Abstract
Phosphatidylinositol 3-phosphate [Ptdlns(3)P] playsan important role in recruitment of various effector proteins in the endocytic and autophagic pathways. In an attempt to follow the distribution of Ptdlns(3)P at the ultrastructural level, we are using the Fab1, YOTB, Vac1, and EEA1 (FYVE) domain, which is a zinc finger motif specifically binding to Ptdlns(3)P. To follow Ptdlns(3)P trafficking during a defined time window, here we have used a mono-meric dimerizable FYVE probe, which binds with high avidity to Ptdlns(3)P only after rapalog-induced dimerization. The probe localized to early and late endocytic compartments according to the time period of dimerization, which indicates that Ptdlns(3)P is turned over via the endocytic machinery. In the functional context of epidermal growth factor (EGF) stimulation, we observed that dimerization of the probe led to clustering of mainly early endocytic structures, leaving most of the probe localized to the limiting membrane of endosomes. Interestingly, these clustered endosomes contained coats positive for the Ptdlns(3)P-binding protein hepatocyte growth factor-regulated tyrosine kinase substrate (Hrs), indicating that the probe did not displace Hrs binding. We conclude that the dimerizer-inducible probe is useful for the time-resolved detection of Ptdlns(3)P at the ultrastructural level, but its effects on endosome morphology after EGF stimulation need to be taken into account.
Keywords
Phosphoinositides are derivatives of phosphatidylinositol that can undergo rapid cycles of phosphorylation and dephosphorylation at the 3-, 4-, and 5-positions of their inositol head groups. These modification processes are regulated by kinases and phosphatases, respectively, and mediate highly localized changes in the levels of phosphoinositides. In this way, the temporal and spatial regulation of effector proteins binding these lipids is carefully controlled. Phosphoinositides represent only minor constituents of the membrane lipid bilayer. However, through specific effector recruitment, they play crucial regulatory roles in guiding membrane traffic and in cell signaling, for instance, as regulators of nuclear functions, cytoskeletal dynamics, and signal transduction.
The phosphoinositide phosphatidylinositol 3-phosphate [PtdIns(3)P] is restricted to the endocytic pathway and is most abundant on early endosomal membranes and on the internal membranes of multivesicular endosomes (MVEs) (Gillooly et al. 2000). Studies on yeast have shown that PtdIns(3)P is transported to the vacuole (the yeast equivalent of the lysosome) for turnover (Wurmser and Emr 1998). The effector proteins of PtdIns(3)P contain either a FYVE domain (Burd and Emr 1998; Gaullier et al. 1998; Patki et al. 1998) or a Phox (PX) homology domain (Simonsen and Stenmark 2001). The name FYVE is derived from the first letters of the first four proteins containing this domain (Fab1, YOTB, Vac1, and EEA1) (Stenmark et al. 1996). The FYVE domain contains a double zinc finger-binding motif, which consists of ~70 amino acids and binds specifically to PtdIns(3)P (Stenmark et al. 1996; Burd and Emr 1998; Patki et al. 1998). The binding properties to PtdIns(3)P are further regulated by a histidine switch, which enhances binding of the FYVE domain at low cytosolic pH values. In the case of EEA1, only half of the protein is active and bound to PtdIns(3)P at normal cytosolic pH (Lee et al. 2005).
Several FYVE-containing proteins, such as EEA1, rabenosin-5, and hepatocyte growth factor-regulated tyrosine kinase substrate (hrs), are involved in endosomal membrane traffic, whereas others, such as PIKfyve, Fabl, and the MTMR3 and 4 phosphatases, are catalytically active, and a third group comprises proteins, such as hrs and SARA, involved in signaling [extensively reviewed in Kutateladze (2007)].
Binding of these domains to PtdIns(3)P is very specific, and although the exact functions of each of the FYVE- and PX-domain-containing proteins remain to be elucidated, many of these proteins seem to play a role in membrane trafficking, regulation of cytoskeletal function, and signal transduction (Simonsen and Stenmark 2001; Stenmark et al. 2002).
