Abstract
In previous studies, we have shown that the bile salt-dependent lipase (BSDL) associates with the Grp94 molecular chaperone, an association that appears to play essential roles in the folding of BSDL. More recently, combined biochemical and immunocytochemical investigations were carried out to show that the transport of BSDL occurs via an association with the Grp94 all along the pancreatic secretory route (ER-Golgi-granules). The Grp94-BSDL complex is secreted with the pancreatic juice into the acinar lumen and reaches the duodenal lumen, where it is internalized by enterocytes. The dissociation of the complex could take place within the endosomal compartment because BSDL continues further on its way to the basolateral membrane of the enterocyte. To localize the affinity binding sites of pancreatic BSDL in pancreatic and duodenal tissues, we have used an affinity-gold ultrastructural technique. BSDL coupled to gold particles appears to interact with specific sites in tissue sections. This was confirmed by another indirect morphological approach using biotin-labeled BSDL and streptavidin-gold complexes on tissue sections. We have shown that BSDL associates with sites in the pancreatic secretory pathway compartments and in the microvilli, the endosomal compartment, and the basolateral membrane of enterocytes. By biochemical approaches, biotin-labeled BSDL displayed affinities with proteins of 180-190 kD in both pancreatic and duodenal tissues. We have also shown that the Grp94-BSDL complexes, which are insensitive to denaturing conditions, are present in pancreatic homogenate but not in duodenal lysate. Thus, BSDL is able to bind protein complexes formed by either BSDL-Grp94 or Grp94 dimers.
T
In previous studies on the pancreas-derived AR4-2J cell line, we have shown that the secretion of BSDL involves a multiprotein complex including the Grp94 chaperone (Bruneau and Lombardo 1995). Combined biochemical and immunocytochemical investigations were carried out to study the secretion of BSDL by normal pancreatic cells and its transport to the small intestine. Both BSDL and Grp94 antigenic sites were localized and found to be associated along the pancreatic acinar cell secretory pathway. In pancreatic juice, these two proteins appeared as a complex of high molecular mass (180 kD) containing both Grp94 and BSDL molecules. The complex reaches the duodenal lumen, where it is internalized by enterocytes (Bruneau et al. 1998). Finally, the dissociation of Grp94-BSDL complexes could take place in the endosomal compartment because BSDL continues further to the basolateral membrane of the enterocyte. These data may argue in favor of a specific mechanism of transcytosis.
High-resolution cytochemical labeling techniques are available to cell biologists for subcellular localization of most types of biological macromolecules, from antigenic sites to carbohydrate residues, nucleic acids, and other substrate molecules, through their interaction with antibodies, lectins, and/or specific enzymes (Roth 1983; Bendayan 1981, 1984, 1995; Londono and Bendayan 1988; Benhamou 1989). Taking advantage of the affinity properties that exist between macromolecules, we have used the protein-gold electron microscopic affinity cytochemical approach (Bendayan 1984) to localize the affinity binding sites of BSDL in pancreatic and duodenal tissues.
Application of a protein-gold complex for labeling tissue sections consists of a direct one-step postembedding technique. The protein coupled to gold particles interacts with its specific binding site exposed at the surface of the tissue section. Colloidal gold is the electron-dense marker of choice, allowing detection at high resolution (Bendayan 1984, 1995). Therefore, BSDL was tagged with colloidal gold particles and applied to sections of pancreatic and duodenal tissues, allowing the ultrastructural localization of its affinity binding sites. To confirm the results obtained with the BSDL-gold complex, BSDL was conjugated to biotin and its interaction with cellular sites was detected on tissue sections with streptavidin-gold complexes. We have shown that BSDL associates with specific sites localized in compartments of the pancreatic secretory pathway and in microvilli, endosomal compartment, and basolateral membrane of enterocytes. By affinity blotting techniques using biotin-labeled BSDL, we have further shown that BSDL recognizes two complexes of proteins with an apparent mass of 180-190 kD in pancreatic and duodenal tissues.
Materials and Methods
Antibodies
Affinity-purified rabbit polyclonal antibodies against rat pancreatic BSDL (Abouakil et al. 1988) and monoclonal antibodies specific for Grp94 (clone 9G10) from StressGen Biotechnology (Victoria, BC, Canada) were used for immunoblotting.
Preparation of the BSDL-Gold Complex
Colloidal gold particles (10-nm) were prepared using a protocol described by Bendayan (1995). For the preparation of the complex, 100 μg of purified human BSDL (pI 4.5) (Lombardo et al. 1978) was mixed with 10 ml of the colloidal gold suspension at pH 5.6. The complex was centrifuged at 25,000 rpm at 4C for 30 min (Beckman ultracentrifuge, rotor Ti-50). The red sediment, corresponding to the BSDL-gold complex, was recovered in phosphate-buffered saline (PBS 0.01 M; NaCl 0.15 M) at pH 5.6, containing 0.02% polyethylene glycol (MW 20,000), and was stored at 4C.
BSDL Biotinylation
Human BSDL was isolated from pancreatic juice as already described (Lombardo et al. 1978) and biotin-labeled as follows. One mg of protein in 1 ml of 0.1 M sodium bicarbonate, pH 8.0, buffer was biotinylated with sulfosuccinimide 6-biotinamido hexanoate (Pierce; Rockford, IL) (50 μl of a 40 mg/ml stock solution in dimethylsulfoxide) for 2 hr at 4C. At the end of incubation, the reaction mixture was loaded onto a Sepharose G-25 column (Pharmacia; Quebec, Canada) to separate the free biotin from the biotin-conjugated BSDL. The biotinylated BSDL was then stored at 4C and diluted to 4 μg/μl before use. By dot-blot experiments, approximately 1 ng of biotin-labeled BSDL can be detected using streptavidin conjugated to alkaline phosphatase (Bruneau and Lombardo 1995).
