Abstract
Kalirin, a homologue of trio and UNC-73, has been previously demonstrated to cause cytoskeletal rearrangements, enhanced outgrowth of neuritic processes, and altered secretion. In the adult rat, kalirin is specifically localized to the central nervous system, with the main adult isoform, kalirin-7, concentrated in neuronal postsynaptic densities. In this study we examined the expression of kalirin in rat tissue from embryonic Day 10 (E10) through E18, using an antibody that detects all known kalirin isoforms. Kalirin expression in the embryo was more widespread than in the adult, with localization of kalirin protein to both neuronal and non-neuronal tissue, such as muscle, lung, intestinal epithelium, and pancreas. In neurons, kalirin was localized both in cell bodies and axon processes; in muscle tissue, kalirin was highly localized to migrating myogenic cells and at muscle attachment sites. Western blotting analysis indicated that kalirin-7, the major adult isoform, was a minor component of embryonic kalirin; the main isoform expressed in the embryo was kalirin-9. This is the first identification of kalirin expression in embryonic tissue and the first demonstration of non-neuronal expression of kalirin.
P
Kalirin was identified through a screen for interactors with the cytosolic domain of peptidylglycine α-amidating mono-oxygenase (PAM), a membrane enzyme essential to the synthesis of many bioactive peptides. PAM functions in large dense-core vesicles, and mutations in its cytosolic domain cause its mislocalization without altering enzyme activity (Milgram et al. 1996). Using the cytosolic domain of PAM as bait and a hippocampal library as prey, a yeast two-hybrid screen identified three
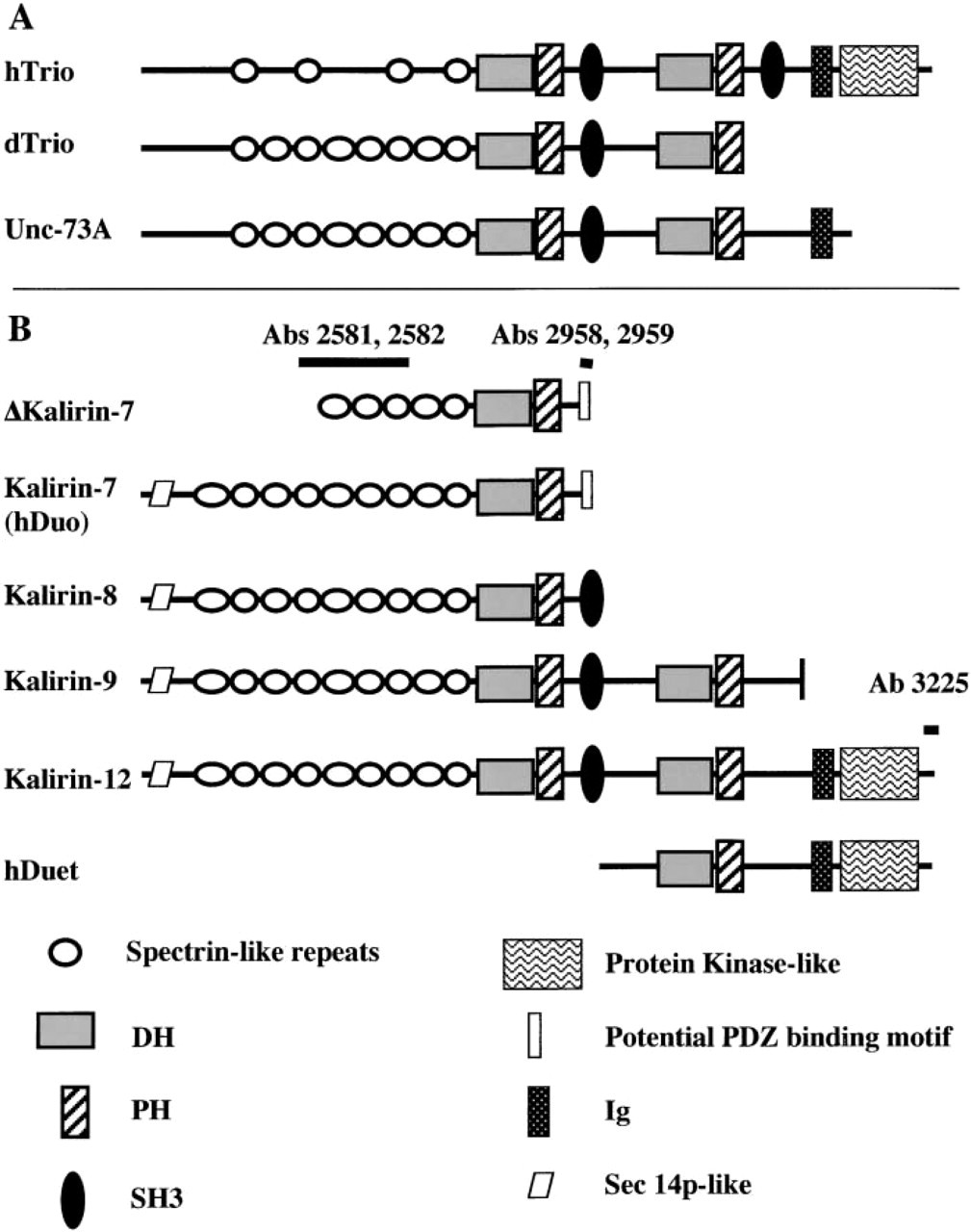
Schematic of kalirin isoforms and homologous proteins. (
The kalirin proteins are heterogeneous (Figure 1; Penzes et al. 2000a; Johnson et al. 2000). Alternative 3′-splicing generates several major kalirin isoforms including kalirin-7, -8, -9, and -12 (named for the approximate size, in kb, of each transcript). All share nine spectrin-like repeats, which are implicated in coupling membrane proteins to the cytoskeleton (Bennett and Gilligan 1993). Kalirin-8, the first kalirin transcript identified, is a minor isoform in adult rat brain (Alam et al. 1997; Johnson et al. 2000). Only kalirin-9 and kalirin-12 contain multiple GEF domains. Three different short NH2-termini, perhaps reflecting alternative 5′-splicing, have been identified and Δkalirin-7 is generated through use of an internal translational start site. Kalirin-7 contains a PDZ-binding motif, which enables this isoform to localize to the postsynaptic density fraction (Penzes et al. 2000a). The Srchomology 3 domain (SH3) present in kalirin -8, -9, and -12 may promote formation of protein–protein complexes (Shaw 1996). Kalirin-12 and human trio contain a protein kinase-like domain, although its function is unknown. dTrio and UNC-73 lack a protein kinase domain.
Overexpression of kalirin-7 in hippocampal neurons leads to the formation of more dendritic spinelike structures and abnormal spine morphology (Penzes et al. 2001). Expressed independently, the first and second DH domains of kalirin have uniquely different effects on transfected neurons (Penzes et al. 2000b). Neither DH domain alone replicates the effect of dual function kalirin. The first DH domain of kalirin is a GEF for Rac 1, but not for Rho A or Cdc42 (Alam et al. 1997; Penzes et al. 2000b). The second DH domain binds both nucleotide-depleted Rac1 and Rho A but is not an exchange factor for Rac1 (Penzes et al. 2000b).