Although FYVE domains bind PtdIns(3)P efficiently in vitro, isolated FYVE domains often fail to localize to endosomes when expressed in cells (Lawe et al. 2000; Raiborg et al. 2001b; Hayakawa et al. 2004). These observations suggest that the affinity of these domains for PtdIns(3)P (K D values usually in the low micromolar range) is too low for efficient membrane recruitment, and additional structural features, such as homodimerization or the presence of additional membrane-targeting domains, may also play a role (Misra and Hurley 1999; Hayakawa et al. 2004). The avidity of FYVE domains for PtdIns(3)P differs greatly for each protein, and this was recently shown to depend on structural differences among FYVE domains (Hayakawa et al. 2004). It has been suggested that these structural differences provide an evolutionary mechanism for controlling the extent and duration of the interaction between specific FYVE domain-containing proteins and early endosomes (Hayakawa et al. 2004).
The ultrastructural localization of phosphoinositides is difficult to study (Rusten and Stenmark 2006). However, it is interesting to clarify the functional localization of PtdIns(3)P in the endocytic pathway in the context of distinctive mechanisms for intraluminal vesicle formation and subpopulations of MVEs, as it has previously been reported that PtdIns(3)P and the lipase-resistant phospholipid lyso-bis-phosphatidic acid (LBPA) localize to distinct endosomal compartments (Gillooly et al. 2000). Previously, Hayakawa et al. (2004) used an approach to homodimerize the monomeric FYVE domain of Hrs by recombinant fusion to the rapamycin-binding protein FKBP (F36V, hereafter called Fv). The green fluorescent protein (GFP)-tagged Fv-FYVE(Hrs) construct homodimerizes in the presence of the small cell-permeable bivalent rapamycin derivative, AP20187, thereby providing the avidity for PtdIns(3)P required to target PtdIns(3)P-containing membranes. We were interested in using this approach to temporally regulate the binding of FYVE domains to PtdIns(3)P and thereby study the spatiotemporal localization of PtdIns(3)P in the endocytic pathway.
We found that the GFP-Fv-FYVE(Hrs) probe localizes to early and late endocytic compartments according to the time period of probe dimerization, which indicates that PtdIns(3)P is turned over via the endocytic machinery. Surprisingly, in epidermal growth factor (EGF)-stimulated cells, we observed that dimerization of the GFP-Fv-FYVE(Hrs) probe led to clustering of early endocytic structures, leaving the probe mainly localized to the limiting membrane of endosomes. These results demonstrate the usefulness of the GFP-Fv-FYVE(Hrs) probe in time-resolved ultrastructural mapping of PtdIns(3)P, but also caution that PtdIns(3)P sequestration may cause unexpected perturbations of endosomal functions.
Materials and Methods
Cell Culture, Plasmid Constructs, and Transfections
HeLa cells were grown and maintained as recommended by the American Type Culture Collection (Manassas, VA). Cells were cultured one day before transfection and were transfected with optimal concentrations of either a GFP2xFYVE construct (Gillooly et al. 2003) or the monomeric GFP-Fv-FYVE(Hrs) construct (Hayakawa et al. 2004) using FuGENE 6 (Roche; Basel, Switzerland) according to the manufacturer's instructions. More specifically, we used 0.2 μg DNA/0.6 μl FuGENE when transfecting cells on cover slips in 24-well plates and 1 μg DNA/6 μl FuGENE for transfection in 6-cm dishes.
