Abstract
The NF-κB/IκB complex is a major transcription regulator of inflammatory and immune responses. Helicobacter pylori infection causes chronic inflammation in gastric mucosa by inducing dissociation of the inhibitory IκB protein from the complex with a resulting increased expression of interleukin (IL)-8. To clarify which of several known IκB proteins could be involved in this inflammatory response, we undertook immunohistochemical examination of normal mouse stomach as well as other murine tissues for comparison, using polyclonal antibodies specific for α-, β-, γ-, and ∊-isoforms of IκB. The results showed strong immunoreactivity for the α-isoform in parietal cells and for the β-isoform in pit cells of the stomach, along with the presence of these proteins in various other sites. Comparative staining revealed a similar but not identical distribution of IκB proteins in the Mongolian gerbil, a rodent model for H. pylori infection. The findings suggest that the α- and β-iso-forms are dominant IκB proteins in gastric parietal and foveolar cells, respectively, and point to a role for these transcription regulators in modulating pathological responses in stomach and other organs.
NF-κB/IκB
NF-κB proteins include p105 which, for example, associates at its C-terminal region with IκBγ as the inhibitory component. Other members of the NF-κB family are associated with different inhibitory IκB isoforms. Members of the IκB protein family include IκBα, IκBβ, IκBγ, IκBδ, IκB∊, and Bcl-3. Each of these isoforms possesses variable ankyrin repeats that connect the inhibitory protein to the Rel-homology domain of NF-κB protein. (Baeuerle and Baltimore 1996; May and Ghosh 1998; Sha 1998).
The NF-κB/IκB system has been studied mainly in immunocompetent cells such as T- and B-lymphocytes and macrophages. However, NF-κB consisting of a p50/p65 heterodimer also has been demonstrated in cultured gastric surface epithelium (Rokutan et al. 1997). Moreover, NF-κB is upregulated in the stomach after H. pylori infection (Keates et al. 1997; Sharma et al. 1998), which commonly leads to chronic gastritis and gastric malignancies. The fact that infection with H. pylori induces production of IL-8 in gastric epithelium and gastric epithelial cell lines (Crabtree et al. 1994; Moss et al. 1994; Noach et al. 1994; Crowe et al. 1995; Fan et al. 1995; Huang et al. 1995; Sharma et al. 1995; Kassai et al. 1999) provides further evidence for a role of the NF-κB/IκB complex in regulating gastric immune responses, because IL-8 expression is known to be regulated by NF-κB under certain conditions.
Although mRNA for IκBα has been demonstrated by Northern blot analysis in gastric homogenates (Rupec et al. 1999), IκB protein isoforms have not been localized to specific cell types in the stomach. Moreover, there is little information concerning the distribution of members of the IκB family in other mammalian tissues. The present study undertook immunolocalization of IκB in stomach and in other organs for comparison. The results show a cell type-specific distribution of different IκB proteins in the gastric epithelium and in other tissues not previously known to express this transcription inhibitor.
Materials and Methods
Animals and Tissue Processing
Tissues harvested from eight healthy 3-month-old mice of the BALB/c strain and four similarly aged Mongolian gerbils, Meriones unguiculatus (half from each gender), were examined. Care and use of the animals were approved by the Medical University of South Carolina's Animal Review Committee under NIH grant DC 00713. The animals were anesthetized by IP injection of sodium pentobarbital (50 mg/kg) and exsanguinated by transcardial perfusion with 10 ml of a 10% formalin solution containing 0.5% zinc dichromate, pH 5.0. The stomach and other organs, including eye, heart, lung, thymus, lymph nodes, liver, pancreas, small and large intestine, kidney, urinary bladder, ovary, uterus, testis, and seminal vesicles, were harvested, sliced into appropriately sized pieces, and immersed in the same fixative for 60 min. The specimens were dehydrated in a graded series of ethanols, cleared in Histoclear (National Diagnostics; Manville, NJ), and embedded in Paraplast X-TRA (Sherwood Medical; St Louis, MO) before sectioning at 5-μm thickness. Organs other than stomach were embedded together in composite blocks for appraising the tissue and cell type-specific distribution in the animal of IκB proteins and the specificity of the antisera.
Immunohistochemistry
Immunostaining of tissue sections was performed as described previously (Schulte and Adams 1989; Schulte and Schmiedt 1992), with slight modification. Deparaffinized and rehydrated sections were immersed for 15 min in 3% H2O2 to block endogenous peroxidase. After washing with distilled water, the sections were treated with 0.1% Triton X-100 in PBS for 30 min, rinsed with PBS, and flooded for 30 min with PBS containing a 5% concentration of normal goat serum. Sections were then incubated overnight at 4C with one of the primary antisera diluted with PBS. The primary antisera and working dilutions tested included the following: rabbit antihuman IκBα (C-21) 1:600; rabbit anti-mouse IκBβ (N-20) 1:300; rabbit anti-human IκBγ (5177-C) 1:600; rabbit antimouse IκB∊ (M-364) 1:100 (Santa Cruz Biotechnology; Santa Cruz, CA). The sections were rinsed in PBS and incubated with biotinylated goat anti-rabbit IgG diluted 1:200. After rinsing with PBS, the sections were flooded with avidin-biotin-horseradish peroxidase complex (Vectastain ABC kit; Vector Laboratories, Burlingame, CA) for 30 min. The sections were again rinsed with PBS and reacted for 10 min in substrate medium containing 3,3′-diaminobenzidine HCl (Sigma Chemical; St Louis, MO) before dehydration and mounting.
