Abstract
Proline-rich synapse-associated protein-1 (ProSAP1) is a neuronal PDZ domain-containing protein that has recently been identified as an essential element of the postsynaptic density. Via its interaction with the actin-binding protein cortactin and its integrative function in the organization of neurotransmitter receptors, ProSAP1 is believed to be involved in the linkage of the postsynaptic signaling machinery to the actin-based cytoskeleton, and may play a role in the cytoskeletal rearrangements that underlie synaptic plasticity. As a result of our ongoing studies on the distribution and function of this novel PDZ domain protein, we now report that the expression of ProSAP1 is restricted neither to neurons and interneuronal junctions nor to the nervous system. Using immunohistochemical techniques in conjunction with specific antibodies, we found that, in the CNS, ProSAP1 can be detected in certain glial cells, such as ependymal cells, tanycytes, subpial/radial astrocytes, and in the choroid plexus epithelium. Moreover, our immunohistochemical analyses revealed the presence of ProSAP1 in endocrine cells of the adenohypophysis and of the pancreas, as well as in non-neuronal cell types of other organs. In the pancreas, ProSAP1 immunoreactivity was also localized in the duct system of the exocrine parenchyma. Our findings demonstrate that, in addition to neurons, ProSAP1 is present in various non-neuronal cells, in which it may play a crucial role in the dynamics of the actin-based cytoskeleton.
(
Keywords
D
The recently described PSD protein ProSAP1 (proline-rich synapse-associated protein-1), also known as cortactin-binding protein-1 (CortBP1; Du et al. 1998) or Shank2 (Lim et al. 1999), is a central element of the postsynaptic actin-based cytoskeleton (Boeckers et al. 1999a). It interacts with the actin-binding protein cortactin (Du et al. 1998) and contains various structural motifs for further protein–protein interactions, including a novel type of PDZ domain (Boeckers et al. 1999a). Recently identified binding partners, such as the group of guanylate kinase-associated proteins (GKAPs)/SAP90-PSD95-associated proteins (SAPAPs) and the Homer protein family, indicate that ProSAP1 is linked to both ionotropic and metabotropic glutamate receptors (Boeckers et al. 1999b; Naisbitt et al. 1999; Tu et al. 1999). In addition to its probable role as a multivalent scaffolding molecule in the mature PSD, ProSAP1 may be involved in the assembly of developing PSDs during the postnatal period (Boeckers et al. 1999a).
Although the molecular dissection of the PSD is naturally focused on neurons, a full understanding of the PSD at the molecular level also requires a detailed knowledge of the possible extraneuronal distribution and function of its protein constituents, even if the PSD is usually regarded as a unique trait of neurons. In this context, it should be recalled that it is already well known that many pre- and postsynaptic proteins of the synaptic signal transduction apparatus are not restricted to neurons. For example, in the CNS several proteins implicated in the regulation of synaptic vesicle trafficking could readily be detected in glial cells (Maienschein et al. 1999). Moreover, several parallels at the morphological and molecular levels can be drawn between synaptic signal transmission of neurons and secretory processes of various endocrine cell types (Thomas–Reetz and De Camilli 1994; Redecker 1999).
In the present immunohistochemical and immunochemical study, we therefore sought to further characterize the novel postsynaptic protein ProSAP1 by asking (a) whether the expression of ProSAP1 in the CNS is really confined to neurons, and (b) whether this protein is also detectable in non-neuronal cells, especially in endocrine cells such as those of the adenohypophysis and of pancreatic islets. The latter endocrine organs were chosen for a detailed investigation because they are already known to be endowed with various protein components involved in presynaptic function.
Materials and Methods
Animals and Tissue Preparation for Immunohistochemistry
Adult Wistar and Lewis rats of both sexes, which had been kept under normal laboratory conditions, were used for the investigation. Principles of laboratory animal care and specific national laws were followed. Anesthetized animals were transcardially perfused with prewash and fixative solutions, as detailed elsewhere (Redecker and Bargsten 1993). The fixative consisted of a solution of 4% depolymerized para-formaldehyde in 0.1 M phosphate buffer (pH 7.4) or a mixture of phosphate-buffered 0.5% picric acid and 4% paraformaldehyde. Fixed tissue specimens (whole rat brain, cervical spinal cord, pituitary, pancreas, lung, liver, kidney) were then dissected out and cryostat sections were cut at a thickness of 5–8 μm. In addition, specimens were embedded in paraffin. For analysis of serial semithin sections, rats were sacrificed by decapitation under CO2 anesthesia. The dissected tissue specimens were then quenched in isopentane precooled with liquid nitrogen, freeze-dried for 48 hr, and fixed by vapor-phase paraformaldehyde. After embedding of the specimens in epoxy resin (Araldite), serial semithin sections were cut at a thickness of 0.5 μm.
