Abstract
Cyclo-oxygenase-2 (COX-2) is believed to participate in the repair of gastric ulcer. Like humans, pigs frequently develop gastric ulcers and thus represent an attractive animal model in which to study the repair process of naturally occurring gastric ulcers. However, expression of COX in the pig stomach has not been reported. The objectives of this study were to determine whether COX isoenzymes are expressed in porcine gastric ulcers and to characterize the porcine COX-2 cDNA. Normal stomachs (n=5) and those with gastric ulcers (n=35) were studied by immunohistochemistry and immunoblotting analysis. Reverse transcription-polymerase chain reaction (RT-PCR) was used to isolate the complete porcine COX-2 cDNA. COX-1 staining was present in normal stomach and in ulcerated areas. No COX-2 was detected in normal stomach, but COX-2 was strongly expressed in the ulcerated area in 28/35 (80%) gastric ulcers (p<0.01). Immunoblotting analysis confirmed the restricted expression of COX-2 in the ulcerated areas. The porcine COX-2 cDNA was shown to code for a 604 amino acid protein that is 89% identical to human COX-2. These results provide the complete primary structure of porcine COX-2 and demonstrate for the first time that the enzyme is induced in naturally occurring porcine gastric ulcers.
Gastric ulcer is a common disease that affects millions of people in the United States each year (Cotran et al. 1999). The pathophysiology of this widespread digestive problem is complex and is still not entirely understood. In pigs, gastric ulcers also represent a widespread problem that annually causes important economic losses (Friendship 1999; Doster 2000). The pathogenesis of porcine gastric ulcers is complex, and the major factors contributing to the development of the ulcerative lesions are multiple and diverse (Friendship 1999; Doster 2000). Alimentary factors and stressful conditions are among the risk factors that have been associated with gastric ulcers in pigs (Friendship 1999; Doster 2000). The presence of a spiral bacterium in porcine gastric ulcers has been reported by some authors (Barbosa et al. 1995; Quieroz et al. 1996) but its pathogenetic role remains to be defined. Although there are differences between human and porcine gastric ulcers, such as the distinct anatomic localization of the ulcer (Lee 2000), the pig represents an attractive animal model in which to study the repair of naturally occurring gastric ulcerations.
Prostaglandins (PGs) play an important role in protecting the mucosa of the stomach (Wilson 1991; Arakawa et al. 1998). By acting on mucus and bicarbonate secretion as well as on blood flow, PGs contribute to maintenance of gastric mucosal integrity. Cyclo-oxygenase (COX), also known as prostaglandin G/H synthase, is the first rate-limiting enzyme in the biosynthesis of PGs from arachidonic acid (Smith 1992; Funk 1993). COX is a homodimer composed of two subunits of about 70,000 Daltons and one heme group (van der Ouderaa et al. 1979; Kulmacz and Lands 1984). Two isoforms of COX have been characterized, i.e., COX-1 and COX-2. Both isoforms share similar catalytic activity as well as important sequence homology at the nucleic acid and amino acid levels (Herschman 1996; Smith and Dewitt 1996) but greatly differ in their pattern of expression. COX-1 is constitutively expressed in many tissues and is believed to mediate physiological responses requiring biosynthesis of PGs. In contrast, COX-2 is generally not expressed in most cells but can be induced in response to inflammatory reactions, growth factors, and tumor promoters (Herschman 1996; Williams and DuBois 1996). The molecular characterization of COX-2 has been reported in various species, including humans, rodents, and various large animal species (Kujubu et al. 1991; Hla and Neilson 1992; Feng et al. 1993; Zhang et al. 1996; Bracken et al. 1997; Guan et al. 1997; Boerboom and Sirois 1998; Song et al. 1998; Liu et al. 2001). Although fragments of the porcine COX-2 cDNA have been sequenced, the complete characterization of porcine COX-2 has not yet been reported.
COX-1, which is constitutively expressed in the stomach, is believed to be the isoform mostly responsible for the synthesis of gastric PGs (Kargman et al. 1996). In the normal stomach of humans and animals, most studies found COX-2 mRNA and protein to be expressed at low or undetectable levels, and the physiological role of COX-2 in normal stomach remains to be defined (Kargman et al. 1996; Ristimaki et al. 1997; Zimmermann et al. 1998; Jackson et al. 2000; Sung et al. 2000). However, COX-2 overexpression has been reported in several different pathological gastric conditions, including ulcer healing (Mizuno et al. 1997; Ristimaki et al. 1997; Kishimoto et al. 1998; Sawaoka et al. 1998; Shigeta et al. 1998; Takahashi et al. 1998; Fu et al. 1999; McCarthy et al. 1999; Jackson et al. 2000; Sung et al. 2000; Tatsuguchi et al. 2000). In the latter, induced COX-2 expression was detected at the ulcer margins or in the ulcer bed by immunohistochemistry in gastric ulcers in humans and in experimentally induced gastric ulcers in rats and mice (Mizuno et al. 1997; Shigeta et al. 1998; Takahashi et al. 1998; Jackson et al. 2000; Tatsuguchi et al. 2000). However, expression of COX-1 and COX-2 in naturally occurring gastric ulcers in animals has never been reported. The objectives of this study were to evaluate the expression of COX enzymes in porcine gastric ulcers, to determine their cellular localization, and to characterize the complete primary structure of porcine COX-2.
Materials and Methods
Materials
The citrate phosphate dextrose solution was obtained from Abbott Laboratories (North Chicago, IL). The Vectastain ABC kit was obtained from Vector Laboratories (Burlingame, CA). Diaminobenzidine tetrahydrochloride was purchased from Sigma-Aldrich (Oakville, Ont, Canada). Hybond polyvinylidene difluoride membranes (0.45 μm) were obtained from ICN Pharmaceuticals (Montreal, PQ, Canada). The horseradish peroxidase-labeled donkey anti-rabbit antibodies and the enhanced chemiluminescence (ECL) detection kit were purchased from Amersham Life Sciences (Arlington Heights, IL). Kodak Bio-Max X-ray film was obtained from Eastman Kodak (Rochester, NY). TRIzol total RNA isolation reagent, 1-kb DNA ladder, and synthetic oligonucleotides were obtained from Gibco BRL (Life Technologies; Gaithersburg, MD). Electrophoretic reagents were obtained from Bio-Rad Laboratories (Richmond, CA). The Prime-a-Gene labeling system, the Access RT-PCR System, and pGem-T Easy Vector were purchased from Promega (Madison, WI). The Gene Clean Kit was obtained from Bio 101 (La Jolla, CA). The porcine genomic library was obtained from Clontech (Palo Alto, CA). [α-32P]-dCTP was purchased from NEN Life Science Products (Mississauga, Ont, Canada).
