Abstract
Members of the cysteine-rich protein (CRP) family are evolutionarily conserved
Keywords
M
Studies in both chick and rat have revealed that CRP2 (also referred to as SmLIM) is present at highest levels in arterial samples (Jain et al. 1996; Louis et al. 1997), and subsequent work has focused on a potential role for CRP2 in the vascular system (Jain et al. 1998). Here we describe the pattern of csrp2 expression in the developing and adult mouse. We demonstrate that csrp2 is broadly expressed in mouse embryos and adults.
Plaque lifts of a mouse embryonic (E) 11.5 day 5′ stretch λgt11 cDNA library (Clontech; Palo Alto, CA) were performed exactly as described (Henderson et al. 1999) using the coding region of the chicken csrp2 cDNA (Weiskirchen et al. 1995) as a probe. A screen of 500,00 pfu provided one positive phage (IC8) that was purified. The cDNA insert of IC8 was excised using EcoRI, cloned into pBluescript (ks−) (Stratagene; La Jolla, CA), and sequenced.
Northern analyses of a mouse embryo multiple tissue northern (MTN) blot and a mouse MTN blot (Clontech) were performed exactly as described previously (Henderson et al. 1999), using the csrp2 3′ UTR (nucleotides 705–850 in Figure 1A) as a probe. According to the manufacturer's instructions (Clontech), MTN lanes contain approximately 1 μg of RNA, and a control experiment using gadph as a probe verifiedthat all lanes of the mouse embryo MTN blot contained RNA.
(
To perform in situ hybridization (ISH) analyses, the csrp2 3′ UTR was cloned into pBluescript (ks−) (Stratagene) and verified by sequencing. Generation of anti-sense radiolabeled riboprobes and ISH of staged E9.5-E16.5 mouse embryos and of adult mouse organs, was performed as described (Henderson et al. 1999). Control experiments using sense riboprobes on whole-mount embryos did not produce a specific signal (data not shown). In situ hybridization of formalin-fixed, paraffin-embedded E7.0 embryos was performed using digoxigenin-labeled probe according to the manufacturer's protocol, followed by incubation with a mouse anti-digoxigenin antibody (Jackson ImmunoResearch; West Grove, PA) conjugated to alkaline phosphatase detected with BCIP/NBT (DAKO; Carpinteria, CA).
