Abstract
Ghrelin is a recently identified hormone with potent growth hormone (GH)-releasing activity. It is produced by rat and human gastric endocrine cells and by the pituitary, hypothalamus, placenta, and by gastroenteropancreatic tumors. No evidence of ghrelin production by foregut-derived organs other than stomach has been provided to date. The aim of the present study was to investigate ghrelin expression by human fetal (20 cases), infant (13 cases), and adult (seven cases) lungs by immunohistochemistry, in situ hybridization, and RT-PCR. Expression of the GH secretagogue receptor, the endogenous receptor for ghrelin, was also investigated by RT-PCR. Ghrelin protein was found in the endocrine cells of the fetal lung in decreasing amounts from embryonic to late fetal periods. Its expression was maintained in newborns and children under 2 years but was virtually absent in older individuals. Scattered positive cells were also found in the trachea and the esophagus. Ghrelin mRNA was detected in adult lung by the more sensitive RT-PCR technique. GHS receptor mRNA was detected in nine cases of infant and adult lungs, possibly indicating the existence of local autocrine circuits. We conclude that the fetal lung is an additional source of circulating ghrelin, whose functions at the respiratory tract level remain to be clarified.
G
In the normal stomach, ghrelin was localized in the neuroendocrine cell compartment of the oxyntic mucosa and was found to be the product of X-like cells, which appear to be the main source of circulating ghrelin in humans (Date et al. 2000). The gastric antrum and small intestine contain very few ghrelin-producing cells and the large bowel is virtually devoid of ghrelin (Date et al. 2000; Papotti et al. 2001). Such apparent foregut-restricted ghrelin expression prompted an analysis of other foregut derivatives, including the bronchial tree and the esophagus (Colby et al. 1995; Williams et al. 1995). In fact, no data were found in the literature, with the exception of the study of Ariyasu et al. (2001), in which ghrelin mRNA was detected by Northern blotting analysis in total lung extracts, among other tissues.
The aim of the present study was to investigate the ghrelin-expressing cells in the human fetal, infant and adult lung by immunohistochemistry (IHC) and in situ hybridization (ISH). We show that neuroendocrine cells of the developing lung contain large amounts of immunoreactive ghrelin and its mRNA as opposed to the adult lung, in which ghrelin is only occasionally expressed.
Materials and Methods
Tissue Samples
Twenty cases of fetal lung tissue (gestational week 7–40) and 20 infant and adult lung specimens (aged 1 day–64 years) were collected from the files of the Departments of Pathology, Universities of Turin and Genoa, and from the “Regina Margherita” Children's Hospital, Turin. Fetal tissue was obtained from voluntary or elective abortions and from autopsies of intrauterine deaths. Infant and adult lung tissues were obtained from both autopsies and surgical specimens. For all but two cases, paraffin blocks of formalinfixed material were available for conventional histology and IHC. All cases of infant and adult lung also had fresh-frozen material for mRNA studies.
Immunohistochemistry
Sections serial to those used for conventional histology were collected on poly-
The expression of ghrelin and chromogranin A in lung tissue was evaluated in serial sections and was quantified by counting the mean number of cells positive for each marker in each bronchial section (at least 20 per section). The mean number of positive cells per bronchial section in each case was recorded.
In Situ Hybridization
Selected cases were studied by a non-radioactive ISH procedure to confirm IHC findings and define ghrelin mRNA localization. Silane-coated slides were hybridized overnight at a working dilution of 33 nM of an equimolar mixture of two 45-nucleotide antisense probes (Kojima et al. 1999) corresponding to positions 90–134 and 421–465 of the prepro-ghrelin sequence. Probes were digoxigenin-labeled with the Boehringer labeling kit (Mannheim, Germany) according to the manufacturer's instructions. Hybridization with an unrelated probe, as well as omission of the specific probe and RNase digestion, served in selected samples as negative controls. Prehybridization treatments included a microwave passage (5 min at 800 W in citrate buffer, pH 6.0) and proteinase K digestion (1 μg/ml) for 4 min. Hybridization products were revealed applying the DAKO GenPoint kit as described elsewhere (Papotti et al. 2000a) with minor modifications, including 1:5 dilution of tyramide solution and hot washing in PBS after tyramide incubation (Volante et al. 2000). Positive control consisted of normal oxyntic mucosa of the stomach.