Tissue Processing
Rat pancreatic and duodenal tissues were fixed by immersion with 1% (v/v) glutaraldehyde in 0.1 M sodium phosphate buffer, pH 7.4, for 2 hr at 4C. The tissue samples were washed in the phosphate buffer, dehydrated by a series of graded ethanol solutions, and embedded in Unicryl or Lowicryl (British Biocell; Cardiff, UK) at −30C as described previously (Bendayan 1995). Ultrathin sections were cut and mounted on Parlodion-carbon-coated nickel grids and processed for cytochemical labeling.
Cytochemistry
Labeling Protocol. The rat pancreatic and duodenal tissue sections were incubated by floating them on drops of PBS, pH 5.6, and were transferred successively to 1% ovalbumin solution for 30 min and to BSDL-gold complex for 30 min at room temperature (RT). Sections were then thoroughly washed with PBS and distilled water, dried, and stained with uranyl acetate. The grids were examined with a Philips 410 electron microscope. A similar protocol was carried out with biotin-labeled BSDL. Tissue sections were incubated on drops of PBS and transferred successively to 1% ovalbumin solution for 30 min and to biotin-labeled BSDL (10 μg/μl) for 30 min at RT. Sections were then washed with PBS. Free binding sites were blocked again by an incubation with the ovalbumin solution for 15 min and sections were finally incubated with the streptavidin-gold complex (Sigma; St Louis, MO) diluted 1:15 with PBS for 30 min. Thin sections were then extensively washed with PBS and distilled water and dried. They were stained with uranyl acetate and examined.
Controls for Specificity. The specificity of the labelings was assessed through competition experiments. Incubations of tissue sections were carried out with a solution of free BSDL (1 μg/μl) for 1 hr at RT before incubation with the BSDL-gold complex. Sections were also incubated with the strept-avidin-gold complex omitting the biotinylated BSDL step. In addition, labelings were carried out in the presence of 1% albumin or 1% gelatin solution before incubation with gold-or biotin-labeled BSDL. In this case, the preincubation or the presence of another protein does not prevent labeling.
Quantitative Evaluations. The labeling intensities were obtained as described previously (Bendayan 1995; Bruneau et al. 1998) using a Videoplan 2 image processing system (Carl Zeiss; Toronto, ONT, Canada).
Polyacrylamide Gel Electrophoresis and Western Blotting
Gel electrophoreses (SDS-PAGE) were performed on slab gels of polyacrylamide (7.5%) and sodium dodecyl sulfate (1.5%) under reducing conditions according to Laemmli (1970) using a Bio Rad mini-protean II apparatus. After SDS-PAGE, proteins were electrophoretically transferred onto a nitrocellulose membrane in 0.2 M Tris-HCl buffer, pH 9.2 (10 % methanol), at 150 mA for 3 hr in a cold room (Burnette 1981). Western blots were performed using specific antibodies for BSDL and Grp94 as primary antibodies (1 μg/ml). The antigen-antibody complexes were revealed by the BM chemiluminescence Western blotting kit (Boehringer; Mannheim, Germany).
Two-dimensional Gel Electrophoresis
Electrophoresis of pancreatic juice proteins was performed as described by Pasquali et al. (1997). In the first dimension, protein samples (25 μg) were separated according to their net charges by isoelectric focusing on Immobiline DryStrips (11 cm, pH 3-10 linear) on the MultiPhor II apparatus (Pharmacia Biotech; Uppsala, Sweden) according to the manufacturer's instructions. In the second dimension, after a standard SDS equilibrating step, proteins were further separated by SDS-PAGE 8%. The gel was run at 15 mA for 16 hr. Proteins were visualized by silver staining.
Ligand Affinity Blotting
Proteins that bind BSDL were detected by ligand affinity blotting on membranes obtained after SDS-PAGE and electrotransfer. Replicas were first blocked overnight with 0.1 M sodium phosphate buffer, pH 6.0 (3% bovine serum albumin, 50 mM NaCl) and incubated with biotin-labeled BSDL (5 μg/ml, 2 hr, 4C). After exhaustive washing (six times) with 0.1 M phosphate buffer (0.3% bovine serum albumin, 0.05% Tween-20), membranes were incubated (1 hr, RT) in 0.1 M sodium phosphate buffer, pH 7.4 (3% bovine serum albumin, 150 mM NaCl) with anti-biotin antibodies conjugated to alkaline phosphatase. Bands were visualized by incubating replicas with 0.5 mM 5-bromo-4-chloro-3-indoyl phosphate and 0.5 mM nitroblue tetrazolium in 0.1 M Tris-HCl, 0.1 M NaCl, 1 mM MgCl2 buffer, pH 9.5.
Results
To morphologically determine the affinity binding sites of BSDL in rat pancreatic and duodenal tissues, we have applied a cytochemical approach for the ultrastructural localization of macromolecules. This technique is based on the affinity properties existing between purified proteins and putative binding sites on tissue sections. For this purpose, BSDL was coupled with colloidal gold particles and tissue sections were incubated with the BSDL-gold complex to reveal BSDL binding sites.
In pancreatic acinar cells, binding sites for BSDL-gold complexes were detected in the rough endoplasmic reticulum (RER), the Golgi apparatus, the zymogen granules, and the flocculent material present in the pancreatic acinar and duct lumen (Figure 1). In control experiments in which the tissue sections were incubated with unlabeled BSDL before incubation with the BSDL-gold complexes, no interaction between BSDL-gold and tissue binding sites was detected. This illustrates the specificity of the recognition of BSDL-gold complexes for binding sites in the different compartments of the secretory pathway of the pancreatic acinar cell.
