Abstract
VEGF-A is a major angiogenesis and permeability factor. Its cellular effects, which can be used as targets in anti-angiogenesis therapy, have mainly been studied in vitro using endothelial cell cultures. The purpose of the present study was to further characterize these effects in vivo in vascular endothelial cells and pericytes, in an experimental monkey model of VEGF-A-induced iris neovascularization. Two cynomolgus monkeys (
Keywords
T
Key processes in angiogenesis, their marker antigens, and staining intensities of these markers of angiogenesis in endothelial cells (ECs) and pericytes (PCs) in monkey iris after VEGF-A treatment or in control eyes (PBS): VEGF-A-induced effects were scored in pre-existing and newly formed vessels a
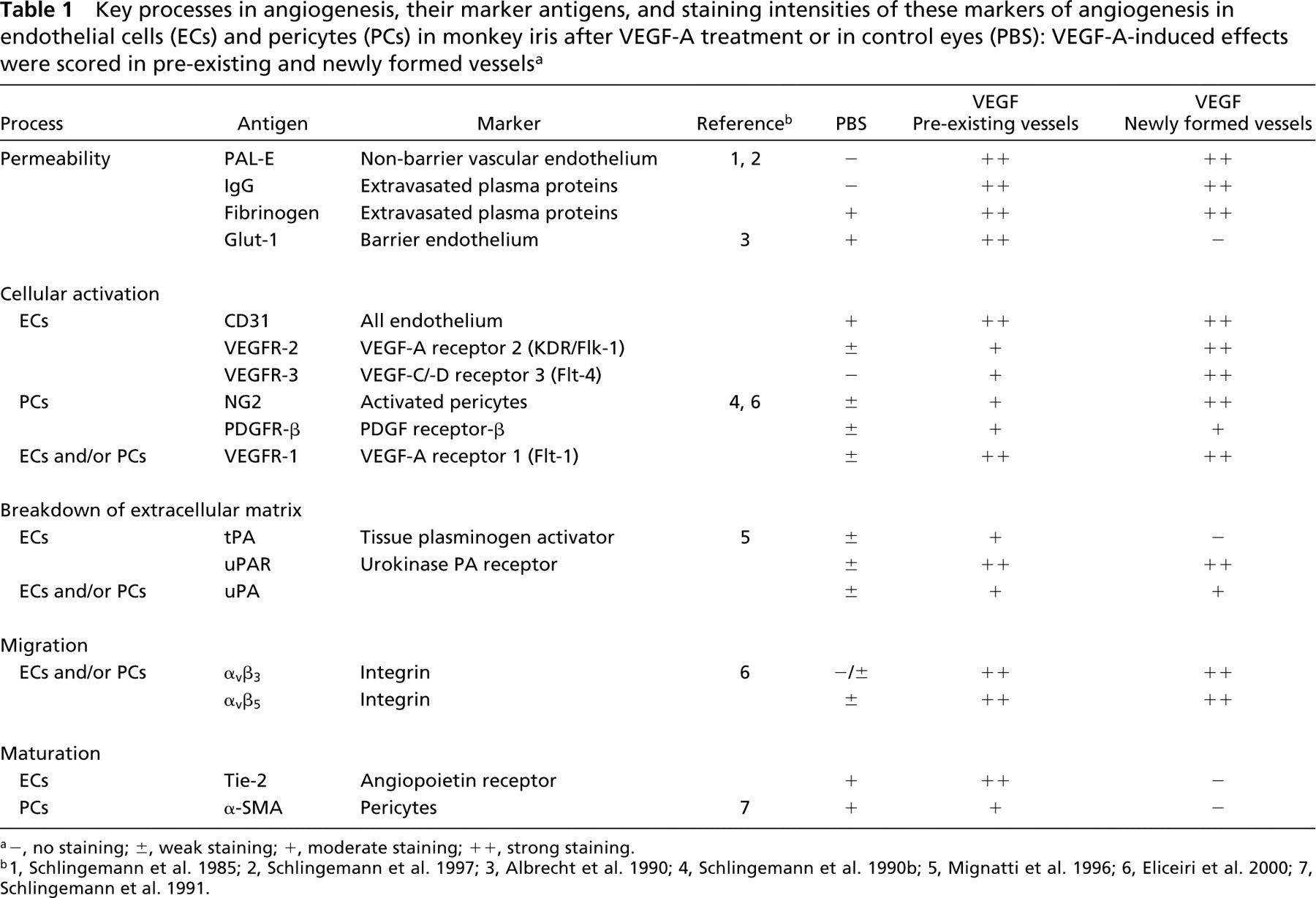
a–, no staining; ±, weak staining; +, moderate staining; ++, strong staining.
b1, Schlingemann et al. 1985; 2, Schlingemann et al. 1997; 3, Albrecht et al. 1990; 4, Schlingemann et al. 1990b; 5, Mignatti et al. 1996; 6, Eliceiri et al. 2000; 7, Schlingemann et al. 1991.
In a monkey model, elevated VEGF-A levels are temporally and spatially correlated with retinal ischemia-induced iris neovascularization (Miller et al. 1994), and VEGF-A has been shown to be necessary for iris neovascularization in this model (Adamis et al. 1996). Injections of VEGF-A into the vitreous of the eye in a primate also induce iris neovascularization (Tolentino et al. 1996). This established model allows easy distinction between pre-existing vasculature in the iris stroma and newly formed vascular sprouts at the anterior surface of the iris. The purpose of the present study was to characterize effects of VEGF-A on a series of molecules involved in key events in angiogenesis in the vasculature of the iris and to define the phenotype of the induced neovasculature.
Materials and Methods
Monkey Tissue Samples
Two cynomolgus monkeys (
The experimental design has been described previously (Hofman et al. 2000). Briefly, the animals received four injections at days 0, 2, 4, and 7 with PBS in the left eye (50 μl) through the pars plana into the center of the vitreous using a 30-gauge needle, and four injections with bioactive human recombinant VEGF-A (Harbor Bio-products, Norwood, MA; 0.5 μg in 50 μl sterile PBS), in the right eye on the same days. Before intravitreal injection, 20 mg/kg ketamine HCl (Ketalar), 0.005 mg/kg acepromazine, and 0.03 mg/kg atropine sulfate were given intramuscularly for general anesthesia. Each day the anterior segment was studied using biomicroscopy and photographed using a handheld camera (Kodak; Rochester, NY). Ten minutes after injection, intraocular pressure was measured using a Schiotz tonometer (Sklar Manufacturing; New York, NY). The two animals were sacrificed on day 9 with a bolus of 7 mg/kg IV pentobarbital (Nembutal). After enucleation, eyes were dissected and parts of the iris and anterior segment were snap-frozen in liquid nitrogen and stored at −70C until used.
