Abstract
Vascular endothelial growth factor (VEGF) plays a major role in angiogenesis, which is essential for both healing of injured tissue and proliferation of carcinoma cells. In this study we elucidated the expression and role of VEGF in rat liver regeneration after partial hepatectomy. VEGF expression was mainly detected in periportal hepatocytes and reached a maximal level 48–72 hr after partial hepatectomy by both immunohistochemistry and in situ hybridization. Similarly, immunohistochemistry for Ki-67 showed that the proliferative activity of sinusoidal endothelial cells was highest in the periportal area and reached a maximal level 72 hr after partial hepatectomy. Moreover, neutralization of VEGF significantly inhibited proliferative activity of hepatocytes (p < 0.0001), as well as sinusoidal endothelial cells (p < 0.001), at 48 and 96 hr after partial hepatectomy. Conversely, injection of VEGF significantly promoted proliferative activity of hepatocytes (p < 0.0001) as well as sinusoidal endothelial cells (p < 0.0005) at 48 hr after partial hepatectomy. These results suggest that VEGF promotes proliferation of hepatocytes through reconstruction of liver sinusoids by proliferation of sinusoidal endothelial cells. Furthermore, these data point to a new therapeutic strategy, the use of VEGF and other hepatocyte growth factors in fulminant or severe acute hepatitis.
Keywords
A
In liver regeneration after partial hepatectomy (PH), both hepatocytes and nonparenchymal cells express VEGF mRNA (Mochida et al. 1996), suggesting that VEGF plays a significant role in liver regeneration. However, the expression and role of VEGF have not been examined in detail in liver regeneration. In this study we investigated the expression of VEGF in regenerating rat liver after PH by in situ hybridization (ISH) and immunohistochemistry (IHC). Furthermore, we elucidated the role of endogenous VEGF by its neutralization and subsequently assessing proliferative activity of SECs and hepatocytes. We found that VEGF was mainly expressed in periportal hepatocytes and we demonstrated that VEGF was significantly involved in proliferation of hepatocytes associated with proliferation of SECs after PH in rats.
Materials and Methods
Animals
Male Fisher rats weighing 200–250 g (Japan SCC; Shizuoka, Japan) were used in all experiments. Rats were kept at a controlled temperature (22C) under a 12-hr light-dark cycle and were maintained on a standard diet and water. All experiments were conducted in accordance with the National Institutes of Health Guidelines for the Care and Use of Laboratory Animals and were approved by the University of Kurume Institutional Animal Care and Use Committee.
Partial Hepatectomy (PH)
Rats underwent 70% PH under light ether anesthesia in the midmorning using the method of Higgins and Anderson (1931). Briefly, the liver was exposed through a 1–2-cm midline abdominal incision and the two anterior lobes were exteriorized, the vascular pedicles were ligated, and the lobes were excised. Rats were sacrificed at 0, 12, 24, 48, 72, 96, or 168 hr after PH. Numbers of rats used in each experiment are indicated in the figure legends.
Immunohistochemistry for VEGF
Liver tissues obtained from sacrificed rats were fixed in 10% formalin and embedded in paraffin. Liver tissue sections sliced at 3 μm were placed on silane-coated slides (DAKO; Kyoto, Japan). Sections were deparaffinized with xylene and immersed in absolute and 95% ethanol for 15 sec. The sections were incubated in methanol with 3% (v/v) H2O2 at room temperature (RT) for 10 min to block endogenous peroxidase activity. After incubation, sections were immersed in 85% and 75% ethanol for 15 sec and washed with water. After rehydration in 10 mM PBS with 0.05% Tween-20 (Wako; Osaka, Japan) (T-PBS), sections were preincubated with Protein Block Serum-Free (DAKO) at RT for 30 min. Sections were then incubated with either an anti-VEGF rabbit polyclonal antibody (Santa Cruz Biotechnology; Palo Alto, CA) diluted 1:250 (v/v) with T-PBS or normal rabbit IgG (Inter-Cell Technologies; Hopewell, NJ) diluted 1:2000 with T-PBS at 4C overnight. Sections were washed three times for 5, 10, and 15 min in T-PBS and incubated with anti-rabbit ENVISION PLUS (DAKO) at RT for 60 min. After washing in T-PBS again, sections were incubated in a solution containing 0.1% (w/v) 3-3′-diaminobenzidine-tetrahydrochloride (DAB) and 0.005% (v/v) H2O2 in 0.1 M Tris-HCl buffer (pH 7.6) at RT for 3 min. Nuclear counterstaining was performed with Mayer's hematoxylin for 30 sec.
