Abstract
Studies of the vascularization of autochthonous rodent mammary tumors are limited in number, and the majority have used Factor VIII staining for blood vessel detection. Moreover, little effort has been directed at measuring the vascularization of tissue immediately adjacent to a tumor despite its central importance in the process of angiogenesis. Thirty-six chemically-induced mammary carcinomas and tissue immediately adjacent to these carcinomas were used to develop a census counting method for quantitative assessment of intra- and extra-tumor vascularization. Blood vessels were identified using antiserum directed against either CD31 or Factor VIII. Techniques used to create digitized images of all tumors and the semi-automated methods for circumscribing the extra-tumoral region are described. For Factor VIII, CD31 allowed greater discrimination of blood vessels with areas <25 μm2 and demonstrated crisp staining of blood vessels, with minimal background and excellent preservation of tissue architecture. Census counting data support the use of CD31 for quantifying both intra- and extra-tumoral vascularization. This method provides a basis for standardizing the approach to evaluation of experimentally induced premalignant and malignant mammary lesions in rodent model systems used to investigate potential anti-angiogenic cancer preventive agents.
IN VIVO, the supply of blood to newly forming tissues and to tumors is a limiting factor that regulates growth (Folkman 1971; Folkman et al. 1989) The process of neovascularization that provides blood to support such growth is referred to as angiogenesis (Folkman and D'Amore 1996). Equally important to homeostatic regulation of tissue size is the maintenance of vascularization once the blood supply has been established. For the mammary gland, there have been a number of reports both on angiogenesis during normal development (ductal extension, lobuloalveolar development, pregnancy, lactation, and involution) (Pepper et al. 2000; Djonov et al. 2001) and tumorigenesis (Heffelfinger et al. 2000) and on the characterization of the vascular networks that exist during these same processes (Kaidoh et al. 1991; Djonov et al. 2001). However, a critical review of these studies revealed a paucity of quantitative data about the spectrum of blood vessel sizes and their frequency of occurrence during mammary gland development or tumorigenesis. Most information that does exist is either qualitative, describing the nature of the vascular beds, or focuses on the occurrence of microvessels. Moreover, the majority of work in the mammary gland using immunohistochemical (IHC) approaches to detect blood vessels has been done using Factor VIII (Ottinetti and Sapino 1988; Fregene et al. 1993; Guidi et al. 1994; Guinebretiere et al. 1994; Heffelfinger et al. 1996, 2000), whereas, work in other model systems indicates that CD31 is the most sensitive and specific endothelial marker for vascular endothelium and is the recommended epitope for these types of studies (Vermeulen et al. 1996).
The work reported here was initiated (a) to develop a method suitable for profiling the vascularization of mammary tumors and the extra-tumoral region of the mammary gland immediately adjacent to a tumor and (b) to determine empirically whether CD31 would yield better staining than Factor VIII in rodent mammary tissue and tumors. No attempt was made to constrain method development based on concerns about direct clinical applicability, particularly as it relates to the amount of time required for the assay to be performed, because the method was intended for use in the research setting. A semi-automated process is described for use in defining a 50-μm perimeter around tumor areas and for utilizing image analysis systems to quantitatively evaluate vascularization of rat mammary carcinomas.
Materials and Methods
Chemicals and Reagents
The carcinogen MNU was obtained from Ash Stevens (Detroit, MI). Pepsin (Fisher Scientific; Pittsburgh, PA) enzyme digestion was performed on sections for the detection of Factor VIII. Donkey serum (Jackson ImmunoResearch; West Grove, PA) was used as blocking serum to prevent nonspecific binding of primary antibodies. Rabbit anti-Von Wille-brand Factor (Factor VIII) antibody was obtained from Dako (Carpinteria, CA) and goat anti-CD31 (PECAM1) antibody from Research Diagnostics (Flanders, NJ). Biotinylated secondary antibodies, donkey anti-rabbit F(ab')2 (Jackson ImmunoResearch) and donkey anti-goat IgG (Jackson ImmunoResearch) were applied for Factor VIII and CD31, respectively. Horseradish peroxidase-conjugated streptavidin (Dako) and Stable DAB (Research Genetics; Huntsville, AL) were used in the visualization of blood vessels.
Animal Protocol and Necropsy Procedures
Twenty-day-old female Sprague-Dawley rats were obtained from Taconic Farms (Germantown, NY). Animals were housed three per cage in polycarbonate solid-bottomed cages in an environmentally controlled room maintained at 22C and 50% relative humidity with a 12 hr light/dark cycle. All rats were maintained on a purified diet, AIN 93G, formulated as described by our laboratory (Zhu et al. 1997), and were provided free access to deionized water. Rats were injected
Specimen Selection
All lesions evaluated were mammary gland carcinomas of cribriform, papillary, comedo, or mixed histological types. Serial paraffin sections of each formalin-fixed carcinoma were cut at 4 μm and mounted on 3-aminopropyltriethoxysilane (APES)-treated slides. Sections were heat-immobilized in an 80C oven for 20 min, deparaffinized in three changes of xylene, hydrated in a series of graded ethanols, and rinsed in several changes of deionized water (DW). Carmine stain was removed by immersing sections in 0.1% lithium carbonate for 5 min, followed by several changes of DW.
Pretreatment
Enzymatic digestion was performed on sections designated for Factor VIII only. Sections were incubated using a modified version of the pepsin digestion protocol established by Hayashi et al. (1988): 0.4% pepsin in 0.01 N HCl for 15 min at 37C. Heat-induced epitope retrieval (HIER) was performed on sections designated for CD31 staining only. Slides were double spaced in a Tissue-Tek (Sakura Finetek; Torrance, CA) 24-slide rack and immersed in a polypropylene Tissue-Tek staining dish filled with 200 ml of 10 mM sodium citrate buffer, pH 6.0. The dish containing the buffer and slides was placed in an 1100-W microwave oven and microwaved for 2 min on high power. Fluid level was checked and an additional 50 ml of DW added if necessary. Sections were microwaved for an additional 18 min at 20% power, followed by a cooling period of 30 min at room temperature (RT) before immunostaining. Sections were rinsed in several changes of DW.
