Abstract
The thymus is a heterogeneous immune organ in which immature T-cells develop and eventually specialize to make certain immune responses of their own. Among various types of stromal cells in the thymus, thymic epithelial cells (TECs) have a crucially important function for presenting self-antigens and secreting cytokines to thymocytes for their maturation into T-cells. In this study we show that the p73 gene, a homologue of the tumor suppressor gene p53, was expressed in the nucleus of the human TEC in vivo and in TEC lines in vitro. Because p73 has the capacity to be a transactivator like p53, it may contribute to T-cell development in the context of TEC biology as regulated in the cell cycle and apoptosis.
T-
TECs have characteristics similar to those observed in keratinocytes of the epidermis, e.g., expression of cytokeratin molecules in addition to a morphological resemblance (Marx and Müller-Hermelink 1997; Shimosato and Mukai 1997). In fact, several reports have demonstrated genes encoding the Whn transcriptional factor or other types of molecules that are commonly responsible for regulation of epithelial cells of both the thymus and the skin (Robles et al. 1996; Frank et al. 1999; Nishio et al. 2000; Su et al. 2001). Several mutations are also known to cause the mutant mouse exhibiting the nude phenotype, which often has abnormal development of the thymus and skin, resulting in immunological deficiencies (Pantelouris 1973). Although the central role of epithelial cells in thymic function is widely recognized, little is known about the molecular mechanisms of organismic growth of TECs to perform functions such as antigen presentation and cytokine secretion.
p73 has been identified as a homologous molecule of the archetypal tumor suppressor p53 (Kaghad et al. 1997). As observed for p53, there is accumulating evidence that p73 has a role as a transactivator and controls the cell cycle and apoptosis (Takada et al. 1999; Irwin and Kaelin 2001). Recent studies have also demonstrated that p73 is epitheliotropic in human tissues, suggesting dependence of epithelial cells on a p73 molecule in regulation of the cell cycle (Lee et al. 1999; De Laurenzi et al. 2000). To examine the expression of p73 in human TECs, we investigated human thymic tissues and TEC lines by molecular and immunohistochemical analyses. The results show that human TECs exhibit nuclear localization of p73 in both tissues and cell lines, as demonstrated by analyses with a polyclonal antibody that we raised against p73 for this study. Together with the functional significance of p73, these findings suggest that epithelial cells in the human thymus depend on p73, at least in part, to regulate the cell cycle and/or cell death, for conserving the microenvironment to foster the development of thymocytes.
Materials and Methods
Tissues and Cell Lines
Normal human thymuses were obtained through the Hokkaido Children's Hospital and Medical Center from patients <3 years of age undergoing cardiovascular surgery for congenital heart disease. These were stored at —80C before cryostat sections were prepared. Six-μm sections of the tissues were cut and placed on glass slides. Then they were fixed in ice-cold acetone for 30 sec and dried. All tissue was obtained with approval by institutional review boards.
Human embryo kidney 293 cells were maintained with DMEM supplemented with 10% heat-inactivated fetal calf serum, 100 U/ml penicillin, and 100 μg/ml streptomycin in a humidified atmosphere at 37C and 5% CO2. These cells were transiently transformed by using the Lipofectamine 2000 reagent (Invitrogen; Carlsbad, CA) with p73α cDNA recombined in expression vector pcDNA3 (courtesy of Dr. G. Melino; University of Tor Vergata, Rome, Italy). TEC lines were cultured in
Antibodies
A rabbit polyclonal antibody against p73 was obtained from New Zealand White rabbits, which were immunized with a synthetic peptide of 18 amino acid residues, CKARKQPIKEEFTEAEIH, at the C-terminal portion of human p73. The IgG fraction purified from the antiserum for the following analyses was named ST-2G. H-79 is a rabbit anti-p73 polyclonal antibody against the N-terminal portion of p73 (Santa Cruz Biotechnology; Santa Cruz, CA). Anti-human cytokeratin-19 and anti-human CD3 monoclonal antibodies were purchased from DAKO (Carpinteria, CA), and the anti-human CD34 monoclonal antibody was from Nichirei (Tokyo, Japan).
Northern Blotting Analysis
A Northern blot membrane on which 2 μg of poly A+ RNA derived from various lymphoid tissues was used (Human Immune System MTN Blot II; Clontech, Palo Alto, CA). An EcoRI fragment of 1380 bp from pcDNA3-p73α was used as a probe. It included exons 7–14 and the 3′ untranslated region. Hybridization was performed as specified in the manufacturer's protocol. Briefly, the membrane was hybridized with radiolabeled probes in ExpressHyb solution (Clontech) at 68C for 20 hr. Then the membrane was washed in SSC containing 0.1% SDS solution with stepwise decreased salt concentrations followed by 2 × SSC, 1 × SSC, and 0.5 × SSC at 50C for 30 min. The membrane was subjected to autoradiography for consecutive days for up to 1 month at most.
