Abstract
In a attempt to improve the sensitivity of the simultaneous use of immunohistochemistry (IHC) with estrogen receptor (ER) and in situ hybridization (ISH) with a neuropeptide receptor, we first applied an existing microwave (MW) irradiation protocol for immunohistochemical detection of the estrogen receptor in frozen brain sections. Regions of interest were the preoptic area and the arcuate nucleus of the hypothalamus. ER signal was effective only after MW heating of sections in the two regions. Control sections without pretreatment exhibited no staining for ER. Second, the MW protocol was applied in a novel procedure that consists of evaluation of the expression of the galanin receptor mRNA with a radioactive riboprobe after MW pretreatment. The galanin receptor mRNA signal intensity obtained after heating was quantitatively at least as good or significantly increased according to the region, with no discernible loss of tissue morphology. Finally, we describe a novel application of MW pretreatment on the same frozen section processed with ER antibody and a radioactive galanin receptor riboprobe. The stainings for estrogen and galanin receptors were intense in many cells of the preoptic area, with very low background. These results show that both IHC and ISH can be significantly improved by subjecting frozen sections to MW heating before the double labeling. This approach may provide a potential method to answer the important question of whether or not estrogen has a direct action on the expression of a peptide receptor.
Keywords
D
The aim of this work was to determine if the use of MW heating pretreatment was (a) efficient to enhance the sensitivity of detection of neuropeptide receptor mRNAs and (b) favorable for double detection, i.e., IHC for detection of estrogen receptor and ISH for neuropeptide receptor. For this investigation, we first experimented on the effects of MW treatment for immunohistochemical detection of ER. In a parallel experiment, we applied the same pretreatment on the detection of a neuropeptide receptor signal to verify that the signal intensity was as good or even enhanced as that obtained with routine ISH. The target RNA was the rat hypothalamic galanin receptor Gal-R1 mRNA expression, chosen as a model because Gal-R1 is expressed in many regions of the rat hypothalamus (Mitchell et al. 1997). Samples were fixed and processed with our unchanged ISH protocol (Mitchell et al. 1997, 1999a,b; Faure-Virelizier et al. 1998; Bouret et al. 2000) in regions involved in the reproductive axis, i.e., the preoptic area and arcuate nucleus. Then we tried similar MW treatment for double IHC detection on the same section of ER, followed by ISH detection of galanin receptor mRNA expression. We suggest a protocol that improves the specific double labeling for two receptors on the same frozen brain section.
Materials and Methods
Animals and Tissue Preparation
Four adult ovariectomized (OVX) female Wistar rats (200–250 g; CERJ, Le Genest St Isle France) were used. They were maintained on a 14-hr light and 10-hr dark cycle (lights on at 0500 hr) with food and water available ad libitum. After 12 days, animals were anesthetized with 20 mg/kg ketamine and 0.2 ml/kg xylazine, then perfused intracardially with 5–10 ml of saline followed by 500 ml of 4% paraformaldehyde in 0.1 M phosphate buffer. The brains were removed and immersed in the same fixative for 2 hr. They were then washed overnight in 0.05 M Coons' veronal buffer (pH 7.4) containing 20% sucrose, embedded in Tissue-Tek (Miles Laboratories; Naperville, CA), and frozen in liquid nitrogen. Frozen 14-μm coronal sections were cut from the level of –1.8 to –4.8 mm relative to Bregma, according to the atlas of Swanson (1992). Four panels of pairs of adjacent sections (one not preheated by MW and the following one preheated by MW) of each specimen were collected for subsequent staining, IHC, ISH, and double labeling. The sections were mounted on gelatin-coated slides and stored at –80C until used. All experiments were carried out in accordance with the European Communities Council Directive of 24 November 1986 (86/609/EEC) regarding mammalian research.
MW Treatment
Before the IHC, ISH, or double labeling IHC–ISH procedures, some sections were submitted to MW treatment. MW heating was performed using a domestic Samsung MW oven. Sections were immersed in a DEPC-treated sodium citrate buffer, 10 mM at pH 6.0, for 5 min at 800 W, immediately followed by two 4-min periods at 400 W. Sections were allowed to cool in the same buffer for 45 min at room temperature. The IHC and ISH procedures were performed as usual.
