Abstract
We examined ultrastructurally the localization of myocilin (formerly called trabecular meshwork inducible glucocorticoid response, or TIGR) protein in cultured human trabecular meshwork (TM) cells and in normal human TM tissues. The TM, a specialized tissue located at the chamber angle of the eye, is believed to be responsible for the development of glaucoma. The myocilin gene has been directly linked to both juvenile and primary open-angle glaucomas, and multiple mutations have been identified. Human TM cells were treated with 0.1 mM of dexamethasone (DEX) to induce myocilin expression. This protein was immunolocalized by colloidal gold electron microscopy using an anti-human myocilin polyclonal antibody. Double labeling with different sizes of gold particles was also performed with additional monoclonal antibodies specific for cell organelles and structures. In both DEX-treated and untreated cultured cells, myocilin was associated with mitochondria, cytoplasmic filaments, and vesicles. In TM tissues, myocilin was localized to mitochondria and cytoplasmic filaments of TM cells, elastic-like fibers in trabecular beams, and extracellular matrices in the juxtacanalicular region. These results indicate that myocilin is localized both intracellularly and extracellularly at multiple sites. This protein may exert diverse biological functions at different sites.
T
The myocilin gene has been directly linked to both juvenile and primary open-angle glaucomas, and multiple mutations have been identified (Sheffield et al. 1993; Stone et al. 1997; Alward et al. 1998). Myocilin was originally identified and named the trabecular meshwork-inducible glucocorticoid response (TIGR) protein as an upregulated molecule secreted by TM cells (Polansky et al. 1997; Nguyen et al. 1998) after treatment with a glucocorticoid such as dexamethasone (DEX). The gene also was cloned by Kubota et al. (1997) from a retinal cDNA library and was termed myocilin for its sequence similarity to myosin.
The myocilin transcript has been detected in the TM (Polansky et al. 1997; Nguyen et al. 1998), retina (Kubota et al. 1997), ciliary body (Escribano et al. 1995; Ortego et al. 1997), and iris (Ortego et al. 1997) in ocular tissues and also in non-ocular tissues including skeletal muscle and heart (Fingert et al. 1998). The protein has also been immunologically detected in the TM of human eyes (Lütjen-Drecoll et al. 1998; Karali et al. 2000). In this study, we ultrastructurally localized myocilin in cultured human TM cells with or without DEX treatment and in normal human TM tissues by immunogold electron microscopy (EM), using an anti-human myocilin polyclonal antibody. Double labeling with different sizes of gold particles (Bendayan 1982) was also performed with monoclonal antibodies (MAbs) specific for intracellular organelles and structures.
Materials and Methods
Cell Culture and DEX Treatment
Normal human donor eyes with no history of glaucoma or other eye diseases were obtained from the Illinos Eye Bank at Chicago. TM tissues from three donors (ages 19, 19, and 29 years) were excised and cultured on Falcon Primaria flasks in complete medium composed of Eagle's minimal essential medium, 10% fetal bovine serum, 5% calf serum, essential and nonessential amino acids, and antibiotics (Zhou et al. 1998). When the cells reached confluence, they were trypsinized. First- or second-passaged cells were seeded onto fibronectin-coated four-well glass chamber slides at a density of 5000 cells per well. Cells in two of the wells were fed with complete medium and those in two others were fed with medium containing 0.1 mM DEX (Nguyen et al. 1998). These cultures were maintained at 37C for 14 days.
Specimen Preparation
Cultured human TM cells without or with the DEX treatment were fixed at room temperature (RT) for 3 hr in a fixative containing 4% paraformaldehyde, 0.1% glutaraldehyde, and 0.05% Triton X-100 in 0.1 M phosphate buffer, pH 7.4. After rinsing three times in PBS, the cells were dehydrated at −20C through a graded series of N,N-dimethyl formamide (DMF). DMF was then exchanged stepwise to glycol methacrylate (GMA) embedding medium that was made up of 65 ml 2-hydroxyethyl methacrylate, 35 ml N-butyl methacrylate, 5 ml ethylene glycol dimethacrylate, and 0.5 g benzoin methyl ether (Spaur and Moriarty 1977). The specimens were then polymerized by UV irradiation for 48 hr at −20C. The resultant GMA blocks were warmed up to RT and were quickly chilled in liquid nitrogen to break the chamber glass for easy removal from the GMA blocks. The sheet of TM cells on the surface of the block was re-embedded in GMA. Ultrathin 80-nm sections were cut parallel to the sheet of cells and were mounted on 150-mesh nickel grids.
For in vivo studies, normal human eyes from three donors (ages 48, 58, and 72 years) with no history of ocular diseases were obtained from the Illinois Eye Bank. The anterior segments that included TM tissues were excised from donor eyes. They were fixed at RT for 3 hr in 4% paraformaldhyde, 0.1% glutaradehyde, 0.1 M phosphate buffer, pH 7.4, and were further dissected into small wedge-shaped pieces. The specimens were dehydrated through a DMF series and embedded in GMA block as described above. Ultrathin sections cut transversely through the entire layers of TM were mounted on EM grids.
