Abstract
Ornithine decarboxylase (ODC) is a key enzyme in polyamine biosynthesis. Increased polyamine levels are required for growth, differentiation, and transformation of cells. In situ detection of ODC in cells and tissues has been performed with biochemical, enzyme cytochemical, immunocytochemical, and in situ hybridization techniques. Different localization patterns at the cellular level have been described, depending on the type of cells or tissues studied. These patterns varied from exclusively cytoplasmic to both cytoplasmic and nuclear. These discrepancies can be partially explained by the (lack of) sensitivity and/or specificity of the methods used, but it is more likely that (sub)cellular localization of ODC is cell type-specific and/or depends on the physiological status (growth, differentiation, malignant transformation, apoptosis) of cells. Intracellular translocation of ODC may be a prerequisite for its regulation and function.
Keywords
O
Polyamines are multifunctional organic bases that play an essential role in cell growth, differentiation, and malignant development (Pegg 1986). Recent studies have shown that polyamines are actively involved in apoptosis (for review see Schipper et al. 2000). For a comprehensive overview of the biochemistry and physiology of polyamines, the reader is referred to the reviews by Pegg (1986), Williams (1997), Morgan (1999), and Wallace (2000) and to the books recently published by Cohen (1998) and Bardocz and White (1999). The biochemical and physiological features of ODC have aroused considerable interest in various fields of scientific research (see reviews by Russell 1985; Pegg 1986; Heby and Persson 1990; McCann and Pegg 1992; Hayashi and Murakami 1995; Hayashi et al. 1996; Shantz and Pegg 1999; Murakami et al. 2000).
The enzyme exhibits rapid and diverse changes in biological activity as an early response to virtually all growth-promoting stimuli to cells, including those of hormones, drugs, growth factors, mitogens, and tumor promoters (Russell 1985). Recent studies showed that overexpression of ODC in cooperation with other cellular events, e.g., activation of oncogenes or triggering of tumor promotion, can cause cell transformation, suggesting that the enzyme can be considered as a proto-oncogene product (Auvinen et al. 1992, 1997; O'Brien et al. 1997; Shantz and Pegg 1998; Tabib and Bachrach 1998).
The native enzyme is active as a homodimer and uses pyridoxal
ODC activity is controlled at the transcriptional, translational, and post-translational levels and is under strict negative feedback control by its polyamine products (Heby and Persson 1990). Rapid and dramatic regulation of ODC expression is possible because of its extremely short half-life of several minutes to 1 hr (the shortest half-life of any known enzyme). Its degradation is regulated by a unique regulatory protein named antizyme, which is induced by polyamine-mediated translational frame shifting (for reviews see Hayashi et al. 1996; Coffino 2001). Antizyme binds reversibly to monomers of ODC and promotes its proteolytic degradation by the 26S proteasome. Antizyme also downregulates polyamine uptake by cells. Activity of antizyme is regulated by another unique protein. This antizyme inhibitor is highly homologous to ODC but lacks its activity completely and is believed to prevent ODC degradation by trapping the antizyme (Murakami et al. 1996). The enzymatic features of the ODC/polyamine system are schematically summarized in Figure 1.
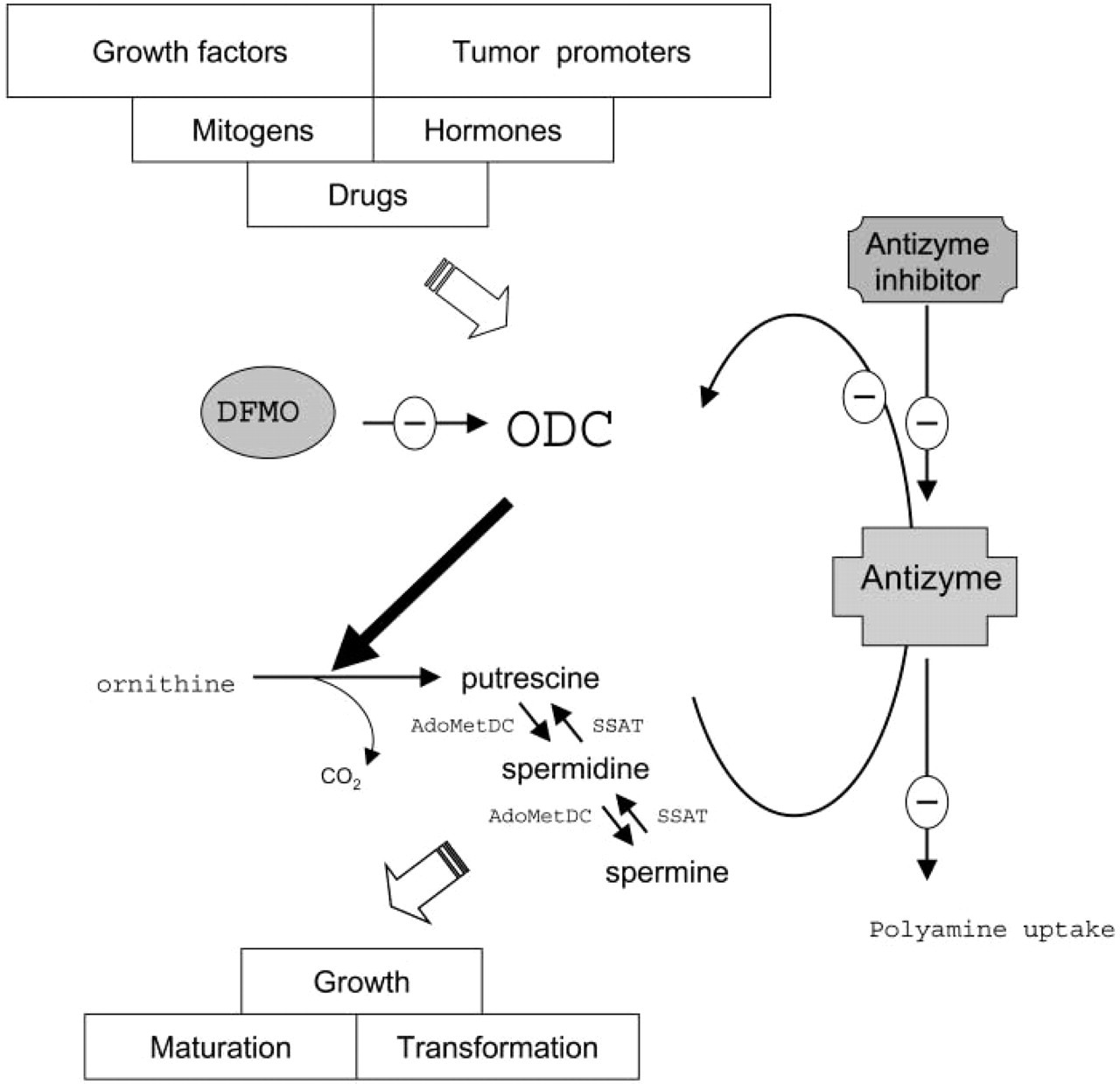
Schematic overview of the enzymatic features of the ornithine decarboxylase (ODC)/polyamine system. ODC catalyzes the conversion of ornithine into putrescine. Biosynthesis and catabolism of the polyamines spermidine and spermine is regulated by adenosylmethionine decarboxylase (AdoMetDC) and spermidine/spermine N 1-acetyltransferase (SSAT), respectively. Difluoromethylornithine (DFMO) is a specific and irreversible inhibitor of ODC. The ODC/polyamine system can be downregulated by antizyme, a polyamine-induced protein that inhibits ODC activity, stimulates ODC degradation, and suppresses polyamine uptake. ODC degradation can be prevented by antizyme inhibitor, which is highly homologous to ODC and traps the antizyme.
