Abstract
This study localized malondialdehyde (MDA, a toxic byproduct of lipid peroxidation), nitrotyrosine [NT, a cytotoxic byproduct of nitric oxide (NO)], and nitric oxide synthase isomers (NOS) in normal and diseased human corneas. Normal corneas (n=11) and those with clinical and histopathological diagnoses of keratoconus (n=26), bullous keratopathy (n=17), and Fuchs' endothelial dystrophy (n=12) were examined with antibodies specific for MDA, NT, eNOS (constitutive NOS), and iNOS (inducible NOS). Normal corneas showed little or no staining for MDA, NT, or iNOS, whereas eNOS was detected in the epithelium and endothelium. MDA was present in all disease groups, with each group displaying a distinct pattern of staining. NT was detected in all keratoconus and approximately one half of Fuchs' dystrophy corneas. iNOS and eNOS were evident in all the diseased corneas. Keratoconus corneas showed evidence of oxidative damage from cytotoxic byproducts generated by lipid peroxidation and the NO pathway. Bullous keratopathy corneas displayed byproducts of lipid peroxidation but not peroxynitrite (MDA but not NT). Conversely, Fuchs' dystrophy corneas displayed byproducts of peroxynitrite with little lipid peroxidation (NT » MDA). These data suggest that oxidative damage occurs within each group of diseased corneas. However, each disease exhibits a distinctive profile, with only keratoconus showing prominent staining for both nitrotyrosine and MDA. These results suggest that keratoconus corneas do not process reactive oxygen species in a normal manner, which may play a major role in the pathogenesis of this disease.
Keywords
The Cornea is a transparent avascular tissue that allows transit of incident light to the more posterior ocular structures. Therefore, this tissue is constantly exposed to a wide spectrum of light, including the ultraviolet (UV) range. UV exposure is a well-documented environmental stress factor that generates free radicals and reactive oxygen species detrimental to most cells and tissues (Wenk et al. 2001). The cornea absorbs most of the UV entering the eye, suggesting that the cornea would be especially susceptible to damage from reactive oxygen species (ROS). In the healthy cornea, a number of defensive mechanisms are present to minimize and reduce this risk. Indeed, 20–40% of the soluble protein content of the cornea is an isoenzyme of aldehyde dehydrogenase (ALDH3), which directly absorbs UV and removes cytotoxic aldehydes produced by UV-induced lipid peroxidation (Abedinia et al. 1990; Gondhowiardjo et al. 1991). Animal studies have shown that this enzyme plays an important protective role in preventing UV damage and that mouse strains expressing enzymatically defective ALDH3 are especially susceptible to UV-induced pathology (Downes et al. 1993,1997; Shiao et al. 1999; Pappa et al. 2001). Furthermore, the cornea is rich in antioxidant enzymes such as superoxide dismutase (SOD), catalase, glutathione peroxidase, and glutathione reductase, all of which help in the removal of free radicals and ROS generated by constant absorption of UV light (Rao et al. 1987). Surprisingly, the potential role of oxidative damage in common human corneal diseases has not been thoroughly investigated.
The most common indications for corneal transplantation are keratoconus and bullous keratopathy (Liu and Slomovic 1997; Lois et al. 1997; Cursiefen et al. 1998). Keratoconus is a non-inflammatory disorder that occurs in young adults (second to fourth decade) and its hallmarks are focal scarring, breaks in Bowman's layer, and thinning of the corneal stroma (Kenney et al. 2000; Zhou et al. 1996). As the cornea thins, patients have irregular corneal astigmatism that can lead to severe visual distortion (Krachmer et al. 1984; Rabinowitz 1998). In contrast, bullous keratopathy patients are typically older (past the fifth decade) with corneas that are edematous, fibrotic, and occasionally vascularized. Bullous keratopathy typically occurs after surgical intervention for cataract removal with (pseudophakic) or without (aphakic) placement of an intraocular lens. Fuchs' endothelial dystrophy is another corneal disorder in which altered fluid balance leads to excessive swelling of the stroma, loss of transparency, and decreased visual acuity. It has a hereditary component, occurring as early as the fourth decade of life, and patients may require a corneal transplant well before the formation of any cataract.
Recently, several lines of evidence have emerged suggesting that corneal components involved in protection against oxidative damage are altered in these disorders. ALDH3 is reportedly abnormal in keratoconus corneas (Gondhowiardjo and van Haeringen 1993; Gondhowiardjo et al. 1993). Extracellular forms of SOD have been reported to be depressed in both keratoconus and bullous keratopathy (Behndig et al. 1998,2001). Although these changes suggest that the corneas are more susceptible to UV-induced damage, no direct evidence of this type of damage is present. It is not clear whether diseased corneas, specifically keratoconus and bullous keratopathy corneas, accumulate byproducts of oxidative damage.
Lipid peroxidation occurs in response to elevated levels of ROS with the liberation of reactive aldehydes, such as malondialdehyde (MDA) and 4-hydroxy-2-nonenal (HNE) (Gutteridge and Halliwell 1990; Esterbauer et al. 1991). In vitro studies show that MDA crosslinks via Schiff-base conjugation with primary amino groups of phospholipids (Bhuyan et al. 1996) and proteins (Ganea and Harding 2000). Therefore, demonstration of the MDA hapten in tissues is considered evidence of lipid peroxidation. Similarly, nitric oxide (NO), which serves as a mediator in diverse processes throughout the eye (Becquet et al. 1997), can have cytotoxic effects. The cytotoxic effects of NO are mediated by peroxynitrite, a potent oxidant produced by the rapid reaction of NO and superoxides (see Figure 1) (Beckman and Koppenol 1996). Peroxynitrite reacts with proteins, non-protein sulfhydryls, methionine, DNA, and phospholipids (Beckman 1996; Kooy et al. 1997; Murphy et al. 1998). Peroxynitrite reacts with the phenolic ring of tyrosine to form a stable product, nitrotyrosine (NT) (Kooy et al. 1995; Beckman and Koppenol 1996). Demonstration of NT residues is therefore considered a specific marker for peroxynitrite and nitrogen-related oxidants (Kooy et al. 1995,1997; Cross et al. 1997; Saleh et al. 1997).
