Abstract
Osteopontin (OPN) has been proposed to act as a substrate for osteoclast adhesion during bone resorption. The aim of the present study was to examine the presence and distribution of OPN at sites of resorption in traumatized radicular pulp. The upper first molars of 6-week-old male Sprague-Dawley rats were luxated and then repositioned in the original sockets. The animals were sacrificed by intracardiac perfusion at 10 and 14 days after tooth reimplantation. The teeth were decalcified in EDTA and then processed for embedding in paraffin for histochemistry or LR White resin for immunocytochemistry. Odontoclasts were identified by their multinucleated morphology and expression of tartrate-resistant acid phosphatase (TRAP). Osteopontin was immunolocalized using postem-bedding colloidal gold labeling with a chicken egg yolk anti-rat OPN antibody. After reimplantation of the teeth, TRAP-positive cells were present along the pulp dentin wall. Osteopontin was not consistently detected at exposed predentin/dentin surfaces. However, gold particles were often found at the margin of resorption lacunae. Labeling was also seen over the Golgi region and cytoplasmic vesicles of odontoclasts and of neutrophils and fibroblast-like cells. The results suggest that accumulation of OPN at the predentin/dentin surface is not a prerequisite for adhesion of odontoclasts to the wall substance and that recruited odontoclasts produce OPN locally to mediate cell and/or matrix events within the resorption lacuna.
Internal resorption on the coronal and root dentin surrounding the pulp tissue is a rare pathological process in human permanent teeth (Trope et al. 1994). The low incidence of internal resorption is in sharp contrast to the common occurrence of resorption along the external root surface under diverse pathological conditions. Dentin resorption is mediated by TRAP-positive multinucleated cells called odontoclasts (Lindskog and Pierce 1998). It is generally accepted that odontoclasts share common features with osteoclasts (e.g., development of clear zones and ruffled borders on the cellular side facing resorption lacunae) (Furseth 1968; Yaeger and Kraucunas 1969; Sasaki et al. 1988, 1989; Matsuda 1992) and expression of TRAP activity (Nilsen and Magnusson 1981; Wedenberg and Lindskog 1985; Wedenberg and Zetterquist 1987; Sasaki et al. 1988; Matsuda 1992; Sahara et al. 1996). The odontoclast, like the osteoclast, derives from mononuclear precursors that belong to the monocyte-macrophage lineage (Brudvik and Rygh 1993, 1994; Alatli et al. 1996). No TRAP-positive multinucleated cells are found in normal pulp tissue (Kondo et al. 1999), and the internal dentin wall is covered by a layer of odontoblasts.
Osteopontin (OPN), a sialic acid-rich phosphorylated glycoprotein, is found in bone and a variety of calcified tissues (reviewed by Sodek et al. 2000). It contains a GRGDS amino acid sequence that can mediate cell attachment through recognition by specific receptors on the cell membrane, such as αvβ3 integrin (Ross et al. 1993; Liaw et al. 1995). Extensive work utilizing immunohistochemistry and immunocytochemistry has identified the localization of OPN in bones and teeth (McKee and Nanci 1996a; Bosshardt et al. 1998) and at pathological calcification sites (Kohri et al. 1993; McKee et al. 1995). mRNA and protein expression studies have shown that various cell types, i.e., osteoblasts, osteocytes, fibroblasts, osteoclasts, macrophages, and bone marrow cells, can produce OPN (Sodek et al. 2000). Osteoblasts play a major role in the production of OPN that accumulates at bone interfaces (McKee and Nanci 1996b). It has also been reported that osteoclasts produce OPN and that the attachment of osteoclasts to bone surfaces is mediated, at least in part, by OPN-integrin interaction (Ross et al. 1993; Liaw et al. 1995; Srivatsa et al. 1997; Ellison et al. 1998). Osteopontin molecules can be either soluble or associated with fibrous matrices. Soluble molecules may participate in a feedback mechanism regulating the development and activation of clastic cells (reviewed in Giachelli and Steitz 2000).
Although there have been a number of studies on the association of OPN with matrix resorption in bone, cartilage, and external root (e.g., dentin and cementum), little is known about odontoclastic resorption along the predentin/dentin surface in the tooth pulp tissue. The aim of the present study was to elucidate the involvement of OPN in the pathogenesis of odontoclastic dentin resorption. We previously developed an experimental protocol to monitor time-sequential events taking place in the pulp chamber after reimplantation of rat molars (Kondo et al. 2000). This model was applied to detect immunocytochemically the appearance and localization of OPN along the disrupted pulp dentin.
Materials and Methods
Surgical Procedure
Six-week-old male Sprague-Dawley rats were used in the study (Charles River; Tokyo, Japan). All surgical procedures were carried out under general anesthesia with an IV injection of sodium pentobarbital (Nembutal, 30 mg/kg body weight; Abbott Lab, North Chicago, IL). The animals were placed in the supine position on a surgical board and the heads were held in a stable position with the aid of a head holder and ear rods. The upper first molars were extracted by luxating the tooth with the aid of a needle holder. The teeth were immediately placed in Hank's buffered solution and verified for signs of root fracture or damage under a stereoscope before repositioning in the original socket. All surgical procedures were carried out under sterile conditions and were completed within 15 min. The experimental protocol was approved by the Animal Care Committee of the Nippon Dental University.