Immunohistochemistry
Endogenous peroxidase activity was quenched by immersing sections in 3% H2O2 for 5 min. Sections were rinsed in several changes of DW followed by PBS, pH 7.4, three times for 5 min. All subsequent steps were performed in a humidity chamber at RT. Donkey serum, 1:5, was applied to all sections for 20 min. Excess serum was drained and primary antibody, rabbit anti-Factor VIII (1:800) or goat anti-CD31 (1:75), was applied to positive sections and incubated for 30 min or 1 hr, respectively. Negative control sections were held in PBS during the primary incubation. Sections were rinsed in PBS three times for 5 min. Biotinylated secondary antibodies, donkey anti-rabbit F(ab')2 (1:1000) or biotinylated donkey anti-goat IgG (1:500) were applied to all sections and incubated for 30 min. Sections were rinsed in PBS three times for 5 min. The enzyme label, horseradish peroxidase-conjugated streptavidin (1:1000) was applied to all sections and incubated for 30 min. Sections were rinsed in PBS three times for 5 min. Stable DAB was applied to all sections and incubated for 6 min. Sections were rinsed in several changes of DW and counterstained as previously described (McGinley et al. 2000). Sections were rinsed in DW, dehydrated in a series of graded ethanols, cleared in xylene, and mounted using a synthetic resin.
Image Acquisition
Images of immunostained sections were captured with a Kodak DCS-420 digital camera (Eastman Kodak; Rochester, NY) mounted on a Zeiss Axioskop microscope (Carl Zeiss; Thornwood, NY) using a ×10 objective. The CCD imaging sensor on the DCS-420 has a focal magnification of ×2.5, yielding images with an approximate total magnification of ×250. The resolution of each image was 1012 × 1524 or 1.5 megapixels. Images were acquired using a 32-bit Kodak TWAIN driver v5.02 within Adobe Photoshop v4.0 graphic software (Adobe Systems; San Jose, CA) running on a 300 MHZ Pentium III PC with 128 MB of RAM. Tumors that exceeded the size of a single imaging area were captured by photographing contiguous microscopic fields in a raster pattern. Each captured image was merged using the layer technique in Adobe Photoshop to form a single composite image for analysis.
Spatial Calibration
CD31 and Factor VIII IHC stains were evaluated using Image Pro Plus (IPP) v4.1 (Media Cybernetics; Silver Spring, MD). Spatial calibration was determined by photographing a 1-mm stage micrometer using the same parameters as those for image capture of immunostains. The distance drawn on the 1-mm calibration image was divided by 1000 to find the number of pixels per micrometers, which equaled 1.12 pixels.
Area Measurement
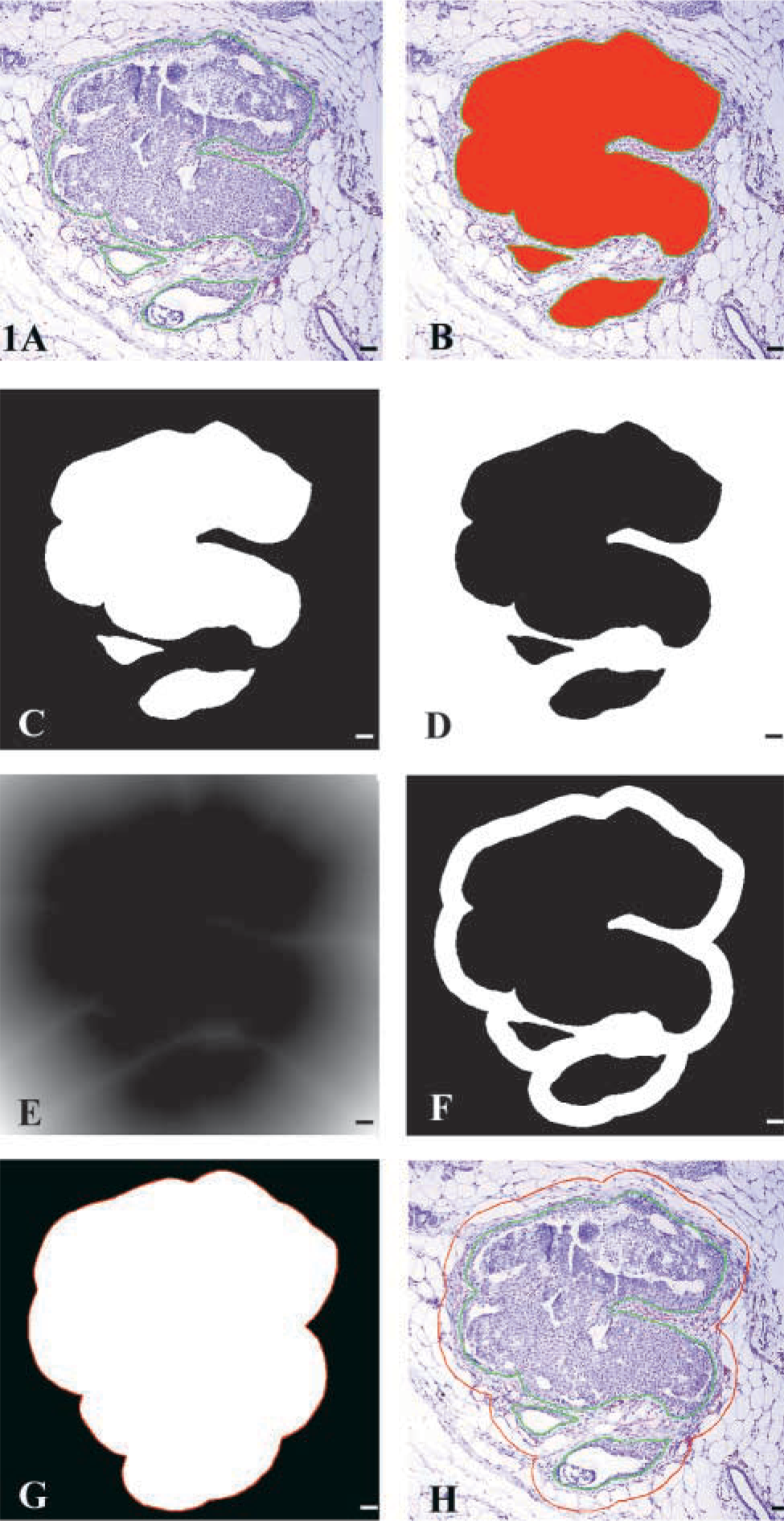