IHC labeling for ER
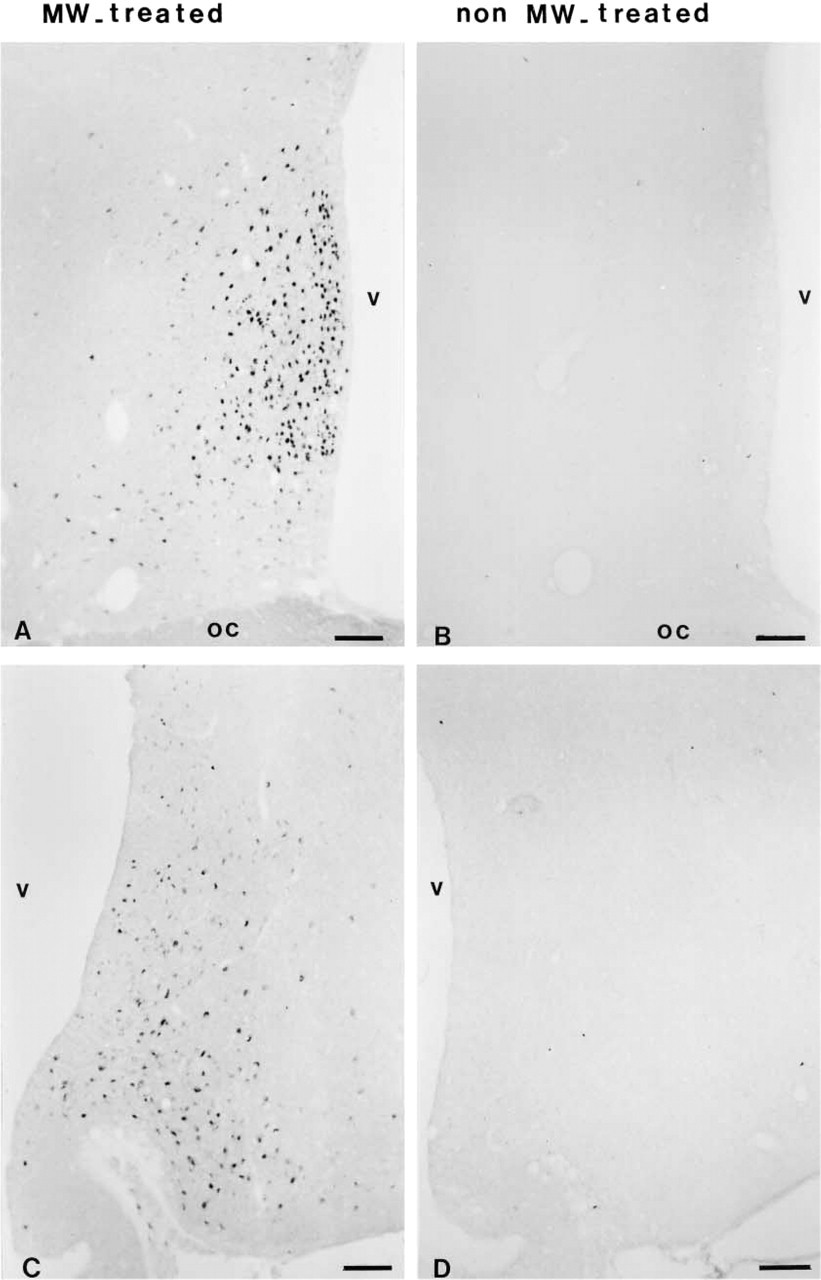
Some sections from each animal were rinsed in 0.1 M Tris-buffered saline (TBS) and incubated for 48 hr at 4C with a mouse monoclonal ER antibody (clone 1D5; Dako, Trappes, France) diluted 1:100 with TBS containing 0.3% Triton. A group of sections was heated by MW as described above and the other group of adjacent sections was not pretreated. Antigen–antibody binding sites were visualized using biotinylated goat anti-mouse serum (1:200, for 90 min; Caltag Laboratories, Burlingame, CA), followed by a peroxidase-conjugated avidin (Vector Laboratories; Burlingame, CA). Sections were processed for the DAB reaction as usual.
