Abstract
Previously, we obtained a protein that has considerable amino acid sequence homology with secretory phospholipase A2 (PLA2) from a bullfrog pituitary fraction obtained during the purification of thyrotropin (TSH). Subsequently, partial amino acid sequence (N-terminal 45 amino acid residues) analysis revealed this protein to be identical to the N-terminal amino acid sequence of otoconin-22, the major protein of aragonitic otoconia in the Xenopus saccule. In this study we developed an antibody against the N-terminal peptide of the bullfrog protein and applied it for immunocytochemical study of the pituitary and its surrounding tissue. Western blotting analysis showed that this antibody recognizes a 20.4-kD protein that has a molecular mass close to that of otoconin-22. Immunohistochemical reaction with the antibody was not found in any anterior pituitary cells but was intense in the monolayer epithelial cells of the endolymphatic sac surrounding the pituitary gland, which is a major storage site of calcium carbonate in amphibians. An electron microscopic study revealed that the cuboidal cells in the endolymphatic sac contained large, polymorphic secretory granules in their apical cytoplasm. Immunogold particles indicating the presence of a PLA2-like protein were observed predominately in these secretory granules. These findings support the view that this PLA2-like protein obtained during purification of TSH was derived from the endolymphatic sac adhering to the pituitary and that this protein is a bullfrog otoconin.
(
Keywords
D
In this study we generated an antibody against the N-terminal peptide of the bullfrog protein and examined the localization of this PLA2-like protein in the pituitary and adjacent tissues, using immunocytochemical techniques, to clarify the origin of the protein obtained during the purification of TSH.
Materials and Methods
Animals
Adult male and female bullfrogs (Rana catesbeiana) were purchased from Ouchi (Misato, Japan). They were kept under normal laboratory conditions for at least 1 week and were fed crickets. Their pituitary glands and surrounding tissues and their paravertebral lime sacs were removed under anesthesia with MS 222 (Nacalai Tesque; Kyoto, Japan), and then processed for light or electron microscopic examinations and for biochemical analysis.
Antisera
To develop an antibody against the bullfrog PLA2-like protein (bPLP), we used a synthetic peptide corresponding to bPLP's N-terminal amino acids 1–13 (ST-135: TPAQFDE-MIKVTT), with an amino-terminal cysteine residue added to assist in coupling the peptide to keyhole limpet hemocyanin (KLH) (Pierce; Rockford, IL). Coupling was done with m-maleimidobenzoyl-N-hydroxysuccinimide ester (Pierce) according to the method of Walker et al. (1985).
A rabbit was injected with the KLH-coupled peptide every 2 weeks for 2 months by the multiple-site method described previously (Tanaka et al. 1991).
Western Blotting Analysis
To separate the proteins from the endolymphatic sac and from the calcium carbonate crystals in the endolymphatic sac, we removed the calcium carbonate from the sac and the crystal by treating them with 10% ethylenediamine-N,N,N,N′-tetraacetic acid (EDTA) in water at 4C for 3 days according to the method of Pote et al. (1993). These samples and anterior pituitary glands were homogenized in cell lysis buffer [50 mM Tris-HCl (pH 8.0), 0.15 M NaCl, 1% Triton X-100, 0.1 mg/ml PMSF, 1 μg/ml aprotinin] and centrifuged in a microcentrifuge for 5 min to remove insoluble materials. The proteins were determined with a BCA Protein Assay Kit (Pierce). The supernatant protein (25 μg) was subjected to electrophoresis on a 10% polyacrylamide gel and transferred to an Immobilon-P membrane (Millipore; Bedford, MA). The proteins in the membrane were reacted sequentially with anti-bPLP serum diluted at 1:20,000, biotinylated anti-rabbit IgG (DAKO; Kyoto, Japan), and streptavidin-conjugated horseradish peroxidase (DAKO). The reaction product on the membrane was visualized using an ECL Western blot detection kit (Amersham Pharmacia Biotech; Uppsala, Sweden). As a control, the primary antibody was replaced with anti-bPLP serum preincubated with 10 μg/ml of the antigen peptide.