RT-PCR for Ghrelin and GHS Receptor
In all infant and adult lung tissues, RT-PCR was performed to reveal mRNA of both ghrelin and its receptor. Total RNA extraction and complementary DNA transcription were performed as described elsewhere (Papotti et al. 2000a). The primers for ghrelin were synthesized according to the sequence reported by Gualillo et al. (2001): 5′-TGAGCCCTGAACACCAGAGAG-3′ for the forward and 5′-AAAGCCAGATGAGCGCTTCTA-3′ for the reverse primer. Those for GHS receptors 1a and 1b were synthesized according to Korbonits et al. (2001) and were employed for RT-PCR using the same conditions described by these authors. The following sequences were used: 5′-TCGTGGGTGCCTCGCT-3′ as the forward primer for both GHS-R 1a and GHS-R 1b, 5′-CACCACTACAGCCAGCATTTTC-3′ for the GHS-R 1a reverse primer and 5′-GCTGAGACCCACCCAGCA-3′ for the GHS-R 1b reverse primer. The expected sizes of the amplicons were 327 bp, 65 bp, and 66 bp for ghrelin, GHS-R 1a, and GHS-R 1b, respectively. β2-microglobulin amplification served as a control of the RNA quality (see details in Papotti et al. 2000a). Positive controls included normal gastric mucosa for ghrelin and pituitary tissue for GHS-R 1a and 1b amplifications. Negative controls consisted of omission of cDNA in the PCR mixture and of reverse transcriptase enzyme during retrotranscription.
To further test the RT-PCR product specificity, Southern blotting analysis (SBA) was performed using the probe sequence previously published by Korbonits et al. (2001). The following sequences were used: 5′-AGGGACCAGAACCACAAGCAAACCG-3′ for ghrelin and 5′-TCCGGTTCAACGCCCCCTTTG-3′ for GHSR-1 probes. Membranes were hybridized overnight at 42C with 25 pmol of digoxigenin-labeled ghrelin and GHS-R oligonucleotide probes. The membranes were then washed with 2 × SSC-0.1% SDS for 5 min at 42C and 0.5 × SSC-0.1% SDS at 42C for 10 min. Digoxigenin-labeled specific hybridization was visualized using an immunological detection system (Boehringer; Mannheim, Germany) employing anti-digoxigenin antibodies conjugated with alkaline phosphatase. Detection was performed using the chemiluminescent substrate disodium 3-(4-methoxyspiro,1,2-dioxetane-3,2-(5-chloro) tricyclo (3.3.1.) decan;-4-yl) phenylphosphatase (CSPD) (Boehringer) according to the manufacturer's instructions. All blots were exposed to X-ray films with intensifying screens at RT for 3 or 5 hr.
Statistics
Statistical analysis of the observed differences of ghrelin expression between groups, including different phases of lung development, was carried out by ANOVA and Student's t-test.
Results
Fetal Tissues
As summarized in Table 1, all cases of fetal lung (7–40 weeks' gestational age) had ghrelin-producing cells scattered in the bronchial mucosa, as shown by IHC using a specific antibody recognizing a C-terminal portion of both acylated and non-acylated ghrelin. The immunostaining was completely abolished in parallel sections by pretreatment of the antibody with excess ghrelin (Figure 1). Ghrelin-producing cells were polygonal or elongated, were intermingled with cubic or columnar epithelial cells of the bronchi, and were basally located against the basal membrane, either isolated or in small clusters. No ghrelin immunoreactivity was ever observed in other structures of the bronchial wall or in the terminal portions of the bronchial tree (e.g., pneumocytes).
The IHC demonstration of ghrelin was confirmed in selected cases (nos. 3, 4, 6, 10, 13, 19, and 20) by ghrelin mRNA detection using ISH in parallel sections. The two procedures identified the same cells in the bronchial mucosa (Figure 1).
The extent of ghrelin expression differed according to the gestational age and was apparently unrelated to the cause of death (although the case series is limited). Ghrelin immunoreactivity was expressed by counting the mean number of positive cells in bronchial sections of the developing respiratory tree. In general, the mean number of ghrelin-producing cells was much higher (5–12.4% of the epithelial bronchial cells) in the embryonic and early fetal periods when the lung is in the pseudoglandular phase of development (Figure 2). Using week 18 of gestation as a cut-off to separate cases in the pseudoglandular vs the canalicular phase, we found that 10 cases of the former stage of development had a mean of 7.7 ghrelin-producing cells per bronchial section as opposed to 3.3 cells counted in 10 cases with a gestational age of 19–40 weeks. This difference was statistically significant (p<0.001).
To better investigate the distribution of ghrelin-producing cells, a comparison with the neuroendocrine cell compartment of the developing lung was made, staining serial sections with panendocrine markers that also recognize also Kultchinski cells (including chromogranin A, synaptophysin, N-CAM, NSE, PGP 9.5) and with bombesin/gastrin-releasing peptide (GRP). This latter was focally positive in endocrine cells apparently containing ghrelin. However, chromogranin A proved the best marker to outline the neuroendocrine cell compartment and was used here to assess ghrelin expression in neuroendocrine cells. In most cases, ghrelin and chromogranin A were produced by the same cells. However, in the first 18 weeks of pregnancy ghrelin-producing cells outnumbered those expressing chromogranin A (and synaptophysin or NSE). Conversely, N-CAM or PGP 9.5 was negative (Figure 2e). Counting the mean number of ghrelin-positive cells and chromogranin A-positive cells per bronchial section, it appeared that the former were two- to 20-Fold more numerous than the latter during the embryonic period and until week 18 of gestation. The opposite was true in the late fetal period, in which the ghrelin-positive cell population was markedly reduced, to such an extent that in some cases it was lower than the chromogranin A-positive neuroendocrine cell population.