Immunohistochemistry for Ki-67
Deparaffinized 3-μm sections of liver were immersed in ethanol and incubated to block endogenous peroxidase activity as above. Sections were then autoclaved in 10 mM citrate buffer at 121C for 5 min and cooled slowly to RT. After washing in T-PBS, sections were preincubated with Protein Blocking Serum-Free at RT for 30 min. Immunostaining for Ki-67, a marker for cell proliferation, was performed to evaluate the proliferation of hepatocytes and SECs (Gerdes et al. 1983, 1984). Sections were incubated with either an anti-Ki-67 mouse monoclonal antibody (Immunotech; Marseille, France) diluted 1:100 (v/v) with T-PBS or normal mouse serum diluted 1:2000 with T-PBS at 4C overnight. Sections were washed in T-PBS and incubated with a biotinylated anti-mouse IgG (Vectastain ABC Kit; Vector Labs, Burlingame, CA) diluted 1:200 (v/v) with T-PBS at RT for 30 min. After washing in T-PBS, sections were incubated with avidin-biotin-peroxidase complex (Vectastain ABC Kit) at RT for 30 min. Sections were then incubated in DAB and H2O2 as above. Nuclear counterstaining was performed with Mayer's hematoxylin for 10 min.
Double Immunostaining for Endothelial Nitric Oxide Synthase and Ki-67
To distinguish SECs from the other nonparenchymal liver sinusoidal cells, SECs were labeled with anti-endothelial nitric oxide synthase (eNOS) mouse monoclonal antibody (Rockey and Chung 1998). Deparaffinized 3-m sections of liver were immersed in ethanol and incubated to block endogenous peroxidase activity as above. Sections were then autoclaved in 10 mM citrate buffer as above and then preincubated with Protein Blocking Serum-Free for 30 min. Sections were incubated with anti-eNOS mouse monoclonal antibody (Transduction Laboratories; Lexington, KY) and anti-Ki-67 mouse monoclonal antibody (Immunotech) diluted 1:100 (v/v) with T-PBS at 4C overnight. Sections were washed in T-PBS and incubated with a biotinylated anti-mouse IgG (Vectastain ABC Kit) diluted 1:200 (v/v) with T-PBS at RT for 30 min. After washing in T-PBS, sections were incubated with avidin-biotin-peroxidase complex (Vectastain ABC Kit) at RT for 30 min. Sections were then incubated in DAB and H2O2 as above. Nuclear counterstaining was performed with Mayer's hematoxylin for 10 min.
In Situ Hybridization for VEGF
Antisense and sense riboprobes for VEGF were generated by subcloning an Sma I-Sma-I fragment (380 base pairs) of rat VEGF cDNA into the pGEM3Zf vector (Promega Biotech; Madison, WI) (Keck et al. 1989). The plasmids were used to generate digoxigenin-labeled antisense and sense riboprobes by T7 RNA polymerase using an RNA labeling kit (Boehringer Mannheim; Mannheim, Germany).
For ISH, liver tissues were fixed with 4% paraformaldehyde at 4C for 6 hr. After immersion in 15% sucrose/PBS at 4C for 6 hr, tissues were embedded in Tissue-Tek OCT compound (Miles; Elkhart, IN), and frozen at –75C. Liver tissue sections sliced at 6 μm were placed on silane-coated slides (DAKO). Sections were treated with 1 mg/ml proteinase K (Sigma; St Louis, MO), acetylated with 0.25% acetic anhydride, and incubated with a prehybridization solution containing 50% deionized formamide/2 × SSC at 42C for 1 hr. The hybridization solution contained 0.3 M NaCl, 1 mM EDTA, 10 mM Tris-HCl (pH 7.6), 120 mg/ml herring sperm DNA, 200 mg/ml yeast tRNA, 1 × Denhart's solution, 10% (w/v) dextran sulfate, 50% deionized forma-mide, and 50–100 ng/ml antisense- or sense-labeled riboprobe. After hybridization at 42C for 16 hr, the sections were washed twice with 50% formamide/2 × SSC at 42C for 20 min and treated with RNase A (Boehringer Mannheim) at 37C for 30 min. The sections were washed once with 2 × SSC and twice with 0.1 × SSC at 42C for 30 min. Probes were detected with a sheep polyclonal anti-digoxigenin Fab fragment conjugated to alkaline phosphatase, followed by development in nitroblue tetrazolium and 5-bromo-4-chloro-3-indolyl phosphate (all from Boehringer Mannheim).