The functions of kalirin in development and adulthood are not yet clear, although the unique subcellular localization of different isoforms suggests form-specific functions (Johnson et al. 2000). In ACTH-producing corticotrope tumor cells, expression of kalirin-8 increases the rate at which newly synthesized PAM is transported through the early secretory pathway, increases ACTH secretion, and causes development of longer and more highly branched neuritic processes (Alam et al. 1997). These data suggest a role for kalirin in coordinating membrane flux for secretion and neurite outgrowth. Furthermore, kalirin-8 expression in corticotropes and Chinese hamster ovary cells induces formation of filamentous actin and spike-like protrusions of the cell membrane. Kalirin's ability to affect cytoskeletal rearrangement and regulated secretion may be key steps in its physiological function (Mains et al. 1999). Genetic studies in Drosophila implicate the Rac/Cdc42-activated serine/threonine protein kinase Pak1 and the Abl tyrosine kinase in the dTrio pathway that affects axon guidance (Winkelmann et al. 1990; Bloom et al. 1993; Debant et al. 1996; Colomer et al. 1997; Steven et al. 1998; Liebl et al. 2000).
To investigate the potential roles of kalirin during embryonic development, we evaluated kalirin protein expression in the embryonic rat using immunohistochemistry and Western blotting analysis. We found that kalirin expression in the rat embryo is predominantly restricted to kalirin-9, which is not the major form of kalirin in the adult cortex. Kalirin-12 expression is readily detected, whereas kalirin-7 expression is not detectable during embryonic development. In contrast to the adult, kalirin expression during embryonic development is not restricted to the nervous system. On the bassis of its sites of expression, kalirin may play a role in axon outgrowth, myoblast migration and fusion, and endocrine development.
Materials and Methods
Animal Preparation
All experimental protocols were approved by the Johns Hopkins University Institutional Animal Care and Use Committee, and all applicable guidelines from the National Institutes of Health Guide for the Care and Use of Laboratory Animals were followed. Timed pregnant Sprague–Dawley rats were obtained from Harlan (Indianapolis, IN). Embryonic Day 0 is defined as the date of conception. For preparation of tissue extracts, timed pregnant female rats were anesthetized with 400 mg/kg chloral hydrate, the embryos removed, washed in PBS, placed in MEM-AIR, and dissected using a dissecting microscope. Tissue was extracted with 20 mM PIPES, pH 6.8, 2 mM Na2EDTA, 50 mM NaF, 10 mM Na4P2O7, 1 mM Na3VO4, 1% Triton X-100, 300 μg/ml phenylmethanesulfonyl fluoride, 2 μg/ml leupeptin, 16 μg/ml benzamidine, 10 μg/ml lima bean trypsin inhibitor, and 2 μg/ml pepstatin, homogenized, and centrifuged for 15 min in a microfuge at 4C. Supernatant was collected and assayed for protein concentration using the bicinchoninic acid protein reagent kit (Pierce Chemical; Rockford, IL).
Antibodies and Antibody Purification
Rabbit polyclonal antibodies to the COOH-terminus of kalirin-12 (Ab3225 and Ab3226) were affinity-purified (Johnson et al. 2000). Peptide [2 mg rat kalirin(Pro2943–Thr2959); synthesized by Dr. Henry Keutmann (Endocrine Unit, Massachusetts General Hospital)] was coupled to 2 ml Affi-Gel 10 resin (Bio-Rad; Hercules, CA) for 2 hr at room temperature (RT), blocked for 1 hr with 0.1 M ethanolamine HCl, pH 8.0, and washed three times in 0.1 M NaOAc/1 M NaCl, pH 4.0, followed by 0.1 M Tris HCl/1 M NaCl, pH 8.0. Before application to the resin, immunoglobulin was enriched by precipitation with 45% ammonium sulfate. The precipitate was solubilized and dialyzed into 100 mM NaPi, pH 7.4, bound to affinity resin (1 ml serum/1.5 ml resin) for 3 hr at 4C, washed, and antibody was eluted with 0.2 M glycine/0.1 M NaCl/0.1% Triton X-100, pH 2.3. Affinity-purified antibody was dialyzed overnight against 100 mM NaPi, pH 7.4, and overall recovery of binding activity, quantified by an ELISA using alkaline phosphatase-conjugated second antibody, was greater than 50%. Affinity-purified Ab3225 was diluted 1:100 for use. Only four of the 17 residues in the kalirin peptide used to generate Abs3225/3226 are common to hTrio; when tested on recombinant trio, neither Ab3225 nor Ab3226 showed any crossreactivity. Rabbit polyclonal Abs2581 and 2582 were generated against spectrin-like repeats 4–7 [rKalirin (517–976)]; Ab 2581 was diluted 1:500 for use (Penzes et al. 2000a). When tested using Gal-4 fusion proteins, neither kalirin-spectrin antibody crossreacted with the corresponding region trio (Penzes et al. 2000a). Antibody blocking experiments were performed by incubating antibody in 100 μg/ml of the appropriate antigen for 1 hr in PBS before slide incubation. Rabbit polyclonal antisera Ab2958 and Ab2959 to the unique COOH-terminus of kalirin-7 were affinity-purified as described (Penzes et al. 2000a). Recovery of binding activity was typically about 50%. Trio contains no sequence homologous to the COOH-terminus of kalirin-7, eliminating concerns about crossreactivity.
Immunohistochemistry
Embryos fixed in 4% paraformaldehyde, embedded in paraffin, and sectioned at 12 μm were obtained from Novagen (Madison, WI). Slides were washed in xylene to remove paraffin, then rehydrated through a series of graded ethanol solutions from 100% to 50%. Slides were microwaved for 15 min, cooled, and postfixed in Bouin's fixative for 15 min. Tissue was washed several times in PBS to remove fixative, incubated in PBS for 15 min, and permeabilized in 0.1% Triton X-100 in PBS for 30 min. Slides were washed twice in PBS for 5 min each, blocked with 4% normal goat serum (Vector Laboratories; Burlingame, CA) for 1 hr at RT, and incubated with primary antibody diluted in PBS at 4C overnight. The following day, slides were washed twice in PBS for 5 min each, incubated in Vectastain biotinylated antibody (1:1000; Vector Laboratories) for 30 min, washed twice in PBS, quenched with 0.5% H2O2 in PBS for 10 min, washed twice in PBS, and incubated in the BC reagent for 30 min. Finally, slides were rinsed in PBS and developed using 3,3′-diaminobenzidine tetrahydrochloride (Gibco BRL; Gaithersburg, MD) in 50 mM Tris, pH 7.4. Slides were viewed using a Zeiss Axioskop. Photography was performed using the Spot Camera (Diagnostic Instruments; Sterling Heights, MI).