Ghrelin expression in fetal lung a
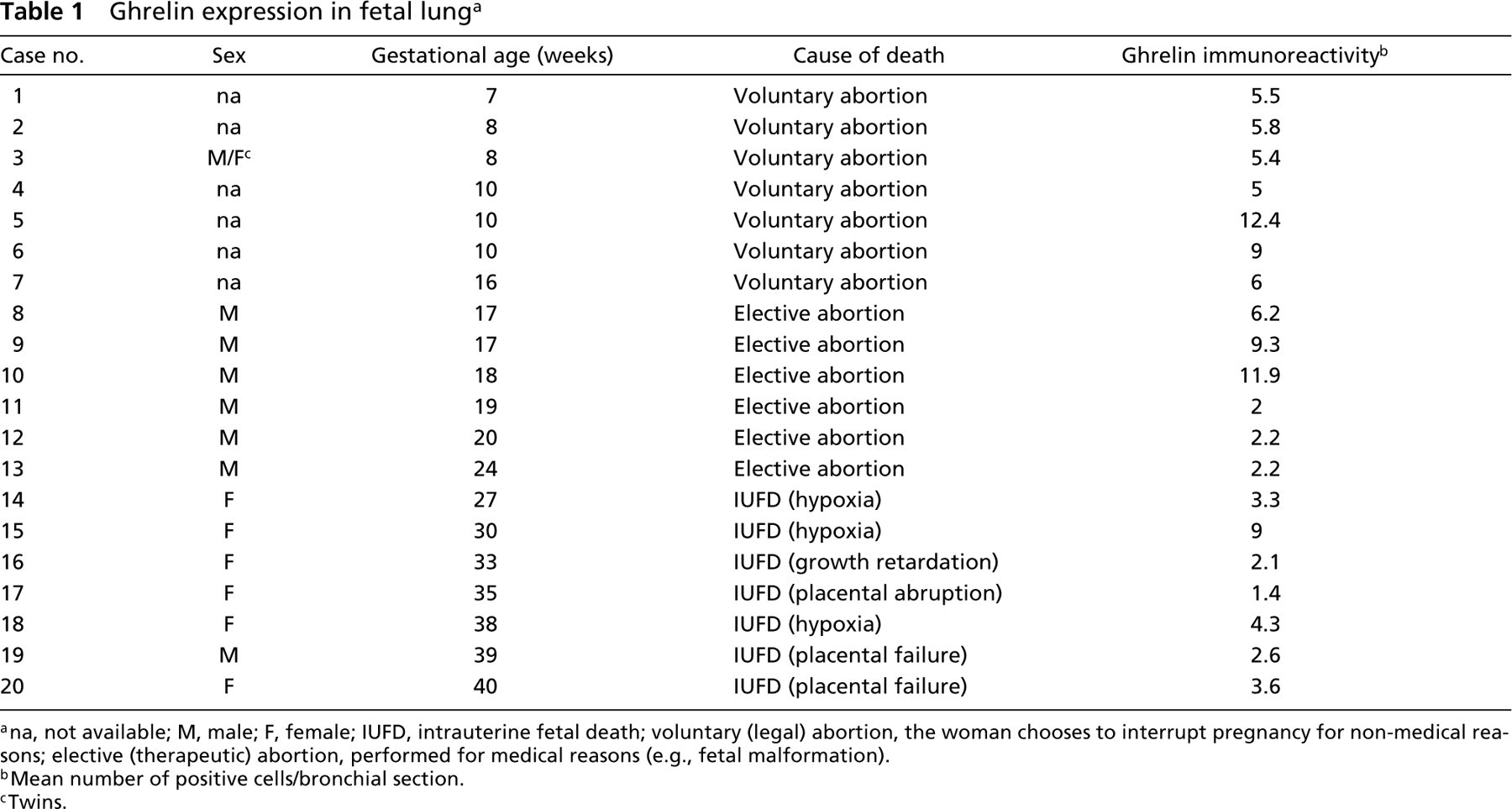
ana, not available; M, male; F, female; IUFD, intrauterine fetal death; voluntary (legal) abortion, the woman chooses to interrupt pregnancy for non-medical reasons; elective (therapeutic) abortion, performed for medical reasons (e.g., fetal malformation).
bMean number of positive cells/bronchial section.
cTwins.