Antibodies and Reagents
Human anti-early endosomal antigen (EEA1) antiserum was a gift from BanHock Toh (Monash University; Melbourne, Australia). Mouse anti-human lysosomal-associated membrane protein (LAMP2; clone H4B4) and mouse anti-human CD63 were obtained from the Developmental Studies Hybridoma Bank of the University of Iowa (Iowa City, IA). Mouse monoclonal antibodies against LBPA were kindly provided by Jean Gruenberg (University of Geneva; Geneva, Switzerland). Sheep antibodies against epidermal growth factor receptor (EGFR) were obtained from Fitzgerald (Concord, MA). Antiserum against Hrs has been described previously (Raiborg et al. 2001a). Antiserum against GFP was kindly provided by Terje Johansen (Tromsø University; Tromsø, Norway). Cy3 and Cy5 antibodies were obtained from Jackson ImmunoResearch (West Grove, PA). ProteinA-gold (PAG) was obtained from Cell Microscopy Center (Utrecht, The Netherlands). Secondary antibodies against mouse IgG and sheep IgG were obtained from Dako (Carpinteria, CA). The ARGENT regulated homodimerization kit was obtained from ARIAD Pharmaceuticals (Cambridge, MA), and the monomeric GFP-Fv-FYVE(Hrs) construct was kindly provided by Silvia Corvera (University of Massachusetts Medical School; Worcester, MA).
Confocal Immunofluorescence Microscopy
HeLa cells were grown on cover slips and transfected with the GFP-tagged constructs. Transfections were performed using FuGENE transfection agent according to the manufacturer's recommendation. Twenty-four hr after transfection, cells transfected with the monomeric GFP-Fv-FYVE(Hrs) construct were either incubated or not with the homodimerizer AP20187 for 30 min, unless mentioned otherwise, and stimulated with 50 ng/ml EGF. Cells were fixed in 3% formaldehyde, perme-abilized in 0.05% saponin, and stained with antibodies as described previously (Simonsen et al. 1998a). Cover slips were observed using a Zeiss LSM 510 META confocal microscope (Jena, Germany), and image processing was done with Adobe Photoshop CS2. All confocal pictures used for quantification were scanned using same pinhole, offset gain, and amplifier values below pixel saturation. Colocalization of GFP-Fv-FYVE and EEA1, CD63, or LBPA was determined using the colocalization tool of the Zeiss LSM 510 software (version 3.2).
Electron Microscopy (EM)
Cells for immuno-EM were fixed in 4% formaldehyde, 0.2% glutaraldehyde in 0.1 M phosphate buffer at room temperature for 40 min, washed, scraped, and pelleted in 12% gelatine at 10,000 rpm. Specimens were infiltrated with 2.3 M sucrose, mounted on silver pins, and frozen in liquid nitrogen. Ultrathin cryosections were cut at -HOC (Leica EM FCS ultramicrotome; Wetzlar, Germany) and collected with a 1:1 mixture of 2% methyl cellulose and 2.3 M sucrose. Sections were transferred to Formvar-carbon-coated grids and labeled with primary antibodies followed by labeling with a bridging secondary antibody, if necessary, and PAG conjugates essentially as described (van Genderen et al. 1991). Sections were observed at 60-80 kV in a JEOL JEM-1230 electron microscope (Tokyo, Japan). Micrographs were recorded with a Morada digital camera using iTEM (SIS) software (both from Soft Imaging Solutions; Muenster, Germany).