Immunohistochemistry
Air-dried cryostat sections (8 μm thick) were fixed in cold acetone for 10 min and stained by an indirect immunoperoxidase procedure. Endogenous peroxidase activity was quenched by incubating sections for 20 min in a solution of 0.1% sodium azide and 0.03% H2O2 in PBS. To reduce nonspecific staining and to block endogenous biotin, sections were preincubated with a solution containing 10% normal goat serum (Jackson ImmunoResearch Laboratories; West Grove, PA), 5% avidin (DAKO; Glostrup, Denmark), and 0.05% saponin (Sigma; St Louis, MO) in PBS for 15 min. Sections were subsequently incubated overnight at 4C with the following monoclonal antibodies: PAL-E (dilution 1:1000; Schlingemann et al. 1985,1997), anti-CD31 (EN-4; Sanbio, Uden, The Netherlands; dilution 1:500); anti-NG2 proteoglycan (formerly known as high molecular weight-melanoma associated antigen (HMW-MAA), a chondroitin sulfate proteoglycan; clone 225.28S, a kind gift from Prof. S. Ferrone, Roswell Park Cancer Institute, Buffalo, NY; dilution 1:500; Schlingemann et al. 1990b), anti-αvβ3 integrin (clone 23C6; a kind gift from Prof. M.A. Horton, Bone and Mineral Center, University Center, London, UK; dilution 1:500), anti-αvβ5 integrin (P1F6; Gibco BRL, Rockville, MD; dilution 1:500), anti-PDGFR-β (a kind gift from Dr. K. Rubin, Uppsala, Sweden; dilution 1:500), anti-VEGFR-1 and anti-VEGFR-2 (kind gifts from Dr. H.A. Weich, Braunschweig, Germany; dilution 1:400), anti-VEGFR-3 (a kind gift from Prof. K. Alitalo, Haartman Institute, Helsinki, Finland; dilution 1:1500), anti-uPAR (Boehringer-Mannheim, Weidel, Germany; dilution 1:250), anti-tPA (a kind gift from Prof. V.W.M. van Hinsbergh, Gaubius Laboratory TNO-PG, Leiden, The Netherlands; dilution 1:200), anti-fibrinogen (Y-18, a kind gift from Dr. J.J. Emeis, Gaubius Laboratory TNO-PG, Leiden, The Netherlands; dilution 1:100), anti-Glut-1 (a kind gift from Dr. L. Andersson, Pharmacia & Upjohn, Uppsala, Sweden; dilution 1:250; Harik et al. 1993), and anti-α-SMA (DAKO; dilution 1:250; Schlingemann et al. 1991), and the polyclonal antibodies: rabbit anti-uPA (a kind gift from Prof. V.W.M. van Hinsbergh; dilution 1:1000) and anti-IgG conjugated with horseradish peroxidase (HRP; DAKO; dilution 1:100).
Sections were rinsed, incubated with goat anti-mouse immunoglobulins conjugated with biotin (Histostain Plus kit; Zymed, San Francisco, CA) for 15 min except for the sections incubated with anti-uPA, which were incubated with goat-anti-rabbit IgG conjugated with HRP (GAR-PO; DAKO), and the sections incubated with anti-IgG-HRP. All sections except for those incubated with anti-uPA and anti-IgG were incubated with a streptavidin-HRP complex. Peroxidase activity was visualized using 3-amino-9-ethylcarbazole (AEC; Sigma) and 0.01% H2O2. The reaction was terminated by rinsing the sections with distilled water. Counterstaining was performed with hematoxylin. Sections were washed and coverslipped in glycerol/glycerin (Sigma). Control sections were incubated in the absence of primary antibodies.
Results
Biomicroscopy
In the two VEGF-A-injected monkey eyes, transient fibrin deposition in the anterior chamber (AC) was detected at day 4. Overt neovascularization of the iris was observed at days 8 and 9 (Figure 1A). In the PBS-injected eyes, these effects were not found (Figure 1B). Apart from fibrin deposition in the AC of VEGF-A-injected eyes on day 4, the eyes did not show signs of inflammation. Intraocular pressure always remained below 25 mmHg.
Immunohistochemistry
Results of the phenotypic IHC characterization of the iris vasculature are summarized in Table 1. The VEGF-A-injected eyes and PBS-injected eyes of the two monkeys showed identical staining patterns for all markers studied. A schematic representation of a cross-section of the anterior segment, with a detail of the iris, is shown in Figures 1C and 1D.
In VEGF-A-injected eyes, CD31 (EN4; Figures 2A and 2B) staining of endothelial cells confirmed the presence of the biomicroscopically observed iris neovascularization and demonstrated hypertrophy of pre-existing iris vasculature. Formation of many sprouting vascular structures in the superficial (anterior) stroma and at the surface of the iris was the most important evidence for neovascularization. In PBS-injected eyes, few or no microvessels were present at the surface of the iris. The pre-existing iris vasculature developed a more tortuous (Figure 2A) and hypertrophic morphology under the influence of VEGF-A compared with iris vessels of control eyes.