Polyclonal Antibody and Western Blotting Analysis
Polyclonal anti-myocilin was generated in rabbits against a synthetic polypeptide corresponding to the amino acid sequence DKSVLEEEKKRLRQ (residues 148–161) of human myocilin. The peptide was coupled to keyhole limpet hemocyanin via a carboxy-terminal cysteine residue not present in myocilin. The sythetic peptide and the antibody were prepared by Alpha Diagnostic International (San Antonio, TX). The antibody was purified by an affinity column and its specificity was confirmed by ELISA. To further verify its specificity, Western blotting analysis was performed. Briefly, protein extracts from normal human TM tissues (donor ages 43, 52, and 53 years) were subjected to SDS-PAGE and were transferred to nitrocellulose membranes. The blots were immunoblotted with either anti-myocilin (1:5000) or the antibody preabsorbed with the immunogenic peptide. The immunoblots were further incubated with biotinylated goat anti-rabbit IgG (Jackson Immuno Research; West Grove, PA), horseradish peroxidase-conjugated streptavidin (Jackson Immuno Research), and Super Signal West Pico (Pierce; Rockford, IL). Using a Fluoroimager Image Station 440CF (Eastman Kodak; Rochester, NY), the typical 66- and 57/55-kD myocilin bands (Nguyen et al. 1998) were detected when the membrane was probed with anti-myocilin (Figure 1). These bands were abolished when the peptide-preabsorbed antibody was used.
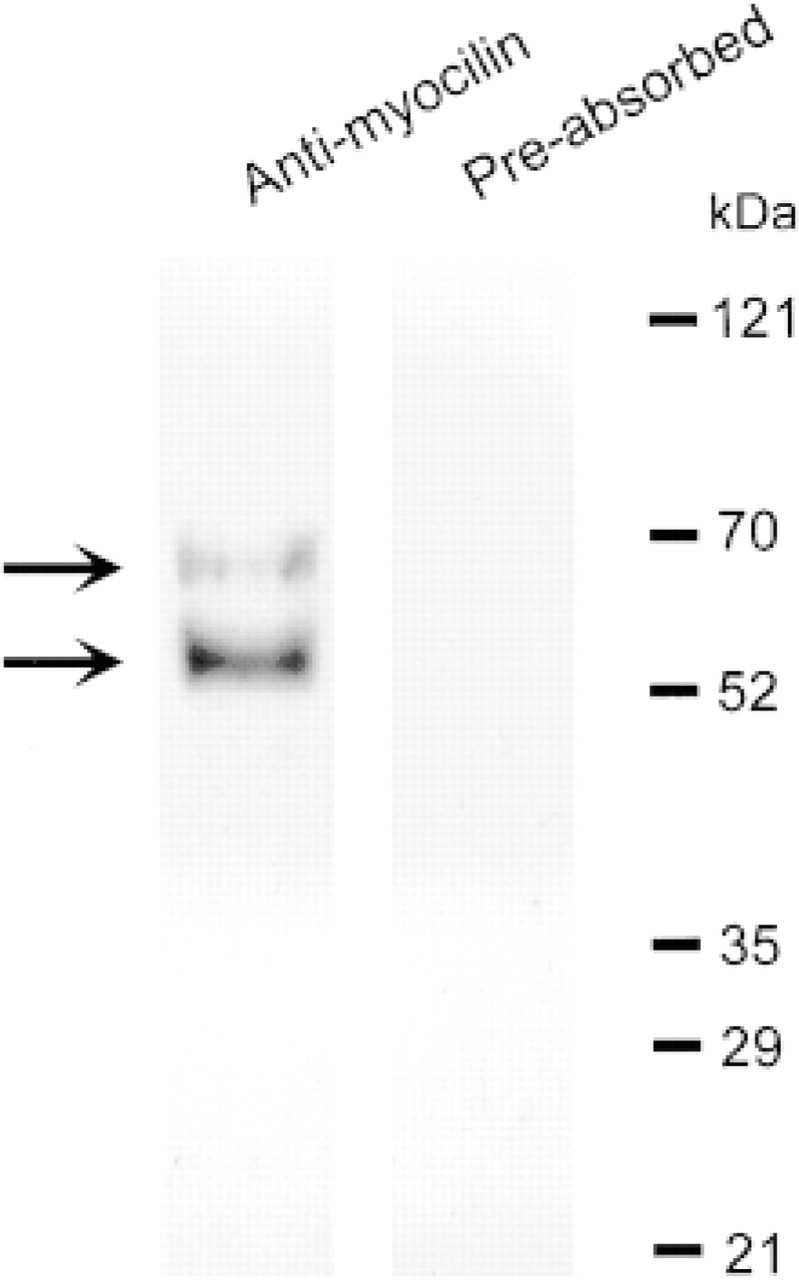
Western blotting analysis. Extracts of human TM tissues were subjected to electrophoresis and the proteins resolved were transferred to nitrocellulose membranes. Immunoblotting was performed with either anti-myocilin or the peptide-preabsorbed antibody. Molecular weight markers are shown at right and the 66-and 57/55-kD myocilin bands are indicated by arrows. These bands were not observed when the membrane was probed with the peptide-preabsorbed antibody.