Light Microscopy
The animals were sacrificed at 10 or 14 days after reimplantation of the molars because it had previously been shown that internal radicular resorption showed a high incidence at these time intervals (Kondo et al. 1999). They were anesthetized with Nembutal 30 mg/kg and perfused through the heart with 4% paraformaldehyde in 0.1 M cacodylate buffer (pH 7.2). The treated molar with the surrounding maxillary bone was dissected out en bloc, further immersed in the same fixative for 24 hr, and then demineralized at 4C in 10% ethylene-diamine-tetraacetate (EDTA) at pH 7.2 for 42 days. The specimens were dehydrated with ethanol and embedded in paraffin. Consecutive deparaffinized sections (4 μm thick) were cut in the sagittal plane and stained with either hematoxylin/eosin or Azan, or processed for histochemical detection of tartrate-resistant acid phosphatase (TRAP) activity using the azo dye method with 0.8% naphthol AS-BI phosphate as substrate (Yagishita et al. 2000).
Electron Microscopy
Tissue specimens for transmission electron microscope observation were fixed by perfusion with 1% glutaraldehyde in 0.08 M sodium cacodylate buffer (pH 7.4). The dissected maxillae were decalcified in 4.13% EDTA, pH 7.4 (Warshawsky and Moore 1967) for 28 days at 4C. The demineralized specimens were postfixed with potassium ferrocyanide-reduced osmium tetroxide (Neiss 1984) and processed for embedding in Epon 812. Semithin sections for survey (1 μm thick) were then cut with glass knives on a Reichert Ultracut E microtome, stained with toluidine blue, and observed under a light microscope. Ultrathin sections (80–100 nm thick) were cut with a diamond knife, double-stained with uranyl acetate and lead citrate, and then examined in a JEOL-2000FXII at 80 kV.
Immunocytochemistry
Some of the specimens fixed with glutaraldehyde were processed for immunocytochemistry. After demineralization, non-osmicated specimens were embedded in LR White resin (McKee and Nanci 1996a). Postembedding colloidal gold immunocytochemistry (reviewed in Bendayan 1995) was applied to localize OPN. All incubations were performed at room temperature. Thin sections of LR White-embedded tissues were mounted on formvar- and carbon-coated nickel grids and then placed for 10 min on a drop of 0.01 M PBS containing 1% ovalbumin, pH 7.2, to saturate nonspecific binding sites. The treated sections were then incubated for 12 hr at 4C on a drop of chicken egg yolk anti-rat osteopontin (diluted 1:150; Nanci et al. 1996). The grids were rinsed with PBS, floated on PBS–1% ovalbumin for 10 min, and incubated with gold-conjugated rabbit anti-chicken IgG (diluted 1:10; Electron Microscopy Sciences, Warrington, PA) for 60 min. As controls, some sections were incubated with gold-conjugated IgG alone or with non-immune serum followed by IgG-gold. Before observation in the transmission electron microscope, sections were stained with uranyl acetate and lead citrate.
Results
As the collection consisted of 17 roots of rat first molar (four medial root, six middle roots, and seven distal roots), the selected teeth showing signs of resorption amounted to six roots (two medial roots, three middle roots, and one distal root) in this study. Figure 1 illustrates representative morphological features of resorption lacunae developed along the pulp-side wall of root dentin at 10 and 14 days after tooth reimplantation. The pulp tissue in root canals of reimplanted teeth was severely degenerated or necrotic and was gradually replaced by granulation tissue infiltrated with inflammatory cells such as neutrophils and macrophages. The odontoblasts were damaged and detached from the predentin/dentin surface, but the predentin layer appeared intact. Various stages of resorption by TRAP-positive multinuclear cells were discerned along the exposed root wall. Histological examination of several root samples at 14 days after reimplantation showed the presence of fibroblast-like mononuclear cells in the resorption lacunae and the deposition of new matrix. A large portion of the exposed predentin remained free of resorption activity and was associated with spindle-shaped mononuclear cells. The following phases of resorption were observed: (a) initial contact phase, evidenced by the presence of multinucleated giant cells contact to the predentin; (b) active phase, evidenced by the presence of odontoclasts attached to the exposed dentin; (c) intermittent or resting phase, featured by the detachment or disappearance of TRAP-positive cells from the lacunae; and (d) repairing phase, characterized by the filling of lacunae with newly deposited matrix, on which a layer of cuboidal mononuclear cells was frequently seen.
