Abstract
We previously found an abnormal deposition of an extracellular matrix glycoprotein, tenascin-C (TN-C), in human corneas with pseudophakic/aphakic bullous keratopathy (PBK/ABK). In this work, we studied cellular TN-C receptors in normal and PBK/ABK corneas. Cryostat sections of normal and PBK/ABK corneas were stained by immunofluorescence for TN-C receptors: α2, α8, α9, αVβ3, β1, and β6 integrins, and annexin II. β6 integrin mRNA levels were assessed by semiquantitative reverse transcription-polymerase chain reaction (RT-PCR) using β2-microglobulin gene to normalize the samples. In PBK/ABK compared to normal corneas, relatively minor changes were observed for α2 and β1 integrins, and for annexin II. α8, α9, and β6 subunits of TN-C receptors, α8β1 α9β1, and αVβ6, respectively, were absent from normal central corneas but were found in the central epithelium of PBK/ABK corneas. β6 integrin showed the most significant accumulation. It correlated best with the expression of TN-C rather than with the expression of other αVβ6 ligands, fibronectin, and vitronectin. RT-PCR analysis also showed elevated levels of β6 mRNA in PBK/ABK compared to normal corneas. Therefore, accumulation of TN-C in PBK/ABK corneas was accompanied by an increased expression of its three binding integrins, especially αVβ6 in the corneal epithelium. The interaction of tenascin-C with these integrins may contribute to the fibrotic process that occurs in PBK/ABK corneas.
P
We have previously shown that PBK/ABK corneas presented signs of an ongoing fibrotic process characterized by excessive deposition of specific extracellular matrix (ECM) components (Ljubimov et al. 1996, 1998b,c). The most conspicuous changes concerned an ECM glycoprotein, tenascin-C (TN-C). Unlike normal corneas, in which it was present only in the limbus, TN-C was deposited in the stroma and basement membranes (BMs) of central PBK/ABK corneas with a respective increase of mRNA levels (Ljubimov et al. 1996, 1998b). Smaller splice variants of TN-C were mostly found in PBK/ABK corneas (Ljubimov et al. 1998b; Saghizadeh et al. 1998).
TN-C may be adhesive for some cell types and antiadhesive for others (Mackie 1997; Vollmer 1997; Jones and Jones 2000; Orend and Chiquet-Ehrismann 2000). It has a number of splice variants that may differentially affect cell migration, proliferation, and neurite outgrowth (Murphy-Ullrich et al. 1991; Leprini et al. 1994; Meiners and Geller 1997; Ljubimov et al. 1998b; Joester and Faissner 1999; Meiners et al. 1999). TN-C has binding sites for various cell surface receptors that mediate its interactions with cells (Jones and Jones 2000). Except for annexin II (Chung and Erickson 1994), these receptors belong to the integrin family of heterodimer cell surface receptors and include α2β1, α8β1 α9β1, αvβ3, and αvβ6 (Joshi et al. 1993; Prieto et al. 1993; Sriramarao et al. 1993; Schnapp et al. 1995b; Yokosaki et al. 1996; Jones and Jones 2000). We hypothesized that elevated expression of TN-C in PBK/ABK corneas would be accompanied by a concomitant increase in its specific receptors. To explore this possibility, we have studied the expression of all known TN-C receptors in normal and PBK/ABK corneas by immunofluorescence. The results show that several epithelial TN-C-binding integrins (α8β1, α9β1, αvβ6) appeared in PBK/ABK corneas. Of these integrins, αvβ6 integrin showed the most significant correlation with TN-C overexpression.
Materials and Methods
Tissue
A total of 15 normal and 38 age-matched PBK/ABK corneas were used. Diseased corneas removed during corneal transplantation were received from local collaborating surgeons. Tissue collection was conducted under the Cedars-Sinai Medical Center IRB protocol number 3201. Normal autopsy corneas were received from the National Disease Research Interchange (Philadelphia, PA), which has a human tissue collection protocol approved by a managerial committee and subject to National Institutes of Health oversight. All tissues were processed within 30 hr after death or corneal transplantation. For PBK/ABK corneas, only a central part was available that had been removed during transplantation. Because of this, all ECM abnormalities in PBK/ABK corneas were previously documented in the central part only, where Bowman's layer lies underneath the epithelium (Ljubimov et al. 1996). Therefore, for gene expression study by RT-PCR, normal corneas were trephined before freezing and central parts only (without limbus) were analyzed to ensure adequate comparison between groups. In immunostaining experiments, again, only central parts of normal and PBK/ABK corneas were compared. Because of the small amount of material available from a single cornea, immuno histochemical and RT-PCR studies were performed with different sets of normal and PBK/ABK corneas.