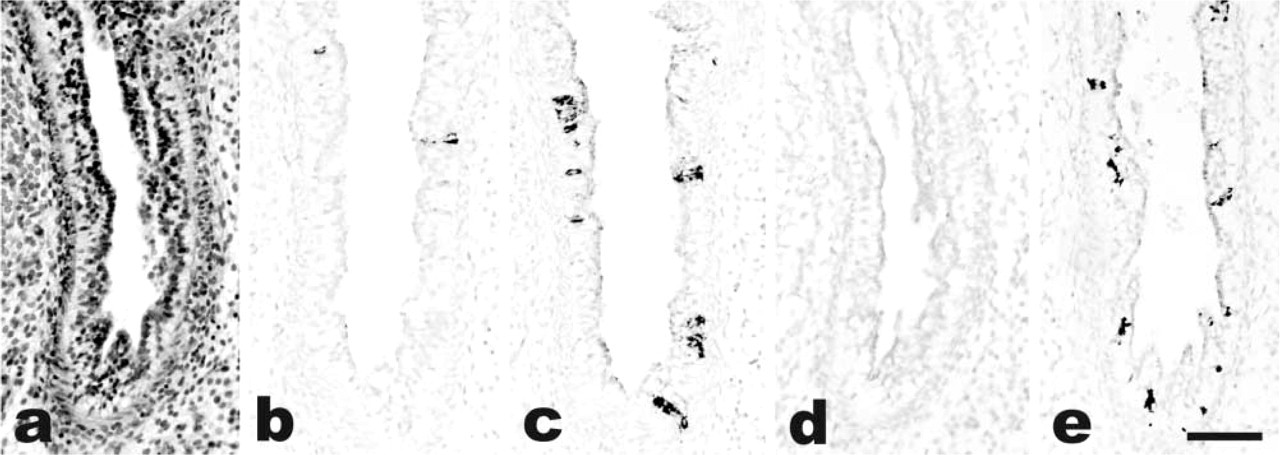
Case 4. Fetal lung in the pseudoglandular phase of development (
In the specimens analyzed here, other fetal tissues were occasionally observed adjacent to the lung. The trachea was present in five cases and showed single immunoreactive basal cells in all cases. The esophagus was present in five cases only, and ghrelin-positive cells were found in the mucosa of three cases having a gestational age of 10–18 weeks (Figure 3). As expected, immunoreactive ghrelin was found in the endocrine cells of the stomach, in the whole gut (with a decreasing number of cells towards the hindgut portion), and in the pancreas, in all cases in which gastroenteropancreatic tissues were available for immunostaining. No IHC reaction was observed in other fetal tissues, i.e., ear, bone, pharynx, thymus, liver, lymph node, bladder, gonads, mesonephrus, salivary gland, skeletal muscle, kidney, trophoblast, heart, cartilage, and blood vessels.
Infant and Adult Tissues
The lungs of children or adults who underwent surgery or died of different causes were found to contain the expected fraction of neuroendocrine cells in the bronchial wall, as confirmed by positive immunostaining of panendocrine markers (including chromogranin A, synaptophysin, and NSE) and of bombesin. N-CAM was negative in the neuroendocrine cells but stained many nerve endings of the bronchial wall beneath the epithelium. Some of the neuroendocrine cells also contained immunoreactive ghrelin in cases aged 1 day to 2 years (mean count of 4.0 ghrelin-positive cells per bronchial section; range 0.3–11.1). In older infants and in adult lung specimens, the number of ghrelin-producing cells was very low. After a careful search, two or three ghrelin-positive cells were identified in each slide (containing at least 20–30 bronchial branches). The mean count was as low as 0.2 ghrelin-positive cells per bronchial section (range 0–0.6).
All the above cases were also analyzed for ghrelin mRNA expression by RT-PCR and were found to be positive in the majority, probably due to the known higher sensitivity of this procedure. Comparing the results of IHC and mRNA analysis, all cases but one were concordant. In case no. 8, no ghrelin mRNA amplification was obtained despite the fact that ghrelin-positive cells were found in the corresponding tissue section.
Finally, RT-PCR was also employed to analyze ghrelin receptor (GHS-R 1a and 1b) mRNA. In the pediatric age group, 2/13 and 7/13 cases expressed the GHS-R type 1a and 1b, respectively. In seven lungs of adult patients, only four expressed the type 1b receptor, while type 1a was negative (Figure 4; Table 2).
