Abstract
The expression of a novel calcyclin (S100A6) binding protein (CacyBP) in different rat tissues was determined by Western and Northern blotting. Polyclonal antibodies against recombinant CacyBP purified from E. coli exhibited the highest reaction in the brain and weaker reaction in liver, spleen, and stomach. CacyBP immunoreactivity was also detected in lung and kidney. Densitometric analysis showed that the concentration of CacyBP in the soluble fractions of total brain and cerebellum is approximately 0.17 and 0.34 ng/μg protein, respectively. Northern blotting with a specific cDNA probe confirmed the high level of CacyBP expression in the rat brain and lower levels in other tissues examined. Immunohistochemistry and in situ hybridization of rat brain sections revealed strong expression of CacyBP in neurons of the cerebellum, hippocampus, and cortex. The in situ hybridization detected CacyBP in hippocampus as early as P7 (postnatal day 7) and a peak of expression at P21, and the expression signal was preserved until adulthood. In the entorhinal cortex, the peak of expression was observed at P7, whereas in the cerebellum it was seen at P21. The results presented here show that CacyBP is predominantly a neuronal protein.
C
The new search for Ca2+-dependent targets of calcyclin has led us to the identification of a novel protein (Filipek and Wojda 1996). This protein was found predominantly in the cytosolic fraction of Ehrlich ascites tumor cells and mouse brain. Recently, a 1.4-kb cDNA clone of this protein (named CacyBP) was isolated from a mouse brain cDNA library and sequenced (Filipek and Kuźnicki 1998). The analyzed clone contained an open reading frame followed by a 3′-untranslated region of 662 bp containing a polyadenylation signal 23 bp upstream of the poly(A) sequence. The clone could be translated into a protein containing 229 amino acids with a calculated pI of 7.6. The nucleotide and predicted amino acid sequences indicated that CacyBP was a novel protein, because there was no similar sequence in databases (GenBank and SwissProt). Using a pGEX vector, the recombinant CacyBP was produced in E. coli as a fusion protein with glutathione-S-transferase (GST) and was shown to bind to calcyclin in a physiological range of calcium concentrations (Filipek and Kuźnicki 1998).
In this study we analyzed the distribution of this novel calcyclin binding protein, CacyBP, in different rat tissues using specific antibodies developed against the recombinant CacyBP and using a cDNA probe encoding this protein. We also performed in situ hybridization and immunohistochemistry on rat brain sections to identify CacyBP-positive cells. Because the strongest expression of calcyclin binding protein was found in the brain, particularly in neurons, we estimated the amount of CacyBP in this tissue.
Materials and Methods
Antibodies
Polyclonal antibodies were raised in rabbits. Recombinant GST–CacyBP fusion protein was expressed in E. coli as described by Filipek and Kuźnicki (1998). Before injection, the protein was purified on glutathione–Sepharose beads following the procedure outlined by the manufacturer (Pharmacia; Uppsala, Sweden) and eluted with 10 mM reduced glutathione. Additional purification was achieved by elution from the nitrocellulose as described by Szewczyk and Summers (1988). About 0.2 mg of GST–CacyBP was injected with complete Freund's adjuvant and then, after 4 weeks, the same amount of antigen emulsified in incomplete Freund's adjuvant was injected. Serum was collected 4 weeks after the second injection and was stored at −70C.
Tissue Extracts
Various rat tissues, as well as parts of the brain such as hippocampus, cerebellum, cortex, and striatum, were homogenized using a Polytron at 6000 rpm in a solution containing 1 mM EDTA, 1 mM dithiothreitol, 10 mg/liter leupeptin, 5 mg/liter aprotinin, 20 mg/liter soybean trypsin inhibitor, 1 mM phenylmethylsulfonyl fluoride, and 40 mM Tris, adjusted to pH 7.5 with HCl. The extracts were centrifuged for 1 hr at 4C and 12,000 rpm in an Eppendorf centrifuge. The supernatants were subjected to SDS-PAGE. In all studies that employed animals, the rules established by the Ethical Committee on Animal Research of the Nencki Institute and based on disposition of the President of Polish Republic were strictly followed.
