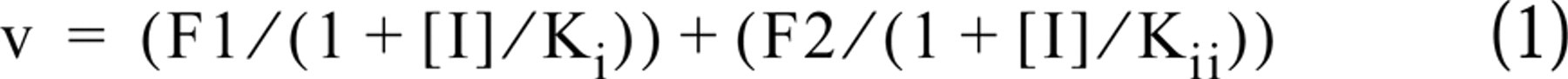Abstract
In addition to the three isoforms of the catalytic subunit of the Na,K-ATPase originally identified (α1, α2, and α3), a fourth α polypeptide (α4) has recently been found in mammalian cells. This novel α-subunit of the Na,K-ATPase is selectively expressed in male gonadal tissues. In the testes, α4 is functionally active and comprises approximately half of the Na,K-ATPase activity of the organ. At present, the pattern of expression of the α4 polypeptide within the cells of the male gonad is unknown. By in situ hybridization, immunocytochemistry, and the ouabain inhibition profile of Na,K-ATPase activity, we show that the α4-subunit is expressed in the germ cells of rat testes. The highest amounts of the isoform are found in spermatozoa, where it constitutes two thirds of the Na,K-ATPase activity of the gametes. The other Na pump present in the cells is the ubiquitously expressed α1 polypeptide. The characteristic localization of α4 in the gonad is further supported by the drastic reduction of the polypeptide in mice that are infertile as a consequence of arrest in maturation of the germ cells. In addition, GC-1spg cells, a murine cell line derived from testis spermatogonia, also contain the Na,K-ATPase α4 polypeptide. However, the level of expression of the isoform in these cells is much lower than in the spermatozoa, a fact that may depend on the limited ability of the GC-1spg cells to differentiate in vitro. The particular expression of the Na,K-ATPase α4 isoform we encounter and the specific enzymatic properties of the polypeptide suggests its importance for ionic homeostasis of the germ cells of the testes. (J Histochem Cytochem 48:1023–1032, 2000)
The Na,K-ATPase or Na pump exists as multiple isozymes consisting of different α- and β-subunits (reviewed in Sweadner 1989; Lingrel and Kuntzweiler 1994; Levenson 1994; Blanco and Mercer 1998). At present, four distinct α-subunits (α1, α2, α3, and α4) and three different β-subunits (β1, β2, and β3) have been identified in vertebrates (Schneider et al. 1985; Shull et al. 1986; Sverdlov et al. 1987; Martin-Vasallo et al. 1989; Gloor et al. 1990; Malik et al. 1996; Besirli et al. 1997). The highly regulated pattern of expression of the α and β polypeptides and their ability to associate in different pairs provides each cell with a particular array of Na,K-ATPase isozymes (reviewed in Lingrel et al. 1990; Lingrel and Kuntzweiler 1994; Ewart and Klip 1995; Blanco and Mercer 1998). The structural heterogeneity and functional diversity of the various αβ complexes (Jewell and Lingrel 1991; Munzer et al. 1994; O'Brien et al. 1994; Blanco et al. 1995a,b; Therien et al. 1996; Crambert et al. 2000) suggest that, rather than being redundant, the Na pump isozymes perform specific physiological roles (reviewed in Blanco and Mercer 1998). The least-characterized Na,K-ATPase isozymes are those composed of the α4-subunit. Recently, studies of the function of this polypeptide have confirmed that it can function as a catalytic subunit of the Na,K-ATPase. Thus, the α4 isoform is able to bind [3H]-ouabain, become phosphorylated from ATP, hydrolyze ATP, and transport 86Rb, in a manner typical for a Na,K-ATPase (Blanco et al. 1999; Woo et al. 1999), although clearly an Na pump α isoform, α4, exhibits enzymatic characteristics that distinguish it from the other α isoforms. For example, α4 is very sensitive to ouabain, has a high apparent affinity for Na+, a low affinity for K+, and an intermediate affinity for ATP (Blanco et al. 1999). Another property unique to the α4 polypeptide is its limited tissue localization. The isoform appears to be selectively expressed in the testes and epididymis (Shamraj and Lingrel 1994; Underhill et al. 1999). The Na,K-ATPase from the testes consists exclusively of the α1 and α4 isoforms (Shamraj and Lingrel 1994; Blanco et al. 1999). In the gonadal tissues, the Na,K-ATPase has been identified in the epithelial cells of the seminiferous and epididymal tubules and in the germ cells (Uesugi and Yamazoe 1966; Quinn and White 1967; McGrady 1979; Ashraf et al. 1982; Byers and Graham 1990; Kenneth and Hess 1992). In the epithelial cells, the enzyme helps create the appropriate medium inside the tubule lumen for normal development of the spermatozoa (Byers and Graham 1990). In the germ cells, Na,K-ATPase activity maintains the cell membrane potential and motility of the spermatozoa (Nelson and McGrady 1981). At present, the functional relevance of the α4 isoform in the physiology of the testes is unknown. An important goal in understanding the role of this isoform is to determine the distribution of the polypeptide within the cells of the gonad. On the basis of in situ hybridization analysis, immunocytochemistry, and the ouabain inhibition profiles of the Na,K-ATPase activity, we present evidence that, in the testes, the α4 polypeptide is expressed in the germ cells and predominantly in spermatozoa.
Materials and Methods
Tissue and Cells
Testes and sperm cells or the germ cell line GC-1spg were used in this study. Testes were obtained from adult Sprague-Dawley rats weighing 200–240 g or from adult mice. In the case of mice, normal and transgenic mice deficient in the translation factor Egr4 (Tourtellotte et al. 1999) were used. Spermatozoa were collected from rat epididymis by standard techniques (Vermouth et al. 1986). Briefly, whole epididymis was dissected, minced, and suspended in 100 mM KCl, 12 mM KH2PO4, 3 mM MgCl2, 0.4 mM EDTA, and 20 mM Tris, pH 7.4. After stirring for 20 min, the suspension was filtered through a fine mesh cloth and centrifuged at 800 × g for 10 min. The sperm pellet was resuspended and recentrifuged as before. Finally, the sample was resuspended in 320 mM sucrose, 25 mM imidazole (pH 7.4), and 0.1 mM EGTA. GC-1spg cells, derived from germ cells of mouse testis, were purchased from the American Type Tissue Culture Collection (ATCC; Rockville, MD). These cells were grown in Dulbecco's modified Eagle's medium (DMEM) supplemented with 1 mM sodium pyruvate, 10% fetal bovine serum, penicillin (100 U/ml), streptomycin (100 mg/ml), and Fungizone (0.25 mg/ml).