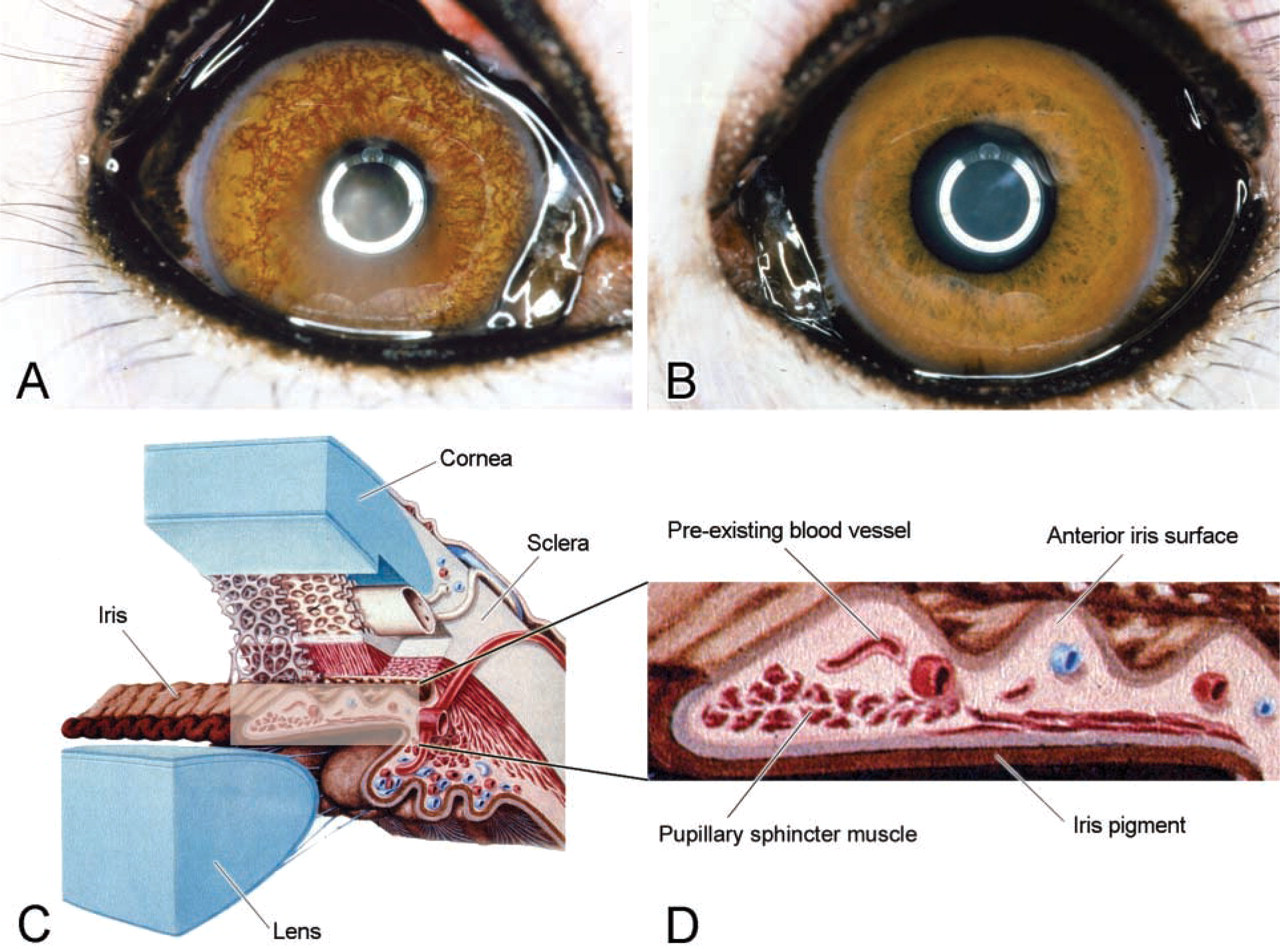
Biomicroscopy of VEGF-A-injected eye (
Markers of Vascular Permeability
In VEGF-A-injected eyes, marked staining of endogenous IgG (Figure 2E) and fibrinogen (Figure 2G) was found in iris stroma, indicating increased permeability of the iris microvasculature. These “leaky” vessels stained for endothelial antigen PAL-E (Figure 2C) whereas staining of this antigen and IgG was absent in the iris of control eyes (Figure 2D and 2F). In VEGF-A-injected eyes, capillaries in the iris base and some capillaries in the ciliary muscle adjacent to the iris base also showed increased staining of PAL-E, with a clear gradient of decreasing vascular staining from the iris base to the ciliary muscle (not shown). As uveoscleral outflow of aqueous humour occurs through this area, this staining gradient suggested an outflow-related concentration-response effect of the exogenous VEGF-A on local blood vessels. Staining for fibrinogen was confined to the blood vessels in the PBS-injected eye (Figure 2H).
Staining of the blood-brain barrier marker Glut-1 was observed in the iris microvasculature of control eyes (Figure 2J). In VEGF-A-injected eyes, Glut-1 staining was more intense in the pre-existing vasculature (Figure 2I). The newly formed vessels did not stain with anti-Glut-1 (Figure 2I).
Markers of Cellular Activation
In VEGF-A-injected eyes, staining of CD31 was more prominent in pre-existing iris vasculature and newly formed vessels at the iris surface compared to iris vessels in the PBS-injected eyes (Figures 2A and 2B).
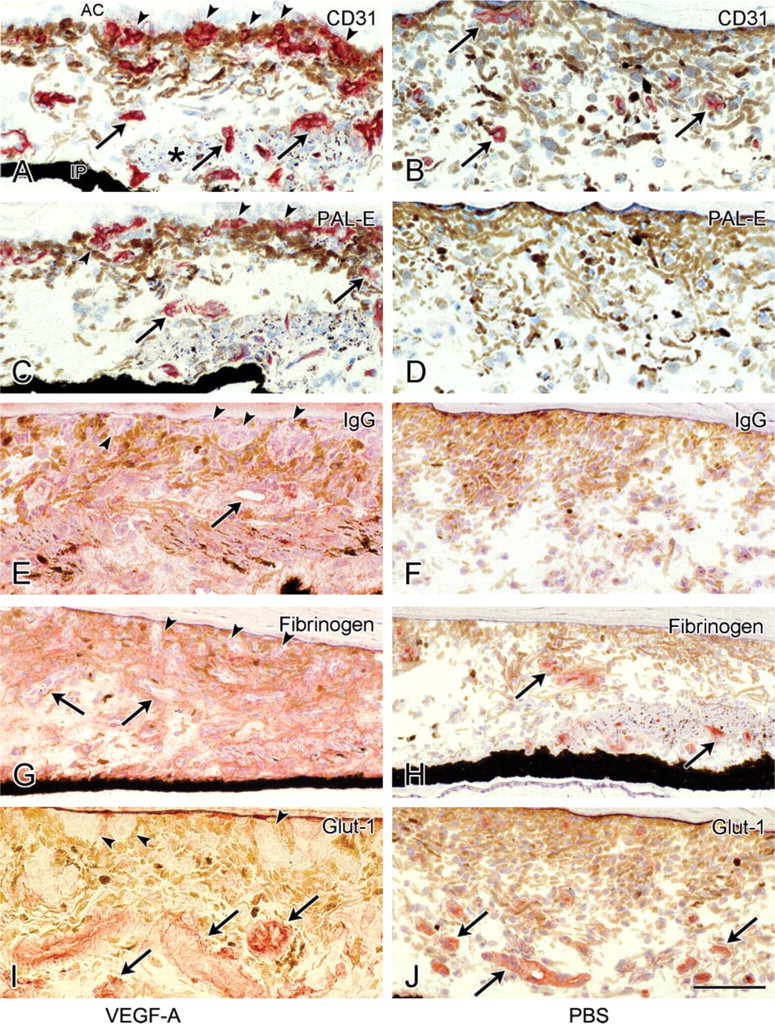
Immunohistochemical staining of endothelial antigens CD31 (
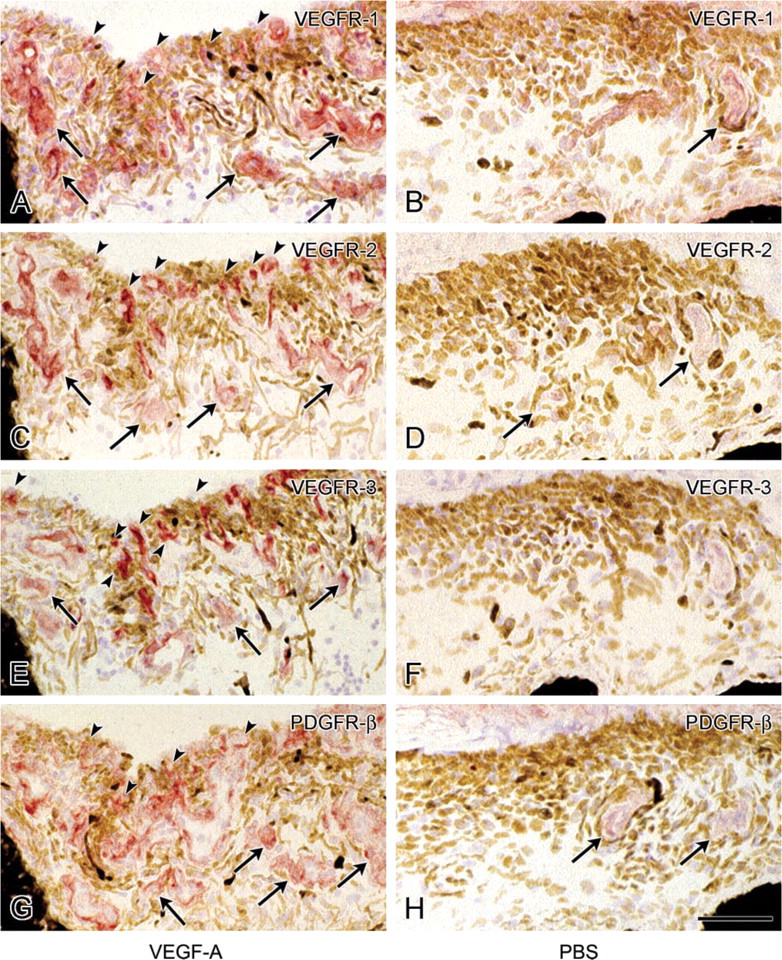
Activated pericytes were recognized by increased staining of the marker for this cell type, NG2 proteoglycan, in the iris of VEGF-A-treated eyes (Figure 3C). Staining of this antigen was also found in newly formed vessels at the iris surface, indicating that these sprouts consisted of both endothelial cells and pericytes (Figures 3C and 6I). Pericytes in control eyes were weakly stained for NG2 proteoglycan (Figure 3D). In both control eyes and VEGF-A-injected eyes, staining of VEGFR-1 was observed in pre -existing iris microvasculature, in a pattern that