Abstract
Estrogen, largely produced in testis and adrenal gland, may play important roles in male reproduction. Most of the effects of estrogens are mediated by binding of estrogen to one or both of the two estrogen receptor (ER) subtypes α and β. Recently, they have been described in testis, prostate, and efferent ducts, mostly in rodents. The goal of this study was to prove the evidence of ERs in human corpus cavernosum and male urethra, exploring the protein expression of these receptors by immunohistochemistry. Corpus cavernosum and corpus spongiosum smooth muscle was immunoreactive for the androgen receptor (AR), ERα, and strongly for ERβ. Endothelial cells were negative for AR, sporadically positive for ERα, and positive for ERβ. Urethral epithelium showed strong nuclear expression of AR, predominantly in the basal cell layer, and nuclear expression of ERα in the intermediate cells. ERβ was highly expressed in almost all urethral nuclei and, much more weakly, in cytoplasm. Progesterone receptor (PGR) was negative in all cases and all tissues. These results represent the first report that ERα and particularly ERβ are regularly expressed in human penile tissue.
T
Estrogens are known to stimulate growth, blood flow, and water retention in sexual organs (Jenner et al. 1972; Batra et al. 1985). There, and in the lower urinary tract, ERα and ERβ have been described in testis, prostate, and efferent ducts (Bodker et al. 1999; Makela et al. 2000; Salmi et al. 2001). Most of these studies have been performed in rodents, but there is only one piece of evidence of ERs in the penis (rat; Jesmin et al. 2002). The goal of our in vitro study was to prove the evidence of the two ER isoforms, as well as of the androgen receptor (AR) and the progesterone receptor (PGR), in human corpus cavernosum and male urethral tissue, exploring the protein expression of these receptors by immunohistochemistry (IHC). The results represent the first report that ERα and particularly ERβ are regularly expressed in human penile tissue.
Materials and Methods
Tissue Samples
Specimens (n = 23) were obtained from patients who underwent partial penectomy in Vienna University hospital because of squamous cell carcinoma of the penis. All cases, within 8 years from 1994 to 2001, for which representative material was available were enrolled in this study. Concerning other clinical diagnosis, probably affecting the vasculature, patients had at time of surgery eight arterial hypertension, five coronary heart disease, three non-insulin-dependent diabetes mellitus, one chronic veinous insufficiency and one adipositas. The ages of the patients at surgery were 21–87 years (mean 65.6, SD 17.5). Tissue was fixed in formalin and routinely processed in paraffin.
Immunohistochemistry
Four-μm-thick sections of the tumor-free resection edge were dewaxed and rehydrated. Deparaffinization was carried out in xylene and graded ethanols. Endogenous peroxidase was blocked by incubation in 5% H2O2 in methanol for 15 min. Heat-mediated antigen retrieval was performed by microwaving three times for 5 min at 500 W in Citra-Plus solution (BioGenex; San Ramon, CA) for ERβ staining or citrate buffer (pH 6.0) for all others. Unspecific bindings were blocked using the biotin blocking system (DAKO; Carpinteria, CA). Incubation was carried out with the following antibodies in a humidified chamber:
AR: mouse monoclonal AR441, dilution 1:100 (DAKO), 1 hr at RT.
ERα: mouse monoclonal 1D5, dilution 1:200 (DAKO), 1 hr at RT.
ERβ: mouse monoclonal 14C8, dilution 1:1500 (BioGenex), overnight at 4C.
PGR: mouse monoclonal PgR 636 (DAKO), dilution 1:1600, 1 hr at RT.