Immunohistochemistry
Corneas (10 normal and 26 age-matched PBK/ABK) were bisected and embedded in OCT compound (Ted Pella; Redding, CA). Six-μm cryostat sections were prepared and stained by indirect immunofluorescence for ECM components and integrins, as previously described (Ljubimov et al. 1995). Routine specificity controls (without primary or secondary antibodies) were negative. Sections of normal and PBK/ABK corneas were exposed to the same dilutions of antibodies simultaneously. Hybridoma supernatants were used undiluted, and purified antibodies were used at 10–20 μg/ml. Well-characterized mouse monoclonal antibodies were used to total TN-C (clone BC8; Balza et al. 1993; a kind gift from Dr. L. Zardi, Istituto Nazionale per la Ricerca sul Cancro, Genoa, Italy), total fibronectin (clone 568; Ljubimov et al. 1995), annexin II (clone 5; Transduction Laboratories, Lexington, KY), vitronectin (clone RM68), integrins αvβ3 (clone LM609), α3β1 (clone M-KID 2), α1 I domain (clone FB12), α6 (clone NKI-GoH3), β1 (clone HB1.1), β4 (clone 3E1) (all from Chemicon International; Temecula, CA), and β6 (clone E7P6; Weinacker et al. 1994). Rabbit polyclonal antibodies were used to TN-C and integrins α5β1, α2 (all from Chemicon International), α8 (Schnapp et al. 1995a), and α9 (Palmer et al. 1993; Sta Iglesia et al. 2000). Both antibodies to α9 reacted in an identical way. Cross-species adsorbed fluorescein- and rhodamine-conjugated secondary antibodies were from Chemicon International. In double-label experiments (not shown here), it was found that in PBK/ABK corneas, affinity-purified polyclonal antibody to TN-C (Chemicon International) reacted identically with the monoclonal antibody BC-8.
Semiquantitative RT-PCR
This was done as previously described (Ljubimov et al. 1998b; Saghizadeh et al. 1998). Briefly, total RNA was isolated from five normal and 12 age-matched PBK/ABK corneas with TRIZOL (Invitrogen Life Technologies; Carlsbad, CA). cDNA samples were subjected to polymerase chain reaction (PCR) using specific primers for β6 integrin and for β2-microglobulin (β2-MG) that served as a housekeeping gene for sample normalization. The primers for RT-PCR were the following: β6 integrin forward, 5′-GTGAGATCAACATCAATGGAGC-3′; β6 integrin reverse, 5′-TTACACACTCCTGGGAACACTTC-3′; product size 181 base pairs (bp); β2-MG (Saghizadeh et al. 1998) forward, 5′-CTCGCGCTACTCTCTCTTTCTG-3′; β2-MG reverse, 5′-GCTTACATGTCTCGATCCCACTT-3′, product size 335 bp.
PCR was carried out with 25 ng of reverse-transcribed RNA, using 1.25 U Taq polymerase (Invitrogen Life Technologies) and 2 mM Mg++. Each cycle consisted of 30-sec denaturation at 94C, 30-sec annealing at 65C, and 45-sec elongation at 72C. Samples normalized by β2-MG amplification were amplified in a linear range established using serial cDNA dilutions and varying the number of cycles. A total of 33 cycles were used for β6 integrin and 31 cycles for β2-MG. Amplified products were separated by electrophoresis in 3% agarose gels and visualized under UV light after staining with ethidium bromide. Routine RT-PCR controls without reverse transcriptase were negative.
Southern blotting analysis was done as described previously (Ljubimov et al. 1998b) using the following oligonucleotide probes: β6 integrin, 5′-GGAAGCCCGTGCACCCTATGCCAAGTTGATCCCATAT-3′; β2-MG (Saghizadeh et al. 1998), 5′-GTCTTTCAGCAAGGACTGGTCTTTCTATCTCTTGTA-3′. Band densities were measured by scanning densitometry and the ratios of β6 integrin to β2-MG for each sample were determined.
Statistical Analysis
Immunostaining data were analyzed by the two-sided Fisher's exact test using In Stat software program (GraphPad Software; San Diego, CA). To this end, the number of cases with abnormal staining pattern in one experimental group (e.g., normal) was compared to the number of cases with abnormal staining pattern in another experimental group (e.g., PBK/ABK). RT-PCR band relative intensities were compared by the two-tailed unpaired Student's t-test using the InStat program and were expressed as mean ± SEM. With both methods, p<0.05 was considered significant.
Results
Appearance of Tenascin-C-binding Integrins α8β1, α9β1, and αvβ6 in PBK/ABK corneas
Normal central corneal epithelial cells and keratocytes expressed the TN-C receptors α2β1 integrin and annexin II, as determined by immunofluorescence (Figure 1). In the corneal epithelium, distinct basal cell surface staining was seen for both components. In central PBK/ABK corneas, these receptors were not changed in about half of cases. In the remaining cases (5/11), α2 integrin appeared reduced in the epithelium (Figure 1). In 6/11 PBK/ABK cases, more (Figure 1) or less staining than normal was observed for annexin II, especially at the basal surface of basal epithelial cells. The stromal staining for both these receptors did not change in diseased corneas. αvβ3 integrin was not found either in normal or in PBK/ABK corneas (Figure 1), in accordance with previous data (Tervo et al. 1991).