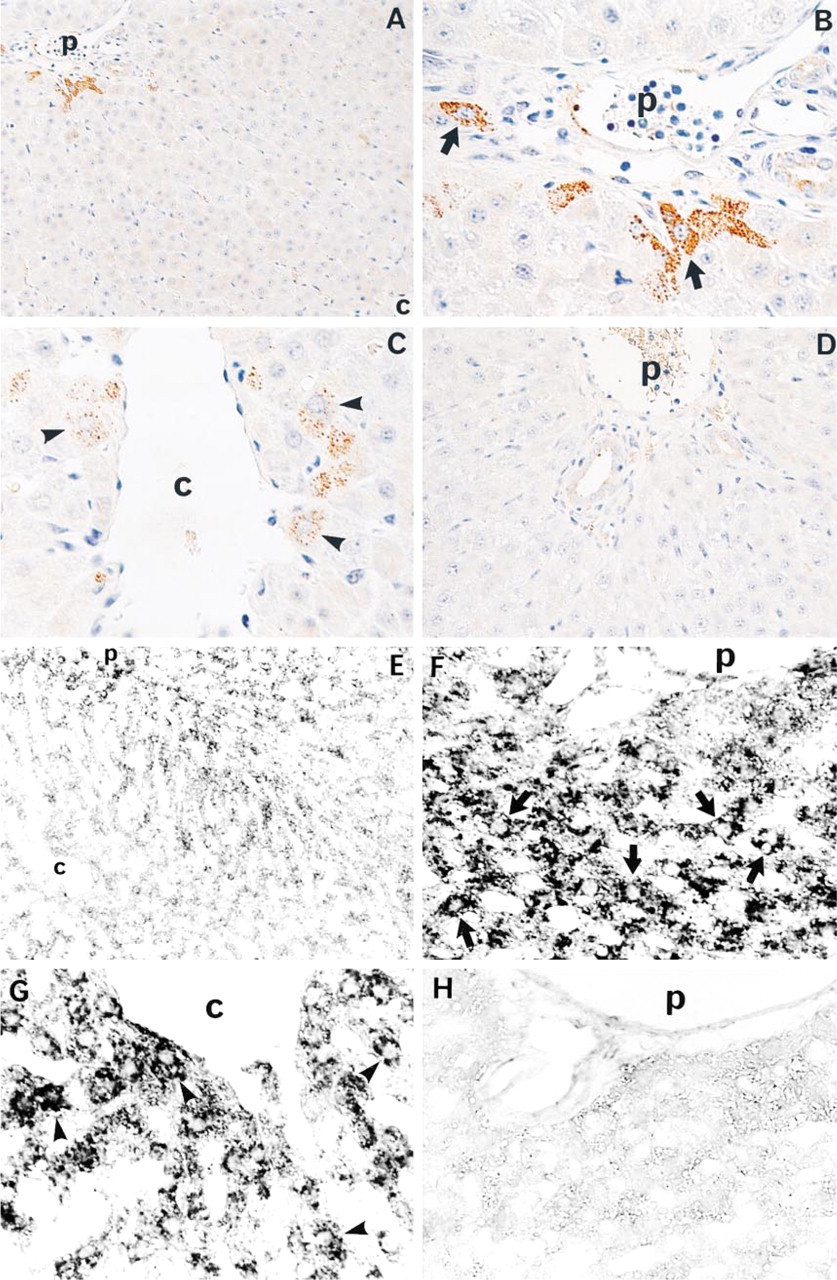
Expression of VEGF and VEGF mRNA in livers after PH. Immunohistochemical staining of VEGF in the liver lobules (
Morphometric Analysis for Cellular Expression of VEGF and Ki-67
We counted the number of hepatocytes positive for VEGF-protein staining and SECs positive for Ki-67 and eNOS staining in the periportal (within 100 μm of the portal area), perivenular (within 100 μm of the central vein), and mid-zonal areas, and the percentage of positive cells was calculated in each lobular area. The staining intensity for VEGF was evaluated by three grades: strongly positive, weakly positive, and negative.
Injection of Anti-VEGF Antibody or VEGF
To investigate the role of endogenous VEGF in liver regeneration, we studied the effect of anti-VEGF antibody on the proliferation of hepatocytes and SECs before and after the peak time point of Ki-67 expression in SECs. Moreover, we investigated the effect of exogenous VEGF for liver regeneration. In separate experiments, when rats underwent 70% PH, 200 μg/rat of anti-VEGF rabbit IgG (Toagosei; Tsukuba, Japan) (Asano et al. 1995) or normal rabbit IgG (Inter-Cell Technologies) was administered via the tail vein. These rats were sacrificed 48 or 96 hr after PH. Similarly, 500 ng/rat of VEGF (Toagosei) was administered 0 and 24 hr after PH. These rats were sacrificed 48 hr after PH. Numbers of rats used in each experiment are indicated in the figure legends.
Statistical Analysis
All data are expressed as means ± SD. Differences between groups were analyzed by the Mann-Whitney U-test. Comparisons between multiple groups were performed by oneway ANOVA, followed by Fisher's protected least significant difference post-hoc test. p < 0.05 was considered statistically significant.
Results
IHC and ISH for VEGF
In the IHC study, VEGF was detected in a few hepatocytes immediately after PH. At 48 hr after PH, VEGF was mainly detected in periportal hepatocytes and less commonly in perivenular hepatocytes (Figures 1A–1C). ISH demonstrated VEGF mRNA mainly in periportal and perivenular hepatocytes (Figures 1E–1G). Staining was absent when normal rabbit IgG was substituted for the primary antibody or when sense probe was substituted for antisense probe as a control (Figures 1D and 1H).