Immunohistochemistry
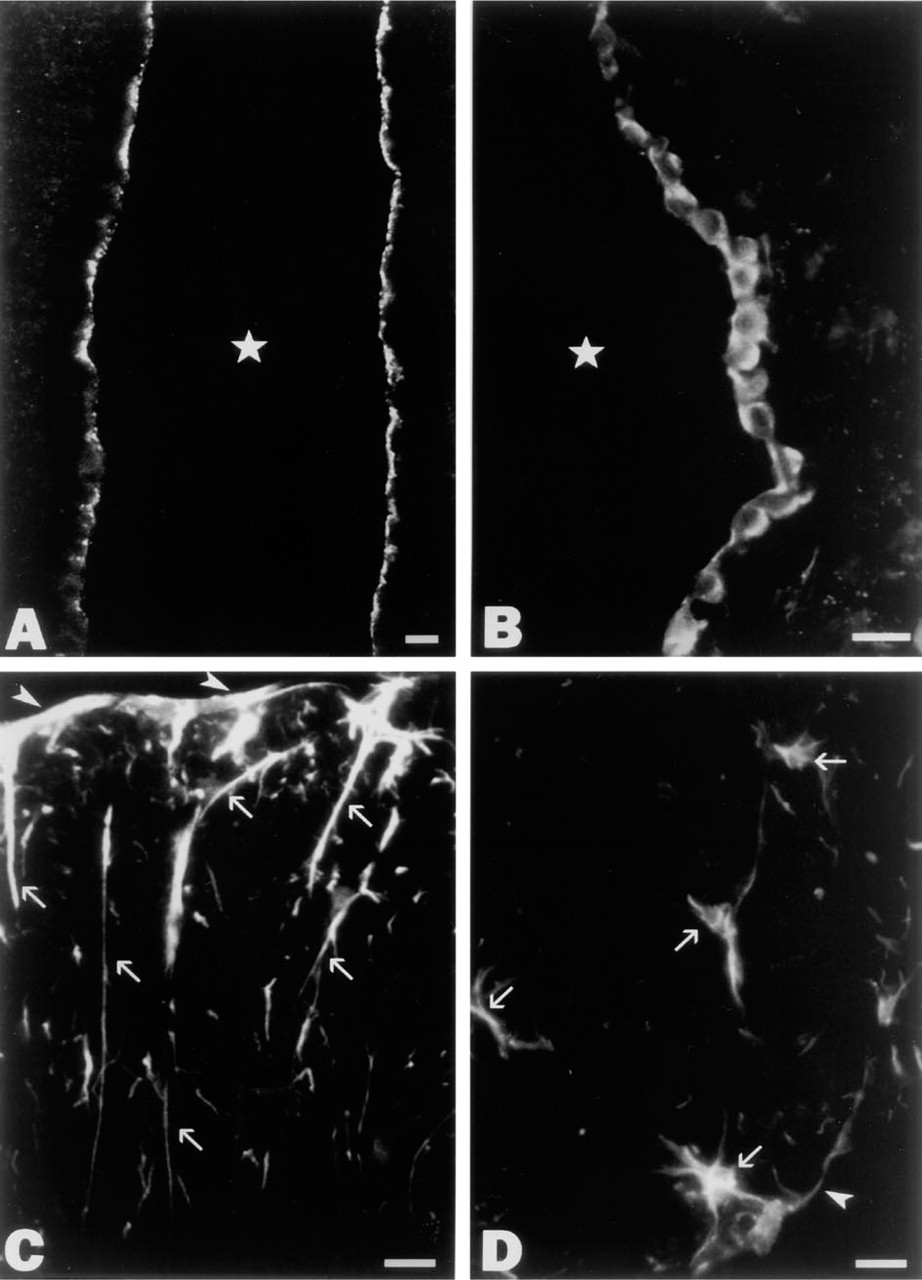
Immunofluorescence microscopic localization of ProSAP1 in the epithelial lining of the cerebral ventricles (
Method controls generally consisted of omission of single steps in the immunohistochemical protocol, application of primary antibodies of unrelated specificities or of non-immune sera, use of ascending dilutions of the first antibody, or use of high-molar (0.5 M) PBS as a rinsing solution between the various steps of the immunohistochemical protocol.
