibroblasts), anti-CCK-BR immunoreactivity was found by blotting. Specific bands were distinguished by the absence of a corresponding band in the blots incubated with the preabsorbed anti-CCK-BR antibody. A common band at ∼162 kD was seen in all hepatocyte lines. Additional bands were seen at ∼137 kD in WRL68, HepG2, and PLC/PRF/5 cells and at ∼79 kD in the MCA RH7777 and HTC cells. A ∼183-kD band was also observed with the MCA RH7777 cells. No specific bands were seen with the NIH3T3 cells (Figure 1).
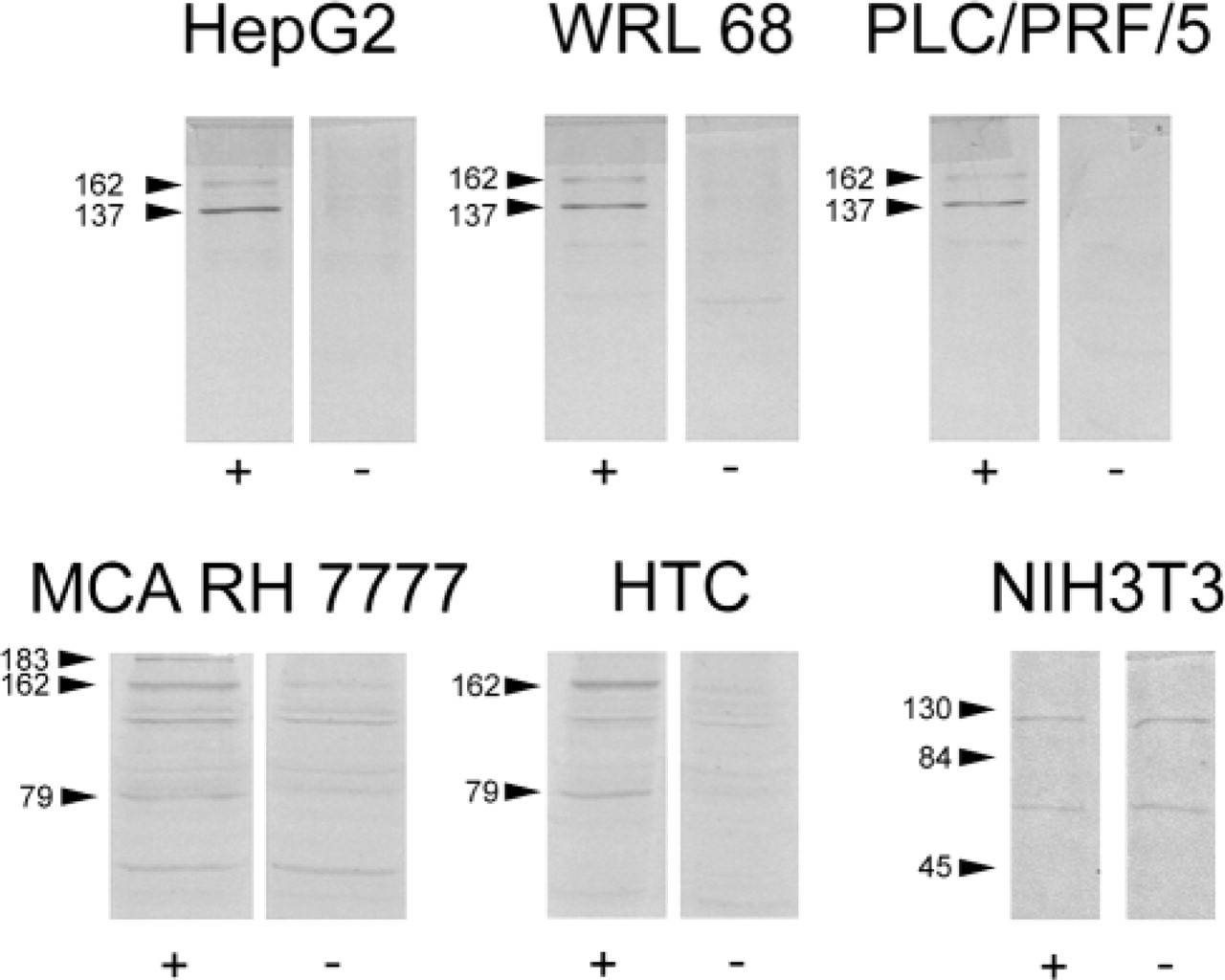
Immunoblot of lysates from HepG2, WRL 68, PLC/PRF/5, MCA RH 7777, HTC, and NIH3T3 cell lines using anti-CCK-BR antibody either untreated (left panels, +) or preabsorbed with the immunizing peptide (right panels, −). Arrows with molecular weights in kD alongside indicate specific bands (HepG2, WRL68, PLC/PRF/5, MCA RH 7777, HTC) or molecular weight markers (NIH3T3).