In intestinal cells, labeling with BSDL-gold complexes was restricted to the microvilli, the endosomal compartment, and the basolateral membrane of the enterocytes (Figure 1), suggesting that BSDL may have affinity binding sites in these compartments. Again, in this case, the preincubation of tissue sections with unlabeled BSDL abolished the reactivity of the BSDL-gold complexes. These data indicate that BSDL-gold complexes are interacting with sites on the enterocyte apical plasma membrane and associated with subcellular compartments of the transcytotic pathway.
To confirm these results, we applied a different cytochemical approach, making use of the streptavidin-biotin system. For this indirect procedure, BSDL was coupled to biotin and used for incubation of tissue sections. Affinity binding sites were detected by the streptavidin-gold complex. As shown in Figure 2, BSDL-biotin complexes were revealed over RER, Golgi, and secretory granules of pancreatic acinar cells (Figure 2A) and over microvilli, endosomal compartment, and basolateral membranes of the enterocyte (Figure 2B). Control experiments in which the tissue sections were incubated only with the streptavidin-gold complex (Figure 2C) showed no reactivity.
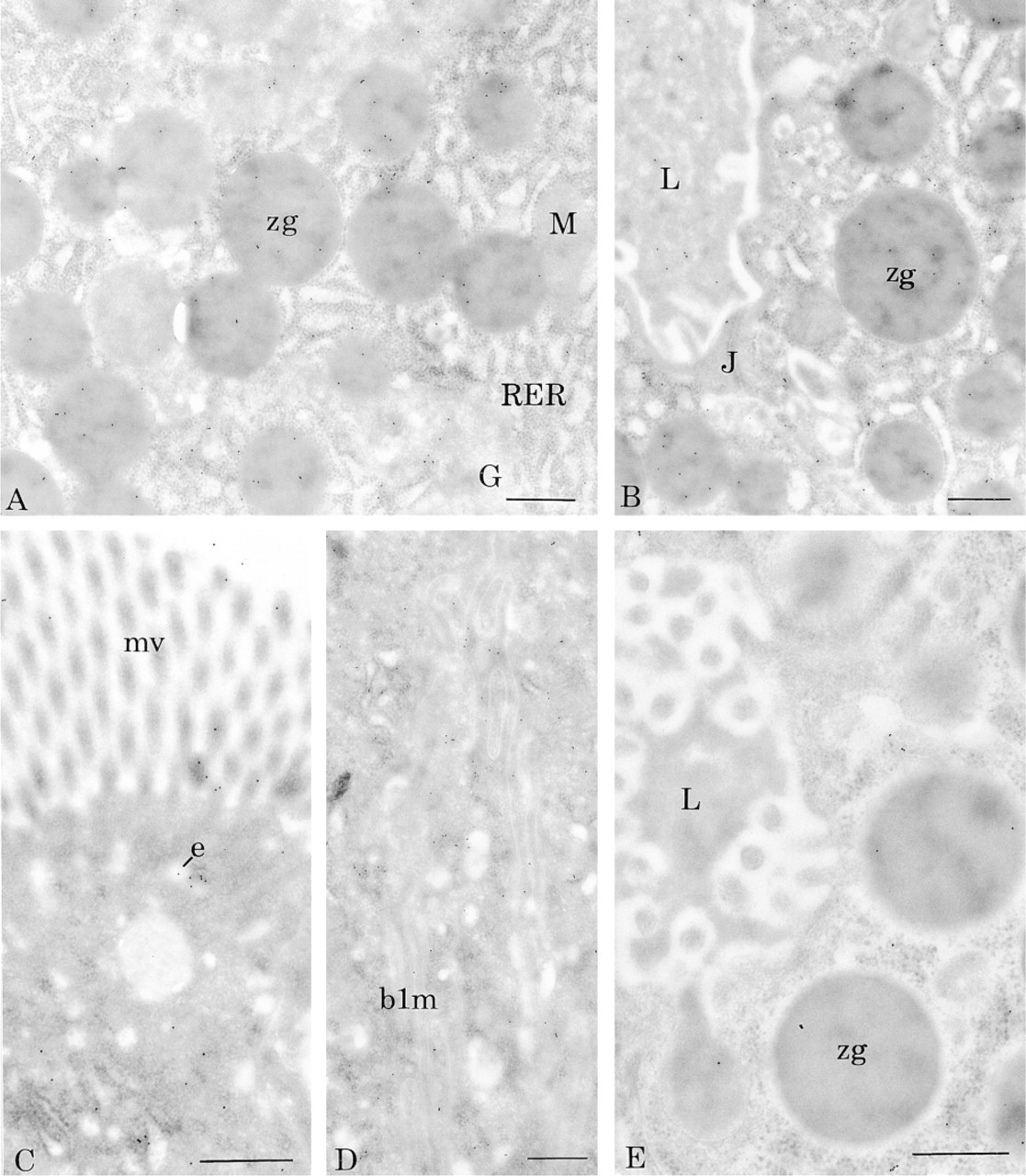
Cytochemical affinity of BSDL-gold complex on rat tissues. (
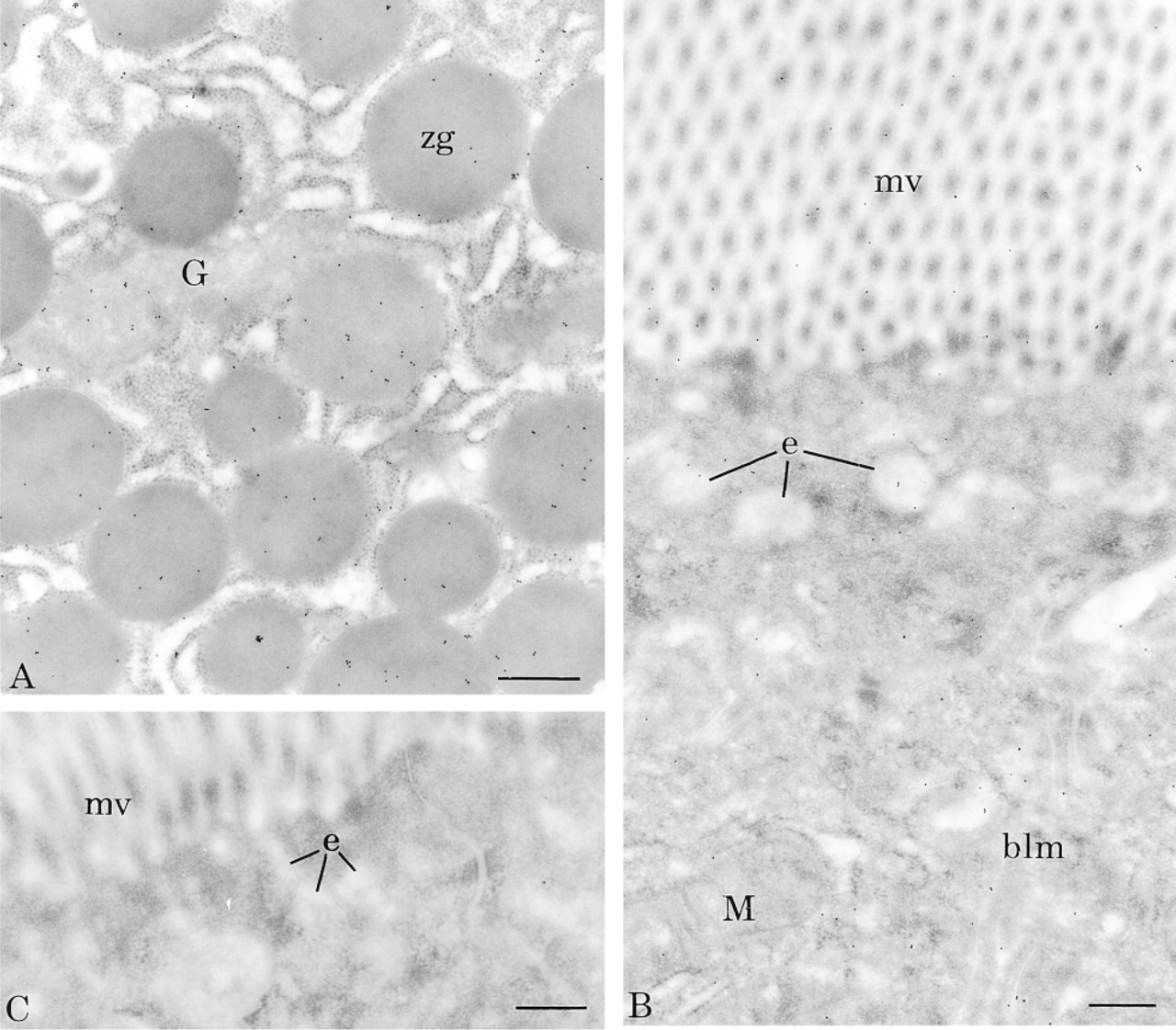
Cytochemical affinity of BSDL-biotin complex on rat tissues. (
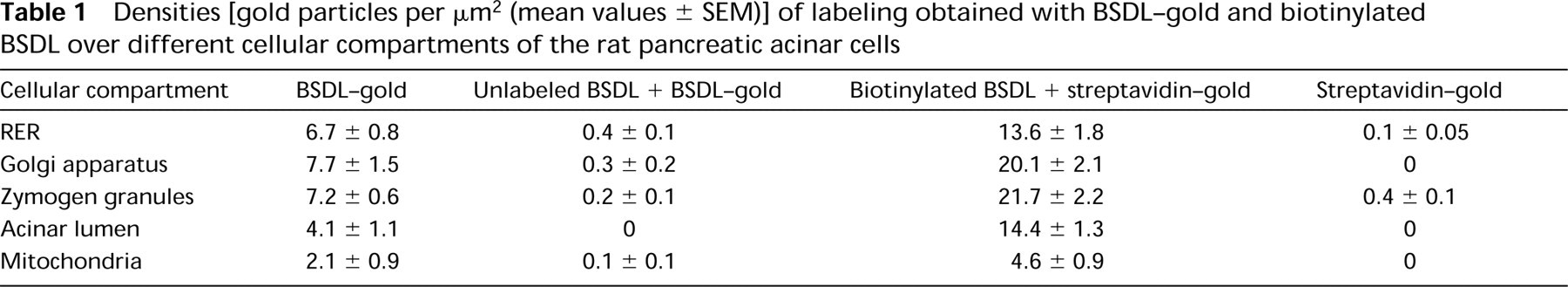
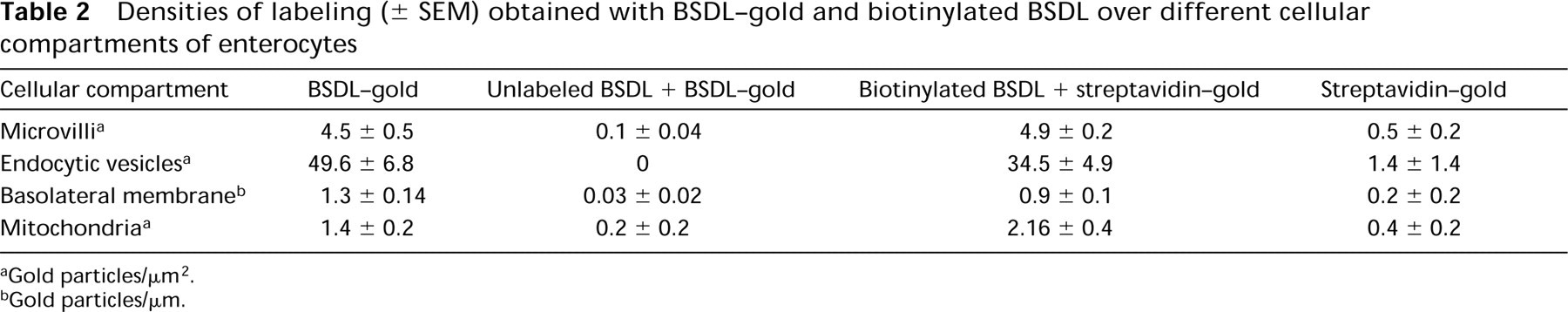
Quantitative evaluations were performed and are shown in Tables 1 and 2. They support the subjective observations showing that BSDL interacts with specific affinity binding sites in both tissues.