Results
Immunohistochemistry
Our immunohistochemical study revealed a punctate pattern of immunoreactivity in many regions of the rat brain, compatible with the predominant synaptic localization of ProSAP1 described previously (Boeckers et al. 1999a). In addition, we consistently noted conspicuous immunoreactivity within the lining of the cerebral ventricles, the aqueductus cerebri, and the central canal of the spinal cord. Systematic analysis showed that, in the cells lining the ventral region of the third ventricle, strong immunofluorescence for ProSAP1 was restricted to the apical cell surface (Figure 1A). This region of the ventricular lining is known to be occupied by tanycytes. Double labeling with antibodies directed against ProSAP1 and vimentin allowed unequivocal identification of the ProSAP1-positive cells as tanycytes, the cell bodies and long basal processes of which were clearly visualized by the vimentin antibodies (Figure 2A). Apical immunoreactivity for ProSAP1 was also observed in scattered tanycytes elsewhere in the CNS, i.e., in the lining of the aqueductus cerebri, the caudal floor of the fourth ventricle, and the central canal of the spinal cord (Figure 2D). As demonstrated by double labeling experiments, the immunofluorescence for ProSAP1 largely overlapped with immunoreactivities for actin and cortactin (Figure 2B) at the ventricular pole of the tanycytes. In addition to tanycytes, ependymocytes of all brain ventricles consistently displayed ProSAP1 immunoreactivity. However, in contrast to tanycytes, immunofluorescence was mostly not restricted to the luminal domain of ependymocytes but was present in the whole cytoplasm (Figure 1B), although an enrichment of ProSAP1 in the apical cytoplasm was also evident in some ependymocytes (Figure 2C). Moreover, we noted that the choroid plexus epithelium of the cerebral ventricles could be labeled by our ProSAP1 antibodies. Similar to tanycytes, immunoreactivity was largely confined to the apical cytoplasm of the plexus epithelium, where ProSAP1 was co-localized with cortactin (Figure 2E) and actin. In contrast to the cortactin-binding