Epithelial areas within each tumor were circumscribed using the irregular area of interest (AOI) tool in IPP (Figure 1A) in combination with an Intuos digitizing pen and tablet (Wacom Technology; Vancouver, WA). A macro was then used to construct a 50-μm boundary using the AOIs. The macro loaded the spatial calibration and the intensities for all three-color channels, red, green, and blue, were set to maximal range (0–255). The areas of the AOIs were measured using the count feature under the count/size menu (Figure 1B). The sum area of the AOIs in μm2 was written to an Excel spreadsheet as intratumoral area (IA) via dynamic data exchange (DDE). An image mask was created, resulting in a binarized image of white AOIs with pixel values of 255 on a black background with pixel values of zero (Figure 1C). The mask was inverted, resulting in black AOIs on a white background (Figure 1D). A Euclidian distance map image filter was applied to the inverted mask (Figure 1E). The value of each pixel outside each AOI is the shortest distance from that pixel to the edge of the AOI. All pixels along the AOI border have a pixel value of 1 because they are 1 pixel away from the AOI. Pixels that are 2 pixels from the border have a value of 2, and so on. Using the number of pixels per micrometer measurement from the spatial calibration, it was determined that a 50-μm distance would be equal to 56 pixels. Therefore, the intensity range on the distance map image was set to 0 and 56 (Figure 1F). A mask was created, resulting in white AOIs plus a white 50-μm border around each AOI on a black background (Figure 1G). The white area was counted using the “automatic bright objects” feature under the count/size menu in IPP. The sum of the white area in μm2 was written to an Excel spreadsheet as total area (TA) via DDE. An outline of the white area was saved to a file and then superimposed on the original image using the “load outlines” feature (Figure 1H). Extra-tumoral area (EA) was calculated by subtracting IA from TA.
Blood Vessel Identification
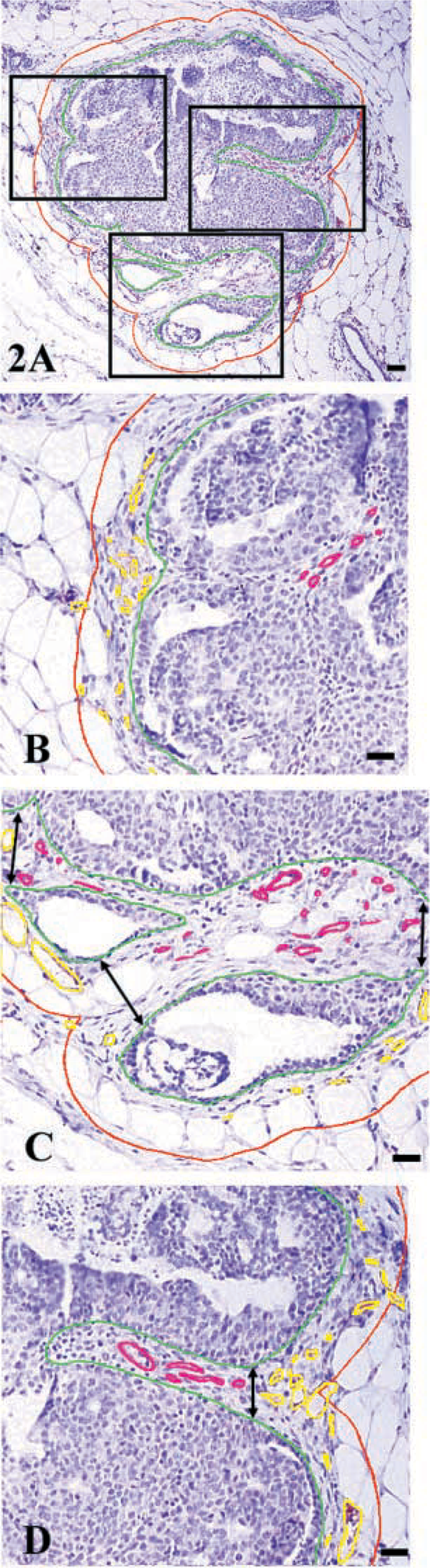
Tumor architecture presented a challenge in determination of extra-tumoral and intra-tumoral vessels (Figure 2A). All vessels were circumscribed manually, using the digitizing pen, to rule out inclusion of artifact or background immunostaining. Criteria established by Weidner et al. (1991) were used to identify blood vessels in immunostained sections. Specifically, positively stained endothelial cells or endothelial cell clusters, regardless of size or shape, that were clearly separate from adjacent blood vessels, tumor cells, or other connective tissue elements were counted. Vessel lumens, although usually present, were not necessary for a structure to be defined as a blood vessel, and red blood cells were not used to define a vessel lumen.