Electrophoresis and Immunoblotting
Gel electrophoresis with 15% (w/v) polyacrylamide containing 0.1% SDS was performed by the method of Laemmli (1970). Proteins separated by SDS-PAGE were transferred electrophoretically to nitrocellulose and CacyBP was detected with serum containing specific polyclonal antibodies diluted 1:1000. Then the blots were allowed to react with secondary antibodies conjugated to horseradish peroxidase, diluted 1:1000 (Sigma; St Louis, MO). Blots were developed with chloronaphthol and H2O2.
Northern Blots
RNA was prepared from various rat tissues using an RNA purification kit obtained from Strategene (La Jolla, CA) and applied on a 1.2% agarose gel in the presence of formaldehyde. Ethidium bromide staining showed similar amounts of RNA in each lane. For detection of CacyBP mRNA, a specific cDNA probe (0.9-kb fragment cDNA between 26 and 931 bp of the original clone) was labeled with [32P]-deoxycytidine 5′-triphosphate ([32P]-dCTP) according to the method of Feinberg and Vogelstein (1983) using the rediprime random primer labeling kit (Amersham Life Science; Poole, UK). Prehybridization and hybridization were performed at 42C. Filters were washed three times for 5 min each in 2 × SSC and 0.2% SDS at room temperature, followed by two 25-min washes at 65C in 0.25 × SSC and 0.2% SDS. The filters were subjected to autoradiography for 48 hr with Amersham film at −70C.
Measurement of CacyBP Concentration in Rat Brain
The proteins from the supernatant fractions of a total brain and cerebellum obtained after centrifugation at 12,000 × g in an Eppendorf centrifuge were separated by 15% SDS-PAGE and transferred to nitrocellulose filters under the conditions described above. Protein concentration was estimated by Bradford's procedure (BioRad reagent; Hercules, CA) with bovine serum albumin as a standard. The intensity of bands identified by the CacyBP antibody were estimated on films after developing the blots with a chemiluminescent kit (Kirkegaard & Perry Laboratories; Gaithersburg, MD). Different time exposures from two independent blots were used for optical density measurements using a Molecular Dynamics (Sunnyvale, CA) densitometer. The concentration of CacyBP expressed in brain and cerebellum was calculated using different amounts of native CacyBP purified as described by Filipek and Wojda (1996) as a quantitative standard.
Immunohistochemistry
Immunohistochemical studies were performed on the rat brain sections. The brain was snap-frozen on dry ice, sliced into 20-μm sections with a cryostat (Reichert–Jung), melted onto slides covered with poly-
In Situ Hybridization
For in situ hybridization analysis (Konopka et al. 1995), brains were dissected and immediately frozen on dry ice as described above. The cryostat sections (20 μm) were prepared and fixed in 4% cold paraformaldehyde in PBS, dehydrated, and prehybridized for 2 hr at 37C in a buffer containing 50% formamide, 2 × SSC, 1 × Denhardt's solution, 200 μg/ml salmon sperm DNA, and 20 mM dithiothreitol. Next, the sections were hybridized overnight in the above solution containing 10% dextran sulfate and α35S-labeled cDNA (0.9 kb fragment cDNA between 26 and 931 bp of the original clone) as a probe at 37C. The sections were washed for 15 min and then for 1 hr in 50% formamide, 2 × SSC at room temperature. Then the sections were exposed for 2 weeks against β-Max Hyperfilm (Amersham). The autoradiograph was analyzed with the aid of the PC-based computer program ProVision.
Results
Tissue Distribution of CacyBP in the Rat
The immune serum was generated against GST–CacyBP fusion protein. Therefore, it contained antibodies against both GST and CacyBP. When immune serum was preabsorbed with CacyBP, it stained only recombinant GST (Figure 1A), but when it was preabsorbed with GST, staining only of purified CacyBP was seen (Figure 1B). Neither GST nor CacyBP was stained on the blots incubated with preimmune serum (not shown). The immune serum recognized only one band of molecular weight 30,000 in the brain extract (Figure 1C). As shown in Figure 1D, this band migrates at the same level as the CacyBP purified from Ehrlich ascites tumor cells and recombinant CacyBP obtained after removing of GST. Thus, despite the fact that immune serum represents a mixture of antibodies specific to both CacyBP and GST, it does not stain proteins other than CacyBP in the brain extract. Therefore, it could be used for immunohistochemical localization in this tissue.