The specificity of the ER antibody has been previously checked in human tissues (Kumar et al. 1987; Al Saali et al. 1993), and had recently been tested for rat ER-α (Nishihara et al. 2000). Control of the specificity of the IHC reaction was verified by replacement of the primary antibody by normal mouse serum, which led to the absence of immunoreaction.
35S-Labeled Gal-R1 Receptor Complementary RNA Probe
The plasmid vector pBluescript IIKS containing a HindIII/BamHI fragment of 1600 bp of the full-length rat Gal-R1 receptor (Burgevin et al. 1995) was used. BamHI and T7 made the antisense probe, and HindIII with T3 RNA polymerase produced the sense probe. The radioactive probes were generated by labeling with 200 μCi of [35S]-CTP (Amersham; Les Ulis, France) using 1 μl of the appropriate RNA polymerase in a 40-μl transcription reaction volume containing 8 μl of 5 × transcription buffer, 2 μl of 0.1 M dithiothreitol (DTT), 1 μg of linearized plasmid (50 ng/ml), 1 μl of E. coli tRNA (5 mg/ml), 20 U of RNasin, and 1 μl of a 10 Mm stock solution of ATP, GTP, and UTP. The transcription reagents were incubated for 4 hr at 39C. Labeled probes were extracted with phenol–chloroform and separated from non-incorporated nucleotides on a Sephadex G-50 column. The 35S-labeled riboprobes were diluted with hybridization buffer to a final concentration of 30,000 dpm/μl.
In Situ Hybridization
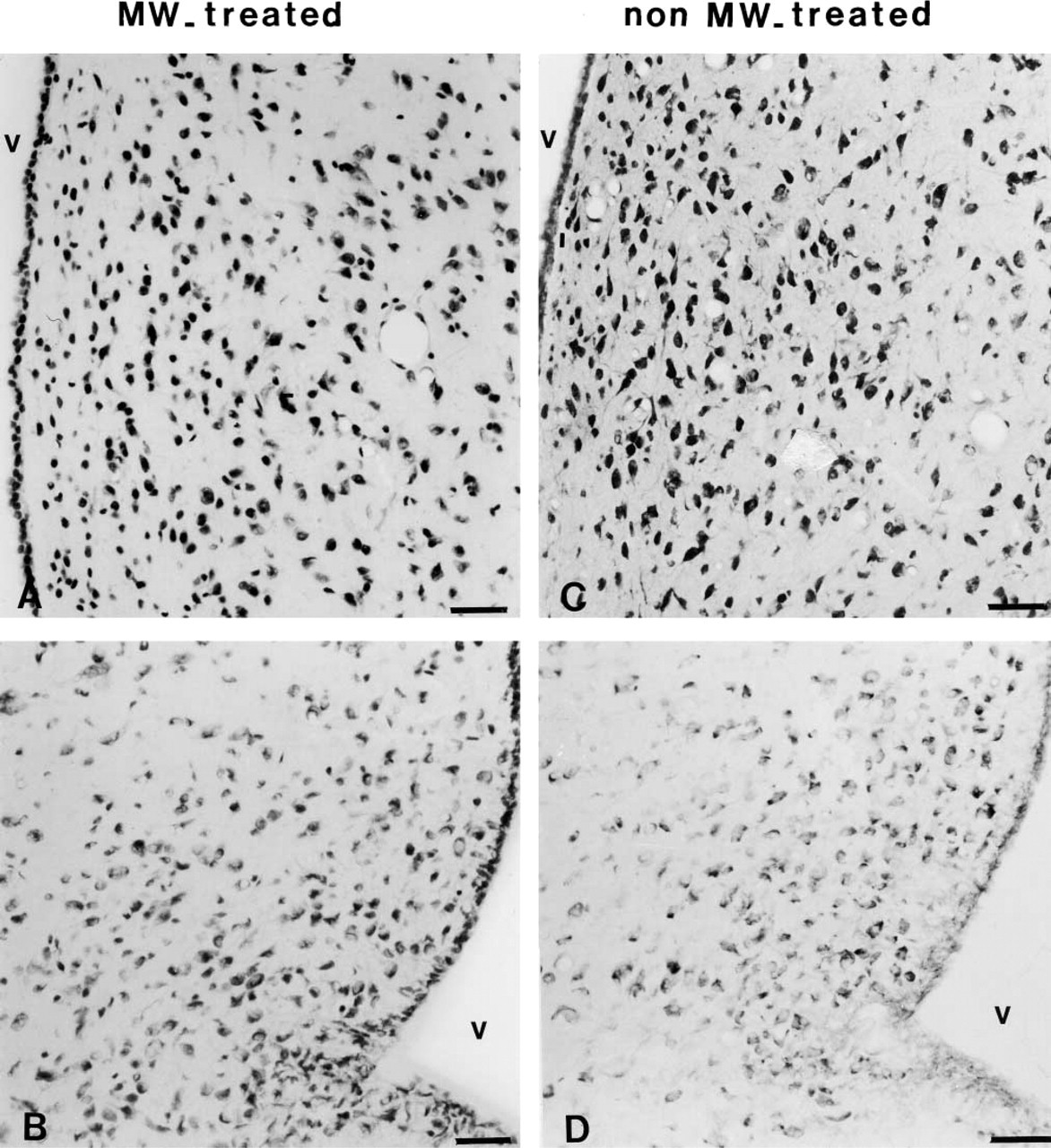
ISH was performed following our well-characterized routine procedure (Mitchell et al. 1997, 1999a,b; Bouret et al. 2000). Briefly, after proteinase K permeabilization, processed sections were hybridized with riboprobe–hybridization buffer mix, which contained the 35S-labeled Gal-R1 cRNA probe (30,000 cpm/μl). Overnight hybridization at 55C was followed by RNase treatment and a series of stringent washes, including a high-stringency wash at 60C. Hybridized slides were dehydrated in 70% and 100% ethanol in ammonium acetate and dipped in K5 emulsion (Integra Biosciences; Eaubonne, France). After a 30-day exposure, slides were developed. The slides were processed under the same conditions within the same experiment and the microphotographs were taken after the same exposure time to ensure an accurate comparison of the labeling signal. For qualitative histochemical assessment of the effects of MW treatment on tissue morphology, some sections that had undergone MW heating followed by ISH were stained with the Laskey's thionin solution. Tissue morphology was compared with sections receiving no pretreatment on Nissl-stained sections.