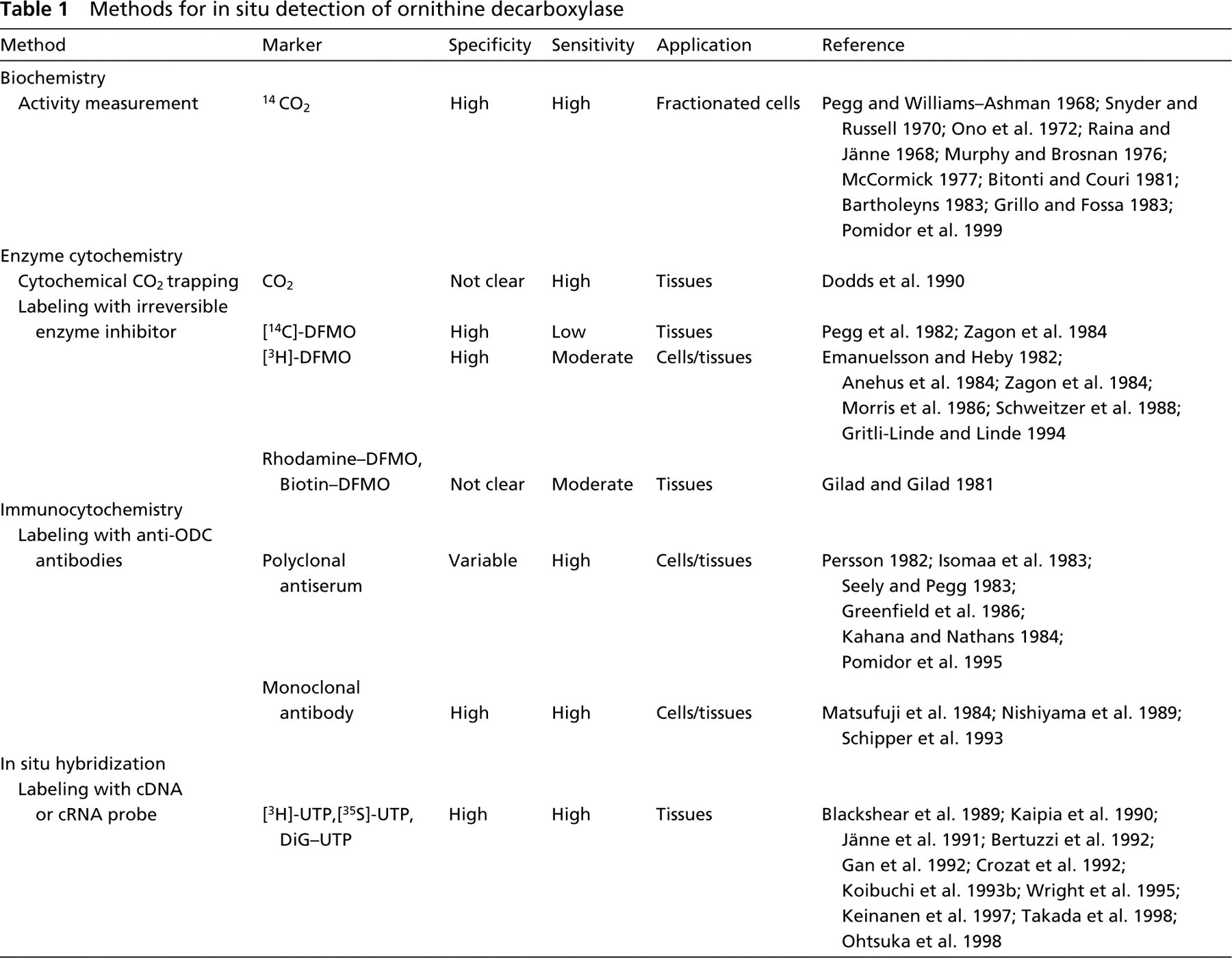
Despite extensive research, the precise role of the ODC/polyamine system in cell physiology remains to be clarified. Insight into the exact cellular and subcellular localization of ODC might provide valuable information on the role of this system in cell physiology. As summarized in Table 1, several approaches have been applied to localize ODC expression or activity in cells using biochemical, enzyme cytochemical, immunocytochemical, and hybridocytochemical methods. In this review, results obtained with the various methods with respect to localization patterns of ODC in cells (Table 2) and tissues in various organ systems (Table 3) in relation to polyamine functions are presented and interpreted.
Distribution Patterns of ODC in Cells and Tissues
Fractionated Cells
Activity of ODC can be determined by measuring production of 14CO2 (Pegg and Williams-Ashman 1968) or [1-14C]-putrescine (Tabib 1998) from [1-14C]-ornithine. These methods allow a linear and quantitative estimate of active ODC protein in homogenates of cells and tissues.
Methods for in situ detection of ornithine decarboxylase
The CO2-trapping technique has been used to determine ODC activity in subcellular fractions of rat prostate (Pegg and Williams-Ashman 1968), rat liver (Ono et al. 1972; Murphy and Brosnan 1976; Bitonti and Couri 1981; Grillo and Fossa 1983; Bartholeyns 1983), and mouse fibroblasts (McCormick 1977). Most studies showed that ODC activity is mainly or exclusively found in the cytosolic fraction (Pegg and Williams-Ashman 1968; McCormick 1977; Grillo and Fossa 1983), with or without induction by growth stimuli (Ono et al. 1972; Murphy and Brosnan 1976). However, some studies demonstrated the presence of ODC in the nucleus (Snyder and Russell 1970; Bitonti and Couri 1981). On the basis of an improved fractionation procedure, Bartholeyns (1983) established that ODC activity is equally distributed over nuclear and cytoplasmic fractions in non-stimulated liver. Nuclear ODC, which was mainly found in the nucleoli, was not induced by growth stimuli, in contrast to cytoplasmic ODC. Subcellular fractionation of human keratinocytes after detergent and salt extractions revealed that ODC protein and activity were detectable in both soluble and insoluble fractions (Pomidor et al. 1999). The insoluble ODC pool was enriched with phosphorylated ODC and appeared to be associated with the keratin cytoskeleton.
The results of fractionation studies to localize ODC activity are rather conflicting and show ODC activity present in both cytoplasm and nucleoplasm or nucleoli. Nuclear and cytoplasmic ODC may be differently regulated because ODC activity induced by growth-promoting stimuli is increased in the cytoplasm only. On the other hand, either contamination of the nuclear fraction with cytosolic ODC or leakage of nuclear ODC to the cytoplasm may have occurred during homogenization of tissues, which led to false conclusions.