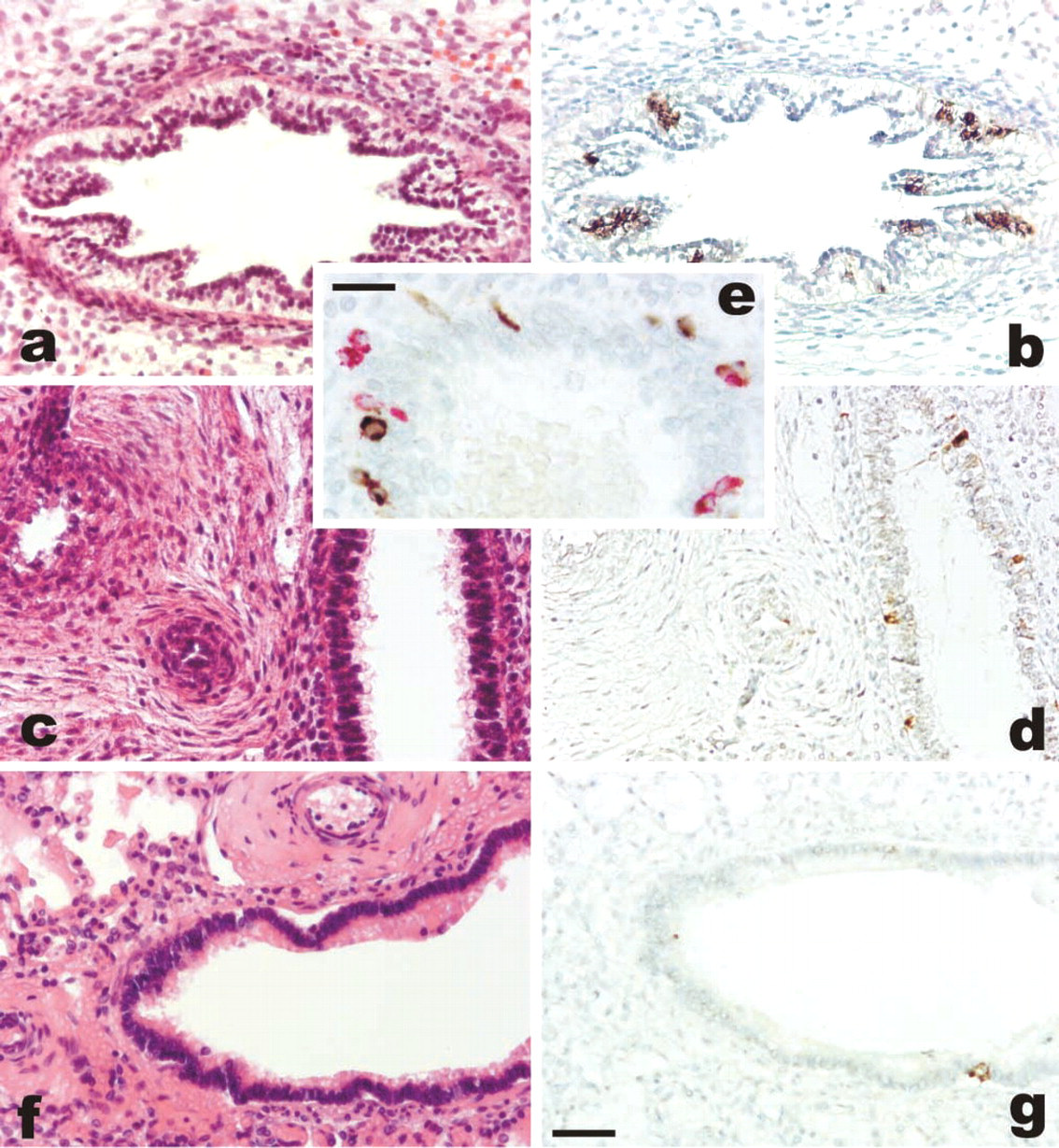
Ghrelin immunoreactivity in the developing lung at 8 (
Discussion
In this study we showed for the first time that the recently identified gastric hormone ghrelin is expressed by neuroendocrine cells of the fetal and infant lung and, to a very limited extent, by the adult lung. Immunoreactive ghrelin was present in single cells or small clusters in the bronchial wall as early as week 7 of gestation and was detected in decreasing amounts in late fetal lungs. In the postnatal period, single endocrine cells scattered in the bronchial mucosa expressed ghrelin during the first 2 years of life. Then they became progressively more rare and were only occasionally encountered in the adult lung parenchyma. The other components of the lung were never found to contain ghrelin, including alveolar spaces of the mature lung, nerve endings of the bronchial wall, blood vessels, and pleura.