Tissue Samples and Platelet Isolation
Thirty-five cases of gastric ulcers from pigs submitted for a necropsy at the Département de Pathologie et Microbiologie of the Faculté de Médecine Vétérinaire (Université de Montréal) were included in the study. All cases were confirmed by examination of hematoxylin-eosin-saffran-stained sections by a veterinary pathologist. Light microscopic examination of Whartin-Starry-stained tissue sections was also done to evaluate the presence of spiral bacteria. Pigs were either brought dead for necropsy or were sacrificed. Five normal stomachs obtained from healthy 6-month-old pigs were used as controls. All tissues studied were fixed in 10% neutral buffered formalin, whereas fresh samples from normal stomachs and from gastric ulcers were frozen at −70C for immunoblotting analysis.
Porcine platelets were isolated from whole blood collected by venipuncture in anticoagulant (citrate phosphate dextrose solution). Platelet-rich plasma was isolated by successive centrifugations of the citrated blood for 3 min at decreasing speeds (700, 650, and 600 × g) as previously described (Catafalmo and Dodds 1989). Platelets were recovered from the platelet-rich plasma by centrifugation at 16,000 × g for 10 min and were stored at − 70C. All animal procedures were approved by the institutional Animal Care and Use Committee.
Anti-COX Antibodies
Two anti-COX antibodies (antibodies 8223 and MF243) were used. Affinity-purified polyclonal antibody 8223 was raised in rabbits against ovine COX-1, and was shown to be selective for COX-1 in various species (Wimsatt et al. 1993; Sirois 1994; Sirois and Doré 1997; Doré et al. 1998; Tremblay et al. 1999). Antibody MF243 was generously provided by Drs. Jilly F. Evans and Stacia Kargman (Merck Frosst Centre for Therapeutic Research; Pointe-Claire-Dorval, PQ, Canada). MF243 was raised in rabbits against ovine placental COX-2 and its selectivity for COX-2 has previously been characterized (Kargman et al. 1995; Tremblay et al. 1999).
Immunohistochemistry
Immunohistochemical staining was performed using the Vectastain ABC kit (Vector Laboratories) as previously described (Doré et al. 1993). Briefly, formalin-fixed tissues were paraffin-embedded and 3-μm-thick sections were prepared, deparaffinized in toluene, and hydrated through a graded alcohol series. Endogenous peroxidase was quenched by incubating the slides in 0.3% hydrogen peroxide in methanol for 30 min. After rinsing in PBS for 15 min, sections were incubated with normal goat serum (1:74 dilution) for 20 min at room temperature (RT). Primary antibodies diluted in PBS were applied (8223 at 1:100 dilution and MF243 at 1:7500 dilution) and sections were incubated overnight at 4C. Control sections were incubated with PBS or with nonimmune rabbit serum. After rinsing in PBS for 10 min, a bio-tinylated goat anti-rabbit antibody (1:222 dilution) was applied, and sections were incubated for 45 min at RT. Sections were washed in PBS for 10 min and incubated with the avidin DH-biotinylated horseradish peroxidase H reagents for 45 min at RT. After a PBS wash for 10 min, the reaction was revealed using 0.5 mg/ml diaminobenzidine tetrahydrochloride (DAB) in Tris buffer (pH 7.6) as the chromogen and 0.05% hydrogen peroxide as the substrate. Sections were counterstained with Gill's hematoxylin and mounted. Immunoreactivity was evaluated in a blinded fashion by two independent observers using a grading system where - = no staining, + = 0–10% positive cells, ++ = 11–30% positive cells, and + + + = ≥31% positive cells.
Solubilized Cell Extracts and Immunoblotting Analysis
Solubilized cell extracts were prepared as previously described (Sirois and Doré 1997). The protein concentration in each extract was determined by the method of Bradford (Bio-Rad Protein Assay). Proteins were resolved by one-dimensional SDS-PAGE and electrophoretically transferred to Hybond polyvinylidene difluoride (PVDF) membranes. Blocking of membranes was done using 5% non-fat dry milk in 0.1% TTBS (0.1% Tween-20, 10 mM Tris-buffered saline, pH 7.5) for 1 hr at RT, then washed twice for 2 min at RT with 0.1% TTBS. After blocking, membranes were incubated with anti-COX antibodies (8223 at 1:100 dilution and MF243 at 1:7500 dilution) diluted in 0.05% TTBS (0.05% Tween-20, 10 mM Tris-buffered saline, pH 7.5) containing 2% non-fat dry milk for 2 hr at RT. Membranes were incubated with a horseradish peroxidase-labeled donkey anti-rabbit secondary antibody (1:15,000 dilution) for 1 hr at RT. The membranes were washed and the bound secondary antibody was detected using the enhanced chemiluminescence (ECL) detection kit. The signal was visualized on Kodak Bio-Max X-ray film.
Cloning and Characterization of the Porcine COX-2 cDNA
The near-complete porcine COX-2 cDNA was cloned by a combination of three reverse transcription-polymerase chain reactions (RT-PCR) using the Access RT-PCR System (Promega) and following the manufacturer's protocol. Three RT reactions were performed with primers corresponding to the 5′-end, mid-portion, and 3′-end of the transcript (antisense primers 2, 4, and 7; Figure 1). RT reactions were performed at 48C for 45 min on 200 ng of total RNA extracted (TRIzol; Life Technologies) from a pig preovulatory follicle obtained 34 hr after IV administration of human chorionic gonadotropin (hCG; 1500 IU) (Côté et al. 2001). PCR reactions were performed under the following cycling conditions: 40 cycles of 30-sec denaturation at 94C, 1 min annealing at 54C, and 2 min elongation at 68C in an Omnigene TR3 SM5 thermal cycler (Hybaid; Franklin, MA). Porcine COX-2 sense and antisense primers were designed from exon sequences of porcine genomic clones or from partial cDNA sequences deposited in GenBank. After electrophoresis on a 1.0% TAE-agarose gel, the DNA fragments of interest were excised, purified with the Gene Clean Kit (Bio 101), and ligated into the pGEM-T easy vector (Promega) according to the manufacturer's instructions. DNA sequencing was performed on both strands with vector-based primers (T7 and Sp6) using an ABI autosequencer (Applied Biosystems; Foster City, Ca).