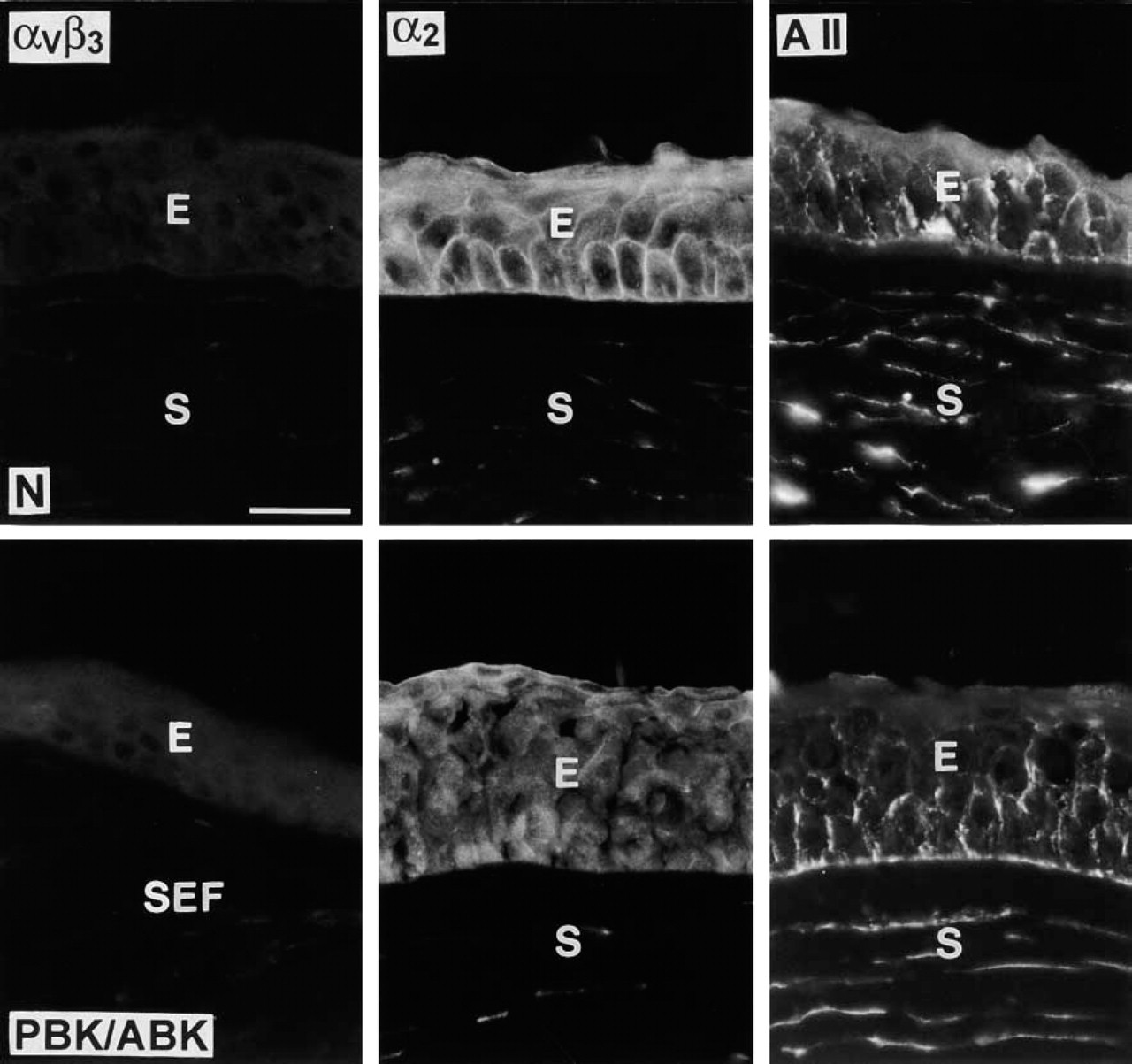
Select TN-C receptors in normal (N) and PBK/ABK corneas. αvβ3 integrin (left column) is absent from normal and diseased corneas. α2 integrin staining is positive in normal corneal epithelial cells and is either unchanged in PBK/ABK corneas (not shown) or decreased (middle column). Annexin II (A II) is present in normal epithelium and keratocytes and shows some increase in epithelial staining in some PBK/ABK corneas (right column). E, epithelium; S, stroma; SEF, subepithelial fibrosis. Immunofluorescent staining. Bar = 40 μm.
Subunits of the TN-C-binding integrins α8 and β6 were absent in normal corneas, both in the central part (Figure 2, upper row) and the limbus (not shown). Only α9 integrin, in accordance with previous data in mice (Stepp et al. 1995; Stepp and Zhu 1997), was observed in normal limbal basal epithelial cells and, in some cases, in the most peripheral corneal basal cells (not shown). However, all these integrins appeared in the central epithelium of PBK/ABK corneas (Figure 2, lower row; Table 1). Ten of 17 PBK/ABK cases (59%) were positive for α8 integrin, 11/17 (65%) were positive for α9, and 12/15 (80%) were positive for β6. All these integrins were mostly expressed in basal epithelial cells, whereas stromal keratocytes and endothelial cells were negative (Figure 2). The expression of these integrins was heterogeneous. Some PBK/ABK cases showed small local areas of positive staining, whereas in other cases the staining for these integrins was observed throughout large parts of the basal epithelium up to a whole cornea. The induction of these integrins was next correlated with the subepithelial expression of TN-C. For α8 integrin, such correlation was found in 8/17 cases (47%), for α9 in 9/17 cases (53%), and for α6 in 11/15 cases (73%).
TN-C-binding β6 integrin, the most increased in PBK/ABK corneal epithelium, also binds to fibronectin and vitronectin. In a separate set of experiments, we tested by double immunofluorescence on serial sections whether β6 expression in these corneas correlated with the expression of either vitronectin or fibronectin or TN-C. The correlation was judged to be positive both in cases where the integrin and the ligand were present (the integrin, in the epithelium, and the ligand, in the subepithelial region and/or epithelial BM), and in cases where the integrin and the ligand were both absent. Double staining for fibronectin and β6 integrin, and also for vitronectin and β6 integrin, was done using pairs of mouse monoclonal antibodies together. This was possible because the patterns of the ligands and the receptor did not overlap. Fibronectin and vitronectin were normally present in keratocytes (fibronectin also in the epithelial BM) but not in the epithelial cells, whereas β6 integrin could be found only in the epithelial cells.