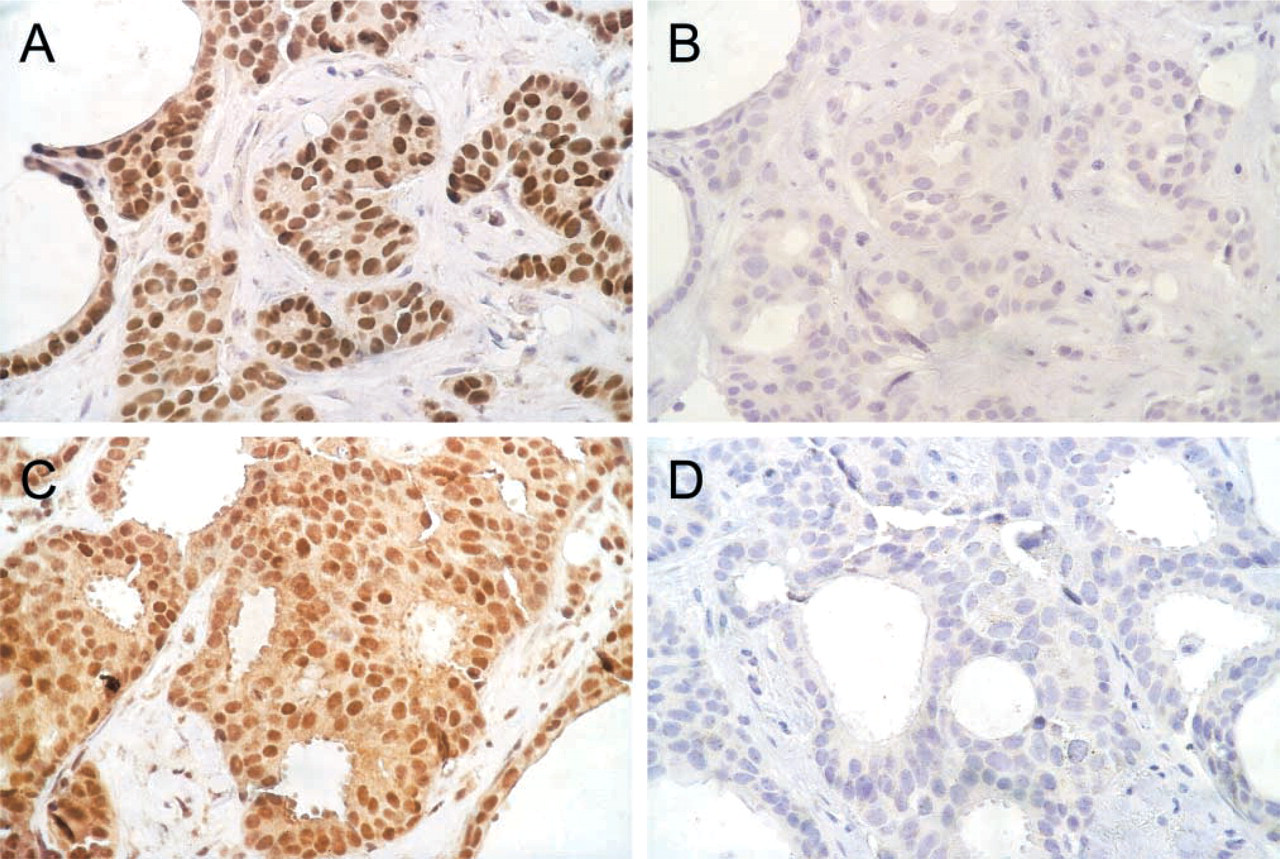
Antibodies against AR, ERα, and PGR have been well characterized by Western blotting on human recombinant receptors (detailed documentation at the manufacturer, DAKO) and are widely used in clinical routine diagnostics. The antibody raised against ERβ has been characterized by absorption studies (Skliris et al. 2002). These authors found it most suitable for IHC on paraffin-embedded tissue compared to six other ERβ antibodies. To further test the specificity of 1D5 and 14C8, antibodies were diluted to working concentration and preabsorbed with a 100-fold excess of the respective antigens, i.e., recombinant human ERα-protein (Affinity BioReagents; Golden, CO) at 23 pmol/100 μl and recombinant human ERβ-protein (Alexis Biochemicals, San Diego, CA) at 44 pmol/100 μl for 1 hr at RT. Solutions were centrifuged at 13,000 rpm for 10 min and the supernatant was used for incubation (see Figure 2).
A staining procedure was carried out for all slides using the ChemMate Detection Kit (DAKO) in which diaminobenzidine is the chromogen. After a 5-min reaction, staining was interrupted by washing in PBS followed by counterstaining with Gill's hematoxylin (Merck; Darmstadt, Germany). Positive controls for ERα, ERβ, PGR, and AR included previously tested and, respectively, positive breast and prostatic cancers. In the negative controls primary antibody was replaced with the respective isotype immunoglobulin (mouse IgG1 for AR441, 1D5, and PgR636; mouse IgG2a for 14C8) in the same concentration (Coulter Clone; Miami, FL).
Immunostaining of CC/CS smooth muscle cells a

a–, number of samples with no significant staining, +, with scattered or focal intense immunostaining, and ++, with intense immunostaining in 50% or more of respective cells.