When various rat tissues were tested with the immune serum, the 30,000 immunoreactive protein band was found mainly in the brain, liver, spleen, and stomach (Figure 2A). A moderate level of CacyBP was found in lung and kidney, but in heart and skeletal muscle a band with higher molecular weight (~38,000) was stained. This higher molecular weight protein stained by immune serum may represent a new muscle-specific isoform of CacyBP. However, Northern blotting with the CacyBP cDNA probe (Figure 2B) showed that in all tissues examined a band at the same level (1.8 kb) was observed. The highest mRNA level of CacyBP was found in the brain and a good correlation between the CacyBP protein and its mRNA is observed in this tissue. There was not such a good correlation in the protein and mRNA level in other tissues, such as spleen, liver, stomach, and lung.
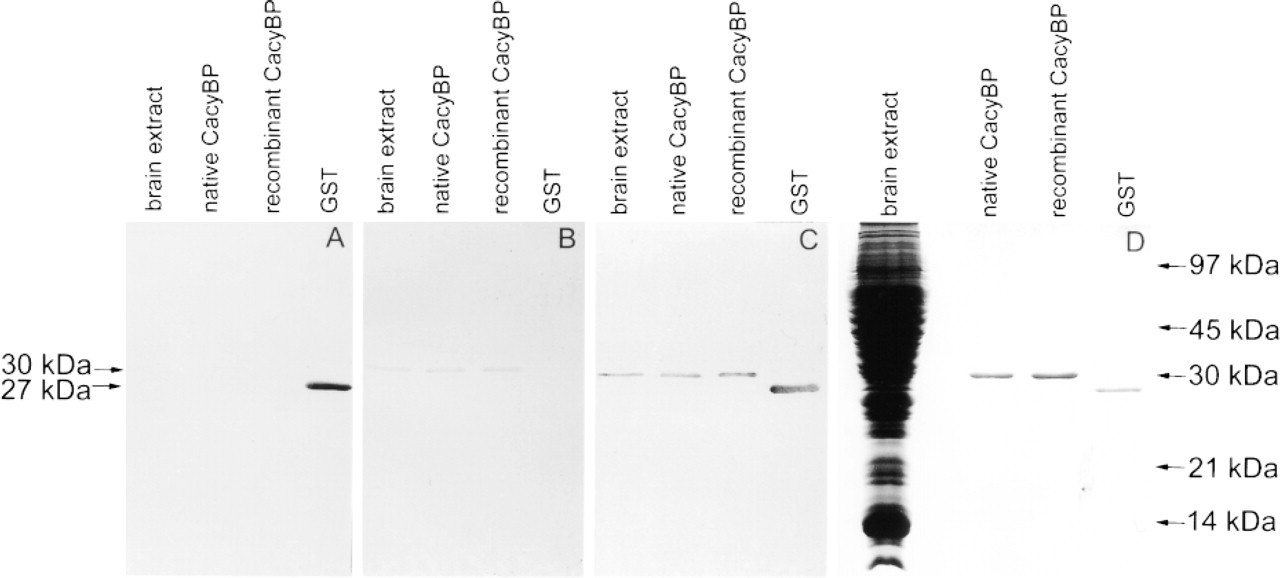
Specificity of CacyBP antibodies estimated by Western blotting (
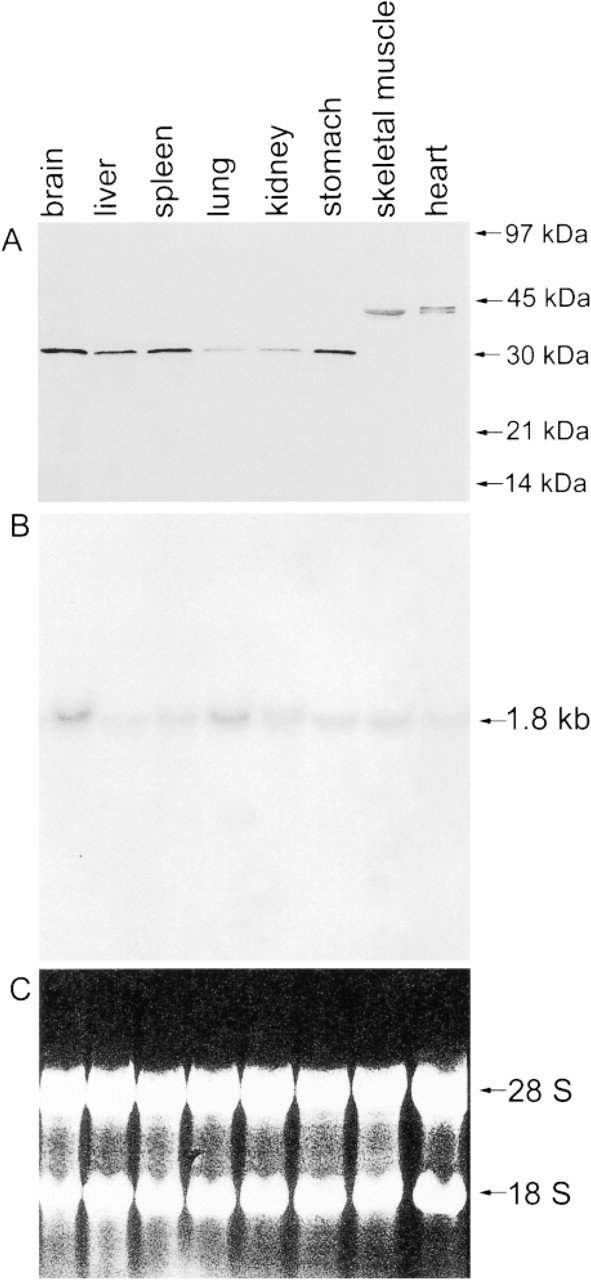
Distribution of CacyBP in various rat tissues. (
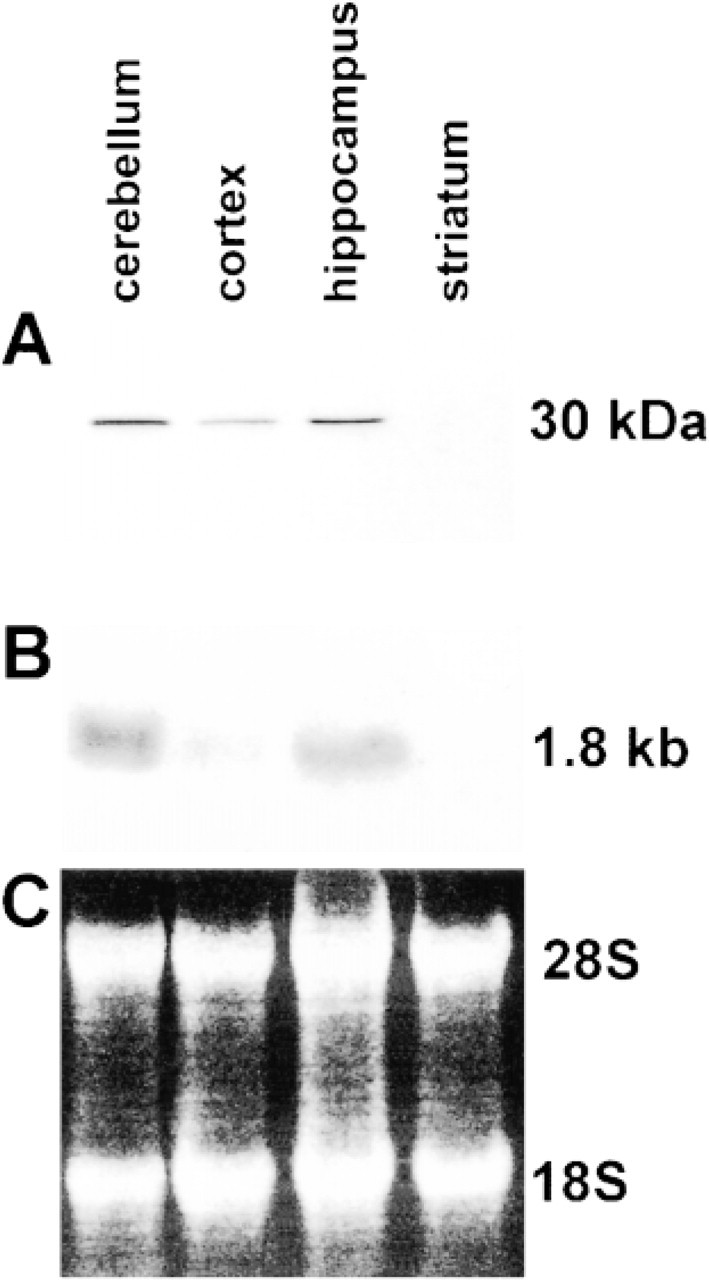
Level of CacyBP in different parts of the brain. (
CacyBP Level in Different Parts of Rat Brain