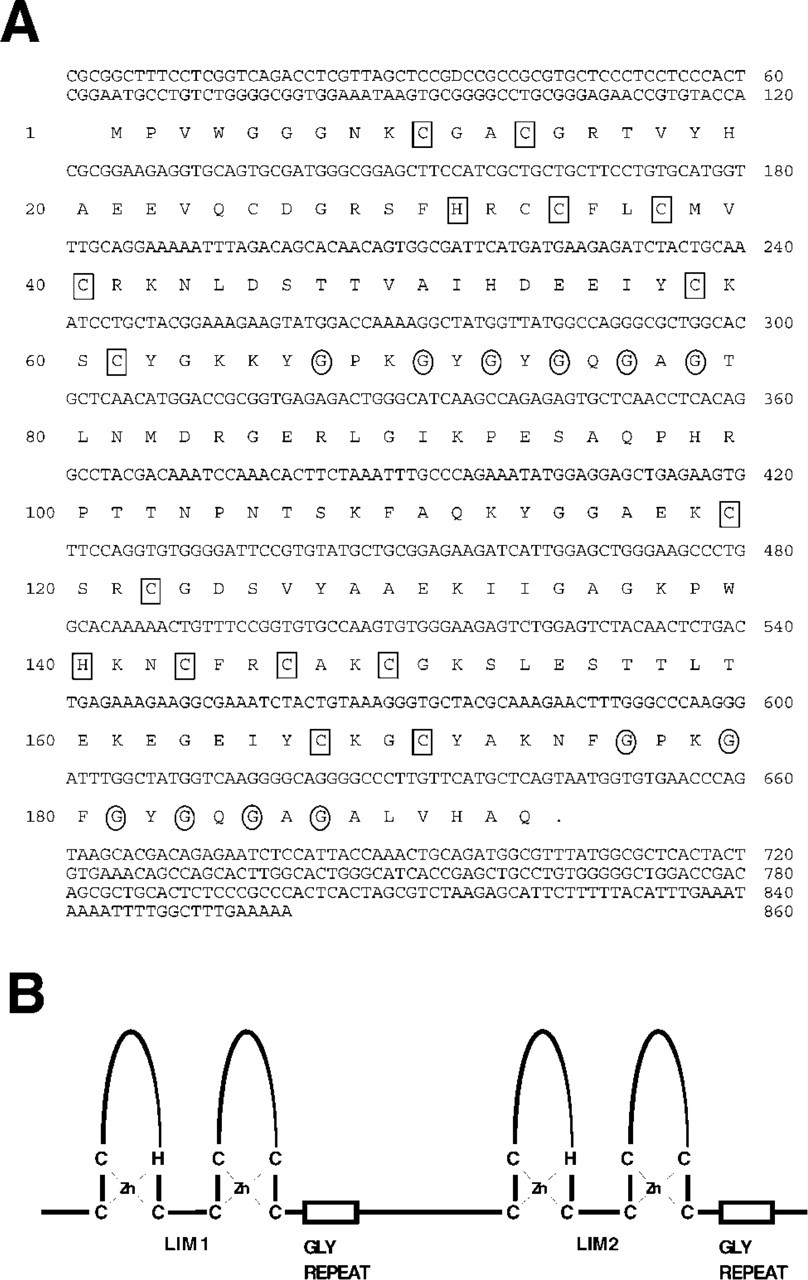
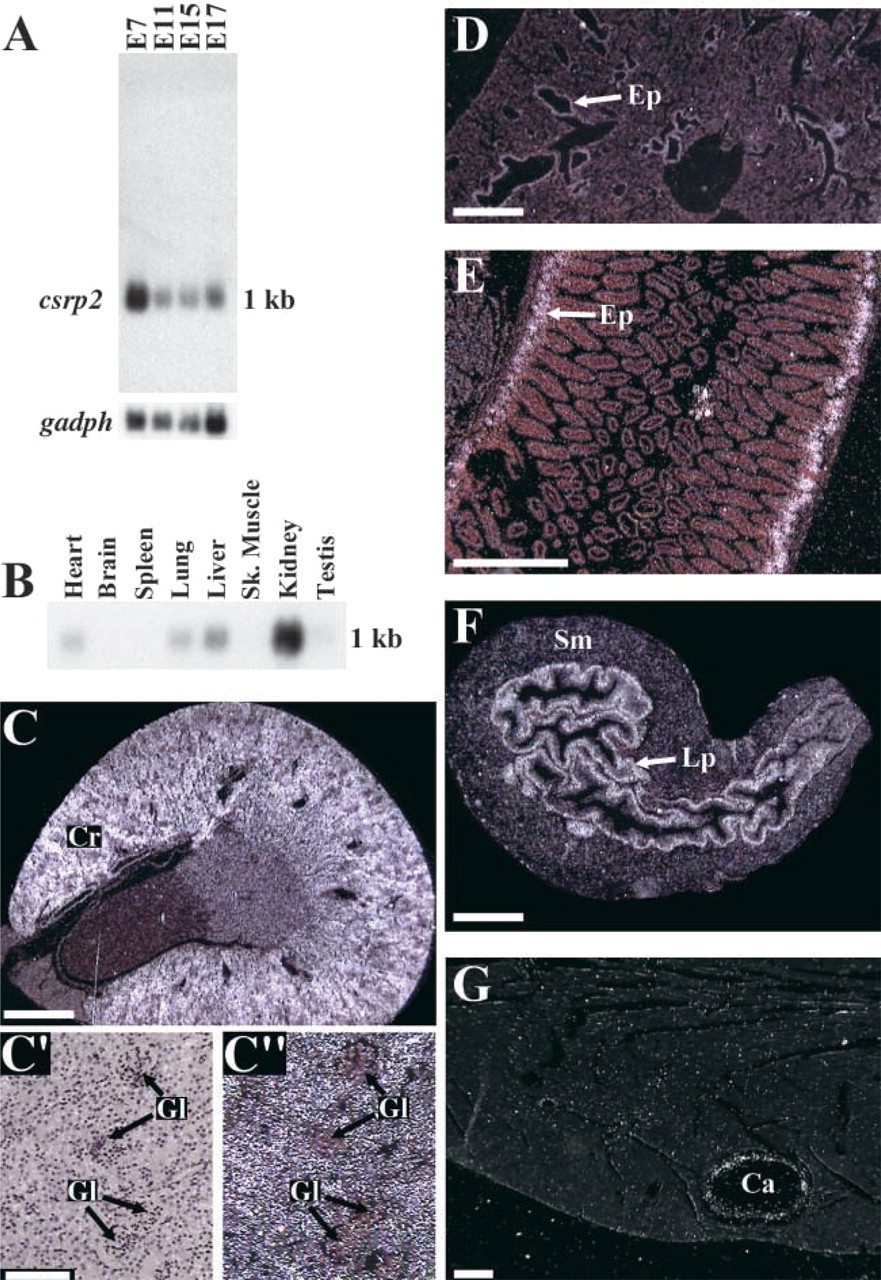
The sequence of mouse CRP2 (Figure 1A) is highly conserved, displaying 95.4% and 99.5% amino acid identity to chicken and human CRP2, respectively. Each LIM domain in CRP2 is followed by a short glycine-rich repeat (Figures 1A and 1B). Northern blotting analysis of total RNA from mouse embryos was performed using the csrp 3′ UTR as a probe. A single csrp2 transcript is detected during development (Figure 2A). The same blot probed for gadph transcripts indicates that all lanes contain RNA (Figure 2A). Analysis of RNA from adult organs (Figure 2B) indicates that csrp2 displays organ-specific regulation, with particularly high levels of expression observed in kidney. We characterized the expression pattern of csrp2 in several adult organs by ISH as described in Henderson et al. (1999). csrp2 is highly expressed in epithelial cells of the kidney (Figures 2C, 2C', and 2C'), lung (Figure 2D), small intestine (Figure 2E), and weakly in the germinal epithelium of the testis (data not shown). Strong expression occurs in the lamina propria of the bladder (Figure 2F). csrp2 expression is also observed in coronary artery smooth muscle cells (SMCs) (Figure 2G), and in the choroid plexus of the brain (data not shown). csrp2 transcripts were not detected in skeletal muscle (data not shown).
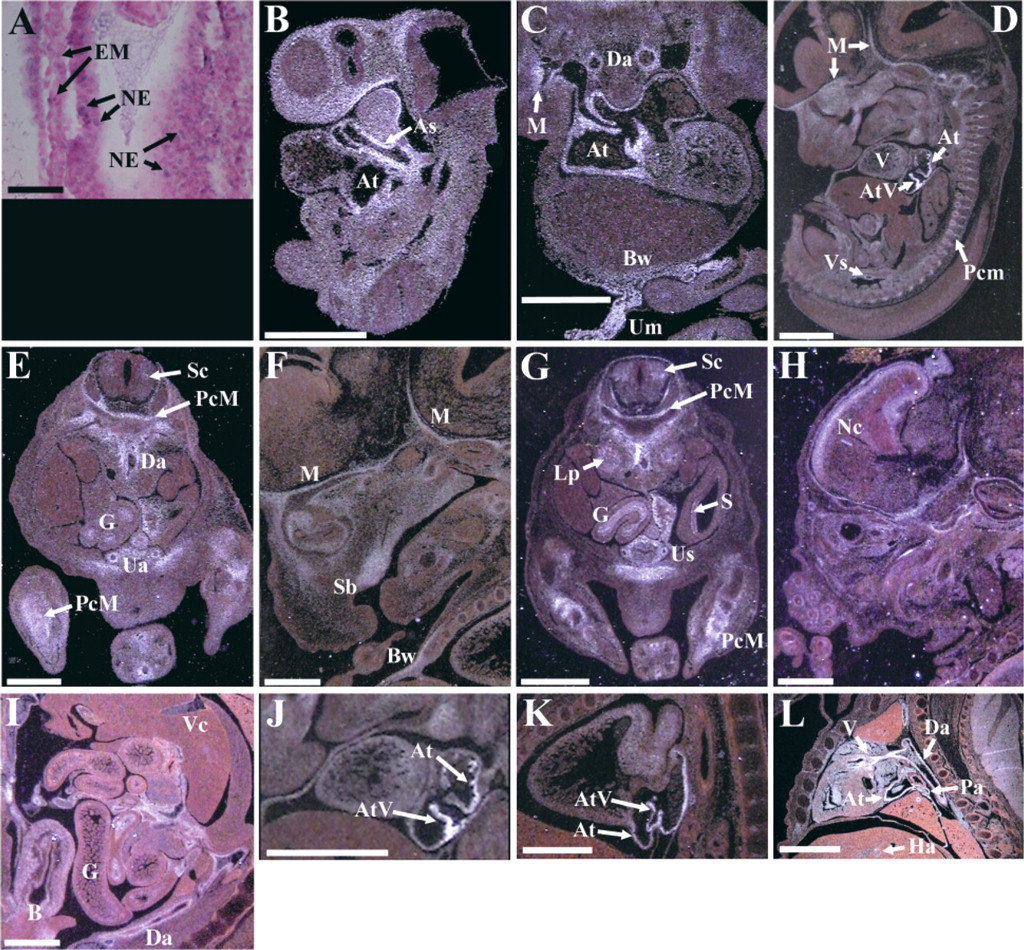
Consistent with the wide csrp2 expression pattern observed in adults, csrp2 transcripts were detected in a number of cell types during embryogenesis, including mesenchyme, vascular SMCs, and epithelial derivatives (Figure 3). In E7.0 embryos (Figure 3A), csrp2 transcripts are apparent in single cells within the embryonic mesoderm and neural ectoderm. As development