Abstract
Leptin is a key mediator in the maintenance of neuroendocrine homeostasis. Recently, leptin and leptin receptor expression were demonstrated in non-tumorous and adenomatous human pituitaries. This study was performed to determine the subcellular localization of leptin in human adenohypophyses (n = 3) and in various types of pituitary adenoma (n = 16). Immunoelectron microscopy showed leptin immunolabeling in most hormone-producing cells of the human non-tumorous adenohypophysis, but not in stellate cells. Labeling was noted over secretory granules. Immunocytochemistry using double labeling revealed leptin expression in GH-, ACTH-, TSH-, and FSH/LH-containing cells but not in PRL cells. The percentage of immunopositive cells and the intensity of immunostaining varied considerably among the various cell types. Immunoelectron microscopy with double gold labeling showed co-localization of leptin and adenohypophysial hormones in the same secretory granules. Among pituitary tumors, leptin immunolabeling was evident only in corticotroph adenomas. Compared to non-tumorous corticotrophs, leptin immunoexpression was less abundant in corticotroph adenomas. The presence of leptin and adenohypophysial hormones in the same secretory granules suggests that leptin is secreted concomitantly with various adenohypophysial hormones and that its release is under the control of hypothalamic stimulating and inhibiting hormones. (J Histochem Cytochem 48:1147–1152, 2000)
Leptin, a circulating hormone secreted by adipocytes, influences body weight homeostasis through effects on food intake and energy expenditure (Campfield et al. 1995; Halaas et al. 1995; Pelleymounter et al. 1995; Levin et al. 1996; Flier 1997). It also modulates other processes, including lipid metabolism, hemopoiesis, pancreatic β-cell function, ovarian activity, thermogenesis, and angiogenesis (Gainsford et al. 1996; Karlsson et al. 1997; Shimabukuro et al. 1997; Sierra-Honigmann et al. 1998). This multiplicity of biological effects on extraneural tissues is also reflected by the fact that the leptin receptor is expressed in many organs, including the hypothalamus. In the hypothalamus, leptin influences the activity of the hypothalamus–pituitary axis by inhibiting neuropeptide Y (NPY) gene expression and increasing expression of corticotropin-releasing hormone (CRH) mRNA (Stephens et al. 1995; Huang et al. 1998; Casanueva and Dieguez 1999). Several lines of evidence indicate the existence of leptin receptor splice variants that are expressed in extra-adipocyte cells (Mercer et al. 1996; Fei et al. 1997; Guan et al. 1997; Baskin et al. 1999).
On the basis of previous studies, the anterior pituitary may play a role in the regulatory effects of leptin. For example, it has been reported that leptin receptor is expressed in rodent anterior pituitary and that its gene expression is regulated by GH and/or GHRH (Zamorano et al. 1997; Cai and Hyde 1998, 1999). Recent studies demonstrated that leptin is also expressed in the rat pituitary (Morash et al. 1999; Jin et al. 2000). Jin et al. (1999) found that leptin and leptin receptor were widely expressed in normal and neo-plastic human anterior pituitary cells, a finding that suggests the existence of a paracrine/autocrine loop involving leptin activity in the pituitary.
The present study is the first to reveal the subcellular localization of leptin in non-tumorous and adenomatous human pituitary cells, using immunoelectron microscopy.
Materials and Methods
Nineteen transsphenoidally resected pituitary specimens were examined. Three represented normal (non-tumorous) adenohypophysis removed in association with an adenoma. On the basis of immunohistochemical and ultrastructural studies, the samples included four GH (two sparsely and two densely granulated), three PRL, three TSH, three FSH/LH, and three oncocytic null-cell adenomas.
All specimens were fixed in 2.5% glutaraldehyde in Sorensen's phosphate buffer (pH 7.4). After a thorough washing in Sorensen's buffer, the samples were dehydrated in a graded ethanol series, embedded in an Epon–Araldite mixture, and investigated by transmission electron microscopy. Portions of the tissues were postfixed in 1% osmium tetroxide, whereas other portions remained unosmified.
