Abstract
Rare disorders related to tight junction (TJ) proteins have been associated with amelogenesis imperfecta. Pathogenic variants of CLDN10, encoding claudin-10b, a cation transport pore, cause the autosomal recessive HELIX syndrome (Hypohidrosis, Electrolyte imbalance, hypoLacrymia, Ichthyosis, Xerostomia). Patients exhibit salivary dysfunction and rapid enamel wear after tooth eruption. Since Cldn10 is expressed in the dental epithelium, this study explores the role of claudin-10b in amelogenesis. We analyzed amelogenesis in constitutive and conditional Cldn10 knockout (KO) murine models, comparing the findings to human HELIX enamel. First, analysis of constitutive Cldn10 knockout (KO) mice, which die within a few hours after birth, showed that claudin-10 is present at the plasma membrane of the stratum intermedium but not at the TJs during the secretory stage. Its absence altered gene expression related to ion transport and pH control, although without major disturbance in cell polarization or enamel matrix synthesis. Examination of later stages of amelogenesis in epithelium-targeted conditional Cldn10 KO mice showed that claudin-10 is present in the papillary layer at the maturation stage. In its absence, the pH of the enamel matrix was more basic during early maturation, suggesting that claudin-10 determines enamel matrix pH. However, at later stage of the maturation process, the pH was corrected and the resulting enamel did not show major structural or elemental alterations. These later findings were confirmed by exploring the enamel of Cldn10 KO transplanted tooth germs, which have developed in a controlled mineral environment. Nevertheless, higher contents of aluminum were detected in the enamel of transplanted germs and in human HELIX enamel, suggesting that claudin-10 deficiency may lead to a loss of enamel organ integrity. These data suggest that while salivary dysfunction is the main cause of enamel wear in HELIX, claudin-10 plays a direct role in amelogenesis by determining pH and enamel organ integrity.
Introduction
Tight junctions (TJs) form the backbone of the paracellular pathway for epithelial ion transport. Recently, the paracellular pathway has been shown to be highly ion specific, tightly regulated, and causally involved in human diseases (Meoli and Gunzel 2023). The specific set of claudins expressed at each TJ is uniquely tailored to the tissue and is responsible for the permeability and selectivity properties of the TJ. Several rare disorders resulting from pathogenic variants in a gene encoding a claudin (CLDN) have been reported, most of which result in disturbances of the ion homeostasis in the kidney (Meoli and Gunzel 2023). We have previously reported that patients with familial hypomagnesemia with hypercalciuria and nephrocalcinosis due to mutations in CLDN16 or CLDN19 displayed amelogenesis imperfecta (AI) (Bardet et al. 2016; Yamaguti et al. 2017). Recently, a novel claudinopathy caused by pathogenic variants in CLDN10 has been reported, resulting in the HELIX syndrome (Hypohidrosis, Electrolyte imbalance-renal loss of NaCl with secondary hyperaldosteronism and hypokalemia- hypoLacrymia, Ichthyosis, and Xerostomia) (Klar et al. 2017; Hadj-Rabia et al. 2018). HELIX patients complained of xerostomia (dry mouth) due to a 98% reduction in salivary flow rate compared with controls (Hadj-Rabia et al. 2018). Claudin-10 exists in 2 isoforms, resulting from 2 alternative exons, 1a and 1b (CLDN10A, CLDN10B) (Gunzel et al. 2009). CLDN10B encodes claudin-10b, an integral membrane-spanning protein of TJs expressed in the kidney thick ascending limb of Henle’s loop (TAL), skin, and salivary glands (Breiderhoff et al. 2012; Milatz 2019; He et al. 2024) and also in the tooth germ (Ohazama and Sharpe 2007; Chiba et al. 2020; Wang et al. 2020). We diagnosed early tooth wear in the 6 HELIX patients reported in Hadj-Rabia et al. (2018). The teeth formed normally, but the enamel eroded rapidly after tooth eruption, suggesting that the enamel wear was mainly due to salivary dysfunction (Obtel et al. 2022). Since claudin-10b has been shown to be expressed in the mouse enamel organ (Chiba et al. 2020), a direct effect of its impairment on amelogenesis cannot be excluded. Therefore, this study aims to investigate the role of claudin-10b in amelogenesis. To this purpose, we studied constitutive and conditional Cldn10 knockout (KO) mouse models, a kidney capsule tooth germ transplantation model (Jimenez-Rojo and Mitsiadis 2023), and human teeth (Obtel et al. 2022).