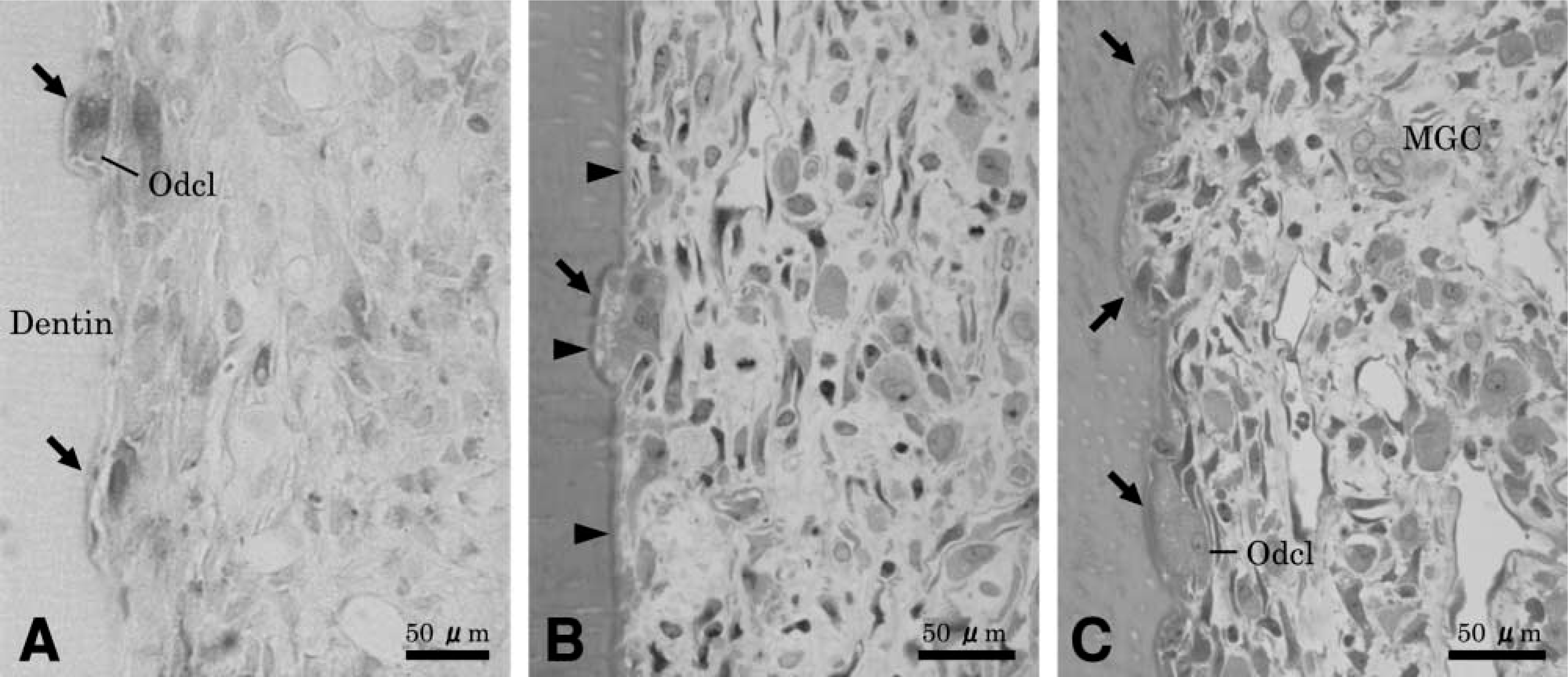
Light micrographs of resorption lacunae and associated cells found along the pulp dentin wall of the medial root of rat first molars at different time intervals after tooth reimplantation. (
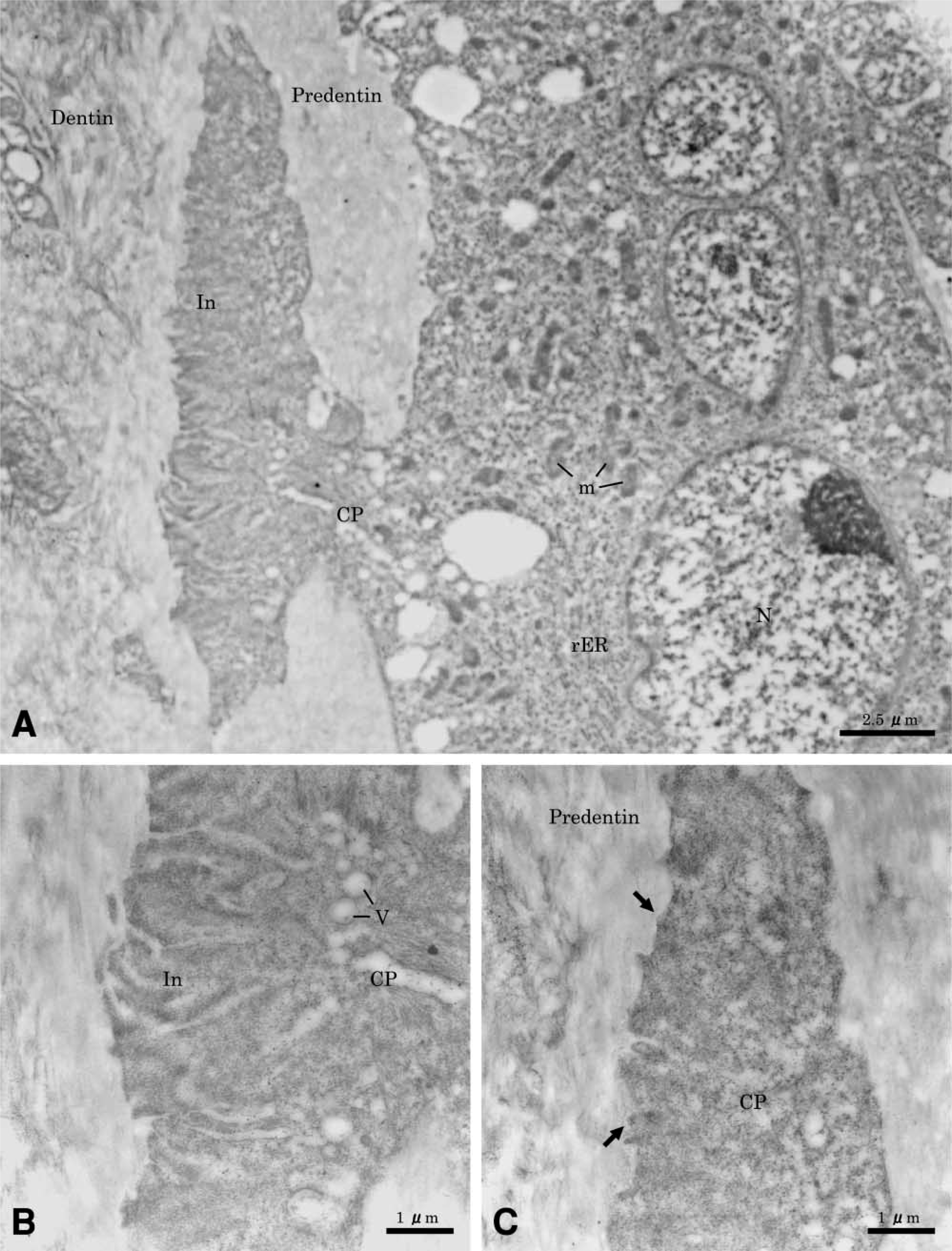
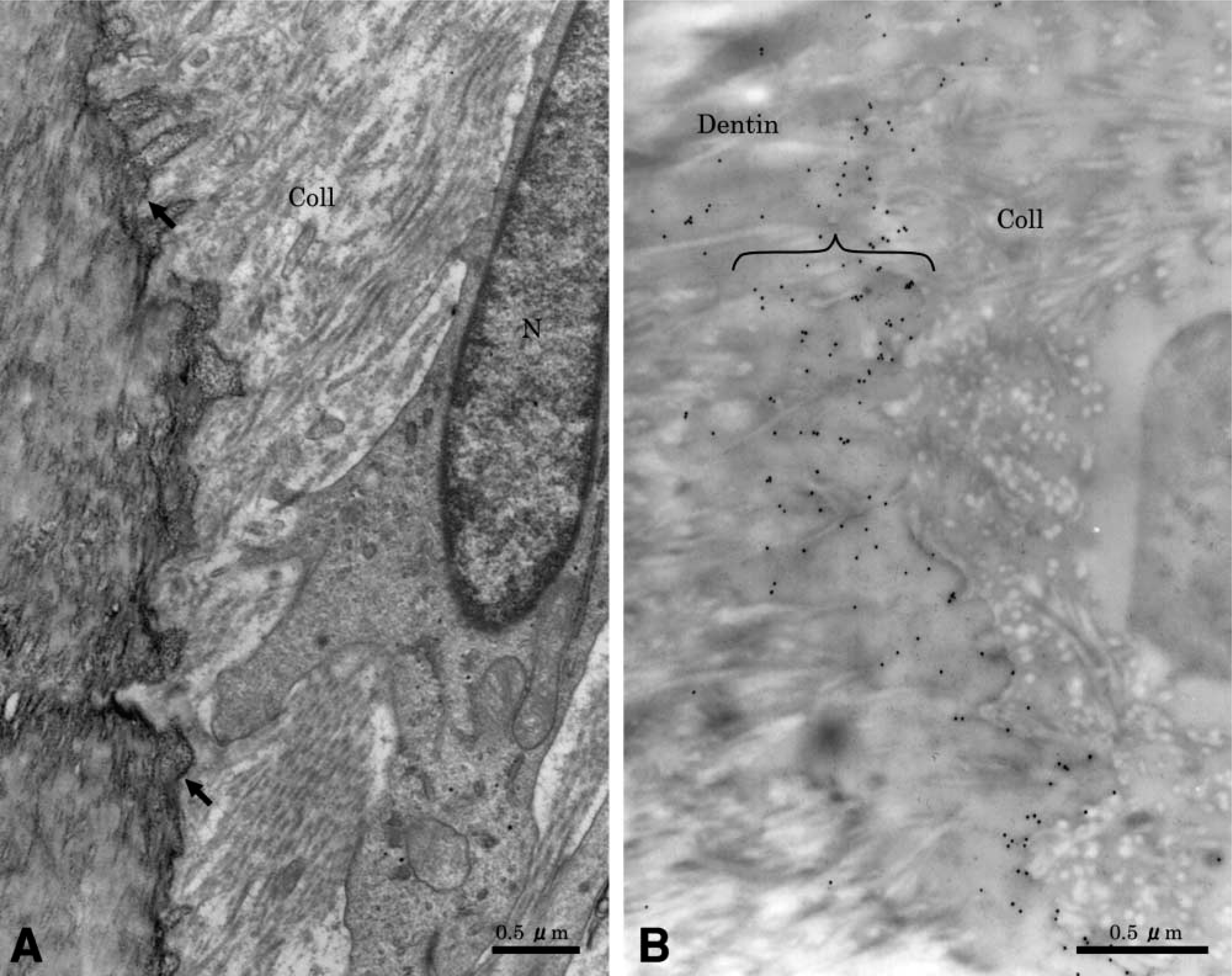
Quantitatively, at 10 days after treatment resorption lacunae were classified as one initial contact phase and seven active phases. At this time intermittent phase and repair phase lacunae were not yet observed. At 14 days after treatment, resorption lacunae were classified in six active phases, four intermittent phases and seven repair phases. At sites of initial contact the odontoblasts covered the root surface, multi-nucleated giant cells extended wide cytoplasmic processes through the dentin, and the body of the cell remained on the predentin surface. Abundant mitochondria, small vesicles, and rough endoplasmic reticulum characterized these odontoclasts. Initiation of ruffled border formation was observed within the area of the clear zone-like structure. Concomitant with these cell surface alterations, many vesicles appeared in the cell processes. There was no significant immunolabeling for OPN along the ruffled border-like and sealing zone-like areas of odontoclast (Figures 2B and 2C), and over the predentin and dentin matrix.