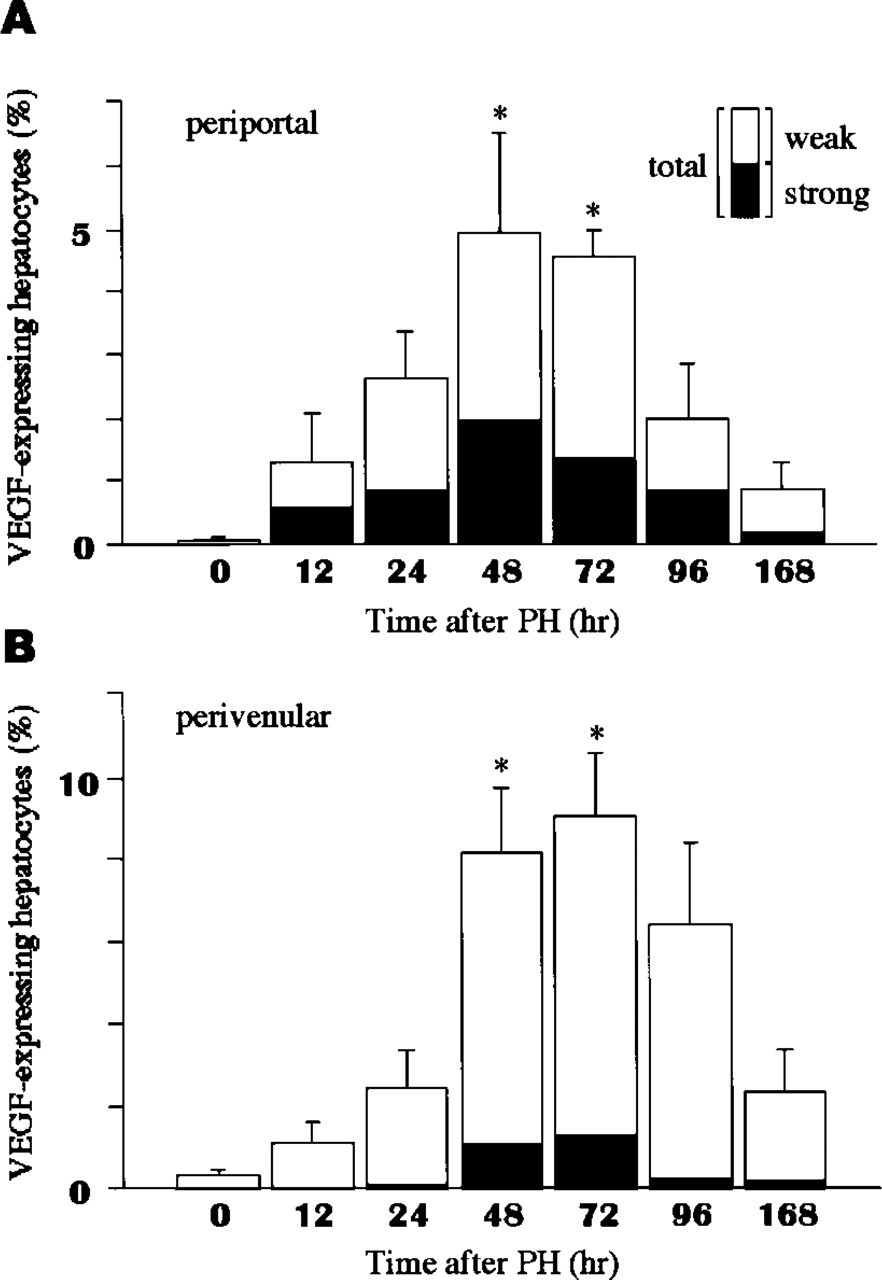
Morphometric analysis for VEGF-expressing hepatocytes in periportal (
Morphometric Analysis for VEGF-expressing Hepatocytes
The percentage of VEGF-expressing hepatocytes progressively increased and reached maximal levels in periportal and perivenular areas 48–72 hr after PH (Figures 2A and 2B). The percentage of hepatocytes strongly expressing VEGF was significantly higher in periportal areas than in perivenular areas (p < 0.005).
Immunohistochemistry for Ki-67 and eNOS
Immediately after PH, Ki-67 was detected in a few hepatocytes and non-parenchymal cells in the liver. At 48 hr after PH, Ki-67 was mainly detected in periportal and midzonal hepatocytes and in some nonparenchymal cells. Endothelial NOS was detected in SECs and the other endothelial cells (Figures 3A and 3B). Staining was absent when normal mouse serum was substituted for the primary antibody (data not shown).
