Abstract
Reactive oxygen species (ROS), including nitric oxide (NO•), are associated with all steps of carcinogenesis. Hyaluronan (HA), a high-molecular-mass glycosaminoglycan over-expressed in a variety of human malignancies also has ROS-scavenging properties. We histochemically studied the level of HA in breast carcinoma cells and their stroma and compared it with the expression of NO• synthases (NOSs), major antioxidant enzymes, and nitrotyrosine. We also assessed whether the level of HA correlates with traditional prognostic factors of breast cancer and survival. Stromal HA level was moderate or high in all the samples studied (n=185), and 84% of the lesions showed HA-positive carcinoma cells. Intense stromal HA signal was associated with high neuronal NOS expression (p=0.009), whereas tumor-cell associated HA was inversely correlated with nitrotyrosine expression (p=0.027). Of the traditional prognostic factors, tumor cell-associated HA was correlated with poor differentiation (p=0.011), and high stromal HA levels were associated with aggressive features of the carcinomas such as large primary tumor (p=0.002), poor differentiation (p=0.019), and estrogen (p=0.012) and progesterone receptor negativity (p=0.009). High stromal HA level also significantly predicted poorer survival. The strong positive correlation between neuronal NOS and stromal HA could reflect NO•-stimulated synthesis of HA, an extracellular matrix alteration that favors breast cancer progression. Furthermore, it is suggested that, while acting as a scavenger of NO•-derived radicals, cell-associated HA undergoes partial fragmentation, release from receptors, and further degradation in lysosomes, and thus becomes undetectable in histological sections.
Keywords
H
As a consequence of aerobic respiration, reactive oxygen species (ROS) are continuously produced in all aerobic organisms. ROS, and especially their subgroup of free radicals, are highly reactive molecules or molecular fragments that play important roles in many physiological processes. However, if the amount of ROS exceeds the capacity of the ROS-suppressing machinery, oxidative stress is said to occur. This imbalance in the redox status is known to damage all cellular macromolecules, including DNA and HA (Wiseman and Halliwell 1996; Soltes et al. 2006). ROS are now considered to form a significant class of carcinogens, participating in cancer initiation, promotion, and progression (Klaunig et al. 1998).
Nitric oxide (NO•) is a short-lived free radical, generated from L-arginine by three NO• synthases (NOSs), encoded by three distinct genes. The majority of NO• is synthesized by inducible NOS (iNOS), usually after a challenge by immunological or inflammatory stimuli (Nathan and Xie 1994; Davis et al. 2001). Endothelial NOS (eNOS) and neuronal NOS (nNOS) are grouped together as constitutive NOSs (cNOSs), and their activity is mainly regulated by intracellular Ca2+ concentration. However, eNOS and nNOS also may be inducible under certain conditions (Nathan and Xie 1994; Davis et al. 2001).
Despite being a free radical, NO• infrequently interacts directly with macromolecules. However, when it reacts with another radical, namely superoxide (O2 •-), the reaction yields a powerful oxidant, peroxynitrite (ONOO-). ONOO- is one of the most reactive free radicals and is involved in numerous potentially carcinogenic reactions. It is known, for example, to induce transversion mutations and strand breaks in DNA, to trigger lipid peroxidation, to disturb the mitochondrial respiratory chain, and to influence protein phosphorylation by nitrating tyrosine residues of proteins such as p53 (Haddad et al. 1993; Huie and Padmaja 1993; Radi et al. 1994; Chazotte-Aubert et al. 2000; Cadenas 2004). Because free radicals are short-lived molecules and they cross-react readily with all cellular components, it is difficult and challenging to assess their concentration directly. Immunohistochemical visualization of oxidative damage “footprints,” such as nitrotyrosine, is usable in this respect and widely applied.