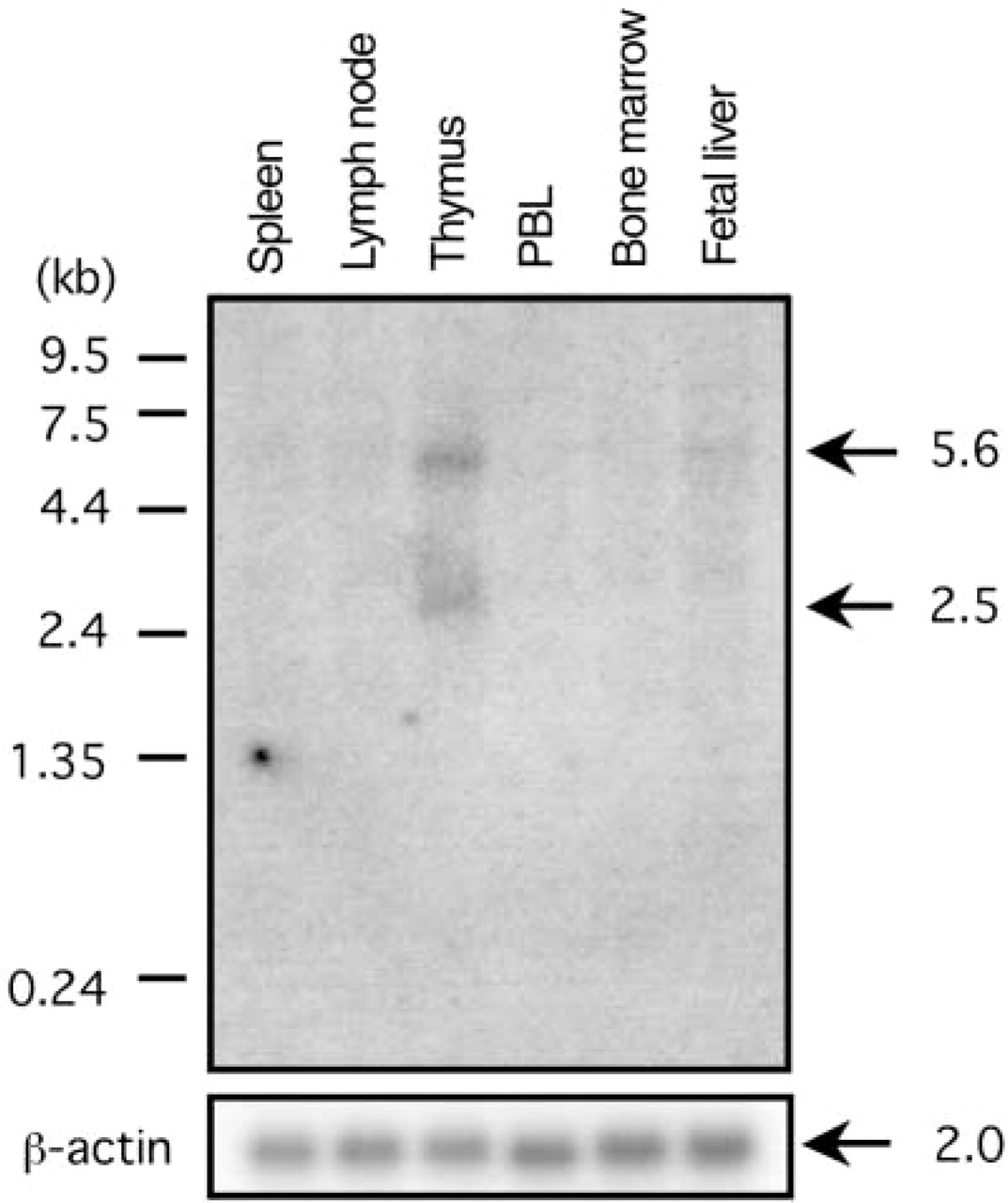
Northern blotting analysis of p73 transcript distribution in lymphoid tissues. The presence of p73 transcripts was analyzed using a 1.4-kb EcoRI-digested cDNA fragment of p73α. The distribution of the transcripts in 2 μg of poly A+ RNA from each of the tissues indicated high levels of p73α transcripts approximately 2.5 kb in length in thymus. Lower levels were noted in fetal liver. The expression of β-actin is shown as a control.
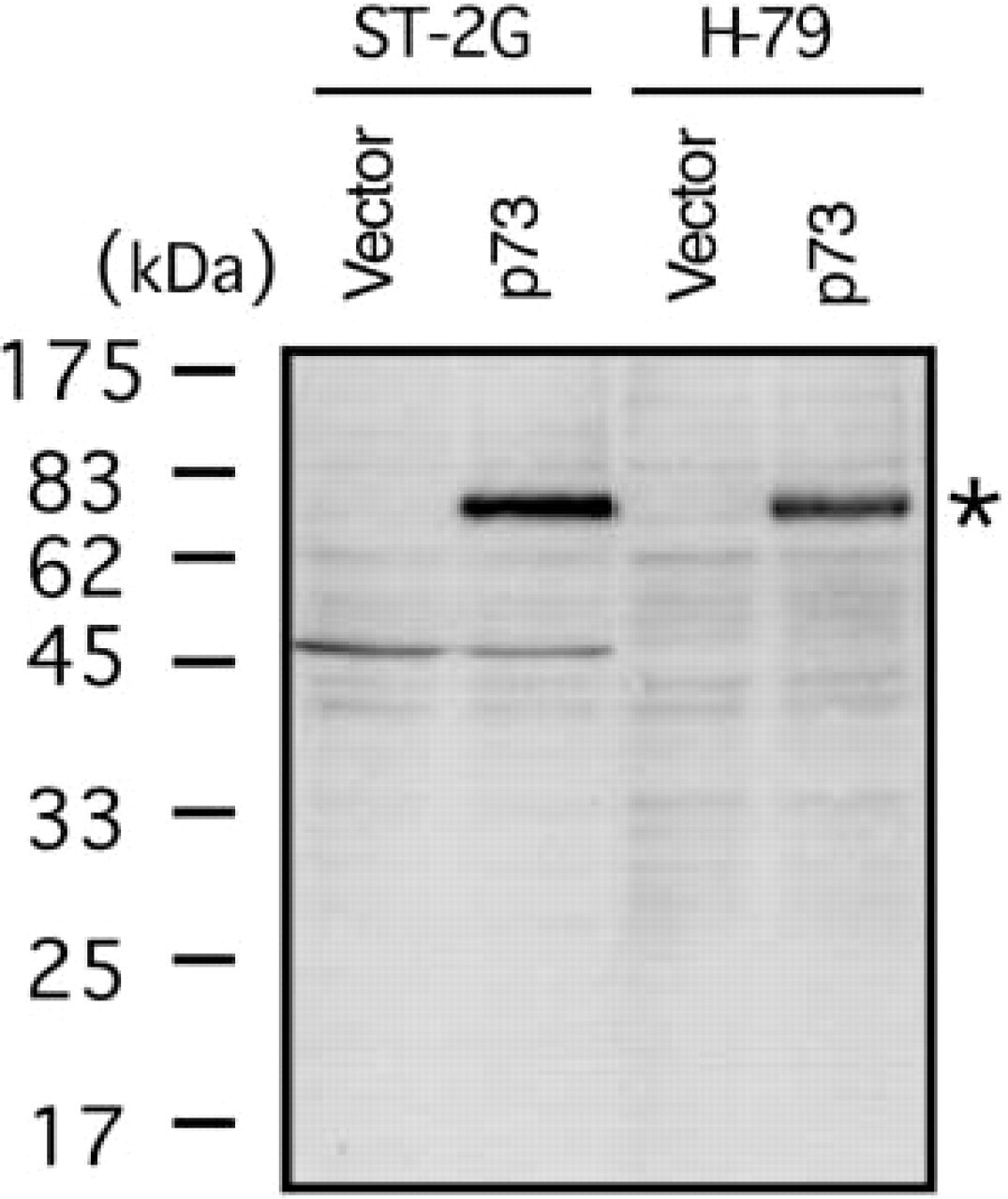
Qualification of a rabbit-polyclonal antibody against p73, ST-2G. Western analysis with ST-2G (10 μg/ml). The antibody reacted with 293-p73 transformants