To see whether the immunoreactive protein(s) was glycosylated, the extracts from the endolymphatic sacs and calcium carbonate crystals in the endolymphatic sacs were treated for 1 hr at 37C with peptide-N-glycosidase F (Daiichi Pure Chemicals; Tokyo, Japan) before SDS-PAGE and Western blotting.
Light Microscopic Immunocytochemistry
The anterior pituitary gland and its surrounding endolymphatic sac and the paravertebral lime sac were fixed by immersion in Bouin–Hollande solution for 2 days. After the tissues had been treated with 10% EDTA in water at 4C for 3 days to demineralize the calcium carbonate crystals, they were dehydrated and embedded in Paraplast. Sections were cut at 4 μm and mounted on gelatin-coated slides.
Immunohistochemistry was performed by the indirect enzyme–antibody method. Deparaffinized sections were incubated sequentially at room temperature (RT) with the following reagents: 5% normal goat serum for 2 hr, rabbit anti-bPLP serum (1:8000) for 16 hr, peroxidase-conjugated donkey anti-rabbit IgG (1:200; Jackson Immunoresearch, West Grove, PA) for 1.5 hr. They were then rinsed three times with PBS, stained with 15 mg of 3,3′-diaminobenzidine tetrahydrochloride (Dojin Laboratory; Kumamoto, Japan) and 0.005% H2O2 in 100 ml of 0.05M Tris-HCl (pH 7.6) to detect peroxidase activity, rinsed with distilled water, stained with Mayer's hematoxylin, dehydrated by passage through a graded ethanol series, and mounted in Entellan (Merck; Darmstadt, Germany). The specificity of the immunostaining was also examined by a preabsorption test. The diluted antiserum was mixed with the antigen peptide at a final concentration of 1 or 10 μg/ml and pre-absorbed for 12 hr at 4C before being used in the specificity test.
Electron Microscopic Immunocytochemistry
The pituitary and its surrounding endolymphatic sac were fixed with a mixture of 0.5% glutaraldehyde, 4% paraformaldehyde, and 0.2% picric acid in 0.1 M cacodylate buffer, pH 7.4, at 4C for 2 hr and then decalcified by the procedure described above. The tissues were postfixed for 2 hr at 4C in 1% osmium tetroxide and then reduced with 1.5% potassium ferrocyanide in the same buffer. They were dehydrated through a graded ethanol series, infiltrated with propylene oxide, and embedded in an Epon/Araldite mixture. Ultrathin sections were cut with a Reichert Ultracut-E microtome (Reichert–Jung; Vienna, Austria) equipped with a diamond knife, mounted on nickel grids, and treated according to the following procedure: (a) etching in 3% H2O2 for 5 min at RT; (b) rinsing well with water; (c) bleaching with saturated sodium metaperiodate solution for 20 min (Bendayan and Zollinger 1983); and (d) rinsing well with water. The sections were immunolabeled using the immunogold method as described previously (Tanaka et al. 1992) and incubated with rabbit anti-bPLP serum (1:2000) and then with goat anti-rabbit IgG conjugated with 10-nm gold particles (Bio-Cell; Cardiff, UK). After immunolabeling, the sections were stained with uranyl acetate and lead citrate and then examined with a Hitachi H-7500 at 80 kV.