Antioxidant enzymes and related redox modulator proteins are the most important system to regulate redox homeostasis of tissues and to protect genome and other critical components against oxidative stress. Superoxide dismutases (SODs) act as a first line to catalyze the reaction of two superoxide anions and two protons to form hydrogen peroxide (H2O2), which still is a reactive molecule. In the light of knockout studies and other experimental studies, manganese SOD (MnSOD) is considered the most important SOD (Li et al. 1995a, b). H2O2 is further reduced to molecular oxygen and water by glutathione peroxidases, catalase, and peroxiredoxins. In addition to antioxidant enzymes, antioxidant defenses include glutathione and the antioxidative vitamins A, C, and E. Also, HA fulfills the criteria of an antioxidant, because free radicals, such as ONOO-, easily react with HA, resulting in the formation of a macroradical and, eventually, chain cleavage (Soltes et al. 2006).
Because HA may act as an antioxidant in cancer cells, apart from its invasion and metastasis-promoting actions, it is possible to hypothesize that this molecule could act in conjunction with other antioxidant systems in breast cancer cells and thus offer additional evidence of the role of HA expression in cancer promotion. The purpose of this study was to determine whether HA expression in stroma or in malignant cells correlates with NO• production, nitrotyrosine, or main anti-oxidant enzymes in breast carcinoma.
Materials and Methods
Study Materials
A total of 185 invasive breast cancer lesions, originating between 1982 and 2001, were collected from the files of the Department of Pathology, University of Oulu. The material used was a randomly selected subgroup of the blocks that have been used in our two previous studies assessing the role of Prxs, catalase, MnSOD, NOSs, and nitrotyrosine in breast carcinomas. These immunostainings were evaluated and reported previously (Karihtala et al. 2003, 2004). The material had been fixed in neutral formalin and further embedded in paraffin blocks. A multitissue microarray technique was applied in Prx, catalase, MnSOD, NOSs, and nitrotyrosine immunostainings. From each paraffin block, the pathologist chose one representative tumor region that was included in the multitissue microarray blocks. The blocks were created with Beecher Instruments Manual Tissue Arrayer (Beecher Instruments; Silver Spring, MD), with 1300-μm sample diameters. HA stainings were processed as individual slides and prepared explicitly for this study.
The histological diagnosis and grading of the 185 cases studied was assessed according to the criteria of the World Health Organization (Tavassoli and Devilee 2003). The material included 157 ductal, 21 lobular, 3 medullary, and 1 tubular carcinomas. Clinical data were obtained from the hospital records, and staging was based on the TNM classification of breast tumors (Elston and Ellis 1998). The mean follow-up time was 68.4 months.
Immunohistochemistry
Prx, catalase, MnSOD, NOSs, and nitrotyrosine immunohistochemistry was performed as follows. Paraffin-embedded tissues were sectioned to slides of 4-μm thickness and placed on SuperFrostPlus glass slides (Menzel-Gläser; Braunschweig, Germany). The slides were deparaffinised in xylene, rehydrated in descending ethanol series, incubated in 10 mM citrate buffer (pH 6.0), boiled in a microwave oven for 10 min, and cooled properly at room temperature before adding the primary antibody. Table 1 shows more details of the stainings for each antibody and HA. The cytochemical and immunostaining methods are also described in our previous works (Auvinen et al. 2000; Karihtala et al. 2003, 2004). Negative controls were prepared with the same procedure except that primary antibodies were replaced by PBS and serum isotype controls (Zymed Laboratories; South San Francisco, CA).
The expression of HA was studied using biotinylated HA binding complex (bHABC) prepared from bovine articular cartilage as described previously (Tammi et al. 1994). The sections were blocked for nonspecific binding with 1% BSA in PBS, followed by overnight incubation with bHABC (3 μg/ml), and treated with avidin-biotin-peroxidase (1:200; Vector Laboratory, Irvine, CA), 3,3′-diaminobenzidine (0.05%; Sigma, St. Louis, MO), and H2O2 (0.03%).
Details of antigens, antibodies, and histochemical methods used in the histochemical assessment
HA, hyaluronan; ER, estrogen receptor; PR, progesterone receptor; iNOS, inducible nitric oxide synthase; eNOS, endothelial nitric oxide synthase; nNOS, neuronal nitric oxide synthase.