Materials and Methods
The detailed methodology is described in the Appendix Materials and Methods.
Results
Claudin-10 Is Expressed in the Stratum Intermedium at All Stages of Amelogenesis
Converging data have indicated that Cldn10/claudin-10 is expressed in the enamel organ at different locations and stages (Ohazama and Sharpe 2007; Chiba et al. 2020; Wang et al. 2020). In wild-type (WT) mice, both frontal and sagittal sections of the continuously growing incisor revealed claudin-10 labeling in the stratum intermedium (SI), throughout all stages of amelogenesis, from the cervical loop to the tip of the incisor at day 1 (P1) after birth (Fig. 1A, Appendix Fig. 1–2A). Similar labeling was observed in the secretory ameloblasts of the first molar germ (Appendix Fig. 2B). The specificity of claudin-10 immunostaining was validated using constitutive Cldn10 KO mice (Appendix Fig. 3A). Co-labeling with the TJ protein ZO-1 showed the expression of claudin-10 in the SI but not at the TJs (Fig. 1A, Appendix Fig. 1). To clarify this later point, we performed stimulated emission depletion microscopy and confirmed that the expression of claudin-10 was restricted to the SI without co-labeling with ZO-1 (Fig. 1B and Appendix Fig. 3). Co-labeling with connexin-43, a marker for gap junctions that are abundant in the SI (Liu et al. 2016), also revealed no co-labeling with claudin-10 (Appendix Fig. 4). Next, we performed claudin-10 immunogold labeling and observed a positive signal in the plasma membranes of the SI but not associated with TJs (Fig. 1C; Appendix Fig. 5). Next, we assessed claudin-10 expression in the dental epithelium at the maturation stage in adult mice and observed positive labeling of the papillary layer (Fig. 1D).