proceeds, high levels of csrp2 transcripts are observed in populations of mesenchymal cells, notably in the head and in condensations surrounding developing bone (Figures 3B–3G). In contrast to csrp1, the SMC-specific CRP family member (Henderson et al. 1999), csrp2 transcripts were not detected in vascular SMCs of the dorsal aorta at E9.5 (Figure 3B), demonstrating that csrp2 is not an early marker of SMCs in the mouse. Instead, csrp2 expression was first observed in vascular SMCs at E11.5 (Figure 3C). Continued csrp2 expression was detected in both vascular and venous SMCs through development (Figures 3D, 3E, 3I, and 3L), as indicated in a previous report (Jain et al. 1998). csrp2 gut and stomach epithelium expression was first noted at E13.5 (Figure 3G) and continued at later stages (Figure 3I). Interestingly, although csrp2 transcripts are not observed in adult heart muscle (Figure 2G), significant csrp2 expression was detected in heart structures throughout development (Figures 3B–3D and 3J-3L).
CRP family members are small, actin cytoskeleton-associated proteins that are enriched in muscle (Arber et al. 1994; Louis et al. 1997; Henderson et al. 1999).
(
In this study we have analyzed the expression of the family member csrp2 in mouse. Consistent with a previous report, we determined that csrp2 is expressed in vascular, but not visceral SMCs (Louis et al. 1997). However, we also detected high levels of csrp2 transcripts in mesenchyme and epithelia during embryogenesis and in adult tissues. These data indicate that csrp2 is the most widely expressed CRP family member in mouse.
During embryogenesis, csrp2 expression was prominently associated with mesenchyme and epithelia. High levels of csrp2 transcripts were observed in a generalized mesenchymal pattern at E9.5, with the mesenchymal signal becoming restricted to cells in the head and in cell condensations surrounding developing bone as embryogenesis progressed. In adults, csrp2 transcripts remained associated with mesenchymal derivatives, including connective tissue in the bladder. csrp2 was also observed in epithelia during development and in adults, with significant levels observed in both the gut and kidney. In adult mouse, csrp2 expression in the kidney was the highest of any organ investigated.