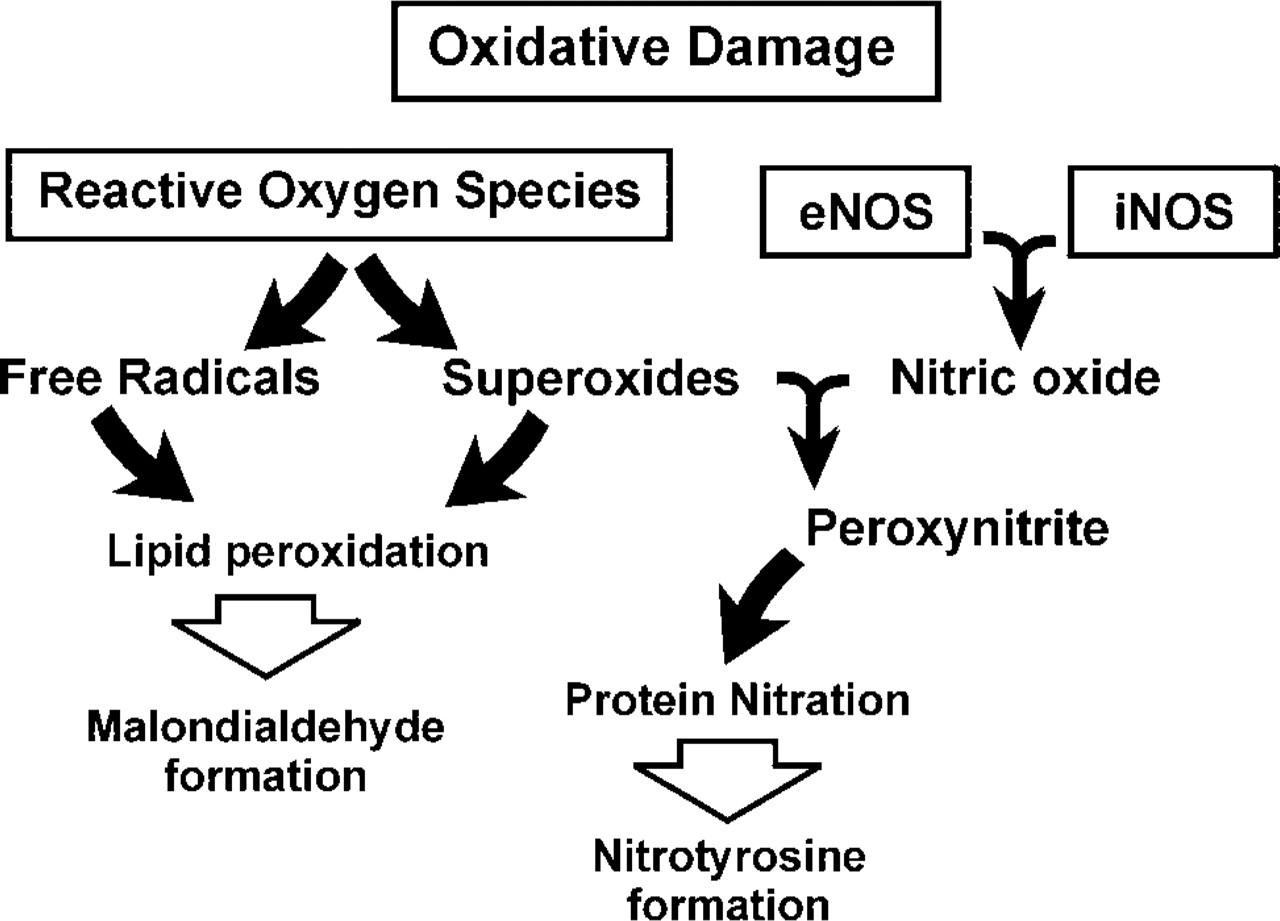
Schematic of oxidative pathways.
In this study we examined over 90 corneas for evidence of oxidative damage. Using antibodies to detect MDA (lipid peroxidation), NOS isoenzymes (nitric oxide production), and NT (peroxynitrite), we demonstrated that (a) NT appeared differentially in diseased corneas (keratoconus > Fuchs' dystrophy > bullous keratopathy > normal), (b) eNOS could be found in both normal and diseased corneas, but iNOS was found only in the diseased corneas, (c) in keratoconus corneas at the site of Bowman's layer breaks, positive staining for NT correlated with the appearance of iNOS, and (d) MDA was also differentially associated with diseased corneas: keratoconus > bullous keratopathy > Fuchs' dystrophy > normal. These observations provide direct evidence that oxidative damage occurs in common human corneal disorders. Importantly, the distinct MDA and NT staining patterns for each disorder may give valuable clues to the underlying mechanisms involved in their pathogenesis.
Materials and Methods
Archived formalin-fixed, paraffin-embedded corneas with confirmed clinical and histological diagnoses of keratoconus (n=26), bullous keratopathy (n = 17), and Fuchs' endothelial dystrophy (n = 12) were obtained for this retrospective analysis. Eleven normal corneas with no histopathological changes were also included in this study. Five-micrometer sections were mounted on poly-L-lysine coated slides. After deparaffinization and hydration, sections were incubated and permeabilized in 0.1% aqueous saponin at room temperature (RT) for 60 min and then rinsed three times in PBS. Endogenous peroxidase activity was blocked in all tissues by a 5-min incubation in 3% hydrogen peroxide (Sigma Chemical; St Louis, MO). The sections were incubated overnight at 4C with the following antibodies: (a) rabbit polyclonal anti-iNOS (the inducible isoform of NOS), raised to a 21-kD protein fragment corresponding to amino acids 961–1144 of mouse macrophage iNOS (#N32030; Transduction Laboratories, San Diego, CA); (b) mouse monoclonal anti-eNOS (endothelial NOS), raised to a 20-kD protein fragment corresponding to amino acids 1030–1209 of human eNOS (#N30020, Transduction Laboratories); (c) mouse monoclonal antibody to nitrotyrosine (clone 1A6; Upstate Biotechnology, Lake Placid, NY) or rabbit polyclonal antibody to nitrotyrosine (raised against nitrated keyhole limpet hemocyanin; Upstate Biotechnology), and (d) rabbit anti-MDA antiserum (#MDA 11-C; Alpha Diagnostics, San Antonio, TX). This was followed by incubation (60 min, RT) in biotinylated anti-mouse or anti-rabbit secondary antibodies (Vector Laboratories; Burlingame, CA). The immunolabeled sites were visualized using an avidin-biotin-peroxidase kit (Vector) with diaminobenzidine tablets (Sigma) used at the development stage. All sections were allowed to develop for 15 min, then immediately washed and counter-stained with Mayer's hematoxylin (Sigma).