Results
Spatiotemporal Tracking of Ptdlns(3)P in the Endocytic Pathway
PtdIns(3)P is known to specifically localize to early endosomal membranes and the intraluminal membranes of MVEs (Gillooly et al. 2000). We were interested, however, to functionally track PtdIns(3)P in combination with an endocytic cargo and a subset of endosomal markers. We transfected cells with a GFP-Fv-FYVE (Hrs) probe that dimerizes when incubated with the specific cell-permeant homodimerizer AP20187 (Hayakawa et al. 2004), thereby increasing the avidity for PtdIns(3)P required to target early endosomes. Formation of the dimerized probe, herein called GFP-2xFv-FYVE(Hrs), was induced for different durations of time, 0 (no homodimerization), 20,60, and 180 min, and the localization of GFP-2xFv-FYVE(Hrs) was studied by immunofluorescence (IF) confocal microscopy. HeLa cells transfected with the monomeric GFP-Fv-FYVE(Hrs) probe showed weak cytosolic staining (Figure 1A) and a varying amount of nuclear staining. Homodimerization led to a punctate labeling pattern, showing the redistribution of GFP-2xFv-FYVE(Hrs) to the endosomal membranes (Figures 1B-1D). We found that after homodimerization for 20 and 60 min, a large amount of GFP-2xFv-FYVE(Hrs) colocalized with EEA1 on the membranes of early endosomes (Figures 1B and 1C). Colocalization with the late endosomal marker CD63, on the other hand, was very apparent after 180 min (Figure 1D). Quantification of the early and late endosomal marker distribution confirmed that GFP-2xFv-FYVE(Hrs) seemed to associate very early with EEA1-positive compartments, whereas colocalization with CD63 increases gradually from 20 to 180 min. These results show that the FYVE-homodimerizing approach can be used for the temporal ultrastructural detection of PtdIns(3)P.
Ultrastructural Analysis of Cells Transfected With the Dimerizing GFP-2xFv-FYVE(Hrs)
To determine the effects of the GFP-Fv-FYVE probe under different conditions on the ultrastructural morphology of the cell, we prepared transfected cells dimerized or not and stimulated with EGF or not, for ICC. Cells were labeled with an antiserum to GFP alone or in combination with an antibody to CD63 (Figure 2). GFP labeling was restricted to the cytosol in transfected cells not treated with dimerizer (Figure 2A), and homodimerization of the probe for 20 min resulted in the relocation of GFP labeling to the membranes of structures with a clearly early endosomal appearance, both