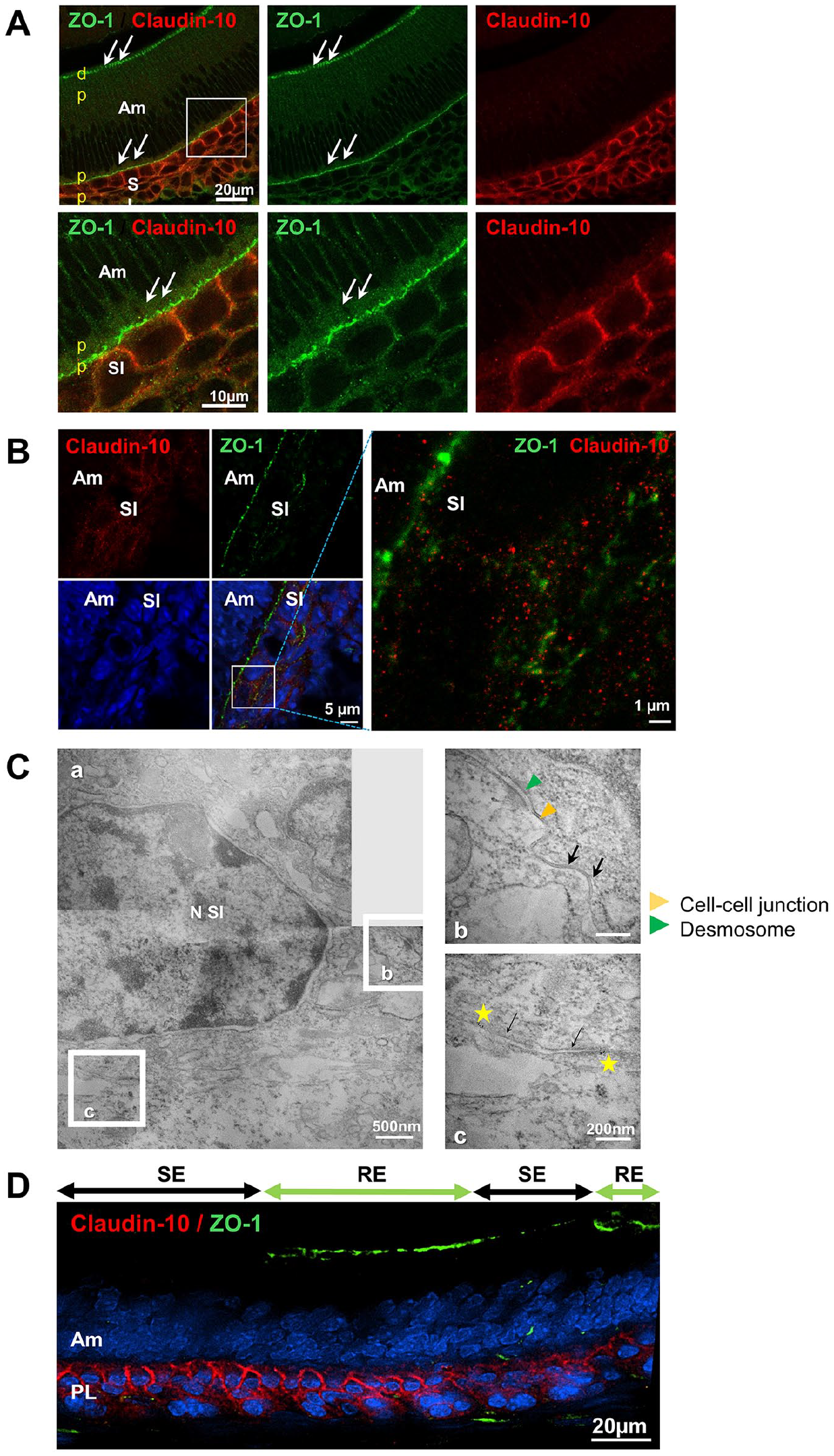
Claudin-10 expression profiling and protein identification in the wild-type (WT) murine incisor germ. (
The Ameloblast Layer Is Disturbed in Constitutive Cldn10 KO Pups
We next investigated constitutive Cldn10 KO mice to study the impact of claudin-10 deficiency on the early stages of amelogenesis, considering that pups die a few hours after birth (Breiderhoff et al. 2012) (Fig. 2). Staining of sagittal sections from the continuously growing incisor revealed disorganization in both the secretory ameloblast layer and the SI in constitutive Cldn10 KO mice (Fig. 2A, right panels). In contrast, littermate WT pups showed well-aligned ameloblasts (Fig. 2A, left panels). Amelogenin–positive forming enamel matrix was observed in both genotypes (Fig. 2A insets; Fig. 2B). Fluorescent labeling for markers of the cell cytoskeleton and the cell polarity confirmed that Cldn10 KO ameloblasts exhibited an irregular alignment but did not reveal loss of polarity or evident disorganization of the cortical actin compared with WT, with ZO-1–positive TJs at both poles (Fig. 2B; Appendix Fig. 6–7). We investigated the basal TJs at the ultrastructural scale as they are adjacent to the SI cells in which Cldn10 is deleted. Measurement of their interspace thickness revealed enlarged paracellular spaces between Cldn10 KO ameloblasts (P ≤ 0.001) (Fig. 2C). Transmission electron microscopy analysis of the SI showed no differences between KO and WT cells, with desmosomes and gap junctions between the cells (Fig. 2D).