depicted as p73 in comparison to a mock transformant (depicted as vector). A similar band was observed in the study with H-79 (5 μg/ml) rabbit polyclonal antibody to p73. The band was little greater than that of the expected core protein (69.5 kD).
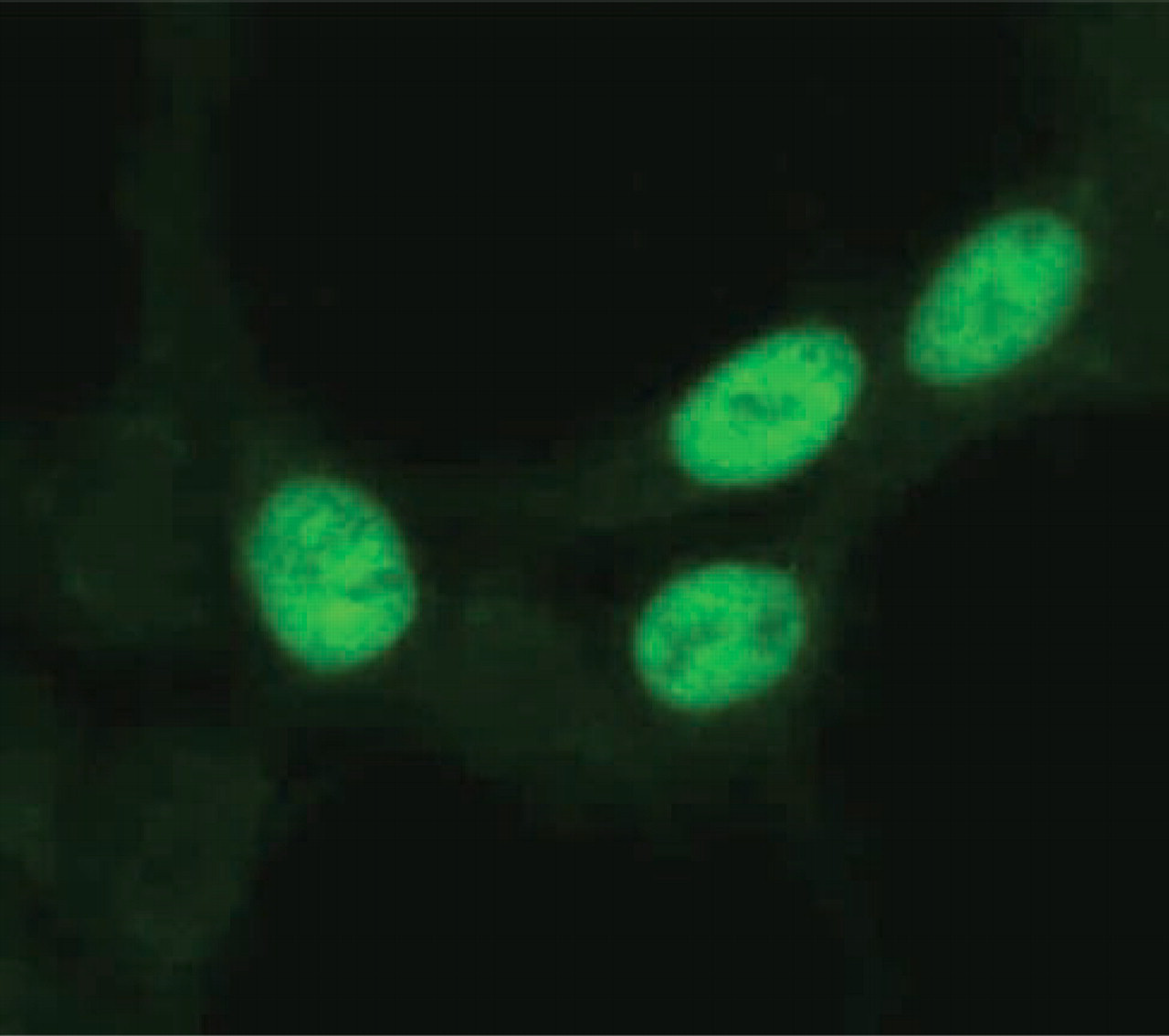
Immunostaining analysis of a transient transformant, 293-p73, with ST-2G antibody. 293-p73 cells were cultured on glass slides and fixed with ice-cold acetone for 30 sec. After washing with PBS, slides were stained with ST-2G (15 μg/ml). Signals were detected in the nuclei of the transformed cells expressing p73α protein but not in non-transformed cells. Original magnification ×600.