Because CacyBP appears to be expressed predominantly in the brain, we employed Western blotting to analyze its level in different parts of the brain. A nitrocellulose blot containing equal amounts of proteins derived from the soluble fraction of cerebellum, hippocampus, cortex, and striatum was incubated with immune serum containing specific polyclonal antibodies. As shown in Figure 3A, a high level of CacyBP was found in the cerebellum and hippocampus and a low level in the cortex. No positive reaction was seen in the striatum. Similar results were obtained by Northern blotting, confirming the different expression level of CacyBP in various brain regions (Figure 3B).
Estimation of CacyBP Concentration in Rat Brain and Cerebellum
To estimate the concentration of CacyBP, the proteins from the supernatant fraction derived from total brain and cerebellum were blotted from SDS-PAGE onto nitrocellulose and CacyBP was detected with an immune serum containing specific polyclonal antibodies. Different amounts (3 μg, 15 μg, and 75 μg) of total brain and cerebellum protein were loaded onto the gel. Densitometric analysis of bands from the Western blot containing a known amount of CacyBP were used to obtain a calibration curve from which the amount of CacyBP was calculated (not shown). It was found that the CacyBP concentrations in the soluble fractions of total brain and cerebellum were 0.17 and 0.34 ng/μg protein, respectively.
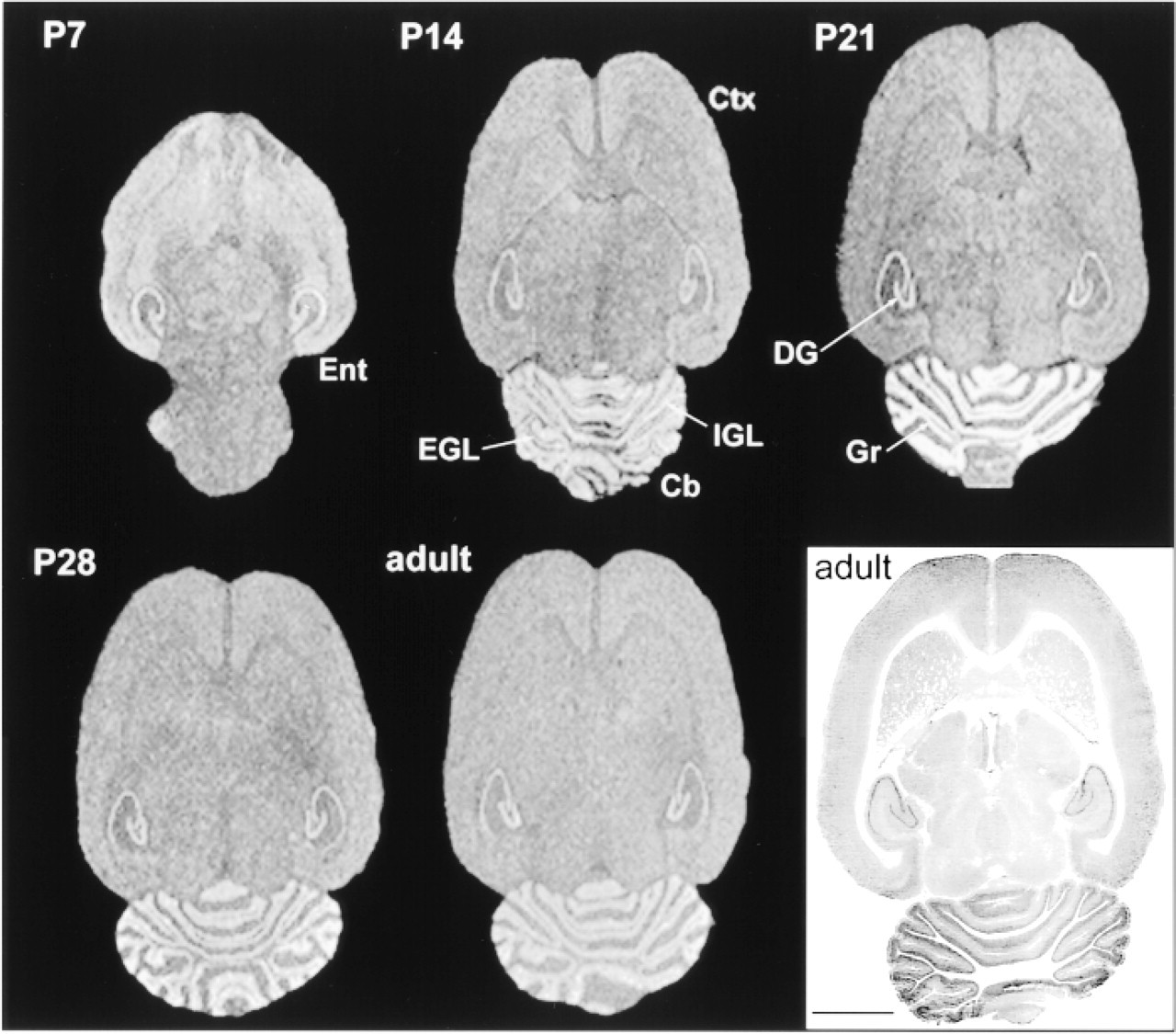
Overall distribution of CacyBP in developing rat brain. Horizontal sections were subjected to in situ hybridization and exposed to X-ray film. (
Cell-specific Expression of CacyBP in Rat Brain
To identify the CacyBP-positive cells, we employed both in situ hybridization and immunohistochemistry. Cryostat sections of adult rat brain, as well as brains at different stages of development, were hybridized with a α35S-labeled cDNA probe. As shown in Figure 4, a high level of expression was observed in the entorhinal cortex (Ent) at P7 (postnatal day 7). At P14 and later until adulthood, no obvious differences in the mRNA level in cortical regions could be seen. The expression was quite uniform throughout the thickness of the cortex (Ctx). No laminar pattern of CacyBP mRNA expression could be revealed. In cerebellum (Cb) at P14, a high level of mRNA was observed in the internal granule cell layer (IGL) and disappeared in the external granule cell layer (EGL). At P21, a marked increase of CacyBP mRNA expression was seen in the granule cells (Gr). This increase was transient; expression declined and reached an adult level as early as P28. In hippocampus, the specific mRNA was present in all fields of Ammon's horn and dentate gyrus (DG). No clear developmental changes of either pattern or intensity of labeling were observed. The immunoreactivity pattern was similar to that obtained with in situ hybridization. Slight differences were observed in the hippocampus, in which the most intense staining was seen in the CA3 region, whereas in situ hybridization revealed the highest level of CacyBP mRNA in the dentate gyrus.
The high-magnification micrographs demonstrated that, in the dentate gyrus, CacyBP-immunopositive cells were neurons located in the granule cell layer (Gr). Many immunopositive cells were also found in the polymorphic cell layer (Po) (Figure 5A). In the CA1 field of the hippocampus, virtually all pyramidal layer neurons (Py) were immunostained (Figure 5C). Some cells in the molecular layer (M) were also positive (Figure 5C, arrow). Pyramidal neurons (Py) of the CA3 region were very intensely stained (Figure 5D), but cells located outside the pyramidal layer were also clearly positive (Figure 5D, arrow).