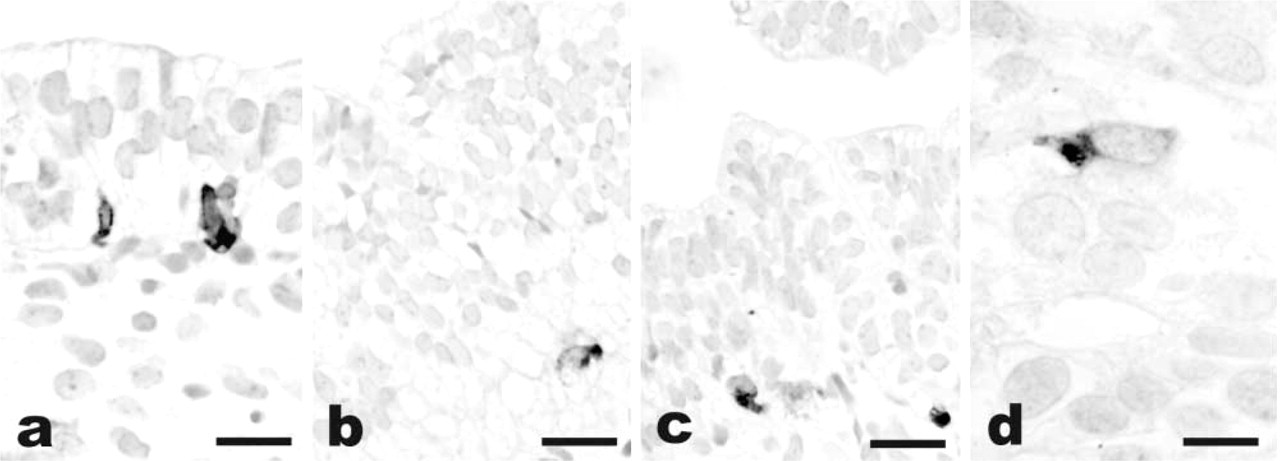
Ghrelin immunoreactivity in fetal tissues other than lung. Strong staining of scattered ghrelin-producing cells is present in the esophageal (
The development of the respiratory tree occurs through several phases that give rise to the original groove and primitive lung bud from the pharynx, followed by progressive dichotomous bronchial division into distal airways (Williams et al. 1995). The embryonic phase is followed by the pseudoglandular, canalicular, saccular, and alveolar phases. The crucial step is the formation of primitive respiratory bronchioli from weeks 16–20 (canalicular phase). Ghrelin was intensely expressed in bronchial cells of cases with a gestation age of 7–18 weeks, corresponding to the pseudoglandular phase of lung development and the transition to the canalicular phase (16–20 weeks of gestation). From gestational week 19 to birth and in the first 2 years of life, the number of ghrelin-immunoreactive cells in the lung was approximately half of that of the cases dated weeks 7–18.
In all cases, the intensity of the ghrelin immunoreactivity was similar to that of the oxyntic mucosa of the stomach, indicating that during fetal life the lung may be an additional important source of circulating ghrelin.
The cells producing ghrelin in the fetal lung were neuroendocrine cells, as confirmed by the co-localization of ghrelin with bombesin/GRP or with panendocrine markers expressed by neuroendocrine cells of the lung (chromogranin A among others). However, especially during the embryonic phase of development, it was evident that ghrelin was produced in cells lacking chromogranin A and other endocrine markers. Whether these represent immature endocrine cells not yet expressing endocrine markers or non-endocrine cells that produce ghrelin is not clear.
The IHC and ISH procedures did not enable us to distinguish between acylated and non-acylated ghrelin because the commercial antibody was directed to the C-terminal portion of the molecule (amino acids 13–28). The significance of ghrelin expression in the developing lung is not known, and its activities may be sustained by both acylated and non-acylated forms of ghrelin. In fact, although the classical GH release-associated activities are mediated by the octanoylated compound des-octanoyl ghrelin, which contributes the largest part of circulating ghrelin, this compound was recently found to have other functions, including anti-proliferative effects on tumor cell lines (Cassoni et al. 2001; Ghè et al. 2002).
In addition, it is not known whether the mechanism mediating ghrelin activities is the result of a direct effect on specific receptors or whether it follows a more complex pathway via GH release. To address this point, the demonstration of specific receptors for ghrelin in the fetal lung is a crucial step. GH secretagoguebinding sites were demonstrated in the adult lung parenchyma by a binding assay (Papotti et al. 2000b; Ghè et al. 2002). In the same study, GHS receptor mRNA was not found in the few cases of lung parenchyma tested by RT-PCR (Ghè et al. 2002).
Unfortunately, frozen specimens from early phases of the developing lung were very hard to obtain. Only one case was tested and found positive for the only currently known ghrelin receptor, GHS-R types 1a and 1b (data not shown). This receptor has also been evaluated in postnatal lungs. Two of 13 pediatric cases studied by RT-PCR contained the specific GHS-R 1a mRNA and approximately half of the cases had that of GHS-R 1b. Far from having demonstrated a direct (possibly autocrine or paracrine) mechanism of ghrelin in lung cells, the presence of the specific receptor in at least a fraction of cases may indicate that ghrelin not only regulates GH release at the central level but may also exert other functions in peripheral tissues via specific receptors, as also shown in the gastroenteropancreatic tract (Papotti et al. 2001; Volante et al. 2002) and in pituitary adenomas (Kim et al. 2001; Korbonits et al. 2001).