(
(
Extra-tumoral vessels were measured first for all tumors. Extra-tumoral vessels (yellow) were defined as vessels existing outside the IA and lying within or in direct contact with the 50-μm border (red) surrounding the tumor (Figures 2B-2D). Intratumoral vessels (magenta) were defined as vessels within the epithelial portions of the tumor (Figure 2) or areas within opposing AOI boundaries (green) (Figure 2). Measurements for all vessels were recorded in μm2 and exported to an Excel spreadsheet via DDE. All data were exported to Systat v9.01 (SPSS; Chicago, IL) for statistical analysis. The specific statistical tests used are described in the table and figure legends.
Results
Qualitative Assessment
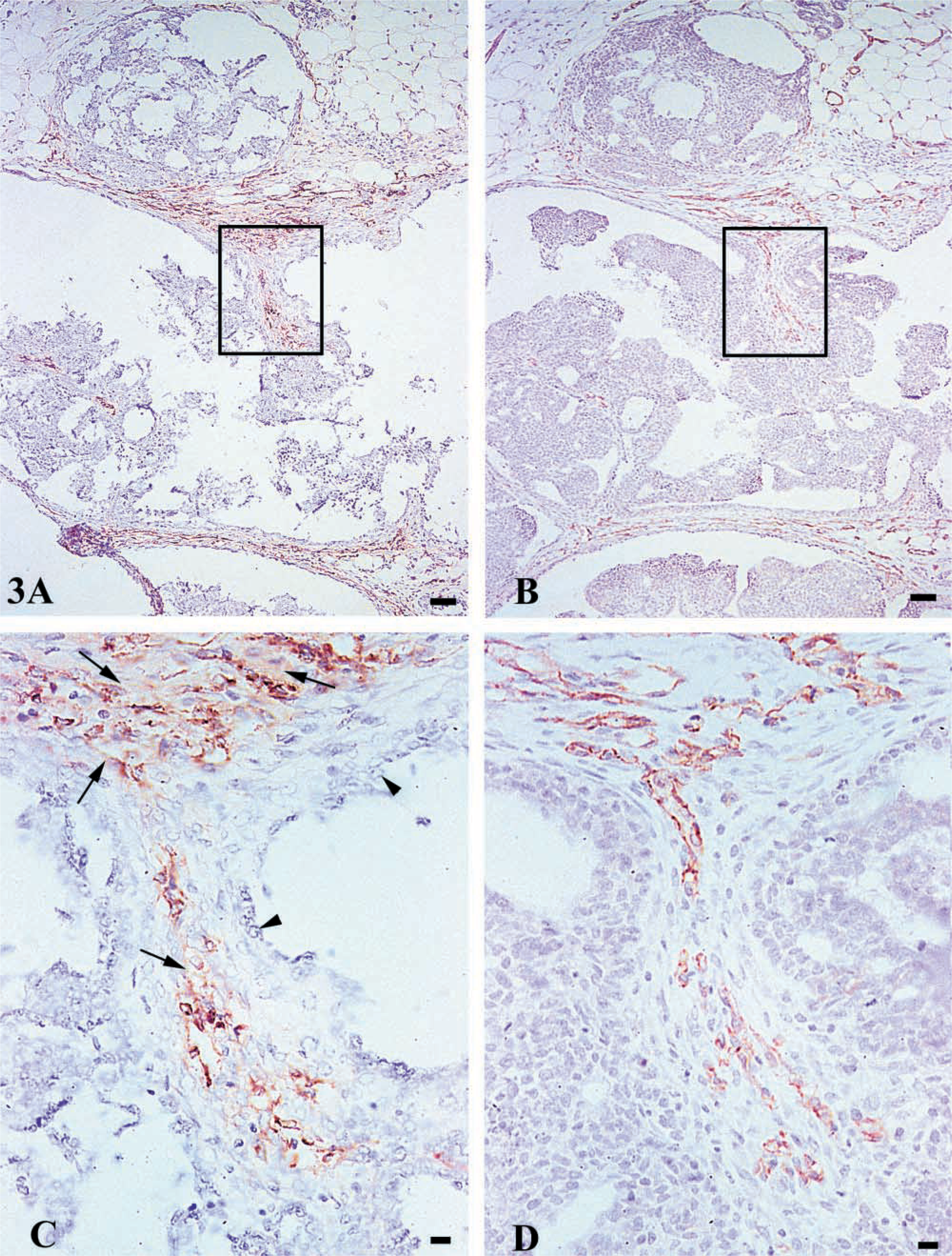
Blood vessels were detected immunohistochemically from serial sections of 36 mammary carcinomas with the use of CD31 and Factor VIII antibodies. Images were captured according the procedures outlined in Materials and Methods. As shown in Figure 2, IHC staining protocols provided specific staining of blood vessels with limited background staining. However, background staining was somewhat higher in Factor VIII-stained sections. The procedure used for Factor VIII involved digestion of the tissue with pepsin, which caused tissue detachment and altered morphology, particularly noticeable intra-tumorally (Figure 3A). Conversely, the CD31 protocol used HIER for epitope retrieval, resulting in crisp vessel staining with only minor tissue detachment while maintaining morphological integrity (Figure 3B).