The controls for specificity of the probe have been previously checked (Mitchell et al. 1997, 1999a,b). Incubation of some sections with 35S-labeled sense probes, pretreatment with ribonuclease, and co-incubation with a 100-fold excess of unlabeled antisense probes gave no specific labeling.
Quantitative Analysis of ISH Labeling
All sections were processed together during hybridization and autoradiographic procedures. Quantitation of Gal-R1 signal intensity was performed by light microscopy under darkfield illumination to evaluate the pattern and intensity of the hybridization signal. The estimation of the Gal-R1-labeled cells was performed by using the azure blue nuclear stain. Analysis was conducted on the entire preoptic area and arcuate nucleus according to the Swanson rat brain atlas (plates 16–30). Four tissue sections per area of interest from each animal were analyzed (about 5000 cells for the preoptic area and 2000 for the arcuate nucleus). The quantification of the Gal-R1 grain density was performed using an image-analysis program (Densirag; Biocom, Les Ulis, France) interfaced to an Axiophot microscope (×60 epiillumination darkfield objective; Carl Zeiss, Göttingen, Germany) run on a PC computer with a high-resolution camera.
The mean density of grains per cell (± SEM) was calculated. Differences among levels of Gal-R1 receptor mRNA expression in the pretreated and control groups were assessed with a Student's t-test and were regarded as significant when p≤0.05.
Double Labeling for ER and Gal-R1
A group of sections was submitted to the MW treatment and was then processed for the double labeling ER and Gal-R1. After the IHC labeling for ER and the DAB reaction as described above, sections were rinsed in TBS and then processed for ISH labeling as also described for detection of Gal-R1 mRNA expression. After a 30-day exposure time, sections were revealed, fixed, and observed under the Axiophot microscope. The Gal-R1 signal intensity was quantified (see above) and the results were expressed as mean density of grains per cell ± SEM.