Controls for immunostaining included the deletion of primary antiserum from the above-described staining procedure.
In addition, each of the primary antisera against the α-, β-, and γ-isoforms was preabsorbed with its homologous blocking peptide and with the other two available heterologous peptides to access crossreactivity. The α, β, and γ blocking peptides were purchased from Santa Cruz Laboratories. Blocking peptide for the ∊-isoform was not available.
Results
Immunostaining demonstrated IκB proteins in various histological sites in the mouse (Table 1) and gerbil. Specimens from mice and gerbils stained comparably, with the exception of a few minor differences noted below.
Gastrointestinal Tract
Strong immunoreactivity for IκBα was observed in cytoplasm of parietal cells in stomach (Figure 1a). The staining intensity varied slightly among individual cells, but not in accord with any known gradient. In contrast, IκBβ was localized in pit cells of the glandular epithelium (Figure 1b). In some deep foveolar cells the nuclei stained more intensely than did cytoplasm, whereas the most superficial cells exhibited pancellular reactivity (Figure 1b, inset). In the gerbil but not in the mouse, the peripheral cytoplasm and/or plasma-lemma of parietal cells was weakly positive for IκBβ. The antiserum to IKBγ stained superficial foveolar cells (Figure 1c), but the staining for IκBγ differed from that for IκBβ in staining some but not other clusters of surface mucous cells.
Distribution of IκB proteins in normal mouse tissues a
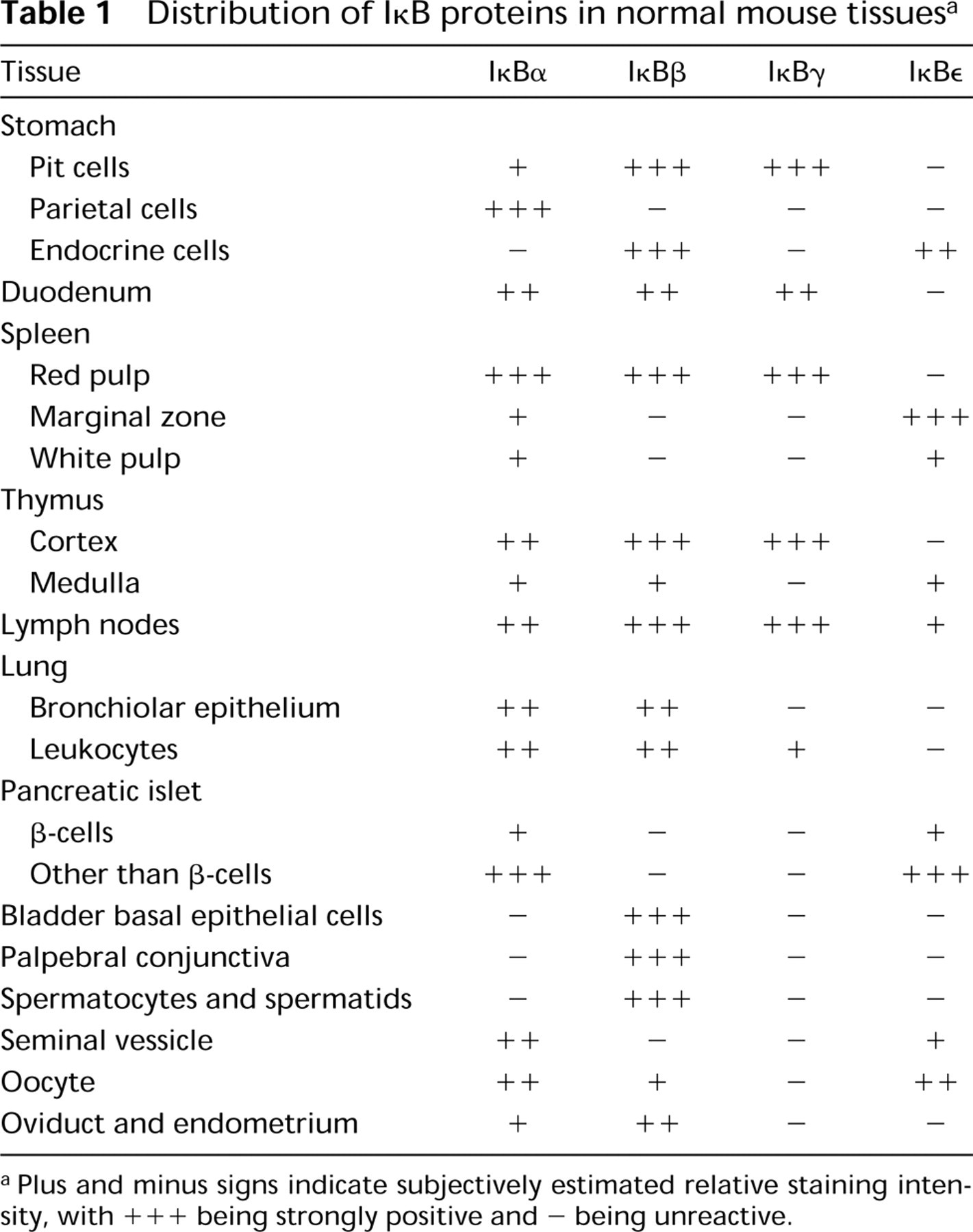
Plus and minus signs indicate subjectively estimated relative staining intensity, with +++ being strongly positive and - being unreactive.