suggested expression in more than just endothelial cells compared to CD31, i.e., also in pericytes (Figures 4A, 4B, and 6A). VEGFR-1 staining was more distinct in VEGF-A-injected eyes, both in pre-existing vessels and vascular sprouts. VEGFR-2 staining was weak in iris vessels of control eyes. It was upregulated in pre-existing vessels but more strongly in newly formed vessels in VEGF-A-injected eyes (Figures 4C, 4D, and 6B). A different staining pattern was found for VEGFR-3 (Figures 4E, 4F and 6C). It was absent in control eyes whereas in VEGF-A-injected eyes strong expression was observed, particularly in newly formed vessels. Staining of VEGFR-2 and VEGFR-3 appeared to be confined to endothelial cells (Figures 6B and 6C). When VEGFR-2 and VEGFR-3 staining with VEGFR-1 staining were compared (exemplified in Figures 4A, 4C, 4E, and 6A-6C), more elements of a vascular wall were stained for VEGFR-1 than for the two other receptors.
Staining of PDGFR-β was found in pericytes of preexisting iris microvasculature in control eyes (Figure 4H), and was stronger in VEGF-A-injected eyes (Figure 4G). PDGFR-β staining was observed in pericytes in vascular sprouts at the iris surface in a pattern similar to that of NG2 proteoglycan (Figure 6F).
Proteins Involved in Fibrinolysis and Breakdown of Extracellular Matrix
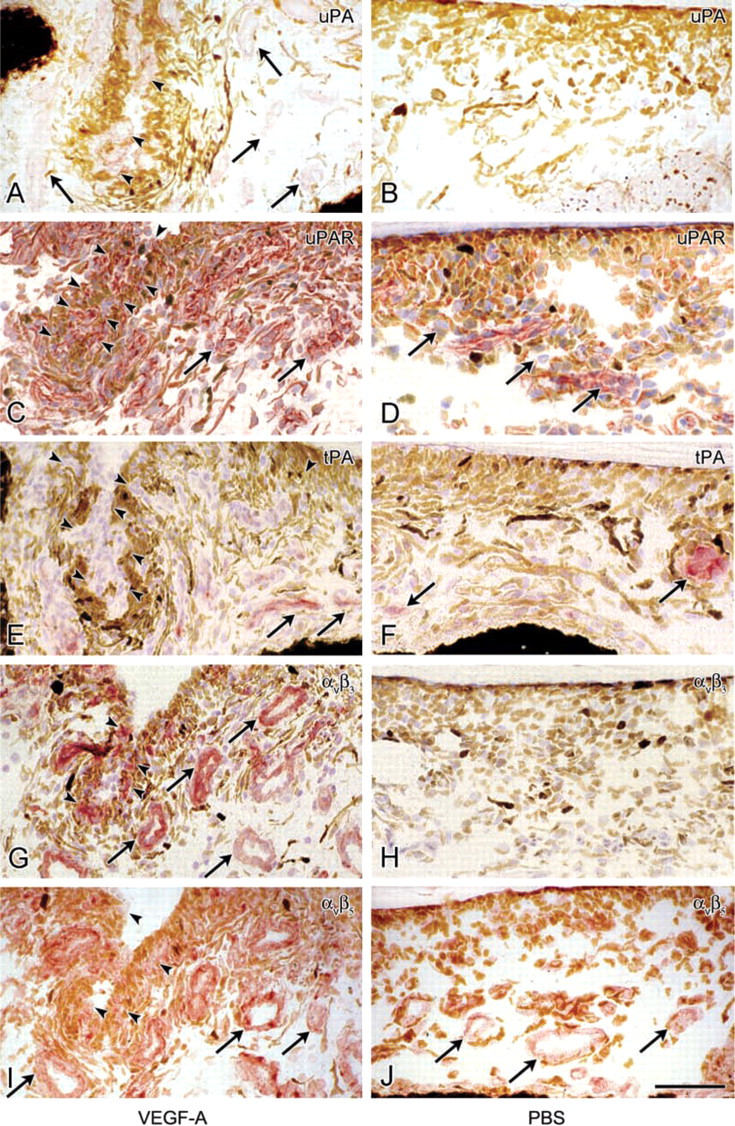
tPA was present in a number of iris vessels of control eyes. It was localized in endothelial cells but also in a more diffuse distribution pattern in the vascular wall (Figure 5F). In hypertrophic iris vessels of VEGF-A-injected eyes, tPA staining was slightly stronger, but newly formed vessels and sprouts at the iris surface were distinctly negative (Figures 5E and 6J). In contrast, staining of uPA was marked in both pre-existing vessels and newly formed vessels, including the tips of sprouts at the iris surface, indicating expression of uPA by both quiescent and migrating cells (Figure 5A). The staining pattern suggested localization of uPA in endothelial cells and pericytes in positive vessels, and it was always co-localized with uPAR. Staining of uPAR was observed in all cell types in control and VEGF-A-injected eyes, but expression was stronger after VEGF-A treatment (Figures 5C and 5D).