protein ProSAP1, cortactin itself was also distributed to the basolateral cytoplasm of both tanycytes and epithelial plexus cells (Figures 2B and 2E). We also encountered some ProSAP1-positive astrocytic glial cells. Thus, in cross-sections of the white matter of the spinal cord, several processes, sometimes of considerable length, became visible, which exhibited distinct immunoreactivity for ProSAP1 and were oriented perpendicularly to the pial surface, extending into the glia limitans (Figure 1C). The co-expression of GFAP in these processes confirmed their astrocytic nature (not shown). Subpial astrocytic cell bodies giving rise to several processes could also be visualized by the ProSAP1 antibodies (Figure 1D). As in the spinal cord, we also found scattered ProSAP1-positive processes of subpial astrocytes in the brainstem.
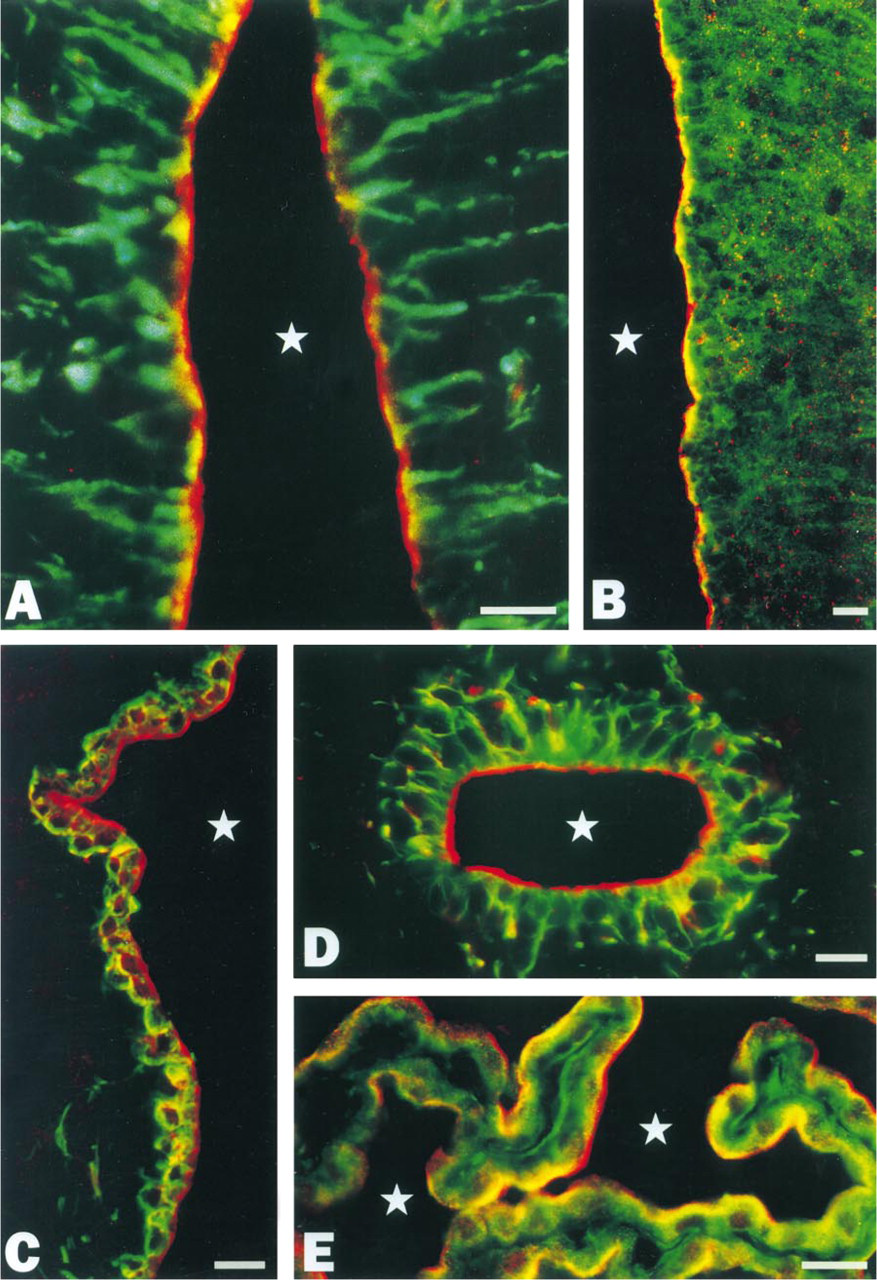
Color micrographs demonstrating the simultaneous localization by immunofluorescence microscopy of ProSAP1 (red)/vimentin (green) (
Our immunohistochemical investigation also disclosed the presence of ProSAP1 in endocrine cells. In the adenohypophysis, many ProSAP1-immunoreactive cells were distributed throughout the anterior lobe (Figure 3A), whereas only faint immunoreactivity could be observed in the intermediate lobe. In addition, strong immunoreactivity was detectable in the non-endocrine marginal layer cells lining the hypophyseal cleft (not shown). In most ProSAP1-positive adenohypophyseal cells, immunostaining was visible in the whole cytoplasm, including the cell cortex, and frequently appeared in a punctate pattern (Figures 3A, 3C, and 3D). For identification of the immunopositive anterior lobe cells, serial semithin sections were alternatively immunostained for pituitary hormones or ProSAP1. We found that somatotrophs were most densely stained by the ProSAP1 antibodies (Figures 3D and 3E), whereas gonadotrophs expressing LH and/or FSH displayed only a moderate degree of immunoreactivity. Thyrotrophs, lactotrophs, and corticotrophs were at most faintly immunopositive or appeared unreactive to the ProSAP1 antibodies.