Immunostaining for IκB proteins in mouse stomach. (
Cells positive for IκBβ and IκB∊ occurred sparsely in the corpus near the forestomach but were more abundant near the bottom of pyloric glands in mice (Figures 1d and 1e). These cells corresponded with cells having clear, teardrop-shaped cytoplasm in H&E-stained sections (not shown), facilitating their designation as endocrine cells. In the gerbil these cells were positive for IκB∊ but not for IκBβ, and were scattered evenly in the lower region of the entire glandular epithelium.
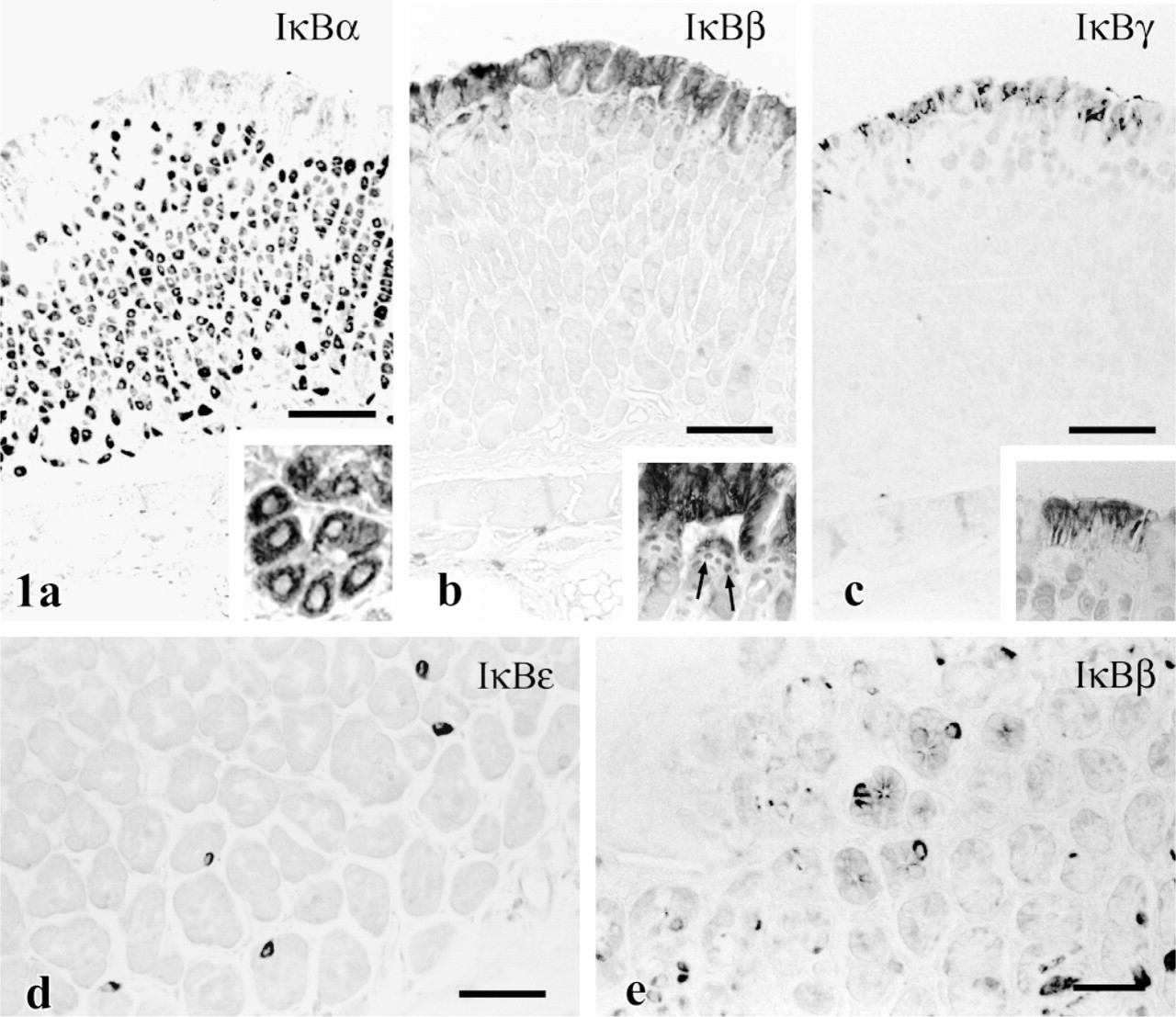
Columnar cells in the upper half of villi in the mouse duodenum were reactive with antisera against IκBα and IκBβ (Figures 2a and 2b). Staining with anti-IκBα was stronger in nuclei than in cytosol in the positive cells. Moderate immunoreactivity for IκBγ was detected in some cells at the apex of duodenal villi (Figure 2c). Although this pattern was similar in mice and gerbils, the gerbil tissues showed higher background staining and less intense nuclear staining for IκBα.