Proteins Involved in Endothelial and Pericyte Migration
Control eyes showed absent to weak vascular staining of αvβ3 (Figure 5H) and αvβ5 integrins (Figure 5J). The staining pattern suggested expression of αvβ3 and αvβ5 in pericytes and/or at the abluminal side of endothelial cells (Figures 6K and 6L). In VEGF-A-injected eyes, staining of αvβ3 and αvβ5 was much stronger, but staining intensity was similar in pre-existing and newly formed vessels, indicating expression of both integrins by quiescent and migrating cells (Figures 5G and 5I).
Markers of Maturation and Differentiation
α-SMA, a marker for smooth muscle cells and resting pericytes (Schlingemann et al. 1991), was stained in a few iris vessels in PBS-injected eyes and pre-existing vessels in VEGF-A-treated eyes. However, activated pericytes in angiogenic sprouts at the iris surface in VEGF-A-injected eyes showed no or only weak and fragmented staining of α-SMA (Figures 3E, 3F, 6G, and 6H).
Tie-2 was present in iris microvasculature in control eyes. In eyes injected with VEGF-A, staining of pre-existing vessels was more intense, whereas Tie-2 staining was absent in newly formed vessels (Figures 3A, 3B, 6D, and 6E).
Discussion
The results of our IHC study in the monkey iris after VEGF-A treatment show prominent changes in expression patterns of molecules that are functional in angiogenesis. Our results allow characterization of an in vivo angiogenic phenotype of endothelial cells and pericytes in VEGF-A-induced neovascularization (Figure 7). This in vivo study confirms a number of in vitro studies (Pepper et al. 1991; Friedlander et al. 1995; Van Hinsbergh et al. 1997; Plate and Warnke 1997; Suzuma et al. 1998) that investigated effects of VEGF-A on cultured endothelial cells, but several of our observations may help to shed new light on functional aspects of angiogenesis in vivo.
The various stages of angiogenesis have been defined in morphological studies (Ausprunk and Folkman 1977; Paweletz and Knierim 1989; Schlingemann et al. 1990b,1991; Paku and Paweletz 1991; Nehls et al. 1992). In an early phase, i.e., within a few hours to a few days after exposure to an angiogenic stimulus, capillary permeability increases and the normally highly differentiated, flattened endothelial cells and adjacent pericytes become activated, displaying hypertrophy and an increased number of organelles. In line with these observations, at 9 days after repeated VEGF-A exposure, the pre-existing iris vasculature was hypertrophic in our model, as was shown by staining patterns of the pan-endothelial marker proteins CD31. Capillary permeability was increased, as was shown by the extensive leakage of plasma proteins IgG and fibrinogen into the iris stroma. This VEGF-A-induced increased permeability of the preexisting vasculature was accompanied by expression of PAL-E, a non-barrier vascular endothelium marker absent in normal iris vasculature (Schlingemann et al. 1997). Glut-1 is a membrane glucose transporter, which is characteristically expressed on blood-brain barrier endothelium (Harik et al. 1993). However, Glut-1 is upregulated in several pathological conditions in the eye associated with VEGF-A activity (Kumagai et al. 1994). In vitro, Glut-1 was found to be translocated from the cytoplasm to the plasma membrane under the influence of VEGF-A, which enhanced glucose transport into retinal endothelial cells (Sone et al. 2000). Our in vivo results of stronger Glut-1 staining of pre-existing vessels of the iris in VEGF-A-injected eyes compared with those in control eyes are consistent with these in vitro findings. The newly formed vessels at the anterior surface of the iris did not express this marker, which is in agreement with the observation that immature blood-brain barrier endothelium does not express Glut-1 (Albrecht et al. 1990).