Tissue and Cell Preparations
Membrane fractions were prepared from adult rat and mouse testes according to the protocol described previously (Blanco et al. 1999). Briefly, samples were homogenized on ice in 320 mM sucrose, 25 mM imidazole (pH 7.4), and 0.1 mM EGTA, using a glass-glass homogenizer. The lysate was then centrifuged at 1000 × g for 10 min. The pellet was re- suspended, homogenized again, and recentrifuged at 1000 × g for 10 min. The supernatants from the first and second centrifugations were combined and centrifuged for an additional 30 min at 30,000 × g. The final pellet was resuspended in the original homogenization buffer. For spermatozoa and GC-1spg cells, crude homogenates were prepared by disrupting the cells with a glass-glass homogenizer. Both the tissue membranes and cell homogenates were used for determination of Na,K-ATPase activity with various concentrations of ouabain.
RNA Blotting Analysis
RNA was isolated from tissue and cells using RNAzol according to the suppliers protocol (Tel-Test; Friendswood, TX). RNA content was quantified spectrophotometrically. Equal amounts of RNA (15 μg) were separated by electrophoresis in a formaldehyde gel and transferred overnight by capillary action to nitrocellulose (Micron Separations; Westboro, MA). The blots were UV-crosslinked and prehybridized at 65C according to Sambrook et al. (1989). [32P]-dCTP-labeled probes for the Na,K-ATPase α isoforms were made from N-terminal fragments of the corresponding cDNAs, using the Rediprime random primer labeling kit from Amersham (Poole, UK). For the α1 and α3 isoforms, a cDNA segment corresponding to the 5′ end of each isoform to the AflII restriction site was used as template. For α2, a region of the isoform spanning from the 5′ end to the NheI site was used. For α4, the segment between base pairs 1314 and 1592 was utilized. Blots were washed in 0.1 × SSC (15 mM NaCl, 0.15 mM sodium citrate, pH 7.0), containing 1% SDS at 65C and exposed for autoradiography.
In Situ Hybridization
Localization of the Na,K-ATPase α4 isoform RNA was performed on 10-μm-thick sections of rat testes that had been fixed in 2% paraformaldehyde and embedded in paraffin. In situ hybridization was performed using the RNA Colour kit from Amersham. The fluorescein-11-UTP-labeled probes for the α1 and α4 isoforms were made using as template the full-length cDNA for the α1-subunit or a 301-bp cDNA fragment (bases 1314–1592 of the coding region of α4) for the α4-subunit. Transcription from both α isoform cDNAs, subcloned into pGEM-T Easy vector (Promega; Madison, WI) was performed using SP6 RNA polymerase. Sense probes using as template the same α1 and α4 cDNAs were made and used as control. Hybridization with the probes was performed for 2 hr at 55C. Samples were washed twice for 10 min each with 0.1% (v/v) of sodium dodecyl sulfate (SDS) in SSC at room temperature (RT), followed by two other washes at 55C for 10 min with 0.1% (v/v) SDS in SSC. The labeled probes were detected using an anti-fluorescein antibody conjugated to alkaline phosphatase at a dilution of 1:100.
Immunocytochemistry
Paraffin sections (10 μm) of rat testes, were treated with xylene and ethanol to remove the paraffin. After hydration, permeabilization of the sections was achieved by a 15-min incubation at 25C with 0.3% Triton X100 in HBS (25 mM Hepes, pH 7.4, 150 mM NaCl, and 1 mM EGTA). The tissue was then blocked for 2 hr at RT in 0.2% BSA, 5% normal goat serum (NGS) in HBS. The primary antibody in HBS containing 0.1% BSA, 2% NGS was added and allowed to attach overnight at 4C. After three 5-min washes in HBS, the secondary antibody in HBS containing 0.1% BSA, 2% NGS was added. After 1 hr, sections were washed three times for 5 min each with HBS and mounted. The α1 polypeptide was detected with a 1:100 dilution of an antiserum that recognizes a N-terminal peptide (DKYEPAAVSEHGD) of the isoform (Blanco et al. 1994). For the α4 isoform, a specific antiserum against a peptide (SEQKPRPTLRASNTNRQPK) corresponding to a sequence at the N-terminus of the α4 polypeptide was used at a dilution of 1:100 (Blanco et al. 1999). As secondary, an anti-rabbit antibody conjugated to fluorescein isothiocyanate (FITC) was used. Images were obtained using a Zeiss Axioskop microscope equipped with a Nikon UFX-II camera attachment.
Biochemical Assays
Protein assays were performed using the bicinchoninic acid/copper sulfate solution as described by the supplier (Pierce Chemical; Rockford, IL) after lysis of the cells in 1% 3-[(3-cholamidopropyl) dimethylammonio]-1-propanesulfonate (CHAPS). Na,K-ATPase activity was assayed through determination of the initial rate of release of 32Pi from γ[32P]-ATP as described previously (Blanco et al. 1995a). Membrane fractions from rat and mouse testes containing a total of 5 μg protein were assayed, whereas for sperm cell and GC-1spg cell homogenates, 10 μg and 20 μg total protein was used, respectively. Na,K-ATPase activity was measured in a final volume of 0.25 ml of medium containing 120 mM NaCl, 30 mM KCl, 3 mM MgCl2, 0.2 mM EGTA, 2.5 mM sodium azide, 30 mM Tris-HCl (pH 7.4), 3 mM cold ATP, 0.2 μCi γ[32P]-ATP ± different concentrations of ouabain. The reaction was started by the addition of ATP and incubated at 37C for 30 min. Radioactivity of 170 μl of the isobutanol-phosphomolybdate phase was measured by liquid scintillation counting. The hydrolysis of ATP in all assays was linear over the time course of the reaction. The total hydrolysis of ATP among the various preparations ranged from 30 to 50 μmoles of Pi released/mg protein/hr. Specific Na,K-ATPase activity was approximately 3 and 7 μmoles of Pi released/mg protein/hr for the Na,K-ATPase from rat and mouse testes, respectively. The germ cell line in culture and the spermatozoa showed lower specific activity, with values of approximately 1 and 2 μmoles of Pi released/mg protein/hr, respectively. Na,K-ATPase activity was determined as the hydrolysis of ATP that was dependent on Na+ and K+ and sensitive to 1 mM ouabain. Thus, total Na,K-ATPase activity was calculated by subtracting the ATPase activity obtained in the presence of 1 mM ouabain from that measured in its absence. For the ouabain dose-response curves, activity values were standardized to compare the relative contribution of each Na,K-ATPase isozyme to the total activity of each sample. For this, data were expressed as a percentage of the total Na,K-ATPase sensitive to 1 mM ouabain.