Western Blotting Analysis
Cell lysis was performed using subconfluent cultures by incubating 10-cm2 dishes with 1 ml of lysis buffer containing 0.5% NP-40, 10 mM Tris-HCl (pH 7.4), 150 mM NaCl, 1 mM EDTA, and protease inhibitors (Roche; Basel, Switzerland) for 30 min at 4C. Aliquots of the supernatants were applied to 10% SDS-PAGE gels under reducing conditions and transferred onto a polyvinylidene fluoride membrane. After blocking nonspecific protein binding using Tris-buffered saline-Tween-20 (TBST) containing 5% fat-free dry milk, the membrane was incubated with antibodies overnight at 4C, followed by reaction with a secondary antibody conjugated with peroxidase (Kirkegaard & Perry; Gaithersburg, MD). After washing with TBST, signals were detected by chemiluminescence with an ECL kit (Amersham; Piscataway, NJ).
Immunohistochemical Analysis
In the immunohistochemical analysis with two different types of antibodies (double-staining method), frozen sections of normal thymic tissues were fixed with ice-cold acetone and stained with ST-2G and a certain monoclonal antibody as previously described (Kojima et al. 2001). These specimens were visualized by Alexa 488 (green)-conjugated anti-rabbit IgG and Alexa 594 (red)-conjugated anti-mouse IgG (Molecular Probes; Eugene, OR), respectively. They were examined with a laser scanning confocal microscope (MRC 1024; Bio-Rad, Hercules, CA).
For immunofluorescent microscopic examination of cultured cells, cells were fixed with ice-cold acetone and stained with an anti-p73 polyclonal antibody (ST-2G), followed by reaction with FITC-conjugated anti-rabbit IgG (Kirkegaard & Perry). These specimens were analyzed with a fluorescence microscope (Carl Zeiss; Göttingen, Germany).
Results
p73 Transcripts Expressed in Normal Thymus
To examine p73 expression in the thymus, we performed Northern blotting analysis of multiple lymphoid tissue blots. As previously noted, the p73 gene is constituted of 14 exons at chromosome 1p36.33, of which the last four exons are mainly responsible for its alternative splicing, and the dominant spliced form is p73α, having all exons (Ichimiya et al. 2000). Results from the analysis with the fragments of p73α cDNA as a probe demonstrated transcripts in the thymus of 2.5 kb and 5.6 kb (Figure 1). According to the cDNA length of p73 transcripts in the database, the 2.5-kb band was expected to be a p73α transcript, as shown by RT-PCR analyses in somatic tissues, including thymus (Kaghad et al. 1997; Ueda et al. 1999). Therefore, the 2.5-kb band was presumed to represent a p73α isoform. Although we could not fully explain the nature of the 5.6-kb band, it might have been due to cross reaction with transcripts from the p63 gene, as previously reported (Senoo et al. 1998). In addition to the thymus, p73 transcripts were less abundantly detected in fetal liver when the filter was exposed for 1 month. In other tissues, including spleen, lymph node, peripheral blood leukocytes (PBLs), and bone marrow, p73 transcripts were not observed.