Localization of ornithine decarboxylase in cells a
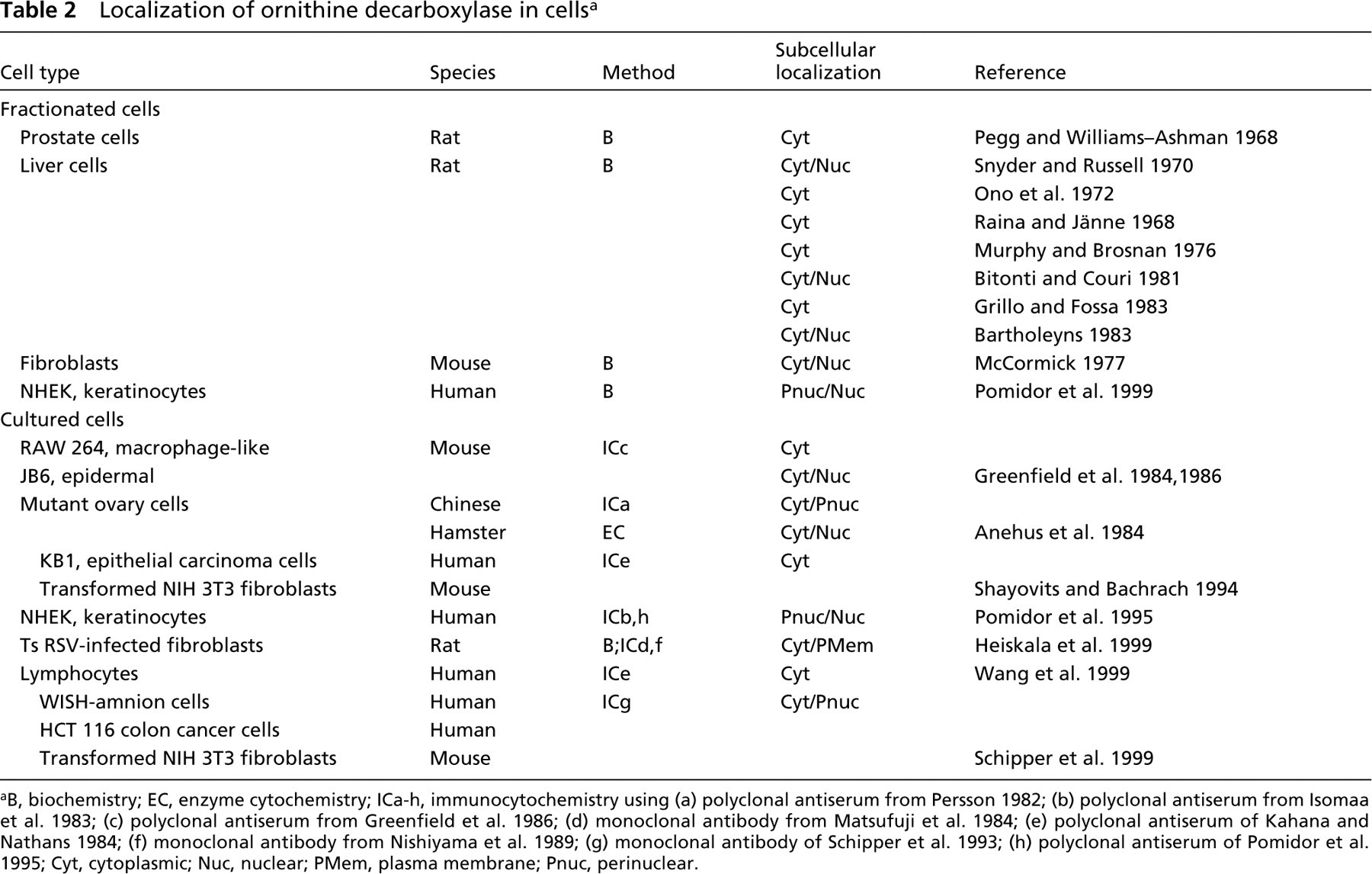
B, biochemistry; EC, enzyme cytochemistry; ICa-h, immunocytochemistry using (a) polyclonal antiserum from Persson 1982; (b) polyclonal antiserum from Isomaa et al. 1983; (c) polyclonal antiserum from Greenfield et al. 1986; (d) monoclonal antibody from Matsufuji et al. 1984; (e) polyclonal antiserum of Kahana and Nathans 1984; (f) monoclonal antibody from Nishiyama et al. 1989; (g) monoclonal antibody of Schipper et al. 1993; (h) polyclonal antiserum of Pomidor et al. 1995; Cyt, cytoplasmic; Nuc, nuclear; PMem, plasma membrane; Pnuc, perinuclear.
Cultured Cells
Intracellular localization of ODC in cultured cells was studied for the first time in stimulated murine macrophage-like RAW 264 cells using a polyclonal antiserum against ODC that had been purified from this cell line (Greenfield et al. 1984, 1986). ODC immunoreactivity was found exclusively in the cytoplasm, whereas in a cell line of epidermal origin (JB6), ODC protein was found in both cytoplasm and nucleoplasm but not in nucleoli.
Anehus et al. (1984) compared an immunocytochemical and an enzyme cytochemical method using radiolabeled difluoromethylornithine (DFMO), a specific irreversible inhibitor of ODC (Metcalf et al. 1978), to determine the intracellular localization of ODC in ODC-overproducing CHO cells. The immunocytochemical method using polyclonal antibodies of Persson (1982) showed considerably more ODC in the cytoplasm than in the nucleus. In contrast, ODC detected by autoradiography using radiolabeled DFMO was evenly distributed over the cell.
Bachrach and co-workers immunocytochemically detected ODC in transformed mouse fibroblasts, human epithelial carcinoma cells, and lymphoblasts from leukemia patients (Shayovits and Bachrach 1994; Wang et al. 1999). ODC immunoreactivity was decreased after treatment with cell growth-inhibiting drugs. In the growing cell fraction of human keratinocytes, ODC showed a perinuclear/nuclear distribution pattern, but it became more diffusely localized in the cytoplasm when cell growth was arrested (Pomidor et al. 1995). Recently, a combined biochemical and immunocytochemical study demonstrated translocation of a fraction of cytosolic ODC to the plasma membrane during cell activation and transformation of rat fibroblasts (Heiskala et al. 1999).
With our monoclonal antibody MP16-2, we examined localization of ODC in various human and murine cell lines (Schipper et al. 1999). Strong immunoreactivity of ODC was detected in the proliferating fraction of cultured cells, especially in cells showing mitotic figures. In confluent cells, ODC staining was faint and diffusely distributed throughout the cell, predominantly in the cytoplasm with the highest density in the perinuclear region, as revealed by confocal laser scanning microscopy (Figure 2A).
We have investigated the subcellular distribution patterns of ODC in more detail by using fusion proteins consisting of ODC and enhanced green fluorescent protein (EGFP). The expression vector pEGFP-N1 was subcloned with or without a human ODC cDNA fragment. Subsequently, EGFP and ODC-EGFP were expressed in cultured HeLa cells. Typical results of expression patterns of EGFP alone or the fusion protein are shown in Figures 2B–2D and 2G. Expression of EGFP alone in these cells was homogeneous throughout the cytoplasm and nuclei of the cells (Figure 2B). In contrast, expression patterns of ODC-EGFP constructs were heterogeneous (Figures 2C and 2D). Confluent cells showed cytoplasmic perinuclear staining, whereas dividing cells showed heterogeneous staining of the nucleus. When cells were treated with polyamines to induce antizyme-regulated ODC degradation, ODC was predominantly localized in the nucleus (Figure 2G). Immunostaining of antizyme in these cells showed that antizyme co-localizes with ODC in the nucleus (Figures 2F and 2G). These results suggest that ODC accumulates in the nucleus after antizyme induction. Further experiments using inhibitors of proteasomal degradation and/or nuclear translocation are needed to clarify the role of ODC translocation to the nucleus in antizyme-induced degradation of ODC.