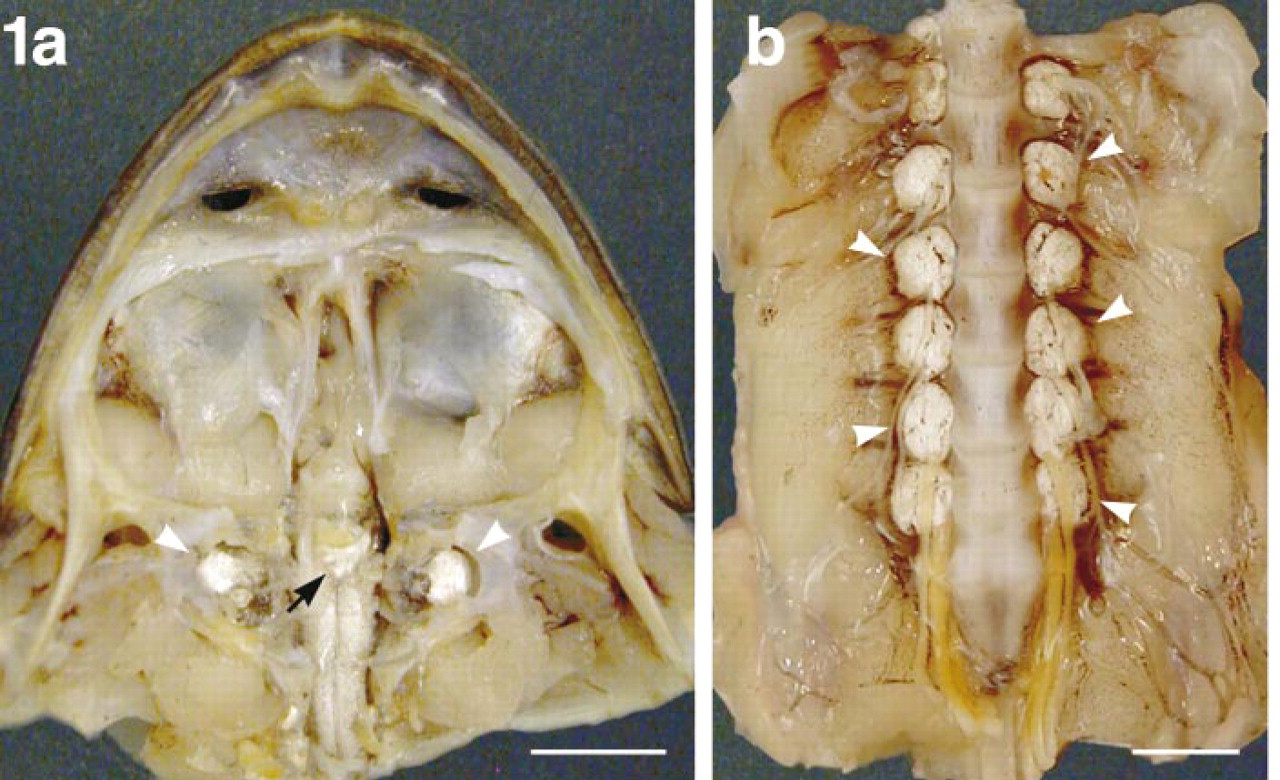
Endolymphatic sac system. (
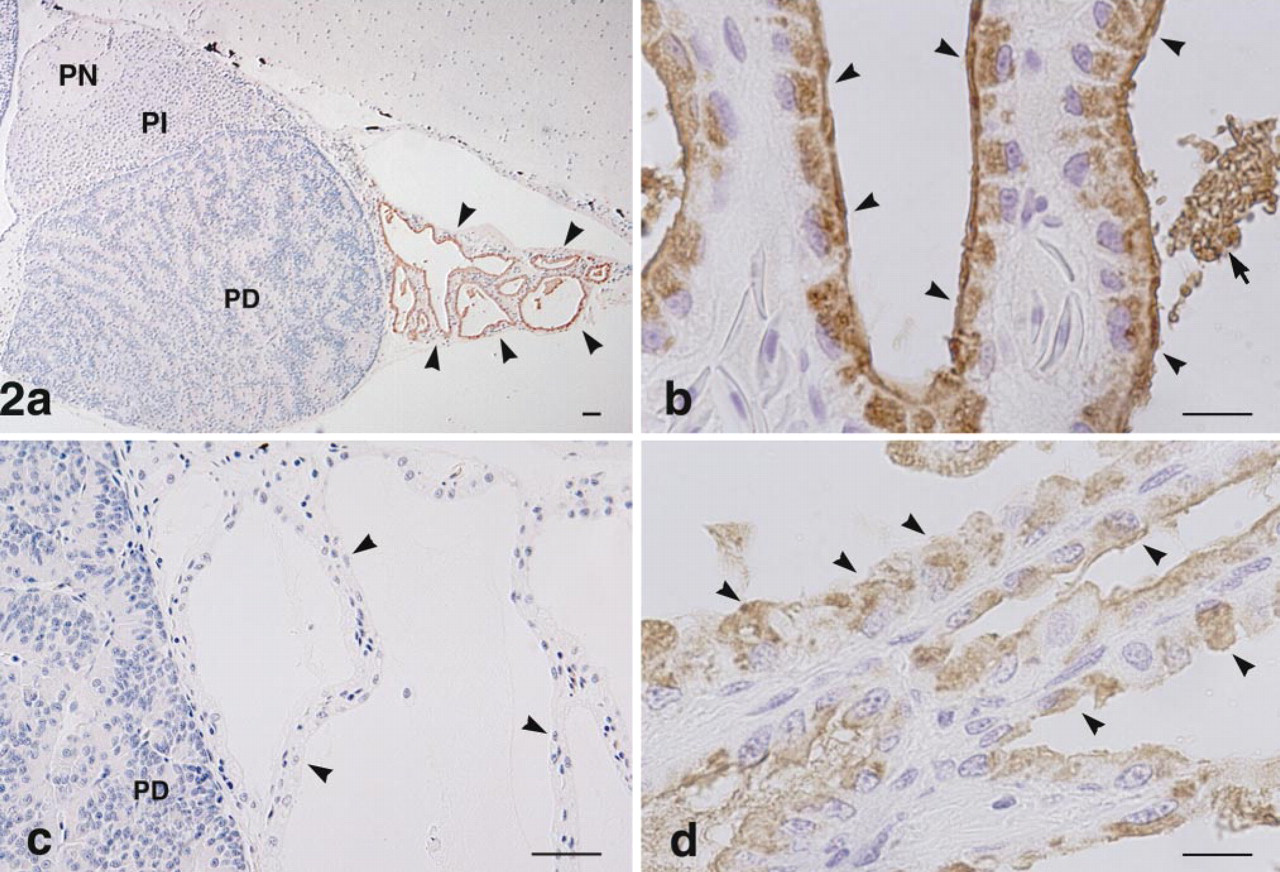
Immunocytochemical localization of bPLP in the pituitary gland and endolymphatic sac adhering to the caudal portion of the pituitary (
Results
Gross anatomy revealed that the caudal part of the bullfrog pituitary gland was covered by a sac containing a large quantity of calcium carbonate. This sac was connected to the membranous labyrinth in the inner ear and also to the endolymphatic sac (referred to as the paravertebral lime sac) which extends along the entire length of the spinal canal (Figure 1). In the tissue sections, the endolymphatic sac attached to the caudal region of the pars distalis was observed as a tissue with a follicular structure. This follicular structure consisted of a monolayer of cuboidal epithelial cells, with the basal side surrounded by a thin layer of connective tissue and blood vessels. When sections, including the pituitary gland and its surrounding endolymphatic sac, were immunostained with antiserum against bPLP, no immunoreaction was observed in any of the cells in any part of the pituitary gland (pars distalis, pars intermedia, and pars nervosa). However, the endolymphatic sac was immunopositive for this antiserum (Figure 2a). The apical side of the cytoplasm in the epithelial cells of the sac displayed an intense positive reaction with the antiserum (Figure 2b). The inclusion materials in the