In the cerebellum, the most intense immunostaining was found in Purkinje cells (Pu) (Figure 5B). A lower level of immunoreactivity was observed in granule cells (Gr). In the neocortex, no specific layer was distinguished. Positive cells were both pyramidal and nonpyramidal in shape and were uniformly distributed (Figure 5E). In all experiments, control sections were incubated with preimmune serum or with PBS and no positive reaction was observed (not shown).
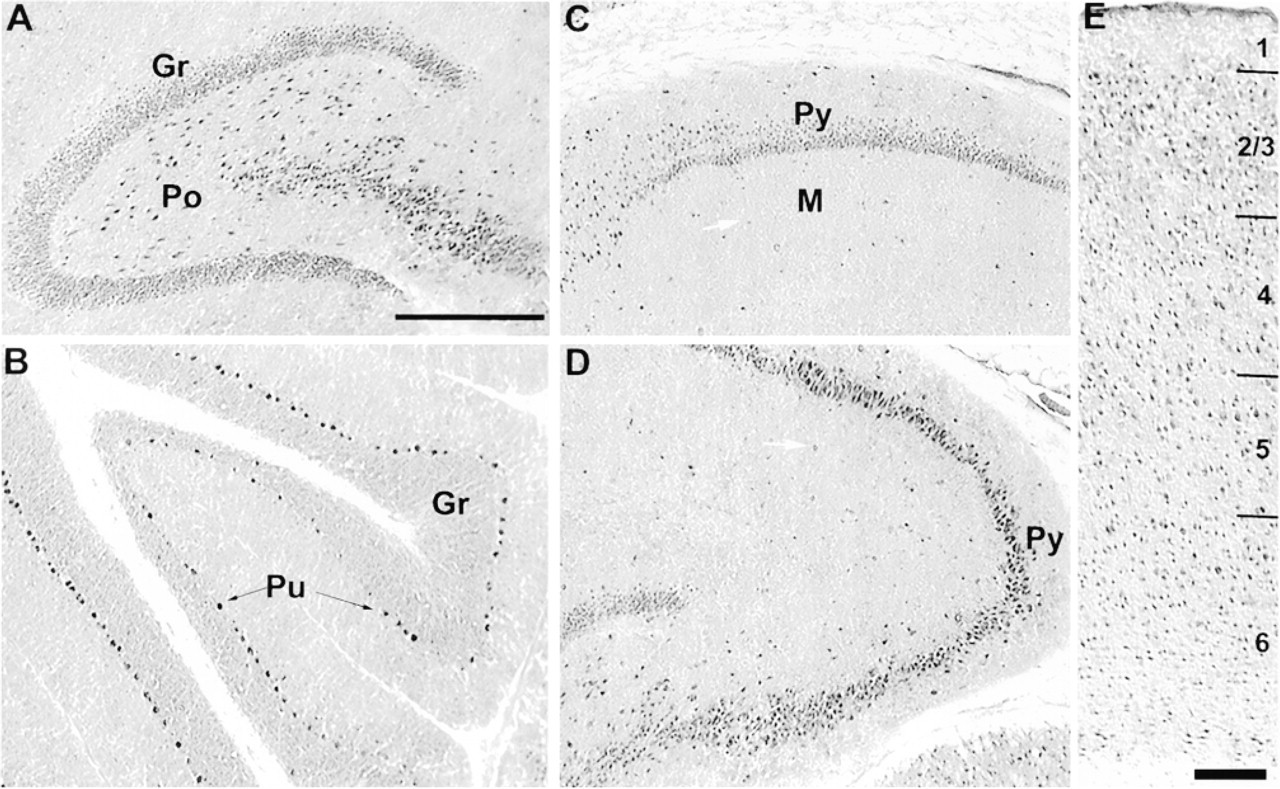
Light micrographs of CacyBP-immunostained horizontal sections of rat brain. (
Discussion
In this study we investigated the expression pattern of CacyBP, recently cloned and showed to bind to calcyclin in a physiological range of calcium concentration (Filipek and Kuźnicki 1998). For the analyses of CacyBP protein expression, we employed an immune serum raised against the fusion protein. The specificity of the serum was documented by the fact that Western blotting on various rat tissues produced a single protein band of the expected molecular weight of 30,000. Additional control experiments performed on rat brain extract using preabsorbed or preimmune serum confirmed the high quality of this serum. Although this serum was found to be specific and in a large number of tissues reacted with a 30,000 protein band, in heart and skeletal muscle it recognized a protein with a molecular weight of approximately 38,000. This is probably a striated muscle isoform of CacyBP or an SDS-stable complex of 30,000 CacyBP and another ligand. The latter is suggested by the observation that mRNAs of all tissues, including those from skeletal and cardiac muscle, are of the same size.