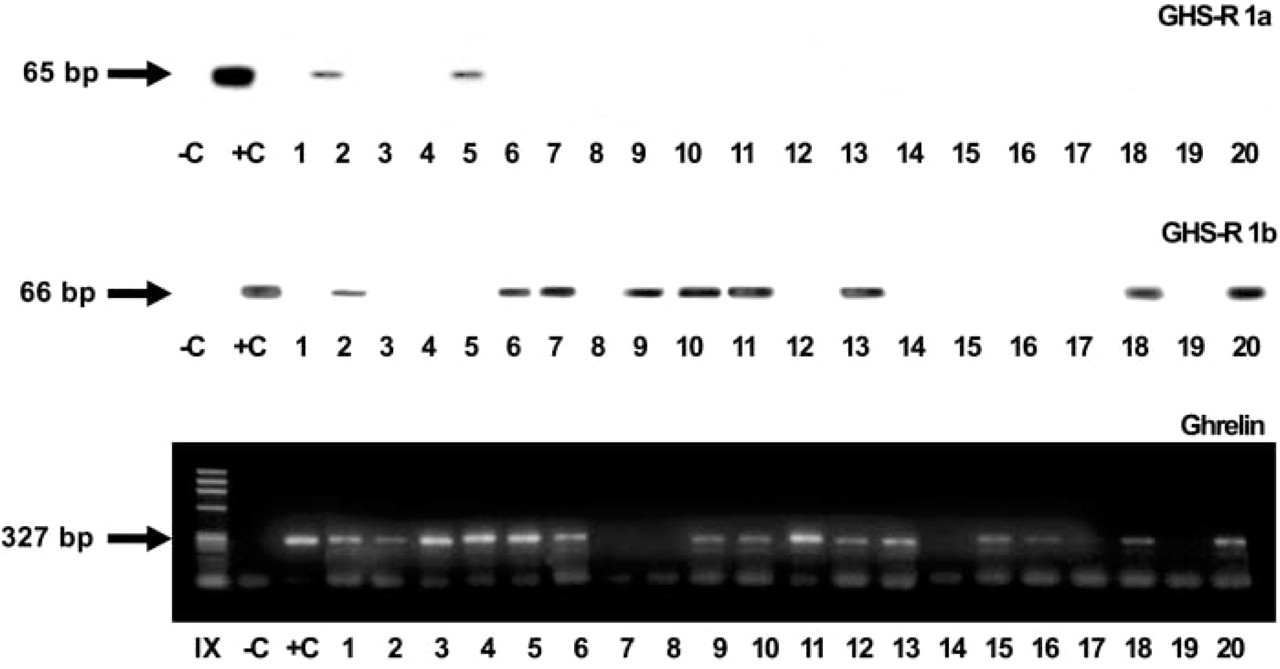
Ghrelin and GHS receptor mRNA expression in infant and adult lung tissues. RT-PCR analysis shows the amplified product of GHS-R 1a and 1b at the expected band (65 and 66 bp, respectively) in two and nine cases. Ghrelin mRNA amplification gives a specific product of 327 bp in 15 cases. C, omission of cDNA in the PCR mixture; C+, positive control, i.e., pituitary for GHS-R 1a and 1b and gastric mucosa for ghrelin amplification.
The findings reported here will require further studies on the ultrastructural localization of ghrelin-producing cells, on the specific activities mediated by the locally produced ghrelin in the developing lung, and on the significance of the marked ghrelin decrease during late pregnancy and infancy. Obviously, it remains to be ascertained whether the decrease is physiological or is related to the pathological condition which led to fetal death. It can be speculated that ghrelin may cooperate in elongation and maturation of the bronchial tree, with special regard to the transition from the pseudoglandular to the canalicular phase, during which the terminal bronchiolar structures are being formed.
Ghrelin and growth hormone secretagogue receptor expression in infant and adult lung a
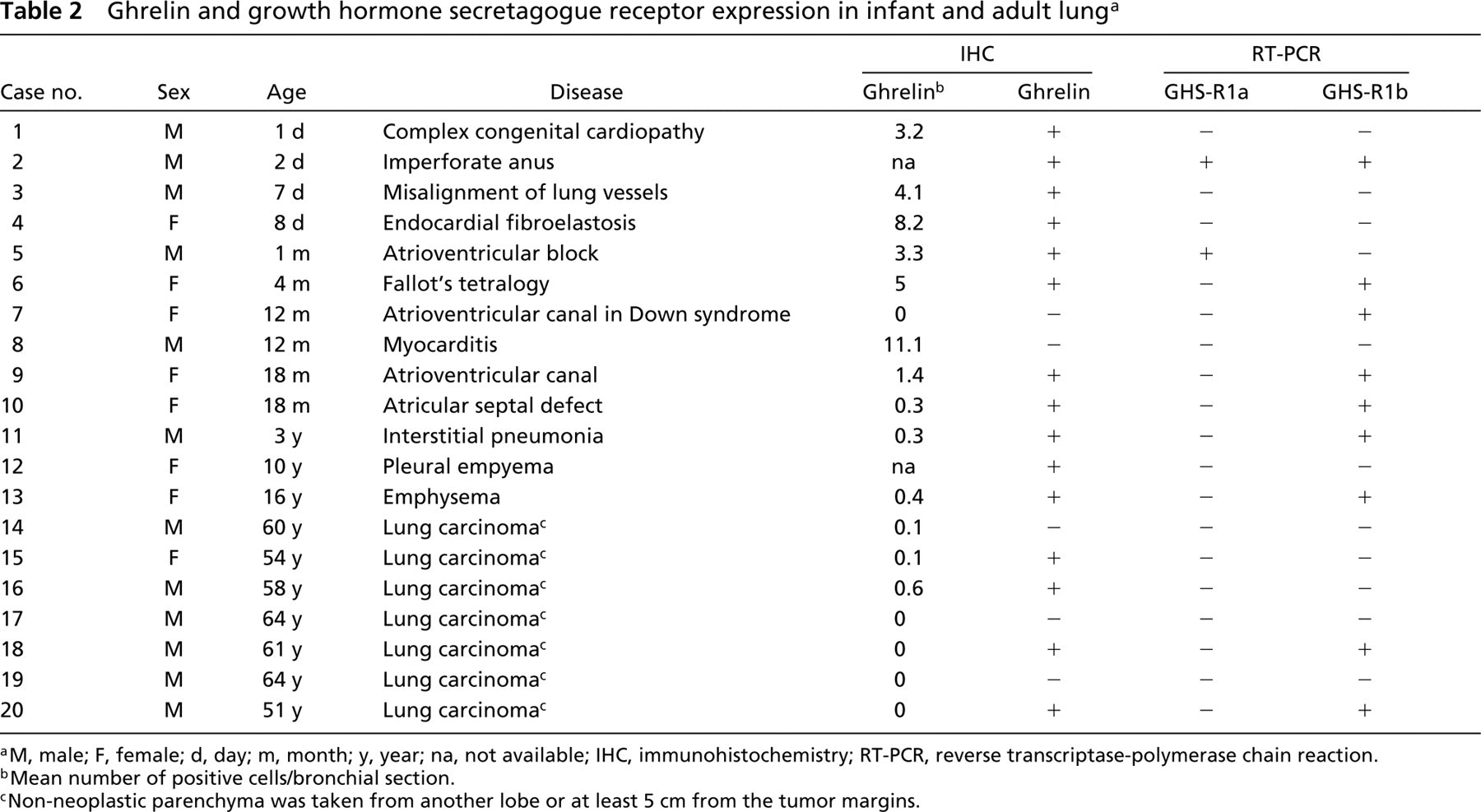
aM, male; F, female; d, day; m, month; y, year; na, not available; IHC, immunohistochemistry; RT-PCR, reverse transcriptase-polymerase chain reaction.
bMean number of positive cells/bronchial section.
cNon-neoplastic parenchyma was taken from another lobe or at least 5 cm from the tumor margins.
A similar pattern of reactivity was also found in the few esophageal walls identified in the tissue blocks. Several ghrelin-producing cells were present in the mucosa, with a basal location. Interestingly, this finding was restricted to cases from the embryonic period because two cases from the fetal period having ciliated or stratified epithelium apparently lacked ghrelin immunoreactivity. With regard to the other tissues, we have