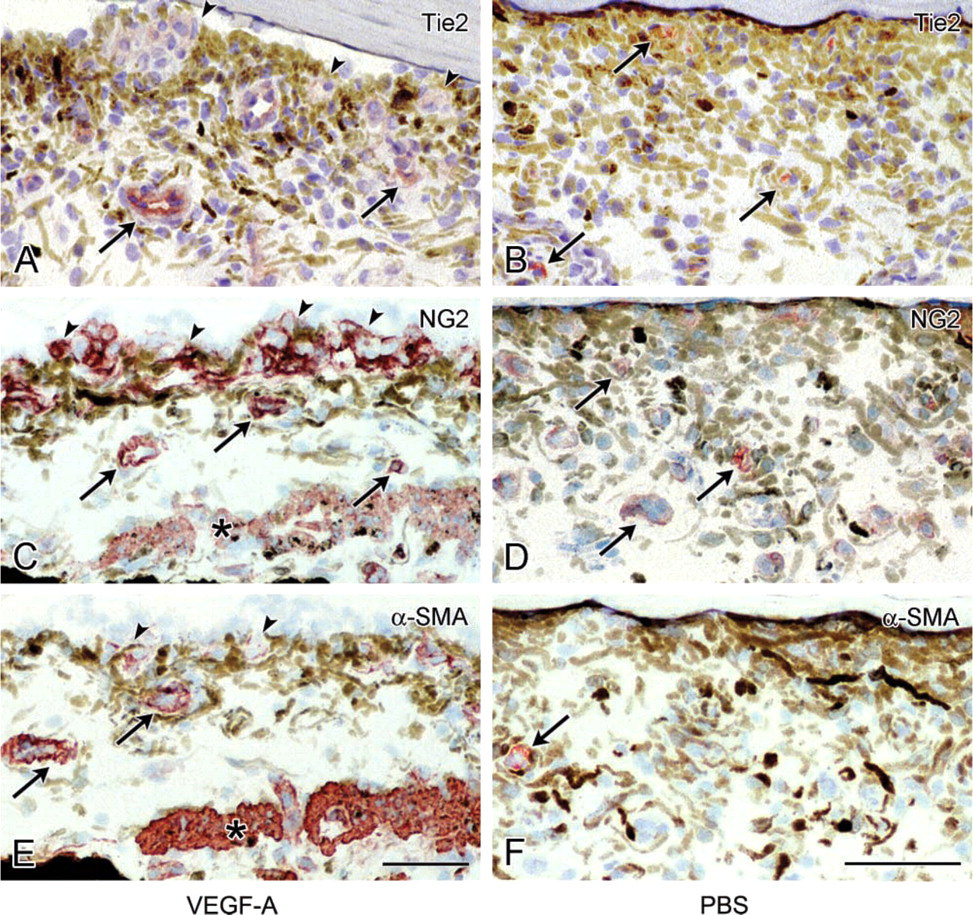
Immunohistochemical staining of angiopoietin receptor Tie2 (
Immunohistochemical staining of VEGFR-1 (
Immunohistochemical staining of uPA (
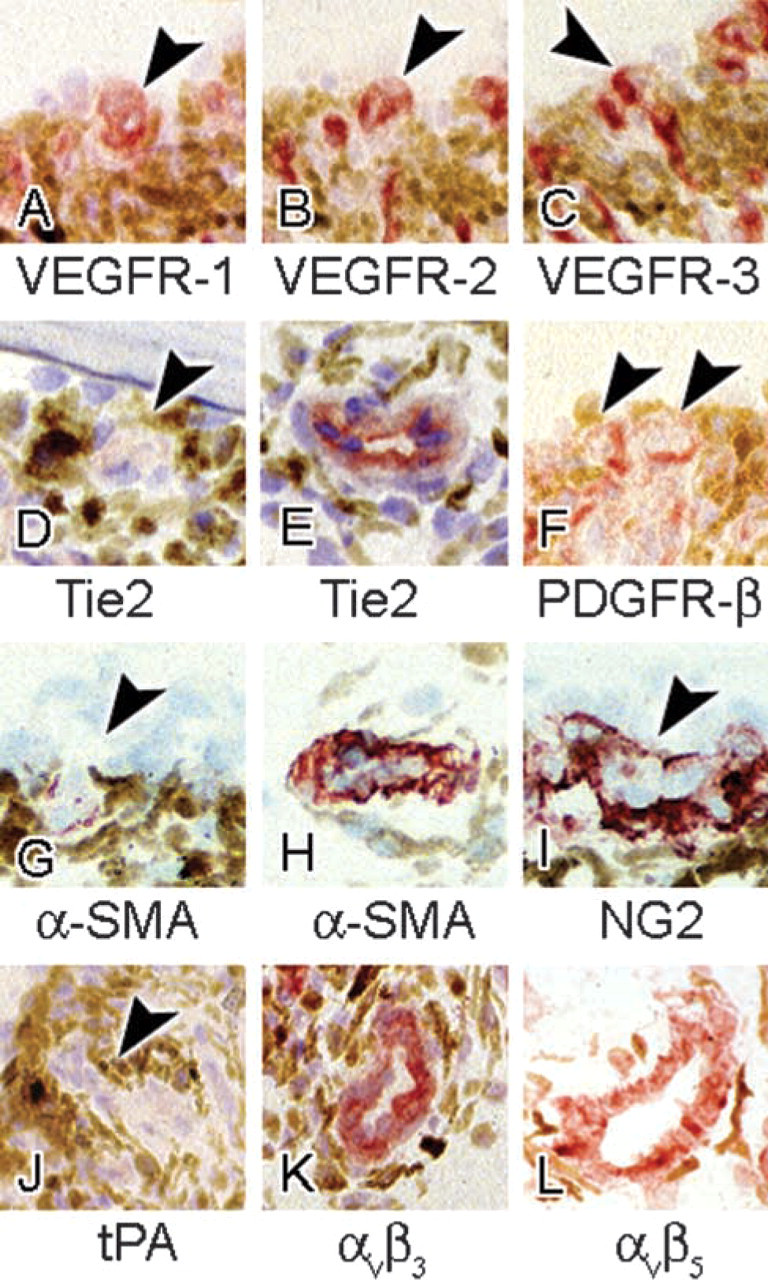
High-power magnifications of staining of VEGFR-1 (
Activation of endothelial cells and pericytes in preexisting iris vessels after VEGF-A treatment was indicated by increased staining of several antigens, including VEGFRs. In vivo, quiescent endothelial cells and pericytes are considered to lack expression of VEGFRs (Ortega et al. 1999), although we have found recently that VEGFRs are also expressed under physiological conditions (Witmer et al. 2002b). However, when VEGF-A is present in vivo, e.g., in tumors, VEGFRs are invariably expressed at a high level (Dvorak et al. 1995; Veikkola et al. 2000). Cultured human microvascular endothelial cells constitutively express all three VEGFRs (Hewett and Murray 1996), which may be a sign of cellular activation associated with culture conditions. These expression patterns are not altered after stimulation with VEGF-A of endothelial cells in vitro (Hewett and Murray 1996). In our in vivo model, all three members of the VEGFR family were upregulated in pre-existing iris vessels after VEGF-A treatment, including VEGFR-3, for which VEGF-A has no affinity.