Western Blotting
Samples from tissue extracts were prepared for electrophoresis by making them 2% in SDS and 5% in 2-mercaptoethanol and boiling for 5 min. Proteins were fractionated on SDS-polyacrylamide gels containing 6% acrylamide, 0.19% bis-acrylamide. Proteins were transferred to Immobilon-P membranes (Millipore; Bedford, MA) in 25 mM Tris-HCl (pH 8.5), 200 mM glycine, 20% methanol for 2 hr at 500 mA. Blots were blocked with 5% skim milk diluted in 50 mM Tris-HCl (pH 7.5) and 150 mM NaCl containing 0.05% Tween-20 (TTBS), incubated in a 1:1000 dilution of rabbit antiserum (Ab2581 or Ab3225) for 2 hr at RT, and rinsed. Blots were then incubated for 1 hr with HRP-conjugated donkey anti-rabbit IgG antibody (1:10,000; Amersham, Piscataway, NJ) and visualized using the Enhanced Chemiluminescence Kit (Amersham). Exposure times ranged from 30 sec to 10 min.
Results
Kalirin-9 Is the Major Embryonic Isoform of Kalirin
Kalirin protein expression in E18 embryonic cortex was examined by Western blotting analysis to determine the major kalirin isoforms expressed (Figure 2A). Antibodies against spectrin-like repeats 4 through 7 were used because they recognize all forms of the kalirin protein except Duet, and adult cortex was analyzed for comparison (Figure 2A). The E18 cortex demonstrates predominant expression of kalirin-9, with readily detectable amounts of kalirin-12; the minor band at approximately 200 kD is thought to represent kalirin-8 (Figure 2A). Essentially identical patterns were obtained with two different kalirin-spectrin antisera (Ab2581 and Ab2582). A protein the size of kalirin-7, the major isoform in adult cortex, was not detected in E18 cortex (Figure 2A). Western blotting analysis of E18 and adult cortex using a kalirin-7-specific antibody verified kalirin-7 protein expression only in the adult CNS (Figure 2B, Ab2959).
We next examined extracts prepared from other E18 tissues, including the heart, lung, kidney/adrenal, intestine, tongue, and liver, for kalirin expression. Kalirin-9 represents the major isoform of kalirin in these tissues (Figure 2C). Although proteins the size of kalirin-8 and kalirin-12 are detectable, levels are generally lower than levels of kalirin-9. Kalirin expression was detectable only at very low levels in the embryonic liver. The amounts of kalirin-9 and kalirin-12 detected in the other tissues examined were quite similar. A very similar pattern was observed using both kalirin–spectrin antisera. Preincubation of Ab2581 with excess antigen demonstrated the specificity of the signal observed.
Kalirin Is Expressed Early in Embryonic Development (E10)
Using an antibody to the spectrin-like regions of kalirin, which identifies the major kalirin isoforms, we next performed immunohistochemistry on developing rat embryos to determine the major sites of kalirin expression. By E10 in the rat, the neural tube and rudimentary heart have formed (Kaufman 1992). At this age we detected expression of kalirin protein only in the developing heart, including the common atrium and common ventricle, but not in the enclosed blood cells (Figures 3A and 3B). In addition, the surrounding embryonic sac appeared immunopositive (Figure 3A, upper right corner). Kalirin protein was not detectable in the neural tube at this time (Figure 3C). Immunodepleted Ab2581 yielded no staining and Ab2582 demonstrated immunoreactivity similar to that of Ab2581 (not shown).
Kalirin Becomes Widely Expressed in Developing Neural and Muscle Tissue by E12
By E12, kalirin protein expression expands to include not only the heart but also multiple nervous system tissues and newly formed, migrating myogenic cells (Figure 4A). Kalirin expression remains elevated in the cardiac region (H), similar to the pattern observed at E10, with the atrium, ventricle, and bulbus cordis demonstrating immunoreactivity. In addition to the heart, at E12 kalirin is uniformly expressed in the newly formed myogenic cells of the tongue (Tn; Figures 4A and 4B). These myogenic cells are first identifiable as an outgrowth from the first five or six somites of the murine embryo and migrate along the hypoglossal cord before eventually taking their place in the tongue, which develops from the mandibular component (Figure 4A, M; Dietrich et al. 1998; Brand–Saberi and Christ 1999). Interestingly, kalirin expression is also identifiable in migrating hypaxial myogenic cells (Brohmann et al. 2000), although the levels are significantly lower than in the hypoglossal cord.