The postembedding double-immunogold labeling technique of Bendayan (1982) was employed for simultaneous detection of leptin and adenohypophysial hormones. One side of the grids was incubated at 37C for 12 hr with specific antisera directed towards either leptin or adenohypophysial hormones. For immunocytochemical demonstration of leptin, a 1:100 dilution of rabbit polyclonal antibody raised against human leptin (Sigma–Aldrich; St Louis, MO) was used. Details of immunocytochemistry for adenohypophyseal hormones, including sources of antibodies, dilutions, duration of exposure, and control procedure have been described in previous publications (Kovacs et al. 1989, 1991). Next, the grids were treated at 37C for 1 hr with gold-labeled goat anti-rabbit IgG (Biocell Research Laboratories; Cardiff, UK). The gold particles utilized measured either 10 or 20 nm in diameter. Between each step, grids were washed in 0.2 M PBS (pH 7.5) admixed with 0.2% coldwater fish gelatin (Sigma–Aldrich). For double immunostaining, the procedure was repeated on the other side of the grid using another specific antibody and a colloidal gold conjugate of a different size. After immunolabeling, sections were stained with uranyl acetate and examined in a Philips 410 LS electron microscope.
To ensure specificity, three control procedures were used successively with each immunolabeling procedure. (a) The specific primary antibody was replaced with the antibody diluent (0.2 M PBS admixed with 0.2% coldwater fish gelatin). (b) The specific primary antibody was substituted by normal serum rabbit (not immune serum). (c) The specific polyclonal antiserum was preabsorbed with homologous and heterologous antigens. Absorption tests were carried out as described previously (Vidal et al. 1997; Vidal et al. 2000).
After staining, immunoreactive cells were counted to express the percentage of leptin-immunopositive cells in each pituitary sample. In such cells, the percentage of leptin-labeled secretory granules was also determined.
Data were tested for statistical significance using the SPSS statistical computer program (SPSS; Chicago, IL). All data were evaluated by one-way analysis of variance and the Student's t-test as a multiple comparison method. Differences of p < 0.05 were considered statistically significant.
Results
Immunoelectron microscopy demonstrated leptin immunolabeling in most hormone-producing glandular cells of the human non-tumorous adenohypophyses. No positive reaction was found in the stellate cells. Leptin staining was detected only in unosmified tissue and was abolished by absorption of the specific primary antibody with 50 μg/ml purified leptin (Eli Lilly; Indianapolis, IN). Positive cells showed specific immunolabeling over secretory granules (Figures 1–4). Leptin-positive secretory granules were fewer in number than immunonegative granules.
Because immunopositive normal adenohypophysial cells varied in ultrastructural appearance, the double immunogold method was applied to specifically identify those cells expressing leptin. The double immunocytochemical method demonstrated that, with the exception of lactotrophs, all forms of hormone-producing adenohypophysial cells showed leptin immunolabeling (Figures 1–4). The percentage of immunopositive cells varied considerably within each of the various cell types. Studies of the co-localization of leptin and pituitary hormones showed that corticotrophs were most frequently leptin labeled (70–80% of ACTH-immunopositive cells). Significantly lower frequencies of leptin immunopositivity were observed among other pituitary cell types, including somatotrophs (10–15%), thyrotrophs (20–25%), and gonadotrophs (25–30%). Immunocytochemistry also showed that intensity of staining varied significantly in each pituitary cell type. Lastly, whereas 20–25% of secretory granules in corticotrophs were labeled for leptin, only 5–10% were positive in somatotrophs. The frequency of leptin-labeled granules in both in gonadotrophs and thyrotrophs was 15–20%.
The double immunogold method showed co-localization of leptin and specific pituitary hormone in the same secretory granules. A few secretory granules that were usually smaller in size contained only leptin (Figure 2).