Immune System
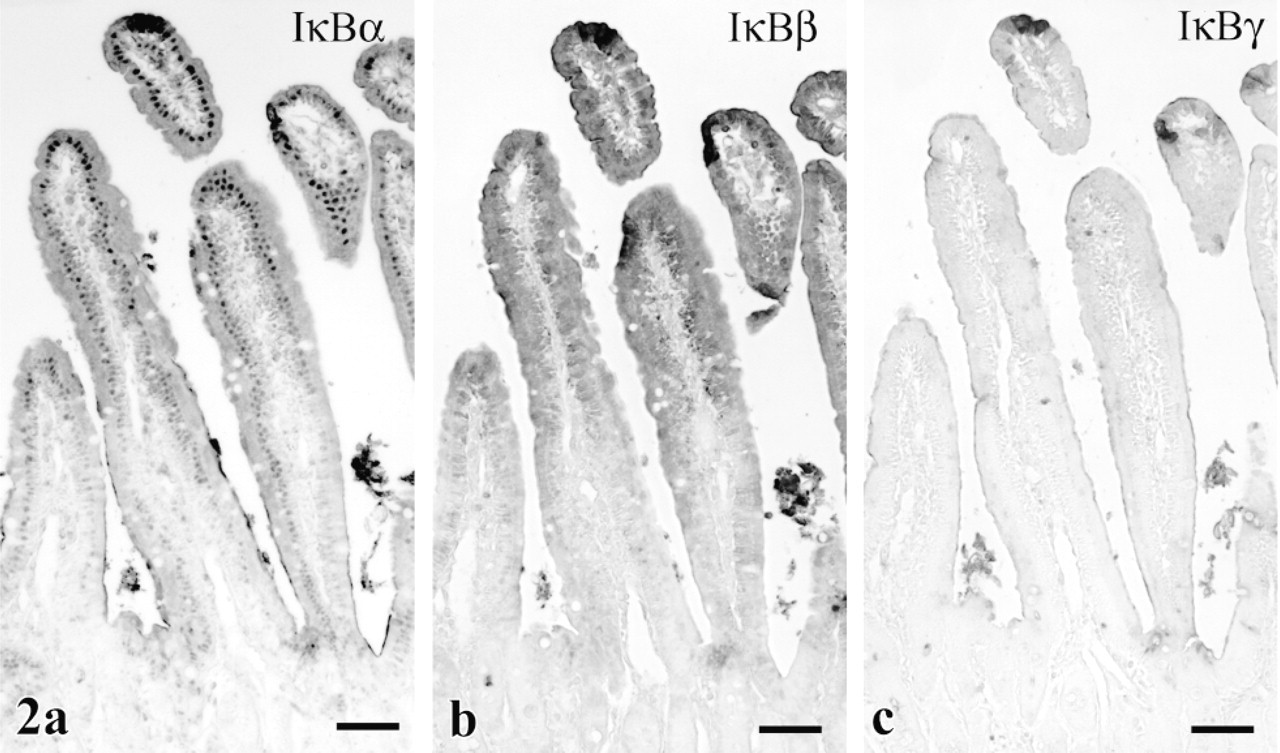
All four known IκB isoforms were present in various regions of the spleen, thymus, and lymph nodes of both mouse and gerbil. Clusters of cells in splenic red pulp were strongly positive for IκBα (Figure 3a), whereas many isolated cells stained only weakly. Splenic cells expressed IκBβ and γ similarly. IκB∊ immunostaining was confined to the marginal zone of white pulp in the spleen (Figure 3b).
Immunostaining of duodenum. (
In the thymus, occasional cells with strong affinity for antibodies to IκBα (Figure 3c), β, and γ lay scattered just under the capsule. Sporadic cells in the medulla of lymph nodes also showed strong immunoreactivity for IκBα, β, and γ (Table 1). These positive immune cells appeared more numerous in gerbil than in mouse.
Many IκB-immunoreactive cells in immune system organs could not be clearly differentiated as lymphocytes, neutrophils, or macrophages. On the other hand, large reactive cells with abundant cytoplasm in pulmonary alveoli (Figure 3d), peritoneal fat around the ovary, and splenic red pulp were interpreted as macrophages. The presumed macrophages in these sites expressed both IκBα and p.