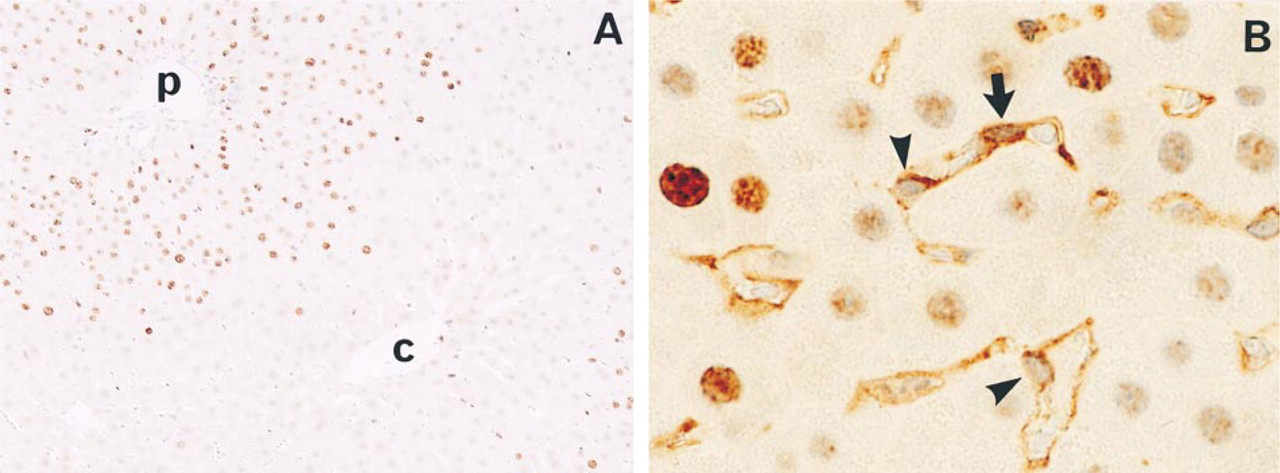
Immunohistochemical staining of Ki-67 and eNOS in livers after PH. Ki-67 was detected in the nuclei and eNOS was detected in the cytoplasm. Double-positive cells for Ki-67 and eNOS indicated proliferative endothelial cells. Note Ki-67-positive (arrow) and -negative (arrowheads) SECs (
Morphometric Analysis for Ki-67-expressing SECs
The percentage of Ki-67-expressing SECs was progressively increased after 24 hr and reached maximal levels 72 hr after PH in all three areas. At 72 hr after PH, the percentage of Ki-67-expressing SECs in the periportal area was significantly higher than in the perivenular or midzonal areas (Figure 4).
Effects of Anti-VEGF Antibody and VEGF on Proliferative Activity of SECs and Hepatocytes
The percentage of Ki-67-expressing SECs 48 hr after PH was significantly lower in anti-VEGF antibodytreated rats and, conversely, was higher in VEGF-treated rats than in untreated rats or control rabbit IgG-treated rats, respectively (p < 0.001 and p < 0.0001) (Figure 5A). Similarly, at 96 hr after PH, the percentage of Ki-67-expressing SECs was significantly lower in anti-VEGF antibody-treated rats (p < 0.0005) (Figure 5B). The percentage of Ki-67-expressing hepatocytes 48 hr after PH was significantly lower in anti-VEGF antibody-treated rats and, conversely, was higher in VEGF-treated rats than in untreated rats or control rabbit IgG-treated rats, respectively (p < 0.0001 and p < 0.0005) (Figure 5C). Similarly, at 96 hr after PH, the percentage of Ki-67-expressing hepatocytes was significantly lower in anti-VEGF antibody-treated rats (p < 0.0005) (Figure 5D). The decreased percentage of Ki-67-expressing SECs in anti-VEGF antibody-treated rats compared with untreated rats was almost similar at 48 and 96 hr after PH. However, the decreased percentage of Ki-67-expressing hepatocytes was higher at 96 hr than at 48 hr after PH.
Discussion
This study is the first to demonstrate that VEGF expression in regenerating rat liver occurs predominantly in periportal hepatocytes. In addition, IHC staining for Ki-67 demonstrated cell proliferation of both SECs and hepatocytes after PH. Finally, anti-VEGF rabbit IgG reduced and, conversely, VEGF promoted the proliferative activity of hepatocytes as well as SECs. These results suggest that VEGF plays a significant role in promoting rat liver regeneration.