The histochemical staining of HA in cells was evaluated as a percentage of all stained malignant cells in the samples. The percentage indicates the proportion of malignant cells expressing HA in the cytoplasm, cellular membranes, or nucleus. When comparing HA staining in carcinoma cells with other stainings, clinicopathological parameters, patient survival, and cell differentiation, these results were divided into two groups according to the median percentage of positively stained cells: 0 = positive staining in 0-39% of carcinoma cells and 1 = positive staining in 40-100% of carcinoma cells.
HA staining in the stroma was divided into three groups according to the amount of strong HA expression: − = no strong stromal HA staining present; + = less than one half of the stroma showing strong HA staining; and ++ = more than one half of the stroma showing strong HA staining. The staining results of Prxs I-VI were evaluated semiquantitatively by dividing the intensity of the staining in tumor cells into four different categories: − = no staining present; + = weak staining; + + = moderate staining; and + + + = strong staining. Tumor cell-associated expression of NOSs, nitrotyrosine, MnSOD, and catalase was subclassified into three categories: − = no immunostaining present, + = weak immunostaining; and ++ = strong immunostaining. Estrogen and progesterone receptor status was also based on immunohistochemistry (10% cut-off level for the hormone receptor-negative and -positive groups). From estrogen receptors, molecular form alpha was assessed.
Statistical Analysis
SPSS 13.0 for Windows (SPSS; Chicago, IL) was used for statistical analysis. The significance of associations was determined using the Fisher exact probability test (two-sided) and Mann-Whitney U-test. A Kaplan-Meier curve was used in survival analysis, and the significance was measured by the log-rank, Breslow, and Tarone-Ware tests. Probability values of p<0.05 were considered significant.
Results
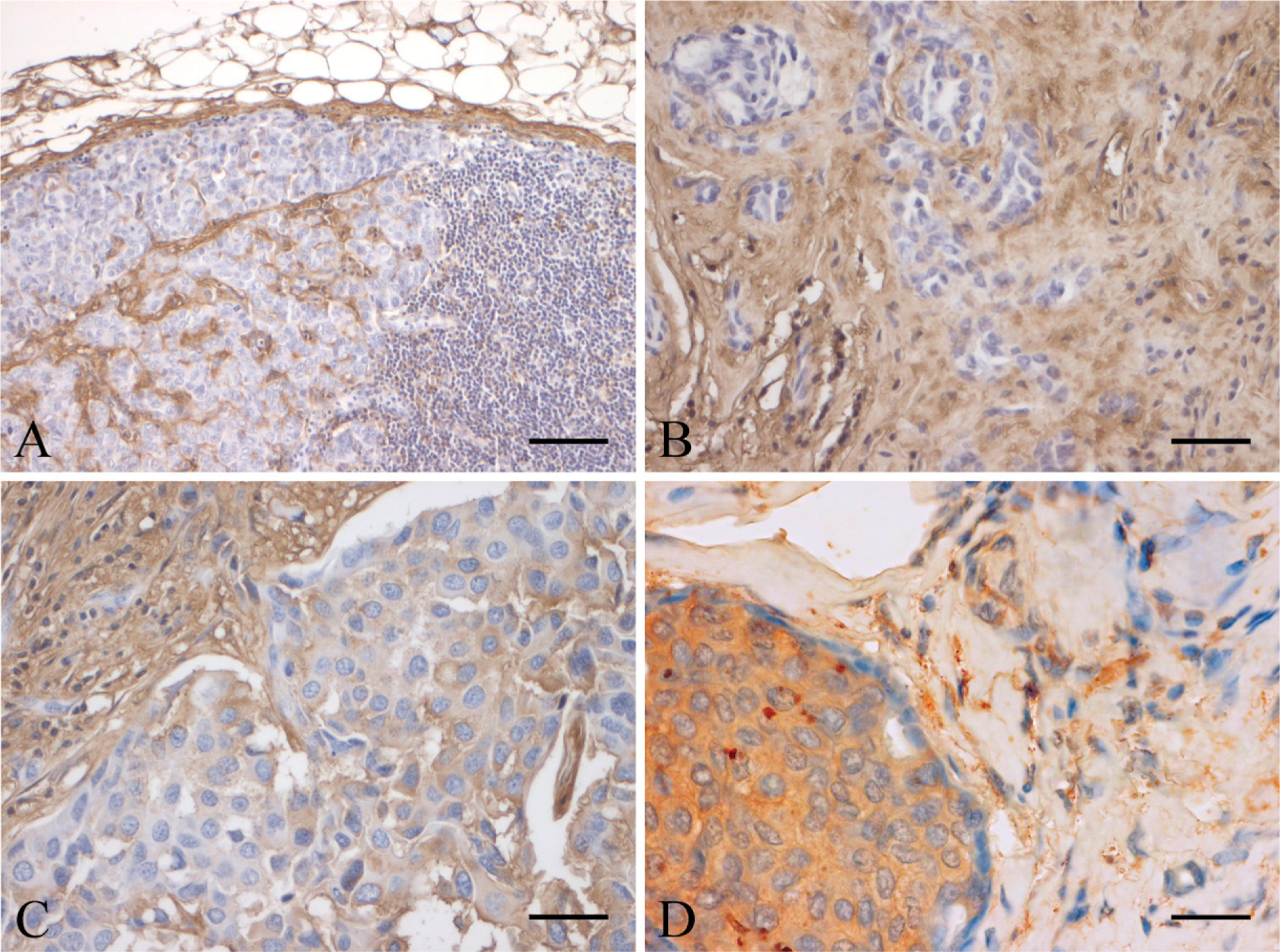
The staining of HA was assessed separately in stromal tissue and in the carcinoma cells. HA level was at least moderate in the stroma of all the lesions studied: 32 (17.3%) were rated moderately positive, and 153 (82.7%) showed strong staining (Figure 1). When examining cancer cell-associated HA, we found that 30 (16.2%) of the samples were completely negative and 155 (83.8%) were at least weakly positive. The median percentage for HA-positive carcinoma cells was 40%. There was a very strong association between HA in the stroma and HA in malignant cells (p=2.15 × 10-7). The detailed description of the immunostaining results of Prxs, MnSOD, catalase, NOSs, and nitrotyrosine are found in the references (Karihtala et al. 2003, 2004).