Vascular Area
All carcinomas were census-counted, i.e., the entire four micron section of the carcinoma was evaluated for intra-tumoral blood vessels. In addition, a 50-μm-wide band that circumscribed all elements of each carcinoma (referred to as extra-tumoral blood vessels, shown in Figure 2) was assessed. Area measurement for each blood vessel was recorded in addition to location, i.e., intra- or extra-tumoral. Table 1 shows the total area (tumor area plus 50-μm boundary around tumor) and the total vascular area that were assessed using each detection antiserum. The distribution of these areas by location of the blood vessel (intra- or extra-tumoral) is also presented. Overall, essentially identical total areas were assessed for vessels using CD31 or Factor VIII (areas differed by only 0.6%). Similarly, the total vascular area identified using CD31 or Factor VIII differed by only 4.4%. However, striking differences were noted when the distribution of vascular areas was categorized according to vessel location. Overall, vascular area constituted approximately 2.0% of the total area assessed. However, this indicator of the degree of vascularity was markedly different by region. The percent of total area that was composed of blood vessels was approximately 6.0% in the extra-tumoral region, whereas it was only 0.7% intra-tumorally. While similar total intra- and extra-tumoral areas were assessed for blood vessels detected by either CD31 or Factor VIII (differing only by 6.3 and 3.2%, respectively), the total vascular area identified by CD31 and Factor VIII differed by region. Total extra-tumoral vascular area was 13.9% higher with CD31 than with Factor VIII (6.2% and 5.18%, respectively), whereas total intra-tumoral vascular area was 45.9% higher with Factor VIII vs CD31 (0.48% and 0.91% for CD31 and Factor VIII, respectively). One likely reason for this difference is illustrated in Figure 3. As noted above, either detection antiserum resulted in specific vessel staining with limited background staining. However, as shown in Figures 3C-3D, background staining was higher with Factor VIII than with CD31. This background staining, in combination with the distortion of tissue morphology observed in Factor VIII-stained sections, made vessel detection and image analysis more error prone with Factor VIII, whereas with CD31 the stain was crisp, with well-defined outlines of blood vessels. Absence of background staining facilitated the identification of individual small blood vessels in the CD31-stained section (Figure 3D), whereas the small vessels were more difficult to distinguish in the Factor VIII-stained serial sections (Figure 3C). This resulted in an underestimation of the number of small blood vessels that were present because they were recorded as a single larger vessel. It is noteworthy that this problem was primarily encountered with the intra-tumoral blood vessels and is probably due to the nature of tissue processing required for retrieval of tissue and tumors from carmine-stained mammary gland whole-mount preparations. Therefore, this particular observation may be specific to the processing used in evaluating tissue from mammary gland whole mounts in this model system.
Vascular Evaluation
A total of 12,138 blood vessels were detected in the analysis of CD31-stained carcinomas; a similar number, 11,732 was detected in the Factor VIII-stained sections (Table 2). The smallest blood vessel detected by either stain was 2.4 μm2. The largest vessel detected using CD31 was 0.08 mm2, whereas the largest vessel detected using Factor VIII was 0.12 mm2. Overall, approximately twice as many vessels were detected in the extra-tumoral vs intra-tumoral region, irrespective of the type of antiserum used to detect the vessels.
These findings led to the analysis of average vessel size. Both the mean and the median average vessel area was higher with Factor VIII than with CD31 (
Range of Vessel Sizes
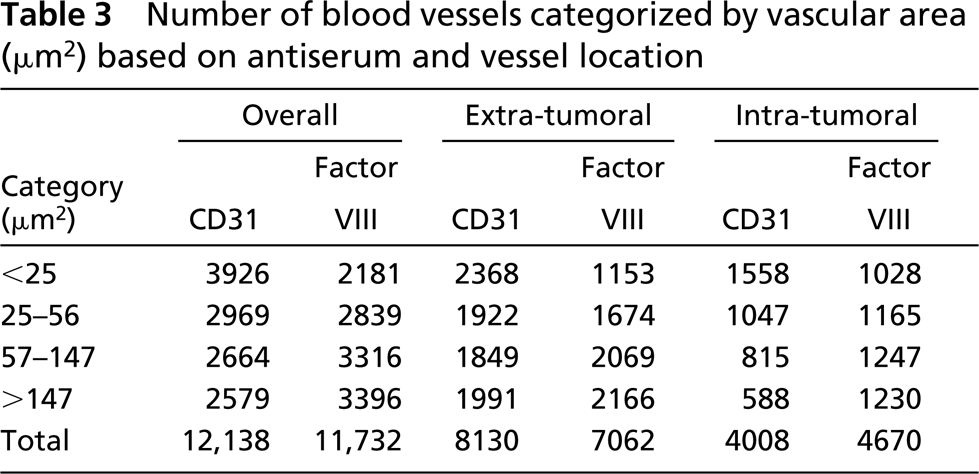
To pursue the observation that CD31 appeared to detect more smaller blood vessels, a stem-leaf plot analysis was performed on all data, irrespective of vessel location or detection system. This provided information on the distribution of vessel area by quartiles. Twenty-five percent of all vessels detected were <25μm2. Interestingly, the diameter associated with this area is 5.6 μm, slightly larger than the average diameter of a formalin-fixed, paraffin-embedded red blood cell in rats, which was determined to be 5.5 μm by image analysis (data not shown). The next 25% of vessels had an area that was ≥25 and <56 ≤μm2. The third quartile of vessel sizes was >56 and ≤147 μm2. The fourth quartile included vessels with an area >147 μm2. The numbers of blood vessels falling into each of these size ranges, as categorized by location and detection antiserum, are shown in Table 3. CD31 detected approximately twice as many vessels that were <25 μm2 as were detected by Factor VIII. More of these small vessels were detected by CD31 in the extra- vs the intra-tumoral region. The numbers of vessels detected using either CD31 or Factor VIII and whose area ranged from ≥25 and ≤56 μm2 were similar. For the remaining vessel area categories, more vessels were detected by Factor VIII than by CD31.