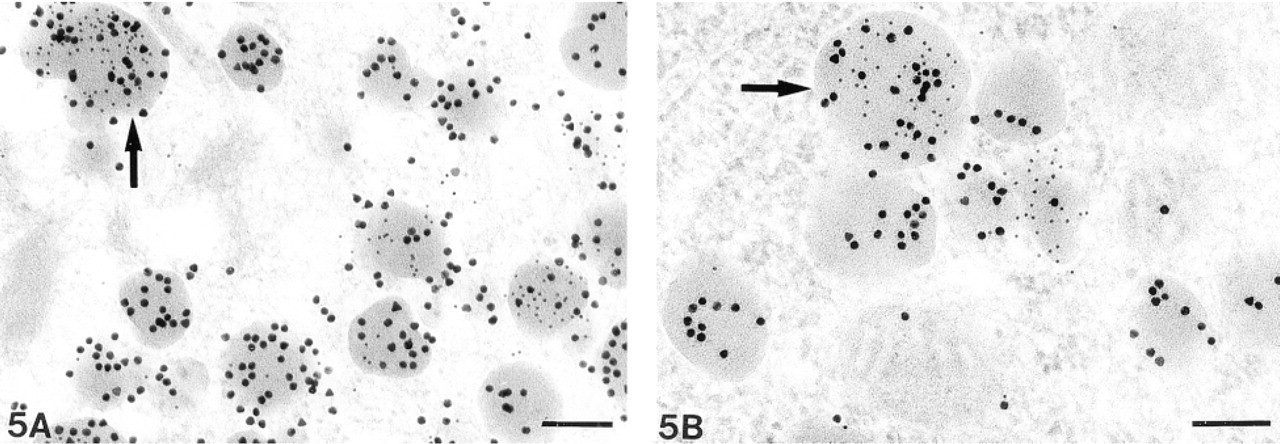
Analysis of 16 pituitary adenomas by immunoelectron microscopy demonstrated leptin immunolabeling only in corticotroph adenomas (Figure 5). Positive immunostaining was not identified in somatotroph, lactotroph, thyrotroph, gonadotroph, and oncocytic null-cell adenomas. Furthermore, leptin immunoreactivity was significantly less in corticotroph adenomas than in non-tumorous corticotrophs; only approximately 30–40% of adenomatous coticotrophs were leptin-immunopositive.
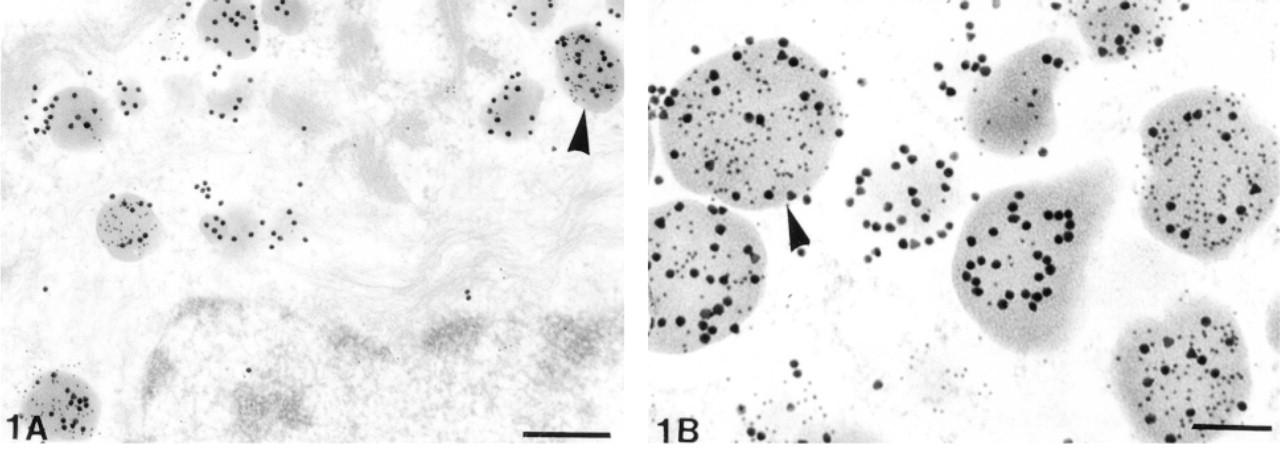
(
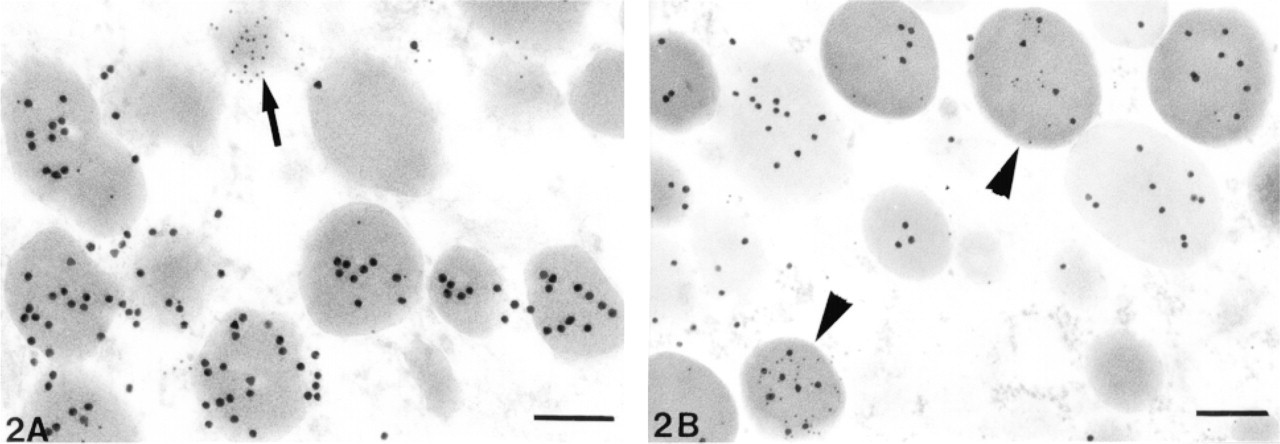
Double immunogold demonstrated monohormonal (arrow) and bihormonal (arrowheads) granules labeled for leptin (10-nm gold particles). (
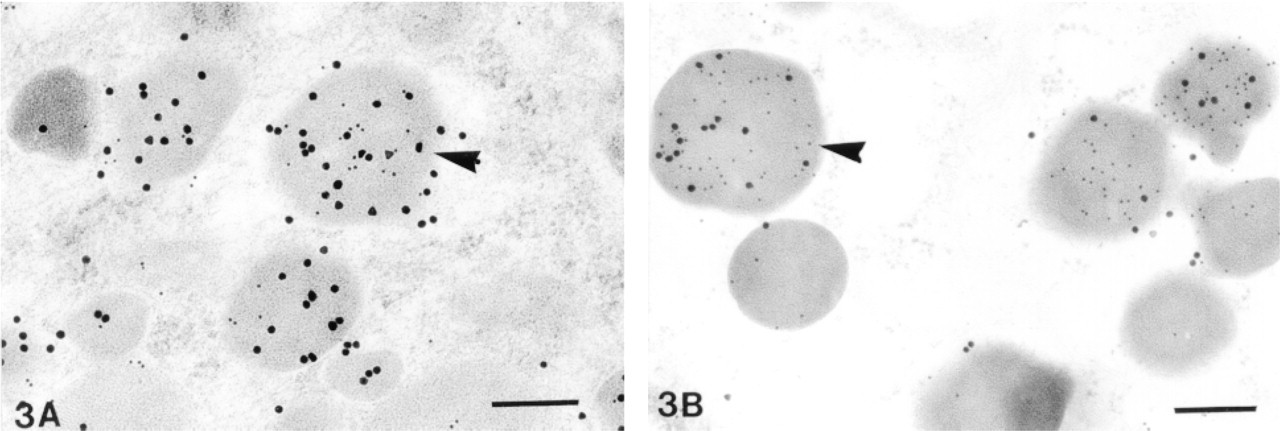
(
Discussion
All endocrine cells possess secretory pathways for the transportation of their products to the plasma membrane. In pituitary cells and in various other endocrine cells that store proteins, the secretory pathway includes their accumulation in secretory granules. Previous studies have shown that secretory granules of adenohypophysial cells store not only pituitary hormones but other proteins as well. These include vascular endothelial growth factor (Vidal et al. 1999), renin, angiotensinogen (Vila–Porcile and Corvol 1998), and deoxyribonuclease I (Shimada et al. 1998). Contained within the granule matrix, these substances must be simultaneously released with the various pituitary hormones (Vila–Porcile and Corvol 1998; Vidal et al. 1999). To our knowledge, the present study is the first to demonstrate leptin storage in the secretory granules of adenohypophysial cells. This observation suggests that leptin secretion in the pituitary follows a different secretory pathway than in adipocytes. In contrast to adipose tissue, in which newly formed leptin is immediately released (Hardie et al. 1996), the storage of leptin in the secretory granules of the adenohypophysis indicates that the