Immunogold Labeling and EM Study
Ultrathin sections were incubated in 1% bovine serum albumin in PBS for 15 min at RT to minimize nonspecific binding. The blocking solution was removed and the grids were incubated at RT for 3 hr with anti-myocilin (1:200). After the antibody incubation, the grids were rinsed by shaking thoroughly in a mixture of 0.5 M NaCl and 0.05% Tween-20 in 0.1 M phosphate buffer. The sections were incubated at RT for 1 hr with 12-nm colloidal gold-conjugated goat anti-rabbit IgG (Jackson Immuno Research) at a dilution of 1:30 in blocking solution. They were then washed, counter-stained with uranyl acetate, and examined under a transmission EM (JEM-1220; JEOL, Tokyo, Japan) at 80 kV accelerating voltage. For negative controls, anti-myocilin at the same dilution preabsorbed with the immunogenic sythetic peptide was used.
To identify the structures with which myocilin was associated, double labeling (Bendayan 1982) was carried out with several MAbs specific for cell organelles. The MAbs included anti-cytochrome c oxidase subunit II (1:100; Molecular Probes, Eugene, OR), anti-vimentin (1:100; BioGenex, San Ramon, CA), anti-actin (1:20; Chemicon, Temecula, CA), and anti-β-tubulin (1:25; BioGenex). Colloidal gold (6 nm)-conjugated goat anti-mouse IgG (Jackson Immuno Research) was used as the secondary antibody.
Quantification of Immunogold Labeling
To quantify the density of gold labeling, 10 photomicrographs at a magnification of × 10,000 were taken and a total of 50 images were randomly chosen and enlarged to ×50,000. The gold particles associated with intracellular structures such as mitochondria, vesicles, acting filaments, and intermediate filaments in both control and DEX-treated cultured cells were counted using a Microtek imaging system and the MetaMorph software (Universal Imaging; West Chester, PA). The labeling density was expressed as either number of particles/μm2 