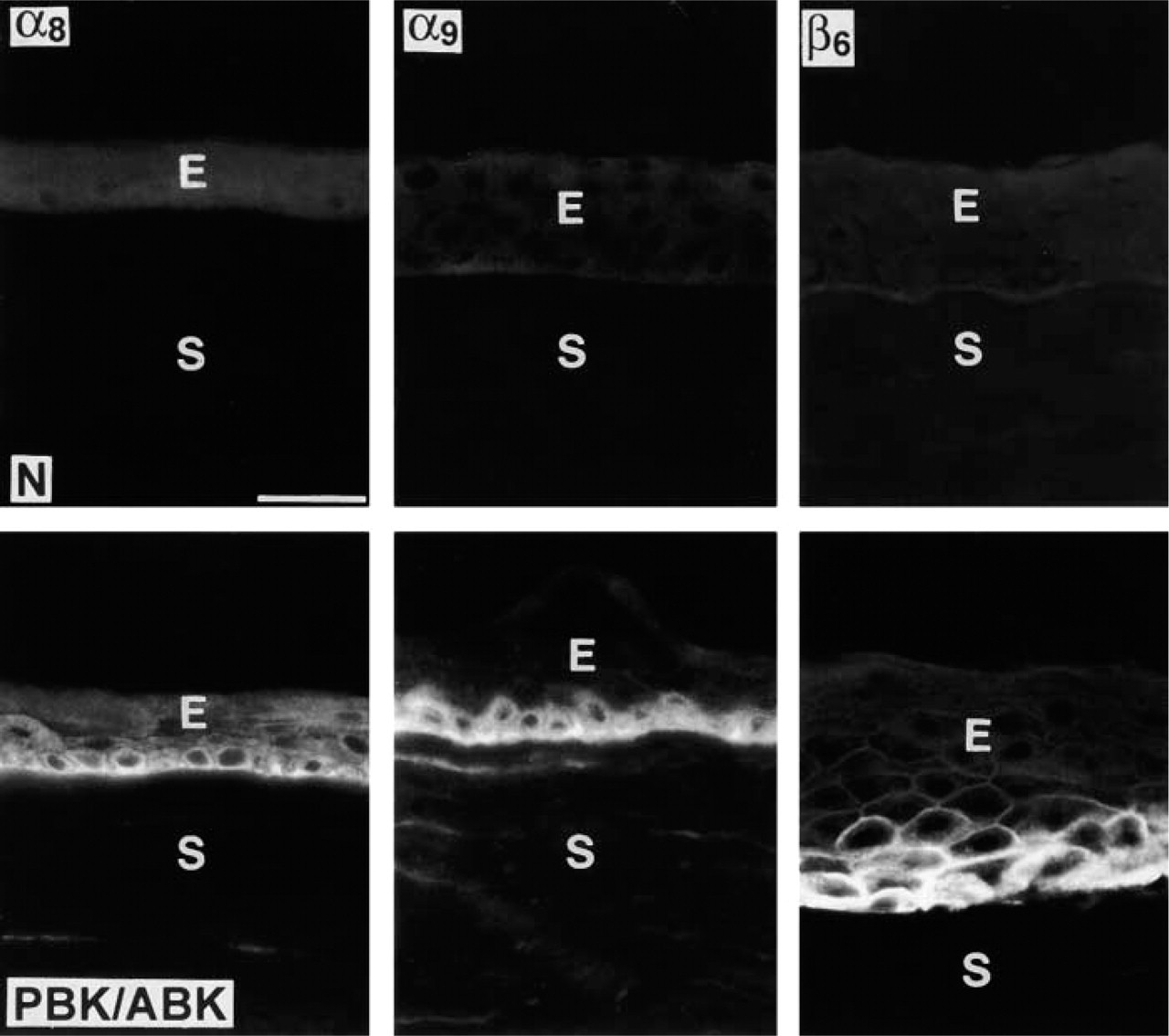
Appearance of TN-C integrin receptors in PBK/ABK corneas. Note the absence of α8, α9, and β6 integrins in normal corneas (N, upper row) and their presence in the epithelium of PBK/ABK corneas, mostly in the basal cells (lower row). E, epithelium; S, stroma. Immunofluorescent staining. Bar = 40 μm.
The results showed that β6 expression in PBK/ABK corneal epithelium correlated with the expression of subepithelial fibronectin in 8/21 cases. Correlation of β6 with vitronectin expression was seen in 7/20 cases. The correlation of β6 with TN-C expression was significantly higher than with fibronectin or vitronectin (p<0.0008) and was observed in 21/24 cases (three of these cases were negative for both β6 and subepithelial TN-C) (Figure 3). These results suggested that β6 integrin appeared coordinately with TN-C in PBK/ABK corneas.
We have previously shown that PBK/ABK corneas also displayed a decreased and discontinuous staining of the epithelial BM for fibronectin, Type IV collagen, laminin, and nidogen-1/entactin (Ljubimov et al. 1996). Therefore, in control experiments we analyzed the distribution of respective integrin receptors for these components in normal and diseased corneas. The distribution of α5β1 (fibronectin receptor), α6β4 (laminin receptor), and α3β1 (collagen/laminin/entactin receptor) did not change in PBK/ABK epithelium (Table 1). α1β1 (collagen receptor) was not found in any of the central corneas (not shown). These results suggest that epithelial cell integrin changes in PBK/ABK may reflect the abnormal presence of TN-C rather than alterations of major epithelial BM components or nonspecific corneal damage during storage and handling.