When we compared the results to traditional prognostic factors, high stromal HA staining was associated with large primary tumor size (p=0.002), estrogen receptor negativity (p=0.021), progesterone receptor negativity (p=0.01), and higher histological grade (p=0.019) (Table 2). High levels of HA in carcinoma cells were also correlated with an aggressive histological grade (p=0.011). No grade I tumors expressed HA in carcinoma cells. Thirty-six grade II tumors and 32 grade III tumors were negative for carcinoma cell-associated HA. The corresponding distribution for samples showing carcinoma cell-associated HA were 9 samples of grade I, 36 samples of grade II, and 32 samples of grade III tumors. Despite an obvious trend, the association between large tumor size and high carcinoma cell-associated HA staining was not statistically significant (p=0.087).
Intensive HA signal in the stroma was associated with high nNOS (p=0.009) expression in cancer cells. The expression levels of catalase, Prxs, or MnSOD did not correlate significantly with either stromal HA or HA in cancer cells. However, catalase had an obvious trend to higher expression in tumors with high stromal HA levels (p=0.069). There was a significant inverse correlation between nitrotyrosine and carcinoma cell-associated HA (p=0.027). In Kaplan-Meier analysis, stromal HA positivity predicted shorter survival according to the Breslow (p=0.041) and Tarone-Ware (p= 0.043) tests but not according to the log-rank test (p= 0.075; Figure 2).
(
Discussion
The tumor promotion by HA may be mediated in several ways. First, accumulation of HA creates highly hydrated, gel-filled spaces in extracellular matrices. The formation of this less dense matrix separates collagen layers apart and enhances tumor cell migration and penetration through physical barriers in the matrices (Knudson et al. 1984; Götte and Yip 2006). Second, a number of papers have reported that interaction of HA with its cell surface receptors CD44 and RHAMM influences cellular processes such as differentiation, growth, migration, and invasion, supporting carcinogenesis both in vitro and in vivo (Toole 2002; Fjeldstad and Kolset 2005). Furthermore, the HA coat on tumor cells may protect malignant cells against immunological response (Delmage et al. 1986), and HA fragmentation into oligosaccharides may enhance tumor angiogenesis (Rooney et al. 1995).