secretory cells retain leptin in granules until stimulated. On discharge of their contents, through either exocytosis or transmembrane diffusion, large amounts of leptin may be rapidly made available on demand. On the basis of the present data, it is not possible to determine the neuroendocrine signal that regulates leptin production in adenohypophysial cells. Co-storage of leptin with pituitary hormones in the same secretory granules of various cell types suggests that a similar response is possible to a variety of stimuli. However, regarding the low percentage of leptin-immunopositive granules observed in most adenohypophysial cells (5–25%), it may well be that secretory granules containing or lacking leptin can be differentially affected, leptin being preferentially retained or released. This suggestion is consistent with published observations regarding rat gonadotrophs (Farnworth 1995; Thomas and Clarke 1997). In these cells, FSH and LH are packaged in separate secretory granules, but all gonadotrophs contain both types. However, extrusion of FSH- and LH-containing granules is regulated differently, i.e., they do not respond to the same stimuli. Whereas more FSH than LH is released in the basal state, stimulation releases more LH than FSH. It is possible that some granule membrane components preventing exocytosis in the absence of large increases in Ca2+ are less effectively packaged in FSH-containing granules (Dannies 1999). Similar mechanisms may affect differential secretion of leptin.
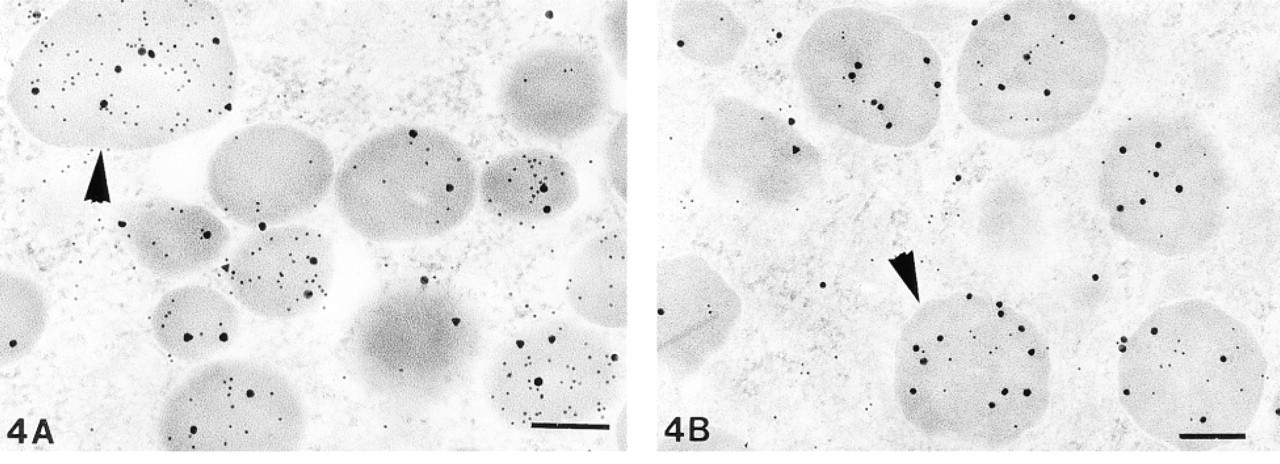
(
(
Our immunoelectron microscopic findings indicate that in the non-tumorous adenohypophysis leptin is expressed in all secretory cells except lactotrophs. Similar observations were previously reported using double-label immunohistochemistry and in situ hybridization at the light microscopic level (Jin et al. 1999). However, our results differ from those of Jin et al. (1999), who found leptin expression in the stellate