lumen of the follicular structures were also positively immunostained. These immunostainings were completely eliminated by preabsorption of the antiserum with 1 μg/ml of the antigen (Figure 2c). Similarly, when we examined the paravertebral lime sac, a positive reaction for bPLP was seen in the apical region of the simple cuboidal epithelial cells (Fig. 2d).
To test the specificity of the antiserum toward the bullfrog endolymphatic sac, the calcium carbonate crystals in the sac, and the anterior pituitary, we conducted Western blotting analysis of their extracts. In the extract of endolymphatic sac and calcium carbonate crystals in the endolymphatic sac, the antiserum detected a major band at 20.4 kD and a minor smear band between 24.6 and 30 kD with the sample from the endolymphatic sac, and two bands at 20.4 and 21.7 kD, and several diffuse bands between 24.6 and 30 kD with that of the calcium carbonate crystals. No band was detectable in the anterior pituitary gland extract (Figure 3A). The bands above were not detected when anti-bPLP was preabsorbed with the peptide used as immunogen (Figure 3B). To confirm that the immunoreactive bands are glycosylated, we performed a digestion experiment using pepide-N-glycosidase F. After digestion, most of the stained bands were converted to a band of 16.4 kD, suggesting that the bands of apparent higher molecular mass represented glycosylated forms of the 16.4-kD PLA2-like protein (Figure 3C).