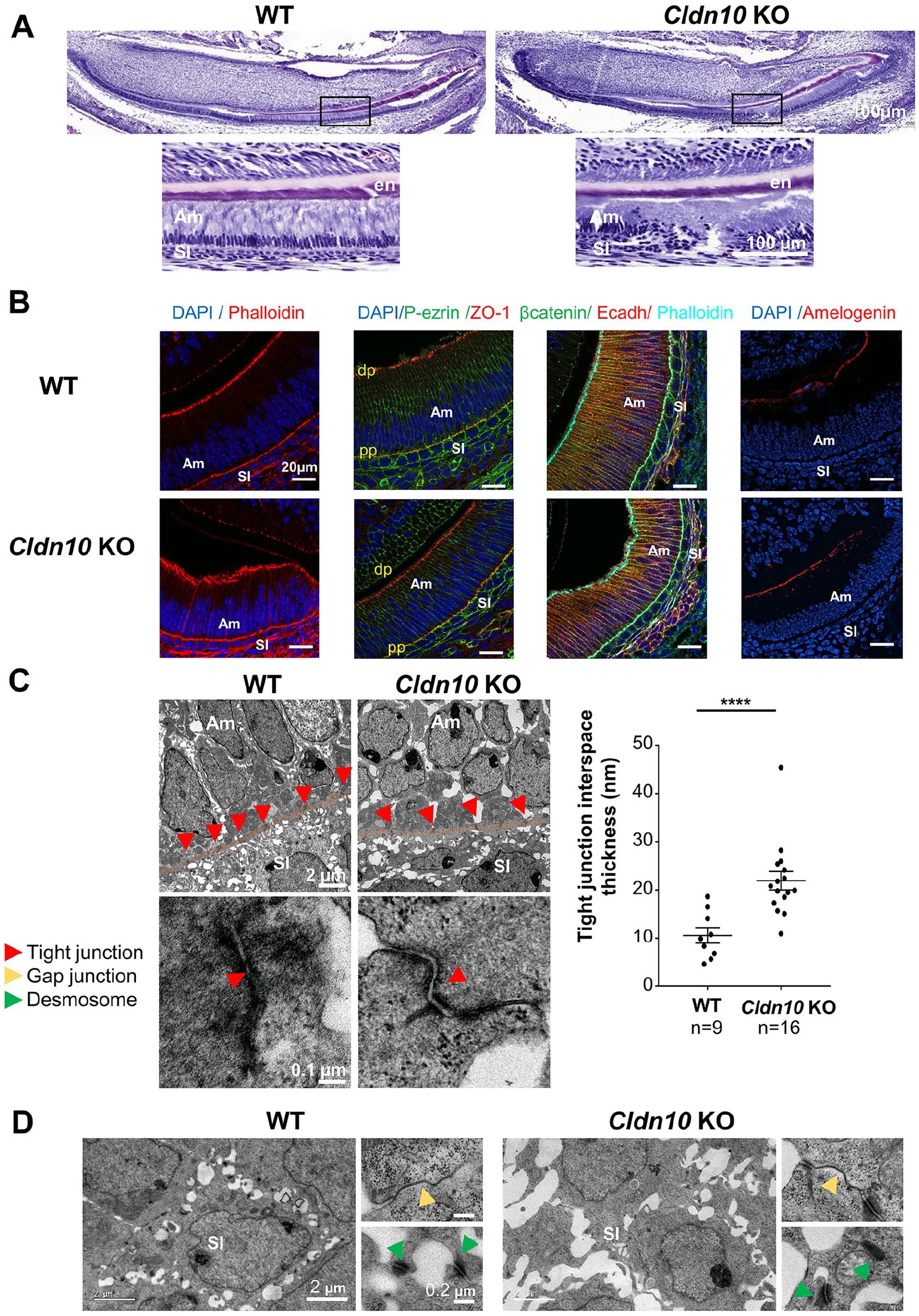
Analysis of the incisor enamel organ in P1 Cldn10 knockout (KO) mice. (
Genes Involved in Ion Transport Are Changed in the Cldn10 KO Enamel Organ
We next investigated the consequences of Cldn10 deletion on amelogenesis by performing reverse transcription quantitative polymerase chain reaction (RT-qPCR) analysis on laser micro-dissected ameloblasts and SI cells (Fig. 3A). In ameloblasts, we explored genes related to ion transport and showed that Slc4a4, which encodes NBCe1, a sodium bicarbonate cotransporter (Jalali et al. 2014; Bronckers 2017), was significantly decreased in KO ameloblasts. In contrast, the expressions of Slc20a2 and Slc27a4, which encode the inorganic phosphate transporter Pit2 (Merametdjian et al. 2018) and a fatty acid transporter, respectively, were increased in KO ameloblasts compared with WT (Fig. 3A, left panel). We investigated genes related to enamel synthesis by ameloblasts (Amelx, Ambn, Mmp20), which were unchanged, as confirmed at the protein level for amelogenin (Fig. 2B). We also explored the expression of Cldn1 and Cldn3, which encode 2 claudins essential for ameloblast TJ integrity (Bardet et al. 2017). We found a significant decrease in Cldn3 expression, confirmed at the protein level by immunofluorescence (Appendix Fig. 8).