It is not yet clear whether the distinct changes in ODC expression or localization patterns actually reflect progression through the cell cycle. The ODC gene has been described as a cell cycle-dependent gene that is only weakly expressed in quiescent cells. Its expression is strongly increased in proliferating cells (Kaczmarek 1986). ODC is an “early gene,” i.e., its transcription is increased early in the cell cycle (Kaczmarek et al. 1987; Pena et al. 1995). ODC mRNA increases at the beginning of the G1-phase both in cycling and stimulated quiescent smooth muscle cells (Campan et al. 1992). ODC activity increases in late G1 and peaks in G2 before the onset of mitosis, suggesting that ODC expression is biphasic during the cell cycle (Heby et al. 1976; Fredlund et al. 1995). Recent studies show that ODC translation in the G2/M transition is regulated in a cap-independent way by using a ribosomal entry site (IRES) (Pyronnet et al. 2000). Therefore, general inhibition of ODC translation in the G2/M-phase of the cell cycle can be overcome and polyamines can be produced when needed for mitotic spindle formation and chromatin condensation.
In general, higher levels of ODC were detected in most studies in smaller, more rapidly growing cells. ODC expression appears to be correlated with cell proliferation and cell transformation. Translocation of ODC in conjunction with cytoskeletal systems might be an important regulatory event in these cellular processes. In situ detection of ODC may therefore be useful as a biomarker for (malignant) cell proliferation or, conversely, as an indicator for sensitivity to anti-proliferative drugs, as suggested by Shayovits and Bachrach (1994).
Nervous System
Considerable evidence exists for a role of the ODC/polyamine system in neurobiological systems (for review see Bernstein and Müller 1999). In brain, temporal relationships between ODC activity and developmental or behavioral patterns have been found (Slotkin 1979; Slotkin and Bartolome 1986). In addition, a detrimental function of brain-associated ODC in response to mechanical, chemical, and ischemic injuries has been documented (Paschen 1992).
In rat and mouse brain, ODC activity is highest during birth and declines after the first week to a low adult level (Slotkin and Bartolome 1986). Localization studies of ODC during brain development in rat (Gilad and Gilad 1981; Morris et al. 1986; Dorn et al. 1987) and human (Müller et al. 1993) correlate with these biochemical findings. ODC expression was highest and ubiquitously distributed in rat cerebellum during the first week of development (Gilad and Gilad 1981; Dorn et al. 1987; Ichikawa et al. 1997) and remained present in discrete neural structures, e.g., the molecular layer of the cerebellum (Morris et al. 1986; Dorn et al. 1987) or spinal moto neurons (Cintra et al. 1987; Junttila et al. 1993). Similarly, during human brain development, high ODC immunoreactivity was found in the first stages that are characterized by neuroblast proliferation and migration. In adult human brain, immunoreactivity was restricted to motor and hippocampal neurons (Müller et al. 1993).
A sharp rise in brain ODC activity has also been found in various pathological conditions of the brain (Paschen 1992, and references therein). During cerebral ischemia, ODC gene expression and ODC protein synthesis are markedly increased after reflow, which is followed by a vast and prolonged increase in putrescine levels. The post-ischemic rise of ODC was found only in neurons in all brain structures, but its expression was most pronounced in vulnerable brain areas (Dempsey et al. 1988; Schweitzer et al. 1988; Müller et al. 1991b). The close relation between ODC activity and the density of neuronal necrosis argues for a role of the ODC/polyamine system in cell injury after ischemia. Polyamine levels may play an important role in the breakdown of the blood-brain barrier and formation of vasogenic edema processes, which occur after ischemic and traumatic brain injury (Baskaya et al. 1996). In addition, polyamines have deleterious effects on nerve cells by interactions with the N-methyl-
Localization of ornithine decarboxylase in tissues a
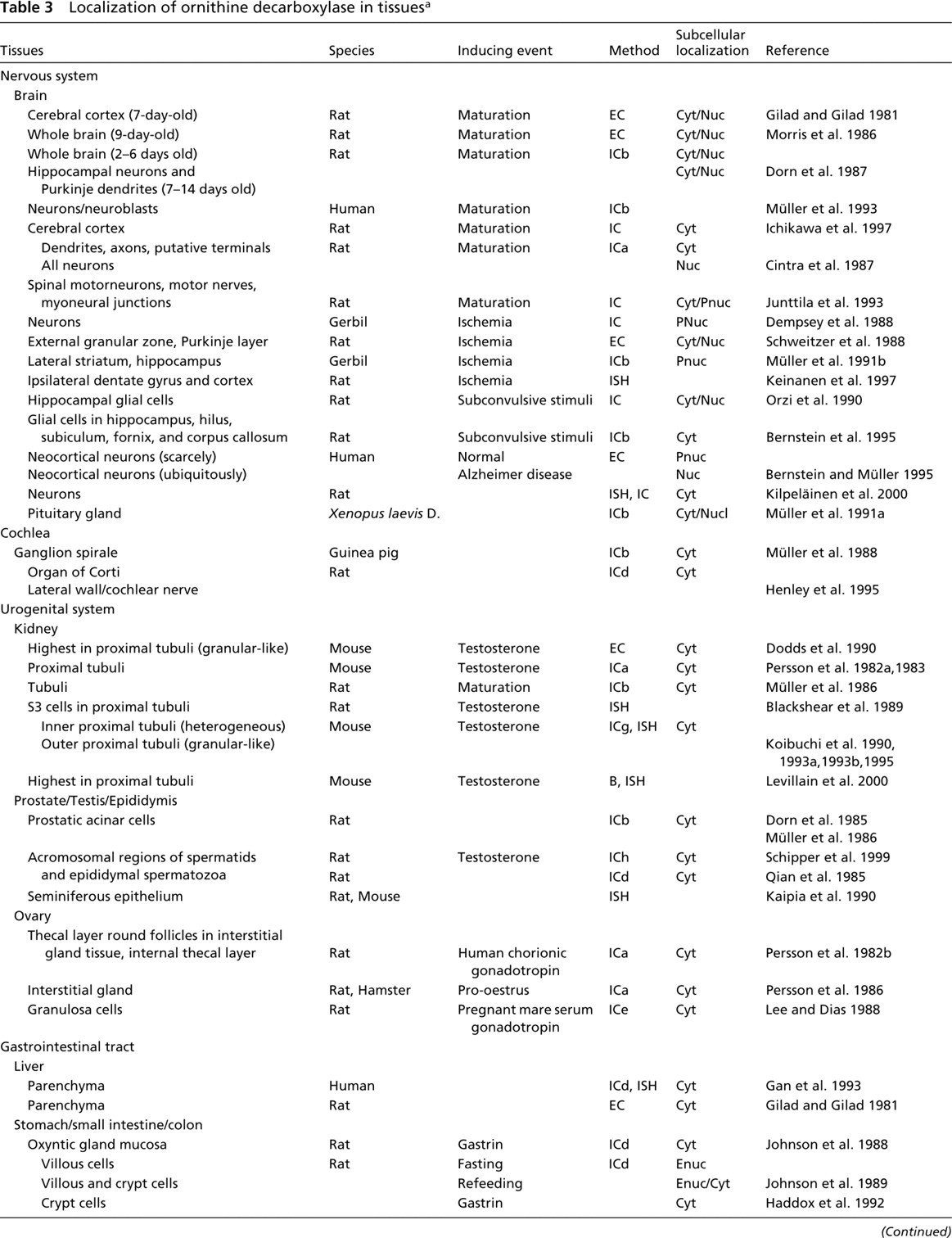
B, biochemistry; EC, enzyme cytochemistry; FC, flow cytometry; ICa-i, Immunocytochemistry using (a) polyclonal antiserum from Persson 1982; (b) polyclonal antiserum from Isomaa et al. 1983; (c) polyclonal antiserum from Seely and Pegg 1983; (d) polyclonal antiserum from Greenfield et al. 1984; (e) polyclonal antisera from Kahana and Nathans 1984; (f) monoclonal antibody from Matsufuji et al. 1984; (g) polyclonal antiserum from Nishiyama et al. 1989; (h) monoclonal antibody from Schipper et al. 1993; (i) polyclonal antiserum of Pomidor et al. 1995; ISH, in situ hybridization; Cyt, cytoplasmic; Nuc, nuclear; Nucl, nucleoli; Enuc, epinuclear; Pnuc, perinuclear; ∗by electron microscopy.