Four studies using clinical samples from invasive breast carcinomas have previously reported a link between elevated HA levels and poor differentiation, high occurrence of local lymph node metastases, and large primary tumor sizes (Auvinen et al. 1997, 2000; Wernicke et al. 2003; Suwiwat et al. 2004). In addition, we previously showed that HA expression in both the stroma and breast cancer cells are significant prognostic factors for poor patient survival (Auvinen et al. 2000). In contrast, no general correlation between tumor HA content and breast cancer prognosis was found when tumors were assayed with a radioimmunoassay-type method, and a slightly more favorable outcome was found with high HA in ductal and node-negative tumors (Corte et al. 2006). Unfortunately, these data are not comparable to the histochemical assays, because only soluble (“cytosolic”) HA, extractable with low ionic strength buffer, was included in the radioimmunoassay (Corte et al. 2006).
Association between stromal HA expression and primary tumor size, local lymph node metastase status, distant metastases, differentiation of tumor, ER status, and PR status
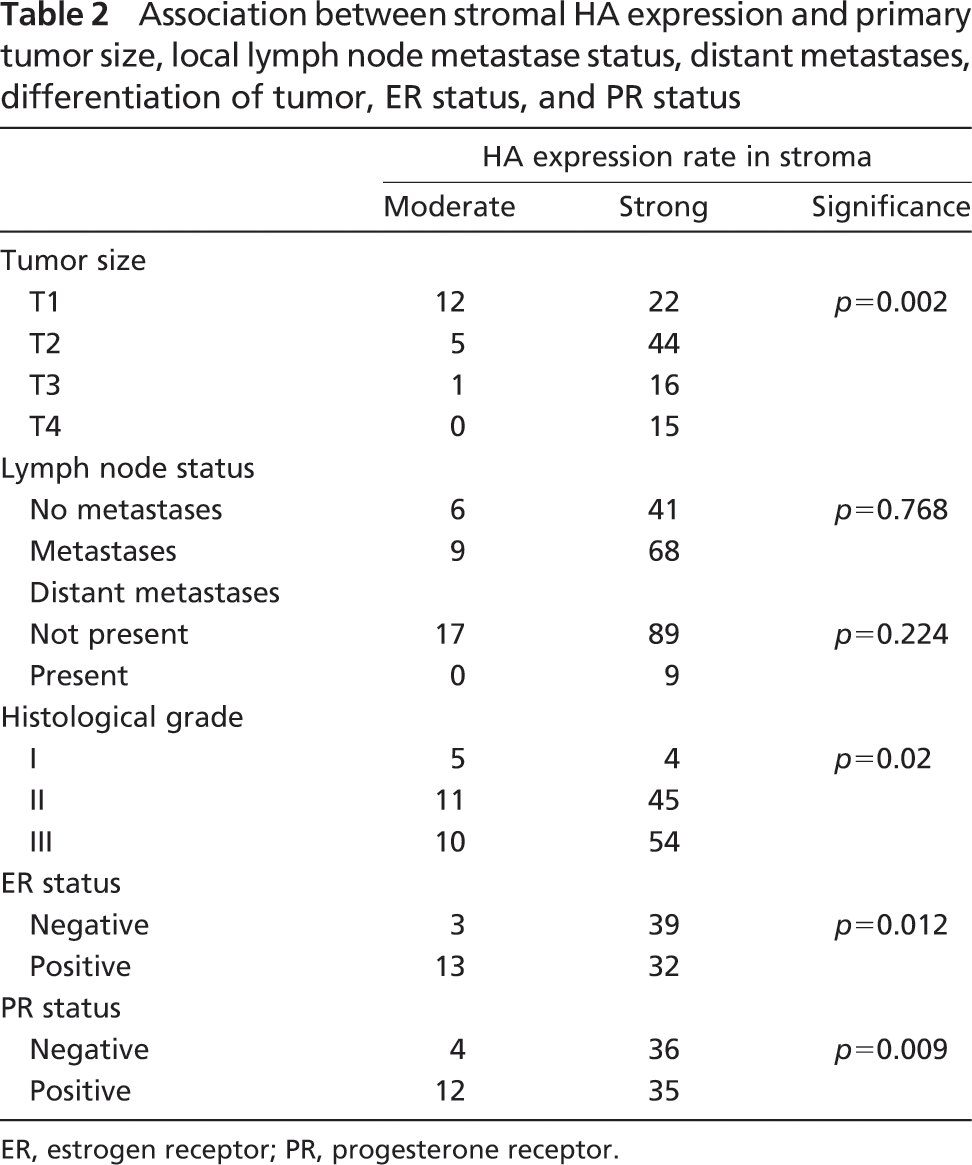
ER, estrogen receptor; PR, progesterone receptor.
