Abstract
C
In human epidermal keratinocytes and MCF-7 breast cancer cells, CYP1B1 is inducible by aryl hydrocarbon receptor ligands (Christou et al. 1994; Sutter et al. 1994). Therefore, CYP1B1 is believed to be an extrahepatic route of xenobiotic metabolism. It has been shown that CYP1B1 is involved in activation of many human promutagens and procarcinogens (Shimada et al. 1996). Bioactivation of promutagens and procarcinogens can irreversibly damage macromolecules and thus contribute to tumorigenesis (Guengerich 1994). High expression of CYP1B1 has been detected in a wide range of human tumors (Murray et al. 1997).
Studies on human uterine myometrium, MCF-7 cells, and a human lymphoblastoid cell line demonstrated that CYP1B1 protein is involved in the metabolism of 17β-estradiol and testosterone (Spink et al. 1994; Liehr et al. 1995; Hayes et al. 1996; Crespi et al. 1997). The metabolism of 17β-estradiol, preferentially by 4-hydroxylation by CYP1B1, forms 4-hydroxyestradiol, a metabolite suggested to be important in estrogen-induced carcinogenesis (Liehr et al. 1995; Hayes et al. 1996).
The wide distribution of CYP1B1 in human organs and its involvement in biotransformation of exogenous compounds as well as endogenous substrates, such as steroid hormones, suggest that this enzyme may have a significant role in tumorigenesis and in endogenous hormone metabolism. Although the distribution of CYP1B1 mRNA and protein in number of human normal tissues has been well documented (Sutter et al. 1994; Shimada et al. 1996; Hakkola et al. 1997; Rieder et al. 1998; Tang et al. 1999), the cells that express CYP1B1 in individual tissue have not yet been determined. To evaluate the functional role of CYP1B1 in certain tissues, it is necessary to identify specific cells that express this enzyme.
In this study, using in situ hybridization (ISH) and immunohistochemical (IHC) techniques, we show the following: (a) CYP1B1 mRNA and protein are expressed in a variety of human normal tissues; (b) CYP1B1 mRNA and protein are expressed in parenchymal and stromal cells of tissues examined; and (c) CYP1B1 protein is localized mainly in the nuclei of the cells, although in some cell types it is also present in cytoplasm.
Materials and Methods
Tissue Samples
Samples of human normal tissues including brain, kidney, prostate, breast, ovary, uterus, cervix, lymph node, and liver were obtained from Cooperative Human Tissue Network (University of PA Medical Center; Philadelphia, PA). Each tissue sample was fixed in 10% neutral buffered formalin for 24 hr, embedded in paraffin, sectioned, and processed for ISH or IHC.
CYP1B1 Antisense RNA Probe
The pRc/CMV plasmid containing a 1.7-kb fragment of the human CYP1B1 open-reading frame of DNA (Sutter et al. 1994) was linearized with HindIII and purified by phenol/chloroform extraction and ethanol precipitation. The linearized plasmid was then used as a template for antisense digoxigenin (DIG)-labeled riboprobe synthesis using SP6 polymerase (Roche Molecular Biochemicals; Indianapolis, IN). The synthesized RNA probe was then fragmented to a size of around 200 bases by alkaline hydrolysis.