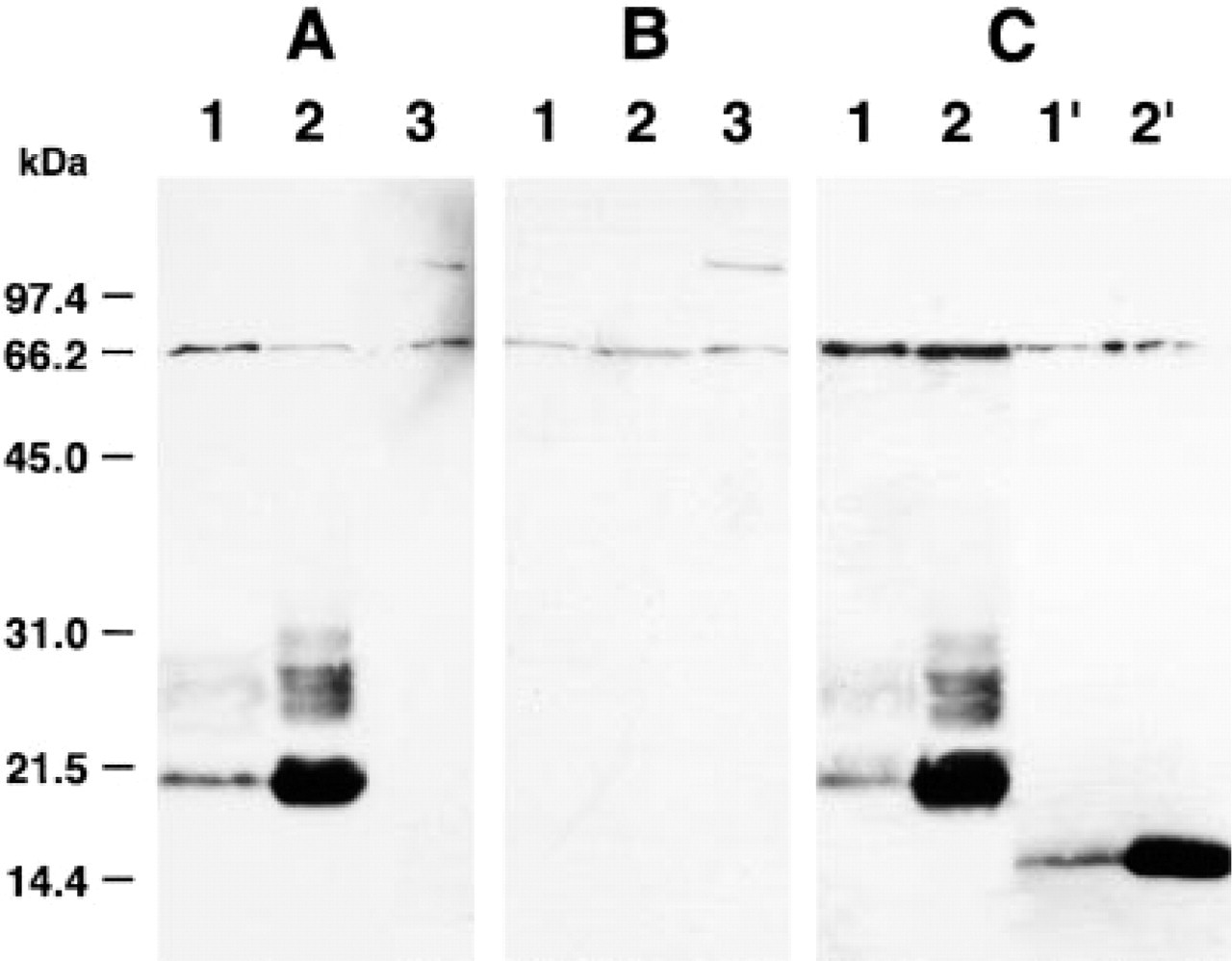
Characterization of anti-bPLP serum by Western blot analysis. (
Electron microscopy revealed that the cuboidal cells in the endolymphatic sac contained two kinds of secretory granules in their apical cytoplasm: one was large, polymorphic, and electron-lucent; the other type was dense and was fewer in number. The cuboidal cells in the sac had many mitochondria in its apical cytoplasm and well-developed microvillus-like structures at its apex. The surface of the follicular epithelium was covered by an amorphous thin layer, which appeared to have been derived from secretory materials (Figure 4a). These ultrastructural characteristics are very similar to those of the endolymphatic sac of tree frogs reported by Kawamata et al. (1987). When ultrathin sections were immunolabeled for bPLP, immunogold particles were seen on both types of secretory granules, but more intense labeling was found in the secretory granules with the dense matrix (Figure 4b). No immunolabeling for bPLP was detectable in the anterior pituitary glands (data not shown).