Increased Expression of β6 Integrin mRNA in PBK/ABK Corneas
Semiquantitative RT-PCR from total corneal RNA demonstrated that normal corneas had relatively low levels of β6 integrin mRNA that significantly increased in PBK/ABK corneas (Figure 4). The average increase of β6 integrin mRNA expression in PBK/ABK vs normal corneas was approximately twofold and statistically significant (p<0.008). No correlation was found between β6 integrin mRNA levels and the degree of visual acuity loss in PBK/ABK corneas examined before corneal transplantation.
Discussion
Tenascins form a family of five related large, hexameric ECM glycoproteins with modular structure. They play a role in various cell functions, including adhesion, migration, differentiation, and proliferation. In the extracellular space, tenascins can bind a plethora of other ECM molecules. Tenascins also bind to the surface of many cell types using several defined receptors, most of them belonging to the integrin family (reviewed in Orend and Chiquet-Ehrismann 2000; Jones and Jones 2000). Although prominent in developing embryos, tenascins become rather scarce in the adult state. However, they (especially TN-C) are often up-regulated during wound healing and in disease conditions, including tissue fibrosis and cancer (Crossin 1996; Vollmer 1997).
ECM receptor alterations in PBK/ABK corneal epithelium a
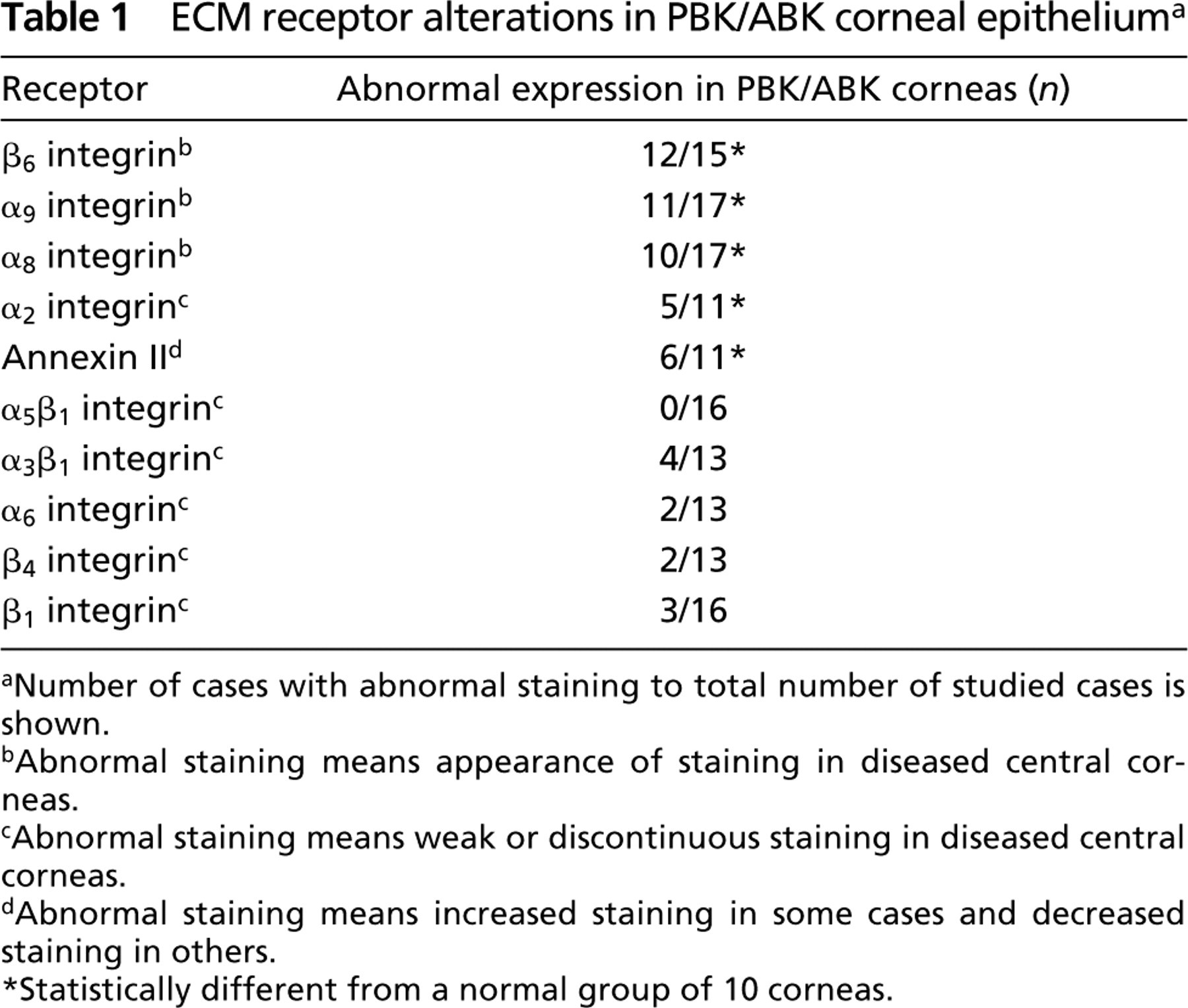
Number of cases with abnormal staining to total number of studied cases is shown.
Abnormal staining means appearance of staining in diseased central corneas.
Abnormal staining means weak or discontinuous staining in diseased central corneas.