or number of particles/structure. The density in the surrounding cytosol in cultured cells (nonspecific labeling) was also counted. Differences in labeling between each structure and the cytosol were statistically evaluated by Student's f-tests. In normal TM tissues, gold particles associated with TM cells and the ECM were analyzed in the same manner. The labeling density was statistically compared with that in the intertrabecular space.
Results
Myocilin in Cultured Human TM Cells
In both DEX-treated (Figure 2A) and control TM cells, immunogold labeling of myocilin was localized to mitochondria. This protein was also associated with cytoplasmic filaments (Figures 2A and 2B) and, to a lesser degree, with intracellular vesicles (Figure 2B). When the peptide-preabsorbed antibody was used in place of anti-myocilin, the gold labeling was reduced to a minimal level (Figure 2C). Quantification of the gold labels (Table 1) confirmed that the mitochondrion was a major site of myocilin association. The cytoplasmic filaments and vesicles were also significantly (p < 0.0001) more densely labeled than the surrounding cytosol.
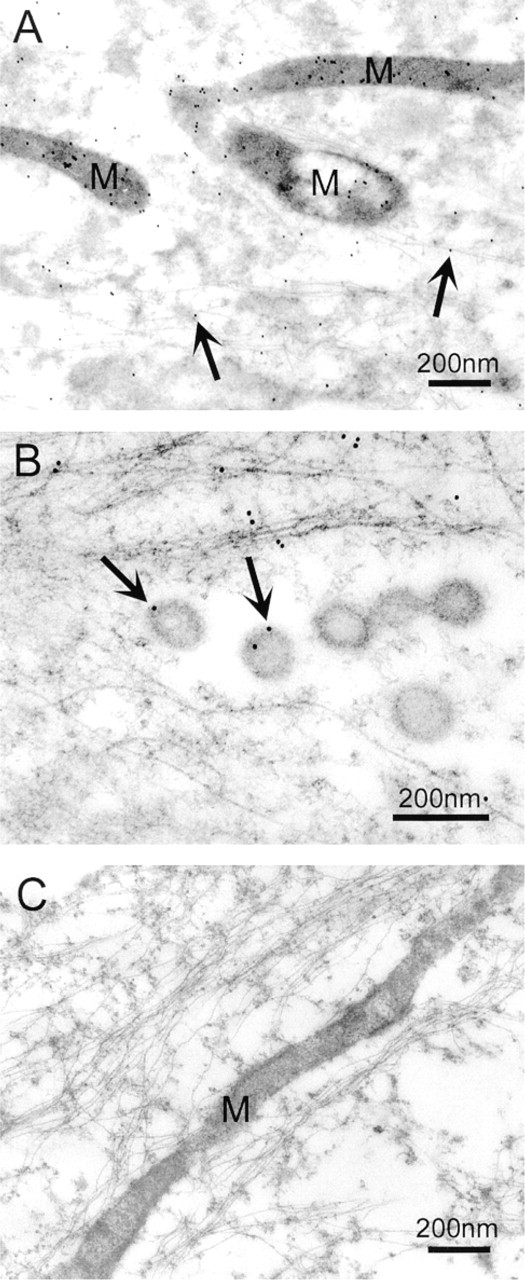
Immunogold labeling of myocilin in DEX-treated cultured human TM cells. (
Labeling density of myocilin in control and DEX-treated TM cells in culture a
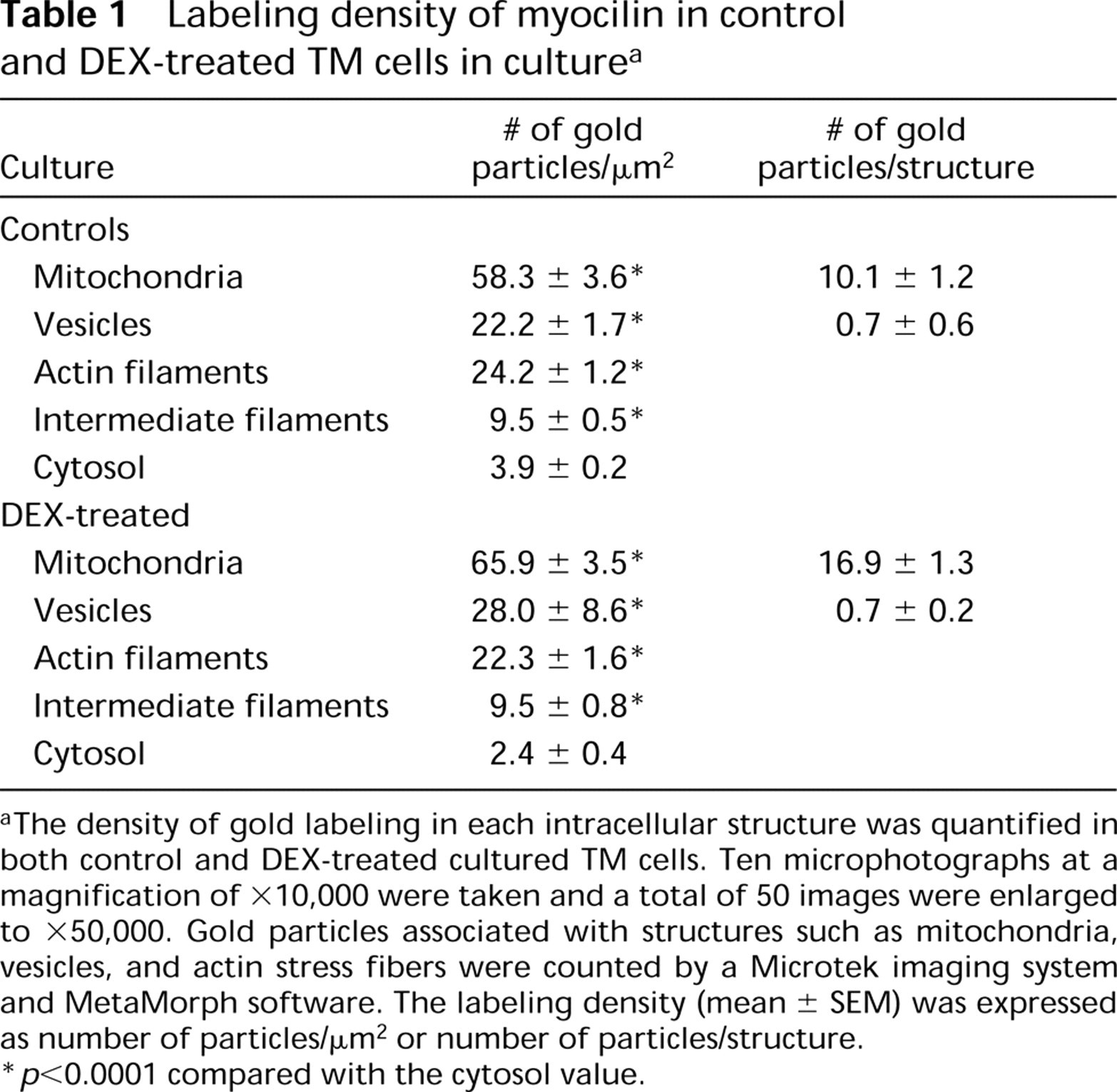
aThe density of gold labeling in each intracellular structure was quantified in both control and DEX-treated cultured TM cells. Ten microphotographs at a magnification of ×10,000 were taken and a total of 50 images were enlarged to ×50,000. Gold particles associated with structures such as mitochondria, vesicles, and actin stress fibers were counted by a Microtek imaging system and MetaMorph software. The labeling density (mean ± SEM) was expressed as number of particles/μm2 or number of particles/structure.
∗ p < 0.0001 compared with the cytosol value.
