Abstract
Phosphatidylinositol 3,4,5-trisphosphate [PtdIns(3,4,5)P3] is a second messenger produced in response to agonist stimulation. Traditionally, visualization of phosphoinositide polyphosphates (PtdInsPn) in living cells is accomplished using chimeric green fluorescent protein (GFP)-pleckstrin homology (PH) domain proteins, while PtdInsPn quantitation is accomplished by extraction and separation of radiolabeled cellular PtdInsPns. Here we describe preparation of a covalent protein-PtdIns(3,4,5)P3 immunogen, characterization of binding selectivity of an anti-PtdIns(3,4,5)P3 IgM, and immunodetection of PtdIns(3,4,5)P3 in stimulated mammalian cells. This antibody has greater than three orders of magnitude selectivity for binding PtdIns(3,4,5)P3 relative to its precursor, phosphatidylinositol 4,5-bis-phosphate (PtdIns(4,5)P2), and is therefore optimal for studies of cell function. The immunodetection in platelet-derived growth factor (PDGF)-stimulated NIH 3T3 cells was bench-marked against HPLC analysis of [3H]-myo-inositol-labeled cellular PtdInsPns. In addition, the changes in subcellular amounts and localizations of both PtdIns(3,4,5)P3 and PtdIns(4,5)P2 in stimulated NIH 3T3 fibroblasts and human neutrophils were observed by immunofluorescence. In insulin- or PDGF-stimulated fibroblasts, PtdIns(3,4,5)P3 levels increased in the cytoplasm, peaking at 10 min. In contrast, increases in the PtdIns(4,5)P2 levels were detected in nuclei, corresponding to the production of new substrate following depletion by phosphoinositide (PI) 3-kinase.
Keywords
PtdIns(3,4,5)P3 is a crucially important second messenger produced in response to cell stimulation, and a growing number of targets are being identified for this lipid signal (Toker and Cantley 1997; Scharenberg and Kinet 1998; Leevers et al. 1999; Czech 2000; Payrastre et al. 2001; Standaert et al. 2001). It is nominally absent in unstimulated cells and rapidly accumulates after stimulation with various agonists. Inhibition of the production of this lipid results in inhibition of many acute cell responses (Carpenter and Cantley 1996; Toker and Cantley 1997; Rameh and Cantley 1999). The physiological events associated with PtdIns(3,4,5)P3 include cell proliferation and transformation (Cantley et al. 1991; Valius and Kazlauskas 1993; Leevers et al. 1997), downstream effects of insulin action and alteration of glucose transport (Hara et al. 1994; Standaert et al. 2001), effects of growth factors on cell shape and motility (Wennstrom et al. 1994), T-cell signaling (Exley et al. 1994; Pages et al. 1994), apoptosis (Franke et al. 1997; Marte and Downward 1997), and endocytosis (Corvera 2001). Receptor-triggered signals have been shown to activate phosphoinositide 3-kinase (PI 3-kinase) (Vanhaesebroeck et al. 2001) and to generate PtdIns(3,4)P2 and PtdIns (3,4,5)P3. Although PtdIns(3,4,5)P3 is not a substrate for phospholipases, its level is controlled by both production (PI 3-kinase) and degradation by two phosphatases. SHIP, a phosphoinositide 5′-phosphatase (Pesesse et al. 1998), converts PtdIns(3,4,5)P3 into PtdIns(3,4,)P2, which can be either activating or inactivating depending on the downstream target (Aman et al. 1998). In addition, the involvement of PtdIns(3,4,5)P3 in cell proliferation emerged in connection with the role of the tumor suppressor protein, PTEN, as a 3-phosphatase that dephosphorylates PtdIns(3,4,5)P3 (Maehama and Dixon 1998,1999).
Membrane-associated PtdIns(3,4,5)P3 can be visualized in individual cells using a GFP fusion of the PtdIns(3,4,5)P3-specific (Baraldi et al. 1999) Btk-PH domain (Varnai et al. 1999; Marshall et al. 2001). The GFP-PH domain fusion strategy is powerful but has limitations (Balla et al. 2000). For example, it is unclear if all the pools of PtdIns(3,4,5)P3 can be detected, and the GFP-PH approach is useful only in cells that can be readily transfected. These fusions have not been employed in fixed cells for immunolocalization.
To quantify PtdIns(3,4,5)P3, the most common protocol requires cell stimulation in the presence of γ-[32P]-ATP, lipid extraction, separation by TLC, and quantification of the PtdIns(3,4,5)P3 produced by fluorography or scintillation counting. This approach is tedious and is limited to bulk analysis of millions of cells. Therefore, although one can detect PI 3-kinase in cells with isoform-selective anti-PI 3-kinase antibodies, there has not been a corresponding antibody to detect the lipid product itself. The availability of a reagent to permit direct determination of the cellular compartmentalization of PtdIns(3,4,5)P3 would provide an important new tool for studying PtdInsPn lipid signaling.
Antibodies against PtdIns(3,4,5)P3 would be indispensable for the study of signal transduction in cells and tissues. Unlike protein antigens, lipids are poorly recognized by antibodies. Therefore, the development of anti-lipid antibodies has progressed slowly. Antibodies against phospholipids have been produced by immunization with liposomes (Banerji and Alving 1990) or by adsorption of monomeric phospholipids to proteins (Tamamura et al. 1971; Maneta-Peyret et al. 1989), bacteria (Umeda et al. 1989), or acrylamide (Maneta-Peyret et al. 1989). Because of the apparent lack of specific lipid adsorption, antibodies produced by these methods may crossreact with different lipids (Umeda et al. 1989; Banerji and Alving 1990) and with other phosphate-containing moieties (Alving 1986).
For phosphoinositide and inositol derivatives, several research teams have reported antibodies and their uses in ELISA and cell-based studies. The first monoclonal antibody (MAb) specific to PtdIns(4,5)P2 was obtained by immunization with liposomes containing native PtdIns(4,5)P2. This MAb inhibited oncogene-induced mitogenesis (Fukami et al. 1988). In this liposomal preparation, the antigenic determinants were dominated by the inositol head group phosphorylation pattern with little diacylglycerol recognition. A second antibody was generated in an analogous fashion (Miyazawa et al. 1988) and has been employed in localization of nuclear PtdIns(4,5)P2 speckles that co-localized with PtdIns(4)P 5-kinase speckles (Boronenkov et al. 1998). More recently, an MAb that was highly PtdIns(4,5)P2-specific was prepared to liposomal PtdIns(4,5)P2 and provided striking evidence for detergent-inaccessible PtdIns(4,5)P2 in the nuclei of cells (Thomas et al. 1999). This nuclear PtdIns(4,5)P2 assembled in cell cycle-dependent structures involved pre-mRNA splicing (Osborne et al. 2001). An antibody to Ins(1,4,5)P3 was prepared by coupling a 6-O-amino-hexyl ether of Ins(1,4,5)P3 to bovine serum albumin (BSA) using glutaraldehyde (Shieh and Chen 1995). This polyclonal antiserum showed high selectivity for Ins(1,4,5)P3 and affinity-purified antibodies displayed IC50 values of 12 nM and 730 nM for InsP3 and InsP4, respectively. There have been no previous reports of antibodies elicited to liposomal PtdIns(3,4,5)P3, and efforts to produce these have been generally unsuccessful (unpublished results).
A different approach was required to convert PtdIns(3,4,5)P3, which appears to be a non-immunogenic hapten, to an immunogenic form. We selected an approach that was modeled on the successful preparation of anti-phosphatidylserine antibodies using an acyl-linked antigen (Diaz et al. 1998). We reasoned that an appropriate lipid-protein conjugate should elicit a more potent and specific immune response. We therefore modified the synthetic routes developed in our laboratories (Prestwich 1996) to synthesize sn-1-O-acyl-linked fluorophores and biotinylated PtdInsPn derivatives (Chen et al. 1996) to prepare an acyl-tethered derivative of PtdIns(3,4,5)P3. We describe here the preparation of this new PtdIns(3,4,5)P3 immunogen, the production of an MAb specific for PtdIns(3,4,5)P3, and the evaluation of this antibody in biochemical and cell-based assay systems. One particularly important finding with our anti-PtdIns(3,4,5)P3 antibody is that PtdIns(3,4,5)P3 levels remain elevated for several minutes in PDGF- and insulin-stimulated cells.