In this study, HA expression showed moderate to high expression levels in the stroma, and cancer cell-associated HA was found in 81% of the lesions. Consistent with previous studies from different human carcinomas (Ropponen et al. 1998; Auvinen et al. 2000; Lipponen et al. 2001; Hiltunen et al. 2002; Paiva et al. 2005), both cell-associated and matrix-associated HA expression increased along with histological grade in this study. In this material, high stromal HA expression provided indirect evidence of growth advantage to the primary tumor but did not correlate with metastasis formation either locally or at distant sites. There was also a trend between tumor cell-associated HA and large tumor size, but this association did not reached statistical significance.
Estrogen and progesterone receptor expressions are powerful prognostic factors and form an authoritative criterion for the choice of treatment. In line with our previous study (Auvinen et al. 2000), high stromal HA levels were associated with the negativity of both hormone receptors. In vitro studies have previously shown that the receptor-negative breast cancer cell lines produce more HA than otherwise comparable estrogen receptor- and progesterone receptor-positive lines (Heldin et al. 1996). These findings, suggesting increased growth of breast cancer and risk of unfavorable disease outcome, are supported by a recent report showing that HA synthesis enhances breast carcinogenesis in vivo, with a significant contribution by stromal HA (Koyama et al. 2007). Although it seems that at least stromal HA expression creates a tumor-promoting environment in breast carcinomas (Auvinen et al. 1997, 2000; Toole 2002; Wernicke et al. 2003; Götte and Yip 2006), future clinical studies will confirm whether blocking HA functions have therapeutic importance in breast cancer, as suggested by the first reports on experimental animals (Yoshihara et al. 2005). In addition, Li et al. (2007) succeeded in their recent study to reduce tumor invasiveness and cancer cell migration by suppressing the expression of HA synthase 2 enzyme in an invasive breast cancer cell line with the use of RNA interference technique.
In the light of current literature, the main antioxidant function of HA is based on degradation of hydroxyl radical and ONOO-, which are considered the two most noxious free radicals (Campo et al. 2004; Hashimoto et al. 2006; Soltes et al. 2006). Because HA is widely used in the treatment of osteoarthritis, the majority of clinical studies have been performed in this patient group thus far. A large body of literature has shown that HA suppresses NO• levels in synovial fluid and menisci (Fukuda et al. 1997; Takahashi et al. 2001; Karatay et al. 2005; Kalaci et al. 2007). However, according to the authors' knowledge, no previous studies have addressed the question of antioxidant function of HA in malignant diseases or whether oxidative stress markers correlate with tissue HA levels. Nitrotyrosine is a reliable indicator of the formation of ONOO-, which is derived from NO• and O2 •-, both short-lived free radicals (Radi et al. 2001). We have previously shown that high nitrotyrosine expression is associated with large tumor size in invasive breast carcinomas and that nitrotyrosine formation significantly increases in breast lesions from benign hyperplasias to in situ carcinomas and to invasive breast carcinomas (Karihtala et al. 2004, 2006). Based on the results of this study, it is possible that cell-associated HA can detoxify free radicals in malignant cells in vivo, as suggested by in vitro studies (Soltes et al. 2006), although not sufficiently to depress carcinogenesis. In fact, persistent oxidative stress, even without obvious cytotoxic effects, can turn off tumor suppressor genes, activate transcription factors, and promote selective cell death and compensatory proliferation, all harmful effects considering carcinogenesis (Toyokuni et al. 1995; Klaunig et al. 1998). Because O2 •- is formed mainly as a by-product of mitochondrial respiratory chain and since NO• production is principally intracellular, it is rather logical that stromal HA is less involved in antioxidant activity. Neither stromal HA nor that associated to carcinoma cells correlated with the expression levels of “traditional” redox-state regulating enzymes such as peroxiredoxins, MnSOD, or catalase, suggesting that these systems have no shared regulatory mechanisms.