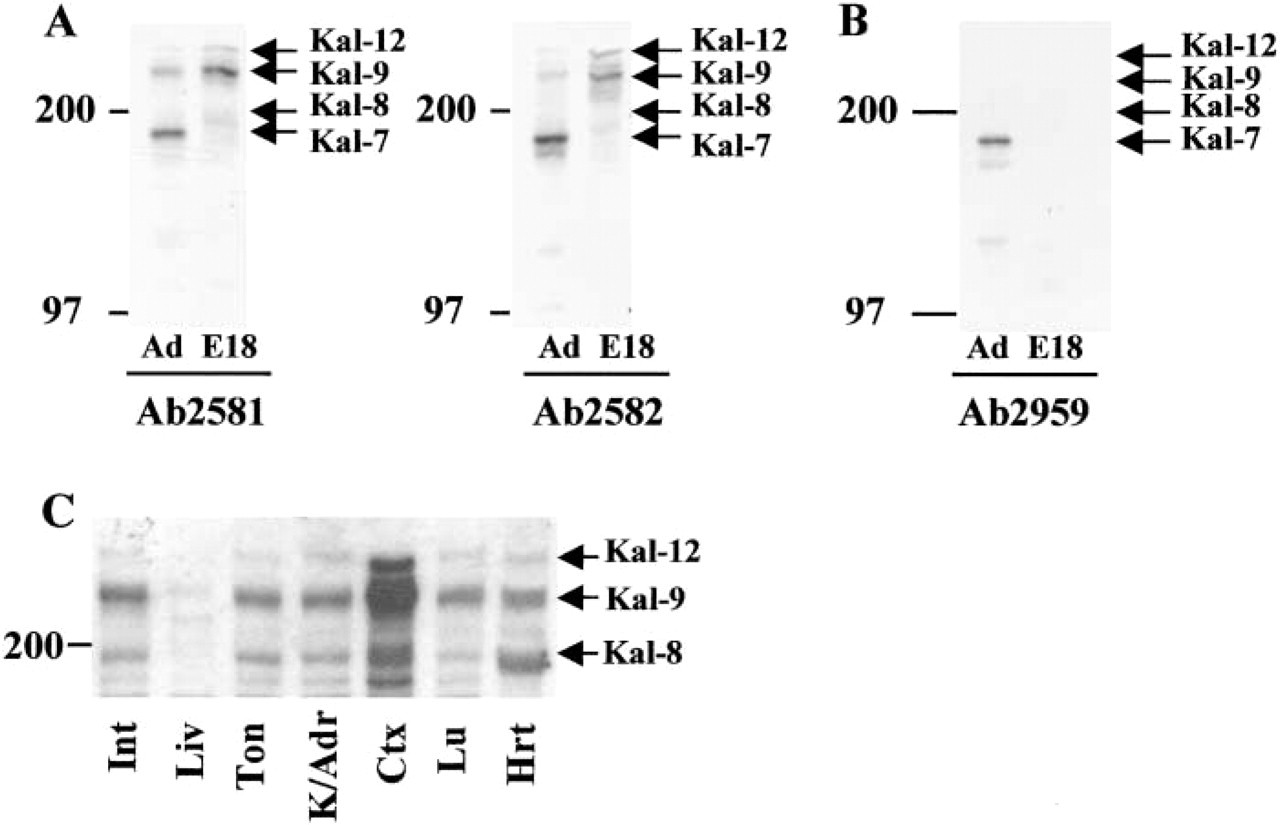
Western blots identifying kalirin isoforms in the developing embryo. The 200-kD and 97-kD molecular weight markers are shown. All samples analyzed contained 15 μg total protein; equal loading was verified by staining the membranes. (
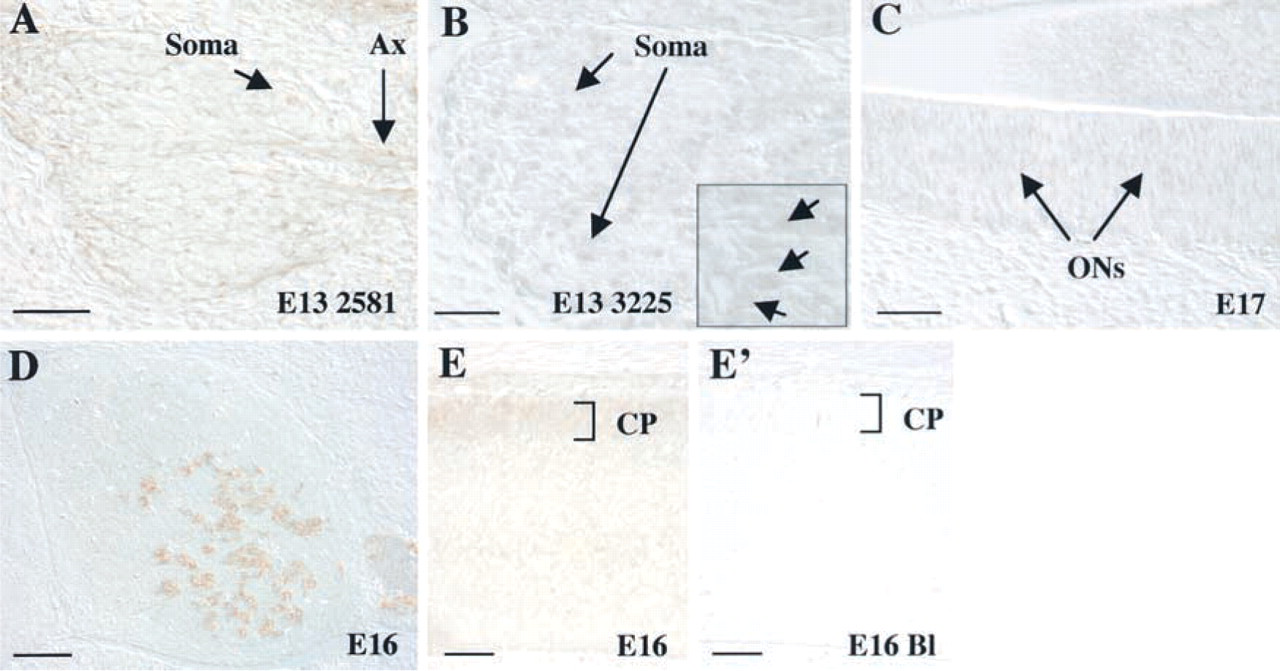
At E12, many regions of the nervous system begin to differentiate (Kaufman 1992). Embryonic events include increased differentiation of the telencephalic vesicle, closure of the caudal neuropore, and appearance of the cranial nerve ganglia. Kalirin expression is evident in the dorsal root ganglia (DRG) along the entire extent of the spinal cord (Figure 4A). Specifically, kalirin protein is detected in both the cell bodies (Soma) in the DRG and in the developing axons (Ax) (Figure 4C). At E12, kalirin immunoreactivity is detectable in the developing cortex (Ctx), both in cell bodies and processes (Figures 4D and 4E).
Interestingly, kalirin expression in not detectable in the E12 olfactory placode (OP), the earliest structure formed during development of the olfactory epithelium (Morrison and Moran 1995). The olfactory placode at E12 demonstrates high levels of neuronal proliferation, with very little differentiation. These findings suggest that kalirin may be functionally relevant during early neuronal and muscle differentiation events rather than during proliferative events.
Kalirin Demonstrates Distinct Cortical, Muscle, and Olfactory Neuron Expression at E15 and E16
In the rat nervous system, differentiation continues through E15 and E16, with expansion of cortical volume and increased differentiation of the olfactory lobes and cerebellar region (Kaufman 1992). Kalirin protein expression remains elevated in the developing nervous system and is readily apparent in the cell bodies of the outer layer of the cortex (Figure 5A). This layer of the cortex, called the cortical plate (CP), is composed of newly formed neurons generated by the “inside-out” development of the cortex (Angevine and Sidman 1961), and represents neurons at early stages of differentiation. At the higher magnification shown in the inset in Figure 5A′, cell soma staining is apparent, with the clear area indicating a lack of nuclear staining. Kalirin expression is detectable at low levels in cortical axons. In addition, kalirin expression is evident in the choroid epithelial cells of the choroid plexus (Figure 5B).
Kalirin immunoreactivity becomes apparent at this age in the developing olfactory epithelium. At this stage in olfactory neuron development, proliferation has slowed and a majority of neurons present begin to differentiate (Cuschieri and Bannister 1975; Farbman 1992). Olfactory differentiation involves axon outgrowth from newly-born olfactory receptor neurons through the underlying basal lamina to the forming olfactory lobes. Kalirin immunoreactivity in the olfactory neurons (ON) is detectable in both the cell bodies and the developing axonal processes (Ax; Figure 5C). Kalirin expression in both the outer layer of cortex and the differentiating olfactory neurons appears to parallel the onset of neuronal differentiation as seen at earlier stages.
By E15/E16, many of the rudimentary muscle structures have formed after myogenic cell migration (Kaufman 1992), and kalirin staining is localized to a number of muscular structures, including the heart (not shown), intercostal muscles, and glossopharyngeal muscles, (Figures 5D, 5D′, and 5E). Although kalirin expression is still evident in the glossopharyngeal muscles, the kalirin protein is now concentrated at the tips of the muscle fibers rather than throughout the fibers as demonstrated at earlier stages (Figures 5E, 5E′, and 5E″; arrows; compare to Figure 4B). In addition, kalirin protein is expressed in a similar tip-staining pattern in the intercostal muscles (Figures 5D and 5D′). This tip region of the muscle may correspond to muscle–muscle attachment sites or muscle–bone/tendon attachment sites.