Materials and Methods
Synthesis of PtdIns(3,4,5)P3-cBSA and PtdIns(3,4,5)P3 Conjugate
sn-1-O-succinoyl-sn-2-O-stearoyl-PtdIns(3,4,5)P3 (PtdIns (3,4,5) P3-COOH) was synthesized according to modifications of protocols developed for the corresponding sn-1-aminohexanoyl derivatives (Chen et al. 1996). PtdIns(3,4,5)P3-COOH (4.4 mg) was dissolved in 1 ml conjugation buffer (0.1 M MES, 0.9% NaCl, 0.02% sodium oxide, pH 4.7), then mixed with 4.1 mg of lyophilized 10 mg/ml cationized BSA (cBSA) or keyhole limpet hemocyanin (KLH). Then 200 μl of 100 mg/ml EDC was added, gently mixed, and incubated at RT for 2 hr. After the reaction, the solution was desalted using a Presto Desalting Column (Pierce Chemical; Rock-ford, IL) and the conjugate eluted with 0.2 M ammonium bicarbonate (pH 8.0). The PtdIns(3,4,5)P3-cBSA or PtdIns(3,4,5)P3-KLH conjugate was detected by absorbance at 280 nm. The eluent was collected and pooled, sterilized with a 0.45-μm filter, and stored at —20C for further use. All dipalmitoyl and dioctanoyl PIPn derivatives were provided by Echelon Research Laboratories (Salt Lake City, UT).
Determination of Organic Phosphate
To aliquots (10–100 μl) of PtdIns(3,4,5)P3-cBSA or PtdIns (3,4,5)P3-KLH conjugate solution was added 30 μl of 10% Mg(NO3)2, and the material was ashed by heating with a bunsen burner. The tube was allowed to cool and 3 ml of 0.5 N HCl was added, followed by 0.7 ml of the detection mixture (10% ascorbic acid:0.42% ammonium molybdate, 1:6 v/v). A phosphate standard was employed for calibration. The absorbance at 820 nm was measured after 30 min.
Production of the Monoclonal Antibody
Eight female mice (BALB/c) were immunized with 1 mg of PtdIns(3,4,5)P3-cBSA conjugate in 1.0 ml of ammonium bicarbonate (pH 8.0) emulsified with an equal volume of Freund's complete adjuvant by multiple IP and SC injections. Booster injections were given at 2-week intervals with the same amounts of PtdIns(3,4,5)P3-cBSA prepared with incomplete Freund's adjuvant. After the fourth boost the animals were bled and the antiserum titer was tested by ELISA. Three days after the final booster injection, positive-titer mice were sacrificed, and spleen cells were collected and fused with sp2/0 cells by the standard polyethylene glycol method. Two weeks after fusion the culture supernatants of the hybridomas were examined by ELISA. After cloning three times by limiting dilution, a hydridoma clone named RC6F8 was established. The cells were inoculated into incomplete Freund's adjuvant-primed mice and the resultant ascites fluid was used to prepare purified immunoglobulin according to the instructions of Immunopure IgM purification kit (Pierce).
Enzyme-linked Immunoabsorbent Assay
Ten microliters of 100 μg/ml PtdIns(3,4,5)P3 in 95% ethanol was added to microtiter wells and the ethanol was then evaporated in air, followed by high vacuum for 5 min. The plates were blocked with 5% non-fat milk in TBS (20 mM Tris, 0.15 M NaCl, pH 8.0) at RT for 1 hr. After washing three times with 5% nonfat milk in TBS, 50 μl of anti-PtdIns(3,4,5)P3 antibody RC6F8 in TBS was applied to the wells at RT for 1 h. Wells were washed three times with 5% non-fat milk, and then 50 μl of anti-mouse IgM-peroxidase conjugate was added and incubated at RT for 1 hr. Wells were washed four times with TBS, 100 μl of TBM substrate solution (Sigma Chemical; St Louis, MO) was added, and absorbance at 370 nm was measured after 15 min.
Photoaffinity Labeling of RC6F8 Antibody
The purified RC6F8 IgM and a standard commercial mouse IgM (0.68 μM) were incubated with 20 μl of TBS containing 0.1 μCi (0.28 μM) of [3H]-BZDC-triester-PtdIns (3,4,5)P3 (Gu and Prestwich 1996; Chaudhary et al. 1998; Feng et al. 2001). Samples were equilibrated at 4C for 15 min in a 96-well plate. The wells in the plate were aligned with the axis of a UV light with a 2-cm distance between the bottom of the wells and the bulb, and samples were irradiated (360 nm at 1900 μW/cm2) for 45 min at 4C. Next, 6 × SDS sample buffer was added to the samples and the proteins were separated by SDS-PAGE on 12% Laemmli gels. The gels were stained with Coomassie blue, destained, and impregnated with En3Hance (NEN Life Science; Boston, MA) according to the manufacturer's instructions, dried, and exposed to Kodak BioMax X-ray film for 8 days at — 80C. No covalent incorporation of the photolabel occurred in the absence of UV irradiation. Fluorograms were digitized on a UMAX-UC 840 scanner using Adobe Photoshop.
Measurement of PtdIns(3,4,5)P3 by HPLC
NIH 3T3 fibroblasts were grown to at least 60% confluency in 75-cm2 flasks, labeled, and phosphoinositide levels analyzed essentially as described (Stratford et al. 2001). Cells were labeled for 36 hr with [2-3H]-myo-inositol (20 μCi/ml) in inositol-free DMEM + 10% calf serum. After 24 hr the medium was removed and replaced with fresh medium containing [2-3H]-myo-inositol. Before stimulation with PDGF (50 ng/ml), cells were serum-deprived for 2 hr in inositol-free and serum-free DMEM containing 0.2% BSA and 10 μCi/ml [2-3H]-myo-inositol. Cells were then treated with PDGF as indicated in the figure legends. Extraction and deacylation of lipids were performed as previously described (Batty and Downes 1996) and the glycerophosphoinositol phosphate head groups were analyzed by HPLC (Hama et al. 2000).
Immunofluorescence (IF) Methodology
NIH 3T3 cells at logarithmic stage on coverslips were serum-starved overnight and stimulated with insulin (100 ng/ml) or PDGF (50 ng/ml). Reactions were stopped by washing the cells with cold TBS, and cells were processed for IF. Cells on glass coverslips were fixed with 2% formaldehyde and then permeabilized with 0.5% Triton X-100 in TBS. After blocking with 10% goat serum in TBS, either RC6F8 MAb ascites (1:50 dilution) or 10F8 (1:5000 dilution) was added and incubated at RT for 1 hr. After washing three times with blocking solution, FITC-labeled anti-mouse IgM (1:2000 dilution) was added and incubated at RT for 1 hr. After the cells were washed three times with deionized water, the fluorescence in the cells was observed using a confo-cal microscope (BioRad MRC 1024; Richmond, CA) (see below). Purified RC6F8 anti-PtdIns(3,4,5)P3 IgM and 2C11 anti-PtdIns(4,5)P2 IgM are now available from Echelon Research Laboratories.
Cell Stimulation
Neutrophils (500 μl of 5.5 × 106 cells/ml), freshly isolated by centrifugation (Vlahos and Matter 1992), were used per time point. Cells were activated by adding 5 μl of 10 μM fMLP. Stimulation was terminated by addition of 500 μl of 4% paraformaldehyde. Cells were fixed at RT for 20 min and then cytospun onto slides coated with Vectabond (Vector Labs; Burlingame, CA) and permeabilized with 0.5% Triton X-100 in TBS, then blocked with 10% goat serum in TBS. Samples were incubated with the primary mouse anti-PtdIns(3,4,5)P3 IgM or the anti-PtdIns(4,5)P2 IgM, washed, and incubated with the secondary goat anti-mouse IgM-fluorescein conjugate. Mounting medium and a coverslip was placed over the cells and visualized using a laser scanning confocal microscope.
Microscopy
Cells were examined using an inverted microscope (Nikon) and a BioRad laser scanning confocal microscope system (MRC 1024) with Laser Sharp acquisition software. Images of animal cells were collected using a ×60 oil immersion objective. Post-acquisition enhancement of images was not performed except for gray-scale conversion to color using Laser Sharp software or Confocal Assistant software.