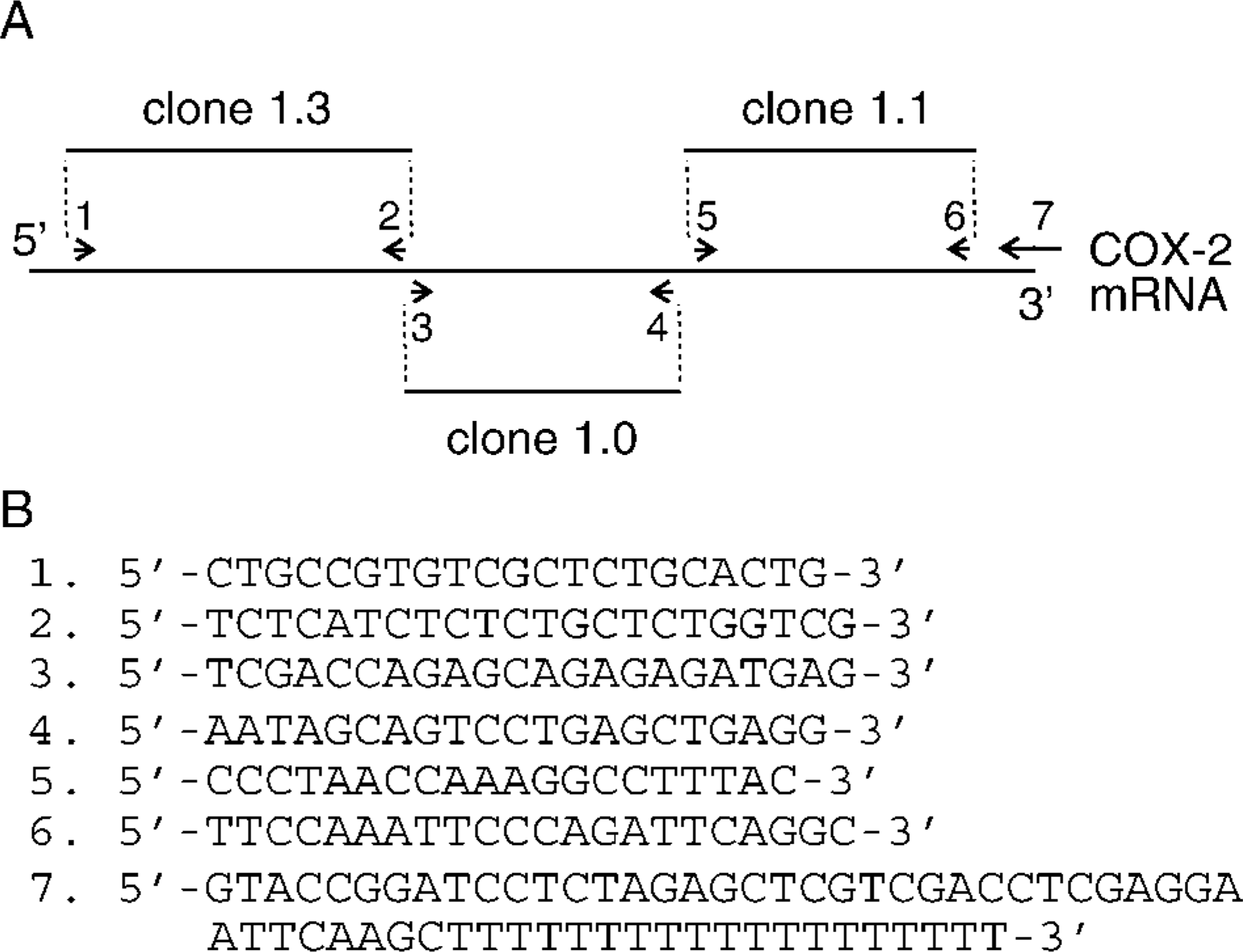
Cloning strategy for characterizing the porcine COX-2 cDNA.
Genomic library screening was used to characterize two small regions of porcine COX-2 that were not obtained by RT-PCR, i.e., the first 28 bp of the coding region as well as a 120-bp portion in the 5′-untranslated region (UTR), and a short 38-bp gap present between two PCR fragments (Figure 1). A porcine genomic library (Clontech) was screened following the manufacturer's protocol with a 5′ 1.5-kb EcoRI fragment of the bovine COX-2 cDNA (Liu et al. 2001) that was labeled with [α-32P]-dCTP using the Prime-a-Gene labeling system (Promega). Two positive clones (clones 2–1 and 3–1) isolated from an initial screen of 450,000 phage plaques were purified through secondary and tertiary screening. Sequencing reactions involving clone 2–1 and antisense primer 5′-GGA CTT GAG TGG TCC AAA CTG-3′ and clone 3–1 and antisense primer 5′-CTA TGA CTG CAG CCT TAA ACC-3′ were used to characterize the 5′-end of the coding region and the gap region, respectively.
Statistical Analysis
The Fisher's exact test was used to compare the frequency of COX-2 expression between normal stomachs and gastric ulcers. Statistical analyses were performed using the JMP Software (SAS Institute; Cary, NC).
Results
Characteristics of Pigs with Gastric Ulcers
The age of pigs with gastric ulcers ranged from 1 month to 4 years, with a mean of 6.9 ± 1.9 months. All ulcers were localized to the pars esophagea. Of the 35 cases, 23 animals (65.7%) were submitted alive and were sacrificed, 11 pigs (31.4%) were brought dead to the necropsy service, and the information was absent from the necropsy report of one animal. In addition to gastric ulcers, most pigs (n=30) were suffering from other pathological processes, including respiratory or gastrointestinal infections (n=28) and polyserositis (n=5). Examination of Whartin-Starry-stained sections to detect the presence of spiral bacteria revealed that bacteria morphologically compatible with Helicobacter spp. were present in the glandular mucosa adjacent to the ulcer in only one sample.
COX Expression in Normal Porcine Stomachs
To determine whether COX-1 and/or COX-2 were expressed under physiological conditions, immunohisto-chemical staining was performed on the pars esophagea of normal porcine stomachs (n=5). Results showed that some COX-1 was present in blood vessels (in endothelial cells and in smooth muscles of the media) and in fibroblasts in the submucosa in all normal stomachs (100%; Figures 2A and 2B). However, no COX-2 expression was detected in normal porcine stomachs (Figures 2C and 2D).