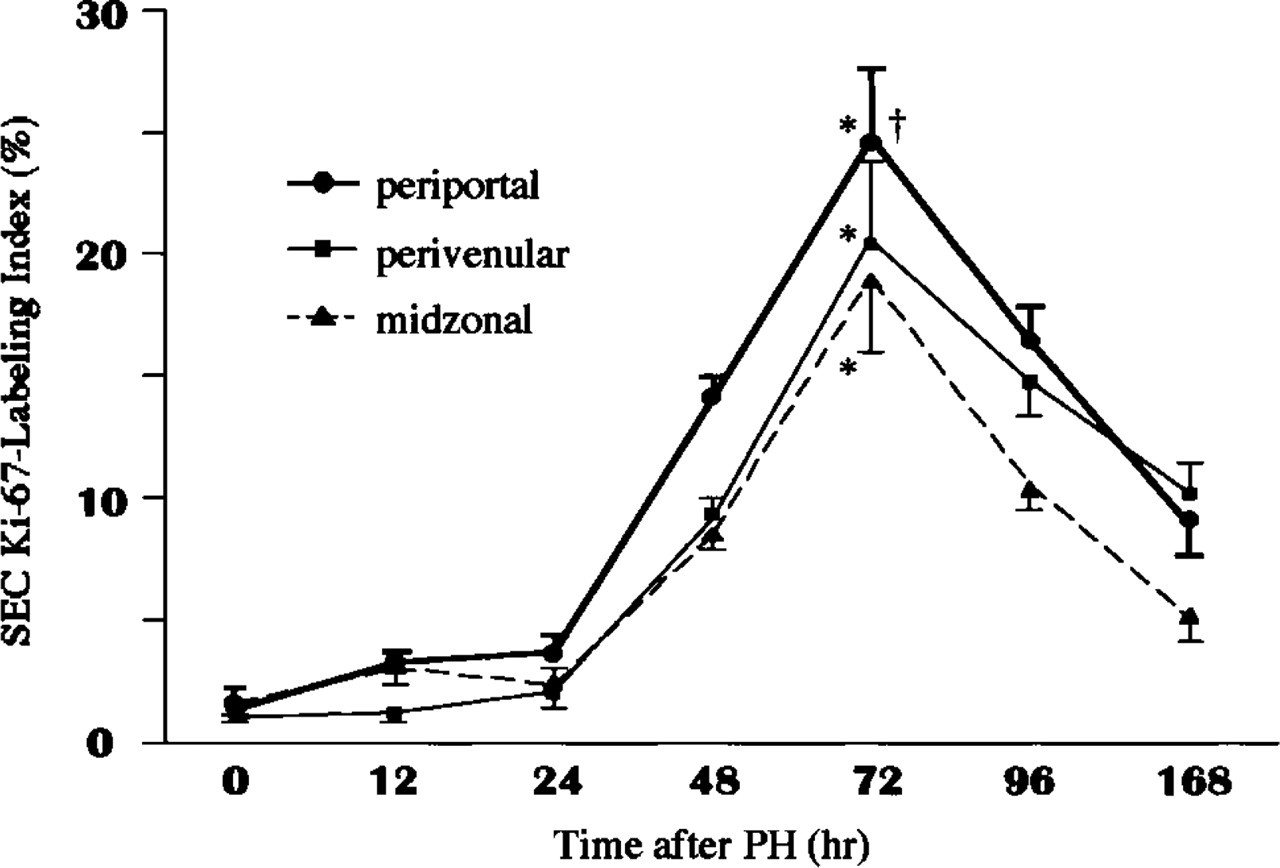
Morphometric analysis for Ki-67-expressing SECs. Liver sections were obtained from four rats in each group at each time point. Ki-67-expressing hepatocytes are expressed as a percentage. Values are expressed as means ± SD (n = 16 liver lobules in each group). •, periportal area; ▪, perivenular area; ▴, midzonal area. ∗p < 0.0001 at 96 hr vs 0 hr after PH in each area. † p < 0.05 for periportal vs perivenular or midzonal areas at 72 hr after PH.
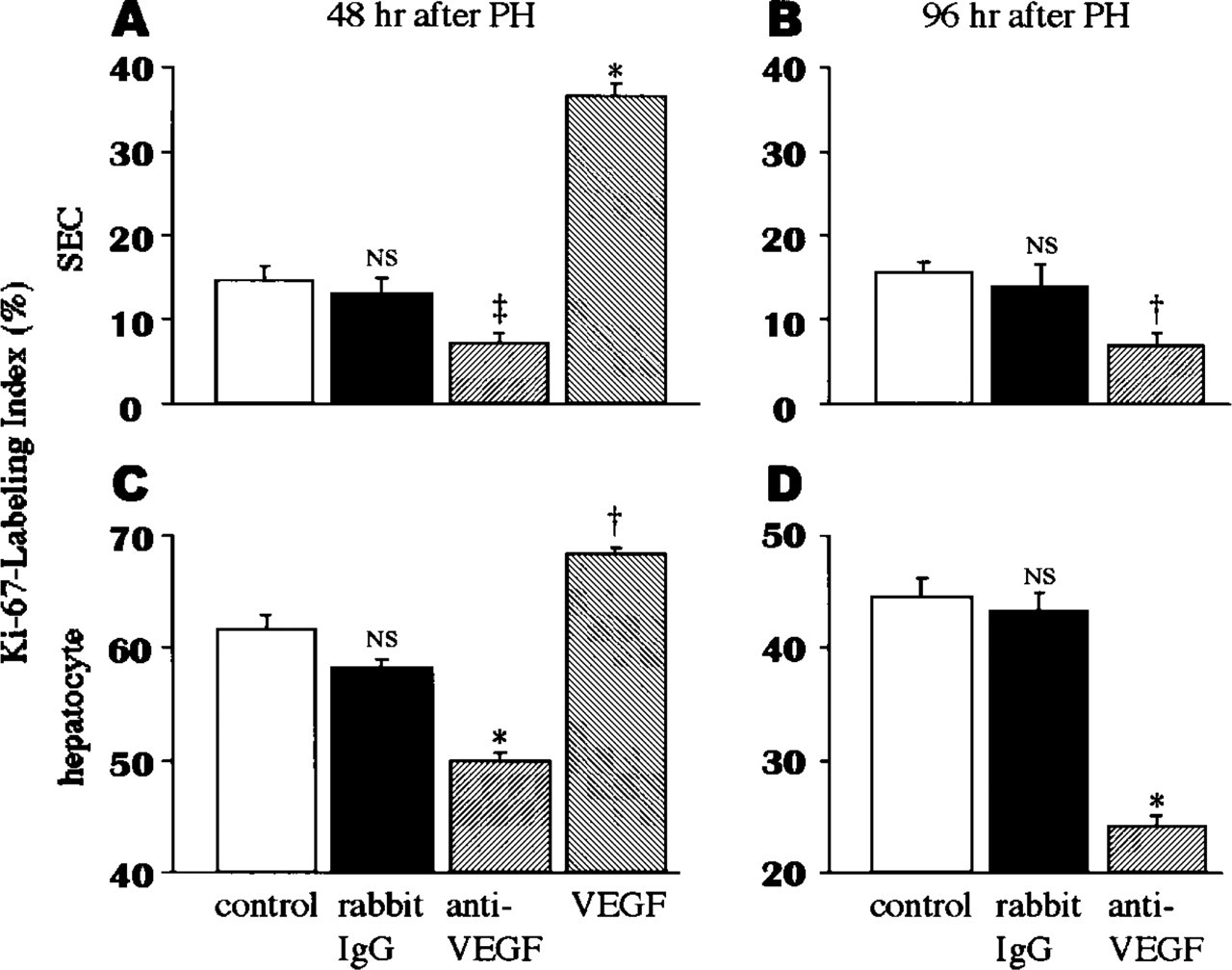
Effects of anti-VEGF rabbit IgG, VEGF, or nonimmunoreactive rabbit IgG on Ki-67-labeling index of SECs (
Although a variety of growth factors and cytokines have been implicated in liver regeneration, the contribution of VEGF is unclear in detail. Recently, it has been reported that VEGF is a potent angiogenic factor (Mustonen and Alitalo 1995; Shibuya 1995), promoting proliferation of carcinoma cells (Shibuya 1995; Brown et al. 1997; Ferrara and Davis-Smyth 1997) and healing of injured tissue (Brown et al. 1997; Ferrara and Davis-Smyth 1997). We hypothesized that VEGF was required for reconstruction of hepatic sinusoids in the regenerating liver and that VEGF sequentially participates in liver regeneration by facilitating hepatocyte proliferation.
Previous studies of VEGF expression in liver homogenates analyzed by Northern blotting, Western blotting, or RT-PCR cannot exclude VEGF production by contaminating platelets (Banks et al. 1998; Wartiovaara et al. 1998). Furthermore, these methods are not able to determine the distribution of VEGF-expressing cells in the liver. Therefore, we examined regenerating liver by ISH with antisense probe for VEGF and by IHC with an anti-VEGF antibody to determine VEGF-expressing cells.
In the present study, VEGF expression was strongly observed in periportal hepatocytes. Previous studies and our results show that hepatocyte proliferation is more active in periportal areas with lobular heterogeneity after PH (Gerlach et al. 1997). Recent studies on expression of key growth factors, such as hepatocyte growth factor (HGF) (Ishiki et al. 1992; Shiota