We next attempted to determine whether BSDL could bind directly to one or several isolated proteins from pancreatic and duodenal tissues. For this purpose, the biotin-labeled BSDL was used in ligand affinity blotting experiments to detect proteins capable of binding BSDL. Rat pancreatic and duodenal tissues were lysed in 10 mM Hepes buffer containing 0.2 M NaCl, 1.5% Triton X-100, 2 mM CaCl2, 2 mM MgCl2, and 2 mM benzamidine. After centrifugation (2000 times; g, 10 min) proteins were separated on SDS-PAGE and electrotransferred onto nitrocellulose membranes. When replicas were incubated with biotin-labeled BSDL, a doublet was detected by anti-biotin antibodies conjugated to alkaline phosphatase (Figure 3A). These bands display an electrophoretic migration corresponding to approximately 180-190 kD in both pancreatic and duodenal tissues. In control experiments in which biotin-labeled BSDL was omitted (Figure 3B), no band at this Mr was detected. These data suggest that biotin-labeled BSDL binds to proteins that have a migration corresponding to 180-190 kD in pancreatic and intestinal tissues.
Densities [gold particles per μm2 (mean values ± SEM)] of labeling obtained with BSDL-gold and biotinylated BSDL over different cellular compartments of the rat pancreatic acinar cells
We then wished to collect information on these proteins and to determine whether BSDL and Grp94 are present in these complexes of high molecular mass. For this purpose, we carried out Western blots on pancreatic and duodenal proteins after SDS-PAGE separation and electrotransfer. Replicas were then probed with antibodies specific for BSDL and Grp94, respectively. In pancreatic and duodenal tissues, BSDL was detected as one major band with an apparent migration of 74 kD (Figure 3C). A second band migrating at approximately 180 kD was also detected in the pancreatic lysate but not in the duodenal one. This latter result indicates that BSDL appears to be part of this high molecular weight complex in pancreatic tissue but not in duodenal tissue.
With regard to Grp94, proteins of both pancreatic and duodenal homogenates were separated by SDS-PAGE, electrotransferred onto nitrocellulose membrane, and finally developed with the monoclonal antibodies to Grp94. As shown in Figure 4D (Lane 1), four proteins reacted with the anti-Grp94, indicating that Grp94 is present in different forms in pancreatic and duodenal lysates. One of these is the monomeric form of Grp94, migrating at 94 kD. The other three display a close migration between 180-200 kD. The intensity of labeling of these latter bands representing complexed Grp94, compared to that of the monomer, suggests that most of the Grp94 in pancreas and in intestinal cells forms heterodimers and/or homodimers.
The band migrating at 180 kD correlates with the heterodimeric complex immunoprecipitated from the rat pancreatic juice containing Grp94 and BSDL (Bruneau et al. 1998). The detection of such complexes on SDS-PAGE indicates that they should be stable and consequently insensitive to denaturing conditions, as suggested for other complexes involving Grp94 (Nicchitta 1998). To assess this specific point, the stability of the heterodimer present in rat pancreatic juice was studied under various denaturing conditions. As shown in Figure 4A, immunodetection of BSDL in rat pancreatic juice with specific antibodies enabled us to detect monomeric BSDL (Mr 74 kD) and BSDL-Grp94 complexes (Mr 180 kD). This detection was independent of the SDS concentration used to preincubate samples loaded onto SDS-PAGE (Figure 4A, Lanes 2-5). Complexes were still present when samples were preincubated in 8 M urea (Figure 4A, Lane 1), indicating that the interaction between proteins probably involved hydrophobic forces. The monoclonal antibody to Grp94 also detected the 180-kD complexes under all conditions. Grp94 monomers appeared to be more strongly represented when samples were pretreated with 0.25 % SDS (Figure 4B, Lane 4).
When rat pancreatic juice proteins were segregated in the first dimension according to their pI and then in a second dimension according to their size, a unique 180-kD band migrating with a pI of 4.6 and probably representing the BSDL-Grp94 complex was detected in 2D electrophoresis (Figure 4C). Note that BSDL and Grp94 have a similar pI (4.5 and 4.6, respectively).