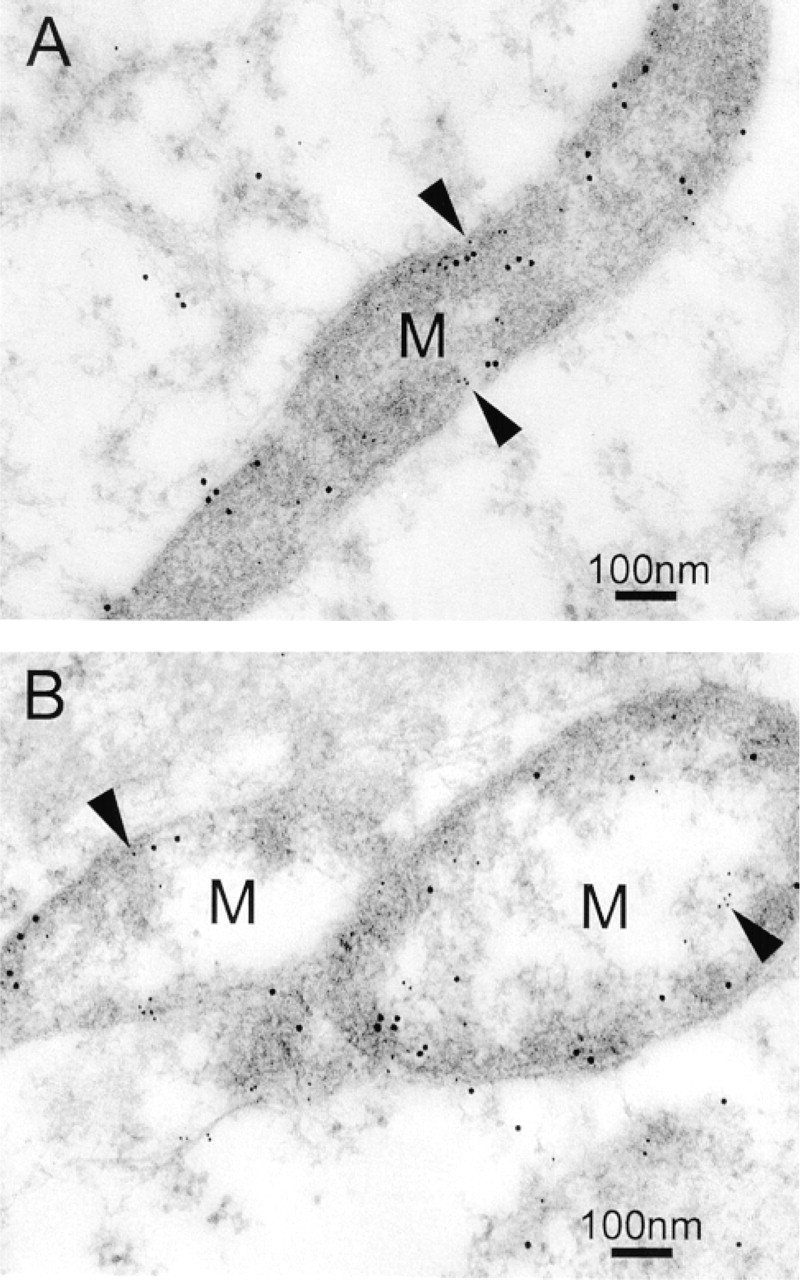
Double labeling experiments performed using different sizes of gold particles further revealed that myocilin co-localized with cytochrome c oxidase subunit II to mitochondria (Figure 3), with vimentin to intermediate filaments in the cell cortex (Figure 4A), and with actin to stress fibers (Figure 4B). The co-localization was observed in all three lines of cultured human TM cells, with or without the DEX treatment. Myocilin did not appear to co-localize with β-tubulin, a component of microtubules, although an association with centrosomes was evident (Figure 4C).
Myocilin in Normal Human TM Tissues
In normal human TM tissues, myocilin-associated colloidal gold particles were associated with TM cells and with elastin-like fibers, as well as their surrounding electron-dense matrices, in the beams of the uveal and corneoscleral meshworks (Figure 5A). In the JCT region, immunogold labeling for myocilin was observed in the JCT cells, the inner wall cells of Schlemm's canal, and in the electron-lucent and electron-dense extracellular matrices (ECMs), including elastin-like fibers (Figure 5C). The labeling density (Table 2) in each region was significantly (p < 0.0001) differently from that in the intertrabecular space. In negative controls, the gold particles were mostly eliminated by replacement of anti-myocilin with the peptide-preabsorbed antibody (Figures 5B and 5D).
Co-localization of myocilin and cytochrome c oxidase subunit II to mitochondria was also seen in the cells of normal human TM tissues (Figure 6A). A giant mitochondrion was observed in a TM cell of the corneoscleral meshwork, in which myocilin was localized particularly to the mitochondrial inner membrane (Figure 6B). Furthermore, in the cells of the corneoscleral meshwork, myocilin was associated with intermediate filaments labeled by vimentin (Figure 6C).
Immunogold double labeling. Cultured human TM cells without (
Discussion
Using a postembedding immunogold EM method, this study provides conclusive evidence that myocilin is localized at both intra- and extracelluar sites in the TM. Of special interest in the association of myocilin with mitochondria. In a previous immunofluorescence study, confocal scanning laser microscopy disclosed that myocilin co-localized with mitochondria, especially in the perinuclear regions of cultured human TM cells either with or without DEX treatment (Wentz–Hunter et al. 1999). This somewhat unexpected finding was subsequently confirmed by subcellular fractionation and Western blotting experiments showing that the previously reported 55/57-kD doublet and the 66-kD forms (Nguyen et al. 1998) of myocilin protein co-sedimented and co-enriched with the purified mitochondrial fraction of TM cells. The current visualization of myocilin labeling in the rod-shaped (Grierson et al. 1986) mitochondria provides a crucial piece of evidence to establish the myocilin–mitochondria association in a more direct manner.