Genitourinary System
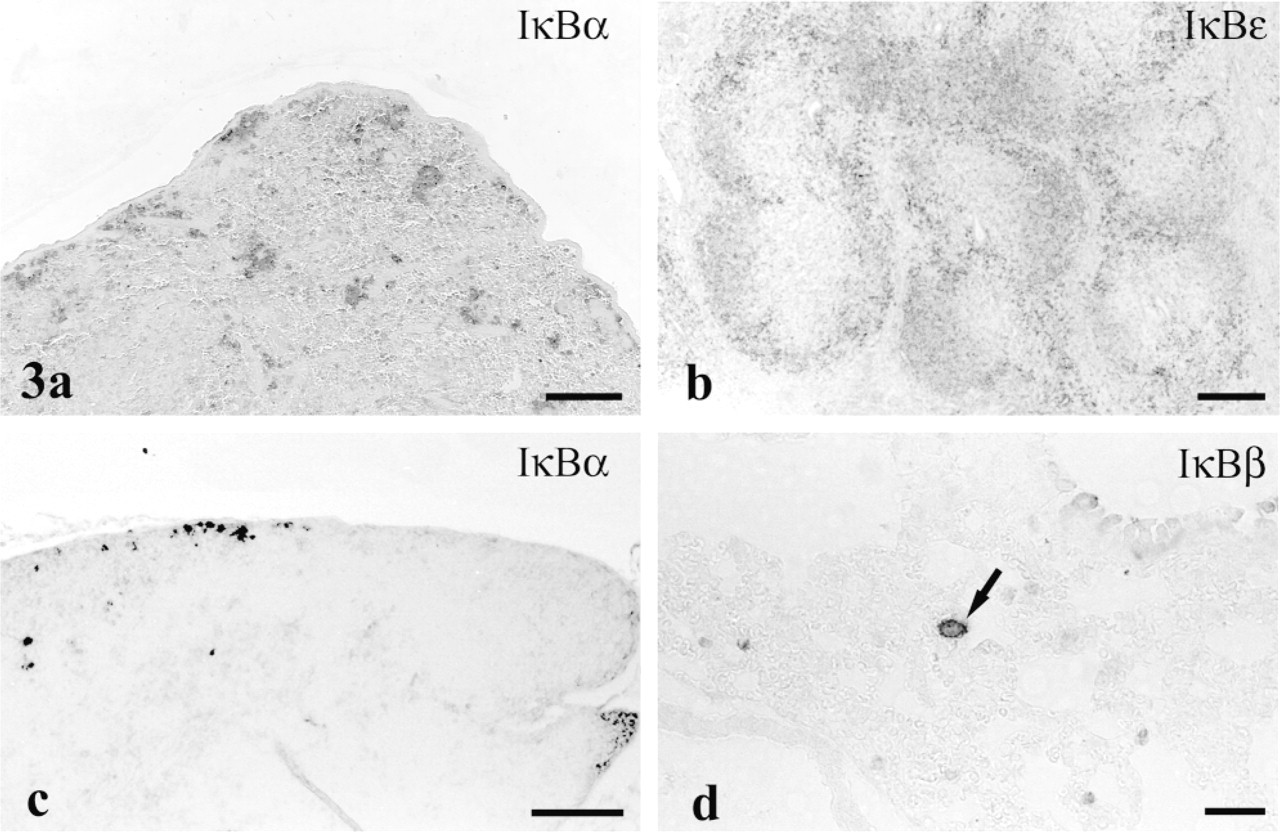
Cytoplasm in epithelium of the murine seminal vesicle showed moderate to intense staining with anti-IκBα (Figure 4a) and weaker staining with anti-IκB∊. in the mouse testis, the heads of spermatids showed moderate reactivity, and a small number of presumably meiotic spermatocytes were intensely stained with anti-IκBβ (Figure 4b and inset). In the gerbil, spermatid/spermatozoon reactivity was abundant, whereas spermatocyte staining was rare.
Cytosol in epithelium of the endometrium (Figure 4c) and oviduct (Figure 4d) stained moderately for the IκBβ -isoform. Cytosol of oocytes revealed moderate staining for both IκB∊ (Figure 4e) and IκBα. in urinary bladder, basal cells of the transitional epithelium expressed strong immunoreactivity demonstrative of IκBβ (Figure 4f).
Miscellaneous Organs
In the lung, some bronchiolar cells showed moderate to strong cytosolic affinity for IκBα (Figure 5a) and IκBβ antibodies, as did occasional cells infiltrating pulmonary alveoli (Figure 5b). Most of the latter cells possessed a polymorphic nucleus and were considered to be neutrophils, but a few with abundant cytoplasm and an oval nucleus were interpreted as alveolar macrophages. Pneumocytes lacked staining.
IκBα and IκB∊ were restricted to cells presumably representing α- and/or Δ-cells in the periphery of pancreatic islets (Figure 5c). Cytoplasm of the positive islet cells was stained intensely. The centrally located β-cells bound antibody to α- and ∊-isoforms only weakly.
Localization of IκB proteins in immune system. (
In palpebral conjunctiva, the most superficial cells exhibited intense staining for IκBβ, far exceeding that in the basal palpebral conjunctiva superficial corneal epithelium (Figure 5d).
Deletion of the primary antiserum resulted in no staining in any tissue. Absorption of either anti-IκBα or anti-IKBβ with its homologous blocking peptide completely eliminated staining in all sites. In contrast, IκBγ blocking peptide diminished but failed to abolish lymphocyte immunoreactivity with anti-IKBγ in the spleen. Cross-absorption of the primary antisera with similar concentrations of the heterologous blocking peptides (α, β, and γ) failed to affect staining reactivity in any site.