on the cisternal membrane and on the membranes of adjacent small vesicles or tubules (Figure 2B). Likewise, after homodimerization for 180 min, GFP labeling was found on early endosomal membranes but also more often on the intraluminal membranes of MVEs, colocalizing with CD63 (Figure 2C). Labeling on these structures was rather weak, however. Additionally, we found that EGF stimulation led to the clustering of structures with either an early endosomal appearance or very small MVE-like structures (Figure 2D).
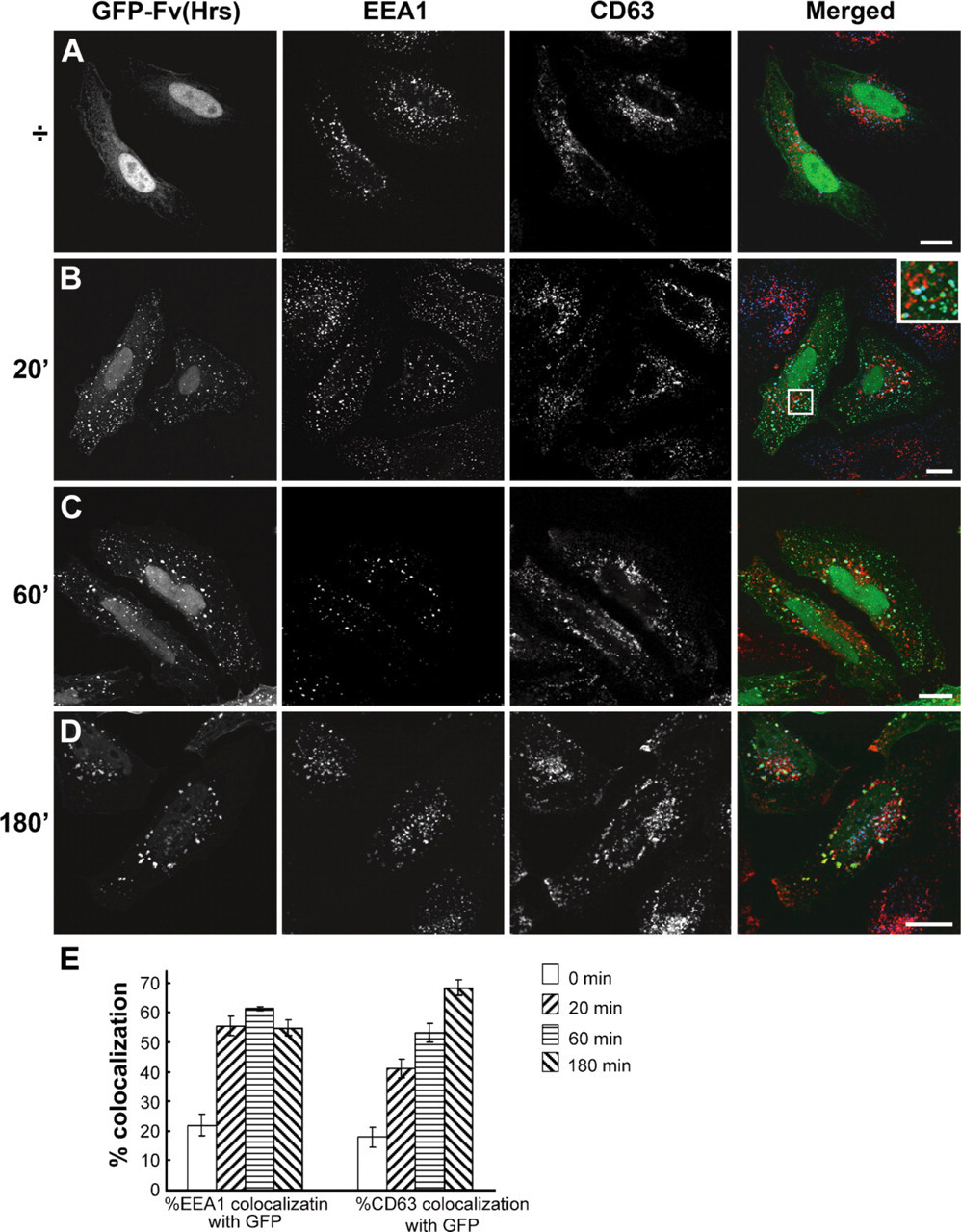
Using a dimerizing FYVE probe to spatially and temporally follow phosphatidylinositol 3-phosphate [Ptdlns(3)P] localization. HeLa cells were transfected with the dimerizing GFP-Fv-FYVE(Hrs) probe, incubated with the crosslinker for 0 min (
Characterization of EGF-induced Clusters of Ptdlns(3)P-containing Endocytic Structures in GFP-2xFv-FYVE(Hrs)-expressing Cells
Next, we wanted to characterize the EGF-induced clusters of PtdIns(3)P-containing endocytic structures in GFP-2xFv-FYVE(Hrs) cells and determine their functionality. To this end, we performed immunolabeling with a number of endocytic markers for IF confocal microscopy and immuno-EM. We found that the GFP-2xFv-FYVE(Hrs) probe colocalized mainly with EEA1 (Figure 3A) and not significantly with CD63 (Figure 3A') and LBPA (Figure 3B). This suggests that the clusters are formed mainly by early endosomal structures that apparently cannot mature into later endocytic structures. Using immuno-EM, we found, however, that although a significant amount of EGFR localized to the limiting membranes of endocytic structures, some of the EGFR was still sorted into intraluminal vesicles (ILVs) of the small, clustered MVEs (Figure 3C). Although GFP-2xFv-FYVE(Hrs) did not colocalize significantly with LBPA in clusters (Figure 3D), it appeared that in some more matured profiles, GFP-2xFv-FYVE(Hrs) did colocalize with LBPA to a certain extent (Figure 3F). Importantly, this suggests that PtdIns(3)P- and LBPA-positive ILVs can reside within the same types of MVEs. Many of the endocytic structures found in GFP-2xFv-FYVE(Hrs) clusters contained electron-dense coats on their membranes. Interestingly, we found that these coats labeled positive for the PtdIns(3)P-binding sorting component Hrs (Raiborg et al. 2002), which strongly suggests that recruitment of Hrs to the endosomal membrane is not impeded by the binding of GFP-2xFv-FYVE(Hrs).