Data Analysis
Curve fitting of the experimental data was performed using a Marquardt least-squares nonlinear regression computing program (Sigma Plot; Jandel Scientific, San Rafael, CA). Dose-response relations for the ouabain inhibition of Na,K-ATPase activity showed a heterogeneous population of enzyme that was best fitted by applying an equation that assumed the existence of two (Equation 1) populations of Na,K-ATPase isozymes with different affinities for ouabain:
where v is the Na,K-ATPase activity corresponding to a certain concentration of the inhibitor ouabain [I], expressed as a fraction of activity in the absence of ouabain. F1 and F2 are the fractional amounts of each Na pump isozyme, whereas Ki and Kii represent the concentration of ouabain that gives the half-maximal inhibition to each of the Na,K-ATPases present in the sample. The best fit of the data to a two, rather than a one or multiple enzyme population model was confirmed by applying an F test as reported previously (Blanco et al. 1995b). This indicated that only two Na,K-ATPase isozymes, with Kis compatible with the existence of α1 and α4, were present in the samples.
Results
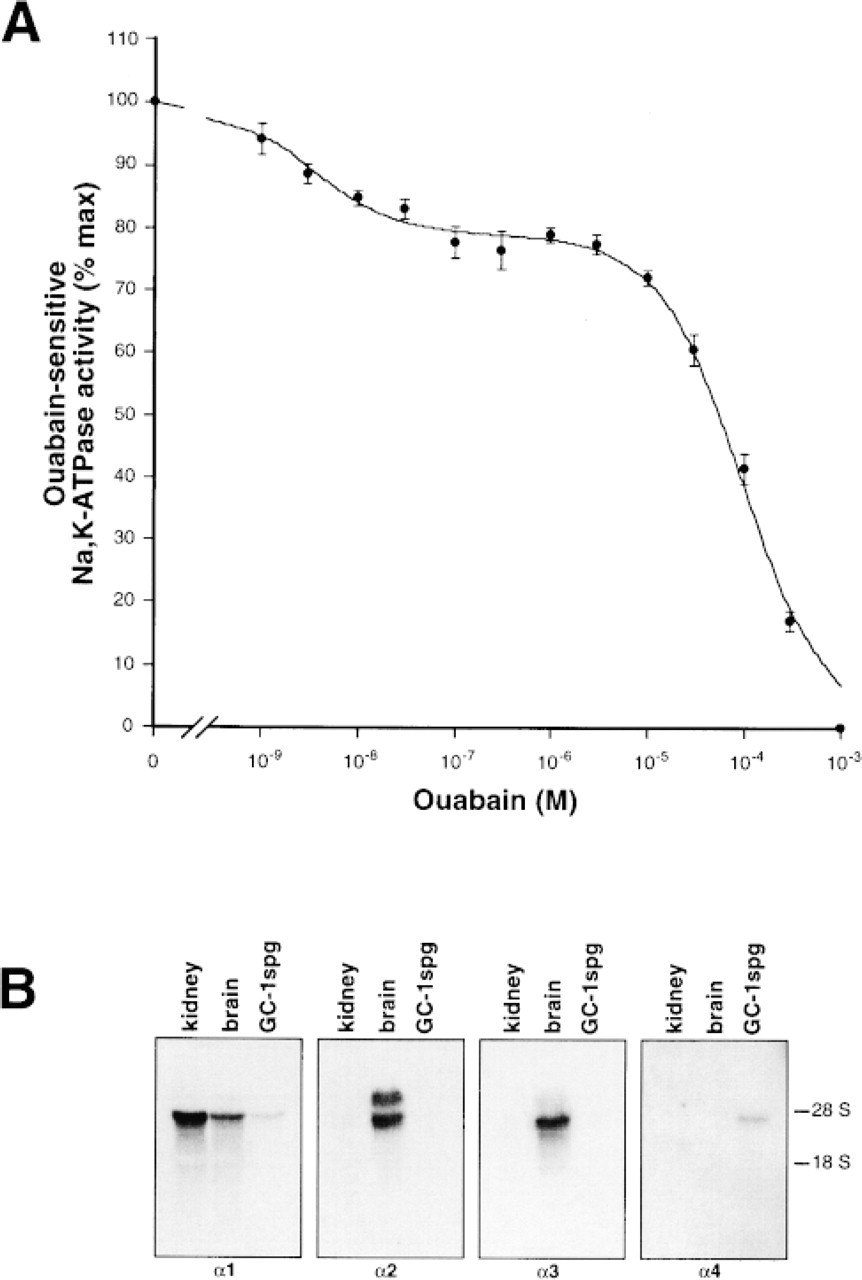
In the testes, the expression of the α-subunit of the Na,K-ATPase has been reported to be limited to the α1 and α4 isoforms (Shamraj and Lingrel 1994; Blanco and Mercer 1998). We confirmed this observation by Northern blotting analysis of total RNA from rat tissue. To ensure the specific identification of the α-sub-units, 32P-labeled probes were made using as template isoform-specific cDNA segments. In addition, hybridization with the probes at 65C and stringent washing conditions using 1% SDS and 0.1% SSC were applied. As shown in Figure 1, transcripts for only the α 1- and α4-subunits of the Na,K-ATPase were detected in the testes. As control, total RNA from rat kidney, which contains only the α1 isoform, and brain, which expresses the α1, α2, and α3 isoforms, were used.
To gain insight into the distribution of the Na,K-ATPase α1 and α4 isoforms in the testes, we determined the localization of the corresponding mRNAs using in situ hybridization. Fluorescein–11-UTP-labeled RNA probes, followed by an anti-fluorescein alkaline phosphatase-conjugated antibody, were used to detect the α1 and α4 transcripts. Specific labeling of the isoforms was obtained by hybridization with the probes at 55C and stringent washes. Figure 2 shows that the α1 mRNA was found in the seminiferous tubules as well as at the interstitial cells of the gonad. In contrast, the message for α4 was localized to cells in the pseudostratified epithelium of the seminiferous tubules and mainly at the inner or luminal portions of these structures. The distribution of α4 mRNA is consistent with expression of the isoform in the germ cells. These cells also express the α1 transcript, and there appeared to be no cells that express solely the α4 mRNA. In contrast, cells at the interstitium, between the seminiferous tubules, contain only the RNA for the Na,K-ATPase α1 isoform.