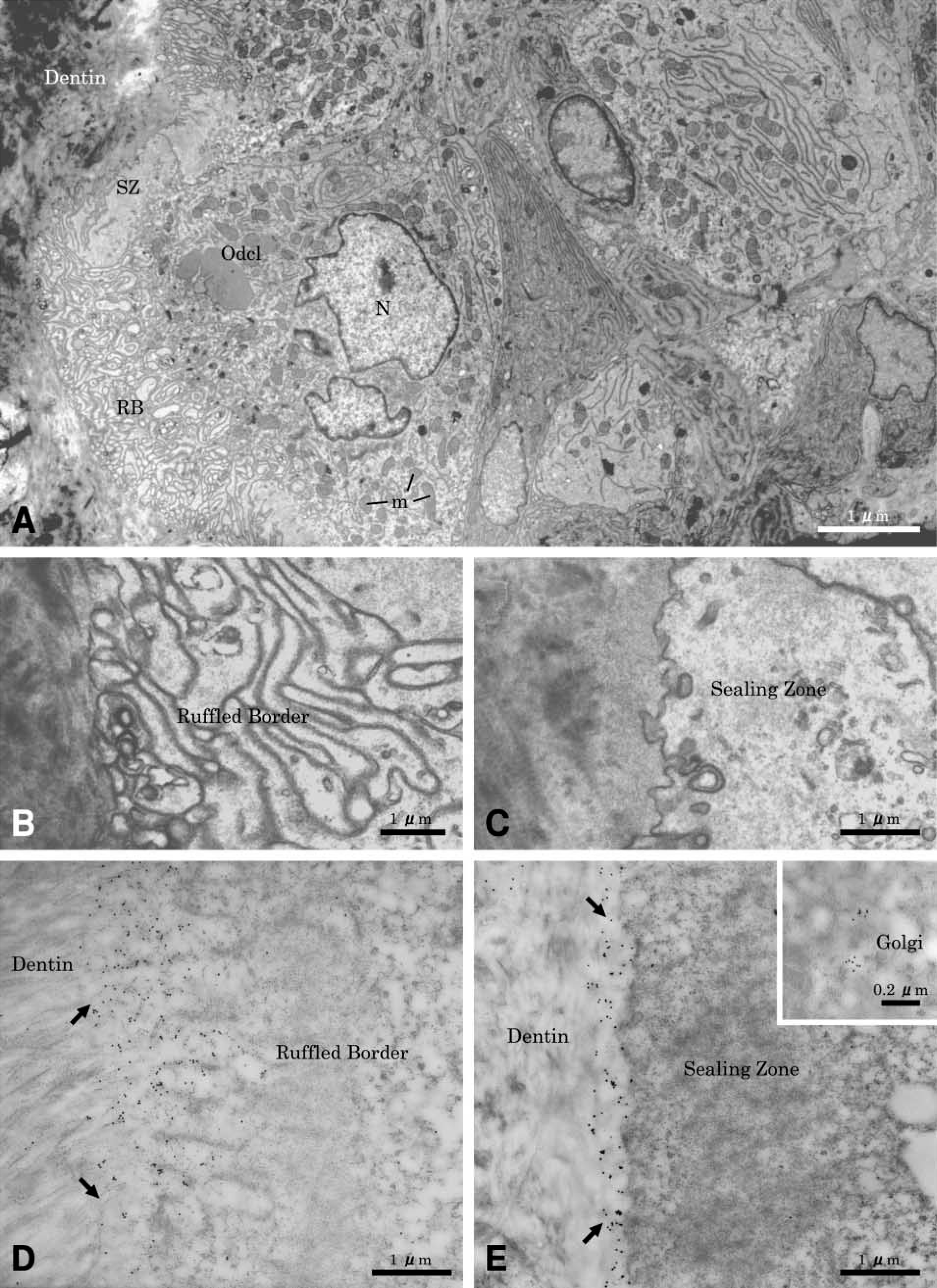
Sites showing active resorption exhibited cells on the root surface with ultrastructural characteristics of clastic cells (Figure 3A). They were multinucleated large cells, containing many mitochondria and several Golgi structures with variously sized vesicles and vacuoles in their cytoplasm. The mature odontoclasts had prominent cell polarity, especially at the resorbing surface. A sealing zone with scant cytoplasmic organelles was found at the periphery of the cell (Figure 3B), and a well-developed ruffled border (Figure 3C) was found. Extracellularly, there was diffuse labeling for OPN near the ruffled border area (Figure 3D), but labeling was more tightly distributed outside of the sealing zone (Figure 3E). Gold particles were also found over the Golgi areas at perinuclear regions of odontoclasts (Figure 3E, inset). In some cases, neutrophils frequently seemed to co-migrate and closely contacted active odontoclasts inside OPN-coated lacunae (Figures 4A and 4B). There was immunoreactivity over cytoplasmic vacuoles in neutrophils and along fine fibrils apposed to the lacunae wall.