In conclusion, ODC expression may be closely associated with normal and pathological processes in the brain. However, whether induction of ODC has real pathological consequences or is merely a trophic response to stress or an epiphenomenon remains to be established. During brain maturation, ODC localization is restricted to neurons, which is in agreement with the proposed role of the ODC/polyamine system in neuronal migration, axogenesis, synaptogenesis, and activity of ion channels (Slotkin and Bartolome 1986; Williams 1997). In pathological conditions, ODC is generally localized in neurons except after subconvulsive stimulation, resulting in the appearance of ODC in glial cells.
Urogenital System
ODC is particularly active in epithelial cells of rodent kidneys and accessory sex organs and is regulated by sex hormones that induce hyperplastic and hypertrophic responses (Pegg et al. 1970; Blackshear et al. 1989; Jänne et al. 1991; Crozat et al. 1992; Fjösne et al. 1992; Calandra et al. 1996). Studies of the murine ODC gene revealed that the ODC promoter contains an androgen-responsive element-like sequence that can bind to the androgen receptor in vitro (Crozat et al. 1992). Increased levels of ODC mRNA and activity induced by androgens have been reported in kidney, testis, prostate, and seminal vesicles (Crozat et al. 1992; Calandra et al. 1996). In addition, ODC has been found in human prostatic fluid, seminal plasma, and sperm (Jänne et al. 1973; Mohan et al. 1999).
Localization of ODC expression in mouse or rat kidney after testosterone treatment has been studied with use of enzyme cytochemical (Pegg et al. 1982; Zagon et al. 1984; Dodds et al. 1990) and immunocytochemical tools (Persson et al. 1982a, 1983; Müller et al. 1986; Koibuchi et al. 1990; Schipper et al. 1999). Consistently, ODC expression was heterogeneously localized in epithelial cells of tubules and predominantly in the epithelial cells of the proximal tubule. This observation is in agreement with biochemical and hybridocytochemical studies in androgen-treated mice and rats showing that ODC is mainly expressed in proximal tubules (Blackshear et al. 1989; Jänne et al. 1991; Crozat et al. 1992; Koibuchi et al. 1993a, b; Levillain and Hus-Citharel 1998; Levillain et al. 2000). ODC was found immunocytochemically in the cytoplasm only, but cytochemical detection of ODC activity with radiolabeled DFMO also revealed ODC activity in the nucleus (Pegg et al. 1982; Zagon et al. 1984).
Prostate cells of human and rat origin produce polyamines at high levels. Its apparent functions are related to cell proliferation and secretory activities. ODC has been localized immunocytochemically in rat prostate gland, revealing heterogeneous cytoplasmic staining of acinar cells (Dorn et al. 1985; Müller et al. 1986; Schipper et al. 1999). Three types of ODC-immunoreactive cells were observed, i.e., strongly positive, weakly positive, and negative cells, which may represent different cell types in the prostate epithelium.
Elevated activity of ODC has been associated with androgen-dependent proliferation of Sertoli cells and germ cells in testis and of principal cells in the epididymal epithelium (Calandra et al. 1996). Qian et al. (1985) found ODC immunoreactivity in the cytoplasm of isolated testicular cells, epididymal spermatozoa, and cultured Sertoli cells. The highest concentration was found in acrosomal vesicles of round spermatids and in the acrosome region of epididymal spermatozoa. Studies in transgenic mice that overexpress ODC suggested that putrescine production has selective local stimulatory and inhibitory effects on DNA synthesis during spermatogenesis (Hakovirta et al. 1993). Spermine has been localized in the middle and top parts of the acrosome and may modulate sperm fertilization competence and the acrosome reaction (Rubinstein and Breitbart 1994).
ODC activity is strongly induced in the ovary of rat, hamster, and chicken during the ovalutory cycle (Persson et al. 1982b, 1986; Armstrong 1987). Immunocytochemical studies in prepubertal rats showed that ODC was localized in the thecal layer and interstitial gland tissue in human chorionic gonadotropin-stimulated rats (Persson et al. 1982b) and in rat and hamster during pro-estrus (Persson et al. 1986). The rapid and strong induction of ODC by gonadotropin was due to an increase in the amount or stability of ODC mRNA (Sertich et al. 1987). In ovaries of immature rats that had been treated with pregnant mare serum gonadotropin, ODC immunostaining was confined to the cytoplasm of granulosa cells. The 15-fold increase in ODC activity after treatment with pregnant mare serum gonadotropin could not be fully explained by the two-fold increase of ODC mRNA, suggesting an additional regulation of ODC at the translational and/or post-translational level (Weiner and Dias 1993). These studies imply that ODC expression in ovary is mediated by different physiological stimuli acting on different target cell types in the ovarian tissue than in other tissues.
Gastrointestinal Tract
Cell growth in the gastrointestinal tract, induced by food, hormones, refeeding after starvation, and intestinal adaptation is paralleled by increases in ODC activity (Luk 1992, and references therein). Furthermore, inhibition of ODC with DFMO has a rather universal inhibitory effect on cell proliferation in liver, stomach, pancreas, small intestine, and colon during postnatal maturation and adaptive growth, suggesting that the ODC/polyamine system plays an essential role in regulating mucosal growth of the gastrointestinal tract.
The liver has been an important organ in the initial studies of regulation of ODC activity. ODC expression was rapidly and strongly elevated during liver regeneration before the onset of nucleic acid and protein synthesis (Raina and Jänne 1968; Russell and Snyder 1969; Höltta 1975). Localization studies of ODC have been performed in human liver specimens, showing a heterogeneous pattern of immunoreactive ODC protein in the cytoplasm of hepatocytes, which paralleled ODC mRNA levels (Gan et al. 1992, 1993).