In Situ Hybridization
Three to six samples of each tissue were processed for ISH. Paraffin sections (5 μm) were deparaffinized in xylene, hydrated in 100, 95, and 70% ethanol, and treated with 0.2 M HCl. Proteinase K (10 μg/ml; Roche Molecular Biochemicals) digestion was performed at 37C for 15 min. The proteinase K reaction was stopped by adding glycine (2 mg/ml in PBS; Sigma, St Louis, MO). Sections were acetylated with 0.25% acetic anhydride (Sigma) in triethanolamine (Sigma) for 15 min at room temperature (RT) to reduce background. Sections were prehybridized for 30 min at 50C with hybridization buffer devoid of probe. Sections were then overlaid with a hybridization mixture consisting of 50% formamide (Sigma), 4 × SSC (Sigma), 500 μg/ml of yeast tRNA (GIBCO BRL; Gaithersburg, MD), 100 mg/ml of dextran sulfate (Sigma), 1 × Denhardt's solution (Sigma), and 50 ng/ml of probe, covered with frame seals, and incubated overnight at 50C. After hybridization, the sections were washed in 2 × SSC and 50% formamide at 50C for 15 min, and treated with RNase A (50 μg/ml in PBS; Roche Molecular Biochemicals) for 30 min at 37C. After washes in Tris-HCl buffer (pH 7.5) and 2 × SSC with 50% formamide, sections were blocked with blocking reagent (Roche Molecular Biochemicals) and 5% normal sheep serum (Sigma) for 30 and 45 min, respectively. The sections then were incubated for 1 hr at RT with alkaline phosphatase-conjugated anti-digoxigenin antibodies (sheep anti-digoxigenin-AP Fab fragments; Roche Molecular Biochemicals) diluted 1:100. Finally, the signal was visualized by overnight incubation with freshly prepared substrate solution (nitroblue tetrazolium chloride and 5-bromo-4-chloro-3-indolyl-phosphate toluidine salt in Tris-HCl buffer, pH 9.5; Roche Molecular Biochemicals) with 0.25 mg/ml levamisole (Sigma). The color reaction was stopped with Tris-HCl buffer, pH 8.0, with 1 mM EDTA (Sigma). Sections were then washed in sterile water, mounted with Crystal/Mount (Biomeda; Foster City, CA) and examined by light microscopy (BX40; Olympus, Japan). Negative controls included (a) hybridization with DIG-labeled Neo RNA (recommended and provided by Roche Molecular Biochemicals) fragmented to a size of around 200 bases by alkaline hydrolysis, (b) omission of either the antisense RNA probe or the anti-digoxigenin antibodies, or (c) RNase A treatment before hybridization.
Anti-CYP1B1 Antibody
An anti-peptide antibody was raised against a 14-mer synthetic peptide (ME001; Alpha Diagnostic, San Antonio, TX) corresponding to the amino acid sequence of human CYP1B1 (Tang et al. 1999). Rabbit-derived antiserum contained high antibody titers as judged by ELISA. In Western blotting analysis, the purified antibody recognized a single 56-kD protein band in microsomes prepared from human and rodent tissues. No significant crossreactivity to either human CYP1A1 or human CYP1A2 protein was detected. Immunoprecipitation and immunoinhibition results indicated that the antiserum recognized the non-denatured hu- man CYP1B1 protein but did not inhibit its enzyme activity (Tang et al. 1999).