Corneas with fungal keratitis were used as a positive control for NT, iNOS, and eNOS. Negative controls included omission of the primary antibody and equimolar concentrations of purified IgG from normal rabbit serum (for the polyclonal primary antibodies). These samples were routinely negative. To confirm the specificity of the staining for nitrotyrosine the antibody was incubated with 10 mM nitrotyrosine as previously described (Beckman 1996; Kooy et al. 1997; Murphy et al. 1998). In addition, some tissue sections were treated with fresh 1 M sodium hydrosulfite (pH 9) for 3 min to reduce any tissue nitrotyrosine to aminotyrosine. Sodium hydrosulfite was prepared immediately before use in 0.05 M Tris-Hcl/0.15 M NaCl buffer and the pH was subsequently adjusted to 9 with sodium hydroxide. In some experiments, the activities of polyclonal antibodies to iNOS and eNOS were neutralized by pre-incubation with an excess of the respective specific blocking peptides against which the antibodies were raised. In both cases, this procedure completely abolished the staining, confirming the specificity of the antibodies.
The sections were examined and photographed with Kodak Ektachrome film using an Olympus BH-2 microscope. The developed prints were digitized using a MicroTek scanner and printed with an Epson Photo printer. Statistical analysis was performed using Fisher's exact test. p<0.05 was considered significant.
Results
Lipid peroxidation occurs in response to elevated levels of ROS. This can lead to cell membrane destruction with the liberation of reactive aldehydes, such as malondialdehyde (MDA) and 4-hydroxy-2-nonenal (HNE) (Gutteridge and Halliwell 1990; Esterbauer et al. 1991). Lipid peroxidation and reactive aldehydes are associated with chronic liver diseases, aging, amyotrophic lateral sclerosis, ischemia, and diabetes mellitus (Paradis et al. 1997; Yoshino et al. 1997; Ohhira et al. 1998; Pedersen et al. 1998). We examined archived human tissue by immunohistochemistry for the presence of MDA and, by extension, lipid peroxidation in diseased corneas.
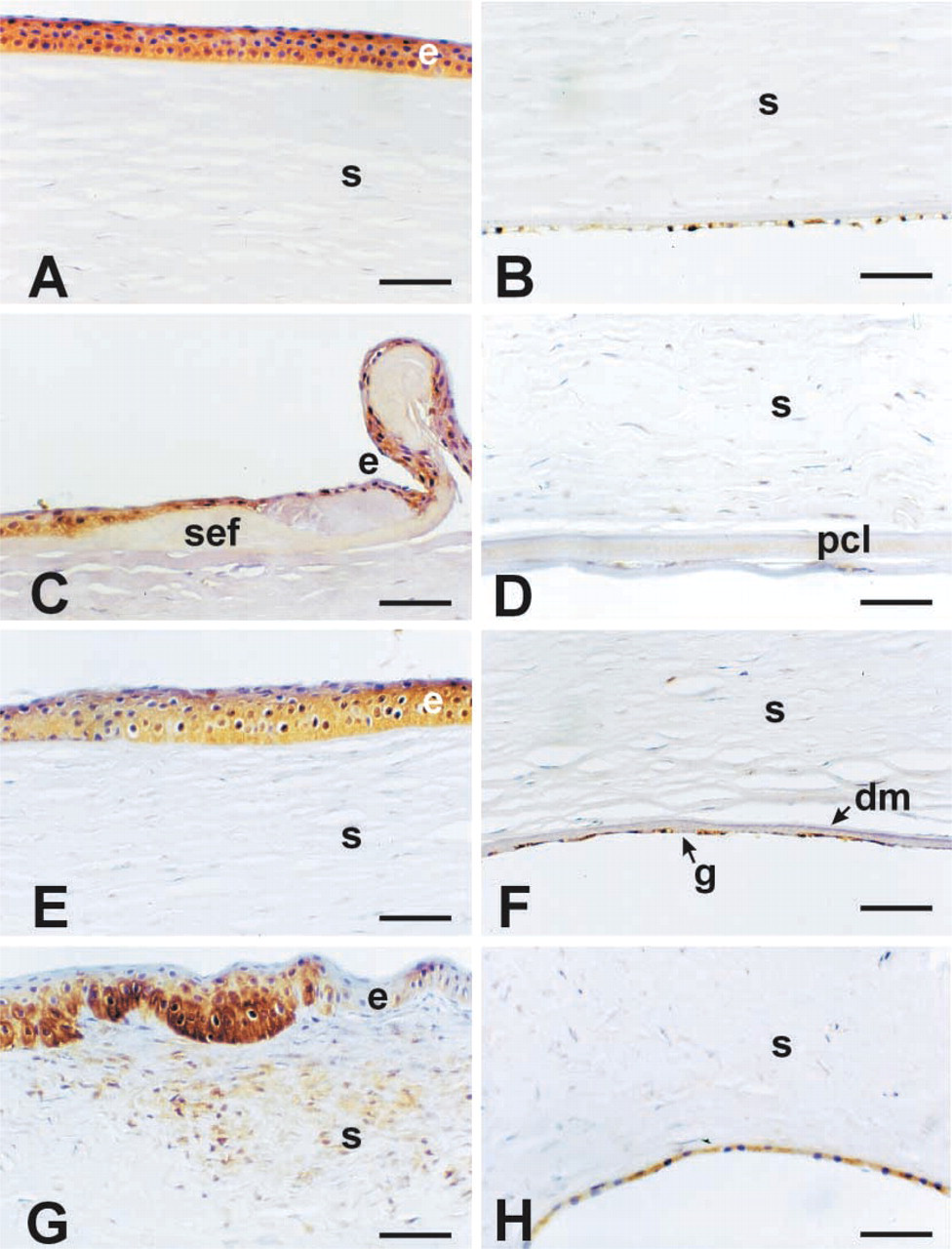
Normal human corneas generally lacked MDA staining in any of the tissue layers (Figures 2A and 2B; Table 1). The single case showing trace staining with this antibody is shown in Figure 2A. Bullous keratopathy corneas consistently had a positively stained epithelium, with strong diffuse staining in areas of sub-epithelial fibrosis (SEF: Figure 2C). The posterior collagenous layer (also referred to as the retrocorneal fibrous membrane) was also strongly positive for MDA in these corneas (Figure 2D. About two thirds of Fuchs' dystrophy corneas stained with MDA in the epithelium (Figure 2E but were consistently negative in the posterior cornea, including the abnormal guttae associated with Descemet's membrane (Figure 2F. Keratoconus corneas were strongly stained for MDA in the epithelium and in areas of stroma adjacent to breaks in Bowman's layer (Figures 2G and 2H). These observations indicate that, in these disease states, the cornea accumulates byproducts of lipid peroxidation.