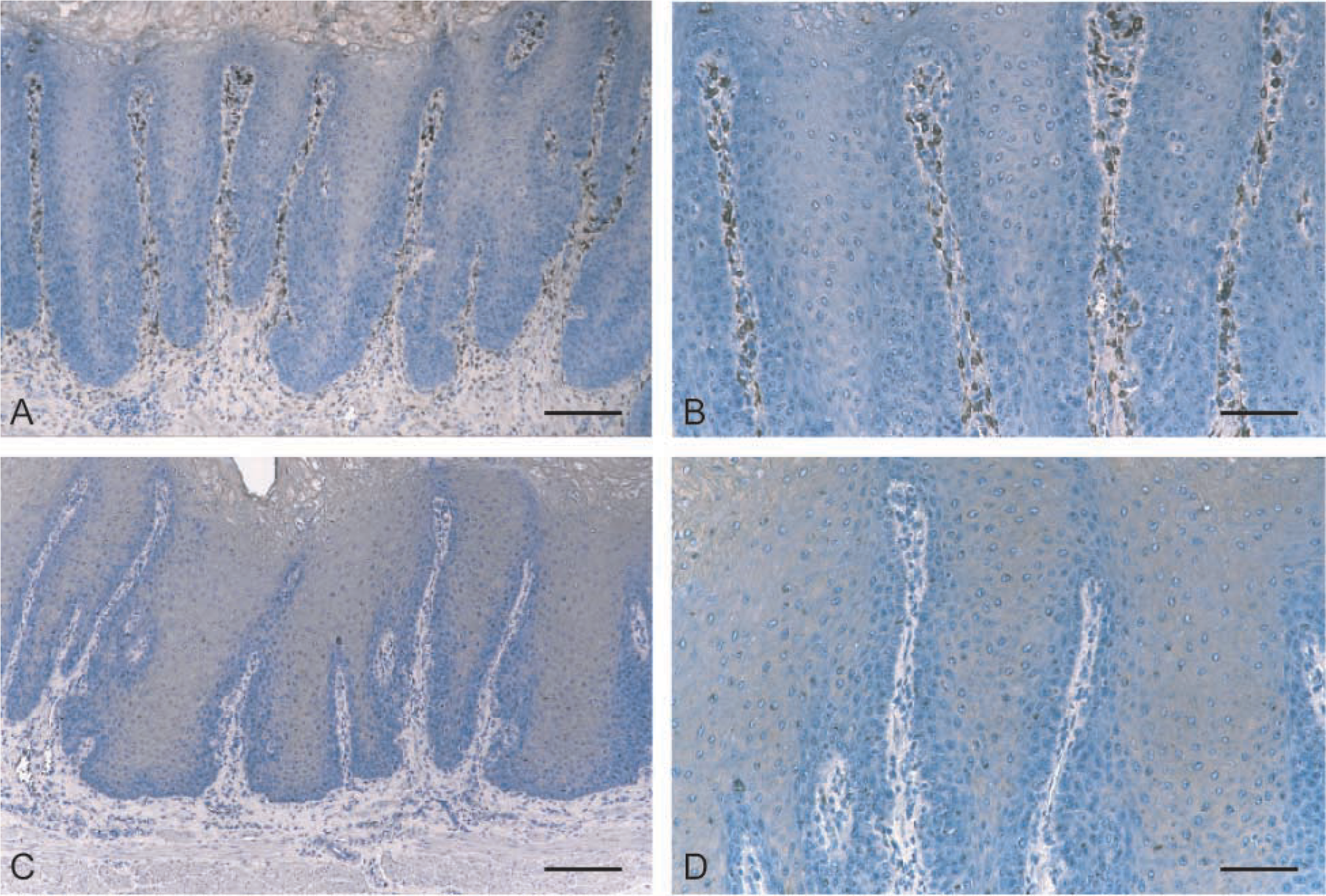
Expression of COX-1 and COX-2 by normal porcine stomach. Immunohistochemistry was performed on formalin-fixed sections of normal porcine stomachs as described in Materials and Methods.
COX Expression in Porcine Gastric Ulcers
COX-1-positive cells were found in the ulcer area in 27 of 35 cases (75%) (Figure 3E), while the ulcerated region from eight cases (25%) contained no COX-1-expressing cells. However, in contrast to normal stomachs in which no COX-2 was detected, 28 of the 35 (80%) gastric ulcers expressed COX-2 (p<0.01; Table 1). The extent and intensity of COX-2 staining varied among samples (Table I). COX-2 immunostaining was predominantly localized in the cytoplasm of elongated fibroblast-like cells that were localized in the granulation tissue proliferating under the ulcerated area (Figures 3A–3D).