All these data strongly suggest that (a) the protein detected in Figures 3C and 3D and migrating at 180 kD is probably the heterodimer BSDL-Grp94, (b) BSDL is able to bind protein complexes composed of either BSDL-Grp94 or Grp94 dimers, (c) the 180-190-kD proteins that bind biotinylated BSDL could represent putative specific binding sites for BSDL that are present in pancreatic tissue and duodenal tissue, (d) the affinity binding sites of BSDL in the pancreatic secretory pathway are probably composed of heterodimer BSDL-Grp94, (e) the BSDL-Grp94 complexes internalized by the enterocytes were probably not detected because of their low amounts, and (f) Grp94 dimers present in the enterocyte are able to bind biotinylated BSDL.
Densities of labeling (± SEM) obtained with BSDL-gold and biotinylated BSDL over different cellular compartments of enterocytes
Gold particles/μm2.
Gold particles/μm.
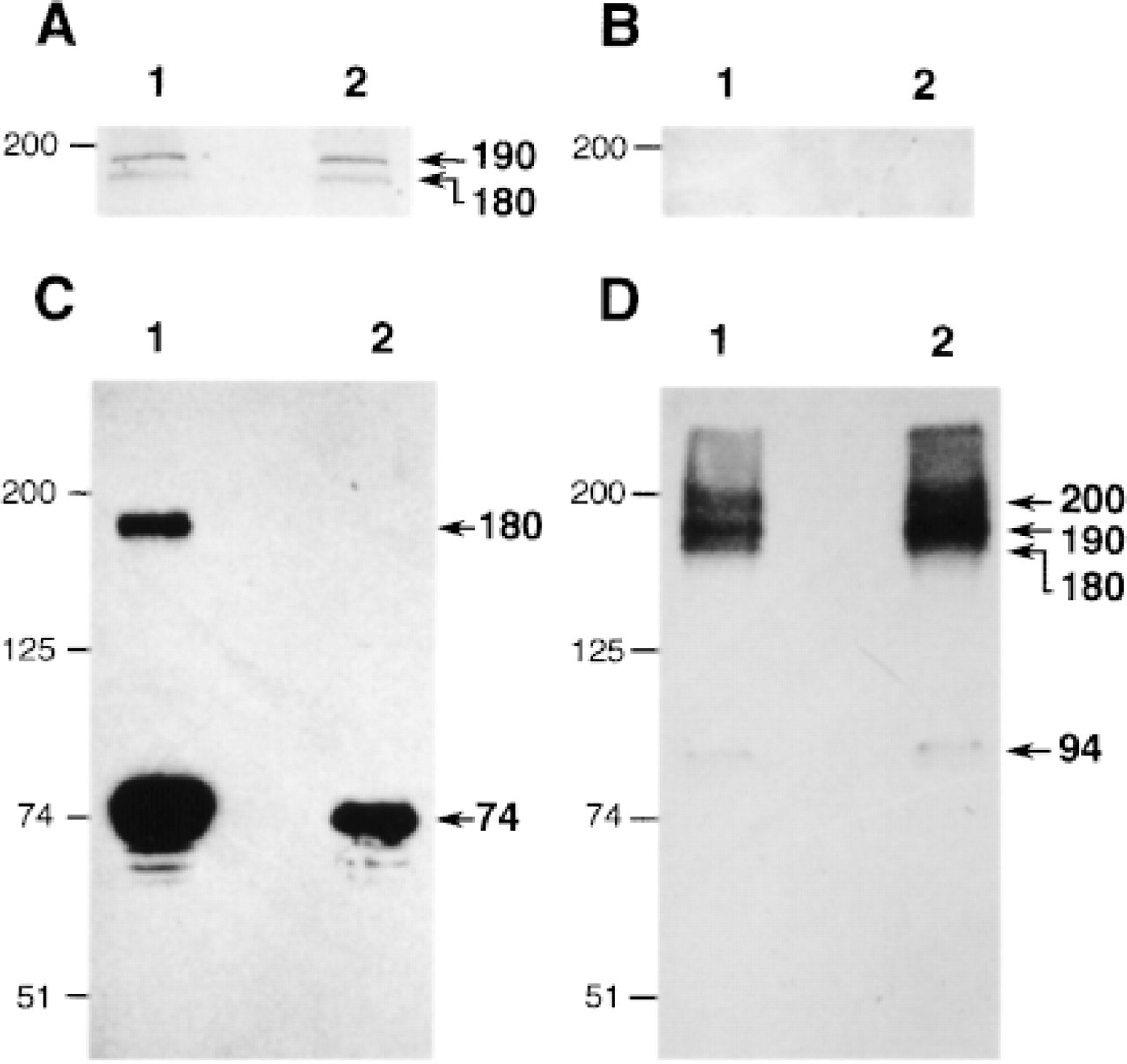
Western and affinity blotting analysis of rat pancreatic and duodenal tissues. The proteins present in pancreatic and intestinal homogenates were separated by 7.5% SDS-PAGE (Lane 1, 30 μg of cell proteins; Lane 2, pancreatic proteins; Lane 3, duodenal proteins) and electrotransferred onto nitrocellulose membrane. (
Discussion
Several molecular chaperones have been shown to participate in the secretion of pancreatic enzymes (Vélez-Granell et al. 1994; Le-Gall and Bendayan 1996; Bruneau et al. 1998; Takemoto et al. 1992). Furthermore, some of these chaperones were found to be associated with secretory proteins and accompanied them along the secretory pathway from the ER to the zymogen granules, to be secreted with the pancreatic juice (Le-Gall and Bendayan 1996; Bruneau et al. 1998).