cells as well as in occasional lactotrophs. One possible explanation for these different results may reside in different sensitivities of the light and electron microscopic methods employed. For example, it is well known that the immunogold technique permits not only precise localization of specific molecules within cells but quantitative evaluation of labeling intensity as well. However, the immunogold method also has limitations. For detection of tissue antigens present in very low amounts, it may have shortcomings in terms of labeling efficiency (Mayer and Bendayan 1999). Concentration of adenohypophysial hormones in secretory granules is not only extensive but varied. It has been shown that prolactin is 200 times more concentrated in the secretory granules than within the endoplasmic reticulum of PRL cells (Farquhar et al. 1978). An analogous explanation may pertain to the detection of leptin in the secretory granules but not in Golgi regions or endoplasmic reticulum. Another possibility is that leptin antigenicity decreases during processing for electron microscopy. Some antigens are sensitive to fixation and postembedding protocols; therefore, standard processing may affect the immunogold method. Indeed, our finding that leptin immunoreactivity was limited to unosmified tissue indicates that its antigenicity was lost during osmification.
The present study revealed stronger leptin immunoreactivity in normal adenohypophysial cells than in adenomas. This is in agreement with the findings of Jin et al. (1999). Furthermore, leptin immunoreactivity was detected only in the secretory granules of all corticotroph adenomas, no positivity being seen in adenomas of other types. These findings are in keeping with the observation of greater leptin expression in RT-PCR studies of corticotroph tumors (Jin et al. 1999).