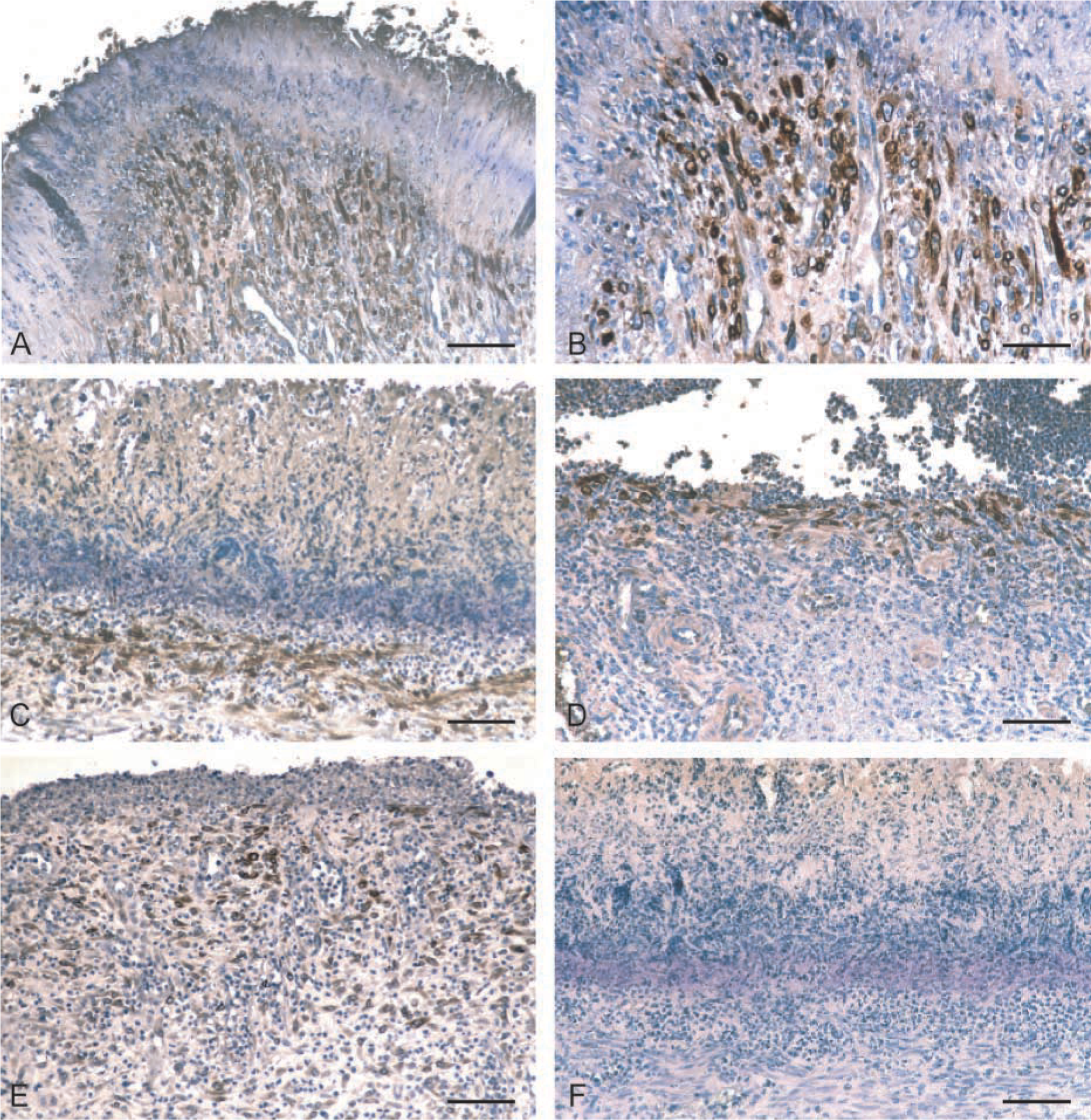
Expression of COX-1 and COX-2 in porcine gastric ulcers. Immunohistochemistry was performed on formalin-fixed sections of porcine gastric ulcers as described in Materials and Methods.
Characteristics of pigs with gastric ulcers a
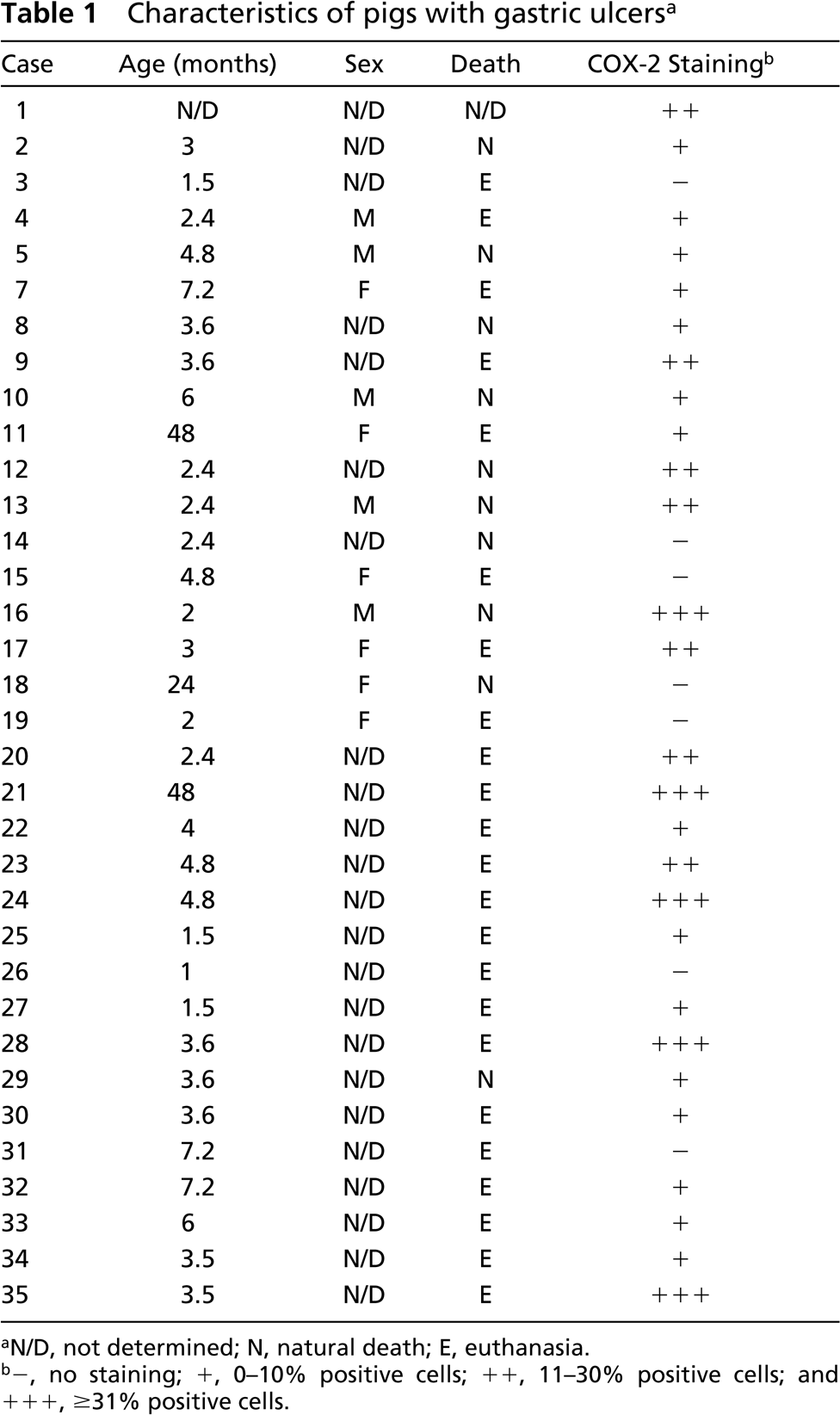
N/D, not determined; N, natural death; E, euthanasia.
-, no staining; +, 0–10% positive cells; ++, 11–30% positive cells; and + + +, ≥31% positive cells.