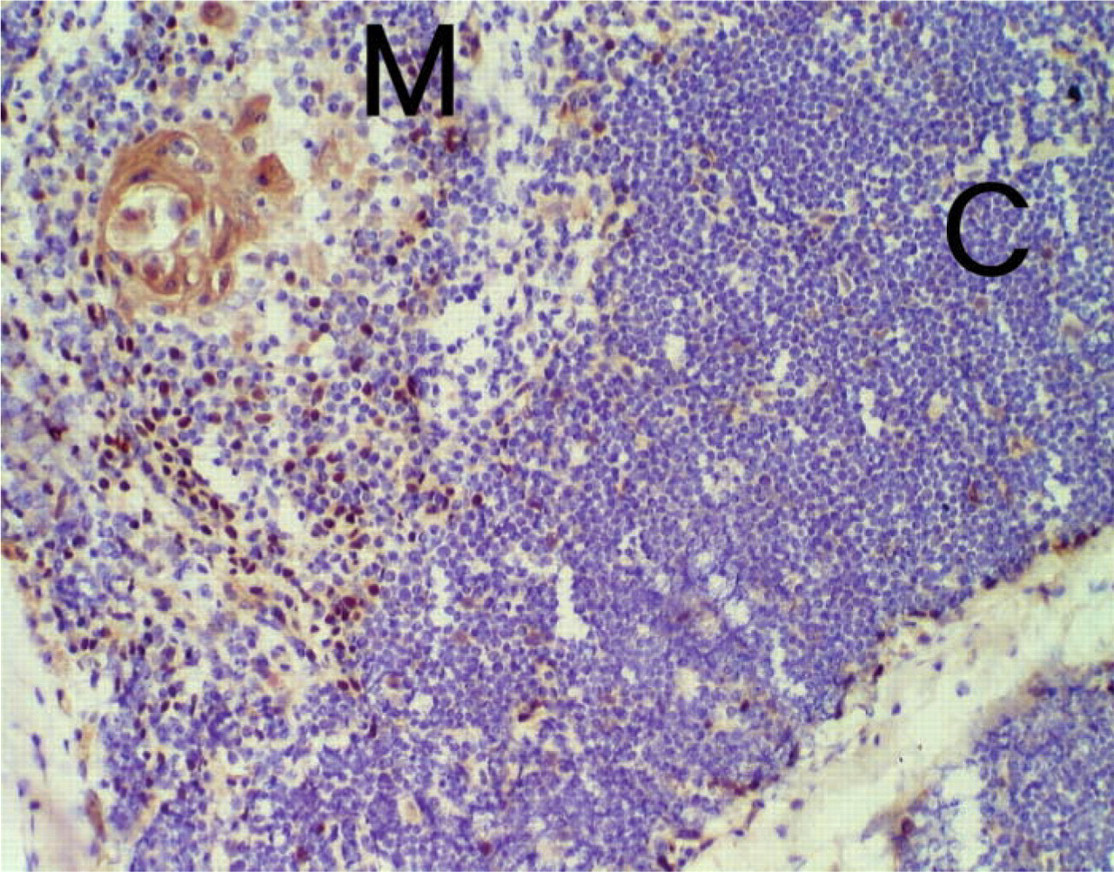
Immunohistochemistry of human thymus. Frozen section of human thymus was analyzed with antibody ST-2G (15 μg/ml). After secondary staining by using goat anti-rabbit IgG conjugated with peroxidase, diaminobenzidine and hematoxylin were used for visualization under a light microscope. p73-positive cells were distributed over the thymus and appeared as nuclear localization. Note that Hassall's body in the medulla was also stained. M, medullary region; C, cortical region. Original magnification ×200.
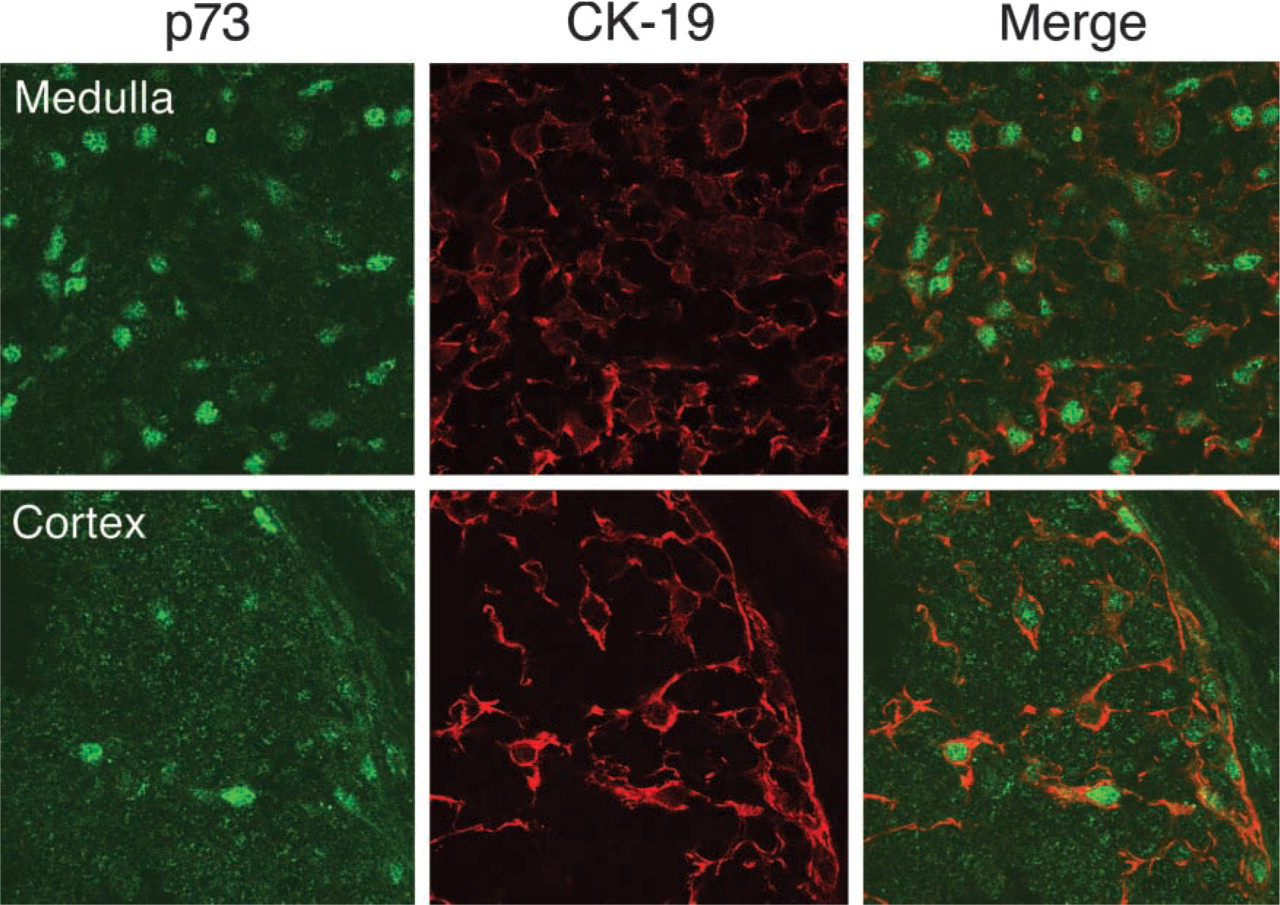
Confocal laser microscopy after double staining with ST-2G (p73) and an anti-CK-19 monoclonal antibody. CK-19 positive cells show nuclear expression of p73 in the thymus. Upper panel represents medullary region and lower panel shows cortical region. Original magnification ×400.