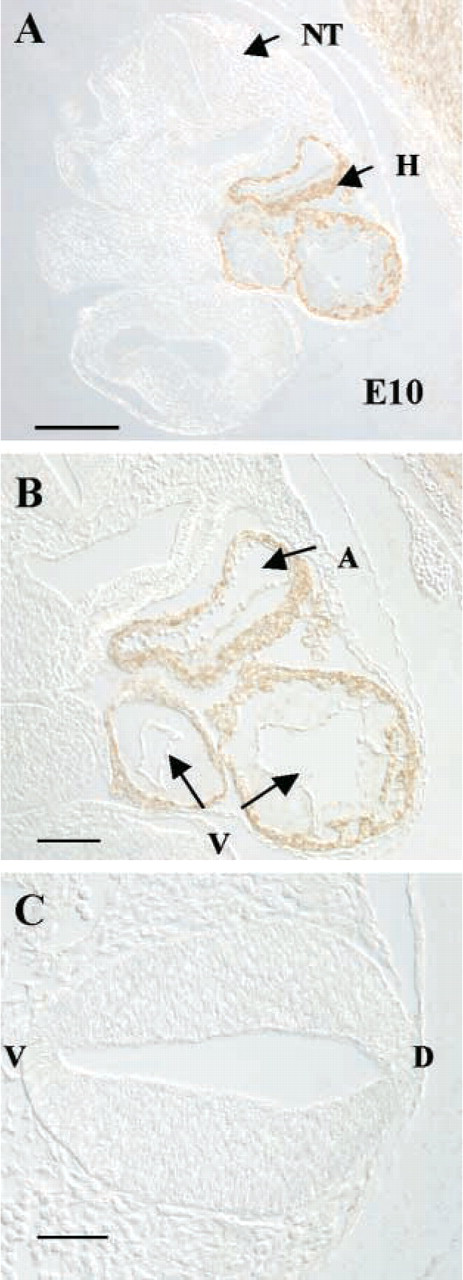
Kalirin expression at E10. Coronal sections through the developing embryo were used. (
To evaluate the specificity of the staining observed using the kalirin–spectrin domain antibody, blocking experiments were performed. Antigen-blocked antibody was unable to recognize kalirin protein in neural tissue (Figure 5F) or in muscle (Figure 5G), although nonspecific staining was evident in the overlying epidermis, which was evident with all antibodies tested. This nonspecific staining may reflect the increased friability of this tissue with processing.
Differential Expression of Kalirin at E18
A dramatic shift in kalirin expression is apparent between E15/E16 and E17/E18. Although the cortex is undergoing continued differentiation at E17, kalirin immunoreactivity in the outer layer of the cortex is decreased, with only a subset of neurons containing detectable levels of kalirin (Figures 6A and 6A′). In addition, there is an increased level of kalirin protein in axon bundles (Figure 6A″). Kalirin expression in a variety of muscles, including the glossopharyngeal and intercostal muscles, has decreased at E17/E18 in comparison to E15/E16 (data not shown).
Kalirin immunoreactivity becomes evident at this age in a number of peripheral tissues, including the intestine (Figures 6B and 6B′), the pancreas (Figure 6C), and the vibrissae (Figures 6D and 6D′). These tissues undergo increased expansion and differentiation at this developmental stage (Kaufman 1992). In the intestine, kalirin protein is expressed in widely scattered cells lining the intestinal lumen (Figure 6B′, arrows and inset). Staining fills the cytosol, with no staining in the nucleus (Figure 6B′ and inset). In the pancreas, kalirin appears in the majority of cells in the developing islets of Langerhans (Figure 6C). Kalirin expression is not detectable in the cells of the exocrine pancreas. Kalirin expression in the vibrissae is localized to the base of the developing hair follicle (HF), with highest expression in the dermal papilla (DP) and lower expression in the root sheath (RS; Figures 6D and 6D′).
Kalirin-12 Exhibits a Distinct Localization Pattern in the Developing Rat Embryo
We examined the expression of one of the embryonic isoforms of kalirin, kalirin-12, in the developing embryo using antiserum specific for the C-terminus of kalirin-12 (Figure 1). At E10, kalirin-12 expression was demonstrated in the embryonic heart, in both the common atrium and the ventricle, and staining was eliminated by immunodepleting the antiserum (data not shown). At later stages of development, kalirin-12 expression was detected in the DRG (Figures 7A and 7B), olfactory neurons (Figure 7C), adrenal medulla (Figure 7D), and developing cortex (Figure 7E). Antibody specificity was established by peptide blocking experiments, which completely eliminated binding to tissue (Figure 7E).
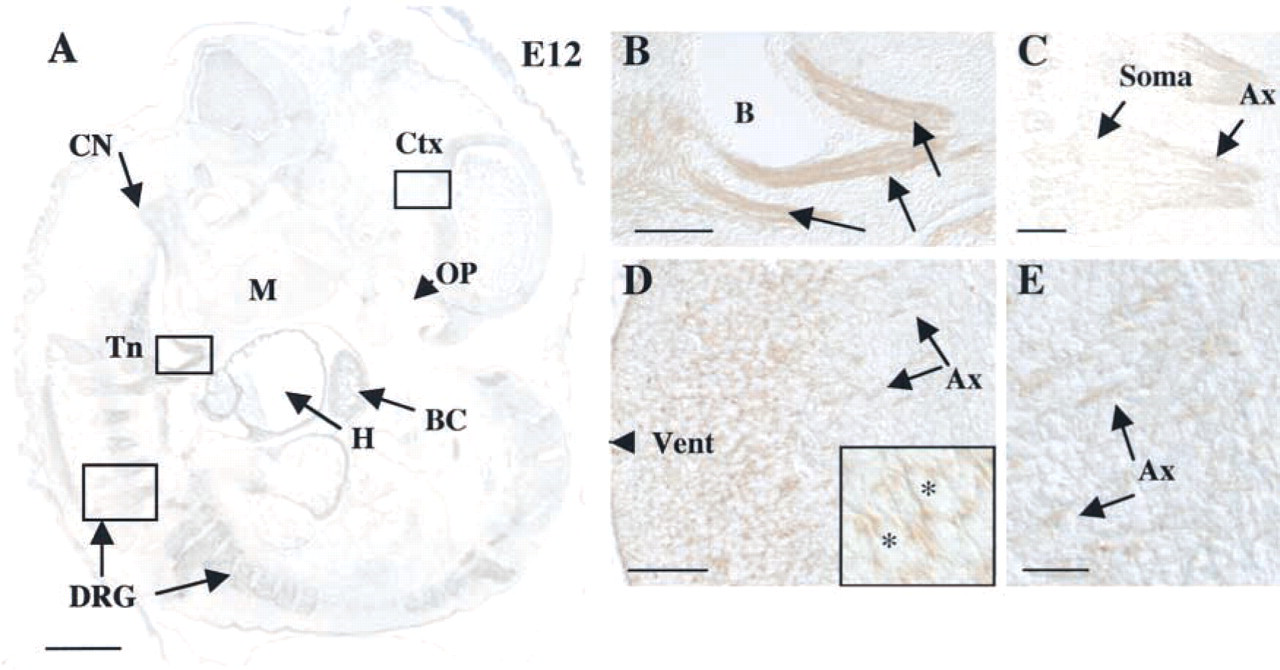
Kalirin expression in nerve and muscle at E12. (
The tissue expression pattern of kalirin-12 parallels that demonstrated previously with the antibody that recognizes all forms of kalirin and agrees with the results of Western blotting analysis. Kalirin-12, however, exhibits a unique localization within cells that express it, occupying only a subset of the sites visualized with the kalirin–spectrin antibody. In neurons, kalirin-12 is specifically localized to cell bodies rather than to axon processes (compare Figure 7A with Figure 7B, and Figure 7C with Figure 4B). The axonal immunostaining demonstrated with the kalirin–spectrin domain antibody presumably reflects kalirin-9, which is the most prevalent isoform, in the processes. Kalirin-9, with only five unique amino acids at its COOH-terminus, is difficult to visualize specifically.