Discussion
The goal of the work reported here was to develop a method suitable for quantifying the vascularization of carcinomas arising in the mammary gland and that would permit the investigator to retain knowledge of the anatomic location of the tumor and its proximity to other mammary gland pathologies and/or anatomic structures within the gland. For this reason, mammary carcinomas were induced using a model system from which mammary gland whole mounts can be prepared and the areas of interest visualized and dissected (Thompson et al. 1995). Thirty-six mammary carcinomas of various histological types were studied. These tumors were dissected with a sufficient margin so that the vascularization of the mammary tissue immediately adjacent to the carcinomas could also be evaluated. Because essentially no quantitative data are available on the vascularization of the rodent mammary gland that could be used to guide our analysis strategy, it was decided to census-count all blood vessels that were identified by immunostaining. Moreover, although the majority of work in experimental models for breast cancer in which immunostaining was used to detect blood vessels has used Factor VIII, the literature indicates that CD31 would provide more consistent detection of small blood vessels. Therefore, antisera directed against both epitopes were studied. To our knowledge, the data presented provide the first comprehensive quantitative assessment of the number of blood vessels, categorized by size, occurring in a large number of chemically induced mammary carcinomas in the rat while providing specific details on an easily implemented, semi-automated method by which these data were collected. The details of our findings are discussed below.
Total area (TA) and total vascular area (VA) measured in [xm2, categorized by antisera and region
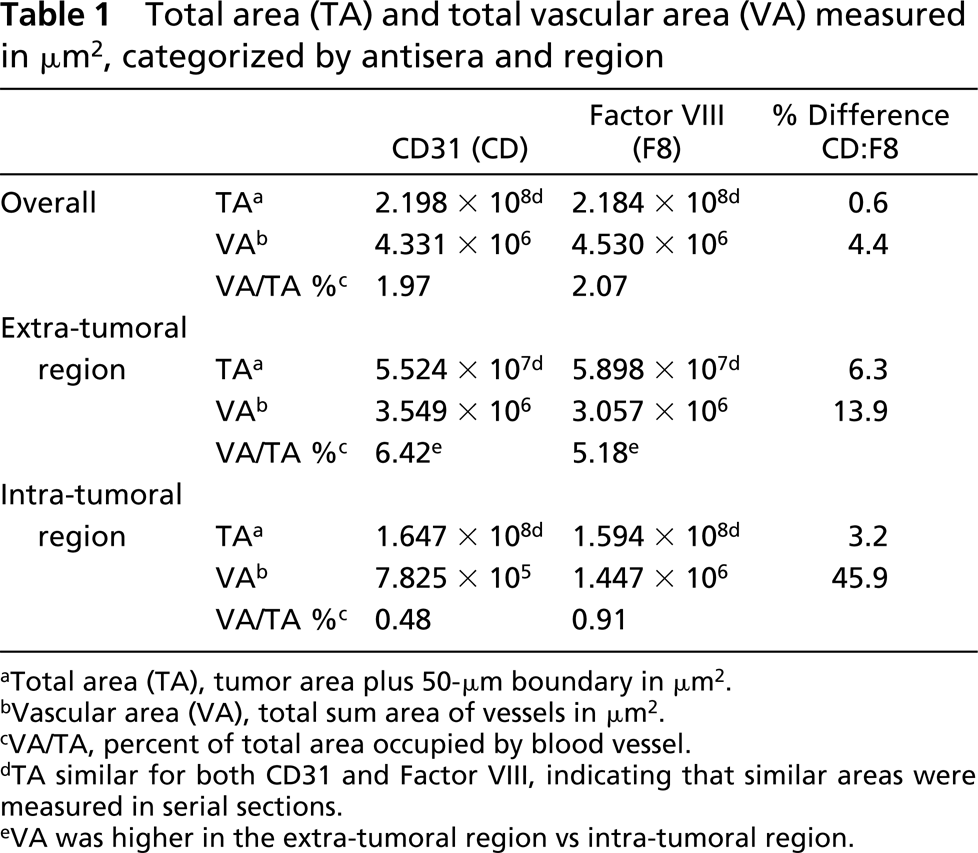
Total area (TA), tumor area plus 50-μm boundary in μm2.
Vascular area (VA), total sum area of vessels in μm2.
VA/TA, percent of total area occupied by blood vessel.
TA similar for both CD31 and Factor VIII, indicating that similar areas were measured in serial sections.
VA was higher in the extra-tumoral region vs intra-tumoral region.
Number of blood vessels and average vascular area based on antiserum and vessel location
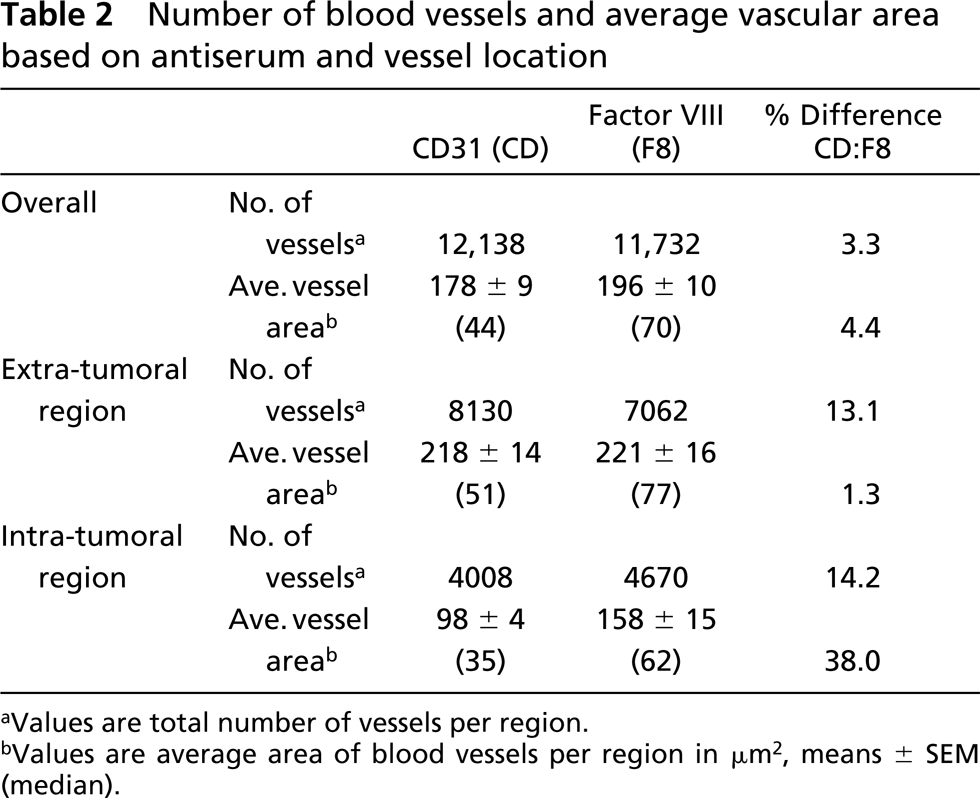
Values are total number of vessels per region.