Discussion
In this study, we have investigated the spatiotemporal distribution of PtdIns(3)P in the endocytic and autophagic pathways using a dimerizable probe (Hayakawa et al. 2004). Dimerization shifted the distribution of the probe from a cytosolic to an endosomal pattern, verifying its functionality in terms of binding avidity. The nuclear localization that was also observed is possibly due to the tendency of GFP to get access to the nucleus by diffusion. The size of the GFP-Fv-FYVE(Hrs) probe could possibly still allow this to occur (Böhm et al. 2006). Our findings are in support of earlier data that localized the bulk of PtdIns(3)P to the limiting and intraluminal membranes of endosomes (Gillooly et al. 2000). However, because of the better time resolution obtained with the dimerizer-inducible probe, we were able to show the localization of PtdIns(3)P on limiting membranes at early time points, whereas longer dimerization periods led to accumulation of the GFP-Fv-2xFYVE(Hrs) probe within MVEs. This indicates that PtdIns(3)P binding does not interfere with internalization into ILVs, although we observed less intraluminal PtdIns(3)P as compared with our earlier studies with a recombinant 2xFYVE probe (Gillooly et al. 2000).
Treatment of GFP-Fv-2xFYVE(Hrs)-expressing cells with EGF induced clustering of endosomal structures with smaller diameter than regular endosomes. Although this finding was unexpected, it is consistent with the idea that PtdIns(3)P effectors are actively involved in the endosome fusion process and that sequestration of PtdIns(3)P interferes with this process (Simonsen et al. 1998b; Petiot et al. 2003). Alternatively, it could be possible that the formation of ILVs, which is induced upon EGF treatment (White et al. 2005), is inhibited by masking PtdIns(3)P. However, our observation that EGF receptors are to some extent still sorted into ILVs in the presence of GFP-Fv-2xFYVE(Hrs) favors the former interpretation.
The observed accumulation of tethered or docked early endosomes indicates that recruitment of the PtdIns(3)P-binding tethering factor EEA1 (Simonsen et al. 1998b) is still functional, despite the presence of the PtdIns(3)P-binding probe. Also, the observed localization of the Hrs-containing coat supports the interpretation that the probe does not displace endosomal PtdIns(3)P effectors at the expression levels used. This suggests that the endosomal pool of PtdIns(3)P is sufficient to allow binding of multiple effectors. It is therefore also intriguing to speculate that the PtdIns(3)P sequestration by the GFP-Fv-2xFYVE(Hrs) probe could inhibit the recruitment of kinases or phosphatases that facilitate PtdIns(3)P turnover (Taylor et al. 2000; Ikonomov et al. 2001). Without PtdIns(3)P turnover, the disassembly of the Hrs-containing coat and termination of EEA1-mediated tethering would be inhibited, thereby explaining why the endosome fusion process seems to be frozen.