Interestingly, csrp2 expression overlaps with that of csrp1 and csrp3/mlp in only a few areas. csrp2 and csrp1 transcripts are both detected in vascular smooth muscle, and even though csrp1 and csrp2 transcripts are not observed in adult heart muscle, all three csrp genes are expressed in the developing heart (Arber et al. 1997; Henderson et al. 1999; and this study). Targeted disruption of csrp3/mlp in the mouse results in cardiac hypertrophy and disorganization of the contractile apparatus. However, these phenotypes are postnatal manifestations. Expression of csrp1 (Henderson et al. 1999) and csrp2 (this study) in the embryonic heart suggests that CRP1 and/or CRP2 may be in a position to compensate for the loss of CRP3/MLP during heart development.
(
In summary, we have detected csrp2 transcripts in both muscle and non-muscle cells, indicating that csrp2 is the most widely expressed CRP family member. The non-muscle csrp2 expression is of particular interest because these are the first data suggesting that CRPs may function outside of striated and smooth muscle. As a result, further characterization of this highly related protein family may offer insight on the cytoskeletal organization of a variety of cell types.
Footnotes
Acknowledgements
Supported by NIH grant HL60591 (to MCB) and by the Huntsman Cancer Foundation.
We thank John M. Shelton and the Molecular Pathology Core laboratory at UTSW and Kelley Murphy at the HCI Tissue Access and Imaging Core facility for expert technical assistance, and the University of Utah Oligonucleotide Synthesis and Sequencing Core facilities. JRH (NIH Developmental Biology Training Grant 5T32 HD07491) thanks the Beckerle lab and T. Wallow for helpful discussions.