been unable to confirm the data of Mori et al. (2000) on ghrelin expression by the kidney. The fetal kidneys (as well as two cases of adult kidney analyzed in parallel; data not shown) were completely unreactive by IHC. The ghrelin expression reported by Gualillo et al. (2001) in the placenta also required better investigation. In only one of 11 cases tested, in fact, small clusters of ghrelin-positive cells were present in the deep portion of the villi, probably corresponding to histyocytic cells of the stroma rather than to trophoblastic cells. In addition, we could not detect ghrelin in fetal gonads, although ghrelin mRNA has been demonstrated to be produced in the rat testis (Tena–Sempere et al. 2002). It should be pointed out, however, that the use of techniques more sensitive than IHC, such as PCR, by the other authors could be responsible for these discrepancies.
In conclusion, we have shown that the developing lung is a major source of ghrelin, with endocrine cells of the bronchial tree strongly expressing ghrelin to an extent similar to that observed in the gastric mucosa. Its expression is maximal in the first 18 weeks of gestation, and decreases during late pregnancy and in pediatric ages. The adult lung is virtually devoid of ghrelin-immunoreactive cells, which are only occasionally present in the bronchial mucosa. A fraction of fetal lung cases also express the ghrelin receptor GHS-R, indicating that ligand–receptor interactions may be actively operating in the developing lung.
Footnotes
Acknowledgements
Supported in part by grants from the Italian Ministry of University and Research (Rome) and the SMEM Foundation (Fondazione per lo Studio delle Malattie Endocrine e Metaboliche) (Turin).
We thank Dr R. Arisio (St Anna Hospital, Turin) for providing one of the cases studied here and Professors G. Bussolati, E. Ghigo, and G. Muccioli (University of Turin) for helpful discussion. The skillful technical assistance of Dr C. Pecchioni, Dr P. Gugliotta, and Mr A. Grua is also gratefully acknowledged.