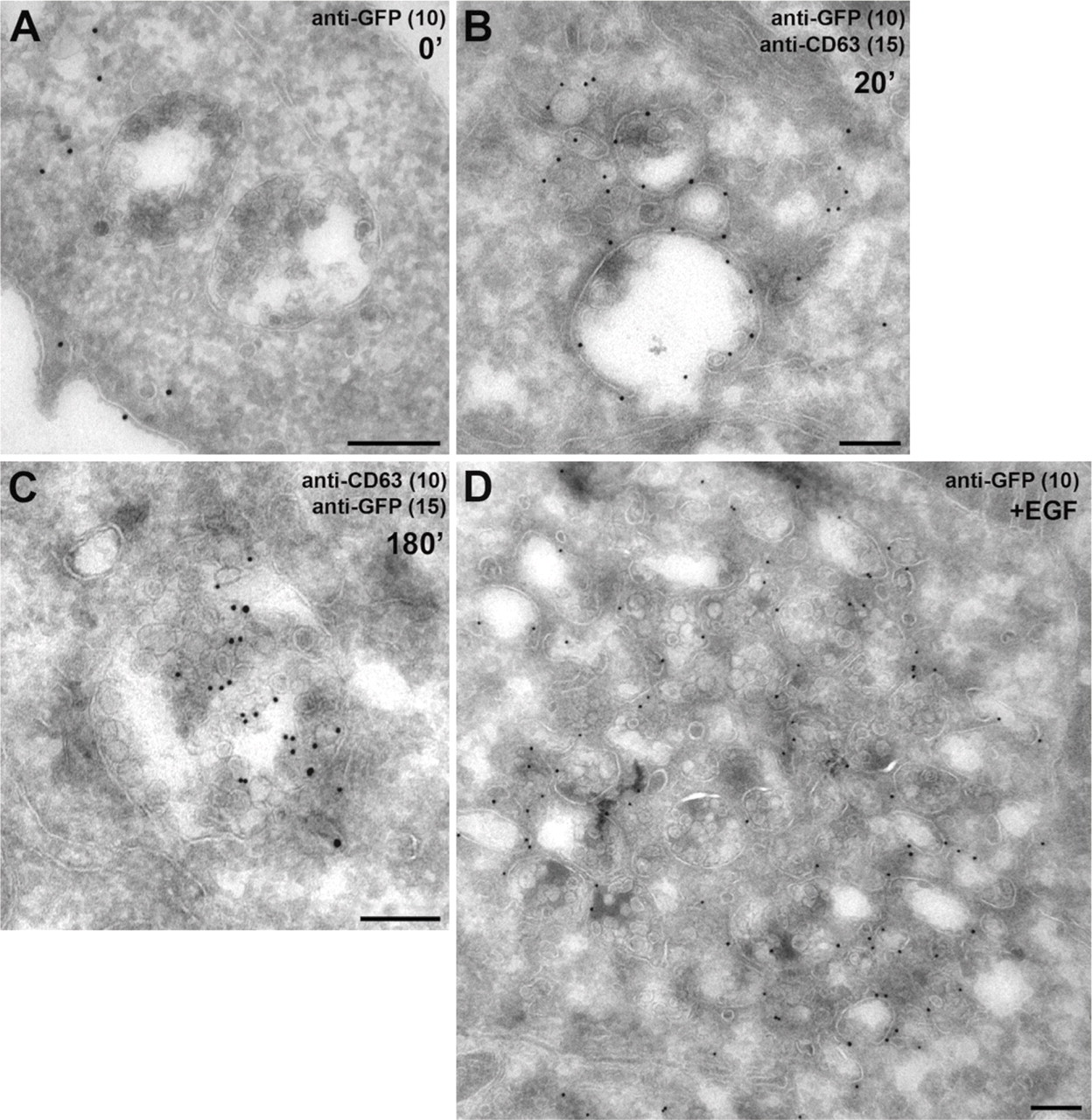
Ultrastructural analysis of cells transfected with the dimerizing GFP-FYVE(Hrs) probe. HeLa cells were transfected with GFP-Fv-FYVE-(Hrs) and incubated with the dimerizer for 0 min (
In conclusion, our studies using a probe that only localizes to PtdIns(3)P-containing membranes after addition of a dimerizer have indicated that PtdIns(3)P translocates from perimeter membranes to ILVs as endosomes mature. Although the GFP-Fv-2xFYVE (Hrs) probe has proven useful in this context, the endosome clustering caused by the probe in EGF-stimulated cells suggests that its sequestration of PtdIns(3)P may also interfere with endosomal functions, at least at high expression levels. The probe therefore has to be used with caution. In future studies, it could perhaps be possible to exploit the PtdIns(3)P-sequestering properties of the GFP-Fv-2xFYVE(Hrs) probe in temporal studies of PtdIns(3)P requirements in endocytic trafficking.