Results
Preparation of Anti-PtdIns(3,4,5)P3 Antibodies
To increase the immunogenicity of PtdIns(3,4,5)P3 and to preserve the structural integrity of phosphoinositides, it is important to construct conjugates that do not modify the polar head group. Although several laboratories have tried to accomplish this by using lipids absorbed non-covalently to carriers (Maneta-Peyret et al. 1993; Rote et al. 1995), the resulting antibodies lacked specificity and crossreacted with many cellular antigens. For our approach, it was of paramount importance to obtain antibodies that could recognize the diacylglycerol-linked head group, not simply the inositol polyphosphate. To this end we employed a strategy in which a reactive carboxylic acid group was introduced into the sn-1-O-acyl position.
A PtdIns(3,4,5)P3-cationized BSA (cBSA) conjugate was synthesized, as illustrated in Figure 1. First, a diacylglycerylphosphoramidite was prepared bearing a benzyl ester of succinic acid as the sn-1-O-acyl group and stearate as the sn-2-O-acyl group. This phosphoramidite was coupled to the protected inositol head group and oxidized to the phosphate to yield the fully protected PtdIns(3,4,5)P3 analogue (Chen et al. 1996). Hydrogenolysis of the benzyl esters and benzyl ether protecting groups furnished the carboxylic acid-functionalized PtdIns(3,4,5)P3 derivative sn-1-O-succinoyl-sn-2-O-stearoyl-PtdIns(3,4,5)P3. The PtdIns(3,4,5)P3 derivative was dissolved in conjugation buffer (0.1 M MES, 0.9% NaCl, 0.02% sodium azide, pH 7.4) and coupled to the carrier protein, cBSA, using [3-(dimethyl-amino)-propyl]3-ethylcarbodiimide hydrochloride (EDCI) at RT. The immunogen was desalted and the presence of the PtdIns(3,4,5)P3-cBSA conjugate in fractions was detected spectrophotometrically at 280 nm. Purified immunogen was sterilized by ultrafiltration and stored at —20C. A bioconjugate of this succinoyl PtdIns(3,4,5)P3 analogue with KLH was prepared in a similar fashion. The molar ratios of cBSA and KLH to PtdIns(3,4,5)P3-COOH were calculated to be 1:190 and 1:400, respectively, based on phosphate and protein analysis (data not shown).
Mice were immunized with the PtdIns(3,4,5)P3-cBSA conjugate and serum was collected after four booster injections given every 2 weeks. Three days after the final booster injection, mice with high polyclonal anti-PtdIns(3,4,5)P3 antibody titers were sacrificed and the spleens were used to prepare hybridomas. Hybridomas were subcloned and selected for anti-PtdIns(3,4,5)P3 binding.
Ligand Binding Selectivity
The monoclonal anti-PtdIns(3,4,5)P3 antibody named RC6F8 (IgM class) was obtained from mouse ascites fluid. Three different methods were employed to test the specificity of the antibody. First, using ELISA, the antibody RC6F8 specifically recognized PtdIns (3,4,5)P3 (Figure 2). The RC6F8 IgM had very low affinity for PtdIns(4,5)P2, for the two monophosphates PtdIns(3)P and PtdIns(4)P, for the nonphosphorylated PtdIns, and for other phospholipids. In these assays, lipids were nonspecifically adsorbed to the plastic microtiter plate wells. The dipalmitoyl (di-C16)-PtdIns (3,4,5)P3 was essentially water-soluble at the low coating concentrations employed, and adhered poorly compared to the essentially insoluble mono- and bisphosphates di-C16-PtdIns(4,5)P2, di-C16-PtdIns(3)P, di-C16-PtdIns(4)P, and di-C16-PtdIns. Therefore, even though the same amount of each lipid was coated in the wells, the amount of the PtdIns(3,4,5)P3 to be tested in the wells was less than other lipids after the washing steps in the ELISA. This screen therefore underestimates both the affinity and the selectivity of the MAb for PtdIns(3,4,5)P3.