Nitric oxide, produced by oxidative deamination of L-arginine by NOS, serves as a mediator in diverse and complex cellular processes throughout the eye (Becquet et al. 1997). Increased production of NO, however, can have cytotoxic effects mediated by peroxynitrite, a potent oxidant produced by the rapid reaction of NO and superoxides (see Figure 1) (Beckman and Koppenol 1996). Demonstration of NT residues is considered a specific marker for peroxynitrite and nitrogen-related oxidants (Cross et al. 1997; Kooy et al. 1995,1997; Saleh et al. 1997). We therefore examined these corneas for immunoreactivity using both monoclonal and polyclonal antibodies to NT.
Specific patterns staining for NT were evident in the keratoconus (Figures 3G and 3H) and Fuchs' endothelial dystrophy (Figure 3E corneas, whereas minimal or no staining was seen in the normal (Figures 3A and 3B) or bullous keratopathy corneas (Figures 3C and 3D). It was interesting that bullous keratopathy corneas, which stained well for MDA in areas of sub-epithelial fibrosis and the posterior collagenous layer, lacked any detectable staining for NT. In keratoconus corneas, NT staining was extensive throughout the epithelial layer and extended into the stroma adjacent to disruptions in Bowman's layer (Figures 3G and 3H). NT staining was completely blocked by pre-incubation of the antibody with 10 mM nitrotyrosine, thus confirming the specificity of the positive staining (Figure 4B. In addition, pretreatment of tissue sections with sodium hydrosulfite (to reduce nitrotyrosine to aminotyrosine) led to either markedly reduced staining or no staining at all (Figure 4C.
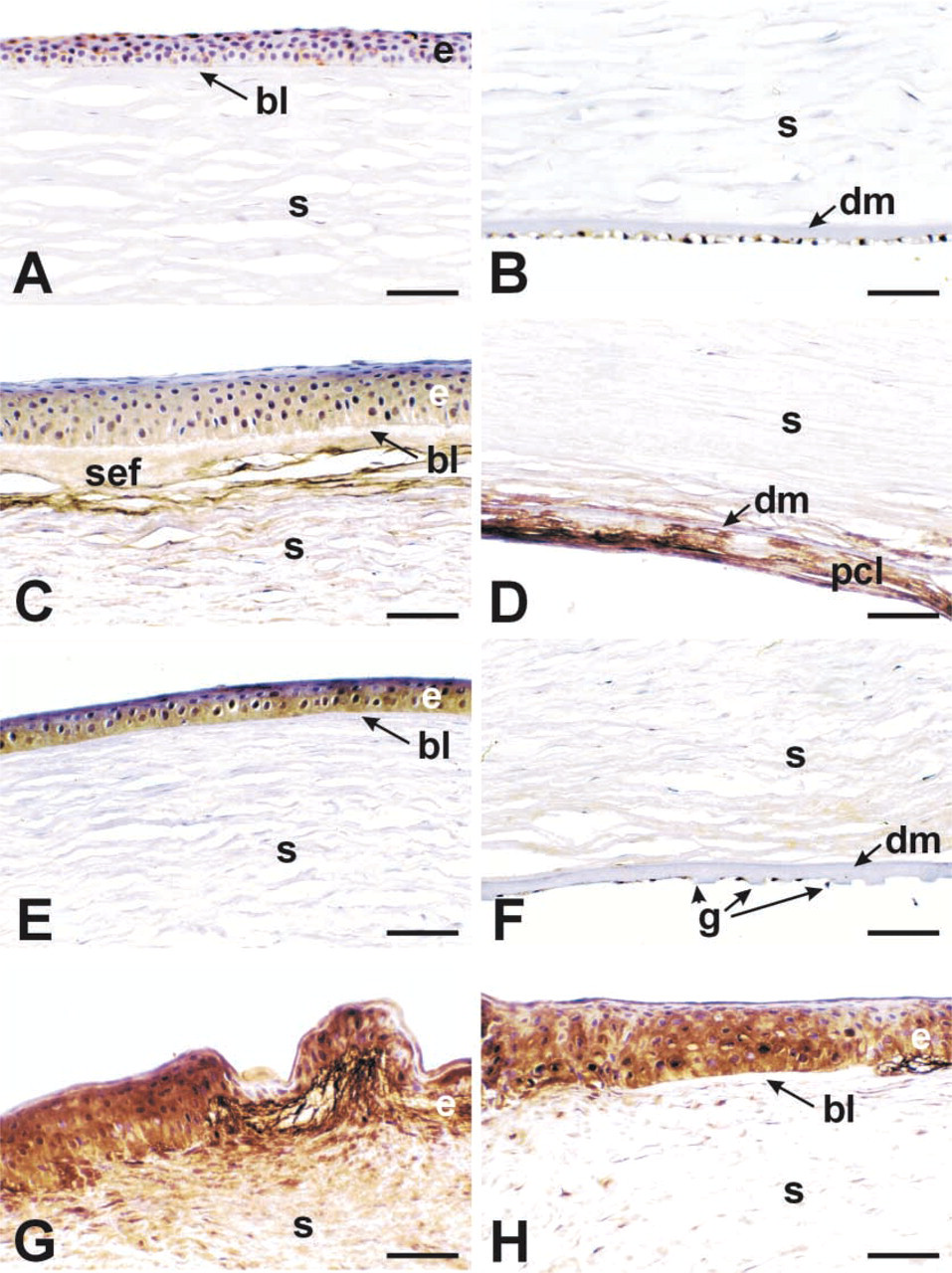
MDA immunoreactivity in the various disease groups. Normal (
Because NT staining suggested that the diseased corneas produced peroxynitrite, it was of interest to determine whether these corneas also produced NO locally. Therefore, each cornea was stained for the presence of both eNOS and iNOS. Immunoreactivity for eNOS was noted in normal corneas in the epithelium and endothelium but was not apparent in stromal cells (Figures 5A and 5B). All other corneas stained clearly with this antibody in both the epithelial and endothelial layers of the tissue (Figures 5C-5H). It was notable that keratoconus corneas displayed eNOS staining in stromal cells in areas adjacent to breaks in Bowman's layer (Figure 5G and bullous keratopathy in posterior stromal cells (Figure 5D. In contrast to eNOS, iNOS antibody staining was absent or minimal in the normal corneas (Figure 6A. All the diseased corneas had increased iNOS staining in the epithelium compared to normal corneas (Figures 6A-6C and 6E). In bullous keratopathy, cells throughout the cornea showed weak but detectable staining (Figures 6C and 6D), which was similar to the eNOS pattern. Fuchs' dystrophy was notable in that the cells near guttae (Figure 6F were stained for iNOS. Again, keratoconus corneas displayed the most intense staining of all the corneas (Figure 6B with all cell layers showing staining.