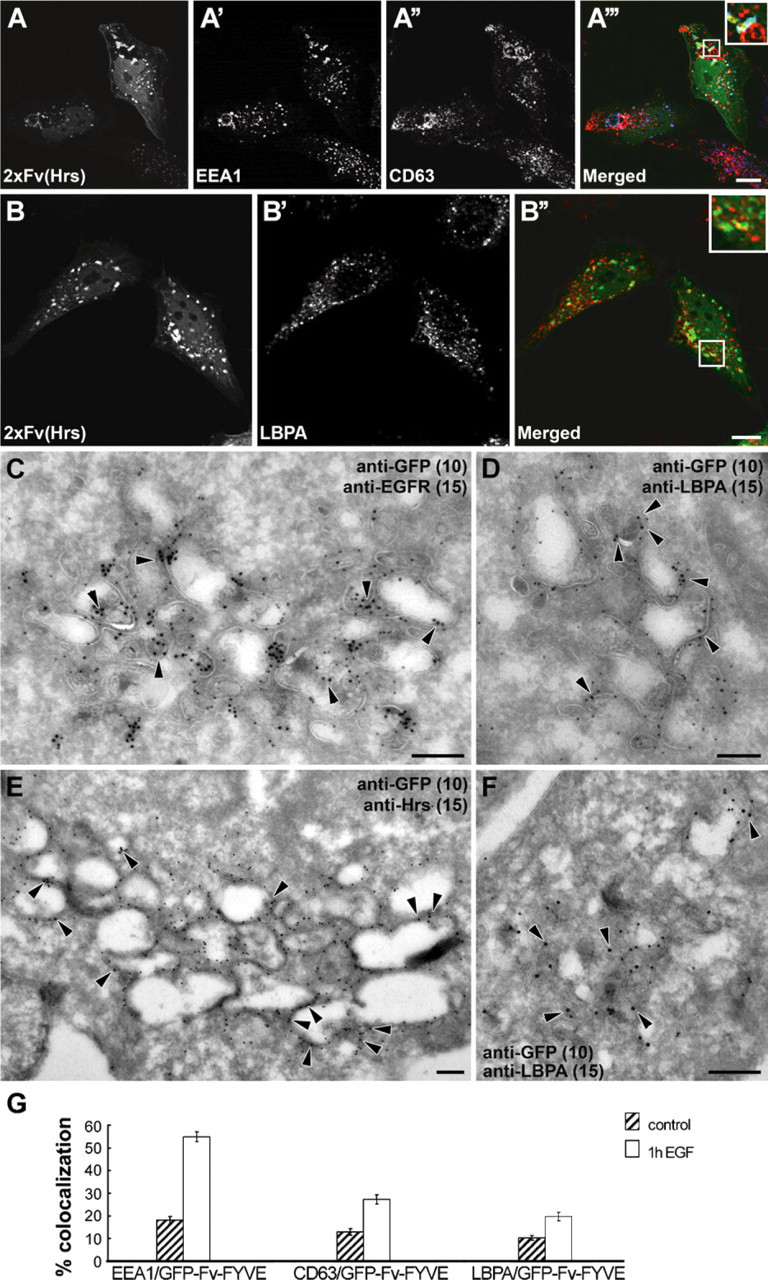
Characterization of the EGF-induced accumulated Ptdlns(3)P-containing endocytic structures in GFP-2xFv-FYVE(Hrs)-transfected cells. HeLa cells were transfected with the dimerizing GFP-Fv-FYVE(Hrs) probe, incubated with the dimerizer and 50 ng/ml EGF for 60 min, fixed, and prepared for immunofluorescence confocal microscopy (
Footnotes
Acknowledgements
S.S. and A.B. are supported by a career grant from the Functional Genomics (FUGE) Programme of the Research Council of Norway (158818). This work was also supported by the Norwegian Cancer Society and the Hartmann Family Foundation.
We thank Terje Johansen for providing GFP antibodies and ARIAD Pharmaceuticals for providing the ARGENT regulated homodimerization kit.