Discussion
Cellular Distribution of IκB Proteins
Although NF-κB proteins were first detected in the immune system, they are now known to exist in other organs such as brain (Meberg et al. 1996; Korhonen et al. 1997; Suzuki et al. 1997), lung/bronchus (Stacey et al. 1997; Aoki et al. 1998), heart (Meldrum et al. 1997; de Moissac et al. 1998), and pancreas (Flodström et al. 1996; Kwon et al. 1998). The NF-κB system has also been observed in fibrocytes (Olashaw 1996; Pan et al. 1996), vascular endothelial cells (Kilgore et al. 1997; Zhang et al. 1998), and certain cancer cell lines. These studies mainly employed electrophoretic mobility shift assays and other blotting methods to detect NF-κB/IκB proteins.
This report presents the first immunohistochemical data mapping the expression of inhibitor IκB isoforms in several major organ systems. The study shows a widespread distribution of immunodetectable NF-κB/IκB proteins. Previous in situ hybridization data (Weih et al. 1994) showing mRNA for IκBα distributed diffusely throughout the spleen appear not to agree with the present observation of focal strong staining for this isoform in red pulp and only weak reactivity in the marginal zone and lymphatic follicles. The reason for a discrepancy between the in situ hybridization and immunohistochemistry results is uncertain, but a similar observation has been made for RelA(p65) and NF-κB1(p50) (Feuillard et al. 1997). A possible explanation could lie in a different rate of synthesis vs degradation for mRNA compared with that of the protein.
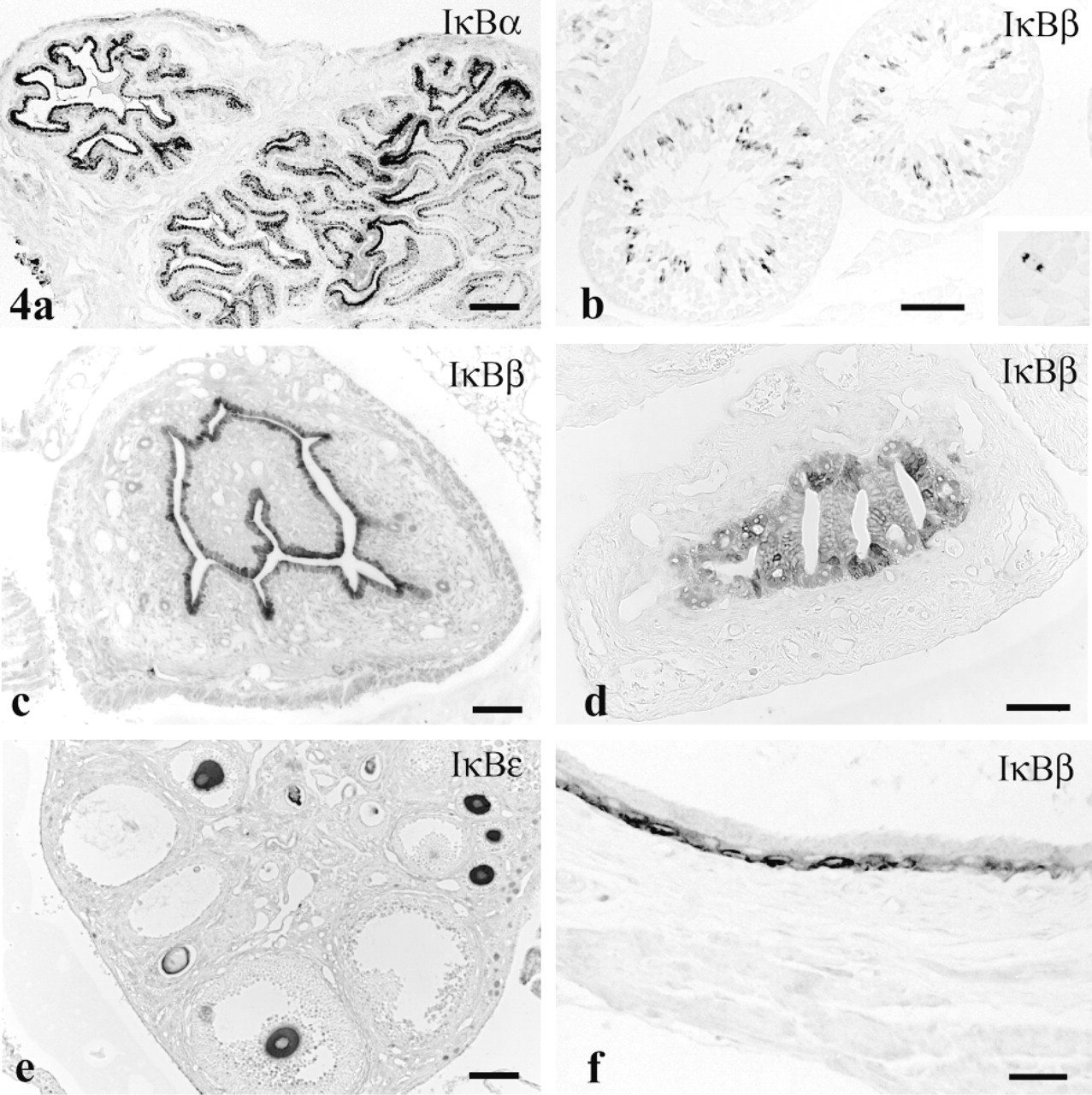
IκB isoforms in genitourinary tract. (
IκBα has been detected in Western blots prepared from a bladder cancer cell line (Sumitomo et al. 1999), but the relevance of this binding to constituents of normal bladder is open to question in view of the IκBβ immunostaining observed here only in basal cells of the transitional epithelium.