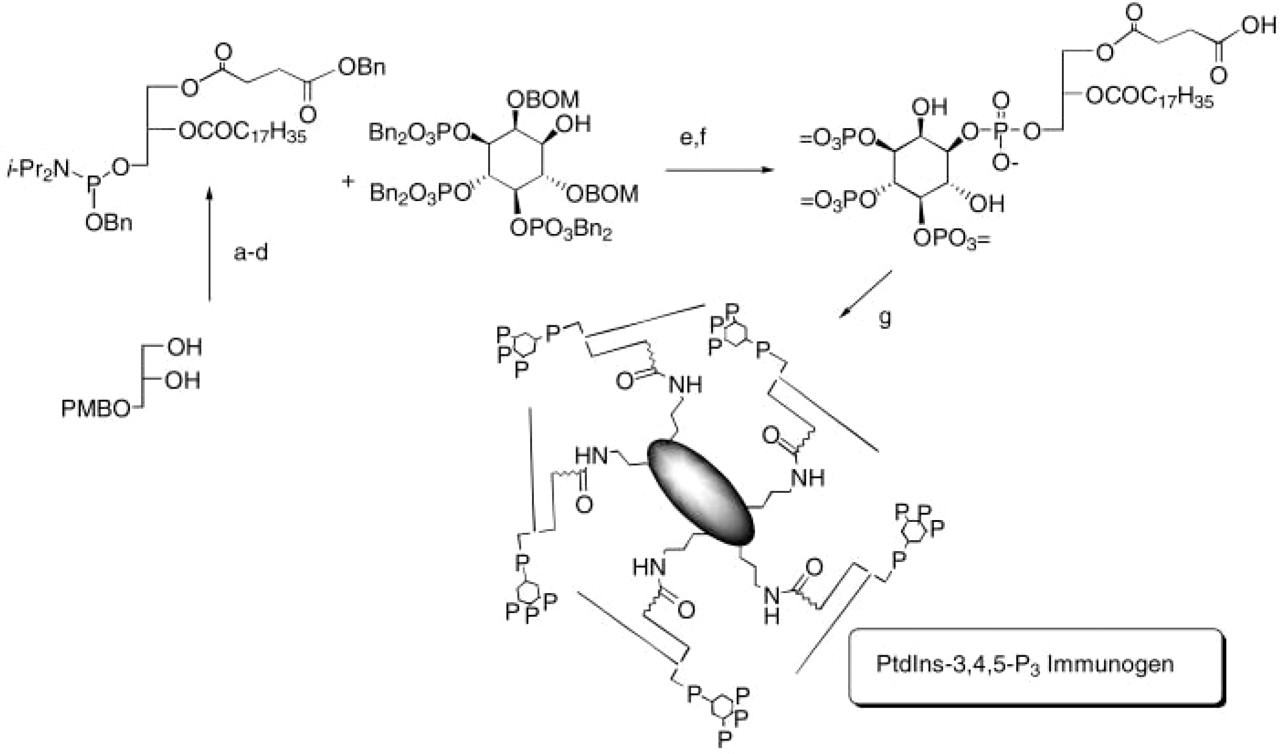
Synthesis of the PtdIns (3,4,5)P3 immunogen. Reagents: (
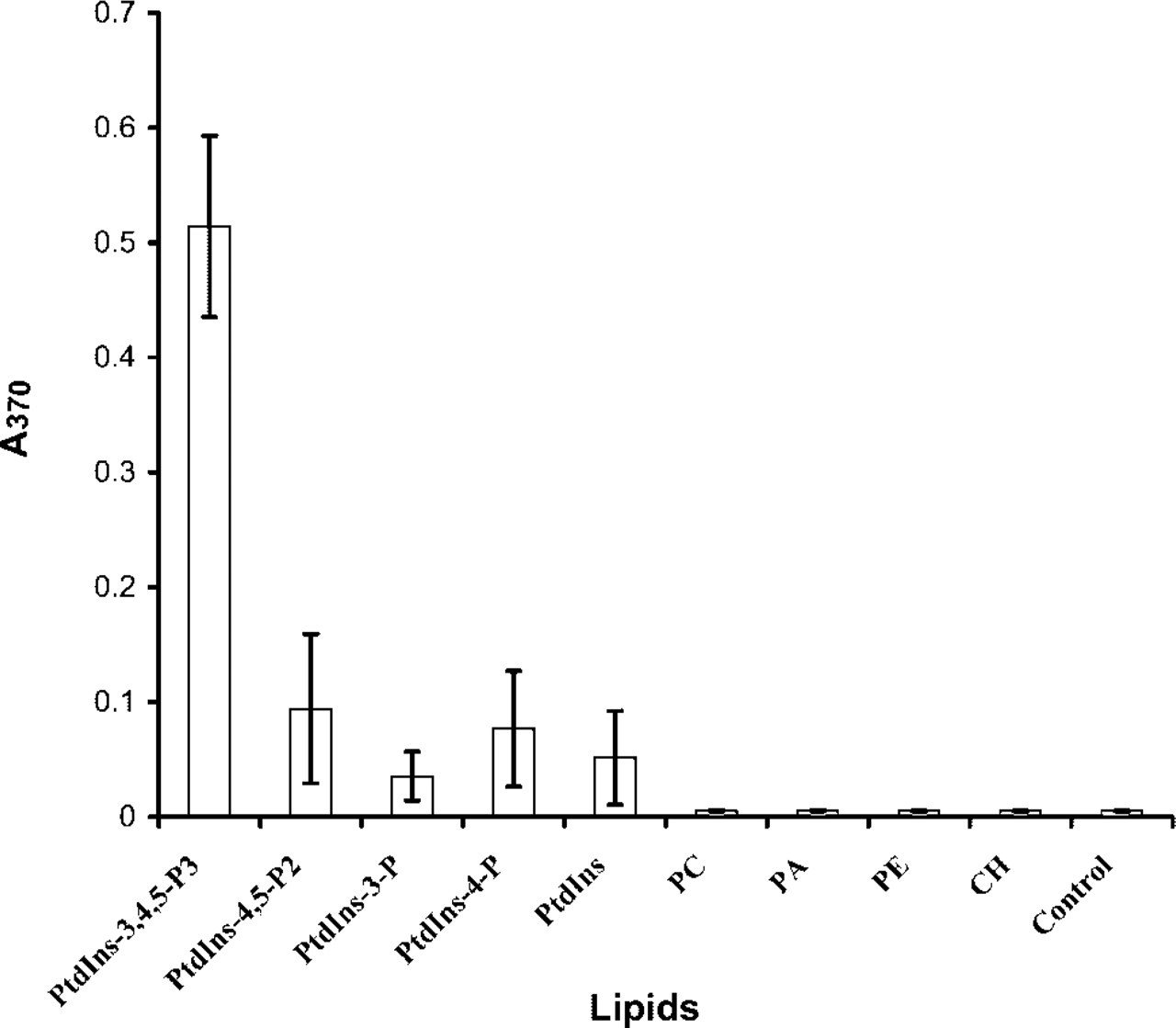
The RC6F8 MAb specifically detects PtdIns(3,4,5)P3. Specificity of the antibody: the wells of a polystyrene microtiter plate were coated with 1 μg of the different lipids from 95% ethanol stock solutions. Solvent was evaporated in air and ELISA detection performed. The results shown summarize data from three independent experiments, each in triplicate. PC, phosphatidylcholine; PA, phosphatidic acid; PE, phosphatidylethanolamine; CH, cholesterol.
Second, we employed ELISA to confirm the antibody affinity and selectivity for PtdIns(3,4,5)P3. Using a competitive ELISA, the RC6F8 IgM was incubated with soluble lipids until equilibrium was reached. For this experiment, we employed dioctanoyl (di-C8) analogues of each of the PtdInsPns. The proportion of antibody that remained unbound at equilibrium was then measured by an indirect ELISA using PtdIns(3,4,5)P3-KLH immobilized as the capture antigen in the wells. The results showed that di-C16-PtdIns (3,4,5)P3 had greater than 100-fold discrimination for di-C8-PtdIns(3,4,5)P3 relative to the other PtdInsPns tested (Figure 3). This result confirms the importance of all three phosphates correctly positioned on the inositol head group in the antibody-antigen recognition. The 3- and 4-monophosphates and the 3,5-, 3,4-, and 4,5-bisphosphates showed significantly reduced affinity for this antibody. Moreover, the soluble head group [Ins(1,3,4,5)P4] showed minimal displacement of the RC6F8-PtdIns(3,4,5)P3 interaction.
Third, we demonstrated ligand selectivity by photo-labeling the MAb with three [3H]-BZDC-triester-modified photoaffinity labels (Prestwich 1996; Chaudhary et al. 1998; Feng et al. 2001). Therefore, a control IgM and the anti-PtdIns(3,4,5)P3 IgM samples were incubated with equal dpm of [3H]-BZDC-triester-PtdIns(3,4,5)P3, [3H]-BZDC-triester-PtdIns(4,5)P2, and [3H]-BZDC-triester-PI(3)P, and a covalent linkage was established by UV irradiation. Probes had identical specific activities, thus allowing direct comparison of relative efficiencies of photoattachment. As shown in Figure 4, no labeling of the control IgM was seen with any of the probes. In contrast, the anti-PtdIns(3,4,5)P3 IgM was specifically photoaffinity-labeled by [3H]-BZDC-triester-PtdIns(3,4,5)P3. Surprisingly, labeling by [3H]-BZDC-triester-PtdIns(3,4,5)P3 occurred exclusively on the heavy chain rather than the light chain of the antibody (IgM), contrary to what might be expected for the location of the binding epitope. Similar results have been observed in the photolabeling of gelsolin with triester probes (Feng et al. 2001), because the probe explores a region of space orthogonal to the head group-diacylglyceryl axis. Apparently, the anti-PtdIns(3,4,5)P3 MAb light chain interaction positions the photoactivatable arm proximal in space to the heavy chain. Such a model is consistent with the wrapping of the heavy chain around the light chain in the three-dimensional antibody structure.
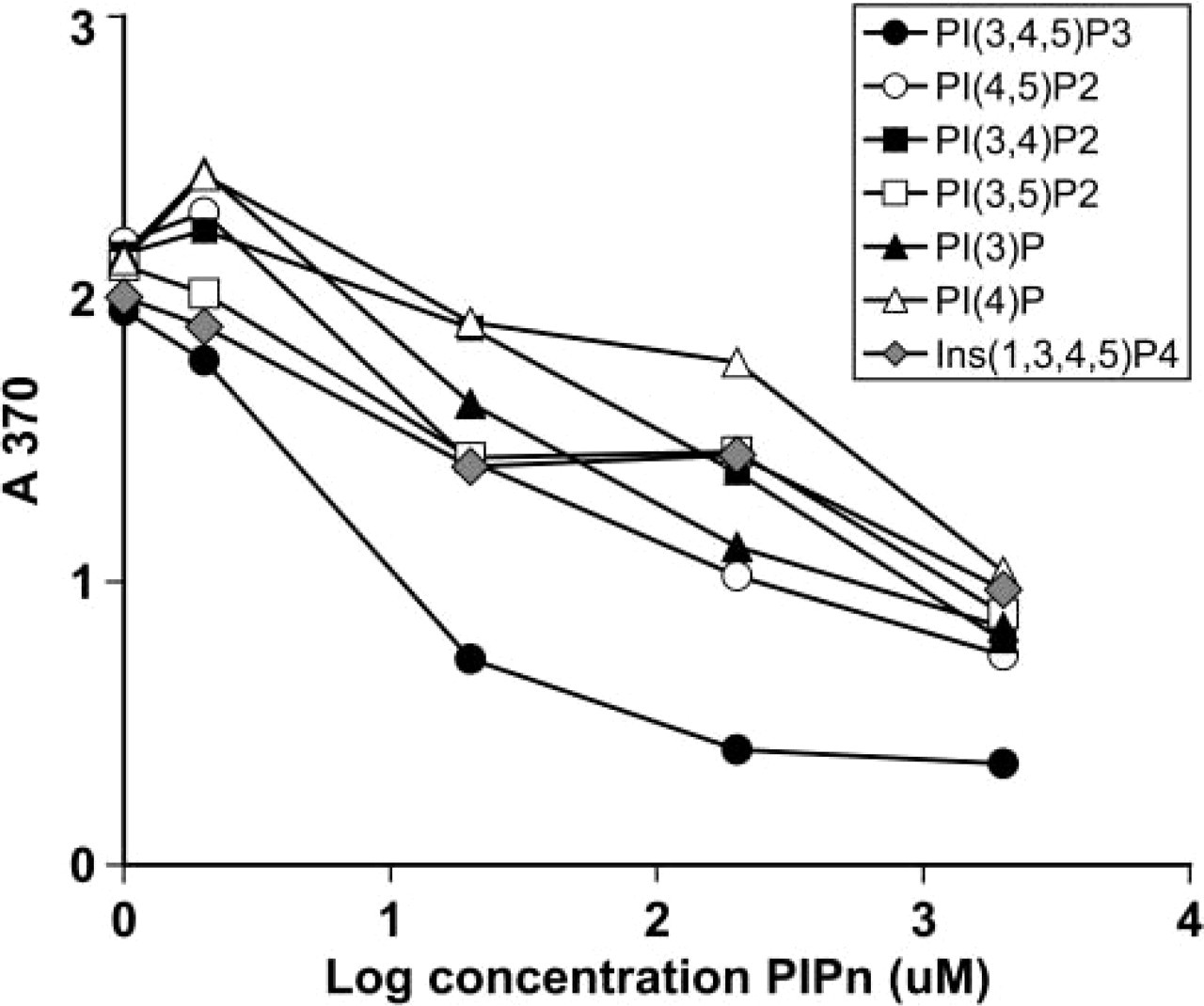
Competitive assays reveal that RC6F8 MAb binds specifically to PtdIns(3,4,5)P3. The purified IgM (100 μl, 0.14 μM) in TBS was added to di-C8 PtdInsPns at various concentration [2, 20, 200, 200 (μM)] and incubated at RT for 1 hr. An aliquot of each mixture (50 μl) was added to the 96-well plate coated with PtdIns(3,4,5)P3-KLH and ELISA detection was performed as described. The results shown are representative of three independent experiments.