Immunofluorescence microscopy demonstrated uptake of anti-CCK-BR in all five cell lines studied (Figure 2). In cells that displayed uptake, nuclear localization of antibody was observed in nearly 100% of PLC/PRF/5, WRL68, Hep G2, and HTC cells but could not be confirmed in MCA RH7777 cells. Using the immunofluorescent TUNEL assay, cells that showed anti-CCK-BR uptake were also found to be positive for apoptosis (Figure 3). No uptake was seen in any of the cell lines studied when exposed to Alexa Fluor 488-labeled non-immune rabbit antibody alone.
Discussion
We have demonstrated that immunodetectable CCK-B/gastrin receptor is present in hepatic tumor and embryonic liver cells but not in normal fibroblasts. We have also shown that anti-CCK-BR antibody is internalized by these cells and that there was an association with apoptosis.
Watson et al. (1998) reported sizes of 40 and 70 kD for the CCK-B receptor using immunoblotting. Here we have found higher molecular weights for the receptor in the hepatocyte cell lines. This discrepancy could be due to differences in glycosylation of the CCK-B receptor molecule, resulting possibly from variation in cell lysate preparation. Larger CCK-B/gastrin receptor sizes of 74 and 115 kD have been reported by other workers (Matsumoto et al. 1987; Morisset et al. 2000).
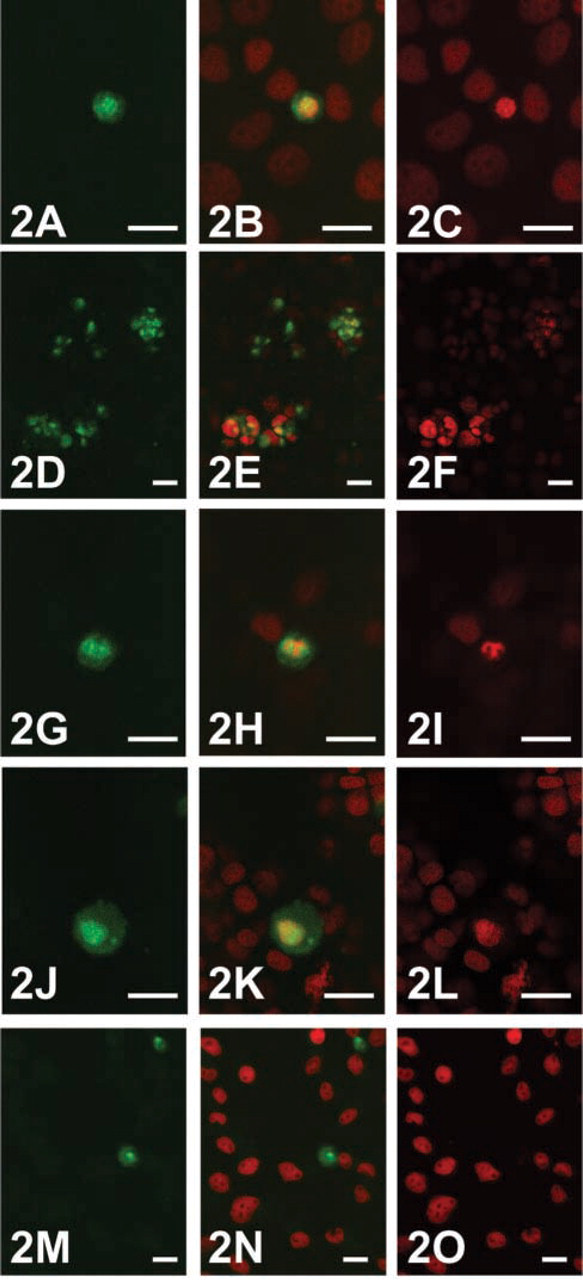
Fluorescent micrographs obtained using confocal laser scanning microscopy showing Alexa Fluor 488-labeled anti-CCK-BR antibody uptake (
Although we observed an association of uptake of labeled anti-CCK-BR antibody and apoptosis in all five hepatocyte cell lines examined, we did not observe nuclear uptake in the MCA RH7777 cells. This suggests that nuclear uptake of the antibody may not be essential for inducing apoptosis.
We do not know if the translocation to the nucleus involves the entire receptor or a fragment of the receptor. Ligand-induced internalization of the CCK-B receptor was demonstrated by Pohl et al. (1997). Deletion of the carboxyl terminus resulted in decreased endocytosis, as did mutation of serine and threonine residues in this region. However G-protein coupling and ligand-stimulated Ca2+ efflux were unaffected by mutation, suggesting that the internalization mechanism is distinct from that involved in stimulus secretion coupling.