Pericytes were previously regarded to be recruited only in the maturation phase of angiogenesis. Several studies, however, show that pericytes accompany endothelial cells in newly formed sprouting capillaries (Schlingemann et al. 1990b,1991,1996; Nehls and Drenckhahn 1991; Nehls et al. 1992; Witmer et al. 2001,2002a,b; Tsai et al. 2002), suggesting an important role for pericytes in early steps of blood vessel formation. In addition to endothelial cell activation, activation and hypertrophy of pericytes in sprouting vessels was demonstrated by increased staining of NG2 proteoglycan and PDGFR-β (Schlingemann et al. 1990b; Sundberg et al. 1993). Previous studies have shown in vitro and in vivo that pericytes express VEGFR-1 as well (Nomura et al. 1995; Takagi et al. 1996; Grosskreutz et al. 1999; Ogata et al. 2001; Witmer et al. 2002b). These findings support our suggestion that VEGFR-1 is expressed on pericytes in iris microvessels, along with endothelial cells. This may imply that VEGF-A acts directly on pericytes (Benjamin et al. 1998).
VEGFR-2 was expressed at low levels in the vasculature of the iris of control eyes, whereas VEGFR-2 is absent in the vasculature of monkey retina (Witmer et al. 2002a). This observation may explain differences in early responses of these two tissues to VEGF-A. In monkey retina, VEGF-A treatment leads to increased permeability, whereas neovascularization is not observed in this model after 9 days (Hofman et al. 2000,2001). However, VEGF-A injections in the eye lead to rapid development of iris neovascularization, which may be mediated by the constitutively expressed VEGFR-2. VEGF-A is considered to mediate iris neovascularization in human patients with retinal ischemia due to central retinal vein occlusion (Aiello et al. 1994) and in other pathological conditions that are not associated with retinal neovascularization, such as uveal melanoma (Vinores et al. 1995). As VEGFR-2 mediates the mitogenic signal of VEGF-A, our observations indicate that different levels of constitutive VEGFR-2 expression in iris and retina could explain these observations.
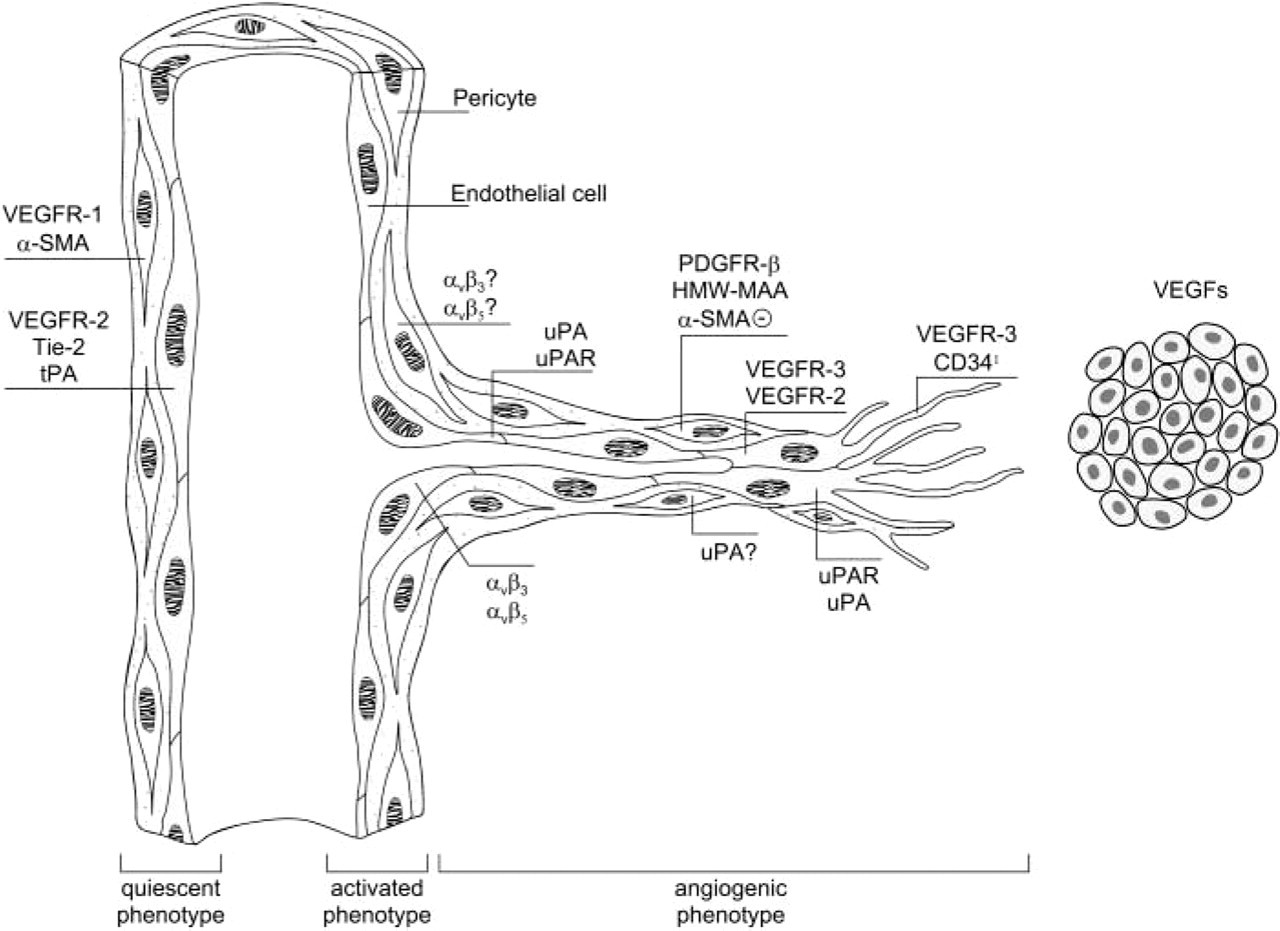
Summary of the findings of the present study. Schematic representation of the endothelial and pericyte phenotype in a pre-existent capillary and adjacent angiogenic sprout. Note the differential expression of key molecules in endothelial cells and pericytes in the quiescent state in comparison with the angiogenic state. (Schlingemann et al. 1990a, Witmer et al. 2003) (Reproduced with permission).
VEGFR-1 and −2 are the functional receptors of VEGF-A, whereas the ligands of VEGFR-3 are VEGF-C and -D. VEGF-C has a strong chemotactic effect on VEGFR-3-overexpressing endothelial cells in vitro (Kroon et al. 1999). In the adult, VEGFR-3 is mainly expressed in lymphatic endothelium and vascular endothelium in several types of tumors (Lymboussaki et al. 1998; Salven et al. 1998; Valtola et al. 1999; Veikkola and Alitalo 1999). In our study, after VEGF-A exposure, VEGFR-3 was expressed by pre-existing vascular endothelium of the iris and very strongly in the sprouting vasculature at the iris surface. This may indicate involvement of VEGFR-3 in migration of endothelial cells in VEGF-A-induced angiogenesis (Witmer et al. 2001).