Sensitivity of the Antibody
We examined the affinity of the RC6F8 MAb for PtdIns(3,4,5)P3. The ELISA can detect 6 pmol/liter PtdIns(3,4,5)P3 (data not shown). Because it has been estimated that the intracellular concentration of PtdIns(3,4,5)P3 can achieve values of up to 200 μM in neutrophils stimulated with formyl-methionyl-leucyl-phenylalanine (fMLP) (Traynor-Kaplan et al. 1989; Stephens et al. 1991), this provides ample sensitivity for detection of the PtdIns(3,4,5)P3 in cells. The confirmation of this sensitivity was then explored directly in several cell systems.
Quantification of PtdIns(3,4,5)P3 in NIH 3T3 Cells
Because growth factors are known to stimulate PI 3-kinase activity, we first measured PtdIns(3,4,5)P3 levels in [3H]-myo-inositol-loaded NIH 3T3 fibroblasts at several time points after PDGF stimulation. We observed that PtdIns(3,4,5)P3 levels increased rapidly. Figure 5 shows significant increases at 3 min, 15 min, and 60 min, with levels over tenfold above the stimulation base line. This finding is intriguing and served as an impetus to examine the localization and levels of PDGF-stimulated PtdIns(3,4,5)P3 accumulation in NIH 3T3 cells using the RC6F8 MAb.
Localization of PtdIns(3,4,5)P3 in NIH 3T3 Cells
To locate PtdIns(3,4,5)P3 in cells, several experiments were performed with NIH 3T3 fibroblasts. Cells were stimulated with either insulin or PDGF and both Ptd-Ins(3,4,5)P3 and PtdIns(4,5)P2 were detected by IF using specific MAbs, FITC-labeled goat anti-mouse IgM secondary antibody, and laser scanning confocal fluorescence microscopy (Figures 6 and 7). In both insulin- and PDGF-stimulated cells, an increase in Ptd-Ins(3,4,5)P3 was detected within 1 min of stimulation (not shown) and was sustained up to 120 min after stimulation. Preabsorption of the RC6F8 MAb with PtdIns(3,4,5)P3 before using the MAb to detect cellular PtdIns(3,4,5)P3 ablated detection, while pre-adsorption with PtdIns(4,5)P2 did not reduce PtdIns (3,4,5)P3 detection. When cellular PI 3-kinase activity was inhibited with LY294002 before growth factor stimulation, no increase in PtdIns(3,4,5)P3 was observed (data not shown).
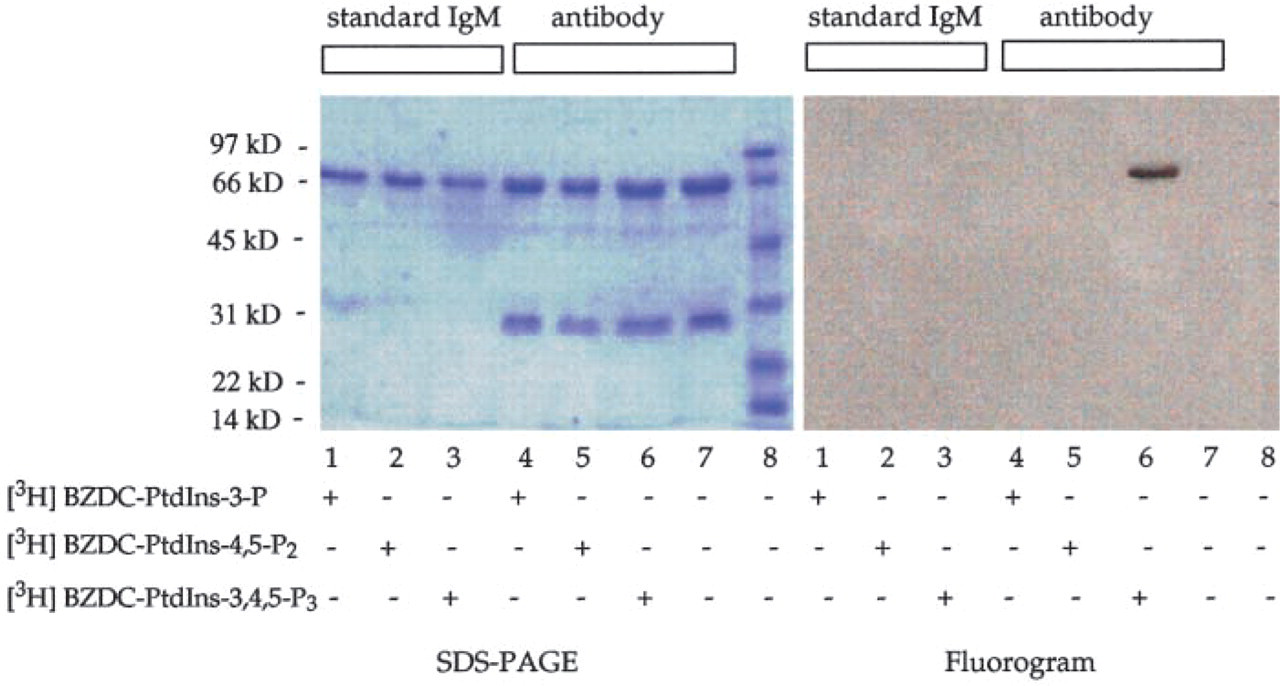
Photoaffinity labeling of the RC6F8 MAb with [3H]-BZDC-triester-PtdInsPn confirms specificity for PtdIns(3,4,5)P3. Either the RC6F8 MAb or standard IgM (0.68 μM) was incubated with 20 μl of TBS containing 0.1 μCi (0.28 μM) of [3H]-BZDC-triester-PtdIns(3)P, [3H]-BZDC-triester-PtdIns (4,5)P2, or [3H]-BZDC-triester-PtdIns (3,4,5)P3. Samples were equilibrated at 4C for 15 min in a 96-well plate and were irradiated (360 nm at 1900 μW/cm2) for 45 min at 4C. The proteins were separated by SDS-PAGE. The gel was stained, destained, and impregnated with En3Hance. The gel was dried and exposed to X-ray film for 8 days at −80C and developed.