Immunoblotting of COX Isoforms in Porcine Gastric Tissues
To characterize each COX isoform, solubilized cell extracts were prepared from normal stomachs, gastric ulcers, and platelets, and proteins were analyzed by Western blotting. When a selective anti-COX-1 antibody was used, a 69,000 Mr band was detected in both normal and ulcerated stomachs (Figure 4A). A band of identical Mr was detected in porcine platelets (Figure 4A) and thus corresponded to porcine COX-1. When a selective anti-COX-2 antibody was used, no signal was detected in normal stomachs but COX immunoreactivity was observed in the two gastric ulcers (Figure 4B). Porcine COX-2 appeared as a 72,000–74,000 Mr doublet and a small 62,000 Mr band (Figure 4B) believed to correspond to a proteolytic fragment, as previously observed in other species (Sirois and Richards 1992; Sirois 1994). The absence of detectable COX-2 in porcine platelets is in keeping with reports in other species (Dubois et al. 1998).
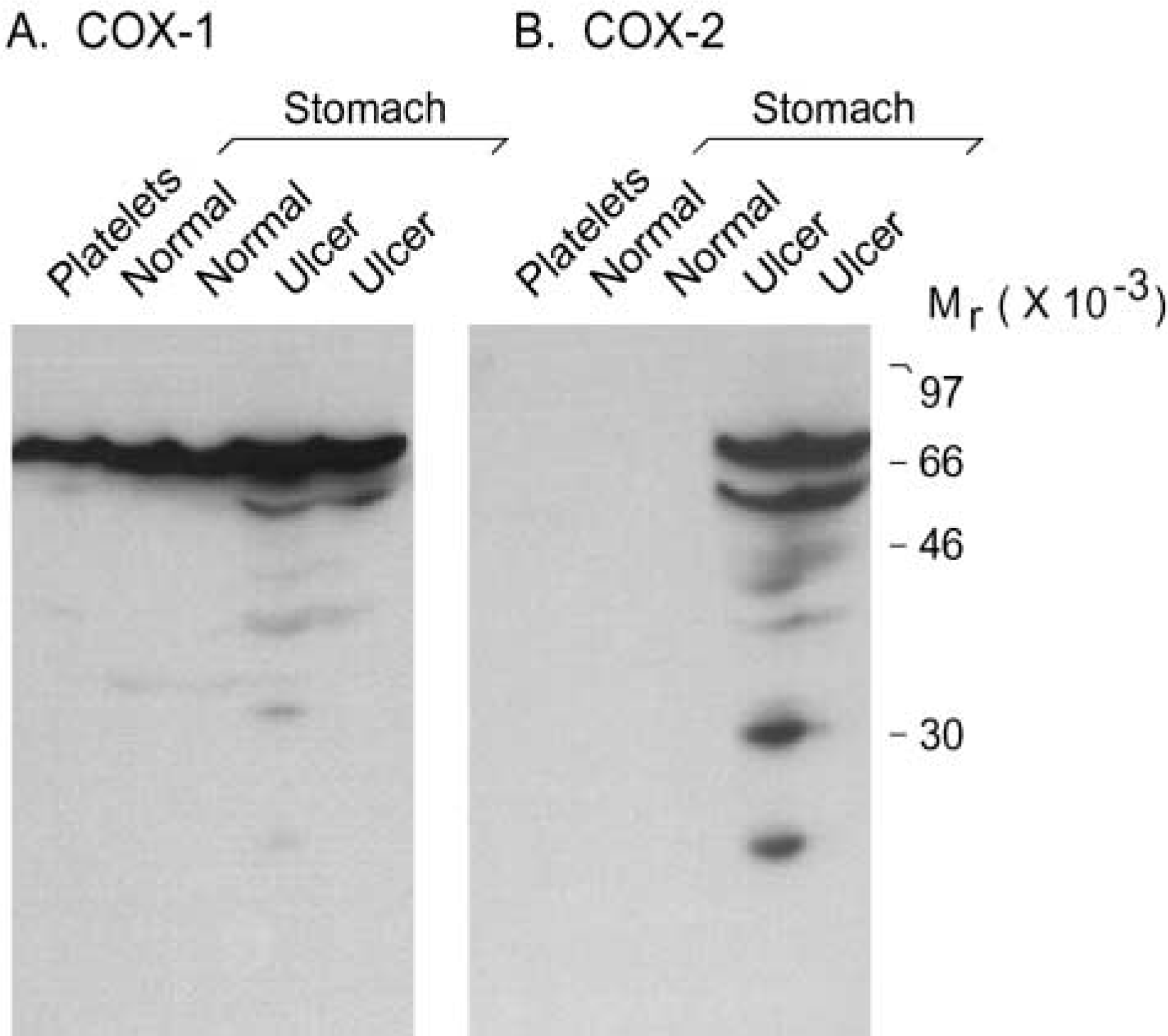
Immunoblotting analysis of COX-1 and COX-2 isoforms in porcine gastric tissues. Solubilized cell extracts were prepared from normal stomachs, two gastric ulcers (cases 24 and 35; Table 1), and porcine platelets, and were analyzed by one-dimensional SDS-PAGE and immunoblotting, as described in Materials and Methods. Duplicate blots were probed with antibody 8223 (selective for COX-1,
Cloning and Characterization of Porcine COX-2
The near full-length porcine COX-2 cDNA sequence was obtained from three RT-PCR reactions designed to generate contiguous DNA fragments (Figure 1). The first fragment (clone 1.3) corresponded to the 5′-region of the cDNA and extended from nucleotides 29 to 1334 of the coding region. The second fragment (clone 1.0), which overlapped with the first one, contained the remaining portion of the coding region as well as 541 nucleotides of the 3′-untranslated region. Lastly, the third fragment (clone 1.1) corresponded to the 3′-region of the cDNA, did not overlap with the second fragment, and contained an additional 1.1 kb of 3′-untranslated region. To complete the characterization of the porcine COX-2 cDNA, the remaining portion at the 5′-end of the cDNA and the 38-bp gap between clones 1.0 and 1.1 were obtained from genomic clones. Collectively, the porcine COX-2 cDNA contains a 5′-untranslated region of 120 bp, an open reading frame of 1815 bp (including the stop codon), and a long 3′-untranslated region of 1686 bp containing multiple repeats of the Shaw-Kamen sequence 5′-ATTTA-3′ (n=12; Figure 5).