Summary of MDA, NT, eNOS, and iNOS immunoreactivity in diseased corneasa
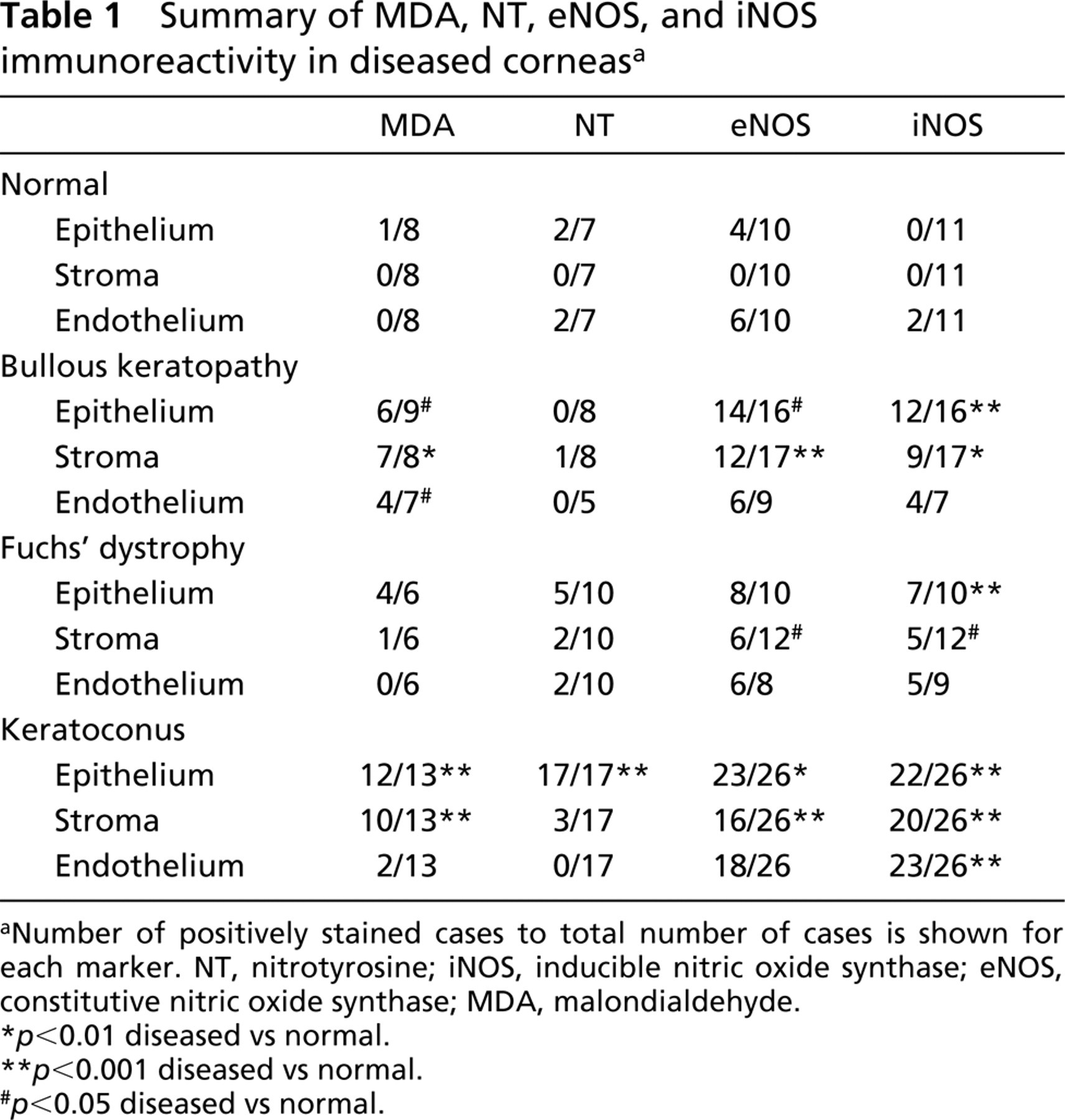
Number of positively stained cases to total number of cases is shown for each marker. NT, nitrotyrosine; iNOS, inducible nitric oxide synthase; eNOS, constitutive nitric oxide synthase; MDA, malondialdehyde.
p<0.01 diseased vs normal.
p<0.001 diseased vs normal.
p<0.05 diseased vs normal.
The staining patterns for iNOS and NT appeared to correlate with each other. To more carefully assess this relationship, serial sections of keratoconus cases were stained for these antigens and examined. As shown in Figure 7, areas staining well with NT (Figure 7A corresponded to areas enriched for iNOS staining (Figure 7B.
Discussion
Because the cornea absorbs approximately 80% of the incident ultraviolet B (UVB) light, there is a potential for generating significant amounts of free radicals and ROS. We undertook this study to look for evidence of oxidative damage in common human corneal disorders. Because there are multiple pathways of ROS/free radical cytotoxicity, we probed for the byproducts of two prominent pathways, MDA (lipid peroxidation) and NT (peroxynitrite formation).
Malondialdehyde (MDA)
Lipid peroxidation results from UV-induced oxidative destruction of cell membranes and the formation of cytotoxic aldehydes. These aldehydes can result in altered enzyme activities, inhibition of DNA/RNA/protein synthesis, and other damaging events (Esterbauer et al. 1991). This is the first study to document the presence of the toxic aldehyde byproduct MDA in human diseased corneas. In normal corneas, high levels of ALDH3 and SOD are present (Gondhowiardjo and van Haeringen 1993; Behndig et al. 1998; Pappa et al. 2001) and presumably function to absorb UV and scavenge both free oxygen radicals (SOD) and the generated aldehydes (ALDH3). Little evidence of MDA staining in normal corneas was obtained in this study. In contrast, all of the disease groups examined here showed significant and distinct patterns of MDA staining (Figure 2). In Fuchs' endothelial dystrophy, only the epithelial layer showed staining, whereas in keratoconus or bullous keratopathy corneas additional staining was observed. In bullous keratopathy, this staining appeared to be associated with areas of subepithelial fibrosis and, in another area of fibrotic change, the posterior collagenous layer. In keratoconus, stromal areas adjacent to regions where Bowman's layer was disrupted also stained for MDA. Because these regions are also associated with fibrosis (Kenney et al. 1998,2000,2001), MDA staining may represent an association with fibrosis in the cornea.