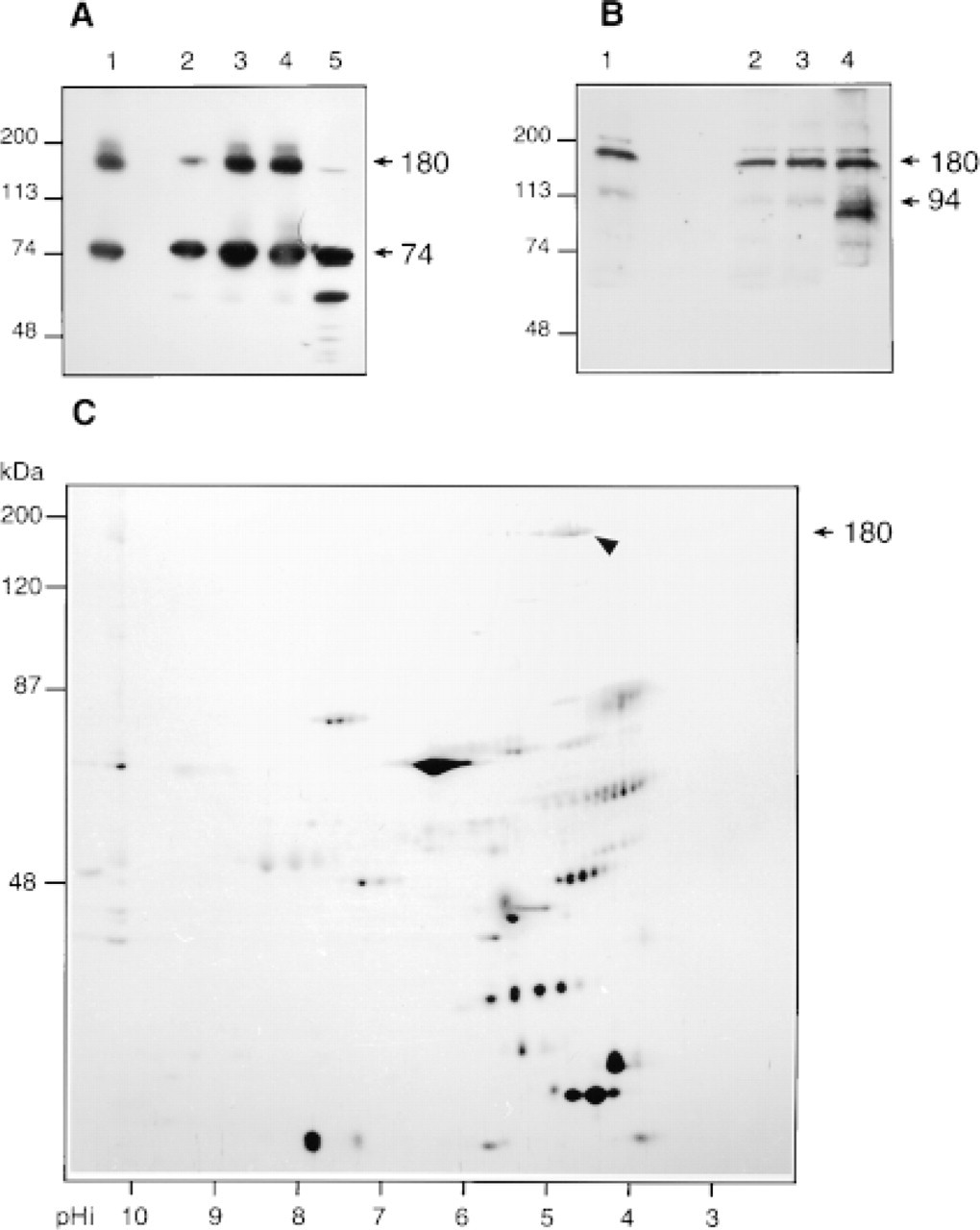
Analysis of pancreatic juice proteins. (
Among these secreted pancreatic chaperones, Grp94 is a glycoprotein that belongs to the glucose-regulated family of proteins. This chaperone, like Grp78 (also called Bip), possesses the KDEL C-terminal sequence, which is a signal for ER retention. Grp94 forms stable homodimers and is part of an oligomeric complex together with Grp78, calreticulin, calnexin, and similar proteins, and participates in the correct folding and assembly of ER proteins and their exit from this organelle (Nemoto et al. 1996; Henle et al. 1998). The orientation of Grp94 dimer is conferred by a carboxyl-terminal oligomerization domain that maintains the amino termini at opposing ends of the molecule (Nicchitta 1998), suggesting that the Grp94 complex may be secreted because the KDEL C-terminal sequence cannot be recognized by the KDEL receptor or because the KDEL sequence is truncated (Nicchitta 1998). Furthermore, once formed, the peptide-Grp94 complex is SDS-resistant (Nicchitta 1998).
Like all digestive enzymes synthesized by the pancreas, BSDL follows the regulated secretory pathway (Bendayan et al. 1980) originally defined by Palade (1975). A particular feature in BSDL secretion is its association with intracellular membranes. The membrane-associated BSDL is O- and N-glycosylated, and consequently the enzyme should be associated with membranes until reaching the trans-Golgi compartment, where it undergoes terminal fucosylation (Mas et al. 1997). This association to membranes involves a multiprotein complex, including Grp94, in which this chaperone is directly linked to BSDL (Bruneau and Lombardo 1995). The association of BSDL with Grp94 appears to be essential for the folding and intracellular transport of the enzyme along the pancreatic secretory route. Multiple reasons can be suggested to explain the particular secretory process of BSDL. The most likely would be the fact that BSDL is a unique secretory pancreatic protein with mucin-like tandem repeated sequences on which O-linked oligosaccharides are clustered. We have shown that the rate of secretion of BSDL depends on the ability of the cell to O-glycosylate the C-terminal repeats of the protein and that only O-glycosylated BSDL is secreted (Bruneau et al. 1997). Furthermore, we have postulated that the association of BSDL with Grp94 is a prerequisite to ensure complete O-glycosylation of the enzyme.