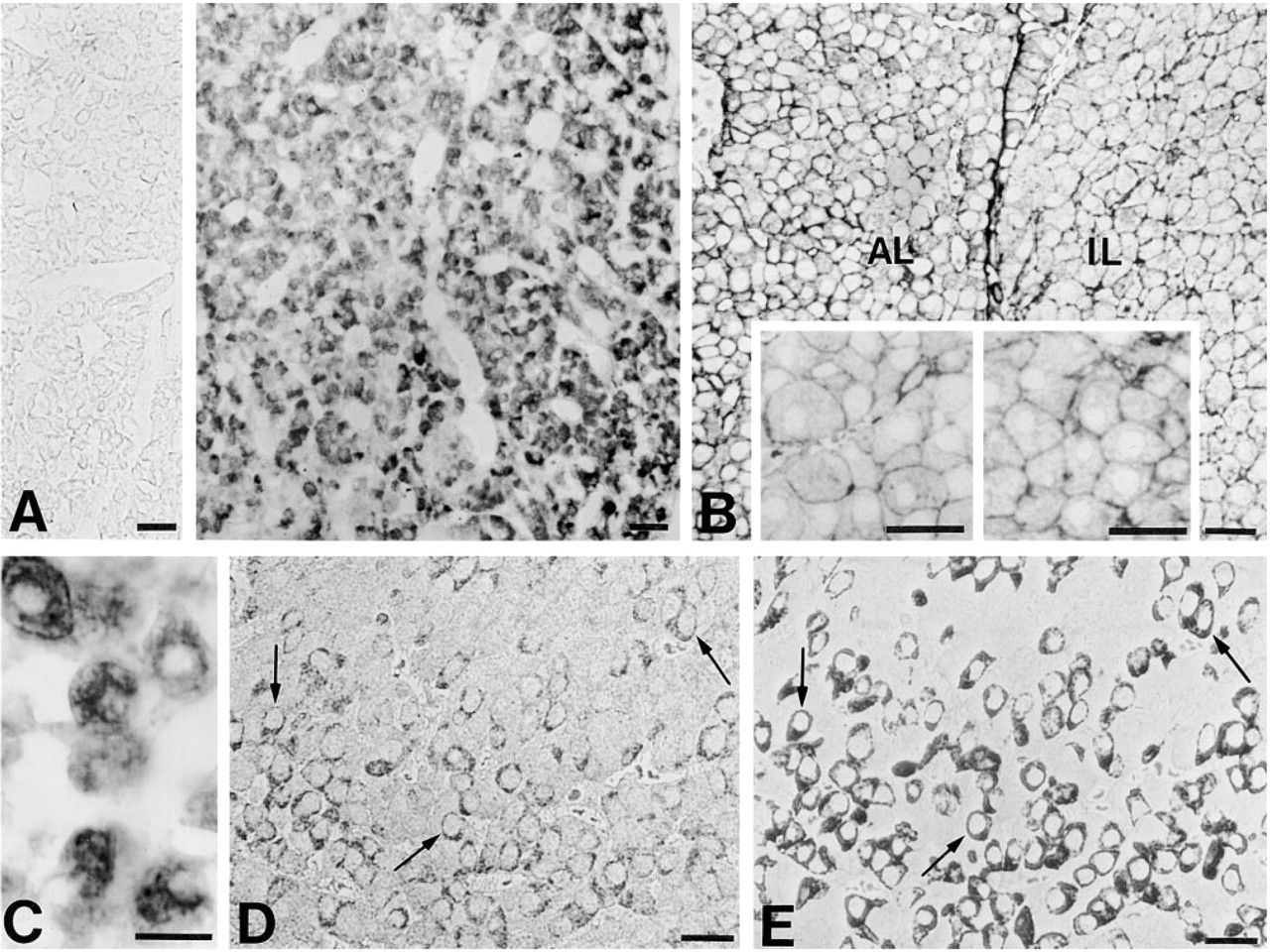
A gallery of micrographs illustrating the immunohistochemical detection of ProSAP1 (
ProSAP1-positive endocrine cells were also discernible in the pancreas, where all islets in the sections were clearly immunostained. Analysis of serial semithin sections demonstrated that ProSAP1 was expressed in both insulin-producing B-cells (Figures 4A and 4B) and glucagon-producing A-cells (not shown), whereas somatostatin-positive D- and pancreatic polypeptide-positive PP-cells were only weakly immunoreactive. In contrast to ProSAP1, in both adenohypophyseal and islet cells cortactin (Figures 3B and 4G) and actin were concentrated in the subplasmalemmal cytoplasm. Interestingly, in the pancreas expression of ProSAP1 was not restricted to the endocrine compartment. In the exocrine parenchyma, ProSAP1 was consistently localized in the duct system. Pronounced immunostaining for ProSAP1 was conspicuous in the luminal cell cortex of centroacinar, intercalated duct, and intra/interlobular duct cells (Figures 4C–4E). Similarly, dense immunostaining in the apical cytoplasm of these epithelial cells could be elicited by antibodies against cortactin (Figure 4F) and actin. Double labeling revealed the co-distribution of the latter proteins and ProSAP1 in the luminal cell cortex (not shown).
In addition to the organs on which we focused in this study, we also noted immunoreactivity for ProSAP1 in other tissues, such as the epithelium of hepatic bile ducts, of the tubule system in the kidney, and the bronchial/bronchiolar tree of the lung.