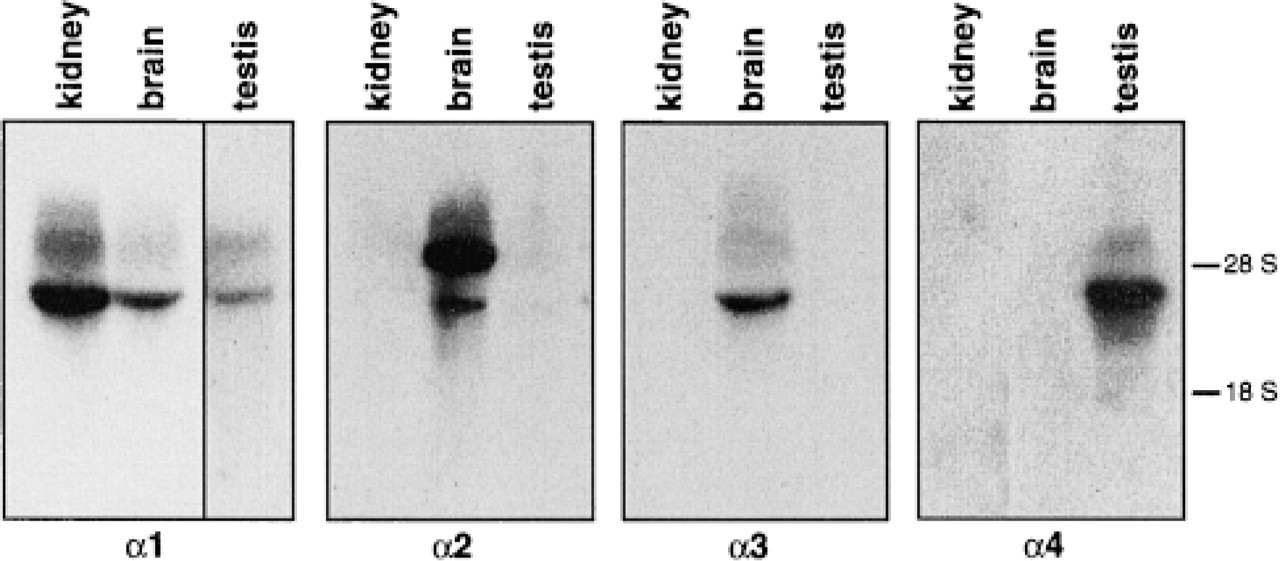
RNA hybridization analysis of Na,K-ATPase α isoform in rat testes. Equal amounts of total RNA (15 μg) were separated on formaldehyde gels and transferred to nitrocellulose. Isoform-specific rat cDNA probes for the indicated α isoforms were used to identify the corresponding transcripts. The specificity of the probes is shown by their hybridization profile with rat kidney and rat brain RNA. Autoradiography exposure time was 14 hr for all samples except for α1 in the testes, which was developed after 25 hr. No signal was seen in testes for the α2 and α3 isoforms even after 72 hr of exposure. The mobility of the 18 S and 28 S RNA has been included for reference.
To investigate the expression of α isoforms at the protein level, sections of rat testes were subjected to immunocytochemical analysis using anti-α1 or anti-α4 antibodies, followed by secondary antibodies conjugated to FITC. As shown in Figure 3, a different pattern of distribution of the Na,K-ATPase α1 and α4 polypeptides was found in the gonad. As expected from the expression of the corresponding RNAs, the α1 antiserum evenly labeled all the cells within and between the seminiferous tubules (Figures 3A and 3D), whereas staining with the α4 antiserum showed a more restricted distribution (Figures 3B and 3E). Immunoreactivity for α4 was primarily located on the adluminal side of the seminiferous tubules, with the highest intensity of label found in the spermatozoa. This suggests that the germ cells are the main source of the Na,K-ATPase α4 isoform. However, the polypeptide is not the only Na pump isoform expressed in these cells because α1 was also detected.
To further study the pattern of expression of the Na,K-ATPase α1 and α4 isoforms in the germ cells of the testis, we determined the profiles of inhibition of Na,K-ATPase activity by ouabain. This approach has been routinely used as a tool to detect Na,K-ATPase isoform content in a sample. The method has the advantage of measuring functionally active Na,K-ATPase molecules and, in addition, it allows a more reliable quantification of the isozymes present (reviewed in Blanco and Mercer 1998). As demonstrated previously in the rat, the α1 and α4 isoforms differ drastically in their affinities for ouabain. Compared to α1, the α4 isoform is over a thousand times more sensitive to the cardiotonic steroid (Blanco et al. 1999). This enzymatic difference, which is the greatest among the Na,K-ATPase isoforms, can readily be used to determine the relative contribution of each α isoform to the total Na,K-ATPase of the sample. The functional Na,K-ATPase isoform composition of germ cells was first determined in GC-1spg cells and in spermatozoa. GC-1spg cells are a nontumorigenic murine cell line derived from germ cells of the gonad that have been immortalized by simian virus 40 large tumor antigen transformation (Hoffmann et al. 1992). As shown in Figure 4A, the dose-response curve for the inhibition of Na,K-ATPase activity by ouabain in the GC-1spg cells was heterogeneous, indicating the presence of two Na,K-ATPase isozymes. The inhibition constants for each of the isoforms, as well as their relative proportions, are shown in Table 1. The Kis in the nanomolar and millimolar range corresponded well with those reported previously for the Na,K-ATPase α1 and α4 isoforms (Blanco et al. 1999). The pattern of expression of the Na,K-ATPase α isoforms in the GC-1spg cells was corroborated by hybridization analysis of total RNA (Figure 4B). As shown, these cells do not express the α2 and α3 isoforms of the enzyme. Therefore, in agreement with the previous results, germ cells express both the α1 and α4 isoforms of the Na,K-ATPase. In addition, the level of isoform activity was unequal, with the α1 isoform making a relatively larger contribution to the total Na,K-ATPase of these cells. Because the turnover number of activity (ATP hydrolysed per min) is similar among the isoforms, their different contribution to the Na,K-ATPase hydrolysis reflect differences in expression of the polypeptides (Blanco et al. 1995a,b, 1999).
Although the GC-1spg cells have all the characteristics of germ cells, they can differentiate only to a certain extent, reaching a stage between spermatogonia Type B and spermatocytes. To investigate the level of expression of the α4 isoform in fully differentiated germ cells, we studied the profile of ouabain inhibition of the Na,K-ATPase from spermatozoa obtained from rat epididymis. As shown in Figure 5, a biphasic dose-response curve was found for the interaction of the cardiotonic steroid with the Na,K-ATPase of sperm cells. The obtained Kis for the sensitive and resistant forms of the enzyme corresponded to those of α1 and α4, confirming the expression of both Na pump isoforms in these cells. The calculated Ki and fractional amounts for each isozyme are indicated in Table 1. As shown, α4 activity constitutes the main Na,K-ATPase of spermatozoa. This contrasts with the relative distribution of the isoforms in the cultured germ cells, in which α1 activity represents the predominant Na pump present.