IHC was performed semiquantitatively on three sections per sample and antibody.
The intensity of nuclear staining and the number of stained cells were examined independently by two investigators (WD and AH). No significant staining was scored as 0, scattered or weak immunostaining as +, and intense immunostaining in approximately 50% or more of respective cells as ++ (Fan et al. 2002).
Results
Corpus Cavernosum (CC)
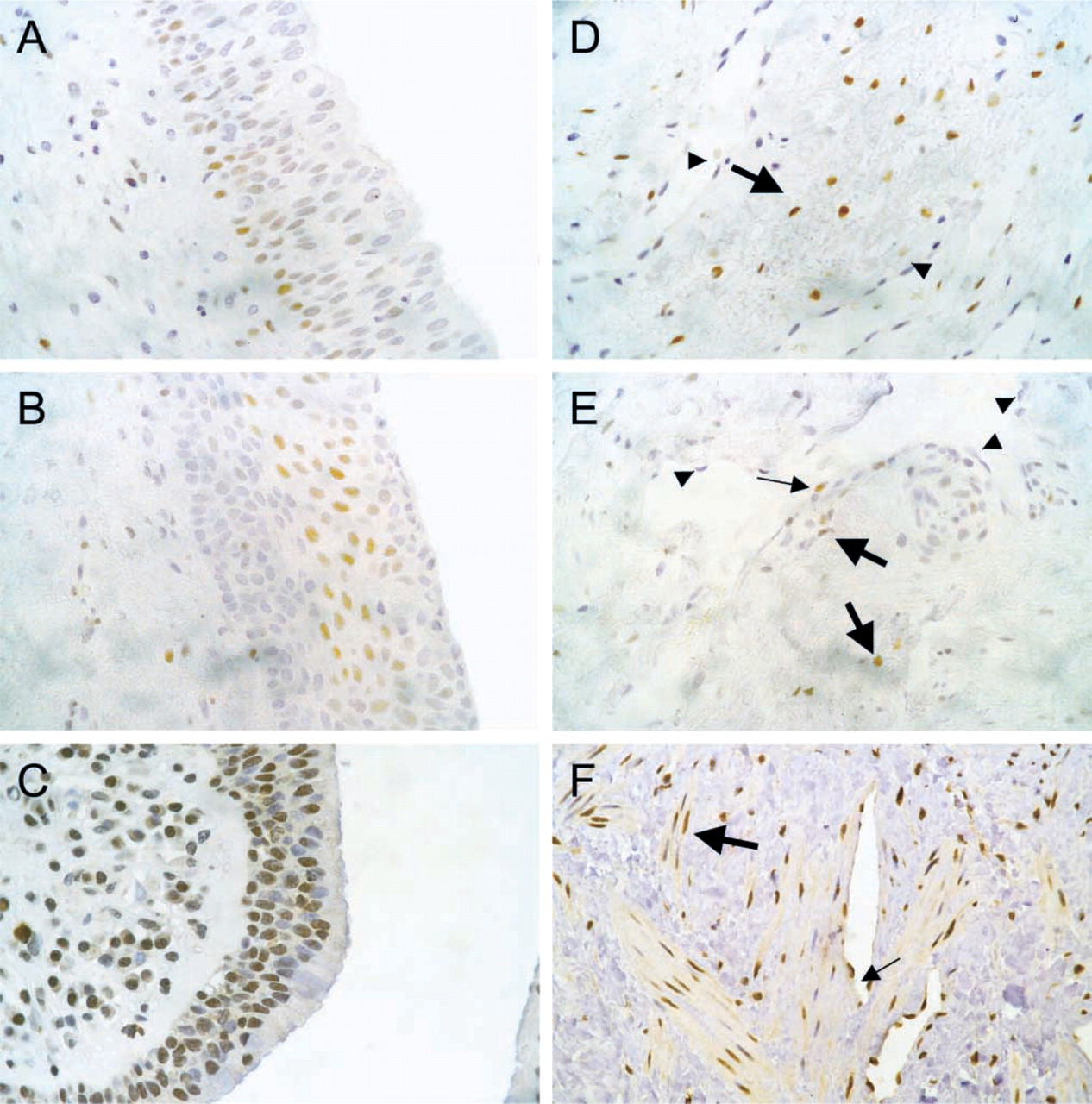
Corpus cavernosum tissue was immunoreactive for AR in all cases, where nuclei of smooth muscle cells showed expression ranging from weak to strong. Endothelial cells were negative for AR in all slides (Tables 1 and 2; Figure 1). ERα protein could be detected in the nuclei of smooth muscle cells and, sporadically, in endothelial cells in seven cases. Immunostaining ranged from moderate to very weak, and the differentiation from negative results was difficult. There was no staining of cytoplasm in any slide (Tables 1 and 2; Figure 1). However, we found moderate to strong staining of ERβ in >80% of the smooth muscle cells in all CCs examined. In addition, weak immunostaining of the cytoplasm could be detected in about one third of cells, which were randomly scattered between smooth muscle cells with cytoplasm not reactive for ERβ. Almost all endothelial cells expressed ERβ in their nuclei but not in the cytoplasm (Tables 1 and 2; Figure 1). PGR was negative in all cases and all tissues examined, even in the positive control.