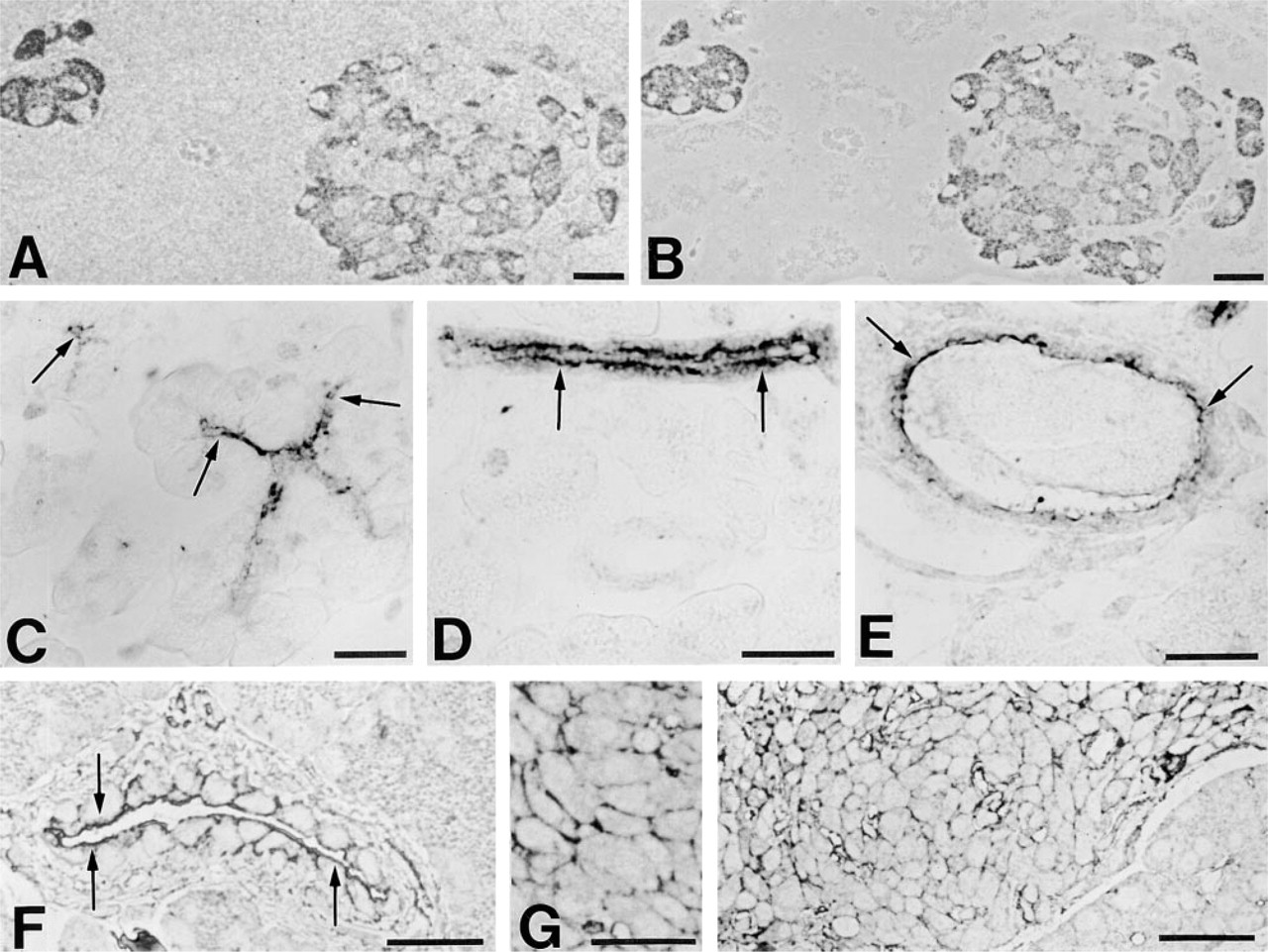
Immunohistochemical localization of ProSAP1 (
Immunoblotting
Similar to previously published results (Boeckers et al. 1999a), all antibodies employed in this study selectively detected a cluster of protein bands with apparent molecular weights ranging from ∼180 kD to ∼230 kD on immunoblots of brain tissue (Figure 5). Protein bands in the same molecular weight range were also revealed on immunoblots of pituitary and pancreatic homogenates (Figure 5A) and of other nonneuronal tissues such as lung, liver, testis, and heart (Figure 5B). Furthermore, corresponding bands became visible in blots of kidney and skeletal muscle homogenates after longer film exposure.