Prior evidence existed for activity of the NF-κB/IκB system in pancreatic β-cells based on production of nitric oxide synthase after NF-κB activation by IL-1β stimulation (Kwon et al. 1998). Data presented here testify further to the presence of this system in pancreatic islets but show IκBα and ∊ at high concentration only in peripheral islet cells, in contrast to a much lower staining intensity in central β-cells.
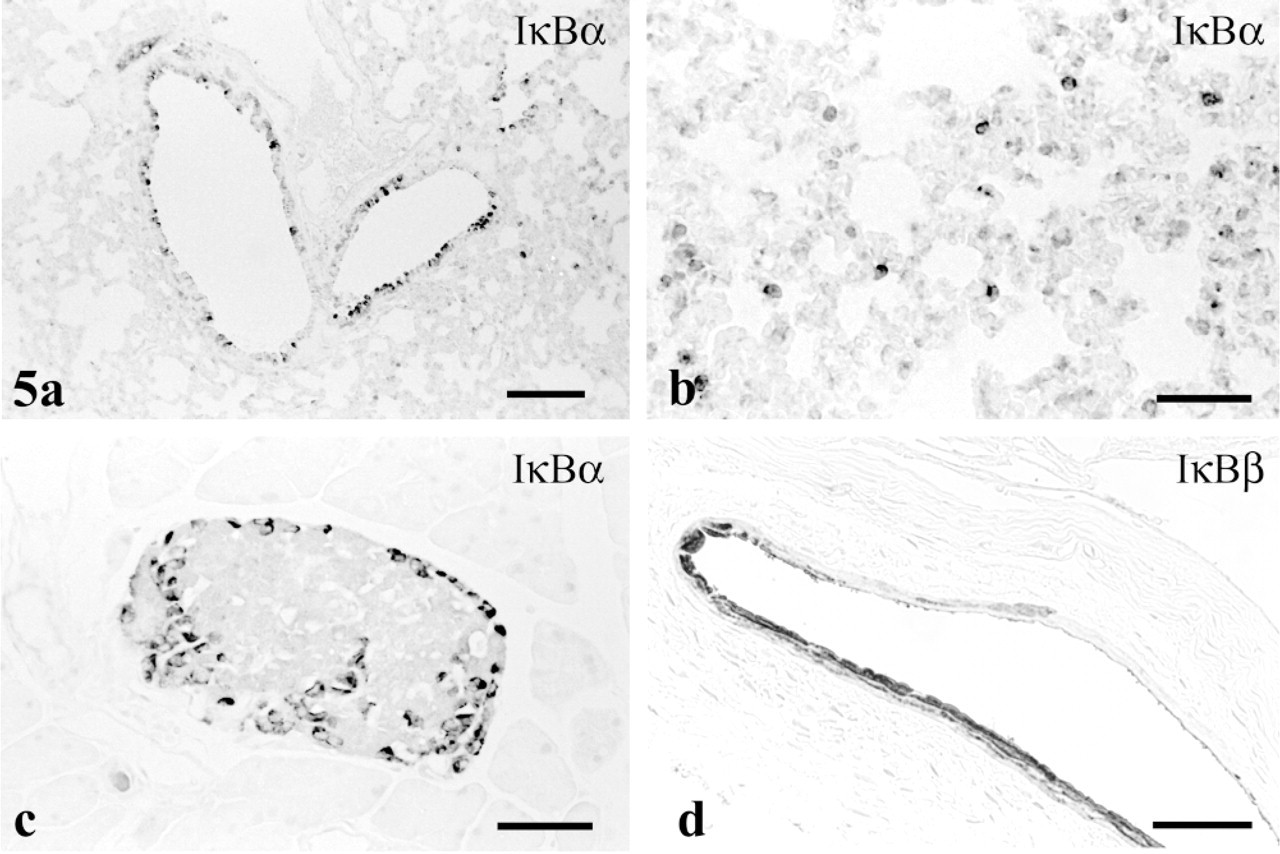
Staining for IκB isoforms in other sites. (
Conjunctival and endometrial adenocarcinoma cell lines possess NF-κB protein (Ray et al. 1997; Tsubota et al. 1999), testifying to the presence of the NF-κB/IκB complex in conjunctival and endometrial epithelia. Our present results concur with this observation and identify the inhibitory component in both cell types as IκBβ.