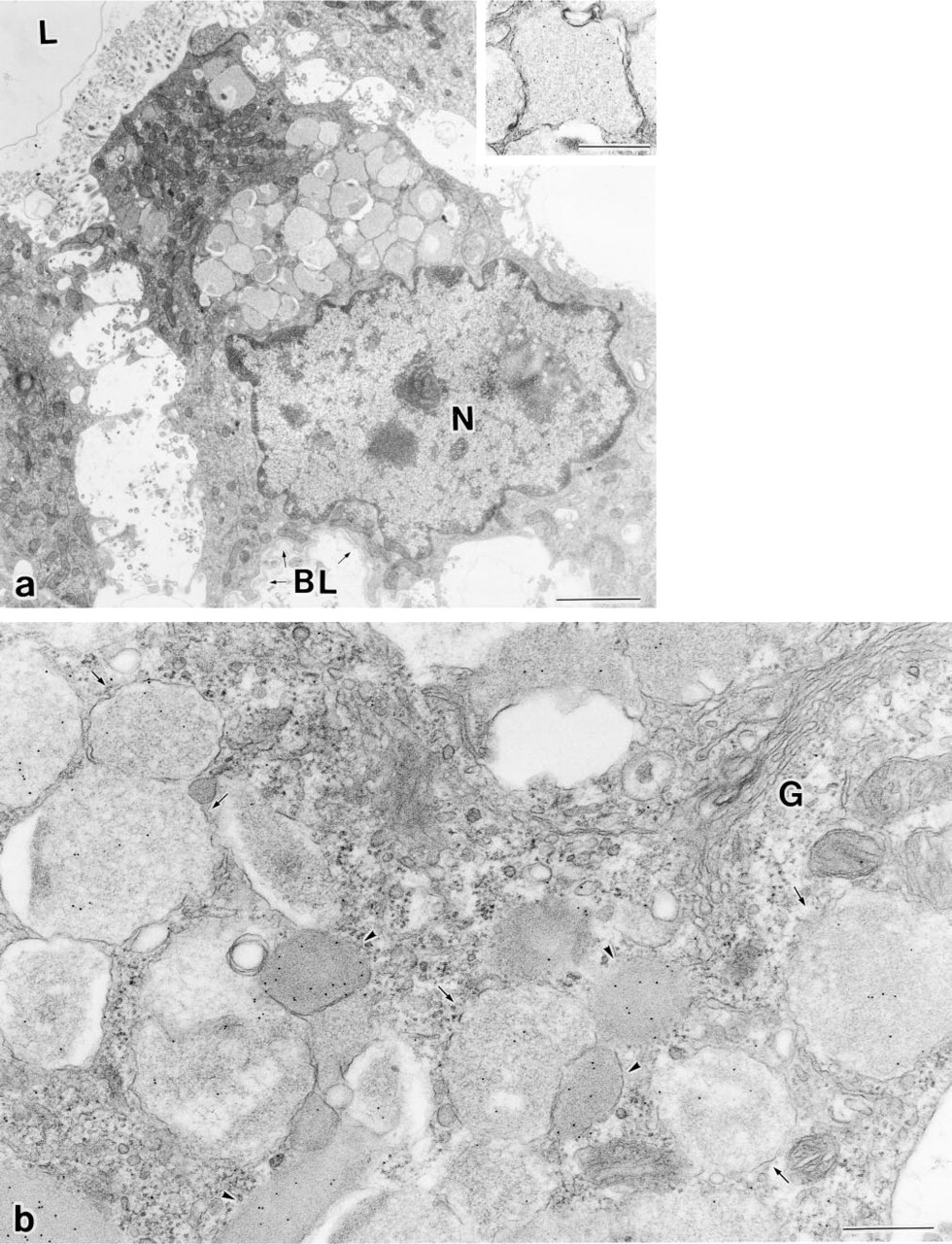
Electron micrographs of epithelial cells of the endolymphatic sac immunolabeled for PLP. (
Discussion
In this experiment bPLP, which was isolated as a minor fraction during purification of bullfrog TSH, was studied immunocytochemically. Immunoreactivity was found in the epithelial cells of the endolymphatic sac surrounding the pituitary gland but not in any anterior pituitary cells, supporting our view that the bPLP was mainly derived from the endolymphatic sac adhering to the pituitary.