A Polyclonal Antibody Against p73
Human thymic tissues consist of various types of cells, such as developing T-cells, epithelial cells, macrophages, endothelial cells, and other stromal cells (Ritter and Boyd 1993; Plum et al. 2000). To identify cells expressing the p73 molecule in the thymus, we produced a polyclonal antibody (ST-2G) against the C-terminal portion of human p73. In Western blotting analysis with ST-2G, a band was detected at approximately 73 kD in a lane of 293 cells that were transiently transfected with p73α cDNA (293-p73) (Figure 2). Seemingly, polyclonal antibody H-79 (purchased from Santa Cruz Biotechnology), which was generated with the N-terminal region of p73α, exhibited the same band as that found in ST-2G. These results indicated that the two antibodies recognized the p73 molecule. Although the molecular weight of the core protein of p73α was expected to be 69.5 kD, this subtle discrepancy in the molecular size can probably be explained by post-translational modification of p73α, such as phosphorylation in cells (Agami et al. 1999). Further immunohistochemical study of 293-p73 also demonstrated that ST-2G detected p73α in the nucleus (Figure 3). Therefore, we concluded that ST-2G was available for the following immunohistochemical studies of p73.
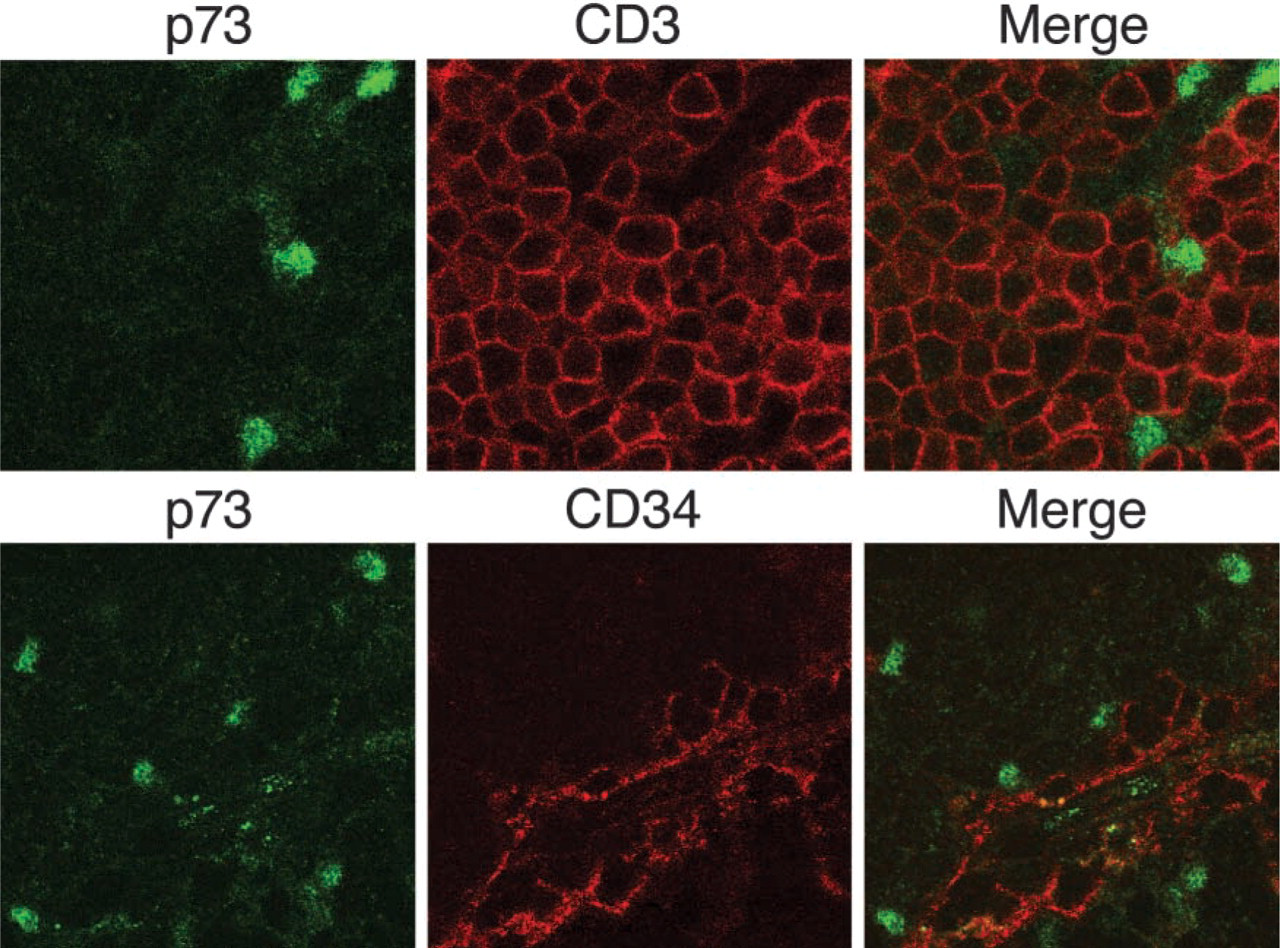
Confocal laser microscopy after double staining with ST-2G (p73) and an anti-CD3 or anti-CD34 monoclonal antibody. Upper panel presents double staining with p73 and CD3 in the cortex, lower panel is double stained with p73 and CD34 in a vascular area of the medulla. In both experiments, p73-reactive nuclei were not co-localized with CD3- or CD34-positive cells. Original magnification ×450.