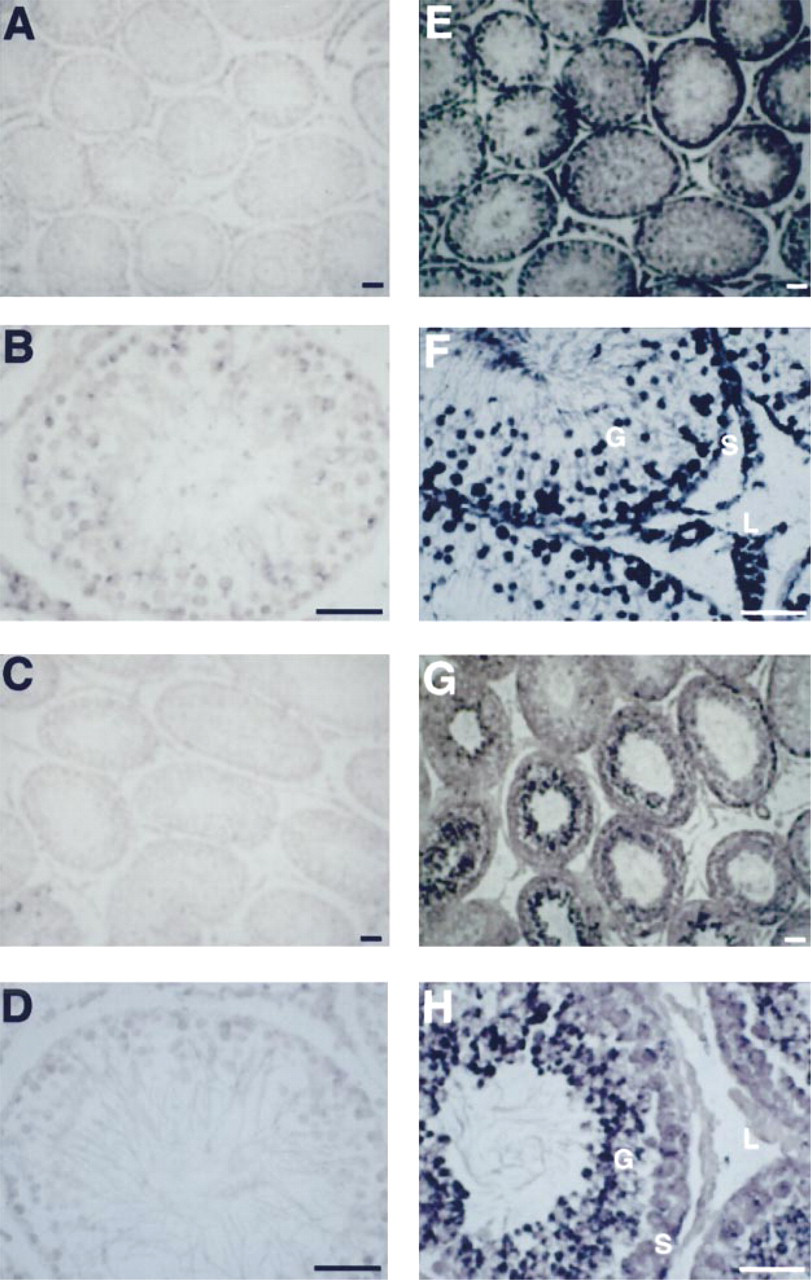
In situ hybridization of Na,K-ATPase α1 and α4 isoforms in rat testis. Testes from adult rats were fixed in 2% paraformaldehyde, embedded in paraffin, and sectioned (10 μm thick). Samples were hybridized with fluorescein-11-UTP RNA probes and detected using an antibody conjugated to alkaline phosphatase. As controls, the sense probes of the cDNA for the α1 (
To further confirm that the expression of the Na,K-ATPase α4 isoform originates mainly from the sperm cells, we characterized the Na,K-ATPase activity in testes from normal mice and mice made infertile through a deficiency in the production of the early growth response transcription factor, Egr4 (Tourtellotte et al. 1999). Egr4 is a member of the Egr zinc-finger transcription factors that regulate cell growth and cell differentiation (Crosby et al. 1991). Egr4 deficiency in male mice causes oligospermia due to an arrest in the maturation of germ cells. Because the Na,K-ATPase α4 isoform is mainly expressed in the mature germ cells, a reduction in the expression of the Na,K-ATPase α4 polypeptide should be expected to accompany the impaired production of spermatozoa in the transgenic mice. Figure 6 shows the ouabain inhibition profiles of the Na,K-ATPase from Egr4-deficient and normal mice testes. Reflecting the presence of both the α1 and α4 isoforms, testes from normal and Egr4-deficient mice exhibited low and high ouabainsensitive Na,K-ATPase activities. The calculated inhibition constants and relative quantity for the activity of each α isoform are summarized in Table 1. As shown, the normal mice gonad has approximately equal amounts of the α1 and α4 isoforms. This is in agreement with the results from rat testes (Blanco et al. 1999). In contrast, the Egr-4 deficient mice exhibit a significantly lower amount of the α4 isoform and, concomitantly, a proportional increase in the α1 isoform. Taken together, these results indicate that the germ cells express two functionally active isoforms of the catalytic subunit of the Na,K-ATPase, α1 and α4, and that the α4 polypeptide is the main functional form expressed in the meiotic cells of the gonad.