et al. 1994), transforming growth factor-alpha (TGF-α) (Webber et al. 1993; Russell et al. 1996), and heparin-binding epidermal growth factor-like growth factor (HB-EGF) (Ito et al. 1994; Kiso et al. 1995), in hepatocyte proliferation after PH have not shown a correlation between the distribution of these growth factor's expression and lobular heterogeneity of hepatocyte proliferation. We demonstrated a correlative distribution between VEGF-expressing hepatocytes and proliferation of SECs. The lobular heterogeneity of VEGF-expressing hepatocytes suggests that reconstruction of hepatic sinusoids by SECs begins from periportal areas at which hepatocyte proliferation occurs in the regenerating liver. Previous studies have demonstrated that VEGF is expressed in epithelial cells of stomach or skin, and is closely associated with wound healing of tissues (Detmar et al. 1995; Frank et al. 1995; Takahashi et al. 1997), suggesting that VEGF expressed in hepatocytes, epithelial cells in liver, had a role in liver regeneration.
VEGF expression was less in perivenular hepatocytes. Although the percentage of VEGF-expressing hepatocytes in both periportal and perivenular areas reached maximal levels 48–72 hr after PH, the percentage of VEGF-expressing hepatocytes in perivenular areas was rapidly increased at 24–48 hr and was even higher 96 hr after PH compared with periportal areas. Therefore, VEGF expression in perivenular hepatocytes may be affected by other factors in addition to PH. It is possible that a relatively hypoxic condition occurs in the regenerating liver because perivenular hepatocytes are susceptible to hypoxia, which is known to induce VEGF (Shweiki et al. 1992; Brown et al. 1997; Ferrara and Davis-Smyth 1997).
VEGF mRNA expression detected by ISH appeared more prominent than VEGF expression detected by IHC. The difference may be attributable to the following possibilities. First, because VEGF is a secreted protein, it may be immediately released from the producing cells. Second, the sensitivity of a VEGF riboprobe against VEGF mRNA may be higher than that of an anti-VEGF antibody against VEGF.