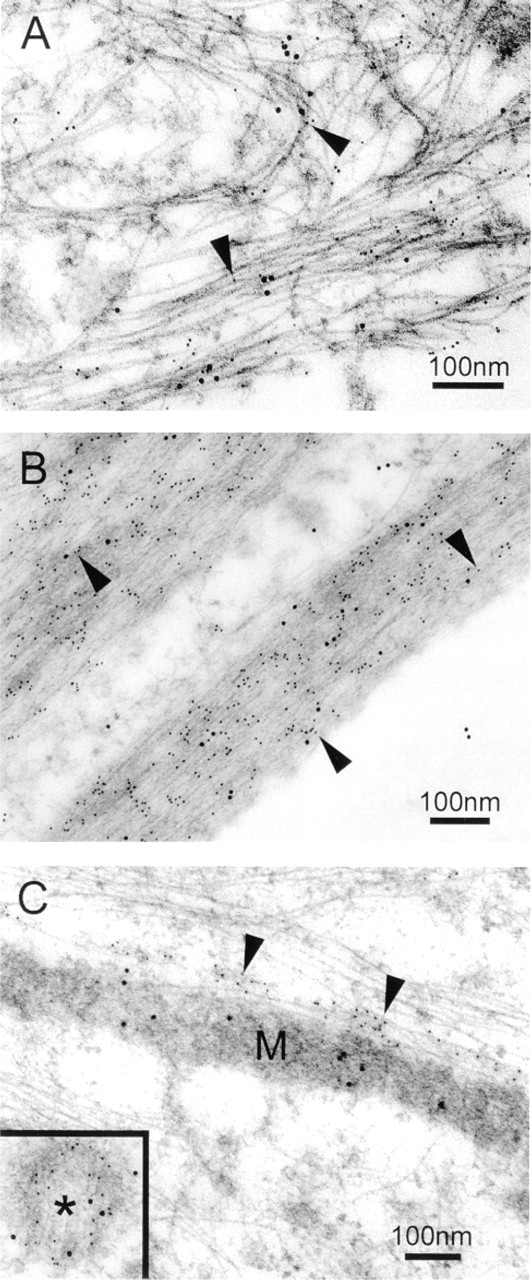
Immunogold double labeling of DEX-treated TM cells (
The mitochondrial localization was observed not only in cultured TM cells but also in cells residing in TM tissues. The biological significance of such a mitochondrial connection is presently unknown. However, because further experiments demonstrated myocilin location in the inner membrane of mitochondria, it is tempting to speculate that myocilin may participate in the energy transfer and metabolic activities that normally take place at this site (Hatefi 1985). The route of mitochondrial translocation also awaits further investigation, although a search of the mitochondria-targeting sequence (Gavel and von Heijne 1990) did identify a signaling segment (amino acid residues 1–47) at the N-terminus of myocilin.
Because of the myosin-like domain in its gene structure, myocilin has been suspected to be connected with actin and/or microtubules (Kubota et al. 1997). This notion is corroborated by the current immunogold study showing that myocilin is observed, both in cultured cells and in TM tissues, to be associated with cytoskeletal structures including intermediate filament vimentin and actin stress fibers. The cytoskeletal elements, especially actin, are known to determine cell shape and polarity and to mediate a variety of essential biological functions, including adhesion and migration in eukaryotic cells (Elson 1988). The possible physiological significance of the TM cytoskeleton in relation to the aqueous humor outflow facility (Epstein and Rohen 1991) and glaucoma also has long been a subject of considerable attention. Chelating agents and cytoskeleton-active drugs that trigger alterations on cytoskeletal elements have been shown to affect the overall shape of TM cells and the aqueous humor outflow facility. For example, EDTA induced an increase in outflow when perfused into monkey eyes (Bill et al. 1980; Hamanaka and Bill 1987). The increase was associated with morphological changes, such as separation of junctions between TM cells and breaks between the cells in the inner wall of Schlemm's canal. Cytochalasins that interfere with the polymerization of actin microfilaments caused retraction of cells, widening of the intercellular spaces (Perkins et al. 1988), and alteration of the outflow facility in monkeys (Kaufman and Erickson 1982). At present, it is unclear whether myocilin binds directly to vimentin and actin or indirectly to intermediate filament-associated and/or actin binding proteins. The interaction, in any case, may have a key role in maintenance of the TM cell integrity and the normal aqueous humor outflow. Changes due to mutations or altered expression levels of myocilin may affect the cytoskeletal structure and lead to pathological consequences.