Northern analysis on the rat brain was performed using mouse cDNA. Although the sequence homology between mouse and rat CacyBP clone is not known (there is no rat sequence of CacyBP in GenBank), there is a high sequence homology between mouse and a human CacyBP clone (accession number AAC 21458). There are also a few sequences found in the EST database that come from different sources and which are highly homologous to the mouse CacyBP clone. Therefore, it was justified to check the possibility of using the mouse cDNA clone to study the expression in rat tissues. This proved to work, because the mouse clone stained only mRNA of CacyBP in the total RNA from rat tissues. Both Western and Northern blotting analysis revealed that the rat brain contains an especially high level of CacyBP, and in this tissue there was the best correlation between protein and mRNA level. Within this tissue, the cerebellum was found to be particularly abundant in CacyBP protein, the level of which was established by densitometric analysis to be in the range reported for brain-specific proteins such as calretinin (Winsky and Kuźnicki 1995).
Using immunohistochemistry and in situ hybridization, we have established that CacyBP is expressed predominantly in neuronal cell bodies, as indicated by high levels noted for Purkinje cells of the cerebellum and for pyramidal and granule cell layers of the hippocampus. In addition, CacyBP-immunopositive cell bodies were also observed scattered throughout the cerebellar cortex, except for Layer I. These data are in agreement with our recent findings showing that CacyBP is highly expressed in the mouse neuroblastoma cell line NB2a but not in glioma C6 cells (not shown).
The pattern of CacyBP expression in the rat brain established by Western and Northern blotting does not correlate well with that of calcyclin (Filipek et al. 1993). Some calcyclin immunoreactivity has been observed in hippocampal pyramidal neurons and granule cells but not in Purkinje neurons of the cerebellum. Furthermore, a recent careful analysis of calcyclin immunoreactivity described by Yamashita et al. (1999) revealed that the protein is present in both neurons and glia of various brain regions, with, however, the notable exception of most of the pyramidal and granule cell layers of the hippocampus. Thus, a clear discrepancy between CacyBP and calcyclin localization in certain regions of the brain and cell types is observed. Furthermore, the tissue-specific distribution of calcyclin does not correlate with that of CacyBP (Kuźnicki et al. 1989a; Filipek and Wojda 1996; Filipek and Kuźnicki 1998). It was also found that there is no quantitative correlation between calcyclin and CacyBP in some tissues and cell lines. The brain is an example of the tissue and mouse neuroblastoma NB2a of the cell line, in which there is much more CacyBP than calcyclin protein. Ehrlich ascites tumor cells are a cell line that expresses high levels of both calcyclin and CacyBP (not shown). It is therefore likely that calcyclin is not the only calcium binding protein that interacts with CacyBP. In Ehrlich ascites tumor cells, calcyclin might be the physiological target of CacyBP, but in other cells and tissues, such as brain, another calcium binding protein may interact with CacyBP and influence its function.
We have also investigated the developmental pattern of CacyBP mRNA expression in the rat brain. The most striking feature was a peak of expression at P21 in the cerebellum. This is interesting because in the same region of the brain several calcium binding proteins change their pattern of expression postnatally (Endo et al. 1985; Cimino et al. 1990; Iacopino et al. 1990; Milosevic and Zecevic 1998). Furthermore, it has also been repeatedly stressed that the regulation of intracellular calcium concentrations may play a major role in maturation of the central nervous system. If indeed the biological function of CacyBP involves binding to calcyclin or to other calcium binding proteins, an understanding of its biological function can be sought in the context of a role in calcium signaling.
Footnotes
Acknowledgements
Supported by grant 6P04 A 05415 from the State Committee on Scientific Research and by statutory funds provided to the Nencki Institute.
We thank Dr P. Groves for critical reading of the manuscript.