Discussion
Kalirin Expression Correlates with Early Neuronal Development, Including Differentiation and Axon Outgrowth
Kalirin is widely expressed in the developing rat nervous system, with high levels of protein in the embryonic cortex, olfactory neurons, and dorsal root ganglia (Figure 8). Interestingly, kalirin expression in the nervous system is first detectable at E12 and appears to parallel neuronal differentiation rather than neuronal proliferation. Olfactory neurons, for example, begin to proliferate at E12, differentiate by E15/E16, and form connections to the developing olfactory bulb by E18 (Farbman 1992). Kalirin expression is not detectable in the embryonic olfactory epithelium until E15/E16, when most olfactory neurons are differentiating and extending axons. Similar patterns of expression were identified in the developing cortex and dorsal root ganglia. Expression of the trio gene in Drosophila is also associated with axon extension (Awasaki et al. 2000; Newsome et al. 2000). The fact that the most prominent abnormalities observed in trio loss-of-function mice involved skeletal muscle and not the nervous system suggests that mammalian kalirin fulfills many of the neuronal functions regulated by Drosophila trio.
In differentiating neurons, kalirin expression may be important in promoting axon outgrowth, because expression of kalirin in AtT-20 corticotrope tumor cells alters cytoskeletal organization and produces spike-like protrusions of actin from the cell surface (Mains et al., 1999). In addition, mutations in unc-73 in C. elegans and trio in Drosophila produce cell and axon growth cone migration defects (Steven et al. 1998; Awasaki et al. 2000; Bateman et al. 2000; Liebl et al. 2000; Newsome et al. 2000). These defects appear to arise from the loss of guanine nucleotide exchange activity needed for Rac activation (Newsome et al. 2000).
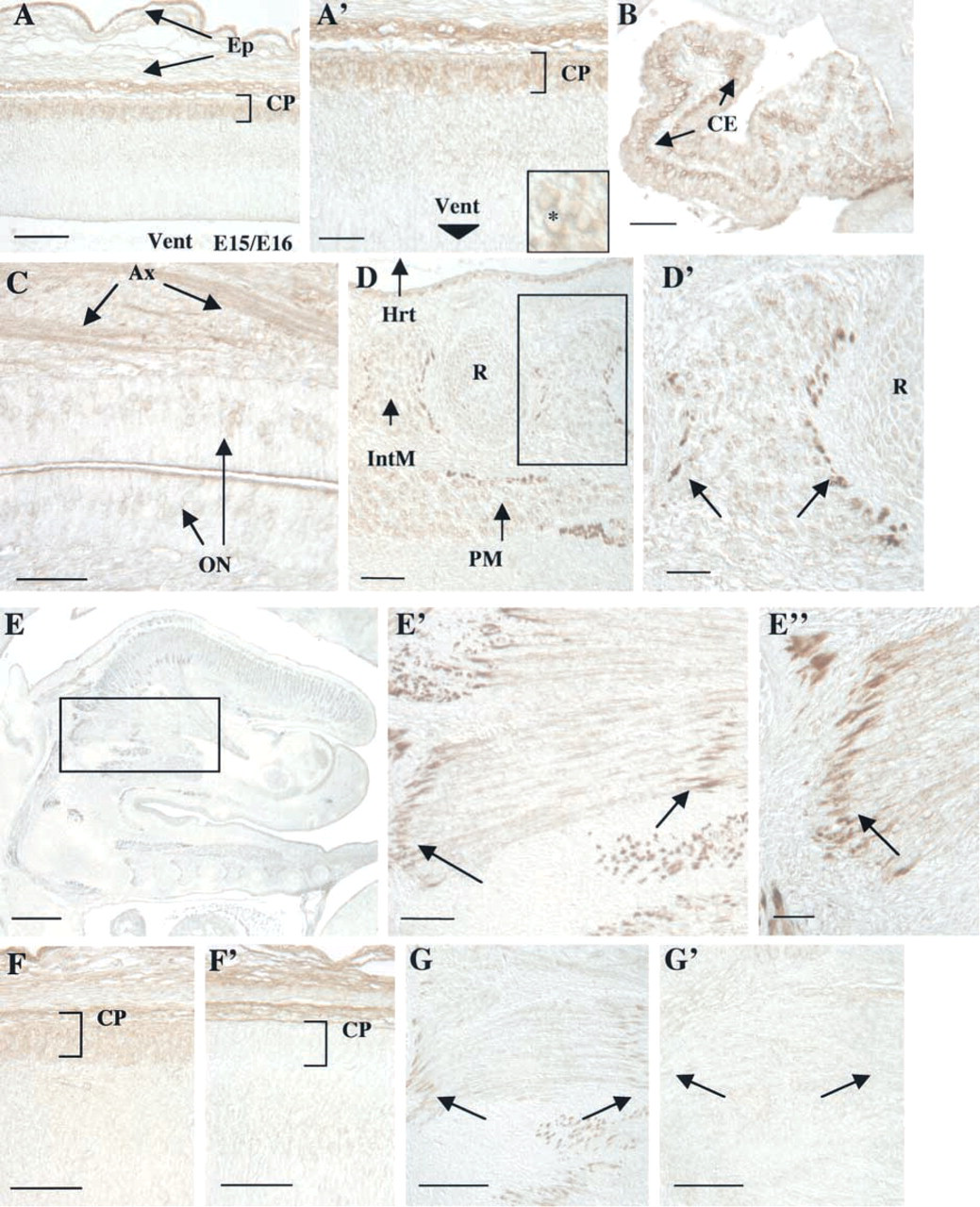
Kalirin protein expression in various tissues at E15/E16. (
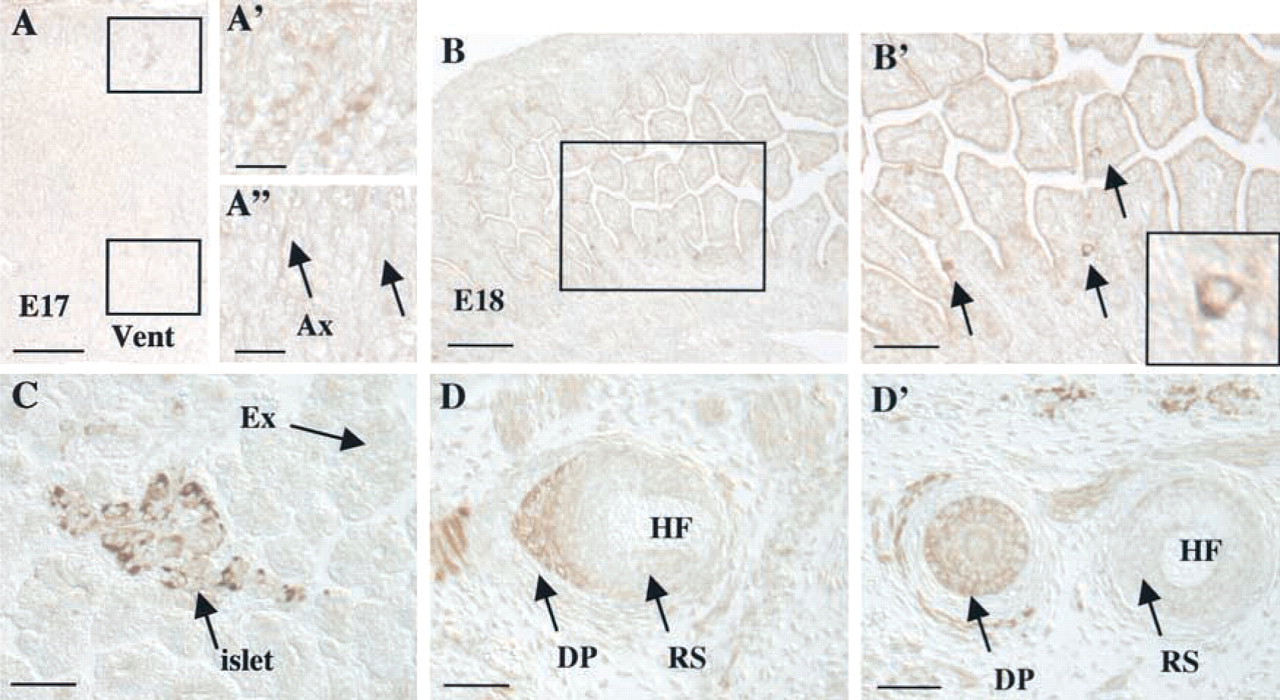
Kalirin demonstrates broad tissue distribution at E17/E18. (
The kalirin isoforms have divergent C-terminal domains. Kalirin-7, the major adult isoform, contains a PDZ-binding motif, while kalirin-8 contains an SH3 domain. Kalirins-9 and -12 both contain this SH3 domain and in addition contain a second DH/PH domain. Kalirin-12 is distinguished by its protein-kinase like domain (Figure 1B). Using antibody against kalirin–spectrin-like repeats 4–7, we identified kalirin protein expression in both cell bodies and axons. However, using an antibody specific for the C-terminal of kalirin-12, we identified kalirin-12 protein specifically in neuronal cell bodies. These results suggest that kalirin-12 may be selectively retained in neuronal cell bodies, or that kalirin-9, which lacks the C-terminal kinase domain, may be targeted to axons. Similarly distinct localizations of endogenous kalirin-9 and -12 were observed in primary neuronal cultures (Johnson et al. 2000). dTrio, which is similar in structure to kalirin-9, is also localized to axons and to growth cones (Newsome et al. 2000).
The isoform-specific localizations observed for different kalirin proteins, which derive from alternative splicing, suggest that kalirin is important in regulating multiple physiological processes. Although kalirin expression in the adult mammal appears to be specific for the nervous system, kalirin is broadly expressed during development. In neurons of the rat embryo, kalirin may regulate growth cone motility and axon growth. In non-neuronal cells, such as endocrine cells and muscle cells, kalirin may regulate cell migration and attachment. Rescue of Drosophila mutant for dTrio requires expression of trio in both neurons and non-neuronal cells (Bateman et al. 2000; Newsome et al. 2000).