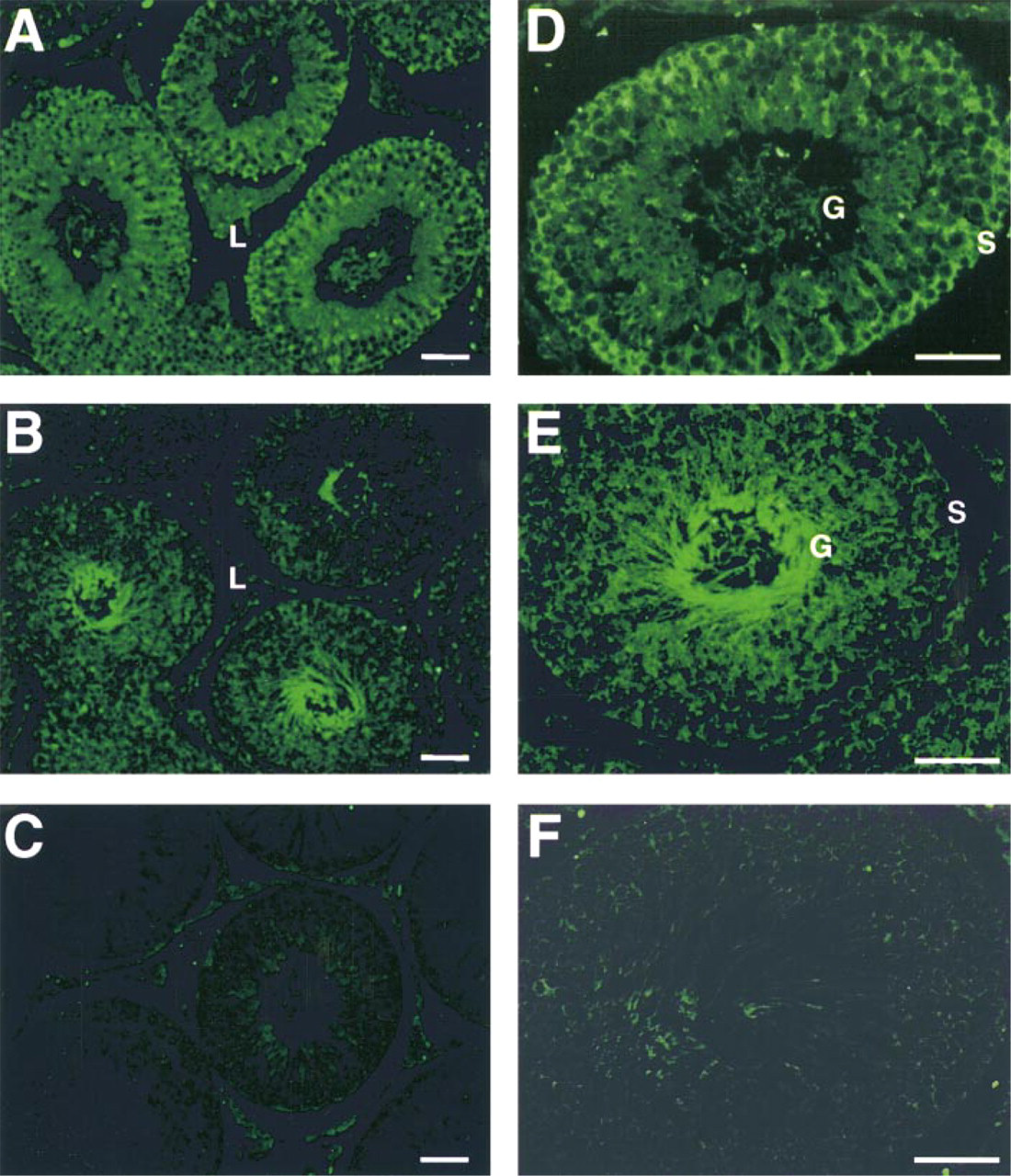
Localization of the Na,K-ATPase α1 and α4 polypeptides in rat testis. Testes were fixed in paraformaldehyde, embedded in paraffin, and sectioned. Immunocytochemistry was done as indicated in Materials and Methods using rabbit antisera that recognize the N-terminus of the α1 (anti-α1) or the α4 (anti-α4) isoform. A goat anti-rabbit antibody conjugated to FITC was used as the secondary antibody. (
Discussion
The α4 polypeptide is the Na,K-ATPase isoform that exhibits the most restricted pattern of expression, having been solely identified in male gonadal tissues (Shamraj and Lingrel 1994; Blanco et al. 1999; Underhill et al. 1999; Woo et al. 1999). In the testis, the expression of the α4 isoform is not exclusive because the α1 polypeptide is present as well (Blanco et al. 1999; Woo et al. 1999). In contrast, the α2 and α3 isoforms are absent (Shamraj and Lingrel 1994; Blanco et al. 1999). We confirmed this observation by hybridization analysis of total RNA from the gonad (Figure 1). Both the α1 and α4 isoforms are catalytically functional in the gonad. Although α1 and α4 contribute approximately equally to the Na,K-ATPase activity of the tissue, the isoforms have enzymatic properties that are different (Blanco et al. 1999). The particular tissue expression and kinetics of α4 suggest that it performs a specific function. An important goal in understanding the role of α4 is to determine its pattern of expression within the cells of the testes. In the present work we have analyzed this by in situ hybridization, immunocytochemistry, and by the profile of ouabain inhibition. Our results indicate that the α4 isoform is expressed predominantly in differentiated germ cells of the testes.
(
Although the polypeptide represents the majority of the Na,K-ATPase in the gametes, it is not the only α-subunit present because the cells also contain lower amounts of the ubiquitously expressed α1 isoform. The presence of the highly ouabain-sensitive and the ouabain-resistant ATPase activity typical of the α1 and α4 isoforms indicates that both polypeptides are functionally competent in the germ cells. This agrees with previous reports indicating that the meiotic cells of the testes are capable of hydrolyzing ATP in an Na+- and K+-dependent manner (Uesugi and Yamazoe 1966). The possibility that minor amounts of the α4 isoform are present in other cells of the testes can not be completely ruled out. For example, the anti-α4 antiserum appears to faintly label the Sertoli cells. However, the level of α4 expression in these cells is sparse. The finding by Muffly et al. (1985) that the Na,K-ATPase activity of rat Sertoli cells is resistant to ouabain agrees with the notion that these cells produce mainly the Na,K-ATPase α1 isoform. In addition, the analysis of the RNA and protein of the Na pump α4 isoform in the space between the seminiferous tubules suggests that the interstitial cells of the testes, such as Leydig cells, express predominantly the α1 isoform. Interestingly, we were unable to identify cells that uniquely express α4. Taken together, these results suggest that the main expression of the Na,K-ATPase α4 polypeptide occurs in the fully mature germ cells. However, the relative levels of activity of the isoform are different when the spermatozoa and the germ cell line GC-1spg are compared. In spermatozoa, the expression of α4 activity clearly predominates over that of the α1 isoform. The opposite occurs in GC-1spg cells, in which the activity of α1 prevails. This dissimilarity in isoform composition between the cultured and native cells may depend on the inability of the GC-1spg cells to fully express the Na,K-ATPase α4 isoform in vitro. This possibility seems unlikely because GC-1spg cells are able to normally express other testis-specific proteins, such as cytochrome ct and the lactate dehydrogenase C4 isozyme (Hoffmann et al. 1992). Alternatively, cell maturation may be involved in the regulation of expression of the isoforms. GC-1spg cells are known to differentiate only partially in culture. On the basis of their morphological and biochemical characteristics these cells have been shown to reach a stage between Type B spermatogonia and primary spermatocyte (Hoffmann et al. 1992). It is possible that upregulation of the expression of the α4 isoform is required at later stages of spermatozoa development. Supporting this notion are the results obtained from testes of Egr4-deficient mice. In these mice, maturation of the germ cells is almost completely blocked at the early-mid pachytene stage of spermatocyte development (Tourtellotte et al. 1999). Concomitant with this, the relative level of activity of the Na,K-ATPase α4 isoform is approximately a third of that found in normal testes. This difference, which suggests a developmental modulation of the expression of the Na pump α4-subunit, is not unique for the isoform, because changes in the levels of other Na,K-ATPase α isoforms have been reported during maturation (reviewed in Sweadner 1989; Lingrel et al. 1990).