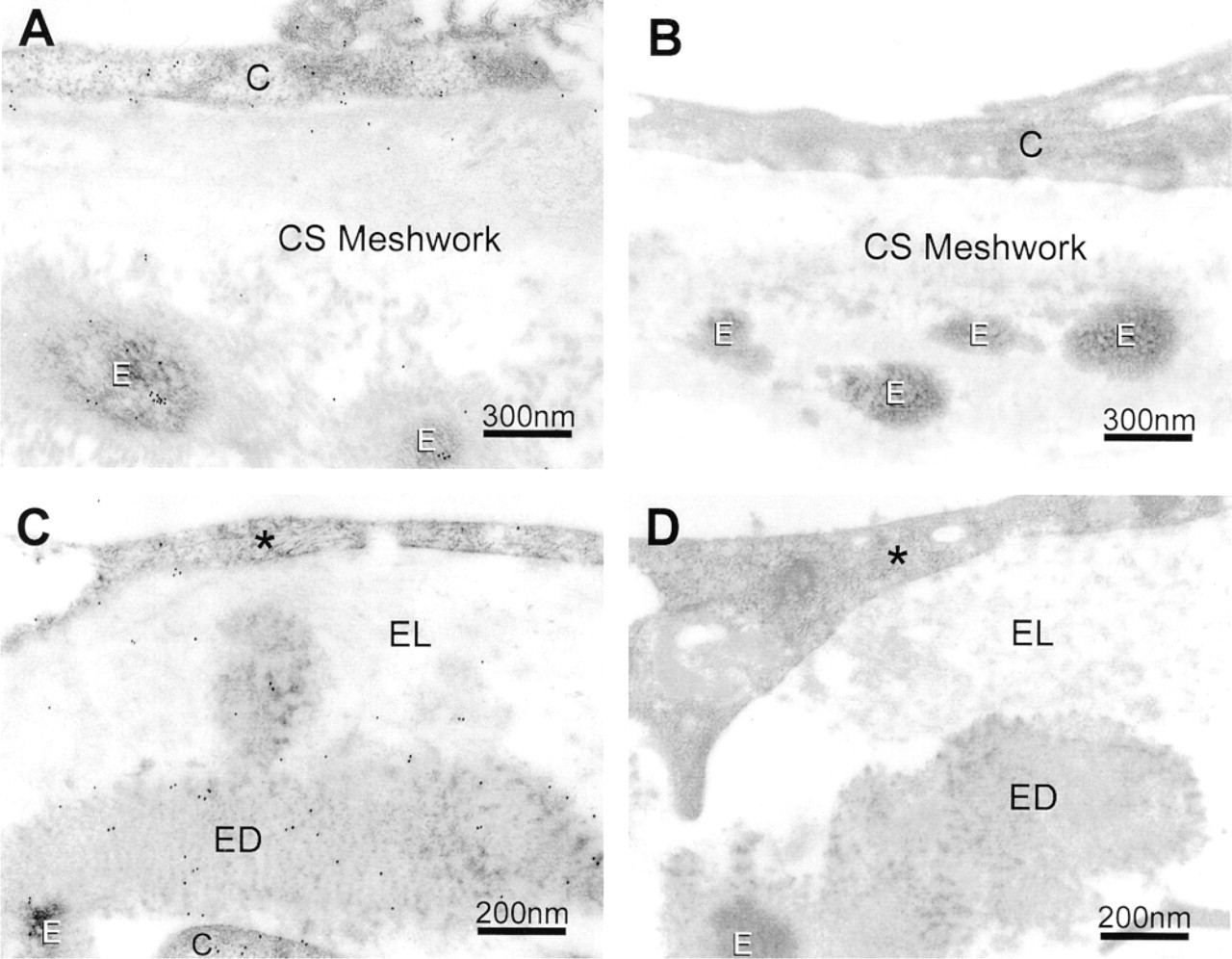
Immunogold labeling of myocilin in normal human TM tissues. (
Our data, although demonstrating the association of the endogenous myocilin with actin and vimentin, indicated no myocilin connection with β-tubulin or microtubules. This observation is in direct contrast to a recent result showing that, with green fluorescent protein as a marker, highly overexpressed myocilin in transfected cells co-localized with microtubules (Mertts et al. 1999). The differences between the two studies may be related to the expression levels of myocilin. It is possible, as has been previously reported for presenilin (Annaert et al. 1999) that, as a consequence of overexpression, the protein processing and the subcellular localization may be modified.
In their study of the retina, myocilin was noted by Kubota et al. (1997) to localize in the basal body of the connecting cilium in photoreceptor cells. The basal body is a microtubule organizing center, also termed the centrosome, that is constituted predominately of β- and γ-tubulins to form ring-like complexes (Zheng et al. 1995). In TM cells, myocilin was likewise detected in centrosomes (Figure 4C), in agreement with that reported by Kubota et al. (1997) and Mertts et al. (1999). Nevertheless, this result was seemingly in conflict with the lack of myocilin–tubulin association discussed above. On the other hand, although tubulin isoforms appear to play a direct role in microtubule nucleation, an increasing number of other components or protein complexes, such as pericentrin, kinases, phosphatases, cyclins (Zimmerman et al. 1999), and a network of keratin filaments (Salas 1999), have also been shown to participate in the organization of centrosomes. Myocilin is likely to be in association with centrosome components other than tubulins.
Labeling density of myocilin in normal human TM tissues a

aThe density of gold labeling in each region was quantified in human TM tissues. Ten microphotographs at a magnificaiton of × 10,000 were taken and 50 images were enlarged to X50,000. Gold particles associated with TM cells and the ECM were counted by a Microtek imaging system and MetaMorph software. The labeling density (mean ± SEM) was expressed as number of particles/μm2.
∗ p < 0.0001 compared with the intertrabecular space value.