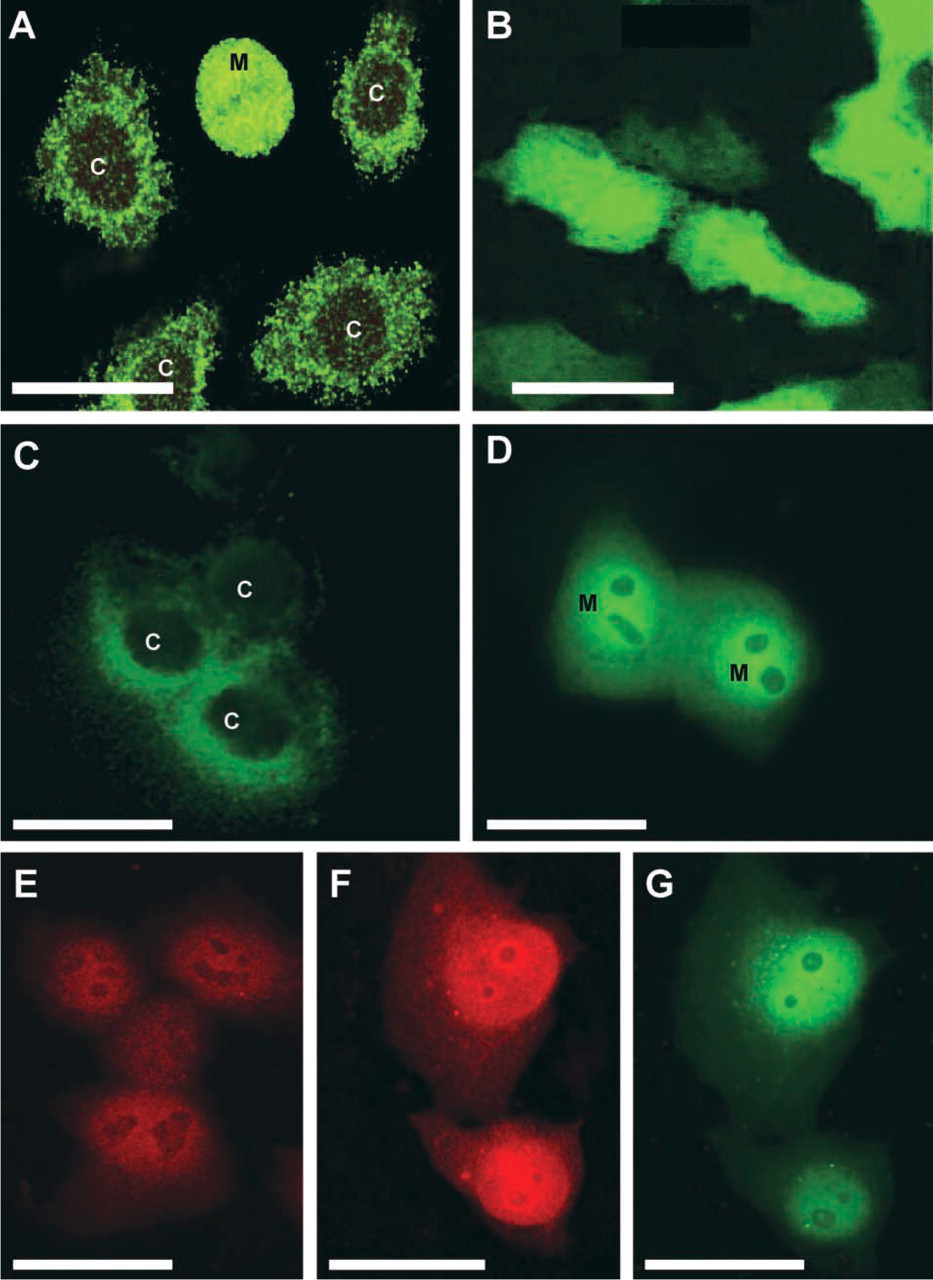
Localization of ODC and antizyme in cultured cells. (
The role of the ODC/polyamine system in proliferation of the gastric oxyntic cell is still a matter of debate. An increase in mucosal ODC activity could not be detected biochemically in response to refeeding or gastrin stimulation. Furthermore, treatment with DFMO had no effect on normal gastric epithelial growth and only partially prevented the trophic response to refeeding, but did inhibit the trophic action of gastrin. However, immunocytochemical analysis of stomach epithelium of fasted animals showed that ODC protein was present only in a narrow band of cells at the base of gastric pits and at the top of oxyntic glands, which increased after feeding or gastrin stimulation (Johnson et al. 1988). A localization study at the ultrastructural level showed that ODC was present in the mucous neck cells, which are the progenitor cells of the epithelium (Haddox et al. 1992). Therefore, growth processes in the epithelium of the stomach are promoted by induction of ODC in the proliferative fraction of gastric cells, which may well be too small a fraction to be detected by biochemical analysis of the heterogeneous mucosa of the stomach. In this case, biochemical analysis is hampered by the fact that stroma is also included in the tissue. Furthermore, the presence of high levels of nonspecific decarboxylases in the stomach may well mask the increase in ODC levels in the activity assay (Johnson et al. 1988).
ODC and polyamine levels are increased during rapid growth phases in maturation and recovery of intestinal epithelium during adaptive hyperplasia, during lactation, after intestinal obstruction, during post-starvation refeeding, and after feeding with lectins (Luk 1992, and references therein). In all studies DFMO treatment suppressed epithelial growth, suggesting a specific role for the ODC/polyamine system in intestinal epithelial growth processes. However, the exact cell physiology of ODC and polyamines in the intestinal mucosa is still not well understood. Several biochemical studies using the fasting-refeeding model have shown that ODC activity is induced after refeeding and is higher in villous epithelial cells than in crypt epithelial cells. Immunocytochemical studies showed that, in fasted animals, ODC was localized in the epinuclear and basal side of the nucleus of enterocytes along the entire villus (Haddox et al. 1992). Refeeding caused an increase in immunoreactive protein in villous cells but also induced ODC expression in proliferative crypt cells. Exposure of isolated jejunal segments to glycine induced an increase in immunoreactive ODC protein, which was confined to cells along the villous. Gastrin, however, increased ODC only in crypt cells near the crypt-villous border (Haddox et al. 1992). In isolated enterocytes of the colon of fasting patients, levels of ODC activity, immunoreactive ODC protein and polyamine concentrations were not different in lower and upper crypt regions (Elitsur et al. 1993).
These studies and studies using cultured enterocyte cell lines (Iwami et al. 1990) suggest that ODC regulation is different in epithelial cells of crypt and villous, which might actually be due to expression of a different ODC (iso)enzyme in maturing villous cells compared with proliferative crypt cells. Induction of ODC and increased levels of polyamines in enterocytes migrating from crypt to villous may be involved in maintenance of mitochondrial function and integrity necessary for nutrient absorption (Madsen et al. 1996).
ODC activity is increased in the early stages of cell growth in intestine and pancreas during the neonatal period (Morisset and Grondin 1987). With the use of an optimized immunocytochemical method to detect ODC in tissues, we performed ODC localization studies in developing rat intestine and pancreas (Schipper et al. 1999). In small intestine of 7-, 14-, and 21-day-old rats, immunoreactivity was present in both crypts and villi, whereas in 28- and 100-day-old rats, strong positivity of ODC was found in crypts only. In the pancreas of neonatal rats, islets of Langerhans showed strong cytoplasmic staining. In the pancreas of 14-day-old rats, heterogeneous staining of exocrine cells was observed (Romain et al. 1998). The observed age-related localization patterns in developing rat small intestine and pancreas may reflect changes in ODC expression during growth and maturation of these organs (Romain et al. unpublished data). An immunocytochemical study at the ultrastructural level showed that ODC was localized in the nucleus and rough endoplasmic reticulum of pancreatic rat acinar cells (Morisset et al. 1986).
Malignancies
ODC expression is upregulated in cancer cells, resulting in high basal levels of ODC and polyamines in many tumor types. Recent studies show that ODC overexpression coincides with expression of oncogenes during cell transformation (Auvinen et al. 1992, 1997; O'Brien et al. 1997; Shantz and Pegg 1998; Tabib and Bachrach 1998).