Values are average area of blood vessels per region in μm2, means ± SEM (median).
At the outset of this study, a review of the literature indicated that antiserum directed against Factor VIII was selected in most work in which immunostaining was being used to detect blood vessels in mammary tissue (Ottinetti and Sapino 1988; Fregene et al. 1993; Guidi et al. 1994; Guinebretiere et al. 1994; Heffelfinger et al. 1996, 2000). Because the goal of methods development was to quantify all blood vessels, which we now know spanned a range from a cross-sectional area of 2.5 μm2 to 0.12 mm2, it was decided to compare Factor VIII staining with that obtained using antiserum directed against the CD31 epitope, which has been reported to be more consistently expressed on developing capillaries than is Factor VIII (reviewed in Vermeulen et al. 1996). As shown in Table 1, similar total areas were assessed for vascularity, and both antisera resulted in the detection of similar vascular areas. However, consistent with the work cited above, CD31 was significantly more effective in detecting blood vessels with a diameter <25 μm2 (Table 3; Figure 3D). For vessels ≤25 μm2, no consistent statistically significant advantage was attributable to the use of either antiserum. We attribute the tendency for Factor VIII to detect larger vessels to a misclassification error due to a tissue processing artifact that occurred with Factor VIII staining. Epitope retrieval for Factor VIII staining included the treatment of tissue sections with pepsin. This process caused areas of a tissue section, particularly tumor epithelial cells, to detach from the slide, resulting in loss of the cells from the section and a decrease in the architectural integrity of the section (Figures 3A-3D). This problem was not encountered using the HIER method with CD31. Although this artifact of sample processing is likely to be limited to tissue excised from carmine-stained mammary gland whole mounts, it is noteworthy because it appeared to decrease the ability to detect small blood vessels (<25 μm2) within tumors and resulted in an increase in the vascular area ascribed to larger blood vessels. Given the greater sensitivity to detect vessels of all sizes in the absence of processing artifacts, the use of CD31 immunostaining is recommended for blood vessel quantification in rat mammary tissue.
(
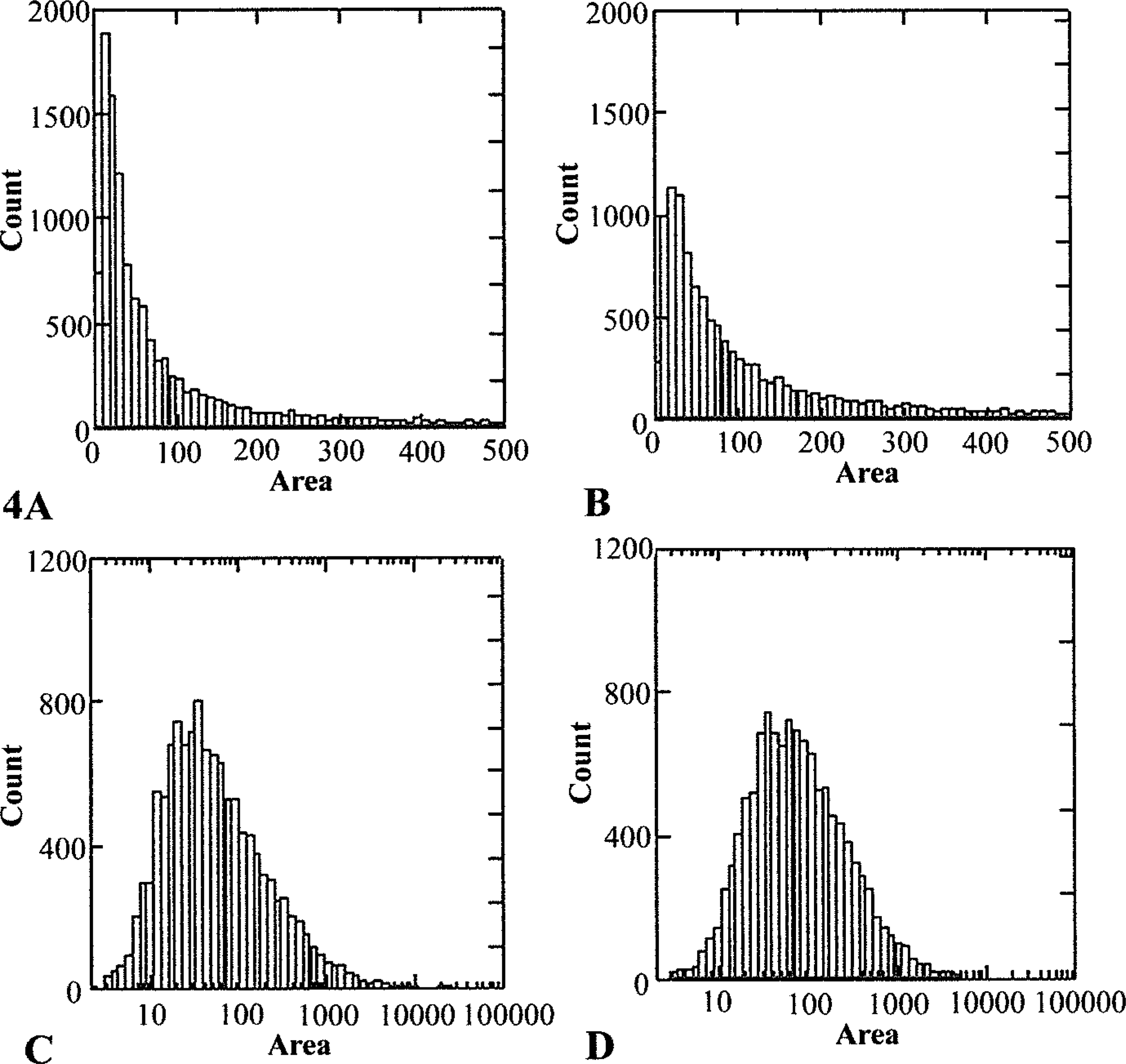
Histograms of total count of vessels by area (μm2) by detection of CD31 (
Census-counting of blood vessels was used in this work and this approach enabled us to determine the frequency with which blood vessels of different sizes occurred. The results, shown in Figure 4, are consistent with circulatory physiology, in that the number of blood vessels of a particular size decreased as the size of the vessels increased. This fact is not likely to be fully appreciated by many scientists studying quantitative aspects of vascularity. However, it has a number of important implications. First, irrespective of the range of blood vessel sizes selected for investigation, the distribution of the vessels counted will be asymmetric, with an extended right-handed tail. Such data violate one of the assumptions underlying most parametric statistical procedures, i.e., that the data are normally distributed. As shown in Figures 4C and 4D, logarithmic transformation of the vessel area data normalized the distribution, making it suitable for parametric statistical analyses such as ANOVA. Log transformation of vessel area data is recommended instead of the use of non-parametric rank tests, which do not fully take into account the magnitude of differences that may exist between two different populations of blood vessels. This issue applies not only to census-counted data sets, such as the one reported in this study, but also to data sets in which vessel density data are obtained by the hot-spot, Chalkley, or random-field methods