Abnormal staining means increased staining in some cases and decreased staining in others.
Statistically different from a normal group of 10 corneas.
TN-C is fairly abundant in normal central fetal corneas (Maseruka et al. 2000). In contrast, adult corneas retain it only in the peripheral limbus area (Tervo et al. 1990; Ljubimov et al. 1996), and the central cornea expresses TN-X only in the stroma (Tuori et al. 1999). However, in scarred, inflamed, and diseased corneas, TN-C becomes expressed again (Ljubimov et al. 1996, 1998a, b; Zhou et al. 1996; Kenney et al. 1997; Maseruka et al. 1997; Tuori et al. 1997; Matsuda et al. 1999a). It is most abundant in corneas with a common edematous disorder, PBK/ABK (Ljubimov et al. 1996, 1998a).
We hypothesized that abnormal expression of TN-C in PBK/ABK corneas might be accompanied by altered expression of some of its binding proteins. A systematic study of these proteins in normal and PBK/ABK corneas was conducted. We found some TN-C receptors in both normal and PBK/ABK corneas, including α2β1 integrin and annexin II. These receptors showed abnormal staining in about half of diseased corneas. The α2β1 integrin is also a laminin/collagen receptor and was usually decreased in PBK/ABK epithelium. This might be due to laminin and/or collagen alterations seen in the epithelial BM of PBK/ABK corneas (Ljubimov et al. 1996). Our first account of the annexin II distribution pattern in human cornea completely agrees with findings recently described for rat cornea (Matsuda et al. 1999b). These authors concluded that annexin II co-distributed with TN-C only in the limbus or during wound healing. In about half of PBK/ABK corneas, the epithelial staining for annexin II was either somewhat stronger compared to normal corneas (Figure 1) or somewhat weaker, with no difference in the stroma. Because of such changes, rather inconsistent with common upregulation of TN-C in PBK/ABK corneas, α2β1 integrin and annexin II were not further studied.
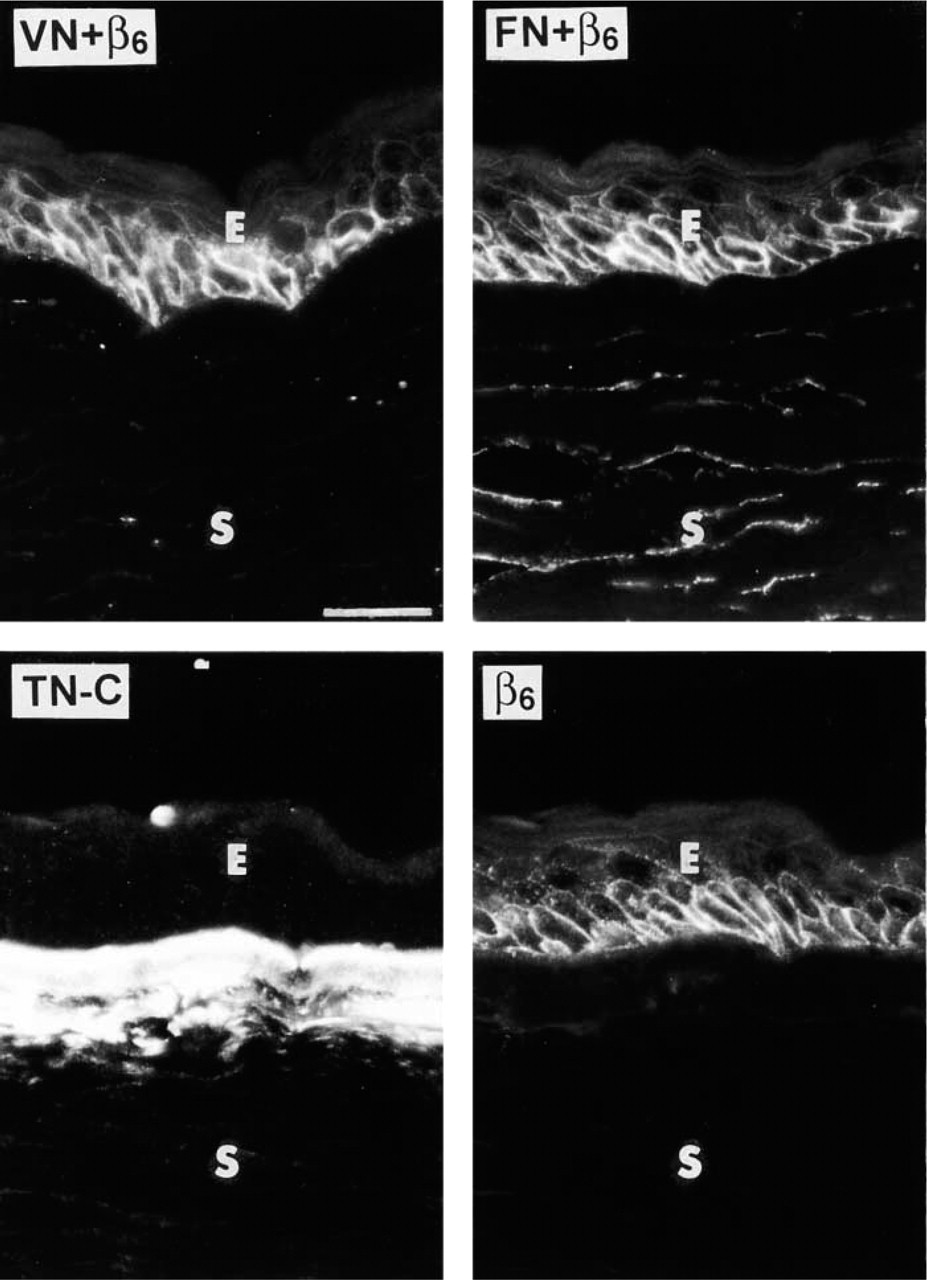
Co-distribution of β6 integrin with TN-C but not with fibronectin (FN) or vitronectin (VN) in PBK/ABK corneas. Representative serial sections are shown. Upper row, left: double staining for β6 integrin and VN. VN is normally absent from the epithelial BM and does not appear there in PBK/ABK corneas even in areas positive for β6 integrin (no co-distribution). Upper row, right: double staining for β6 integrin and FN. FN is normally present in the epithelial BM, Descemet's membrane, and keratocytes. Here, FN is absent from corneal epithelial BM (it is only present in keratocytes) despite the appearance of epithelial β6 integrin in the same area (no co-distribution). Lower row: double staining of one section serial to those in the upper row for TN-C (left, polyclonal antibody) and β6 integrin (right). TN-C is normally absent from adult central cornea. Here, abundant TN-C is revealed under the epithelial cells that express β6 integrin (co-distribution). E, epithelium; S, stroma. Immunofluorescent staining. Bar = 40 μm.