Immunocytochemical preparations for osteopontin (OPN) showing an odontoclast apposed to the predentin wall 10 days after reimplantation. (
(
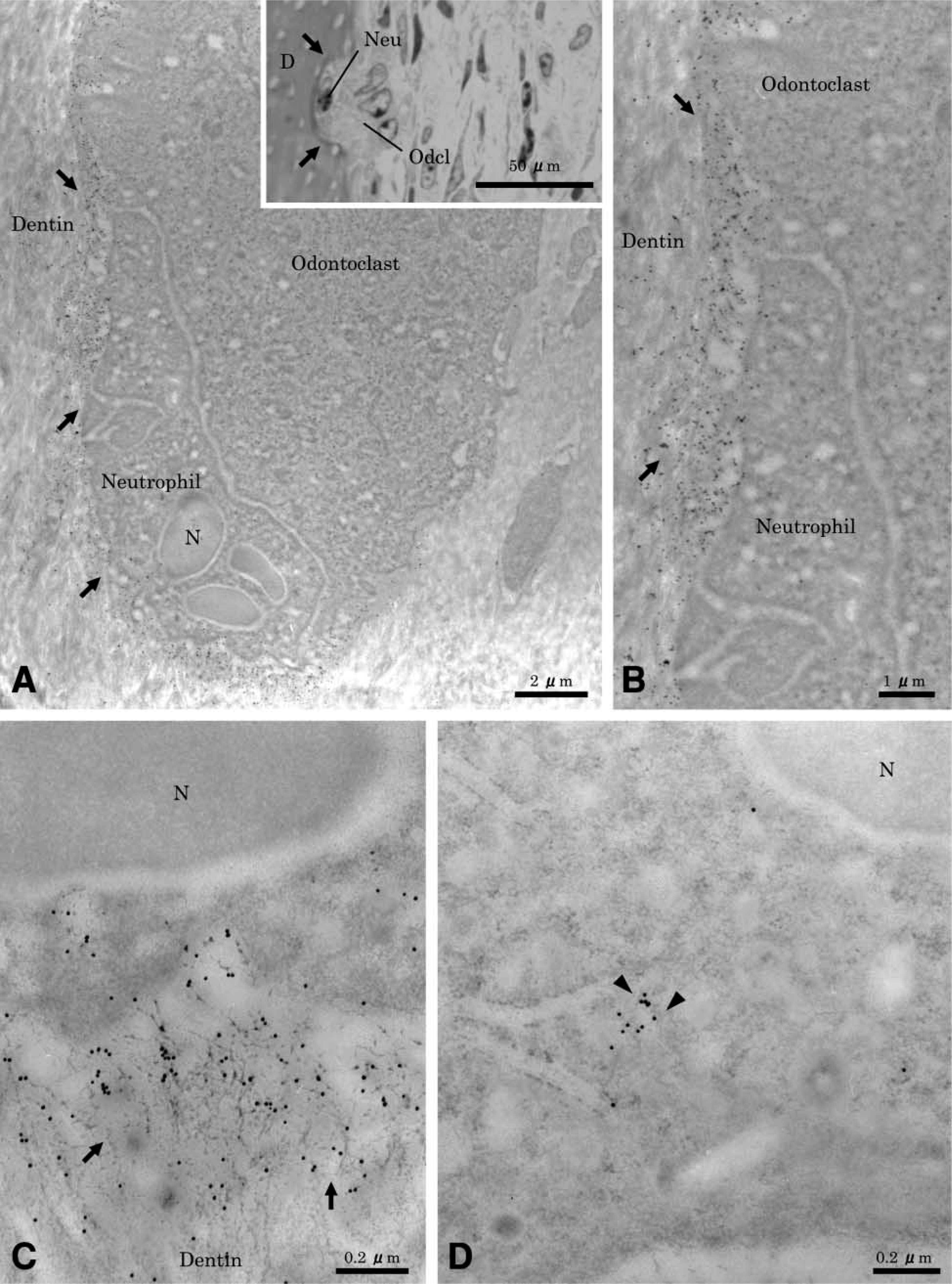
(
(
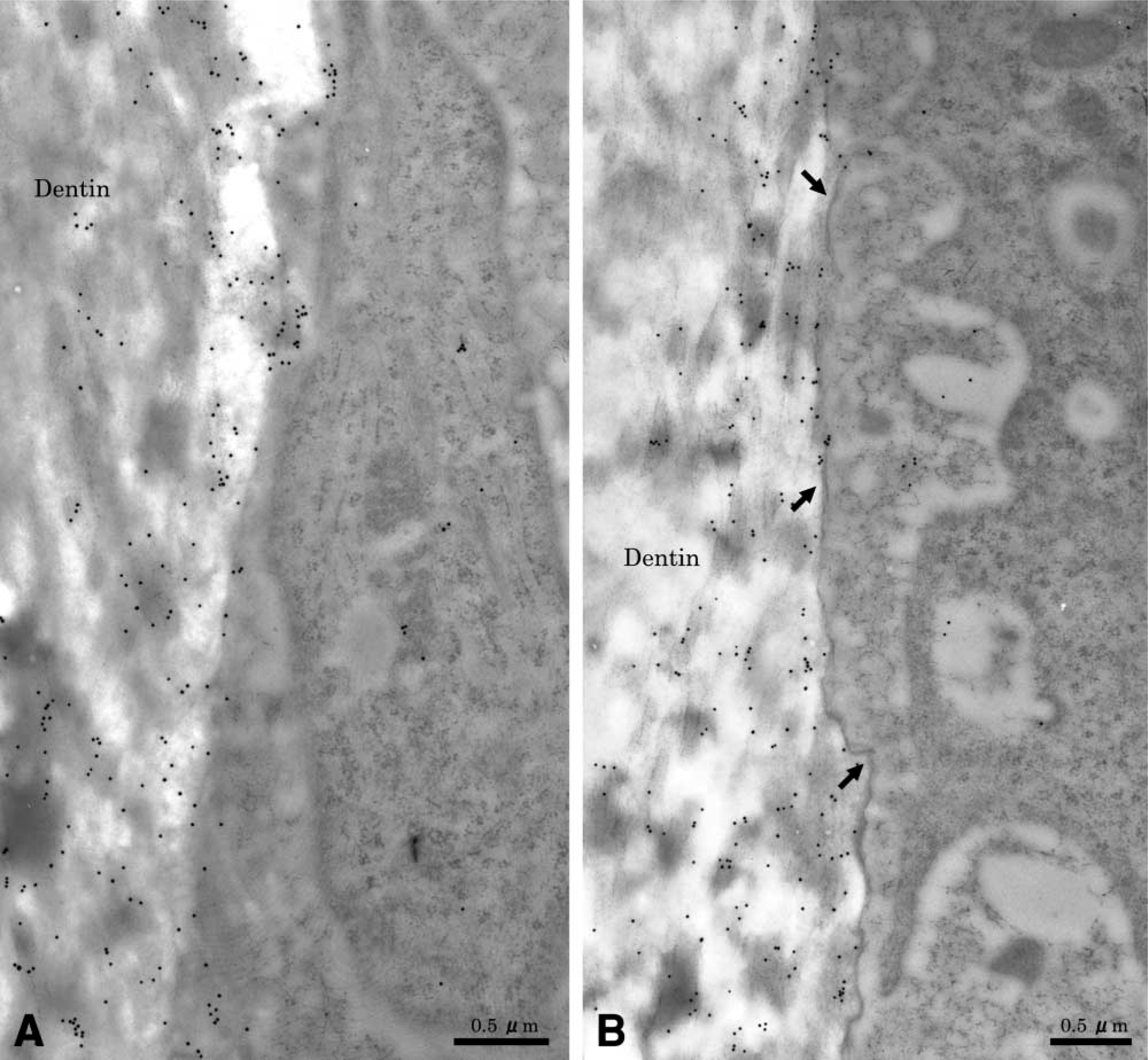
After odontoclasts detached from the resorption lacuna, the exposed dentin surface frequently showed intense metachromatic staining (Figure 1B). It consisted of an irregular dentin edge, an electron-dense line, and a layer of amorphous material, which on occasion was covered with an electron-dense line (Figure 5A). Short and irregular collagen fibers were present within the lacunae. These fibrils were oriented approximately at the surface of the lacunae. Fibroblast-like mononuclear cells were seen close to the resorbed dentin surface. OPN labeling was present over the old root matrix but dentin beyond the superficial region was not immunoreactive. The electron-dense line observed at the dentin surface was intensely labeled for OPN. When present, the amorphous material between old root matrix and the electron-dense line was very weakly or not at all immunoreactive.
(
After the damaged odontoblasts detached from the predentin, fibroblast-like cells without evidence of clastic resorption covered a large portion of the exposed predentin wall. Mononuclear cells close to the denuded dentin frequently established contacts with its surface. Immunostaining of the corresponding region revealed that underneath fibroblastic cells, gold particles were present over the dentin matrix, particularly at its mineralized surface in association with degraded collagen fibrils. In some areas an electron-dense line, which was labeled for OPN, was observed between the dentin matrix and degraded organic matrix. Gold particles were also found over cytoplasmic organelles of various types of infiltrating mononuclear cells, including fibroblastic cells (Figure 6).
Discussion
On the basis of the histological observations in the current and previous studies (Kondo et al. 1999), the following sequence of events appears to characterize the pathogenesis of root resorption in the pulp of rat molars after reimplantation: (a) degeneration and necrosis of pulp tissue; (b) extension of granulation tissue enriched in monocyte/macrophage/stromal cells from the apical