Ki values and relative amounts of the rat Na,K-ATPase isozymes α1 and α4 expressed in GC-1 cells, rat spermatozoa, and mouse testes from normal and oligospermic mice a
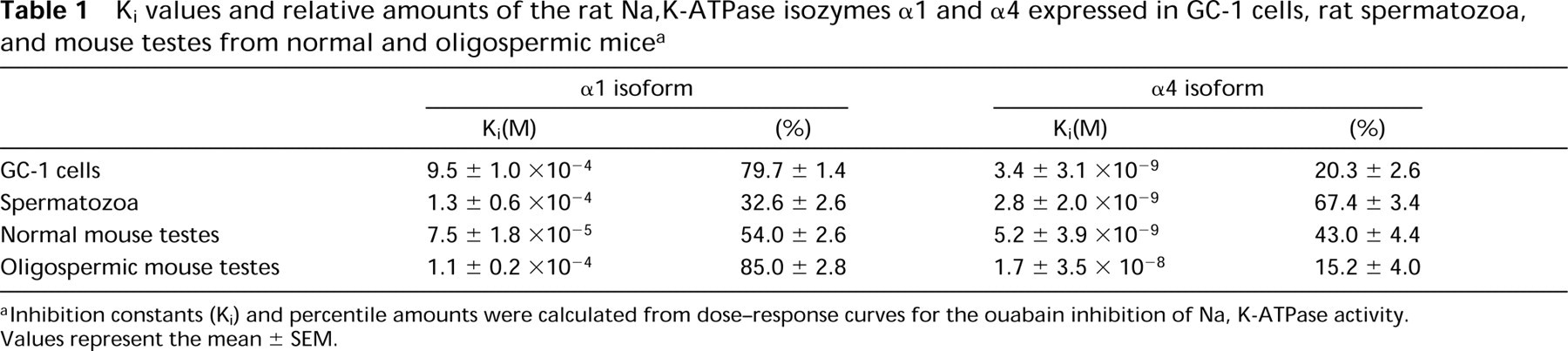
Inhibition constants (Ki) and percentile amounts were calculated from dose-response curves for the ouabain inhibition of Na, K-ATPase activity. Values represent the mean ± SEM.
The high level of expression of the α4 isoform of the Na,K-ATPase in the mature germ cells of testes is intriguing. This occurrence may be related to the particular ionic environment of the germ cells. The medium bathing the cells inside the seminiferous tubules has a concentration of K+ that is higher than that of blood (Sheth and Rao 1968; Polak and Daunter 1984; Rosecrans et al. 1987). Coincidentally, when compared to the other isoforms the affinity of the α4 isoform for K+ is low (Blanco et al. 1999). Therefore, it is possible that the Na,K-ATPase α4 isoform function has evolved to adapt to the medium in the tubules. In addition, the increase of α4 in spermatozoa compared to the immature germ cells may be related to the fact that as the cells differentiate they come in contact with the high-K+ fluid in the lumen of the tubules. Another important enzymatic property of the Na,K-ATPase α4 isoform is its high affinity for ouabain. Differences in ouabain sensitivity of the Na,K-ATPase isozymes have been proposed to have a role in the regulation of Na,K-ATPase activity (reviewed in Blanco and Mercer 1998). This is supported by the identification of endogenous ouabain-like compounds in mammalian tissues (Blaustein 1993, 1994; Kelly and Smith 1994; Doris 1996). Interestingly, endogenous digoxin-like immunoreactivity has been found in the seminal fluid of humans (Vadazs et al. 1983). Ouabain has been shown to elicit various responses when applied to spermatozoa (Nelson and McGrady 1981). For example, changes in the membrane potential and in