Crosslinking of cell surface Fas molecules mediated by binding of either Fas ligand or anti-Fas antibody leads to apoptosis (Nagata and Golstein 1995; Wallach et al. 1999). By analogy, crosslinking of CCK-BR by antibody may be the trigger for apoptosis in the cell lines studied here. Alternatively, the occurrence of receptor endocytosis in the absence of ligand-induced secondary messenger production may be the signal for apoptosis. Recently, the existence of dependence receptors has been postulated in both cancer and neuronal development (Mehlen and Bredesen 2000). Such receptors have a dual function. In the presence of the appropriate ligand, they initiate a signal for proliferation or differentiation. In the absence of ligand, a signal for cell death by apoptosis is generated (Mehlen and Bredesen 2000). The association of antibody uptake and apoptosis demonstrated here could be explained by this hypothesis if CCK-BR existed as a dependence receptor in tumor cells. In this scenario, gastrin produced by the tumor cells would act in an autocrine fashion on the CCK-BR to cause proliferation. By contrast, binding of anti-CCK-BR antibody would mimic an absence of ligand and lead to cell death by apoptosis. If this hypothesis could be proved to exist in hepatic and other cancers, this could have major implications for their treatment.
In summary, this study has demonstrated CCK-B/gastrin receptor expression in hepatocyte cell lines and that binding and uptake of anti-CCK-BR antibody is associated with apoptosis. The therapeutic options for hepatocellular carcinoma are very limited and further studies are ongoing to determine whether the findings of this study can be applied in vivo and subsequently in human subjects.
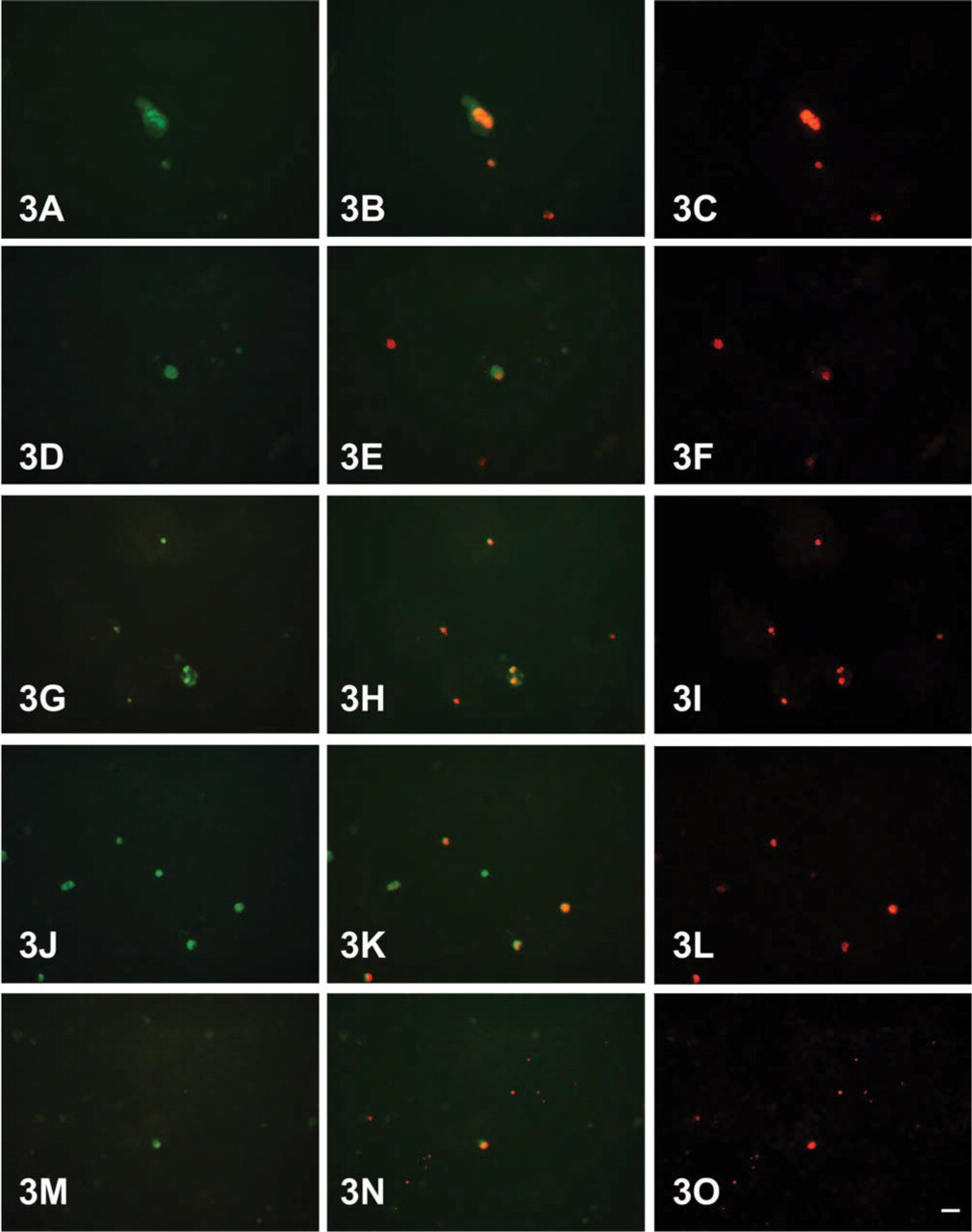
Fluorescent micrographs showing PLC/PRF/5 (
Footnotes
Acknowledgment
Supported by a research grant from Aphton Corporation.