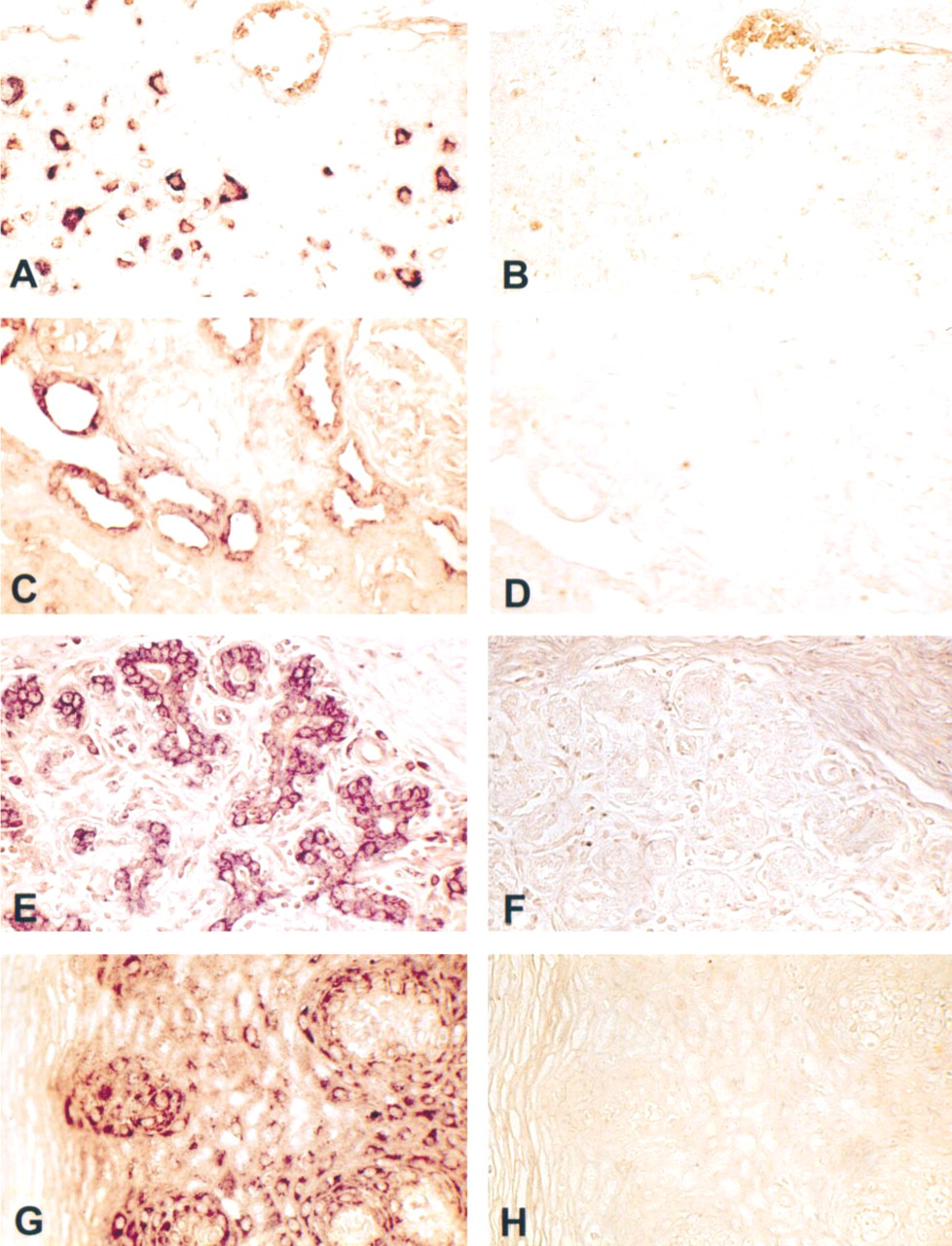
In situ hybridization for CYP1B1 mRNA in human normal tissues. Shown are representative photomicrographs of sections of brain (
Immunohistochemistry
Rabbit-derived anti-CYP1B1 antiserum or purified antibodies were used for IHC detection of CYP1B1 protein in human tissue samples. Formalin-fixed, paraffin-embedded human tissue sections were processed for IHC demonstration of CYP1B1 protein by the Biotin–ExtrAvidin–Peroxidase detection system (Sigma). Antiserum or purified antibodies were used at 1:200 dilution. Endogenous peroxidase was inhibited by incubation with freshly prepared 3% hydrogen peroxide with 0.1% sodium azide. Nonspecific staining was blocked with 0.5% casein and 5% normal goat serum. Tissue sections were incubated with biotinylated goat anti-rabbit antibodies and ExtrAvidin-conjugated horseradish peroxidase. Staining was developed with diaminobenzidine substrate and sections were counterstained with hematoxylin. Negative controls included (a) replacement of antibodies with normal rabbit serum (Sigma) or PBS, and (b) neutralization of antibodies with an excess of antigenic ME001 peptide.
For evaluation of CYP1B1 expression in astrocytes of human brain cortex, immunostaining of brain sections with CYP1B1 antibodies was followed by immunohistochemical detection of glial fibrillary acidic protein (GFAP). Briefly, GFAP antibodies (mouse monoclonal anti-human GFAP; Lab Vision, Fremont, CA) were used at 1:50 dilution. Nonspecific staining was blocked with 5% normal goat serum (Sigma). The brain sections were incubated with biotinylated goat anti-mouse antibodies and ExtrAvidin-conjugated alkaline phosphatase (Sigma). Staining was developed with New Fuchsin substrate (Biogenex; San Ramon, CA). For inhibition of endogenous alkaline phosphatase activity, 0.6 mg/ml levamisole (Sigma) was added to the substrate mix. Sections were counterstained with hematoxylin. Normal mouse serum or PBS replaced GFAP antibodies in negative controls.
Results
In Situ Hybridization
Expression of CYP1B1 mRNA in different human tissues was analyzed by ISH using a DIG-labeled antisense probe. ISH analysis demonstrated that CYP1B1 mRNA was expressed in all extrahepatic tissues examined. However, the intensity of staining varied among the tissues. Intense staining for CYP1B1 mRNA was observed in the cytoplasm of neurons of brain cortex, glandular cells of breast, stratified squamous nonkeratinizing epithelial cells of ectocervix, and epithelial cells of distal tubules in kidney (Figure 1). The signal obtained from other extrahepatic tissues was relatively weak. All three liver samples processed for ISH were negative. None of the negative controls showed any positive staining.