Three TN-C-binding epithelial integrins, α8β1, α9β1, and αvβ6, did show correlation with TN-C expression. All of them, like TN-C, were only found in PBK/ABK central corneas. The corneal α9β1 integrin has been thoroughly studied by Stepp's group (Stepp et al. 1995; Stepp and Zhu 1997; Sta Iglesia et al. 2000). We previously showed elevated expression of the β6 integrin in PBK/ABK corneas by gene array analysis (Spirin et al. 1999) and are now confirming this result using other methods. We also provide the first description of the α8β1 integrin distribution in normal and PBK/ABK human corneas, which was largely similar to that of αvβ6 integrin.
The α8β1 integrin can serve as an RGD-dependent receptor for TN-C, fibronectin, vitronectin, and osteopontin (Schnapp et al. 1995b; Varnum-Finney et al. 1995; Denda et al. 1998a,b). It is expressed on myofibroblasts that contribute to fibrotic response to injury (Levine et al. 2000) and mediates interactions of embryonic chick motor and sensory neurons with TN-C (Varnum-Finney et al. 1995). However, in vitro, α8β1 could only bind to TN-C fragments but not to total protein (Denda et al. 1998a). The importance of this integrin for kidney morphogenesis, as revealed in knockout mice (Muller et al. 1997), is apparently due to its interaction with osteopontin (Denda et al. 1998b) but not with TN-C.
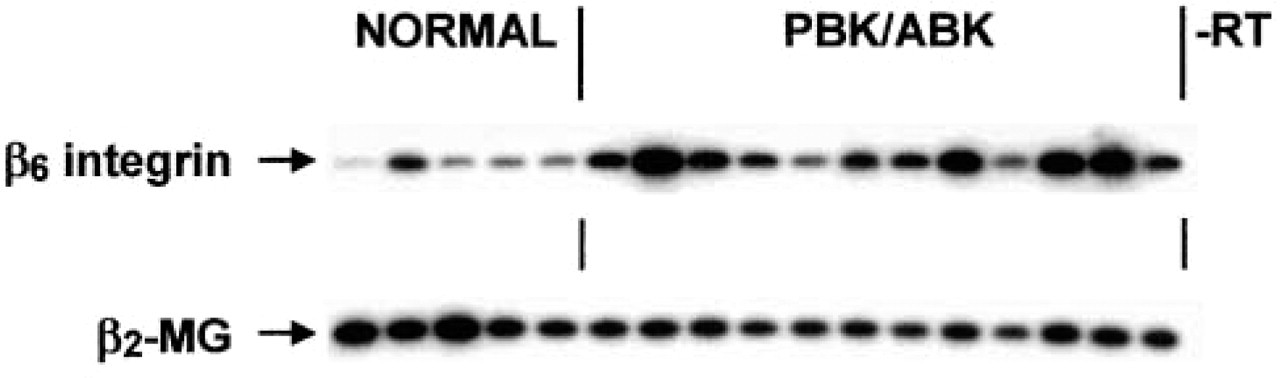
Semiquantitative RT-PCR analysis of β6 integrin gene expression in normal and PBK/ABK corneas. Lanes 1–5, normal corneas; Lanes 6–17, PBK/ABK corneas. Note increased expression of β6 integrin gene in PBK/ABK corneas. Samples were normalized with respect to β2-microglobulin (β2-MG) cDNA amplification. Autoradiographs of Southern blots are shown. -RT, negative control without the reverse transcription step.
The α9β1 integrin is expressed in epithelial, muscle, and neutrophil cells and binds to vascular cell adhesion molecule 1 (VCAM-1), TN-C, osteopontin, transglutaminase, and other ligands (Yokosaki et al. 1994; Smith et al. 1996; Taooka et al. 1999; Marcinkiewicz et al. 2000; Takahashi et al. 2000). It appears to be required for normal development of the lymphatic system (Huang et al. 2000). In neutrophils and activated endothelium, VCAM-1 seems to be the major ligand for α9β1 integrin (Taooka et al. 1999). In the epithelial cells, α9β1 integrin may interact with TN-C. These two proteins co-distribute during oral mucosa wound healing and in proliferative disorders (Häkkinen et al. 1999, 2000), and during healing of injured nasal mucosa (Yoshimura et al. 1999). In the cornea, α9β1 integrin is confined to the limbal basal epithelium (Stepp et al. 1995) but appears, together with TN-C, during restratification after wound healing of central corneal epithelium (Stepp and Zhu 1997). Surprisingly, α9β1 integrin is also induced in the central corneal epithelium after wound healing in TN-C knockout mice (Sta Iglesia et al. 2000). These data show that caution should be exerted in inferring interactions of proteins on the basis of their spatial and/or temporal co-distribution.