motility have been reported after exposure of the male gametes to ouabain. Therefore, the Na,K-ATPase α4 isoform in spermatozoa may be a site of regulation by endogenous ouabain-like compounds. In conclusion, our results provide evidence of a differential cell distribution for the Na,K-ATPase α4 polypeptide in testis cells. This, in addition to the fact that the isoform exhibits unique enzymatic properties, suggests that the α4-subunit may be important in the physiology of the male gonad.
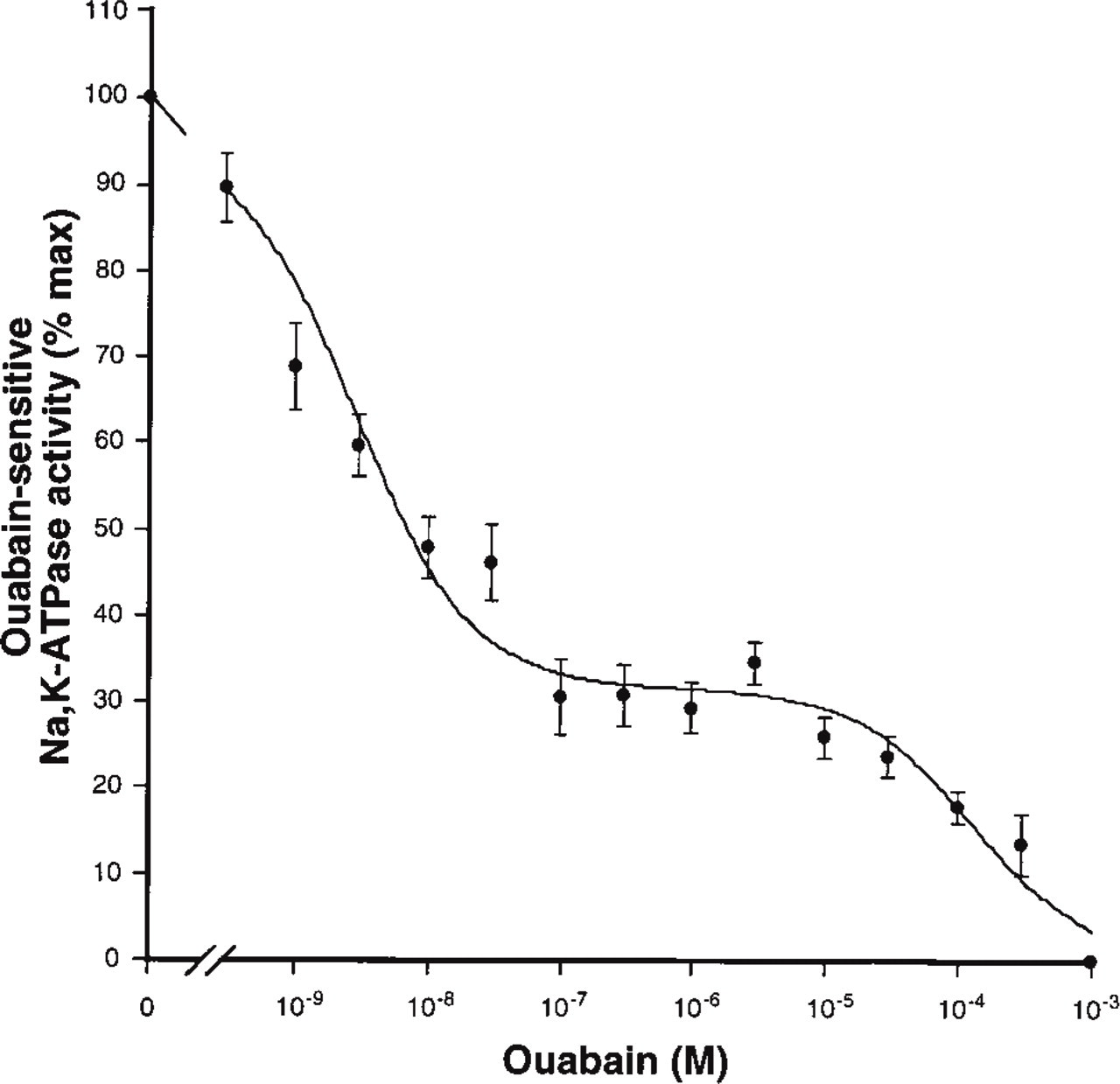
Ouabain inhibition profile of the Na,K-ATPase from spermatozoa. Na,K-ATPase activity of sperm cells from rat epididymis was determined as described in Materials and Methods. Values for specific activity were obtained as described in Figure 4. Data are expressed as percentage of Na,K-ATPase activity determined in the absence of the inhibitor ouabain. The best fit of the experimental data was obtained using an equation describing two enzyme populations with different affinities for the cardiotonic steroid. Each value is the mean and error bars represent SEM of four experiments performed in quadruplicate.
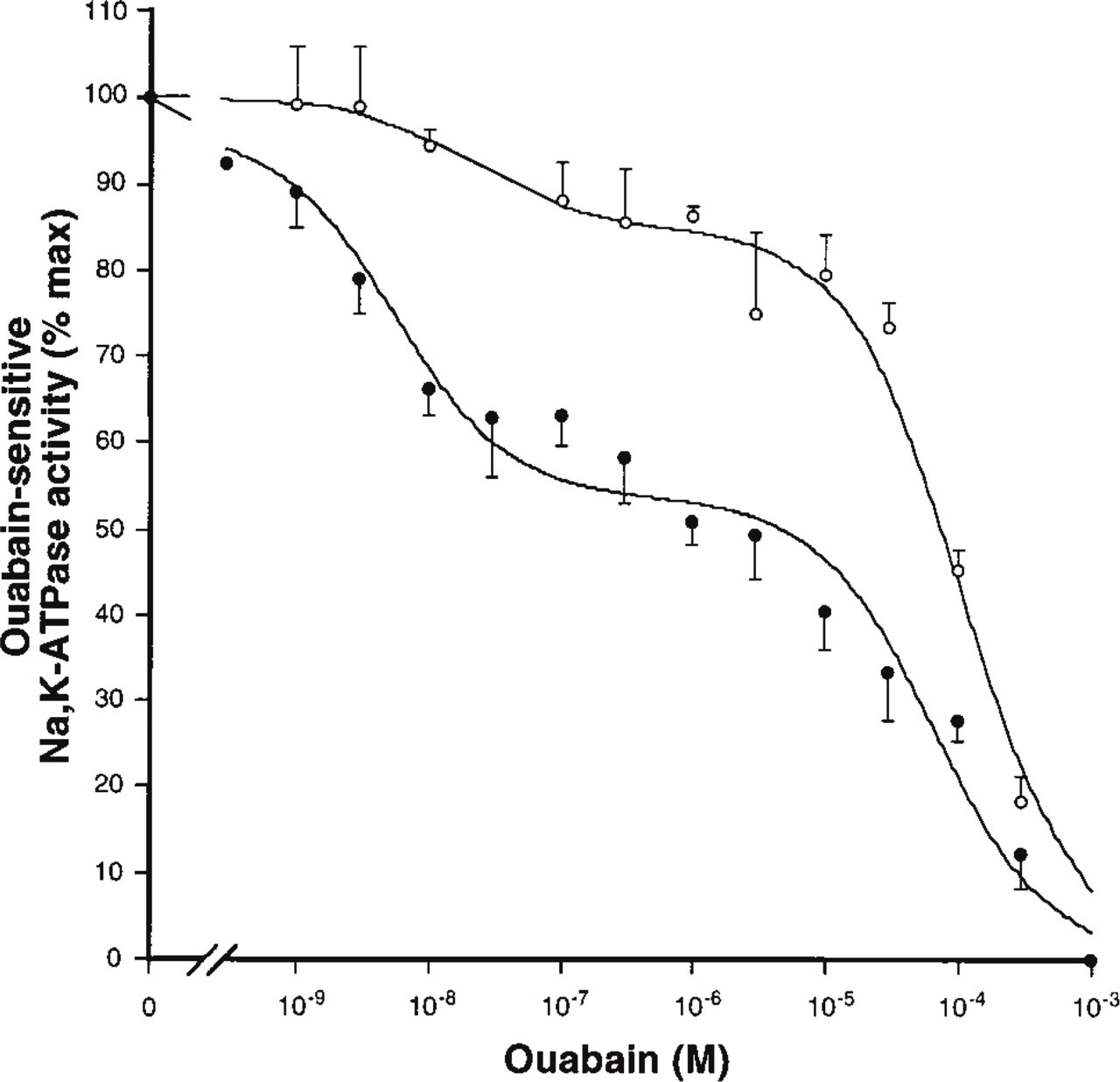
Ouabain inhibition of Na,K-ATPase from normal and Egr4-deficient mice. Na,K-ATPase activity was determined as explained in Materials and Methods. Values are expressed as percentage of the activity obtained in the absence of ouabain. The biphasic curves represent the best fit of the data and indicate the presence of the highly ouabain-sensitive α4 and ouabain-resistant α1 isoforms in normal (•) and oligospermic (○) mice. Each value is the mean and error bars represent SEM of three experiments performed in quadruplicate.
Footnotes
Acknowledgments
Supported by National Institutes of Health grants GM 39746 and DK 45181, and by American Heart Association grant 96012080.
We thank Dr Jeffrey Milbrandt for providing the mice deficient in the Egr-4 transcription factor.