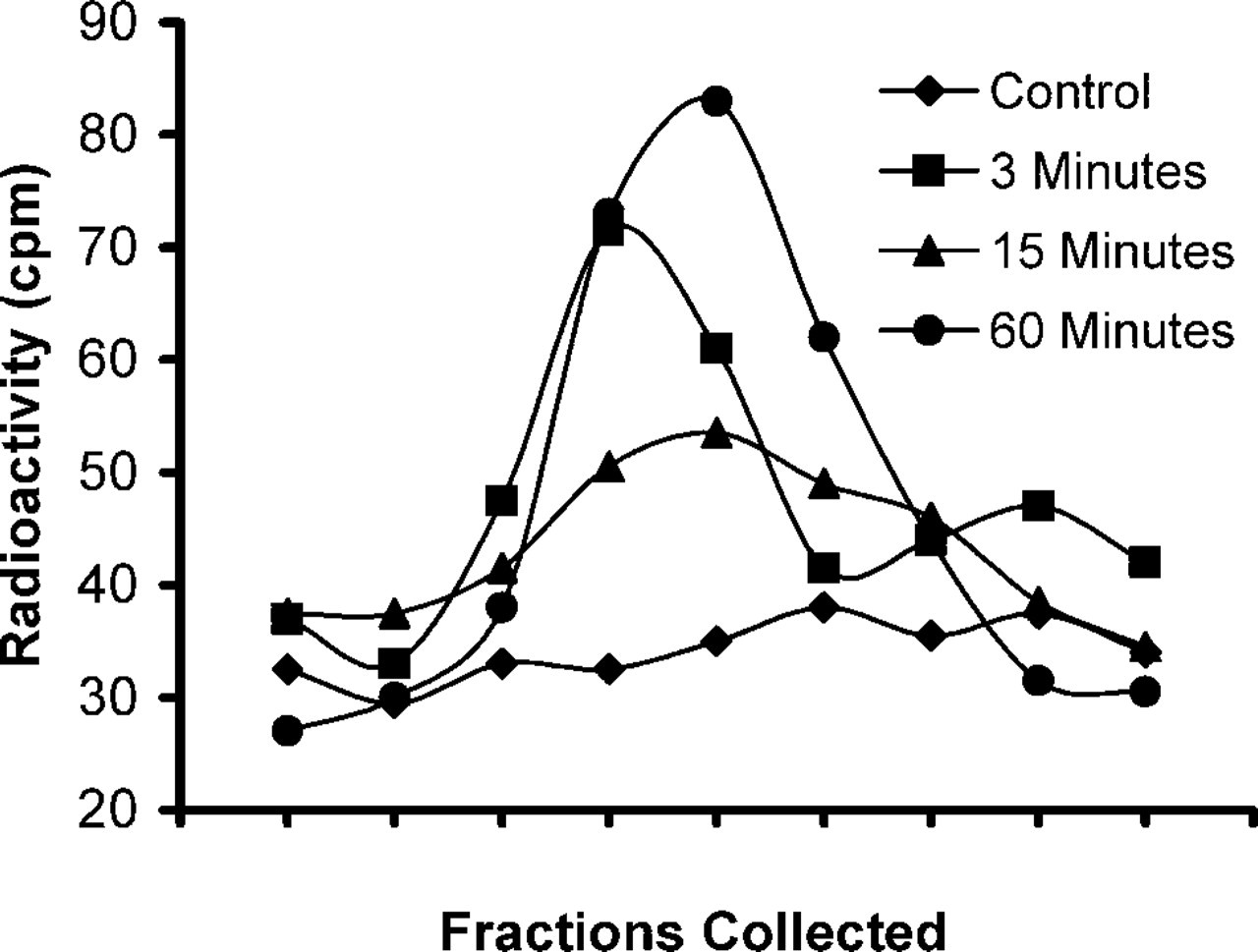
PtdIns(3,4,5)P3 accumulation is sustained in NIH 3T3 fibroblasts. NIH 3T3 fibroblasts were metabolically labeled with [3H]-myo-inositol and serum-starved for 2 hr before treatment with 50 ng/ml PDGF for the times indicated. Cellular phospholipids were extracted, deacylated, and glycerophosphoinositol head groups were analyzed using an HPLC-based assay. The peak in the chromatogram corresponds to glycerophosphoinositol 3,4,5-trisphosphate from unstimulated control cells (diamonds) and after treatment with PDGF for t = 3 min (squares), t = 15 min (triangles), and t = 60 min (circles).
Serum-starved fibroblasts were stimulated with insulin for 0, 5, or 30 min and PtdIns(3,4,5)P3 and PtdIns(4,5)P2 distributions were visualized by IF (Figure 6). Consistent with previous studies (Jones et al. 1999), PtdIns(3,4,5)P3 accumulated rapidly. However, the level was sustained for over 30 min in insulin-stimulated cells (Figure 6C) and over 60 min in the PDGF-stimulated cells (Figure 7C). This was significantly different than reported previously for HepG2 cells (Jones et al. 1999). For both insulin- and PDGF-stimulated cells, PtdIns(3,4,5)P3 localized mainly in the cytoplasm, and a dramatic change in localization on stimulation was not observed. Much less PtdIns(3,4,5)P3 was observed in the nucleus (Figures 6B, 6C, 7B and 7C), but some “nuclear speckles” were visible.
To compare PtdIns(3,4,5)P3 production and localization with another PtdInsPn molecule acting as a second messenger in NIH 3T3 cells, PtdIns(4,5)P2 was detected by IF using the anti-PtdIns(4,5)P2 MAb 10F8 (Thomas et al. 1999) (Figures 6D–6F and 7D-7F). In contrast to the localization of PtdIns(3,4,5)P3, Ptd-Ins(4,5)P2 was detected predominantly in the nucleus of the cells stimulated with either PDGF or insulin (Figures 6D–6F and 7D-7F). Furthermore, we observed significant differences between the localization of PtdIns(4,5)P2 in stimulated and unstimulated cells (compare Figure 6D with 6E and Figure 7D with 7E). The maximal PtdIns(4,5)P2 accumulation in insulin-stimulated (Figure 6E) and PDGF-stimulated (Figure 7E) cells occurred at 15 min, but nuclear staining was observed up to 2 hr after growth factor stimulation (data not shown).
To confirm the specificity of the 10F8 MAb for cellular lipids, we tested whether the antibody reacted with any proteins present in NIH 3T3 cell extracts. For this determination, a lysate of NIH 3T3 cells (1 × 106 cells) was subjected to SDS-PAGE, and then the proteins were electrotransferred to the PVDF membrane and subjected to Western blotting analysis with the antibody. Results showed that the antibody did not react with any proteins present in NIH 3T3 cell lysate (data not shown).
Stimulated Production of PtdIns(3,4,5)P3 and PtdIns(4,5)P2 by Human Neutrophils
It had been shown by metabolic labeling and lipid extraction that PtdIns(3,4,5)P3 levels increase rapidly in neutrophils on stimulation with fMLP (Traynor-Kaplan et al. 1989). To determine the distribution of PtdIns(3,4,5)P3 in intact neutrophils, we stimulated the cells with fMLP for various lengths of time and performed IF with the anti-PtdIns(3,4,5)P3 RC6F8 MAb (Figures 8A–8C) and the anti-PtdIns(4,5)P2 10F8 MAb (Figures 8D–8F). There was a dramatic and transient increase in PtdIns(3,4,5)P3 levels with fMLP stimulation, with a peak production at 5–10 sec (Figures 8A–8C). PtdIns(3,4,5)P3 was clearly excluded from the nucleus both before and after stimulation, in contrast to what was observed in the NIH 3T3 cells. As a comparison, distribution and levels of PtdIns(4,5)P2 were examined at the same time using an MAb shown to be specific for PtdIns(4,5)P2 (Thomas et al. 1999). Ptd-Ins(4,5)P2 levels were at maximum with no fMLP stimulation (t = 0; Figure 8D) and gradually decreased after fMLP for the duration of the experiment (t = 60 sec; Figure 8F). The changes in PtdIns(3,4,5)P3 and PtdIns(4,5)P2 levels correspond closely in time to those reported previously (Sorisky et al. 1996).
Discussion
The distribution of PtdInsPns, their relocalization and association with target proteins upon cell activation, and their metabolism in cells is still not clearly delineated. Answering these questions unequivocally is necessary to obtain a coherent view of phosphoinositide signaling pathways. Here we utilized an anti-PtdIns(3,4,5)P3 MAb as a detection tool and showed approximate cellular level changes and demonstrated distribution of PtdIns(3,4,5)P3 in intact cells. Previously, PtdInsPn level changes have been alluded to, indirectly, as PI 3-kinase activity, or measured by laborious metabolic labeling followed by lipid extraction and HPLC. The PtdIns(3,4,5)P3 MAb enabled us to easily measure and visualize changes in PtdIns (3,4,5)P3 after cell stimulation in vivo. The specificity of the antibody can be further exploited to quantitate PI 3-kinase activity and PtdIns(3,4,5)P3 levels in cells, and can be used to specifically disturb the cellular activity by introduction into living cells.