Although the significance of leptin expression in the adenohypophysis is unknown, various lines of evidence suggest that leptin plays an important role in regulating adenohypophysial activity. Prior studies have demonstrated that leptin acts via specific receptors expressed by most human and rodent adenohypophysial cells. In addition, it was found that leptin stimulates GH, LH/FSH, and PRL release (Barash et al. 1996; Carro et al. 1997; Yu et al. 1997; Tannenbaum et al. 1998). Recently, Jin et al. (1999) reported that leptin is involved in the regulation of growth and differentiation of adenohypophysial cells. The expression of leptin in adenohypophysial cells may also affect the functional activity of peripheral endocrine organs. For example, it has been found that leptin has an important influence on the activity of the hypothalamic–pituitary–gonadal and hypothalamic–pituitary–adrenal axes. Because the pituitary is crucial in the control of the functional activity of adrenals, gonads, thyroid, and many other organs, it is possible that leptin produced in the pituitary may be a key integrator of several neuroendocrine functions related to energy homeostasis. Therefore, the simultaneous release of leptin with pituitary hormones may (a) affect the functional activity of various organs, (b) control body weight homeostasis, and (c) have an autocrine and/or paracrine effect on adenohypophysial cells.
Further studies are needed to establish the mechanism controling leptin expression at the pituitary level and to examine the possible association between decreased leptin expression and secretory activity as well as growth of pituitary adenomas.
Footnotes
Acknowledgments
Supported in part by the Research Council of St. Michael's Hospital, by Mr and Mrs Stephen Jarislowsky, and by the Lloyd Carr–Harris foundation. Dr. Sergio Vidal was supported by a research grant from the University of Santiago de Compostela, Spain.
We are indebted to Ms Sandy Briggs and Ms Elizabeth McDermott for technical assistance and to the staff of St. Michael's Health Sciences Library for their contribution to this study.