Basement membrane degradation and subsequent migration of endothelial cells and pericytes through the extracellular matrix are the next steps in the angiogenic process. Endothelial cells and possibly pericytes need active proteases to hydrolyze the extracellular matrix proteins during migration. Proteases that are upregulated in endothelial cells in vitro by VEGF-A include members of the plasminogen system, such as uPA and its receptor uPAR (Mignatti and Rifkin 1996; Kroon et al. 1999). In addition, several matrix metal-loproteinases (Pepper 2001a,b) and cathepsin B activity have been associated with angiogenesis (Keppler et al. 1996; Hazen et al. 2000). In the present study, the pre-existing iris vasculature in VEGF-A-injected eyes showed a somewhat stronger expression of tPA, but tPA was absent in sprouting vessels at the iris surface. This may be an indication that tPA activity is not involved in migration of endothelial cells in the sprouting phase of angiogenesis, as was previously suggested by in vitro experiments (Koolwijk et al. 2001). In contrast, uPA expression was present in sprouting vessels in a pattern suggesting positivity in both pericytes and endothelial cells. Although protein expression of these proteases is not a direct marker of their activity (Hazen et al. 2000; Bleeker et al. 2000), our IHC observations may indicate that uPA of both pericytes and endothelial cells contributes to protease activation in the angiogenic process.
In addition to proteases, vascular cells express integrins known to be involved in cell migration and cell signaling during angiogenesis (Senger et al. 1996; Max et al. 1997; Byzova et al. 2000). In our study, vascular staining of the integrins αvβ3 and αvβ5 was stronger in VEGF-A-injected eyes than in control eyes. It is remarkable that both uPA and the integrins studied were upregulated at the protein level in both stationary and migrating cells in pre-existing and newly formed vessels in the iris after VEGF-A treatment. From in vitro studies it is known that endothelial cells upregulate the integrins αvβ3 and αvβ5 in response to growth factors (Eliceiri and Cheresh 2000), although upregulation of αvβ5 is less pronounced than αvβ3 (Friedlander et al. 1995; Eliceiri and Cheresh 2000; Walton et al. 2000). It has previously been suggested that different pathways of angiogenesis exist in vivo because angiogenesis in the mouse cornea and chorioallantoic membrane models induced by basic fibroblast growth factor or tumor necrosis factor-α depends on αvβ3, whereas angiogenesis initiated by VEGF-A depends on αvβ5 (Friedlander et al. 1995). Our observation that in iris angiogenesis after VEGF-A treatment staining of αvβ3 was much higher than that of αvβ5 is not in line with this association of VEGF-A and αvβ5.
After formation of vascular sprouts, the final phase of angiogenesis involves maturation and differentiation. Pericytes are considered to appear in angiogenesis only in this final stage, attracted via PDGFR-β signaling by endothelium-derived PDGF-BB (Hirschi and D'Amore 1996; Benjamin et al. 1998), to induce maturation of blood vessels, decrease of permeability (Thurston et al. 2000), and endothelial differentiation, by production of angiopoietin-1 and activation of TGF-β (Yancopoulos et al. 2000). This late role of pericytes was supported by observations that pericytes can inhibit endothelial proliferation in vitro via TGF-β (Antonelli-Orlidge et al. 1989). The absence of α-SMA staining at the front of growing vascular sprouts and in tumor vessels is generally interpreted as a sign of absence of pericytes and thus as a marker of angiogenesis and vessel immaturity (Benjamin et al. 1999). However, this study suggests that NG2 proteoglycanpositive pericytes are already present and activated in the front of newly formed iris vessels, and that these angiogenic pericytes do not express α-SMA (Schlingemann et al. 1991). Therefore, under angiogenic conditions, NG2 proteoglycan and PDGFR-β may be better markers for pericytes, whereas α-SMA is probably only expressed in a more stable and mature phenotype of pericytes, which develops when the newly formed vessels mature. Our results suggest that pericytes have an early role in angiogenesis and that they can differentiate into an angiogenic phenotype in a similar way as do endothelial cells. Expression of VEGFR-1 by pericytes may allow direct induction by VEGF-A or related factors such as placental growth factor (PlGF) of this phenotype and related angiogenic functions.
Angiopoietin receptor Tie-2 is thought to induce stabilization of primitive endothelial tubes, reduce permeability, and induce maturation on binding of angiopoietin-1 (Jain and Munn 2000; Thurston et al. 2000). Mouse embryos lacking Tie2 develop a rather normal vasculature but fail to undergo further remodeling (Sato et al. 1995). In this respect, the absence of Tie-2 in newly formed iris vessels is consistent with this stabilizing and remodeling function, as newly formed vessels are still immature. On the other hand, Tie2, through binding of angiopoietin-2, has also been proposed to be involved in destabilization of vessels allowing angiogenesis (Carmeliet 2000), a notion not supported by our observations.
In conclusion, our study shows that proteins, which play a role in angiogenesis, are upregulated in endothelial cells and pericytes in both pre-existing and newly formed iris vasculature after VEGF-A treatment. In several respects, our findings throw new light on the complex cellular process of angiogenesis as it occurs in vivo. VEGF-A induces hypertrophy and loss of barrier function in pre-existing vessels, and induces angiogenic sprouting, characterized by endothelial cells that lack expression of tPA and Tie-2, and pericytes that lack α-SMA expression, and by marked endothelial expression of VEGFR-3. Pericytes appear to play a significant role, next to endothelial cells, in immature sprouting blood vessels. These insights into the mechanism behind angiogenesis may raise new potential strategies into an attempt to inhibit (ocular) neovascularization.
Footnotes
Acknowledgements
Supported by the Haagsch Oogheelkundig Fonds, the Landelijke Stichting voor Blinden en Slechtzienden, the Rotterdamse Blindenpenning, the Donders Fonds Utrecht, the Edward and Marianne Blaauwfonds, and the Diabetes Fonds Nederland (grants 95.103 and 99.050).
We wish to thank Prof Dr K. Alitalo, Dr H.A. Weich, Prof Dr S. Ferrone, Prof Dr M.A. Horton, Dr K. Rubin, Prof Dr V.W.M. van Hinsbergh, Dr J.J. Emeis, and Dr L. Andersson for providing antibodies, and T. Put for preparing the microphotographs.