By combining immunocytochemical and biochemical techniques, we have recently shown that the association between BSDL and Grp94, which forms a 180-kD complex, takes place in the RER, continues in the Golgi apparatus and the zymogen granules, and is secreted in the pancreatic juice (Bruneau et al. 1998). In the present study we report the localization of affinity binding sites for BSDL. Using a morphological approach consisting of biotin-labeled and gold-labeled BSDL incubation of tissue sections, we show that BSDL interacts with sites along the pancreatic secretory pathway. As shown in Figure 3A (Lane 1), one of these sites could correspond to proteins of 180-190 kD. The immunochemical data suggest (Figure 3C) that in pancreatic tissue BSDL is part of the 180-kD heterodimer containing one molecule of Grp94 and one molecule of BSDL, as already found in pancreatic juice (Figure 4; and Bruneau et al. 1998). In the pancreatic tissue, Grp94 is also present as a monomer (94 kD) and as two more complexes with Mr >180 kD. Because in the Grp94-BSDL 180-kD complex the Grp94 binding site for BSDL is already occupied, it is probable that proteins migrating at 180-190 kD which are able to bind biotinylated BSDL do not represent those Grp94-BSDL complexes. However, they may represent dimers of Grp94 (Nicchitta 1998) which could still be able to recognize biotinylated BSDL. Consequently, the 180-190-kD proteins that bind biotinylated BSDL could represent putative specific binding sites for BSDL which are present in pancreatic tissue, probably along the secretory pathway.
Two molecular populations of BSDL appear to be secreted in the pancreatic juice. One is monomeric BSDL, whereas the second is associated with Grp94 (Figure 4; and Bruneau et al. 1998). In the duodenal lumen, the physiological role of monomeric BSDL is to catalyze the hydrolysis of cholesterol esters, which cannot be absorbed without prior hydrolysis to free cholesterol (Huang and Hui 1990; Shamir et al. 1995; Howles et al. 1996). Concerning the BSDL-Grp94 complex, which also reaches the duodenal lumen, once in the vicinity of microvilli and on binding to the cell surface it is internalized via endocytosis by enterocytes (Bruneau et al. 1998). The Grp94-BSDL complex dissociates in the endosomal compartment and BSDL continues on its way to the basolateral membrane of the enterocyte (Bruneau et al. 1998). The presence of BSDL in the blood of normolipidemic patients (Caillol et al. 1997) could result from such transcytotic transport across the duodenal mucosa.
The mechanism by which BSDL interacts with membranes appears to be specific. Bosner et al. (1988) have demonstrated the interaction of BSDL with heparin-like molecules at the surface of intestinal cells. Using a heterologenous in vitro system (CaCo-2 cells and porcine enzyme), Huang and Hui (1990) have shown that the initial interaction of BSDL with these cells was mediated by the binding of BSDL to low-affinity and high-capacity binding sites at the cell surface. Recently, we have postulated that Grp94 appears to be important in promoting the enterocytic internalization of pancreatic BSDL (Bruneau et al. 1998). In the enterocytic cells BSDL, either biotin-labeled or gold-labeled, binds to sites on microvilli, in endocytic vesicles, and in basolateral membranes (Figures 1 and 2). In rat duodenal homogenate, the biotin-labeled BSDL appears to interact with the 180-190-kD proteins which could be similar to those already characterized in pancreas (Figure 4A). If so, these proteins, which are probably those able to bind biotin-labeled or gold-labeled BSDL on tissue sections of the different compartments of the secretory and endocytic pathways in pancreatic and duodenal cells, could be proteins involved in the trafficking of BSDL within these cells.
As mentioned above, in enterocytes BSDL binding sites are associated with microvilli, the endosomal compartment, and basolateral membranes. However, BSDL-Grp94 complexes were detected up to the endosomal compartment, BSDL alone (not associated with Grp94) being present in basolateral interdigitations (Bruneau et al. 1998). These data suggest that, in the enterocytes, pancreatic BSDL dissociates from Grp94, which could explain why Grp94-BSDL complexes were not detected in enterocytes (Figure 4C, Lane 2), although endogenous Grp94 is present in enterocytes and involved in complexes (Figure 4D, Lane 2), probably with other proteins. Alternatively, the failure to detect Grp94-BSDL complexes in duodenal tissue could be due to low amounts of internalized complexes that are below the threshold of detection.
In conclusion, we have recently shown (Bruneau et al. 1998) that the transport of BSDL all along the pancreatic secretory route occurs via an association with Grp94. These pancreatic complexes are secreted in the pancreatic juice and reach the duodenal lumen, where they are internalized by specific endocytosis into enterocytes. On dissociation from Grp94, BSDL is transferred to the basolateral membrane. We show here that Grp94-BSDL complexes are present in pancreatic tissue but not in duodenal tissue, suggesting that in the latter these complexes are rapidly destroyed or dissociated (being less stable). We further demonstrate that the trafficking of the enzyme in pancreatic and enterocytic cells probably involves specific binding sites which could be protein complexes of 180-190 kD, the nature of which remains to be elucidated.
Footnotes
Acknowledgments
Supported by the Medical Research Council of Canada and by the Institut National de la Santé et de la Recherche Medicale (France). Dr N. Bruneau is recipient of a fellowship from the Fondation pour la Recherche Medicale.
The technical assistance of D. Gingras, C Rondeau, and G. Mayer and the photographic work of J. Léveillé and C. Crotte are greatly appreciated. We thank Dr M. Desjardins for his kind collaboration.