Expression of Kalirin in Migrating Muscle Cells and at Muscle Attachment Sites
In the adult rat, kalirin expression is restricted to the CNS. In the embryonic rat, however, kalirin expression is present in a number of muscle tissues, as well as other non-neuronal tissues. Earliest expression of kalirin in muscle tissue begins in the E10 heart, where it remains elevated throughout development. The fact that heart muscle exhibits normal morphology in trio loss-of-function mice also suggests a major role for kalirin in this tissue (O'Brien et al. 2000). At this embryonic age, kalirin protein expression parallels expression of PAM mRNA in the cardiac region (Zhang et al. 1997). PAM mRNA, however, tends to be more widely expressed, in areas such as the dorsal neural tube and several mesodermal populations (Zhang et al. 1997). The fact that PAM and kalirin are not always co-localized indicates that PAM trafficking does not require kalirin (Alam et al. 1996). Kalirin expression becomes evident in a number of other muscle groups, with the glossopharyngeal and tongue muscles becoming some of the most prominently immunoreactive. Although dTrio is expressed in non-neural tissue in Drosophila larvae (Newsome et al. 2000), the sites at which mammalian trio is expressed during development have not been mapped.
Kalirin-12 is broadly expressed and uniquely localized during embryonic development. (
In muscle cells and tissues, kalirin demonstrates onset of expression during muscle cell migration. This is most dramatically seen with cells that will eventually form the embryonic tongue (Figure 4B). In rats and mice, these cells originate from the first five somites, migrate to the level of the diaphragm as the hypoglossal cord, and move anteriorly to form the embryonic tongue muscles (Dietrich et al. 1998; Brand–Saberi and Christ 1999). Kalirin is homogeneously expressed within these migrating muscle cells. After migration to their final position in the newly forming tongue, these muscle cells fuse to form multinucleate myotubes (Mayo et al. 1992; Yamane et al. 1997). Kalirin expression patterns change after final migration to the tongue from a homogeneous expression pattern to a tip-like staining pattern. This staining pattern localizes kalirin protein to the end tips of the muscle fibers and to points of muscle–muscle contact. Interestingly, this expression pattern is similar to that described for dTrio, which localizes to muscle attachment sites in Drosophila (Awasaki et al. 2000; Bateman et al. 2000). Skeletal and tongue muscle morphology is grossly altered in trio loss-of-function mice, suggesting that both kalirin and trio are expressed.
The kalirin homologue unc-73 is broadly expressed in a number of migrating cells in the developing C. elegans, including the sex myoblasts, and mutations in the unc-73 gene lead to defects in cell migration (Steven et al. 1998). These defects have been proposed to be secondary to loss of Rac activation and actin reorganization. Kalirin may function in a similar manner in mammalian myoblasts to aid in cell migration and also to help with the formation of muscle–muscle and muscle–tendon attachments. Kalirin may act in a similar manner to the Drosophila protein MSP-300, a member of the spectrin superfamily, which participates in the formation of muscle–ectoderm and muscle–muscle attachments, as well as aiding myotube migration (Volk 1992).