portion of the tooth towards the crown; (c) detachment of damaged odontoblasts from the predentin surface; (d) appearance of TRAP-positive mononuclear and multinuclear cells related to blood vessels; (e) recruitment of multinucleated odontoclasts onto the damaged predentin/dentin wall; (f) formation of resorption lacunae; and (g) deposition of repair tissue. Figure 7 summarizes the sequence of events leading to the resorption and repair of dentin.
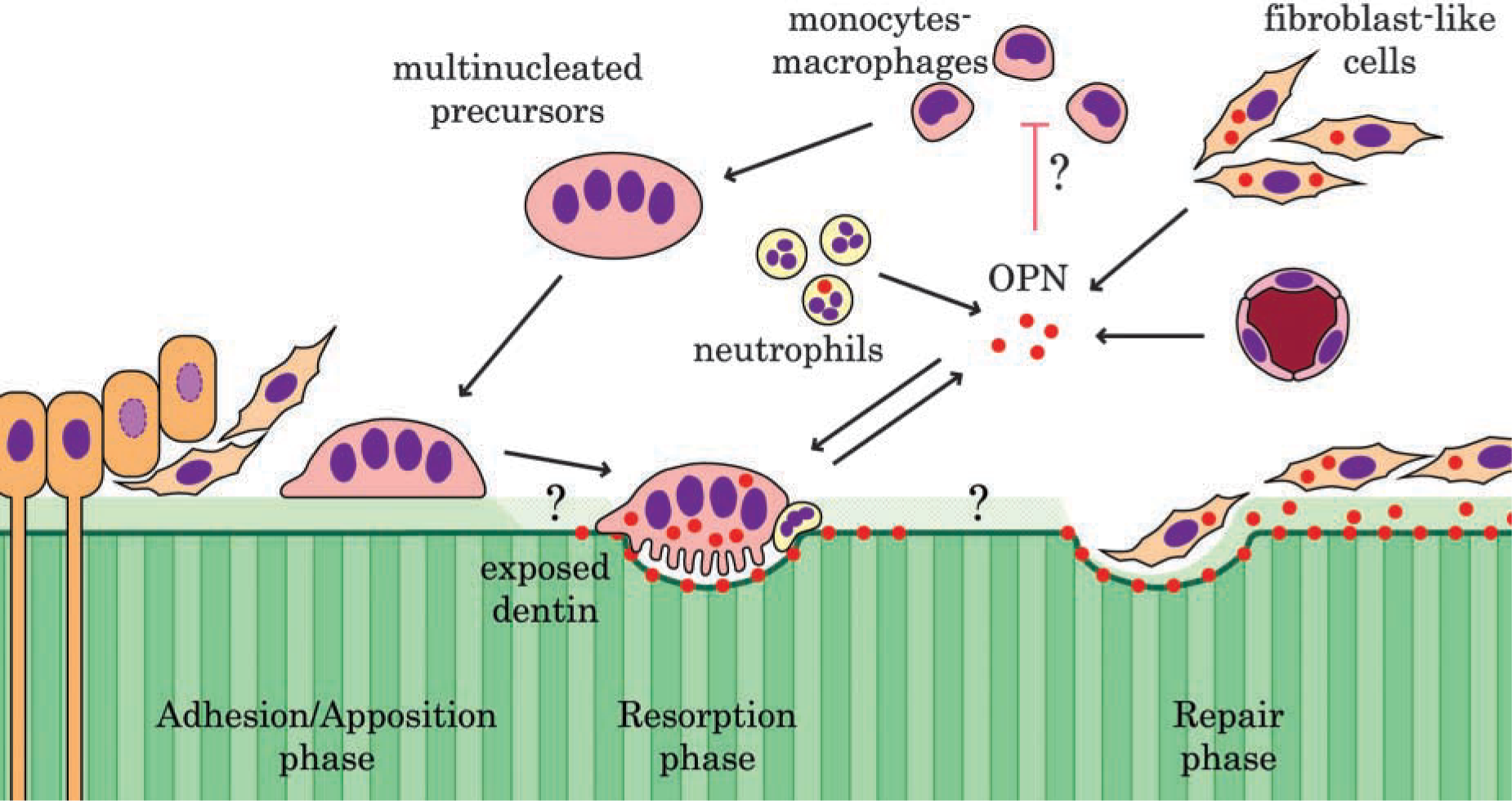
Schematic representation of the putative roles of osteopontin in development of odontoclastic dentin resorption. The mature odontoclasts, as well as fibroblast-like cells and neutrophils, secrete osteopontin (OPN). Osteopontin on the resorbed dentin surface likely facilitates recruitment and activation of mononuclear matrix-forming cells. Soluble OPN may be implicated in sequential cellular events taking place on the odontoblast-free predentin/dentin wall.
Osteoclasts express αvβ3 integrin (Flores et al. 1992; Ross et al. 1993), which binds strongly with the RDG motif of OPN. It has also been documented that OPN contains another cryptic domain, i.e., the SVVYELR sequence, that interacts with α9β1 integrin after exposure to thrombin cleavage (Smith et al. 1996; Yoshitake et al. 1999). Interestingly, α9β1 integrin is highly expressed on neutrophils (Taooka et al. 1999) and may, at least in part, be responsible for their migration onto the OPN-coated dentin surfaces.
Osteopontin-enriched electron-dense lines (i.e., cement line, lamina limitans) were not consistently detected at sites where odontoclasts contacted the exposed dentin wall. When present, OPN showed a diffuse distribution throughout the exposed dentin surface. The infrequent presence of OPN immunoreactivity suggests that the major mechanism for odontoclast adhesion onto dentin is not mediated by this non-collagenous protein. Osteoclasts have been shown to interact with uncalcified bone surface in the absence of a cement line (Mocetti et al. 2000), suggesting that this structure may not be an obligatory substrate for their recruitment. Osteoclasts may bind to native Type I collagen via α2β1 integrin and denatured collagen via αvβ3 integrin (Helfrich et al. 1996). They can also interact with other αvβ3 ligands