The results described here illustrate two major aspects of the utility of this anti-PtdIns(3,4,5)P3 antibody. First, it is clear that the plasma membrane is not the only site at which PtdIns(3,4,5)P3 is produced in fibroblasts and neutrophils. The RC6F8 antibody identifies cytoplasmic pools of PtdIns(3,4,5)P3 that are associated with membrane ruffling (Rickert et al. 2000), coatomer-coated vesicles important in Golgi trafficking (Chaudhary et al. 1998), and other cell transport processes. Second, we demonstrate that the PtdIns(3,4,5)P3 produced in stimulated cells persists for at least an hour. PtdIns(4,5)P2 has been identified at sites other than the plasma membrane, e.g., the nucleus (Thomas et al. 1999; Osborne et al. 2001), but it has generally been accepted that PtdIns(3,4,5)P3 is produced at the plasma membrane and then recycled by movement to the lysosome via endocytosis (Joly et al. 1994,1995; Corvera et al. 1999; Corvera 2001). However, current techniques using GFP-PH domain fusion proteins do not confirm this view empirically, even though this is most consistent with a model depicting PtdIns(3,4,5)P3 as a second messenger. To fulfill a role as a signaling molecule, PtdIns(3,4,5)P3 must be synthesized rapidly to initiate a signaling cascade, and then metabolized or relocalized to downregulate the signaling pathway. If downregulation of PI 3-kinase signaling occurs via intracellular relocalization of PtdIns(3,4,5)P3, then PtdIns(3,4,5)P3 should be detected in vesicles found in the cytoplasm. Cytoplasmic staining was observed, but additional work is necessary to reveal whether immunodetected PtdIns(3,4,5)P3 was associated with vesicles.
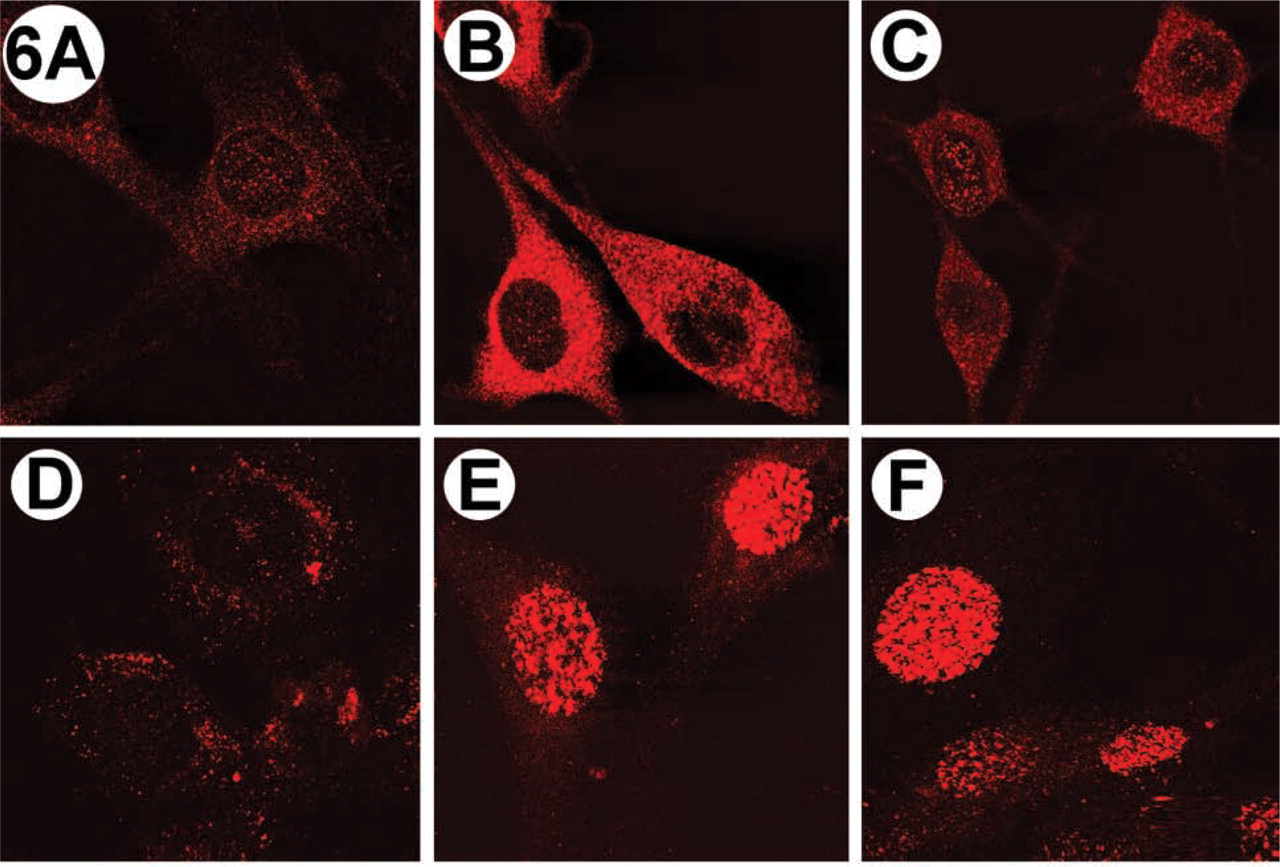
Immunolocalization of PtdIns(3,4,5)P3 (top) and PtdIns(4,5)P2 (bottom) in insulin-stimulated NIH 3T3 fibroblasts. Cells were stimulated with 100 ng/ml insulin and phosphoinositides were visualized by IF as described in the text. (
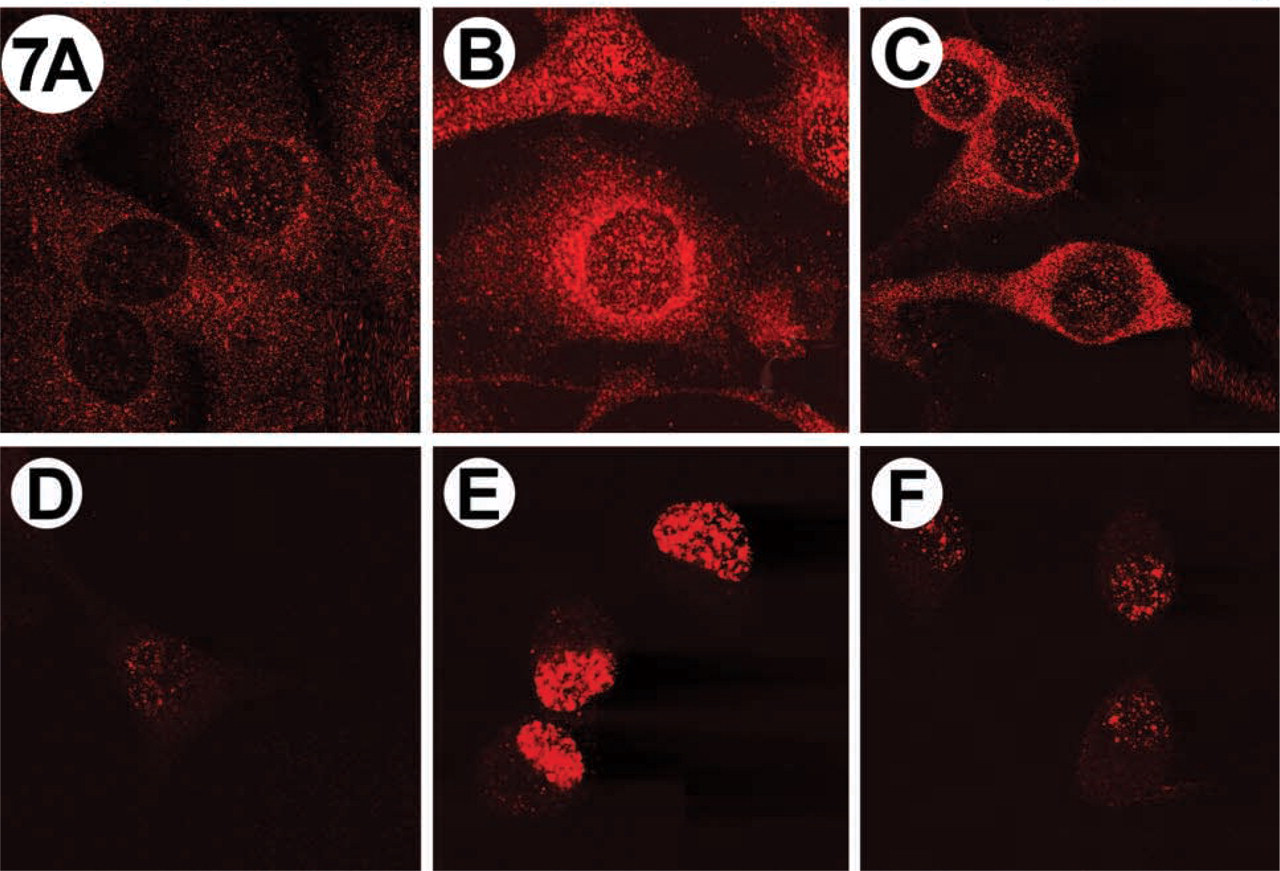
Immunolocalization of PtdIns(3,4,5)P3 (top) and PtdIns(4,5)P2 (bottom) in PDGF-stimulated NIH 3T3 fibroblasts. Cells were stimulated with 50 ng/ml PDGF and phosphoinositides were visualized by IF, as described in the text. (
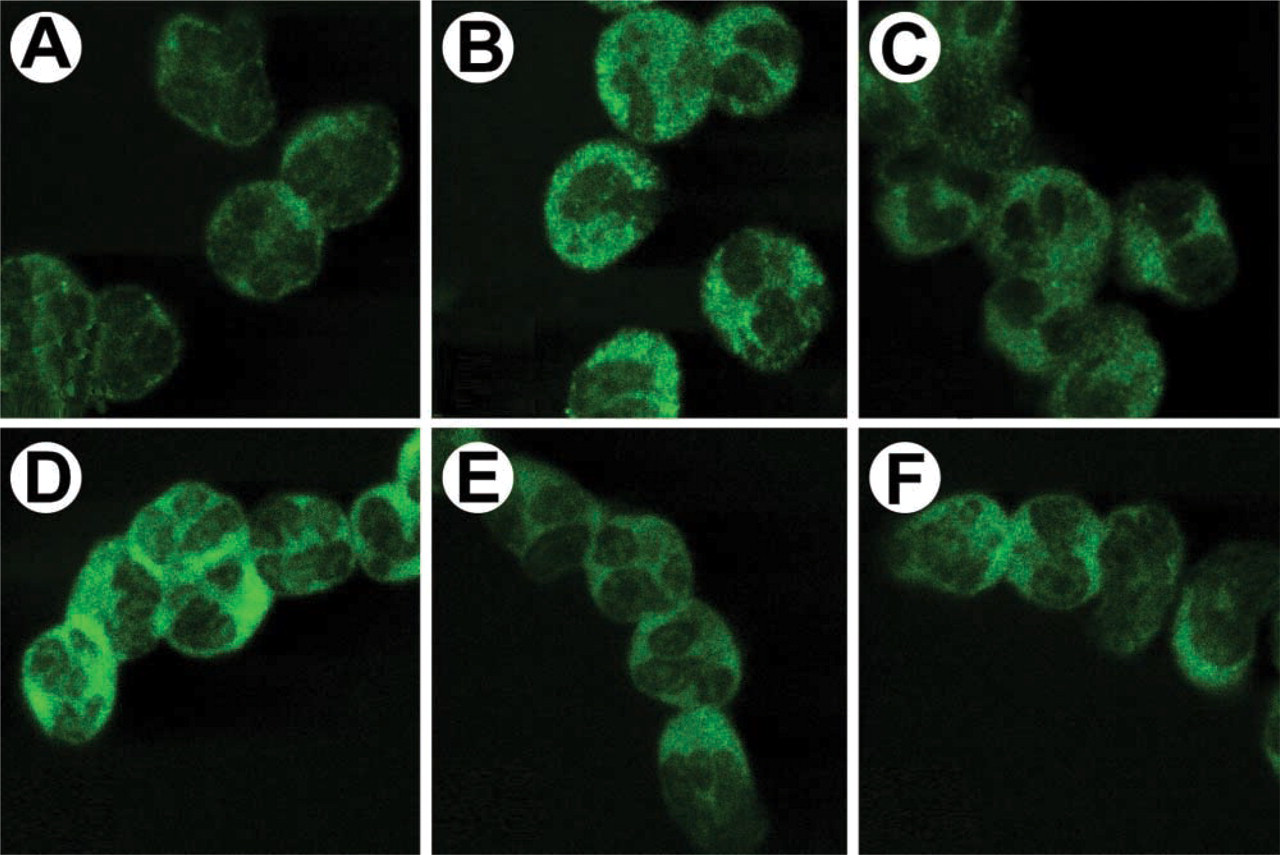
Immunolocalization of PtdIns(3,4,5)P3 and PtdIns(4,5)P2 in human neutrophils stimulated with fMLP. Neutrophils were stimulated with 100 nM fMLP for the times indicated. Reactions were stopped and cells were fixed with paraformaldehyde. (
PtdIns(3,4,5)P3 was also detected in the nucleus of stimulated cells. Interestingly, PtdIns(3,4,5)P3-binding proteins have been identified in the nuclei of the neuronal cells (Tanaka et al. 1999). In addition, the isoforms of PIPKI (Type I phosphatidylinositol phosphate kinase) and PIPKII (Type II phosphatidylinositol phosphate kinase), PtdIns(4,5)P2, and the enzymes required for mRNA processing and transcription are associated with nuclear speckles (Anderson et al. 1999). Our data support the idea that PtdInsPn synthesis and metabolism may be associated with specific protein domains in the nuclear matrix and at sites in the cytoplasm.