The αvβ6 is an epithelial integrin and can bind TN-C, fibronectin, and vitronectin (Busk et al. 1992; Prieto et al. 1993; Breuss et al. 1995; Huang et al. 1998). Many epithelia express this integrin only in the developing embryo but not in the adult state (Breuss et al. 1995). It can be re-expressed during inflammation, tissue repair after injury, tumor growth, at late stages of wound healing, and in fibrotic diseases (Breuss et al. 1995; Weinacker et al. 1995; Clark et al. 1996; Haapasalmi et al. 1996; Cass et al. 1998; Spirin et al. 1999; Häkkinen et al. 2000; Kaminski et al. 2000; Miller et al. 2001). This integrin is not necessary for wound healing, as shown using α6 knockout mice (Huang et al. 1996). However, it plays a role as a downregulator of inflammation and may be involved in tissue fibrosis (Munger et al. 1999). Several growth factors, including transforming growth factor (TGF)-β induce the expression of αvβ6 integrin (Sheppard et al. 1992). In turn, this integrin can bind and activate the latent form of TGF-β1 (Munger et al. 1999).
As we showed here, βvβ6 integrin was absent from normal corneal epithelium but became expressed in most PBK/ABK corneas. Increased mRNA levels accompanied its appearance in diseased corneas. An important finding was that αvβ6 integrin expression specifically correlated with the deposition of subepithelial TN-C. At the protein level, such a correlation was also observed at late stages of oral mucosal healing (Häkkinen et al. 2000) and in lung fibrosis at the mRNA level (Kaminski et al. 2000). On the basis of the available data, we can speculate that αvβ6 integrin may be first expressed in PBK/ABK corneal epithelium as a result of epithelial alterations including edema, remodeling, and proliferation. This may activate corneal TGF-β which, in turn, could induce the expression of subepithelial TN-C as part of the fibrotic process typical for this corneal disorder. Alternatively, TN-C may be up-regulated by IGF-I elevated in PBK/ABK corneas (Saghizadeh et al. 2001; and our unpublished data), which could contribute to the αvβ6 integrin appearance in the epithelium.
TN-C is associated with inflammation and wound healing in remodeling and pathological tissues (Crossin 1996; Maseruka et al. 1997; Ljubimov et al. 1998a; Jones and Jones 2000). Using knockout mice, it was shown that TN-C is not needed for corneal epithelial wound healing (Sta Iglesia et al. 2000). In the absence of TN-C, α9β1 integrin is still induced during corneal wound healing, suggesting that it binds to a different ligand (Sta Iglesia et al. 2000). It would indeed be interesting to study αvβ6 in TN-C knockout mice, but mouse-reacting antibodies to this integrin that would stain tissue or work in Western blotting are not available.
At the same time, TN-C knockout mice have abnormal wound healing in corneal stroma (Mackie and Tucker 1999; Matsuda et al. 1999c). Incidentally, TN-C deposits are also found primarily in the stroma of PBK/ABK corneas. However, these corneas do not appear to have an ongoing wound healing. During wound healing, disruptions of normal tissue architecture are repaired (Clark 1996). However, chronically edematous (because of fluid accumulation) PBK/ABK corneas usually do not display such disruptions and the integrity of the epithelial, stromal, or endothelial layer in these corneas may be compromised only in rare severe cases with rupture of bullae. Although PBK/ABK corneas have a fibrotic accumulation of certain ECM components, including TN-C, no typical scar tissue is formed and only scattered stromal myofibroblasts can be found (Ljubimov et al. 1996; Kenney et al. 2001). Interestingly, we did not observe any changes in the patterns of TN-C receptors in the PBK/ABK corneal stroma. It is possible either that these changes are quantitative and impossible to document by qualitative immunofluorescence or that stromal keratocytes use as yet unidentified receptors to TN-C. Further studies are needed to clarify these issues.
In summary, PBK/ABK corneas show abnormal upregulation of several TN-C-binding epithelial integrins, including αvβ6. The epithelial expression of this integrin spatially correlates with that of subepithelial TN-C (but not fibronectin or vitronectin) in diseased corneas and may be part of and/or contribute to the fibrotic process going on in PBK/ABK corneas.
Footnotes
Acknowledgments
Supported by grants from the NIH (EY10836 to MCK), the Braille Transcribers Guild (AVL), the Discovery Fund for Eye Research, and the Skirball Program for Molecular Ophthalmology (AVL, MCK).
We thank Drs Anthony Nesburn, Ezra Maguen, Ronald Gaster, Wayne Bowman, John Hofbauer, Theodore Perl, and Ira Wong for providing us with bullous keratopathy corneas, and the National Disease Research Interchange for supplying normal human corneas. We are grateful to Drs Mary Ann Stepp (George Washington University Medical Center; Washington, DC) and Luciano Zardi (Istituto Nazionale per la Ricerca sul Cancro; Genoa, Italy) for donating antibodies.