The inverse correlation between cancer cell-associated HA and nitrotyrosine may reflect the ability of free radicals to cleave HA chains. HA is largely retained in tissues by multivalent binding to cell surface receptors (Lesley et al. 2000), its ability to organize molecular complexes with proteoglycans (Ricciardelli et al. 2007), and crosslinking to proteins expressed at sites of inflammation (Day and de la Motte 2005). Even limited cleavage by free radicals could release HA chains from cell surface receptors and result in rapid diffusion into lymph or cellular uptake and lysosomal degradation. It has been shown that scavengers of oxygen free radicals slow down HA turnover in the epidermis (Ågren et al 1997), suggesting that cell-associated HA can be reduced by free radicals. Intermediate-sized HA fragments, previously suggested to be angiogenetic factors (Stern 2005), which are released from cell surface receptors to stroma after the cleavage of free radicals, may also accelerate carcinogenesis through the stimulated neovascularization, although further studies are needed to confirm this hypothesis.
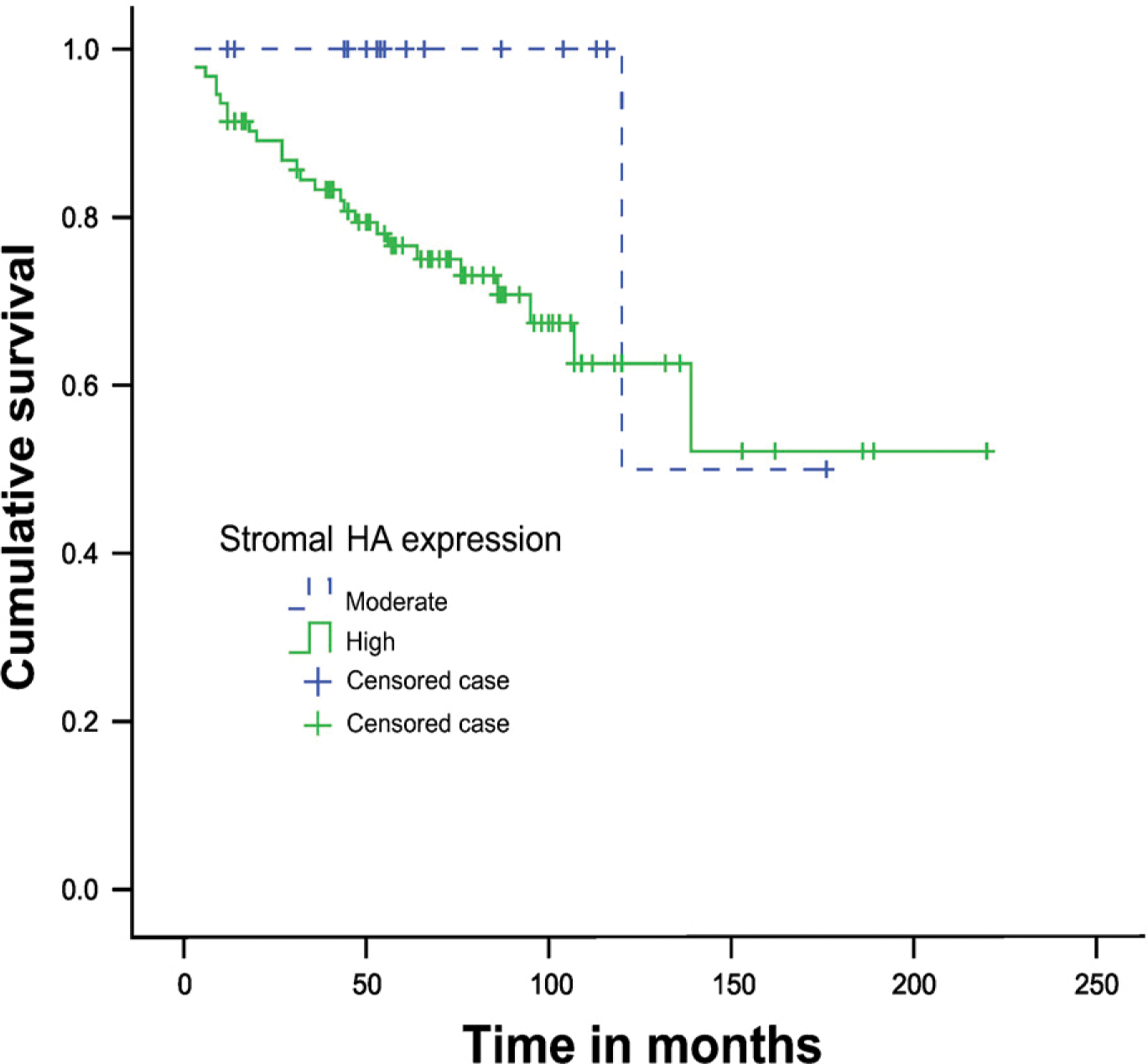
A Kaplan-Meier curve showing patient survival in relation to moderate and high stromal HA staining. Patients with moderate HA staining (dashed line) have a better prognosis. Crosses indicate the censored cases.
The positive correlation between stromal HA and nNOS expression favors the idea that NO• stimulates the synthesis of stromal HA in cancer, such as previously shown in articular cartilage (Hashimoto et al. 2006) and synovial cells (Chenevier-Gobeaux et al. 2004). Despite stimulated synthesis, the level of cell-associated HA may still remain low because of its release and partial degradation by the NO•-induced radicals that are most concentrated in and around the cells producing them, as discussed above. Therefore, enhanced synthesis and turnover of hyaluronan caused by NO• and its combined effect with ROS, respectively, is compatible with these findings.