The perception of the NF-κB/IκB system as a complex that dissociates on activation, releasing NF-κB protein into the nucleus (Baeuerle and Baltimore 1988; Auphan et al. 1995; Scheinman et al. 1995; Faure et al. 1998), is supported by the present data localizing IκB proteins in the cytoplasm in some cells but in the nucleus or in both locations in others. Duodenal epithelium, mitotic spermatocytes, and deep gastric foveolar cells, for example, disclosed nuclear staining for IκBα- or β-isoforms. Although the nuclear distribution of IκB proteins is not fully understood, some recent work indicates that nuclear staining reveals newly synthesized IκBP (Thompson et al. 1995; SuYang et al. 1996). Activated NF-κB in the nucleus induces synthesis in cytoplasm of IκBP protein, which, lacking phosphorylation, binds to but fails to inhibit NF-κB. Translocation of this active NF-κB/IκB complex to the nucleus, where it prolongs transcriptional activity, could explain the nuclear immunostaining for IκBβ. On the other hand, newly produced IκBα migrates into the nucleus and transports NF-κB to the cytoplasm. This NF-κB/lKBα complex, which has the effect of decreasing NF-κB activity in the nucleus (Arenzana-Seisdedos et al. 1995, 1997) could account for the observed nuclear staining for IκBα.
NF-κB/IκB Proteins in Stomach
NF-κB has been detected in gastric mucous cells and parietal cells (Ishikawa et al. 1997; Rokutan et al. 1997). However, the IκB isoform(s) that inhibits NF-κB proteins in gastric mucosa has not been determined. Our present study provides further evidence for the NF-κB/IκB system in the gastric mucosa and identifies IκBβ as the major inhibitory isoform in the pit cells.
One proposed activity of the NF-κB/IκB system in pit cells concerns induction of IL-8, which is a chemotactic cytokine. H. pylori infection increases IL-8 immunoreactivity in pit cells (Crabtree et al. 1994). The infection presumably increases IL-8 production by activating NF-κB, because H. pylori infection has been shown to activate NF-κB in gastric biopsy specimens (Keates et al. 1997). increased IL-8 production in gastric cancer cell lines likewise depends on NF-κB activation (Aihara et al. 1997; Sharma et al. 1998), as may also be the case for increased IL-8 expression in stomach in vivo. The immunostaining data reported here suggest that activation of NF-κB in pit cells results from the dissociation of the IκBβ isoform. That the IκBα and IκBβ isoforms regulate NF-κB activity differently is suggested by their different kinetics (Tran et al. 1997).
IκB protein has not been demonstrated previously in gastric parietal cells, although mRNA for IκBα is present in the stomach (Rupec et al. 1999). The localization of IκBα in parietal cells points to a role for transcriptional regulation in this cell type through dissociation of IκBα from NF-κB. Phosphorylation of IκB proteins mediates this separation. information concerning the phosphokinases that phosphorylate IκB proteins is limited. It is known that casein kinase II phosphorylation of the C-terminus of IκBα and β occurs unrelated to inducible activation of NF-κB, whereas phosphorylation of the N-terminus of these isoforms by IκB kinases (IKKs) induces dissociation of IκB (Régnier et al. 1997; Zandi et al. 1997, 1998). Although mRNA transcripts for IKKs are detectable in gastric homogenates, the level of the enzyme is lower than in other IKK-rich tissues (Hu and Wang 1998) and their cellular distribution in stomach has not been established. Therefore, an as yet unidentified phosphokinase can be speculated to react with IκB protein in parietal cells. Gastric parietal cells are known to contain several phosphokinases, but whether these phosphorylate IκBα has not been determined.
A function proposed for the NF-κB/IκB system in parietal cells is the induction of prostaglandin endoperoxide synthase-2 (COX-2). H. pylori infection induces COX-2 expression in gastric parietal and pit cells (Sasaki et al. 1998; Sawaoka et al. 1998; To et al. 1998; McCarthy et al. 1999). Various stimuli that are known to activate NF-κB have been shown to induce COX-2 in several different cell types (Crofford et al. 1997; D'Acquisto et al. 1997; Jobin et al. 1998). IκBα may play a role in upregulating COX-2 in these systems and, by analogy, also in gastric parietal cells. Because H. pylori infection activates NF-κB, it is likely that this system mediates the increase of COX-2 in parietal cells of H. pylori-infected stomach and that the IκBα observed in these cells functions in this response.
Absorption of antiserum with homologous peptide abolished affinity for antibodies to IκBα and β, whereas absorption with the heterologous peptide did not, establishing specificity of the staining for these two IκB isoforms. The lack of lκB∊-specific peptide precluded absorption studies for this isoform. Although data reported here offer the first indication of the presence of IκBγ in stomach, the specificity of staining for this isoform remains inconclusive because of failure of blocking peptide to completely inhibit staining with anti-IκB.
Footnotes
Acknowledgments
Supported by Research Grant DC 00713 from the National institute on Deafness and Other Communication Disorders, National institutes of Health.
We thank Ms Deborah Brown for editorial and Ms Barbara Schmiedt for technical assistance.