Primary structure of the porcine COX-2 cDNA. The porcine COX-2 cDNA is composed of a 5′-untranslated region of 120 bp (lowercase letters), an open reading frame of 1815 bp (capital letters), and a 3′-untranslated region of 1686 bp. The nucleotide sequence was derived from clones isolated by RT-PCR and genomic screening as described in Materials and Methods. The translation initiation (ATG) and stop (TAG) codons are in bold type, Shaw-Kamen motifs (ATTTA) are underlined, and numbers appearing at left refer to the first nucleotide on that line. The nucleotide sequence was submitted to GenBank (accession number AY028583).
The amino acid sequence of pig COX-2 was deduced from the coding region of the cDNA, and comparisons were made with the human (Hla and Neilson 1992), bovine (Liu et al. 2001), equine (Boerboom and Sirois 1998), rat (Feng et al. 1993), mouse (Kujubu et al. 1991), rabbit (Guan et al. 1997), guinea pig (Bracken et al. 1997), mink (Song et al. 1998), and ovine (Zhang et al. 1996) homologues (Figure 6). The porcine COX-2 coding region encodes a 604 amino acid protein that is identical in length to all other known mammalian COX-2 proteins except for ovine COX-2, which has only 603 residues (Zhang et al. 1996). Comparisons between pig COX-2 and other mammalian homologues revealed more than 87% identity at the amino acid level, and all important structural and functional domains implicated in COX-2 function appeared to be conserved (Figure 6).
Discussion
This study reports for the first time the induced expression of COX-2 in naturally occurring gastric ulcers in pigs. COX-2-expressing cells were found in the majority (80%) of ulcerated stomachs, principally in the granulation tissue at the margins and underneath the ulcer. In recent years, several reports on the expression of COX-2 in experimentally induced gastric ulcers in laboratory animals have appeared, but COX-2 expression had not yet been documented in naturally occurring gastric ulcers in animals. In contrast to COX-2, COX-1 expression was similar in healthy vs ulcerated stomachs, in agreement with most previous reports (Mizuno et al. 1997; Kishimoto et al. 1998; Takahashi et al. 1998).
The implication of COX-2 in gastric ulcer healing has emerged in recent years. Mice and rat models of acetic acid- or ethanol-induced gastric ulcers have been used to document the expression of COX-2 in ulcerated stomachs (Mizuno et al. 1997; Shigeta et al. 1998; Takahashi et al. 1998). In these models, the use of NS-398, a specific COX-2 antagonist, inhibited PGE2 production in the ulcerated tissues and impaired the healing of ulcers (Mizuno et al. 1997; Shigeta et al. 1998; Takahashi et al. 1998). The induced expression of COX-2 has also been documented in rat models of ischemia-reperfusion-induced gastric ulcers and in gastric cryoulcers (Kishimoto et al. 1998; Schmassmann et al. 1998). The process of ulcer healing of the gastric mucosa involves the expression of various growth factors, including hepatocyte growth factor (HGF), which appears to be a key player of the repair process in the stomach (Kinoshita et al. 1995; Takahashi et al. 1996). Interestingly, recent studies have reported a possible association between HGF expression and COX-2 expression in ulcerated gastric tissues and have proposed that the action of HGF is mediated, at least in part, by COX-2 (Bamba et al. 1998; Horie-Sakata et al. 1998; Brzozowski et al. 2000). The present results demonstrating the expression of COX-2 in the granulation tissue of the ulcerated area suggest that COX-2 expression may also be involved in the repair process after gastric ulceration in the porcine species.
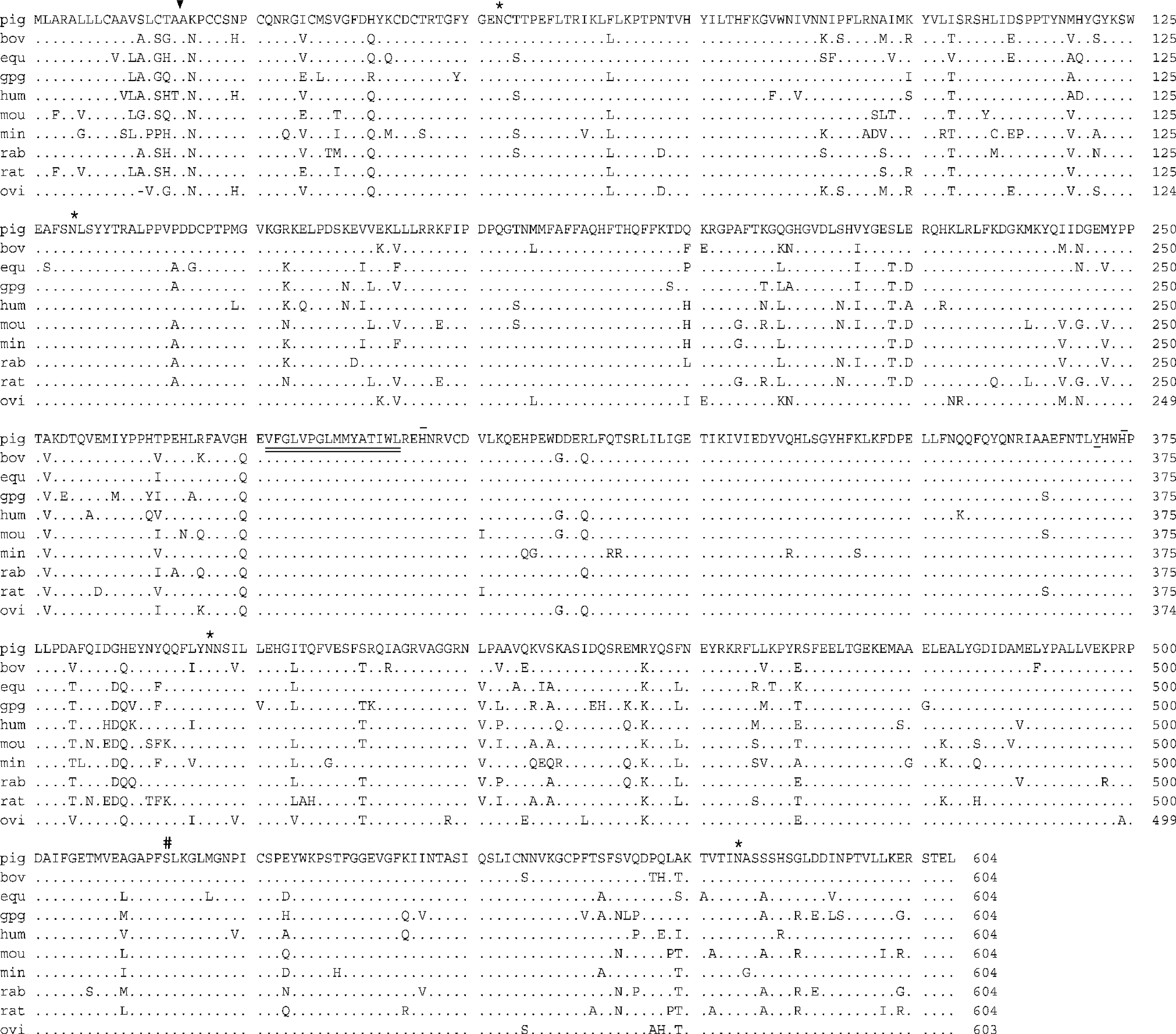
Predicted amino acid sequence of porcine COX-2 and comparison with other mammalian homologues. The deduced amino acid sequence of pig COX-2 is aligned with the human (hum), rat, mouse (mou), bovine (bov), ovine (ovi), equine (equ), rabbit (rab), guinea pig (gpg), and mink (min) homologues. Identical residues are marked with a printed period; the putative signal peptide cleavage site is indicated by an inverse triangle; putative N-glycosylation sites are marked with an asterisk; the putative transmembrane domain is doubled underlined; proximal and distal heme coordination residues are overlined, the tyrosine associated with the cyclo-oxygenase active site is underlined and the aspirin-acetylation site is indicated by a number sign. Numbers appearing at right refer to the last amino acid residue on that line.