Results
Preservation of Tissue Morphology with MW Treatment
The Nissl staining showed characteristically intense dark blue nuclear staining of chromatin on a colorless background (Figure 1) in many cell bodies. MW pretreatment of sections that had undergone ISH for Gal-R1 had negligible effects on cell morphology (Figures 1A and 1B) in comparison with control sections without any pretreatment (Figures 1C and 1D). It demonstrated an acceptable architectural preservation of pretreated sections.
Effect of MW Treatment on IHC ER Staining
Under OVX conditions, MW irradiation before the IHC produced staining with the clone 1D5. The signal was intense in many cells of the preoptic area (Figure 2A) and in some nuclei of the arcuate nucleus (Figure 2C). Background was negligible. On sections not pretreated with MW, labeling for ER was undetectable in the two regions of interest (Figures 2B and 2D).
Specificity of Gal-R1 ISH on MW-treated tissue sections
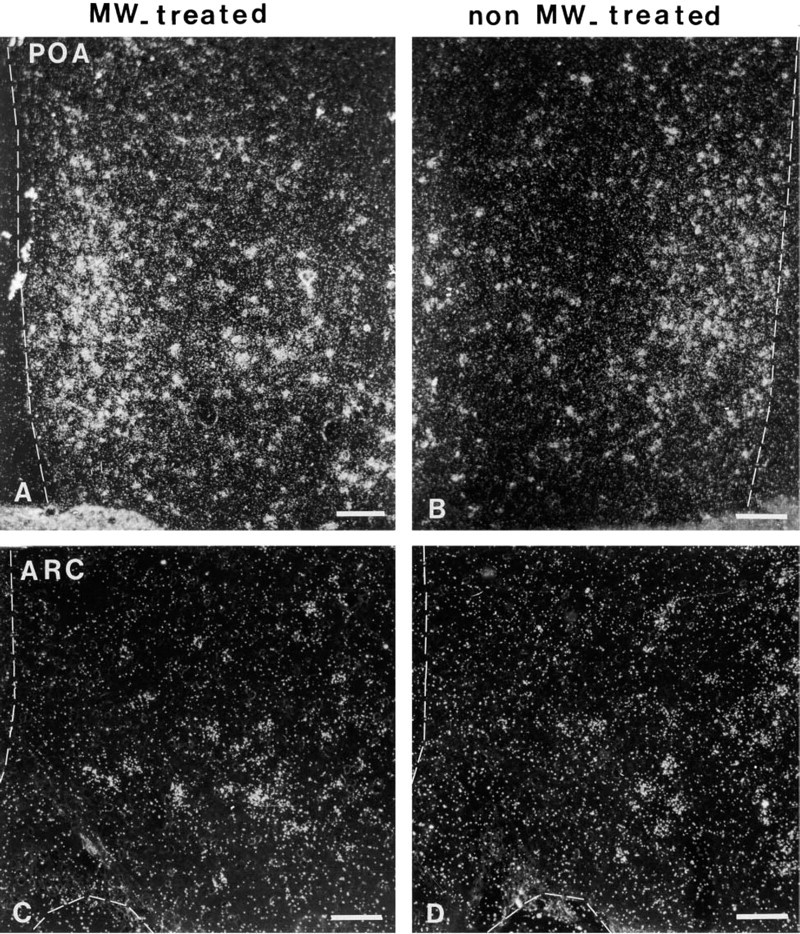
The localization of positive signal from MW-heated and control sections obtained with antisense Gal-R1 mRNAs was consistent with that previously published (Mitchell et al. 1997, 1999a,b; Faure-Virelizier et al. 1998). For example, a great many cells displaying intense labeling were observed in the preoptic region (Figures 3A and 3B) and in the paraventricular and ventromedial nuclei (not shown). The arcuate nucleus contained moderate numbers of labeled cells (Figures 3C and 3D).
The sense probe, which was used at equivalent specific activity in the same concentration as the antisense probe, gave no hybridization signal. RNase pretreatment completely abrogated hybridization signals obtained with the antisense probe.