p73 Expression in the Nuclei of Human TECs
Microscopic examination of human thymus immunostained with ST-2G revealed that p73 appeared in the nuclei of cells that were widely localized in the subcapsular region, cortex, and medulla (Figure 4). This distribution of p73 throughout the thymus was like that of TECs. We also found that Hassall's corpuscles, a particular structure formed by TECs in the medulla, expressed p73. To further identify cells presenting the p73 molecule in the thymus, we employed confocal laser microscopy for double immunostaining analysis with ST-2G and several monoclonal antibodies specific for certain cell types. The results demonstrated that p73 was localized in the nuclei of cells expressing cytokeratin-19 (CK-19), which is a ubiquitously expressed cytokeratin in human TEC (Figure 5) (Marx and Müller-Hermelink 1997; Shimosato and Mukai 1997). This strongly suggested that cells expressing the p73 protein in the thymus were epithelial cells. We also noted that the expression of p73 was mostly exclusive in CK-19-positive cells, but the intensity varied in some CK-19-positive populations. Moreover, p73-positive cells did not co-localize with cells that immunoreacted to CD3 or CD34, which are a pan-T-cell marker and a marker for endothelial cells, respectively (Figure 6). This indicated that p73 was specifically expressed in TECs, but not in any populations of T-cells or other types of stromal cells in the human thymus.
p73 Expression of TEC Lines
To further study p73 expression in TECs, two human TEC lines were subjected to an immunohistochemical analysis. Fernandez et al. (1994) established these cell lines, F2.5B6 and P1.4D6, which originated from TECs of the cortex. Immunohistochemical study with ST-2G showed nuclear localization of p73 in these TEC lines (Figure 7), in agreement with its in vivo localization in thymic tissues as shown in Figure 4.
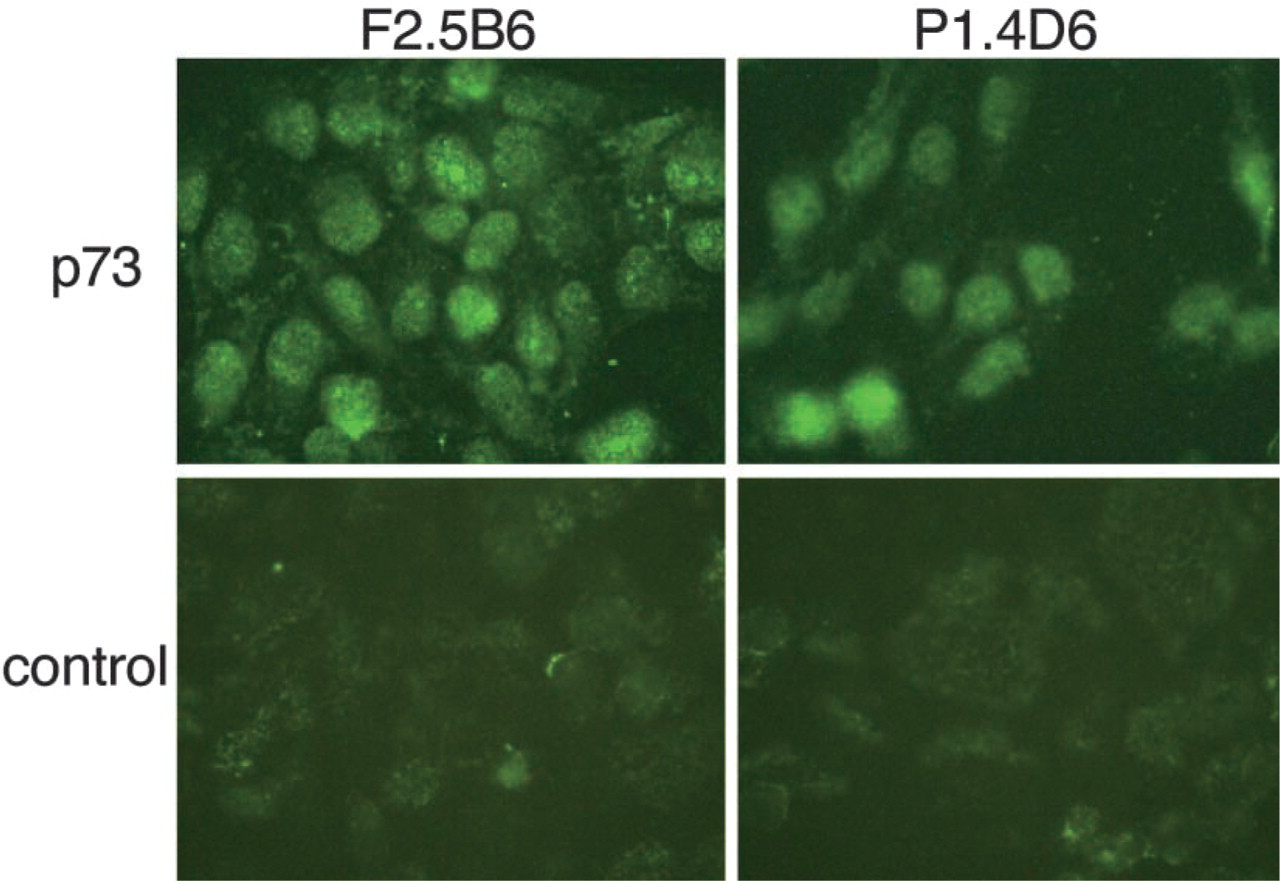
p73 expression in human TEC lines. Immunostaining of F2.5B6 and P1.4D6 with ST-2G. p73 is expressed in the nuclei of both TEC lines as observed in thymic tissue sections. Original magnification ×500.