The difference in duration of PtdIns(3,4,5)P3 levels and PI 3-kinase activity on stimulation poses interesting speculations. Previous studies showed that insulin stimulation resulted in a slow rise and sustained PI 3-kinase activity in 3T3-L1 cells, whereas PDGF resulted in rapid and transient PI 3-kinase activity (Ricort et al. 1997). Visualization of PtdIns(3,4,5)P3 at the leading edge of motile cells can be accomplished with either a GFP-PH domain (Rickert et al. 2000) or with the RC6F8 IgM described herein (H. Bourne, personal communication). Here we showed that PtdInsPn levels are sustained for long periods of time with constant exposure to agonists. Indeed, in Hep2 cells stimulated with PDGF there is a second and sustained wave of Ptd-Ins(3,4,5)P3 products for several hours after stimulation. This second wave was essential for progression of HepG2 cells through the cell cycle (Jones et al. 1999). The sustained production of phosphoinositides with constant exposure to either insulin or PDGF suggested that their half-lives may be longer than previously expected. To determine the half-life of PtdIns (3,4,5)P3, we performed pulse-chase experiments. NIH 3T3 cells were stimulated with insulin for 5 min, and then PtdIns(3,4,5)P3 and PtdIns(4,5)P2 were visualized at various time points by IF. The IF results corroborated the biochemical analyses; PtdIns(3,4,5)P3 production and accumulation occur over several minutes in PDGF-stimulated fibroblasts (Sorisky et al. 1996).
The change in PtdIns(3,4,5)P3 and PtdIns(4,5)P2 levels in neutrophils suggests the immediate precursor of PtdIns(3,4,5)P3 to be PtdIns(4,5)P2. However, in NIH 3T3 cells the primary precursor of PtdIns(3,4,5)P3 may not be PtdIns(4,5)P2, because the accumulation of PtdIns(3,4,5)P3 does not correspond directly to a decrease in PtdIns(4,5)P2. It has been suggested that, in NIH 3T3 cells, PtdIns(3,4,5)P3 formation can occur via the PtdIns(3)P to PtdIns(3,4)P2 to PtdIns(3,4,5)P3 pathway (Cunningham and Majerus 1991).
In summary, the generation of a PtdIns(3,4,5)P3-specific MAb has enabled us to address fundamental issues in phosphoinositide signaling. This tool should now facilitate discovery of potential drug candidates and development of diagnostic tools for cancer and other pathologies (Razzini et al. 2000; Blume-Jensen and Hunter 2001). This MAb will also allow observation of the dynamics of intracellular changes of PtdIns(3,4,5)P3 in space and time.
Footnotes
Acknowledgements
NIH (Grant NS29632), the American Cancer Society (Grant RPG-00-126-01-TBE), and the Utah Centers of Excellence Program provided financial support for the antigen preparation and antibody development.
We thank Echelon Research Laboratories for providing dipalmitoyl and dioctanoyl PIPn derivatives and Dr D.W. Neklason for assistance with manuscript preparation. Drs L. Feng, and J. Peng provided radioligands and assistance with IgM photoaffinity labeling. Special thanks to A. Weyrich (U Utah) for assistance with neutrophil preparations. Mr C. Jones and Ms K. Manabe (Utah State) assisted with HPLC and microscopy. Anti-PtdIns(4,5)P2 IgMs 2C11 and 10F8 were initially obtained from Dr G. Schiavo (Imperial Cancer Research Fund, London). In more recent studies, the 2C11 and RC6F8 antibodies were obtained from Echelon.