Effect of MW Pretreatment on Hybridization Efficiency
The hybridization signal obtained from pretreated tissue sections with MW oven heating was compared to that obtained from control sections without pretreatment after a 30-day exposure (Figures 3 and 4). An important observation was that the background signal obtained after MW pretreatment was not significantly increased in comparison with that of control sections. In the preoptic area, all the slides heated by MW pretreatment showed increased hybridization signal compared with the control slides (Figures 3A, 3B, and 4A; 39.72 ± 1.97 vs 32.77 ± 1.23). This difference was significant (p < 0.05). In contrast, in the arcuate nucleus no significant difference was noted between the control group and that preheated (Figures 3C, 3D, and 4; 19.92 ± 1.21 vs 22.21 ± 1.72; p < 0.05).
Photomicrographs demonstrating the effect of MW treatment (
Pairs of consecutive frozen sections of paraformaldehyde-fixed rat preoptic region immunostained for ER without (
Darkfield photomicrographs of the expression of Gal-R1 mRNA in the preoptic area (POA;
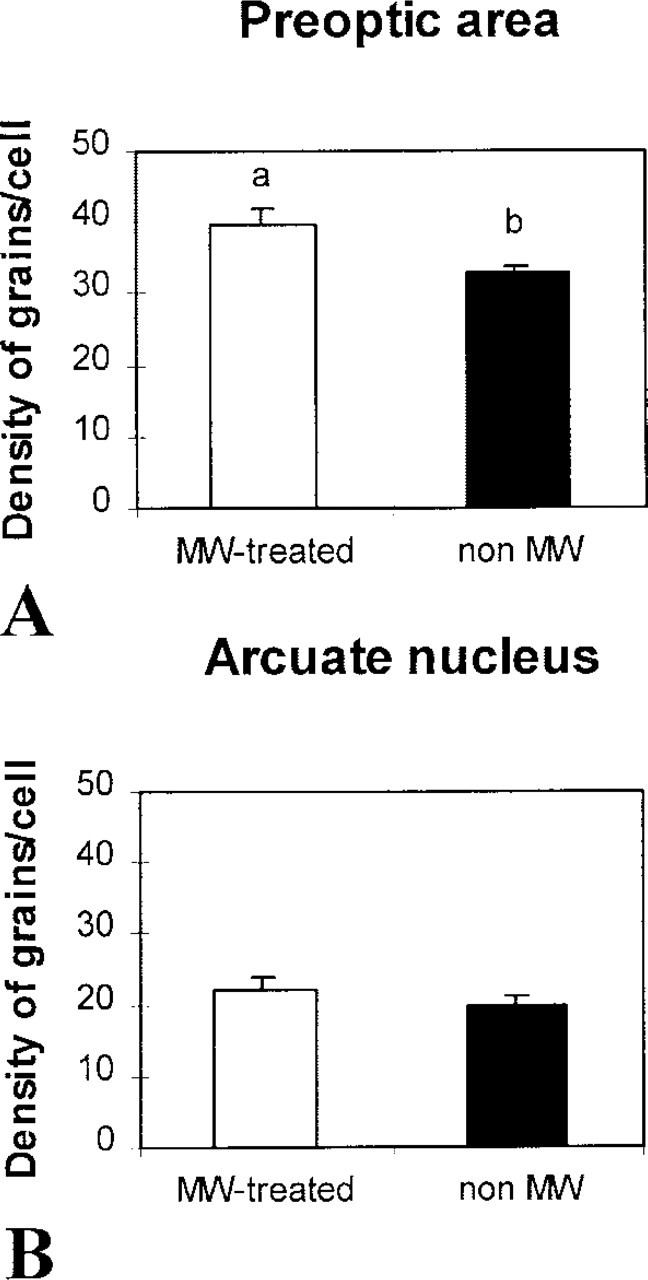
Effects of MW pretreatment on the signal intensity of Gal-R1 mRNA expression in the preoptic area and arcuate nucleus. The values are the mean ± SEM. Significant differences (p < 0.05) among the values are noted (a vs b).