Discussion
ProSAP1 and the closely related molecule ProSAP2 are multidomain proteins that are believed to be central elements of the PSD protein network. Established interactions of both proteins comprise the association with the actin-binding protein cortactin and the binding to members of the GKAP/SAPAP protein family (Boeckers et al. 1999b). The latter interaction is mediated by a new type of PDZ domain contained in the ProSAP sequence and is probably necessary for linking clusters of postsynaptic membrane proteins, such as neurotransmitter receptors or adhesion molecules, to the cytoskeleton (Garner et al. 2000). In this study we extend the current knowledge of ProSAP1 distribution by demonstrating that this protein is not restricted to neurons but also appears to be an important constituent of various non-neuronal cells, in which it often shows a polarized intracellular distribution.
Our immunohistochemical analysis has revealed that ProSAP1 is present in ependymal cells and tanycytes of the cerebral ventricles and of the central canal of the spinal cord. Most conspicuous in tanycytes, the protein is concentrated beneath the apical plasma membrane, where cortactin is also present. Moreover, immunoreactivity for actin is particularly strong in the apical cytoplasm of tanycytes and several ependymal cells (this study; and Gröschel–Stewart et al. 1977).
Therefore, ProSAP1 and cortactin obviously are part of the apical terminal web of these cells, in which microfilament bundles of their many microvilli (Bruni 1974; Wittkowski 1998) are anchored. It has been speculated that cortactin may be involved in the generation of the brush border microvilli in the small bowel (Wu and Montone 1998). Therefore, it is possible that cortactin and ProSAP1 play a role in microvillous-related cytoskeletal rearrangements of ependymocytes and tanycytes. On the other hand, because cortactin and ProSAP1 are held to be engaged in the transduction of signals from the cell surface to the cytoskeleton (Wu and Parsons 1993; Boeckers et al. 1999b), it remains to be elucidated whether comparable functions might also apply to the cells of the ventricular lining. Some circumstantial evidence for such functions comes from recent findings of neurotransmitter receptor expression in ependymal cells and tanycytes (Tang and Sim 1997; Eyigor and Jennes 1998; Kawakami 2000). The tendency of ProSAP1 to accumulate in apical domains of epithelium-type cells is further underlined by its distribution pattern in the choroid plexus epithelium. Thus, ProSAP1 is preferentially localized beneath the apical cell surface of this epithelium where the actin/spectrin-based cytoskeleton is known to be concentrated (Alper et al. 1994).
The results of our immunohistochemical study demonstrate that certain astrocytes located in white matter tracts, obviously corresponding to the particular populations of subpial astrocytes (Bitner et al. 1987) and so-called radial astrocytes (Liuzzi and Miller 1987), must also be included in the list of ProSAP1-expressing cells of the CNS. In these glial cells, the PDZ protein may be part of the molecular machinery that links glial ion channels and neurotransmitter receptors (Hosli and Hosli 2000; Verkhratsky and Steinhauser 2000) to the cytoskeleton. Taking into account its presumptive role during neurite outgrowth in neurons, ProSAP1 may also be implicated in cytoskeletal remodeling during morphological changes of astrocytes. Recent observations of frequent cell divisions and cell differentiation in the white matter of the adult rat spinal cord suggest a higher degree of glial plasticity for the intact spinal cord than was previously thought (Horner et al. 2000).