Nitrotyrosine
Several biochemical processes have been implicated in the pathogenesis of keratoconus (Kenney et al. 2000). In this study, keratoconus corneas consistently showed significant NT staining compared to normal corneas, which were routinely negative (Figure 3). The only other disease group that had NT staining was the Fuchs' dystrophy corneas. Bullous keratopathy corneas lacked NT staining, even if there was a significant amount of subepithelial fibrosis or a thick posterior collagenous layer (Figures 3C and 3D), indicating that fibrosis per se does not involve the accumulation of peroxynitrite byproducts. In contrast, Fuchs' endothelial dystrophy, which clinically has similar signs and symptoms as bullous keratopathy, does accumulate NT in the epithelium.
Nitric Oxide Synthase
The cornea contains all the biochemical pathways necessary for free radical generation as well as free radical quenching. Corneal cells are capable of expressing iso-forms of the NOS, suggesting that significant quantities of NO are produced physiologically in the cornea (Dighiero et al. 1997; McMenamin and Crewe 1997). In the cornea, eNOS activity has been demonstrated in the epithelium and endothelium (Yanagiya et al. 1997). The NO/cGMP pathway may be important in preventing corneal edema and maintaining normal corneal thickness (Yanagiya et al. 1997). iNOS can be found in corneal endothelial cells after induction by lipopolysaccharide (LPS) and cytokines (Dighiero et al. 1997).
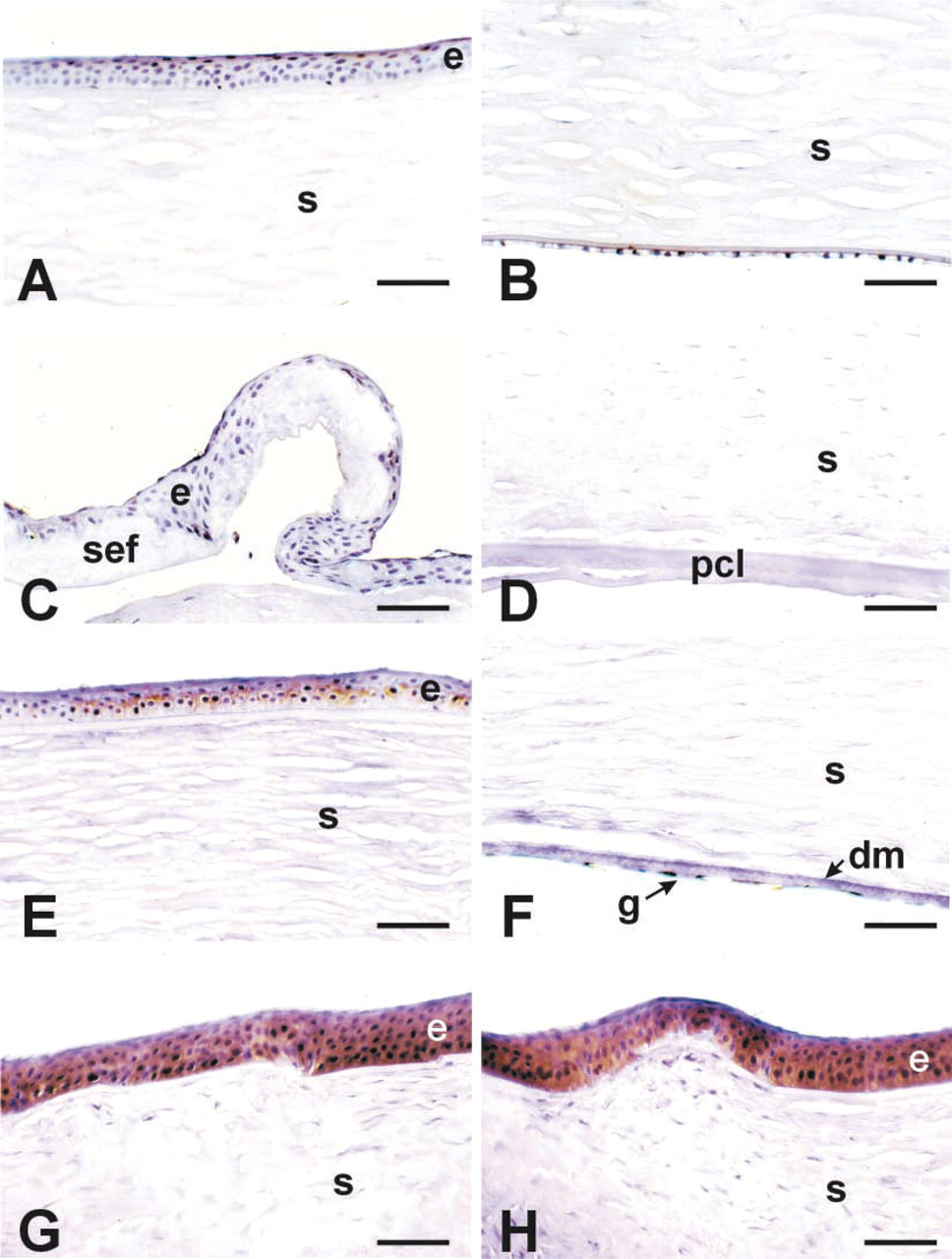
Nitrotyrosine (NT) immunoreactivity in the various disease groups. Normal (
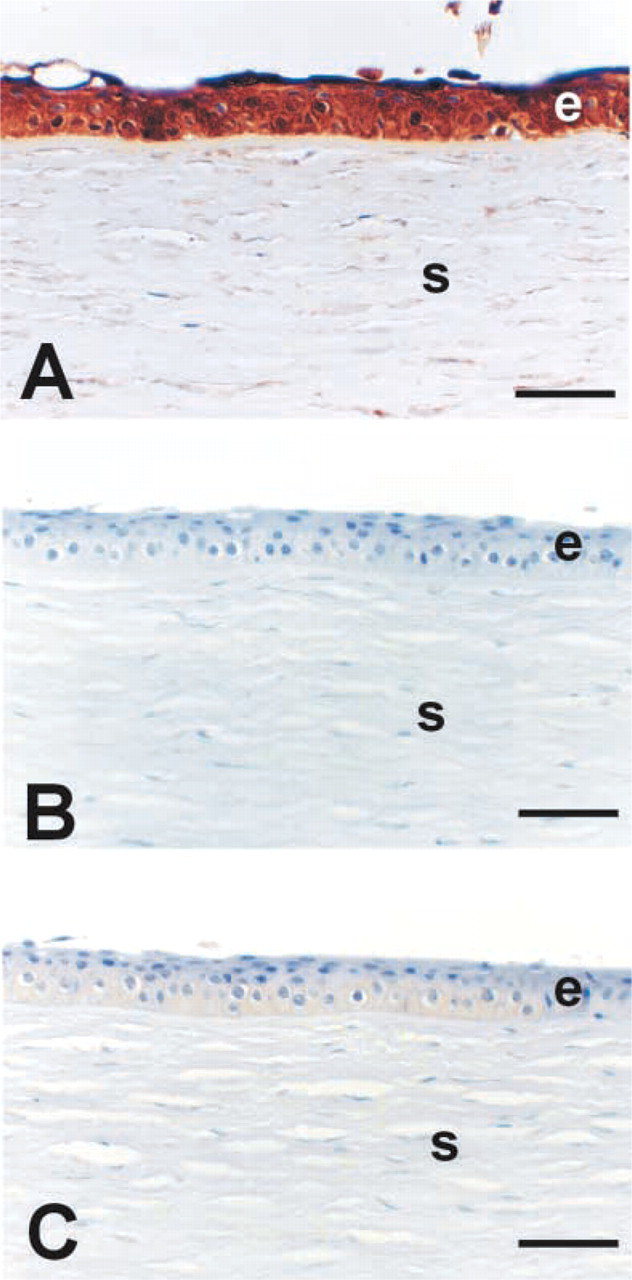
Specificity of nitrotyrosine staining. (
This study provides information about the distribution of eNOS in normal and diseased corneas. In agreement with others (Yanagiya et al. 1997), we found that, in normal corneas, eNOS was expressed only in the epithelium and endothelium (Figures 5A and 5B). Consistent with this, all three diseases showed continued eNOS expression in the epithelium and endothelium. iNOS staining was essentially absent in normal corneas (Figure 6A but was localized to the epithelial, stromal, and endothelial cells in all three disease groups.
Finding both eNOS and iNOS in the diseased corneas certainly suggests that higher amounts of NO are present within the tissue, but this may not translate into increased NT or oxidative stress byproducts. As mentioned previously, the majority of bullous keratopathy corneas had both eNOS and iNOS but lacked NT/peroxynitrite staining. Even Fuchs' dystrophy corneas consistently displayed these NOS isoenzymes, but only a few corneas showed NT staining. This suggests that, even with ongoing disease, human corneas can adequately process the free radicals/ROS so that the cytotoxic peroxynitrite byproduct was not formed. Keratoconus appeared to lack this capacity because all these corneas showed consistent NT staining that correlated with iNOS distribution (Figure 7).