MW Pretreatment Improves Double-labeling Efficiency
As illustrated in Figure 5, MW-pretreated double-labeled sections for ER and Gal-R1 exhibited three populations of labeled cells: ER-immunostained nuclei, Gal-R1 mRNA-expressing cells, and double-labeled ER-Gal-R1 cells. In the preoptic area, the double labeling concerned many cells, as shown in Figure 5A, whereas ER-Gal-R1 cells were sparse in the arcuate nucleus (Figure 5B). The mean optical density for Gal-R1 mRNA as quantified in double-labeled cells of the preoptic area was 26.26 ± 0.47.
Discussion
Our results first confirm that, as in other tissues, the MW procedure before the IHC is very favorable to obtain staining with the ER antibody from fixed frozen sections of hypothalamus. Second, this work demonstrates that the MW preheating before the ISH improves the sensitivity of Gal-R1 expression in the preoptic area. Finally, we show that MW treatment is helpful for double-labeling techniques that originally associate the simultaneous detection of steroid receptor by IHC and neuropeptide receptor mRNA by ISH from frozen brain sections.
Since the initial description of the MW procedure by Mayers (1970), there has been considerable interest in this heating pretreatment, which appears to enhance either the immunostaining of some antigens or the ISH signal, in research and routine histopathological laboratories. However, in contrast with most previous reports, our aim in ISH is not to reduce the hybridization time nor to modify our ISH protocol, i.e., proteinase K permeabilization or paraformaldehyde postfixation. This is exclusively used to obtain better efficiency for the double-labeling IHC and ISH. To date, the MW procedure has been used for single labeling performed on serial sections for comparison of neuronal nitric oxide synthase protein or mRNAs (Garcia-Vitoria et al. 2000). As far as we know, the method reported here is the only one that applies MW oven heating for detection of neuropeptide receptor from brain tissue with a radioactive RNA probe. Only a few works have studied the effect of MW heating on the hybridization signal obtained with a peptide receptor in peripheral tissues (Sibony et al. 1995). In addition, our results clearly demonstrate the advantages of MW in detection of two receptors on the same section in the central nervous system. It is also important to note that MW pretreatment does not alter morphological preservation, as already observed at both light (Boon et al. 1988; Login and Dvorak 1988) and electron microscopic levels (Login and Dvorak 1985; Naganuma et al. 1990).
Our results show an analogous effect of enhancement of the ISH autoradiographic signal composed to that previously observed in the literature (Sibony et al. 1995; Oliver et al. 1997). Previous reports demonstrated that MW heating enhanced the ISH signal from paraffin-embedded (Sibony et al. 1995; Sperry et al. 1996; Wilkens et al. 1996; Oliver et al. 1997) or frozen sections (Slayden et al. 1995; Lan et al. 1996), in various tissues. Several studies have employed oligonucleotides radioactively or digoxigenin-labeled. These short-length DNA probes are comparatively less sensitive than RNA probes that usually do not necessitate MW pretreatment. However, even for RNA probes, MW pretreatment can be necessary for enhancing the signal for detection of low copies of mRNA leading to insufficient signal intensity, as is often the case for detection of receptor mRNAs. Because the MW pretreatment could convey a risk for deactivation of some molecules in the tissue, it has been necessary to apply this protocol in a first step on the signal expression for galanin receptor before the double-labeling. Interestingly, MW pretreatment enhanced the efficiency of the ISH signal in the preoptic area and had no major effect on signal intensity in the arcuate nucleus compared to the preoptic area. The effect of heating appears to depend on the region of the central nervous system, perhaps due to the heterogeneity in mRNA expression of neuropeptides and receptors in the different hypothalamic areas. In contrast, MW pretreatment improved the ER immunostaining, enhancing the intensity of the immunostaining both in the preoptic region and the arcuate nucleus, thus unmasking the immunoreactivity in almost all positive cell types. As previously reported (Leong and Milios 1993), clone 1D5 is effective only on sections subjected to MW irradiation in citrate buffer before immunostaining. The intensity of the reaction product was excellent and background staining was minimal. Altogether, these results allowed easy interpretation for the observation of double-labeled cells, and their numbers can be evaluated easily on a percentage basis. Moreover, a quantitative analysis of Gal-R1 mRNA expression can be performed in ER-immunoreactive cells. More generally, this type of double labeling is very interesting in neuroendocrinology because it can answer a question asked by many laboratories, i.e., the direct action or not of estrogen on expression of peptide receptors in a neuron.