of data acquisition (Vermeulen et al. 1996). A second implication of the results presented in Figure 3 is applicable when a decision is made not to perform census-counting of blood vessels. If an investigator decides to use a random-field approach to estimate the values that would be determined by census-counting, the number of fields that need to be sampled will increase as the size of the vessels to be evaluated increases. The number of random fields selected will depend on the magnitude of the differences among treatment groups that an investigator wishes to detect. A bootstrapping technique can be used to determine the optimal number of random fields to assess.
Number of blood vessels categorized by vascular area (μm2) based on antiserum and vessel location
A final observation emerging from this work relates to the assessment of vascularity in the 50-μm band circumscribing the tumor (extra-tumoral). In previous studies of premalignant breast disease, various approaches to the evaluation of blood vessels in the stroma surrounding a lesion have been reported (reviewed in Heffelfinger et al. 1996). Of the factors that differed among these studies, most notable was the distance from a lesion that has been assessed (range 100–300 μm). In the present study we decided to limit the area evaluated to a 50-μm band circumscribing each tumor element, for the following reasons: (a) vessels providing blood supply to a tumor will lie within this circumference; (b) new blood vessels being induced by angiogenic factors released by tumor cells, which the stroma has been reported to nullify (Ottinetti and Sapino 1988), will occur within this area; and (c) it represents a manageable area for census-counting. As shown in Table 3, a large number of blood vessels of all sizes are observed in the extra-tumoral region. This implies that angiogenic activity is high not only within a tumor but also in the stroma surrounding a tumor. Therefore, for investigators interested in studying changes in vascularization during premalignant breast disease and/or the effects on mammary carcinogenesis of chemopreventive agents that may act by blocking angiogenesis, the census-counting approach as applied to both the extra-tumoral and intra-tumoral regions and described here, is recommended. It is noteworthy that, during methods development, consistent identification of the extra-tumoral band around a carcinoma was found to be very subjective and error prone when the task was performed manually. Consequently, the semi-automated procedure described in Materials and Methods was developed and provides objective criteria for identification of the extra-tumoral region. This approach reduced technical effort and eliminated observer bias in area selection, and can be easily applied to studying the vascularization of premalignant and malignant pathologies in the rodent mammary gland.
In conclusion, maximal information can be derived concerning the vascularization of mammary tissue and tumors within the mammary gland if specimens are harvested from mammary gland whole-mount preparations. CD31 immunostaining was found to be superior to Factor VIII in detecting vessels of all sizes because of minimal background staining and the elimination of processing artifacts observed in Factor VIII-immunostained preparations. Census-counting of blood vessels provides the greatest amount of information and revealed an asymmetric distribution of blood vessel sizes that must be taken into account both in the statistical analyses of data and in selection of histological fields for counting if census-counting is not performed. In characterizing the vascularization of pre-malignant mammary lesions, consideration should be given to evaluating the extra-tumoral pattern of vessel occurrence by census-counting using the semi-automated methods described.
Footnotes
Acknowledgements
Supported by PHS grant CA 52626 from the National Cancer Institute.