Immunohistochemistry
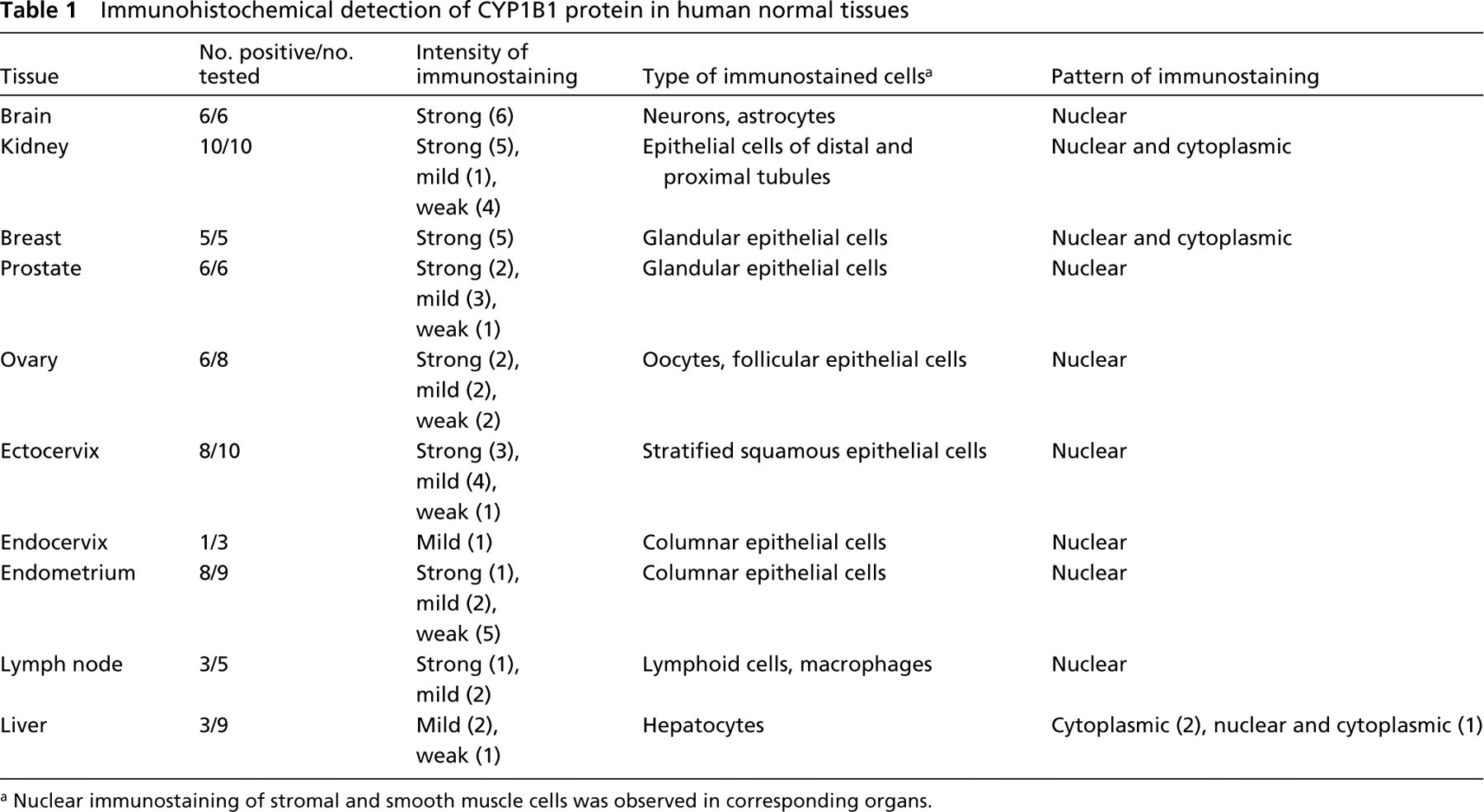
Expression of CYP1B1 protein in different types of human tissues was evaluated by IHC using rabbit anti-CYP1B1 antiserum or purified antibodies. The results demonstrated that CYP1B1 protein was expressed in all tissue types examined. The protein was expressed in most specimens, being undetected in only nine of 62 samples of extrahepatic tissue (Table 1). All tissue samples were categorized according to the intensity of immunostaining and were assigned to one of three immunoreactivity groups: strong, mild, or weak (Table 1). No immunostaining was observed in negative controls.