The increased formation of nitrotyrosine (peroxynitrite) and MDA in keratoconus corneas (and, to a lesser extent, in Fuchs' dystrophy and bullous keratopathy corneas) suggests an important role of free radicals in their pathogenesis. Peroxynitrite-mediated tyrosine nitration as well as peroxynitrite itself can lead to altered biological functions (Estrada et al. 1997; Go et al. 1999; Jope et al. 2000; Kiroycheva et al. 2000). Structural proteins appear to be major targets for nitration by peroxynitrite with resultant disruption of their polymeric structure (Beckman 1996; Beckman and Koppenol 1996). In keratoconus, such a mechanism could, over a period of time, cause alterations of the stromal collagen molecules, perhaps making them more susceptible to digestion by activated enzymes and leading to stromal thinning. In addition to the effect on proteins, both NO and peroxynitrite are involved in multiple cytotoxic pathways. Both NO and peroxynitrite are toxic to DNA, leading to mutations and strand breaks (Szabo and Ohshima 1997). In this context, it is important to note that sustained exposure to low levels of NO or peroxynitrite can trigger apoptosis (Szabo and Ohshima 1997), and increased apoptotic activity has been reported in keratoconus corneas (Kim et al. 1999), possibly playing a role in its pathogenesis.
The relationship between MDA, NT, and NOS isoenzymes has been examined in a rat iron-overload renal system (Zhou et al. 2000). Upregulation of eNOS, iNOS, NT, and MDA was found in these animals. This is interesting because keratoconus corneas, which had increases of both MDA and NT, classically have distinct iron rings (Fleischer's ring) at the base of the cone. Iron accelerates lipid peroxidation of cell membranes (Zhou et al. 2000) and its presence may lead to oxidative damage. It should also be noted that the increased oxidative damage found in keratoconus corneas is probably not related to duration of exposure to UV light (age-related). In general, keratoconus is first diagnosed in mid-teen to early 20-year-old patients, while the bullous keratopathy patients are well into the fifth to sixth decades of life and have undergone cataract surgery before they have corneal problems.
eNOS immunoreactivity in the various disease groups. Normal (
More recently, alterations in the corneal enzymes ALDH3 and extracellular SOD, required to process and scavenge free radicals, have been reported in association with human corneal disease (Gondhowiardjo and van Haeringen 1993; Gondhowiardjo et al. 1993; Behndig et al. 2001). ALDH3 is known to participate in protecting the cornea against UV damage and aldehyde formation. If ALDH3, reported to be altered in keratoconus, were functionally impaired, one would expect increases in aldehyde damage and the accumulation of byproducts such as MDA. Although no changes in ALDH3 have been reported associated with bullous keratopathy and Fuchs' dystrophy, we predict that this may be the case because MDA is present in these diseases. In addition, both keratoconus and bullous keratopathy have reduced levels of SOD (Behndig et al. 2001), an oxygen radical scavenger. This could account for the accumulation of MDA in these tissues. Decreased SOD activity, coupled with increased NO production, could also account for the nitrotyrosine accumulation.