It may seem paradoxical that HA, by protecting tumor cells from oxidative stress that is clearly procarcinogenic, still acts as a promoter of carcinogenesis. However, cell redox state-regulating proteins, including antioxidant enzymes, have a general tendency to be up-regulated in carcinomas (Gasdaska et al. 1994; Grogan et al. 2000; Kahlos et al. 2001; Soini et al. 2001; Lincoln et al. 2003; Kinnula and Crapo 2004; Lehtonen et al. 2004). It has been postulated that persistent oxidative stress is characteristic of carcinoma cells both in vivo and in vitro (Szatrowski and Nathan 1991; Toyokuni et al. 1995; Brown and Bicknell 2001). If redox balance is too oxidative, cells are driven to apoptosis or it may induce the variety of potentially carcinogenic alterations in cellular macromolecules, especially in DNA. However, oxidative stress in tumors in vivo is usually not sufficient to induce cytotoxic effects, because tumor cells have developed increased resistance to persistent oxidative stress (Toyokuni et al. 1995). High HA cellular concentration therefore may lead to the development of an oxidation state that is sufficient to promote carcinogenic mutations but still unable to lead apoptotic cell death.
In conclusion, the interplay between NO•, ROS, and HA seems important in the oxidative stress that initiates and promotes malignant growth. These results are in line with previous reports on the association between HA in tumor stroma and adverse prognosis and suggest a novel “buffering” role of cell-associated HA against excessive oxidative stress that could otherwise lead to apoptosis of the malignant cells. Furthermore, NO• may have a role in stimulating the HA synthesis that leads to its stromal accumulation, and at the same time, fragmenting the cell-associated HA into sizes that support tumor angiogenesis.