Immunohistochemical detection of CYP1B1 protein in human normal tissues
Nuclear immunostaining of stromal and smooth muscle cells was observed in corresponding organs.
In human brain cortex, strong nuclear immunostaining of the majority of neurons was observed in all samples examined (Table 1; Figure 2A). Double labeling of brain sections with CYP1B1 and GFAP antibodies demonstrated that CYP1B1 protein was also expressed in the nuclei of a majority of astrocytes (Table 1; Figure 2B).
In the kidney, strong CYP1B1 immunoreactivity was detected in both nuclei and cytoplasm of distal tubules. However, weak nuclear and cytoplasmic immunostaining was also observed in some of the proximal tubules (Table 1; Figure 2C).
Immunostaining of mammary epithelia was not even. In the same sample, secretory cells of some lobules displayed only nuclear immunostaining, whereas cells in other lobules exhibited both nuclear and cyto-plasmic immunoreactivity (Table 1; Figure 2D).
Epithelial cells of prostate, cervix, and uterus, oo-cytes and follicular cells in ovary, and lymphoid cells and macrophages in lymph nodes exhibited only nuclear immunostaining, which ranged from weak to strong in different samples (Table 1; Figures 2E–2J).
Stromal cells and muscle cells of organs such as uterus and cervix were also positively stained; the protein was expressed mostly in nuclei (Table 1; Figure 2K).
Only three of nine liver samples examined were CYP1B1-positive. Two of them exhibited mild cyto-plasmic immunostaining, and in the third sample both nuclei and cytoplasm were weakly immunoreactive (Table 1; Figure 2L).
Discussion
The present results demonstrated that CYP1B1 mRNA and protein are expressed in different types of human normal tissues (Figures 1 and 2). These results are not consistent with a previous study that found CYP1B1 protein only in tumor tissues (Murray et al. 1997). However, CYP1B1 mRNA has been detected in many human normal tissues by Northern blotting analysis (Sutter et al. 1994; Shimada et al. 1996; Rieder et al. 1998) and by RT-PCR (Huang et al. 1996; Hakkola et al. 1997; Vadlamuri et al. 1998). In a variety of human tissues, expression of CYP1B1 protein was recently demonstrated by Western blotting (Tang et al. 1999). Therefore, our results confirm these observations of CYP1B1 mRNA and protein expression in different human normal tissues.
Earlier, it had been shown that CYP1B1 mRNA was present in human brain tissue (Sutter et al. 1994; Shimada et al. 1996; Rieder et al. 1998), but the cell types expressing the RNA were not identified. Recently, CYP1B1 immunoreactivity was detected at the blood–brain interface areas of the microcirculation in human temporal lobe (Rieder et al. 2000). ISH analysis performed in our study demonstrated that, in human brain cortex, CYP1B1 mRNA is expressed mainly in neurons. Correspondingly, immunohistochemical analysis with anti-CYP1B1 antibodies also showed that neurons express CYP1B1 protein and that it is localized in the nucleus of the cell. These results indicate that CYP1B1 protein is constitutively expressed in human neurons.
According to ISH analysis and immunohistochemistry with CYP1B1 antibodies, the main cellular subset expressing CYP1B1 in human brain is neurons. Morphologically, however, astrocytes are hardly distinguishable from small neurons. To determine if astrocytes also express CYP1B1 protein, we performed double immunolabeling with CYP1B1 and GFAP antibodies. The double labeling clearly demonstrated that most astrocytes also express CYP1B1 protein and that in these cells the enzyme is also localized in the nucleus.
The intracellular pattern of CYP1B1 immunostaining in other tissues was mostly similar to that in brain cells: epithelial cells of prostate, uterus, and cervix, oocytes and follicular cells in the ovary, lymphoid cells and macrophages in lymph nodes, and stromal and muscle cells in different organs displayed mostly nuclear staining. However, in tubule cells of the kidney and secretory cells of the mammary gland, immunostaining for CYP1B1 was observed in both nucleus and cytoplasm. The present study demonstrates for the first time nuclear localization of CYP1B1 protein, suggesting a functional role in the nucleus.