Myocilin labeling was observed to a lesser degree on vesicles. The vesicular association, also suggested recently by Stamer et al. (1998), is not entirely surprising. The myocilin gene contains a signal sequence (Nguyen et al. 1998) and the gene product has been shown to be secreted into the culture medium. This protein presumably is posttranscriptionally modified and translocated through, and thus associates with, various vesicular structures, such as endoplasmic reticulum, at different stages of protein processing.
The investigations using in vivo TM tissues, demonstrating the same pattern of myocilin intracellular distribution as that in cultured cells, validate the in vitro culture findings. More significantly, the tissue studies provide, for the first time, experimental evidence depicting the extracellular localization of myocilin. In the uveal and corneoscleral meshwork, myocilin labeling was detected in the central core of trabecular beams, particularly in association with elastin-like fibers. Myocilin was also seen dispersed in the ECM of JCT regions. ECM components underlying the cells are believed to be important for a properly functioning TM (Acott and Wirtz 1996; Yue 1996). In the TM of patients with primary open-angle glaucoma, excessive abnormal accumulation of ECM materials has been reported, and sheath-like “plaque materials” (Rohen 1983; Lütjen–Drecoll et al. 1986) that are partially made up of elastin-like fibers (Umihara et al. 1994) have been described in a number of electron microscopic, histological, and immunological studies (Segawa 1975; Rodrigues et al. 1980; Lütjen–Drecoll et al. 1981; Umihara et al. 1994). Myocilin, through its interactions with ECM elements, may contribute to maintenance of the normal aqueous outflow and development of the disease conditions.
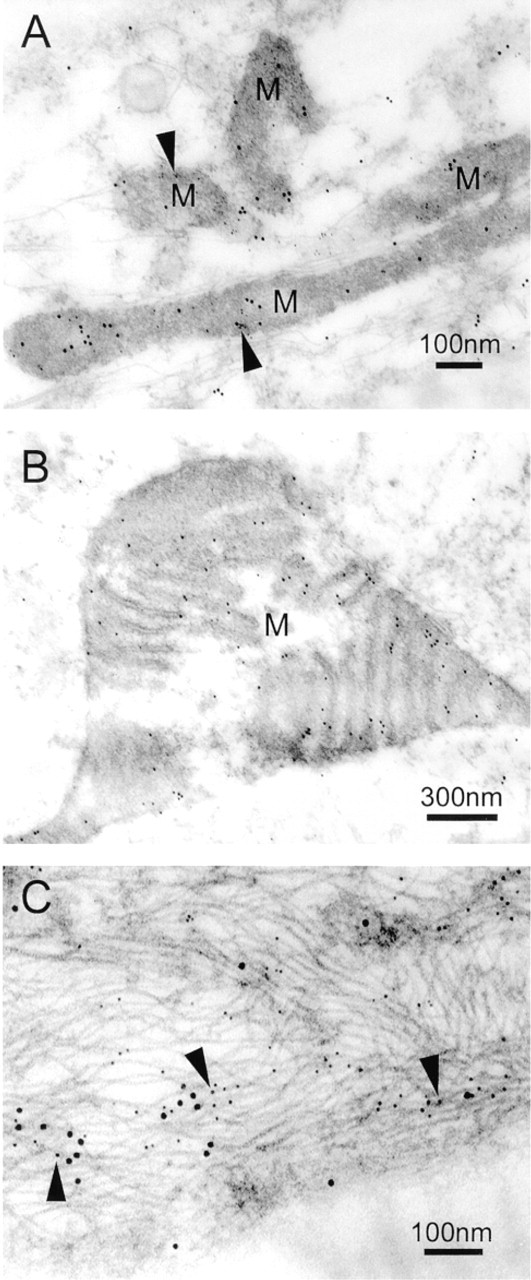
Immunogold double labeling. (
Topical and systemic treatments with glucocorticoids such as DEX affect the fluid dynamics of the eye and raise IOP in normal human eyes (Armaly 1963a,b). DEX is well documented (Polansky et al. 1997; Nguyen et al 1998; Tamm et al. 1999) to induce myocilin expression by TM cells. However, there has been little evidence establishing a direct link between the DEX-induced IOP elevation and the myocilin upregulation. On this note, it is noteworthy that the localization of endogenous myocilin was virtually identical in TM cells with or without DEX treatment. Some mitochondria, however, were somewhat swollen or had irregular shapes (not shown), suggesting that the cells might be midly stressed after exposure to DEX.
In summary, myocilin was ultrastructurally localized to multiple intracellular and extracellular sites in normal human TM. This protein, depending on the sites at which it localizes, may exert different biological functions. The current localization data help shed light into the possible functions of myocilin and provide a basis for future investigative pursuits on this novel protein.
Footnotes
Acknowledgements
Supported in part by research grants EY 05628, EY 03890, and core grant EY 01792 from the National Institutes of Health (Bethesda, MD) and by a Senior Investigator Award from Research to Prevent Blindness, Inc., New York, NY. K.K.W.H. was supported by an Individual NSRA EY 06889 from the National Institutes of Health.
We thank Kira Lathrop for expert advice in imaging and Chan Boriboun for assistance in tissue culture experiments.