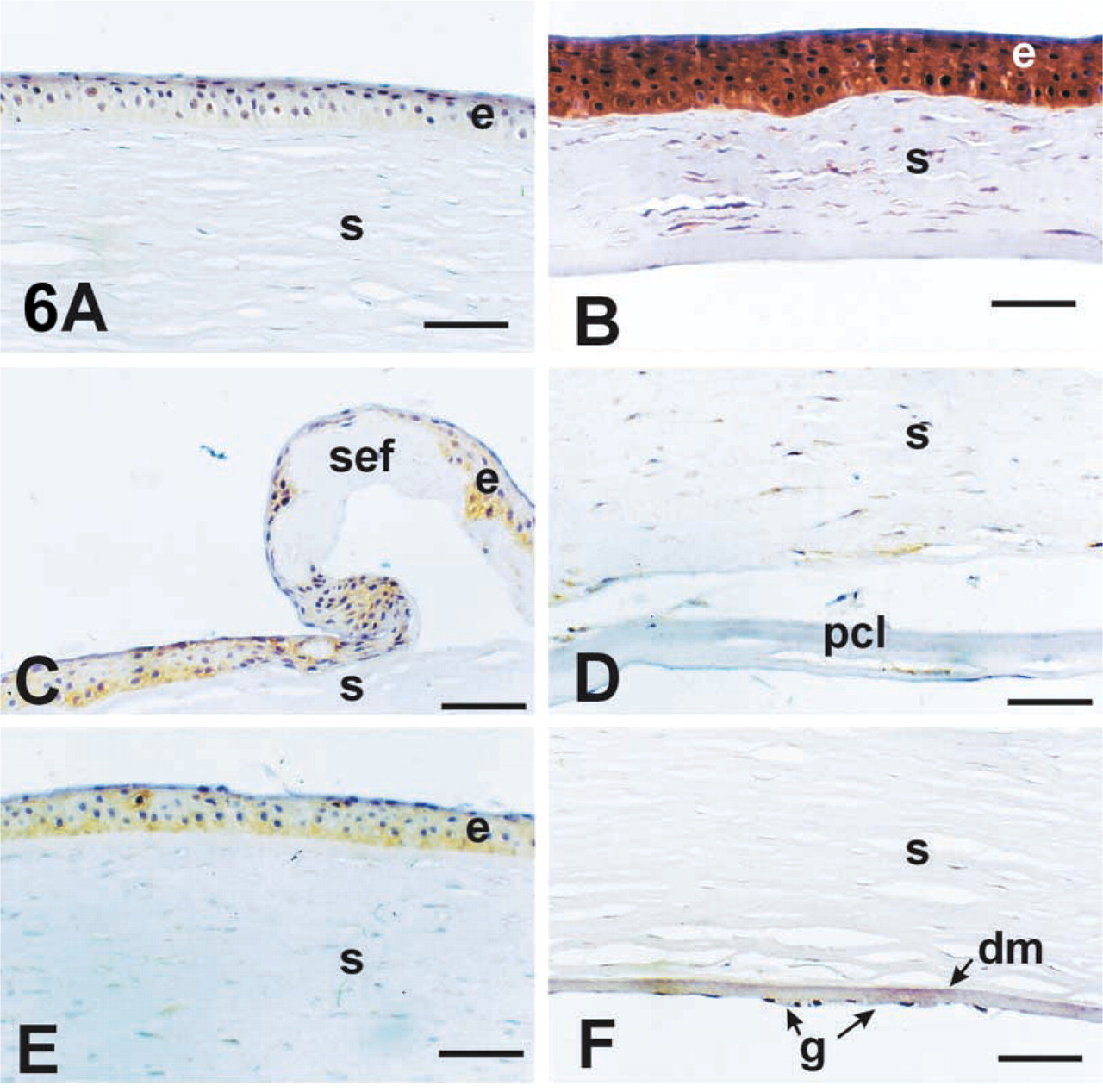
iNOS immunoreactivity in the various disease groups. Normal (
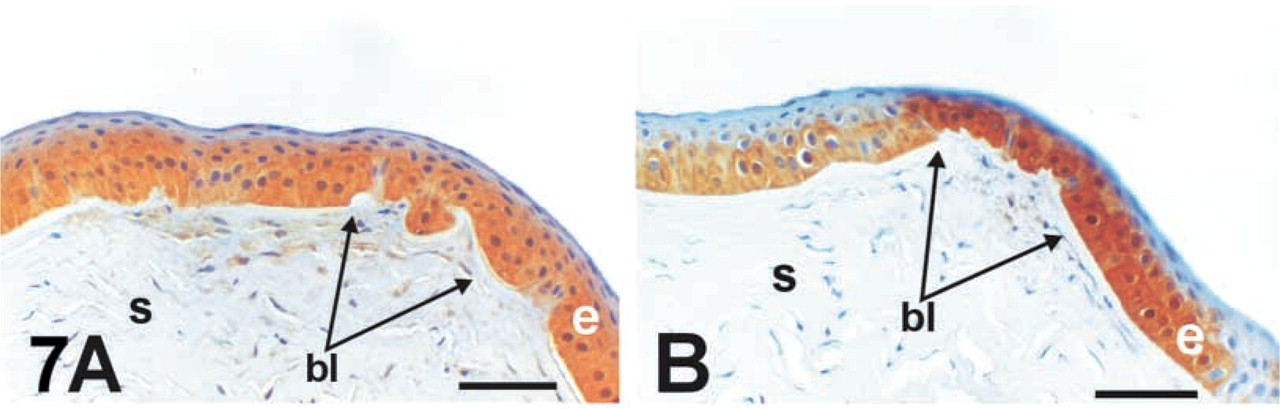
Distribution of NT and iNOS in serial sections. (
To conclude, we demonstrated unique patterns of NT/peroxynitrite and/or MDA distribution in keratoconus, bullous keratopathy, and Fuchs' endothelial dystrophy corneas. The excessive amounts of both NT and MDA in the keratoconus corneas support the hypothesis that oxidative damage plays an important role in this corneal disease. In addition, although bullous keratopathy patients and Fuchs' dystrophy patients often have similar clinical symptoms, their NT and MDA staining patterns are different from each other, with MDA correlating with fibrotic changes and NT correlating with iNOS distribution, suggesting that the pathophysiological pathways may not be identical.
Footnotes
Acknowledgements
Supported by NIH EY06807 and EY10836, the Schoellerman Charitable Foundation, the Discovery Fund for Eye Research, the Skirball Molecular Ophthalmology Program, and the National Keratoconus Foundation.
The authors wish to thank Dr. Narsing Rao (Doheny Eye Institue, Los Angeles) for his helpful comments and provision of tissues.