The human liver exhibits the lowest levels of CYP1B1 mRNA (Shimada et al. 1996) and protein (Tang et al. 1999). In agreement with these observations, we were not able to detect expression of CYP1B1 mRNA in the liver, and only three of nine liver samples examined by IHC tested positive for CYP1B1 protein. Two of these samples displayed mild cytoplasmic immunostaining, and in one case both nuclei and cytoplasm were weakly labeled. Therefore, some hepatocytes were the only cells to contain cytoplasmic immunoreactivity in the absence of nuclear immunoreactivity. It is noteworthy that, unlike CYP1B1-negative liver samples, all CYP1B1-positive liver samples were obtained at autopsy. The absence of the signal in nuclei of hepatocytes in two of three of these autopsy samples suggests that synthesis of the CYP1B1 protein in these livers may have been induced by terminal metabolic events.
The constitutive expression and wide distribution of CYP1B1 mRNA and protein in human normal tissues supports the possibility of an important regulational role and suggests its involvement in local metabolism of xenobiotics. Human CYP1B1 is capable of activation of chemically diverse human procarcinogens (Shimada et al. 1996) and of 4-hydroxylation of estradiol (Spink et al. 1994; Liehr et al. 1995; Hayes et al. 1996). Therefore, CYP1B1 may also be important in bioactivation of non-toxic compounds to reactive toxic substances, which can damage cells and lead to neoplastic transformation or cell death. For example, expression of CYP1B1 in human neurons may be associated with certain xenobiotic-induced neurodegenerative diseases, and the presence of the CYP in epithelial, lymphoid, or stromal cells may contribute to neoplasia. The role of this CYP in the metabolism of estradiol or testosterone in the brain is altogether unclear. However, it is tempting to speculate that it may have some role in gender-related differences in neurobiology.
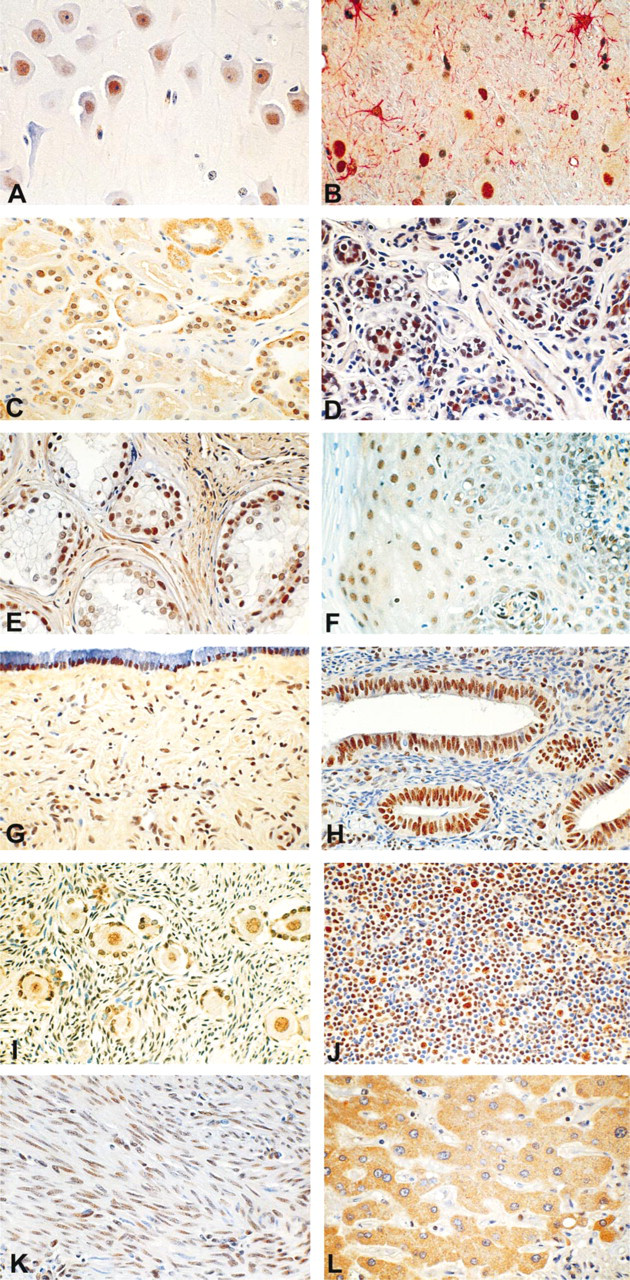
Immunohistochemical detection of CYP1B1 protein in human normal tissues. Shown are representative photomicrographs of sections of brain (