Because the amphibian endolymphatic sac connects with the membranous labyrinth, the tiny crystals of calcium carbonate that are found in the endolymphatic sac are believed to be homologous to the otoconia in the utriculus and the sacculus. Our scanning electron microscopical examination indicated that the crystals in the bullfrog endolymphatic sac were of a prismatic shape (unpublished data), as reported for several species of amphibians (Pote and Ross 1993; Kido and Takahashi 1997), and exhibited the typical morphology of aragonitic otoconia. In fact, the N-terminal 45 amino acid residues of the bPLP were identical to those of otoconin-22, the major protein of aragonitic otoconia in the Xenopus saccule (Pote et al. 1993). The antiserum used in the present study was developed according to the sequence corresponding to the N-terminal region of otoconin-22 as an antigen. Therefore, the protein existing in the endolymphatic sac and showing immunoreactivity with this antiserum is considered to be involved in creating conditions that favor nucleation and then controlling crystal growth of the calcium carbonate lattice in otoconia. Western blotting analysis of the extracts of bullfrog endolymphatic sac and calcium carbonate crystals showed one major band of 20.4 kD, which is consistent with that of Xenopus otoconin-22 (Pote et al. 1993). Analysis of the extract of calcium carbonate crystals revealed an additional band at 21.7 kD and several bands between 24.6 and 30 kD. These are presumed to be glycosylated forms with a different number of sugar side chains. It has been reported that Xenopus otoconin-22 has two potential N-glycosylation sites (Pote et al. 1993). Digestion experiments with peptide-N-glycosidase F revealed a single band of 16.4 kD, its value being close to the molecular mass (14.6 kD) calculated from the amino acid sequence of Xenopus octonin-22 (Pote et al. 1993). Furthermore, the present Western blotting analysis using the preabsorbed antiserum confirmed that these immunoreactive bands were specific to the antiserum. Taken together, these findings strongly suggest that the bPLP recognized by this antiserum is homologous to Xenopus otoconin-22. In addition, the mammalian otoconial protein otoconin-90 (also called otoconin-95), which was recently identified by cDNA cloning, has a molecular mass of 85–105 kD (Wang et al. 1998; Verpy et al. 1999). According to Pote and Ross (1991), the calcitic otoconia of the amphibian utricle may contain the same matrix protein as mammalian otoconia, i.e., otoconin-90, and each crystal polymorph may be associated with a unique matrix protein forming the calcium carbonate crystal.
In this study, the Western blotting analysis and immunocytochemical staining showed that the anterior pituitary gland does not have PLA2-immunoreactive bands, strongly suggesting that the anterior pituitary gland does not contain such materials. Taken together, the present findings indicate that the PLP fraction obtained during TSH purification was most probably derived from the endolymphatic sac contained in the starting material for TSH purification.
There are some arguments about the precise cellular sites at which otoconia are formed. Kawamata (1991) reported that an organic material and calcium carbonate make complexes in the lumen after each has been secreted from supporting cells, whereas Harada et al. (1998) stated that calcium binds to organic material in intracellular compartments and then the complexes are secreted into the extracellular lumen. The present immunoelectron microscopic study revealed that PLP was localized in secretory granules in the epithelial cells, suggesting that PLP is produced in the epithelial cells and is then secreted into the lumen of the endolymphatic sac. Further investigation of PLP in the endolymphatic sac is expected to contribute to the clarification of the process of otoconium formation.
Footnotes
Acknowledgements
Acknowledgments
Supported in part by a Grant-in-Aid for Scientific Research from the Ministry of Education, Science, Sports, and Culture of Japan (ST).