Immunostaining of CC/CS endothelial cells a

a–, number of samples with no significant staining, +, with scattered or focal intense immunostaining, and ++, with intense immunostaining in 50% or more of respective cells.
(
Corpus Spongiosum (CS)
In corpus spongiosum, a more “venous” lacunary tissue, results were nearly comparable to those in CC. ERα could be detected in smooth muscle cells and some endothelia in the same seven cases where CC was immunoreactive for ERα. In all 23 CS, smooth muscle showed strong ERβ immmunoreactivity in the majority of cells, but there were more negative nuclei than in CC. All endothelia showed strong ERβ staining. The only unequivocal difference from CC was found in five cases in which the AR could not be detected in any cells. Generally, in the periurethral stroma layer, which passes into the CS, nuclear immunostaining of all expressed steroid receptors was more impressive than in the surrounding vessels. We could not find any significant influence of age, hypertension, or coronary heart disease on CC or CS receptor status.
(
Urethra
Urethral epithelium showed strong nuclear expression of AR, predominantly in the basal cell layer. Immunostaining decreased in the luminal direction to negative in the superficial cells. ERα showed strongest positivity in the intermediate cells of all urethras explored, but staining was weaker than that of AR. Further, ERβ was highly expressed in almost all nuclei, and, much more weakly, in the cytoplasm of epithelial cells of every urethra explored.
Discussion
In this study we investigated the distribution of AR, ERα, ERβ, and PGR protein in human adult penile tissues including corpus cavernosum, corpus spongiosum, and urethra. AR was expressed in smooth muscle cells of all explored CC and 18 of 23 CS tissues, whereas AR was absent in endothelial cells of all vessels. These findings of AR distribution are in accordance with earlier reports of the expression of AR in developing human fetal penis, e.g., Kim et al. (2002), where in specimens older than 16 weeks of gestation CC showed the strongest AR expression of all penile tissues. This study group revealed a weaker immunoreactivity in CS and glans, which underlines our finding, that 5/23 CS lacked reactivity for AR. Kalloo et al. (1993) observed intense immunostaining of AR in CC in human fetuses. Furthermore, they detected strongest reactivity in the periurethral zone of the CS, exactly what we also observed in adult tissue. This might lead to the assumption that androgen not only regulates development in these areas but also helps to maintain normal cell function in vascular smooth muscle cells of the human penis. Evidence of the distribution of ERα, known as “the” estrogen receptor until the β-isoform was cloned, in penile tissue is diminished. In the interesting study of Kalloo et al. (1993), ERα could not be found in developing human penis, whereas ERα mRNA and protein were detected in rat penis very recently by Jesmin et al. (2002). New advances in antigen retrieval techniques, which increase sensitivity especially for steroid hormone staining of paraffin-embedded tissue, may account for the difference in these studies. Despite the fact that immunostaining was low, we obtained clear evidence of ERα protein in CC and CS in 7/23 cases. Differentiation from negative results was difficult, so it can be assumed that some of the samples counted as negative actually do express ERα protein in such low amounts that they cannot be detected with the antigen retrieval methods known today. On the other hand, variation in the ER status of human vasculature has been shown previously (e.g., Rogers et al. 1996). Chen et al. (1999) showed that ERα can mediate the short-term effects of estrogen on eNOS activity and may assist vasodilatation by releasing NO. Concerning possible interactions between AR and ERα, as in ERα-knock-out male mice, in which lower expression of AR immunoreactivity was found in some areas of the brain by McAbee and DonCarlos (1999), we could not find any association between ERα positivity and the amount of AR protein expression in human penile tissue. Beyond that, there was no significant influence of age or disease, such as hypertension or coronary artery disease, in remodeling the vasculature or on CC or CS receptor status. We must admit that the numbers of samples in this study were too low to provide statistically relevant statements. We showed that ERβ is the predominant ER subtype in human penile cavernous tissue. ERβ is abundantly expressed in both smooth muscle cells and endothelium of the CC and the CS, the urethral epithelium, and the