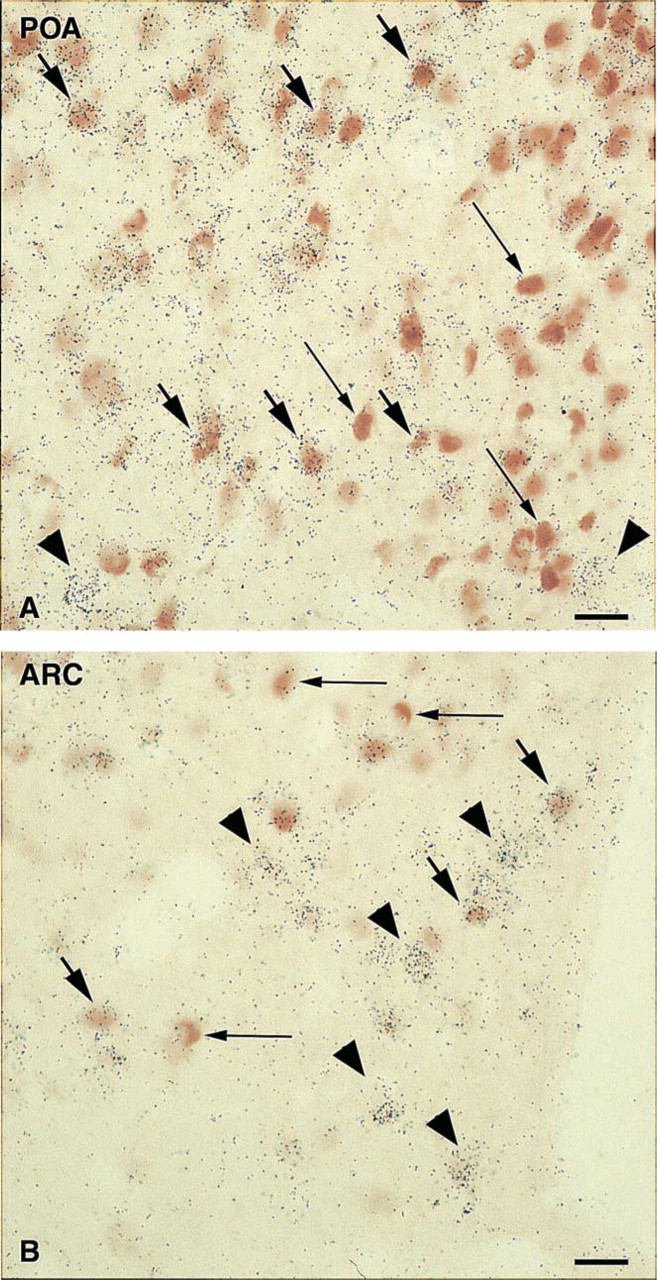
Double-labeling staining for ER and Gal-R1 after MW treatment in the preoptic area (
The mechanisms of enhancement of the IHC or ISH signal had previously been speculated on and discussed in the literature: better penetration of the probe to the target by unmasking the crosslinking effects of formaldehyde fixation and denaturation of protein and nucleic acid structure. In IHC, when MW heating is performed on unfixed fresh tissue, this pretreatment appears to preserve antigens in sections by increasing their resistance to extraction during subsequent aqueous fixation and IHC processing (Slayden et al. 1995). In ISH, the results showed that there was no loss of target mRNA after MW treatment because there was an enhancement of the signal expression. This observation is in favor of increasing the probe's ability to bind to the mRNA target by facilitating this interaction, which is the most common explanation.
In conclusion, MW heating demonstrates its efficiency for hybridization labeling of galanin receptor in frozen brain tissue. This is applied to the double labeling by IHC and ISH techniques on the same section and thus may help detect low levels of mRNA together with difficult detection of nuclear antigens. This protocol appears particularly suitable for simultaneous evaluation of the expression of two receptor states. This study provides evidence of a distinct expression of estrogen receptor in Gal-R1 cell populations of the preoptic area and arcuate nucleus, and of the fact that estrogens may target its α-type receptor in populations expressing receptors for neuropeptides of hypothalamic regions involved in reproduction.