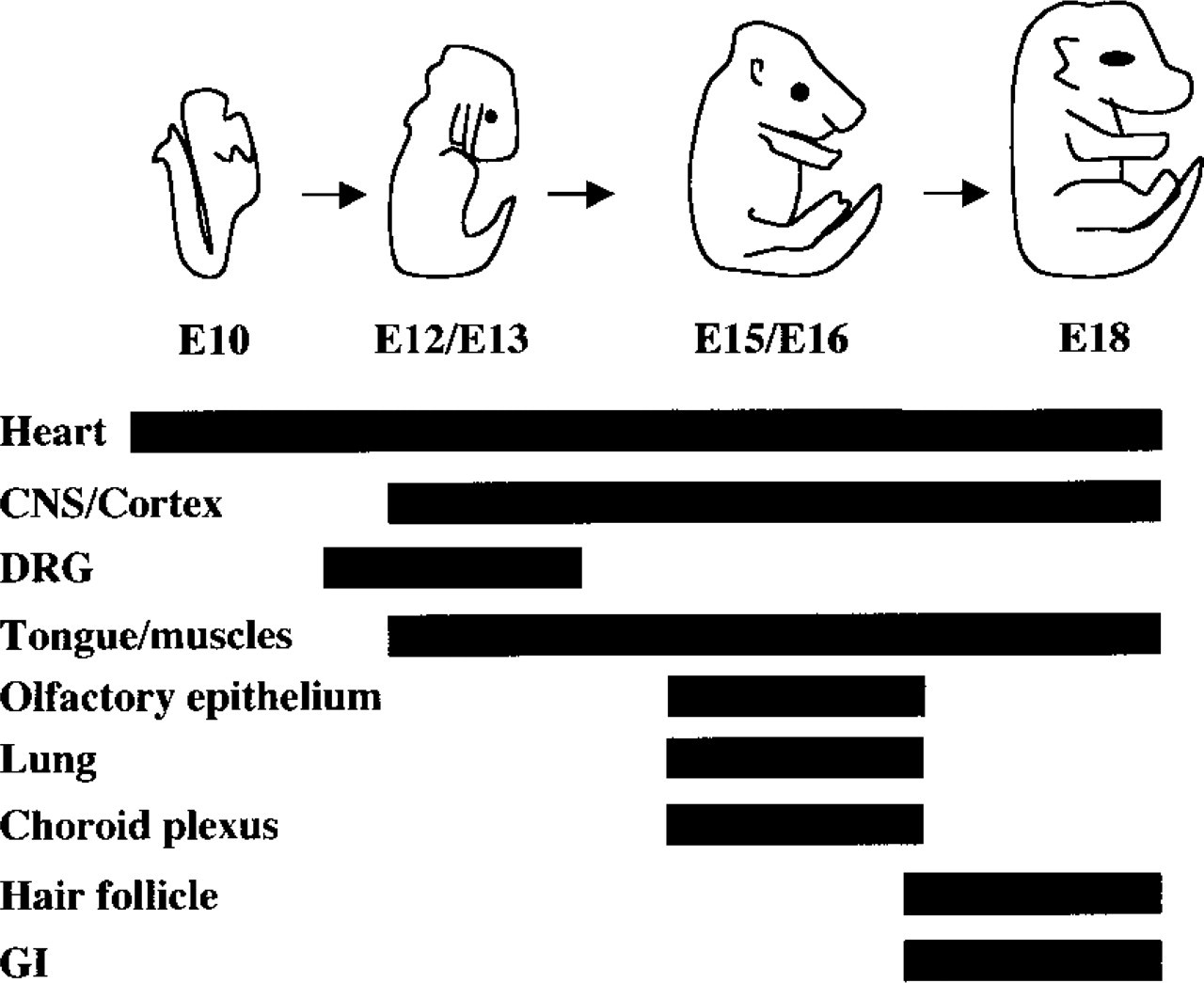
Schematic diagram summarizing kalirin expression patterns at E10, E12/E13, E15/E16, and E18. The heart, central nervous system/cortex (CNS/Cortex), dorsal root ganglia (DRG), tongue/muscles, olfactory epithelium, lung, choroid plexus, hair follicle, and gastrointestinal system (GI) are represented. Solid black bars indicate kalirin expression as identified by immunohistochemistry. Kalirin protein was identified in the heart at E10 and persisted through E18 before declining to background levels. Kalirin protein expression in both the central nervous system/cortex and tongue/muscles was seen from E12/E13 to E18. Dorsal root ganglia expressed kalirin protein within a more restricted window at age E12/E13. Similarly, the olfactory epithelium, lung, and choroid plexus also showed transient patterns of kalirin protein expression at E15/E16. Hair follicles and intestinal epithelial cells showed kalirin protein expression late in embryonic development (E18).
The functional domains of kalirin suggest pathways through which development of nerve, muscle, and endocrine tissue could be affected. Kalirin is a close homologue of a number of proteins demonstrated to function in axon guidance and cell migration, including UNC-73 and Trio (Steven et al. 1998; Lin and Greenberg 2000). Kalirin also shares identity with Duet, which has been shown to interact with the actin cytoskeleton (Kawai et al. 1999). dTrio has been localized either at or near the plasma membrane, suggesting that its homologue kalirin may function to link the actin cytoskeleton to the cell surface (Newsome et al. 2000). The ability of kalirin to activate Rac1 is similar to that of Trio and UNC-73 (Steven et al. 1998; Lin and Greenberg 2000; Penzes et al. 2000a). Mutations in Drosophila Rac1 (Drac1) cause altered initiation and elongation of axon outgrowth, as well as disrupted myoblast fusion (Luo et al. 1994). Furthermore, Drac1 is highly expressed in the nervous system and mesoderm during neuronal and muscle development and differentiation (Van Aelst and D'Souza–Schorey 1997).
During embryonic rat development, kalirin, predominantly the kalirin-9 isoform, may function to promote proper formation and interaction of a number of tissues, including muscle and nervous tissue. Kalirin most likely can transduce these effects in a similar manner to UNC-73 and Trio, by activating Rac1, a member of the Rho GTPase family of proteins (Van Aelst and D'Souza–Schorey 1997). Recently, the Drosophila Trio protein has been demonstrated to activate the Pak protein within spatially restricted domains of the growth cone, and Drosophila Pak mutants appear phenotypically similar to Trio mutants (Newsome et al. 2000). Kalirin may function to mediate similar effects in murines via a downstream activation of Rac1 and the mammalian homologue to Pak. The roles played by kalirin in embryonic development may provide insights into the molecular events underlying axon outgrowth, muscle cell migration and differentiation, and endocrine organ development.
Footnotes
Acknowledgements
Supported by DK-32948, DA-00266, DC-2979, and the Medical Scientist Training Program.
We would like to thank Marie Bell for technical support, Dick Mains for assistance with imaging, and Dr Christian Hansel for invaluable constructive comments on the manuscript.