with lower affinities, e.g., bone sialoprotein, but the weaker interactions may result in less efficient osteoclast function (Yoshitake et al. 1999). During internal root resorption, one of the alternative mechanisms is likely operating, particularly when odontoclasts interact with predentin. However, an accumulation of OPN was consistently observed on the lacunar surface, particularly at more advanced stages of resorption. This suggests that OPN is implicated in events that follow matrix degradation, such as recruitment/adhesion of mononuclear cells implicated in deposition of repair matrix and/or in cohesion between the old and new matrices. Taking into consideration that the OPN within lacunae appears while odontoclasts are still closely apposed to them and that it can be immunolocalized within their Golgi apparatuses, it is likely that odontoclasts are responsible, at least in part, for the OPN that accumulates on the resorbed dentin surfaces.
As regards the origin of OPN that coats the unresorbed exposed dentin wall, it may be produced locally by mononuclear cells (fibroblasts, macrophages, neutrophils) and free odontoclasts. Circulating molecules may also contribute to the formation of surface coatings. In this context, it has been demonstrated that exogenously administered OPN is incorporated into cement lines and newly formed bone (Brunski et al. 2000).
In conclusion, the present results indicate that the internal root surface of rat molars can be used as an experimental model for investigating the recruitment and activation of odontoclasts. The cue to development of odontoclastic dentin resorption is the replacement of the innate pulp tissue with granulation tissue. The initial attachment of odontoclasts to the predentin wall does not necessarily need preconditioning with OPN and appears to be mediated by alternative recognition mechanisms.
Footnotes
Acknowledgements
Supported by grants-in-aid for Scientific Research (12671866) from the Ministry of Education, Science and Culture of Japan (YS) and the Canadian Institutes of Health Research (AN).
We are grateful to Dr K. Sato for her great help in preparation of the manuscript and illustrations, Sylvia Zalzal for assistance with immunolabeling, and Micheline Fortin for technical assistance.