Discussion
In this study we demonstrated the nuclear expression of p73 in epithelial cells of the human thymus as well as in TEC lines. The antibody used in this study, ST-2G, was against an 18-mer peptide of the C-terminal region of p73 observed in both p73α (a full-length form with 636 aa) and a somewhat shorter form, p73ε (a spliced-out form of exon 11 and 13 with 555 aa) among spliced-variant species of the p73 gene (Ueda et al. 1999; Irwin and Kaelin 2001). This raises the question of which p73 is dominant in human TECs. Although several transcriptional species are found in the thymus by RT-PCR analysis, p73α is probably dominant because Northern blotting analysis showed a band presumably corresponding to a p73α transcript (Figure 1), in addition to reported evidence that p73α is the main isoform in the thymus (Kaghad et al. 1997). In TECs, a transcriptional activity of p73α may be regulated by its SAM domain in the C-terminal, which is a regulatory element in cells (Chi et al. 1999; Ozaki et al. 1999). Heteromerization of p73α to other p73 isoforms may also affect its transcriptional activities (Ueda et al. 2001).
It has been reported that a substantial number of genes are activated by p73, like p53. They include mdm2, p21, gadd45, cyclin G, bax, and insulin-like growth factor binding protein (IGFBP3), which regulate the cell cycle and apoptosis (Yang and McKeon 2000). Physiologically, cell cycle control of TECs should be important for maintaining the functional architecture of the thymus because TECs remarkably influence the selection and maturation of developing thymocytes (Viret and Janeway 1999; Annunziato et al. 2001). Some tumors derived from TECs, such as thymoma and thymic carcinoma, were less likely to express p73 in the nucleus (data not shown). Therefore, a p73-associated pathway of TECs may play a central role in thymic function in the context of epithelial growth regulation by transactivating the aforementioned target genes. Nevertheless, several reports indicate that the corresponding loci including the p73 gene do not appear to be responsible for tumorigenesis of TEC-derived tumors (Sait et al. 2001; Zettl et al. 2000), as seen in other human malignancies (Ichimiya et al. 1999).
Most CK-19-positive cells whose subtype is distributed over thymic tissues showed nuclear localization of p73, though some CK-19-positive cells exhibited faint or no expression of p73 in the nucleus. This may imply that there is some population of TECs not under the control of p73. Ontogenically, the thymic epithelial rudiment in humans is widely recognized to be formed from the endoderm of the third pharyngeal pouch and ectoderm of the corresponding branchial clefts, indicating heterogenous epithelial components in the thymus (Manley 2000). In fact, TECs are classified by various keratin subtypes in conjunction with their intrathymic localization (Marx and Müller-Hermelink 1997). The low expressors of p73 may represent certain cell types in the heterogenous TEC population or in a particular state of the cell cycle.
In summary, p73 is expressed in human TECs and may have a role in regulating the cell cycle of these cells. p73 null mice have neurological and immunological defects but no obvious abnormalities of lymphoid populations (Yang et al. 2000). As previously reported, p63 is an another p53 family member that has a role in cell cycle control and shows epitheliotropism in the skin, like p73 (Osada et al. 1998; Celli et al. 1999; Parsa et al. 1999). This implies that p53 family members may redundantly or complementarily work with each other in the cell cycle as well as in phenotypic regulation of epithelial cells in the thymus (van den Elsen et al. 1998; Yun et al. 1999; Gobin and van den Elsen 2000), as is often seen within other families of transcriptional factors, such as the homeodomain proteins or the myc gene family.
Footnotes
Acknowledgements
Supported in part by Grants-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports and Technology of Japan and by the Akiyama Foundation of Japan.
We are grateful to Gerry Melino (University Tor Vergata; Rome, Italy) for p73α cDNA. We also thank Avinash Bhandoola (University of Pennsylvania) for helpful discussions, and Laura Crisá (Scripps Research Institute) and Shigeo Yokoyama (Hokkaido Children's Hospital and Medical Center, Japan) for generous assistance.