A recent study has reported that nonparenchymal cells expressed VEGF in regenerating liver after PH (Mochida et al. 1996), whereas VEGF was not detected in nonparenchymal cells by either IHC or ISH in this study. We suggest that nonparenchymal cells, especially Kupffer cells, take up platelets that express VEGF mRNA (Wartiovaara et al. 1998), or that isolation of nonparenchymal cells artifactually induces the expression of VEGF in these cells.
Widmann and Fahimi (1975) have demonstrated that SECs increased in number and reached a maximal level 96 hr after PH. Our results demonstrated that the percentage of Ki-67-expressing SECs reached maximal level 72 hr after PH. This discrepancy could be explained by different procedures use to analyze SEC proliferation. Although the previous study examined the number of whole SECs, we examined the proliferative SECs using the well-documented cell proliferation marker Ki-67 (Gerdes et al. 1983, 1984). However, proliferation of SECs has been demonstrated only in periportal areas but not in perivenular and midzonal areas. We found that proliferation of SECs was most active in periportal areas, moderately active in perivenular areas, and less active in midzonal areas, suggesting that VEGF produced by periportal and perivenular hepatocytes promoted proliferative activity of SECs, probably in a paracrine fashion. Furthermore, the pro-liferative activity of periportal SECs was significantly higher 48 and 72 hr after PH, corresponding to increased proliferative activity of periportal hepatocytes.
Assy et al. (1999) have demonstrated that injection of VEGF promoted the proliferative activity of hepatocytes, suggesting that VEGF promoted liver regeneration. However, it is unclear how VEGF promotes the proliferative activity of hepatocytes that do not express any of the VEGF receptors KDR/Flk-1 and Flt-1 (Akira et al. 1994; Mochida et al. 1996). Moreover, they have not examined proliferative activity of SECs that expressed VEGF receptors (Akira et al. 1994; Mochida et al. 1996). We first demonstrated that endogenous and exogenous VEGF promoted the proliferation of SECs during liver regeneration after PH and that the reconstruction of hepatic sinusoids appeared to promote the proliferation of hepatocytes. VEGF in transplanted hepatic tissues promotes liver tissue formation accompanied by vascular structures (Ajioka et al. 1999). Taken together, these results suggest that VEGF promotes liver tissue formation, accompanied by reconstruction of hepatic sinusoids by SECs, in liver regeneration after PH or liver injury.
In our results, although anti-VEGF antibody similarly inhibited the proliferative activity of SEC 48 and 96 hr after PH, the effect of anti-VEGF antibody for the proliferative activity of hepatocytes was stronger at 96 hr than at 48 hr after PH. These results indicated that anti-VEGF antibody inhibited significantly but partially the proliferative activity of hepatocytes 48 hr after PH. These results can be interpreted as follows. In the early phase, hepatocytes proliferate at random significantly from periportal areas but rarely accompanying reconstruction of the architecture of hepatic sinusoids. Subsequently, proliferative hepatocytes in ischemic conditions express VEGF to obtain sufficient blood flow. Then hepatocytes proliferate with sufficient blood supplied by accompanying reconstruction of the architecture of hepatic sinusoids. It is likely that hepatocyte proliferation requires growth factors that directly stimulate hepatocytes, such as HGF and TGF-α, in the early phase, and growth factors that indirectly stimulate hepatocytes, such as VEGF, in the late phase.
Fulminant hepatitis is associated with profound liver regeneration when patients recover. Our previous report showed that patients with fulminant hepatitis had serum VEGF levels significantly lower than those of healthy individuals, and that serum VEGF levels in survivors of fulminant hepatitis were significantly increased in the recovery phase compared with levels on admission (Akiyoshi et al. 1998), suggesting that VEGF has a critical role in liver regeneration after fulminant hepatitis. Recently, HGF was implicated in liver regeneration after PH (Sakon et al. 1992; Tomiya et al. 1992) or liver injury (Tsubouchi et al. 1992; Shiota et al. 1995) in humans, and recombinant HGF has been suggested as a therapeutic application for liver diseases that require regeneration (Ishiki et al. 1992; Kaibori et al. 1997; Patijn et al. 1998). For future clinical applications, exogenous VEGF may provide a new therapeutic means for liver regeneration after PH or liver injury, such as fulminant hepatitis by itself or in combination with HGF.
In conclusion, we demonstrated that VEGF is produced predominantly by periportal hepatocytes and that it stimulates the proliferative activity of SECs. Furthermore, angiogenesis, as mediated by the proliferation of SECs induced by VEGF, may play a significant role in the proliferation of hepatocytes after PH. Administration of VEGF could open a new therapeutic approach to promote liver regeneration after PH or liver injury and warrants further investigation.
Footnotes
Acknowledgments
We thank Hironori Koga, Motoaki Kin, Takumi Kawaguchi, Atsuko Goto, and Kaori Maeda for technical assistance.