The role of the ODC/polyamine system in cancer progression has been extensively studied in a murine multistep model of carcinogenesis in the skin (reviewed by DiGiovanni 1992). Treatment of mouse epidermis with the potent tumor promoter phorbolester 12-O-tetradecanoylphorbol-13-acetate (TPA) resulted in a rapid and transient induction of ODC and subsequent accumulation of polyamines. Furthermore, epidermal tumors contain constitutively high levels of ODC activity, with carcinomas expressing higher ODC levels than papillomas. Immunocytochemical examination of ODC in TPA-treated epidermis of mice showed that ODC expression is most intense in the suprabasal keratinocytes located around hair follicles. In papillomas and squamous cell carcinomas, heterogeneous ODC staining was observed (Gilmour et al. 1986; Hietala et al. 1988). However, ODC expression was found to be different in skin of mice after TPA treatment when a different immunocytochemical method was used (Sundberg et al. 1994). Intense diffuse staining of suprabasal cells was found in interfollecular epidermis, with occasional more intense staining around follicles. In addition, ODC staining in papillomas was diffuse throughout the suprabasal layer, in contrast to the staining patterns in scattered areas as was found by Gilmour et al. (1986). Immunocytochemical and flow cytometric studies further supported the hypothesis that specific subpopulations of epidermal keratinocytes may be more sensitive to ODC modulation and neoplastic transformation (Robertson et al. 1990; Gilmour et al. 1992). Differential regulation of ODC may be due to the presence of a structurally and functionally altered ODC in epidermal tumors (Hietala et al. 1988; O'Brien et al. 1988). Studies in transgenic mice in which ODC was targeted to epidermal keratinocytes of the hair follicle further support the hypothesis that hair follicles are the possible origin of skin tumors and that ODC overexpression is sufficient for tumor promotion in mouse skin (Megosh et al. 1995; O'Brien et al. 1997).
Immunolocalization studies of human skin showed that ODC is present in both basal and corneal layers of normal skin (Verrando et al. 1987) and is constitutively increased in skin neoplasms (Hietala et al. 1988; Kagoura et al. 2000). Well-differentiated tumors showed more intense staining compared with poorly differentiated tumors (Kagoura et al. 2000).
Subcellular localization studies of ODC in normal human keratinocytes revealed that ODC is present in the perinuclear/nuclear region (Pomidor et al. 1995, 1999). Intracellular localization becomes more diffuse over the cytoplasm after treatment with TPA. Interestingly, changes in ODC localization were associated with rearrangements of the cytoskeleton, suggesting attachment of ODC to structural elements and/or a role of ODC in cytokinetic processes.
ODC localization studies have also been performed in some other types of malignancies, i.e., hepatocellular carcinoma (Gan et al. 1993), colorectal cancer (Oya et al. 1995), oral neoplasms (Williams et al. 1995), and papillomatosis of bladder epithelium (Takada et al. 1998). ODC expression was increased in these lesions, indicating an important role of ODC in diseases characterized by cell proliferation. Gan et al. (1993) quantitated ODC expression in liver specimens by in situ hybridization and immunocytochemistry. A heterogeneous pattern after immunostaining of ODC protein was found in the cytoplasm of hepatocytes. The amount of immunoreactive ODC protein progressively increased from normal, to normal liver adjacent to carcinoma, to cirrhosis, and to hepatocellular carcinoma. ODC mRNA levels were generally in agreement with protein levels. Recent biochemical studies of ODC activity and polyamine levels in hepatocellular carcinomas further provided evidence that polyamine metabolism may be a parameter of degree of malignancy in hepatocellular carcinomas (Kubo et al. 1998).
On the Subcellular Localization of ODC
In the past 30 years, ODC expression has been found in either the cytoplasm, the nucleus, or both. It is difficult to synthesize these rather inconclusive studies into a general concept of ODC localization and its significance in cell physiology. Interpretation of ODC distribution patterns is further hampered by the little knowledge on the exact molecular actions of its products, the polyamines.
One possibility is that these inconsistent results are based on methodological problems with respect to the (lack of) sensitivity and/or specificity of the various methods used for the localization of ODC. Alternatively, the various localization patterns may reflect differences among cell types, their particular biological behavior (proliferation, differentiation, apoptosis), or the onset of ODC expression. Furthermore, (trans)location of ODC may be an important mechanism to control expression of ODC.
These aspects of subcellular localization patterns of ODC in relation to polyamine function are discussed in detail in the next paragraphs.
Methodological Problems
Early studies focused on biochemical measurement of ODC activity in fractionated cell preparations. Activity of the enzyme was found in cytoplasmic as well as nuclear fractions, and particularly in nucleoli. This biochemical method is highly sensitive, but care must be taken lest improper homogenization procedures or storage of frozen supernatants cause artifacts due to the metabolism of ornithine via the mitochondrial pathway (Gaines et al. 1988). Furthermore, redistribution of ODC during isolation and fractionation procedures may have affected accurate localization.
Development of the highly specific and irreversibly binding ODC inhibitor DFMO enabled cytochemical localization of ODC activity using fluorescent or radioactively labeled derivatives of DFMO. Most of these studies report that ODC is localized in both cytoplasm and nuclear compartments. Because DFMO is highly specific for ODC and does not react with the inactive ODC-antizyme complex (Kitani and Fujisawa 1986a), reliable labeling of catalytically active ODC can be obtained. However, the use of DFMO to localize ODC in vivo has some drawbacks. The accuracy and sensitivity of ODC localization strongly depend on in vivo binding of the labeled inhibitor to ODC. Low levels of ODC are not detected, and it is not clear whether DFMO is taken up in a similar way by the different cell types or subcellular compartments that have been investigated (Seely et al. 1982; Kitani and Fujisawa 1986b).
Immunocytochemical detection of ODC revealed different localization patterns, depending on the cell system studied and the antibody used. These patterns varied from cytoplasmic or (peri)nuclear localization to localization in both cytoplasm and nucleus. A problem in ODC immunocytochemistry is that information on the nature or activity of the detected ODC protein cannot be obtained. ODC antibodies may also react with catalytically inactive forms of ODC, e.g., monomeric subunits of ODC or monomers bound to the ODC-inhibitory antizyme protein. Furthermore, ODC antibodies may crossreact with the antizyme inhibitor, which shows a high homology with ODC protein but lacks its catalytic activity (Murakami et al. 1996). The existence of multiple or modified forms of ODC may further complicate the immunocytochemical localization of ODC. In homogenates of mouse kidney, two species of active ODC protein were found, which might represent true isoenzymes (Kitani and Fujisawa 1988). ODC might also be post-translationally modified by arginylation or phosphorylation, producing heterogeneous ODC molecules that are differently charged and/or have different enzymatic properties (Kopitz et al. 1990; Brown et al. 1994; Reddy et al. 1996; Heiskala et al. 1999; Pomidor et al. 1999). Phosphorylation has been shown to mask an epitope of mouse ODC that is recognized by a monoclonal antibody (Donato et al. 1986).
In addition to differences in specificity of antibodies against ODC, effects of cell and tissue preparation procedures, such as fixation, permeabilization, dehydration, and embedding, can greatly affect preservation and/or accessibility of the ODC protein for the antibodies. We recently attempted to systematically develop a reliable method for immunocytochemical detection of ODC (Schipper et al. 1999). In these studies we observed that fixation and antigen retrieval methods have substantial effects on the intensity and specificity of ODC immunostaining.
In contrast to the findings obtained with biochemical and enzyme cytochemical studies, immunocytochemical studies of ODC showed that ODC is present mainly in the cytoplasm. This discrepancy may mean that catalytically active nuclear ODC exists in a complexed or cryptic form or is less accessible for antibodies to bind. On the other hand, cytosolic ODC may exist in an immunologically active but enzymatically inactive form. Immunocytochemical detection of ODC in ODC-overproducing CHO cells showed considerably more ODC in the cytoplasm than in the nucleus. In contrast, ODC detected by autoradiography using radiolabeled DFMO was evenly distributed over the cell (Anehus et al. 1984). This difference in distribution patterns suggests the existence of a cytoplasmic pool of catalytically inactive ODC protein, which might represent post-translationally modified ODC. Recent evidence for this “dormant” pool of ODC has been found in fractionated human keratinocytes in which a phosphorylated form of ODC was demonstrated in the insoluble fractions (Pomidor et al. 1999). This may be an important post-translational regulation mechanism of the activity of ODC, as is known for other enzymes as well, such as glucose-6-phosphate dehydrogenase (Swezey and Epel 1986; for review see Van Noorden and Jonges 1995). Another possibility is that ODC largely exists as an inactive complex with antizyme protein in the cytoplasm, as was reported in adult mouse brain (Laitinen 1985).