urethral glands. Although this is the first evidence of ERs in human adult penile tissue, it matches one earlier report of ERα and ERβ in developing rat penis by Jesmin et al. (2002), who found less pronounced expression of ERβ in adult rats. In our data on human cavernous tissue, ERβ is dominant in almost all cells. In the endothelium, known to be the key regulator of vascular integrity, ERβ was found in this study to be expressed ubiquitously but ERα only sporadically. A comparable result was described first in vascular endothelium of rat uterus by Wang et al. (1999), and then in vessels of human endometrium (Critchley et al. 2001), where ERβ was the only steroid receptor expressed. Moreover, a number of studies have shown ERα to be important in the functioning and growth of endothelium both in vitro and in vivo, e.g., Brouchet et al. (2001) and Gargett et al. (2002), where estrogen tends to mediate transcription via ERα rather than ERβ. Therefore, estrogen incubation increased VEGFR-2 expression and VEGF binding only in ERα-positive myometrial microvascular endothelial cells, but expression of ERα was low and variable among subjects (13 of 21 samples). In that study, the VEGFR system was not associated with ERβ expression, but ERβ mRNA and protein could be demonstrated at a higher level than ERα in every sample. Beyond this, ERβ has been demonstrated not only in the vasculature of the uterus but also in great vessels (Taylor and Al-Azzawi 2000) and umbilical vein endothelial cells (Stefano et al. 2000). It has been suggested that ERβ mediates some of the direct effects of estradiol on the vasculature. For example, ERβ mRNA is upregulated in endothelial and luminal smooth muscle cells after balloon injury (Lindner et al. 1998) and inhibits the vascular injury response in ERα-deficient mice (Iafrati et al. 1997). Interestingly, all samples were negative for PGR, which has often been found to be co-expressed with ERs. Moreover, there is clear evidence that progesterone can influence the proliferation of endothelial cells (Vázquez et al. 1999). Although extended parts of human vasculature seem to be susceptible to progesterone, some studies have revealed heterogeneous staining patterns for PGR, e.g., in endometrial vascular endothelial cells (Rogers et al. 1996), or completely negative results, e.g. in telangiectasias (Sadick and Niedt 1990). In the male reproductive system, PGR has been demonstrated in prostate and epididymal epithelial cells, but the same study (Ergun et al. 1997) found epididymal vascular endothelial cells ER-positive but PGR-negative in all regions of the organ. Accordingly, progesterone seems to be subordinate for vascular function of human epididymis and penis.
The relevance of androgens and other sex hormones, particularly estrogen, in the modulation of the structure and function of the urothelium and the distal urethra is not yet understood. Although two reports also described strong reactivity of AR in developing human and adult rat urethra by IHC (Salmi et al. 2001; Kim et al. 2002), there is no evidence in adult human male urethra. In our study, the basal cells markedly expressed AR, indicating that androgen is involved in the growth of human male urothelium. Whereas in the male rat urethra, Makela et al. (2000) could not detect ERα by in situ hybridization and IHC, a more recent study found weak ERα protein expression in the proximal and distal female urethra (Blakeman et al. 2000). However, the importance of moderate expression of ERα in the suprabasal layer of male urothelium remains unclear, particularly because ERβ, which is known to exert inverse transcriptional effects, is highly expressed in almost all epithelial cells. Here, our results showing strong expression in nuclei of the epithelial cells and, additionally, moderate cytoplasmic expression are in agreement with all previous studies in male rat urethras.
In view of the observation that ERβ is expressed in human penile vasculature and urothelium in excess to the other sex steroid receptors, we propose that estrogen could play a significant role in penile blood vessel regulation and epithelial function, operating preferably on the ERβ-isoform. It can be suggested that estrogen's action supports resistance to increased mechanical and hemodynamic stress, according to Lindner et al. (1998), and maintains vascular tone and perhaps erectile function. Further studies will be needed to discover estrogen-regulated genes, especially those being controlled by ERβ, in the male reproductive system, and to clarify the possible involvement of aromatization of androgens to estrogen in penile tissue.