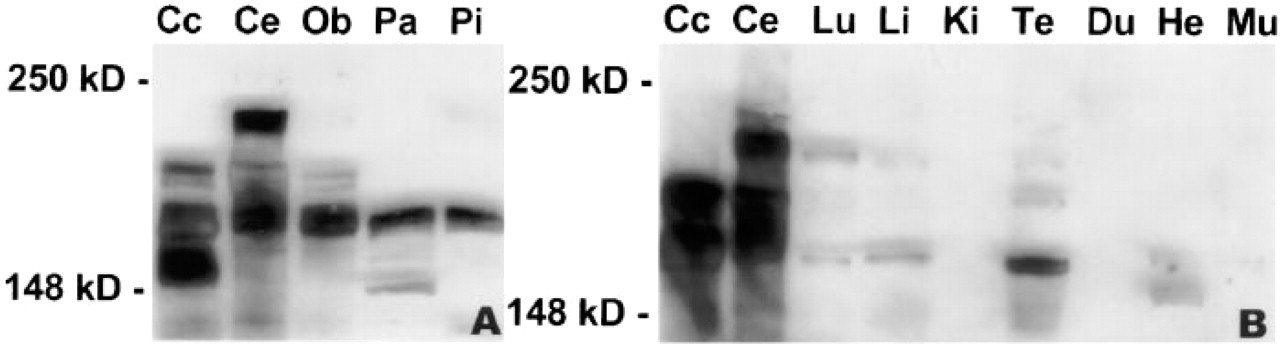
Immunoblots of SDS-PAGE-separated proteins from homogenates of cerebral cortex (Cc), cerebellum (Ce), olfactory bulb (Ob), pancreas (Pa), and pituitary (Pi) (
Interestingly, our immunochemical and immunohistochemical analyses have revealed that ProSAP1 is also expressed outside the nervous system. In endocrine organs, the protein can be localized to various endocrine cell types of the pituitary and the endocrine pancreas as well as to chromaffin cells of the adrenal medulla (our unpublished observations), where it might be engaged in the microfilament-dependent control of secretory activity. A considerable body of evidence indicates that, in both adenohypophyseal and in pancreatic islet cells, cortical actin filaments participate in the regulation of secretory granule exocytosis (Orci et al. 1972; Snabes and Boyd 1982; Castellino et al. 1992; Li et al. 1994; Senda et al. 1994; Carbajal and Vitale 1997) and in membrane retrieval after granule exocytosis (Valentijn et al. 1999). It is conceivable that cortactin is also involved in such microfilament-dependent regulatory mechanisms, taking into account our present new finding of strong cortactin immunoreactivity in the peripheral cytoplasm of endocrine cells. In view of its more widespread cytoplasmic distribution in the endocrine cells investigated, it is possible that in these cells the function of ProSAP1 is not restricted to the cell cortex but extends to other cell compartments, eventually including secretory granules.
In the pancreas, the expression of ProSAP1 is not confined to the endocrine compartment. Rather, strong immunoreactivity also becomes visible in the duct system of the exocrine pancreas after incubation with ProSAP1 antibodies. An apical enrichment of ProSAP1 is conspicuous in centroacinar, intercalated duct, and intra/interlobular duct cells. These cells display several ultrastructural similarities, including the presence of a terminal web beneath their apical surface and the formation of many microvilli with a core of axial bundles of microfilaments (Egerbacher and Böck 1997). According to our results, ProSAP1 and cortactin must be considered as regular constituents of the terminal web in the pancreatic ductule system. It is noteworthy that a PDZ-containing scaffolding protein has recently been described in the pancreatic ductule epithelium, which might link the cystic fibrosis transmembrane conductance regulator (CFTR) to the cortical actin cytoskeleton and could thus be part of a multiprotein complex that leads to a compartmentalization of CFTR (Short et al. 1998). It would be interesting to evaluate whether the PDZ protein ProSAP1 qualifies as a further component of such a presumptive scaffolding complex.
In conclusion, we have established that the expression of ProSAP1, which probably acts as a central organizer of the PSD in neural tissues, is not restricted to neurons. The presence of ProSAP1 in various nonneuronal cells indicates that the function of this protein is neither unique to neurons nor confined to intercellular junctions. It will be interesting to determine all binding partners of ProSAP1 in the former cells and to see whether they differ from the ProSAP1-interacting proteins in neurons. Such an analysis could also give further clues to the targeting mechanisms that specify the polarized distribution of this PDZ protein to discrete cell microdomains, such as the neuronal PSD or the apical cytoplasm of several epithelium-type cells. Therefore, the future elucidation of the function of ProSAP1 in non-neuronal cells not only will provide new insights into the dynamics of their cytoskeleton and its interactions with cell membranes but should also contribute to a comprehensive understanding of the functional microanatomy of the neuronal PSD.
Footnotes
Acknowledgements
Acknowledgments
Supported by the DFG (SFB426/A1 to EDG, Bo 1718/1–1 to TMB), IZKF and IMF (University of Münster to TMB).
We gratefully acknowledge the skillful technical assistance of H. Böning, S. Fischer, and D. von Mayersbach. We thank Prof E. Ungewickell for providing access to the Nikon Eclipse E800 photomicroscope, and Dr A. Gebert for help with the color prints.