In situ hybridization studies using a labeled cRNA or cDNA probe specifically hybridizing with ODC mRNA have been performed on mouse, rat, and human tissues (Table 1). The localization patterns of ODC mRNA were, at least partially, consistent with ODC localization patterns obtained in biochemical and immunocytochemical studies. However, many other experiments have provided evidence for translational and post-translational control of ODC (Heby and Persson 1990). Therefore, localization and concentration of mRNA may not actually reflect ODC expression at the level of ODC protein and/or enzyme activity.
Cell Physiological Aspects
Technical aspects may be responsible for the variety of staining patterns, but it may also be possible that the localization of ODC in cytoplasm and nucleus varies depending on the physiological state of the cell. ODC may be expressed in a particular cell type or subcellular compartment when its products, the polyamines, are needed there. Although intracellular transport of polyamines cannot be ruled out, targeting of ODC to the functional site of polyamines may be the actual transport mechanism.
Polyamines are implied to have an essential role in mitotic spindle organization and modulation of DNA structure and stability. Therefore, nuclear ODC may be an important source for nuclear polyamines. An argument against this proposition is that, to produce nuclear spermidine and spermine in addition to their precursor putrescine, the other biosynthetic enzymes of the ODC/polyamine system also must be translocated/present in the nucleus, for which no evidence (yet) exists. However, putrescine may not serve solely as a precursor for the production of spermidine and spermine but may exert cellular effects of its own, as found recently in intestinal epithelial cells (Bardocz et al. 1998; Löser et al. 1999; Pignata et al. 1999; Farriol et al. 2001).
Localization studies of polyamines using autoradiographic, cytochemical, and immunocytochemical techniques have confirmed the presence of polyamines in the nucleus (Roch et al. 1997, and references therein). A few studies have reported nucleolus-associated localization of ODC (Emanuelsson and Heby 1982; Müller et al. 1991b). It has been suggested that post-translationally modified ODC is translocated to the nucleolus, where it may regulate RNA polymerase I activity (Manen and Russell 1977; Russell 1985). However, more purified fractions of ODC did not affect the latter enzyme activity (Urata et al. 1987; Eichler and Corr 1992), and DFMO was not able to inhibit rRNA transcription (Iapalucci et al. 1984). Therefore, the suggested involvement of ODC in modulating RNA poly-merase activity may actually be untrue, but a possible role of the ODC/polyamine system in other nucleoli-associated functions (e.g., ribosomal RNA synthesis, ribosome biogenesis) cannot be excluded.
The vast majority of the results presented in Tables 2 and 3 show a cytoplasmic localization of ODC. Furthermore, most studies in which ODC localization was examined before and after stimulation report on a cytoplasmic rather than a nuclear localization of stimulated ODC.
The precise functional role of the ODC/polyamine system in the cytoplasm still needs to be elucidated, but many studies suggest that polyamines play essential roles in the machinery of protein biosynthesis. This hypothesis is further supported by immunoelectron microscopic studies of ODC and polyamines in which the label was closely associated with rough endoplasmic reticulum and polyribosomal structures, respectively (Morisset et al. 1986; Fujiwara et al. 1998).
Recent studies in cultured cells using confocal laser scanning microscopy (Pomidor et al. 1995, 1999; Heiskala et al. 1999; Schipper et al. 1999; Figure 2) have revealed the presence of ODC at the plasma membrane and/or nuclear envelope. The plasma membrane as a functional site for ODC is plausible because ODC has been implicated in the formation of signaling complexes in focal adhesion sites (Auvinen et al. 1995), transmembrane calcium and/or calmodulin signaling (Ginty and Seidel 1989), and membrane translocation of protein kinase C (Groblewski et al. 1992). There is no direct evidence that ODC is exported out of the cell, although ODC activity has been found in human prostatic fluid, seminal plasma, and sperm (Jänne et al. 1973; Mohan et al. 1999).
Membrane localization of ODC appeared to be related to structural elements, i.e., actin or keratin cytoskeletal elements (Pomidor et al. 1995, 1999; Heiskala et al. 1999). In activated or transformed fibroblasts, translocation of ODC to the plasma membrane was mediated by a p47phox membrane-targeting motif-related sequence (amino acids 165-172) and was dependent on phosphorylation (Heiskala et al. 1999). It was suggested that cell shape changes transduced via the cytoskeleton translocate ODC and that concomitantly altered ODC activity affects mitotic cytosketal rearrangement events. These findings also show that translocation of ODC is mandatory for regulation of physiological function of ODC because inhibition of ODC activity or irrelevant membrane targeting of the active enzyme led to defective cytokinesis.
Translocation of ODC may also have a functional role in its degradation. It is well established that antizyme binds to ODC and targets it to the proteasome for degradation. Antizyme has been localized in the cytoplasm but also in the nucleus, especially in cells with high ODC expression (Junttila et al. 1995; Kilpeläinen et al. 2000; Gritli-Linde et al. 2001). When turnover of ODC in the nucleus by antizyme was blocked by a proteasome inhibitor, ODC was expressed in the nucleus (Gritli-Linde et al. 2001). In our GFP experiments, induction of antizyme caused accumulation of antizyme and ODC in the nucleus (Figure 2D). These data suggest an antizyme-mediated translocation of ODC to the nucleus for degradation by nuclear proteasomes.
The variety of ODC distribution patterns as observed in different tissue types may be explained by the various roles of polyamines in cell metabolism and growth. As expected, ODC expression is prominent in the proliferating fraction of rapidly growing tissues. For example, ODC activity is particularly high in tissues with high cell turnover, e.g., intestinal epithelium, which require a constant supply of polyamines for growth, renewal, and metabolism. In contrast, highest activity of ODC has been found in the prostate, in which polyamines are synthesized for export rather than for intracellular use. In transgenic mice that over-express human ODC due to a ubiquitous promoter, abnormalities were found only in testis and brain (Halonen et al. 1993). Targeted ODC overexpression using cell type-specific promotors had no phenotypic effects in liver (Alhonen et al. 1996) and intestine (unpublished observations) but resulted in an increased frequency of papillomas in skin (Megosh et al. 1995).
ODC distribution patterns may depend not only on the cell type but also on cell kinetic behavior, because ODC activity and polyamine pools change during cell growth, differentiation, and cell death in a transient manner. Therefore, the onset of ODC expression can vary greatly among cells, depending on their physiological status, creating heterogeneous and apparently inconsistent results in cytochemical studies.
In conclusion, intracellular localization of ODC is not static but is subject to highly dynamic processes. Future studies in living cells using real-time analysis of fluorescence-tagged ODC and ODC regulatory proteins (antizyme, antizyme inhibitor) are needed to further unravel the complexity of the regulation and function of the ODC/polyamine system.
Footnotes
Acknowledgments
Supported by the Dutch Cancer Society (grants NKB-93-599, NKB-98-1807) and the Nijbakker Morra Foundation.
We wish to thank Dr Lo Persson and Prof dr Olli Jänne for generously providing us with the human ODC cDNA. We gratefully acknowledge Prof dr Olle Heby and Dr Jonas Nilsson for kindly supplying antizyme antiserum.