In the present study, COX-2 expression was not observed in normal porcine stomachs. This finding is in keeping with the current dogma that COX-2 is inducible and not constitutively expressed like COX-1, and with reports stating that COX-2 protein is absent from normal gastric tissues (Kargman et al. 1996; Ristimaki et al. 1997; Lim et al. 2000). However, a controversy exists about the expression of COX-2 in normal gastric tissues because some studies have reported that normal gastric mucosa does express constitutive COX-2 (Iseki 1995; Zimmermann et al. 1998; Jackson et al. 2000). The reasons for these conflicting reports are unknown but could reflect, in part, differences in the specificity of the antibodies used and/or species-related differences.
The cellular localization of COX-2 in ulcerated stomachs was associated with cells that were morphologically compatible with fibroblasts, a finding also in agreement with previous reports (Schmassmann et al. 1998; Shigeta et al. 1998; Jackson et al. 2000). Immunolocalization of COX-2 has also been reported in inflammatory cells (macrophages/monocytes and granulocytes) as well as in endothelial cells (Schmassmann et al. 1998; Shigeta et al. 1998; Jackson et al. 2000). No COX-2 immunoreactivity was found in endothelial cells in this study, but several COX-2-positive vascular endothelial cells have been found in the granulation tissue proliferating in bovine gastric ulcerations (our unpublished data), suggesting the presence of potential species-specific differences in the cellular localization of COX-2.
In humans, H. pylori has been closely associated with chronic gastritis and peptic ulcer disease (Marshall and Warren 1984), and upregulated expression of COX-2 has been documented in various H. pylori-related pathologies, including gastritis, gastric ulcers, and premalignant and malignant gastric lesions (Sawaoka et al. 1998; Fu et al. 1999; McCarthy et al. 1999; Jackson et al. 2000; Sung et al. 2000; Tatsuguchi et al. 2000; To et al. 2001). Such an association between bacterial infection and gastric ulcers is not as clear in pigs, even though the presence of a spiral bacterium known as Helicobacter heilmanii has been reported in porcine gastric ulcerations. However, experimental infection of pigs with H. heilmanii did not induce gastric ulcerations (Krakowka et al. 1998). The detection of spiral bacteria in only one case in the present study questions the role of bacteria in the pathogenesis of porcine gastric ulcers.
This study is also the first to document the cloning and characterization of the full-length porcine COX-2 cDNA. Comparative analysis showed that the amino acid sequence of porcine COX-2 is very similar to that of other mammalian homologues, being 89%, 88%, 87%, 90%, 91%, 91%, 90%, 88%, 87% to human, rat, mouse, horse, cow, ovine, rabbit, guinea pig, and mink COX-2, respectively (Kujubu et al. 1991; Hla and Neilson 1992; Feng et al. 1993; Zhang et al. 1996; Bracken et al. 1997; Guan et al. 1997; Boerboom and Sirois 1998; Song et al. 1998; Liu et al. 2001). The porcine enzyme includes all known structural and functional domains involved in COX-2 activity. These domains include a putative transmembrane region, heme-coordinating histidines 295 and 374, the cyclo-oxygenase active-site tyrosine 371, the aspirin acetylation-site serine 516, and four putative N-linked glycosylation sites (DeWitt et al. 1990; Shimokawa and Smith 1991; Lecomte et al. 1994; Wennogle et al. 1995). The presence of multiple repeats of the Shaw-Kamen's sequence (5′-ATTTA-3′) in the 3′-untranslated region in the porcine COX-2 transcript is also a feature reported for COX-2 in several other species (Kujubu et al. 1991; Hla and Neilson 1992; Feng et al. 1993; Bracken et al. 1997; Zhang et al. 1996; Guan et al. 1997; Boerboom and Sirois 1998; Song et al. 1998; Liu et al. 2001). This motif has previously been shown to be present in several immediate early genes and to confer instability to mRNAs (Caput et al. 1986; Shaw and Kamen 1986).
In summary, our results provide the complete primary structure of porcine COX-2 and demonstrate that the enzyme is expressed in the stomach of the majority of pigs with naturally occurring gastric ulcers, being concentrated in fibroblast-like cells in the granulation tissue proliferating in the ulcerated area. These findings suggest that COX-2 is involved in the tissue repair process during gastric ulceration in pigs, and represent the first demonstration of COX-2 expression in an animal model of naturally occurring gastric ulcers.
Footnotes
Acknowledgements
Supported in part by the Natural Sciences and Engineering Council of Canada Grant 195188 (MD) and the Canadian Institutes for Health Research (CIHR) Grant MT-13190 (JS). JS is the recipient of a CIHR Investigator Award.
We thank Drs Jilly F. Evans and Stacia Kargman (Merck Frosst Centre for Therapeutic Research; Pointe-Claire-Dorval, Québec) for kindly supplying antibody MF243, Dr Bruce Downey and Fabienne Côté for providing porcine follicular tissues, and Dr Khampoune Sayasith and Nadine Bouchard for technical assistance.