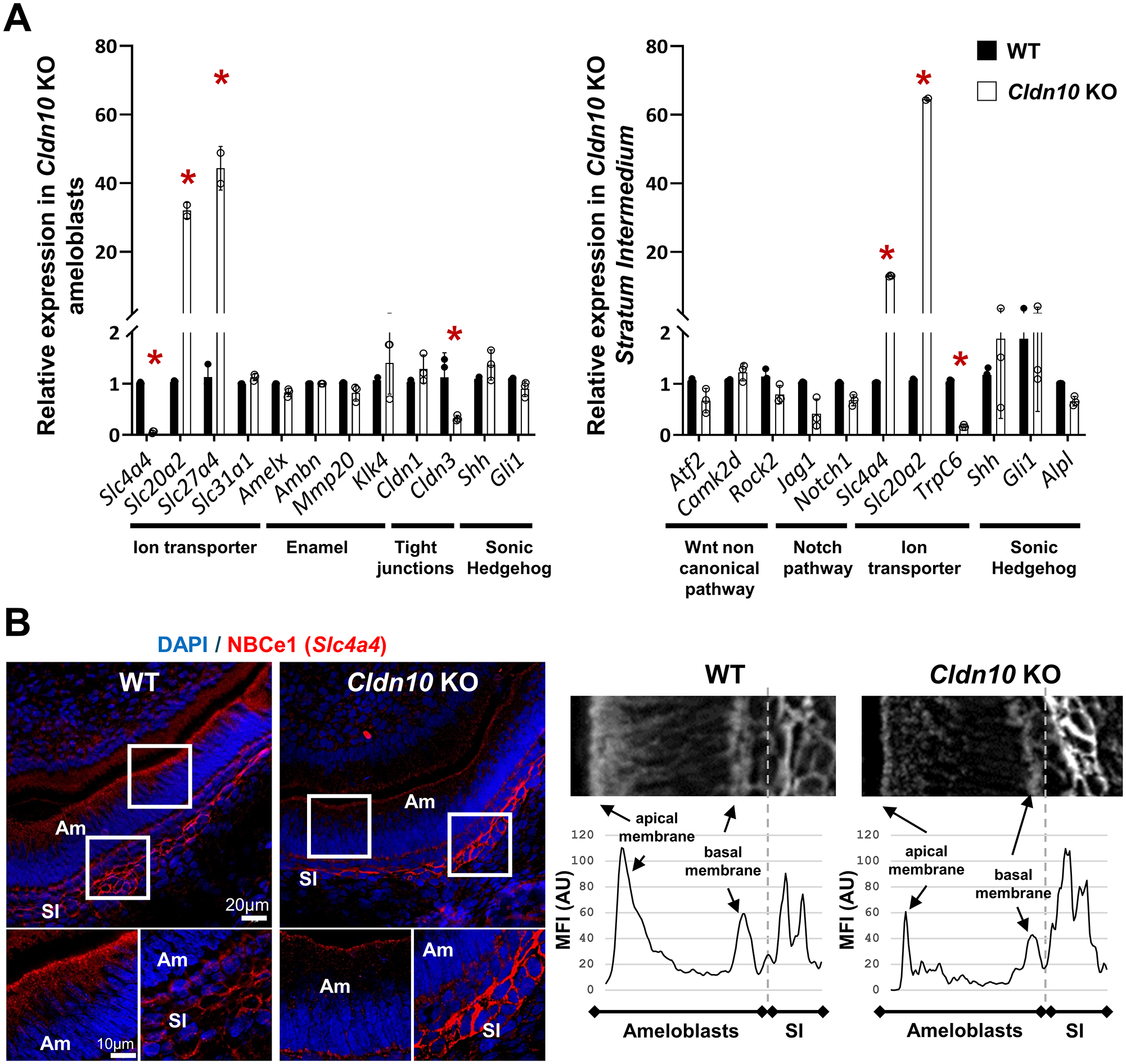
Consequences of Cldn10 deletion on amelogenesis in P1 Cldn10 knockout (KO) mice. (
In the SI, we showed that neither the gene expression related to the noncanonical WNT signaling pathway nor the Notch pathway were changed as a direct consequence of Cldn10 deletion in SI cells (Fig. 3A, right panel). We investigated the genes related to SI activity, showing that the expression of Slc4a4 and Slc20a2, which encode NBCe1 and Pit2, respectively, were significantly increased in Cldn10 KO SI cells, while TrpC6, which encodes the canonical transient receptor potential 6 that is important for enamel formation (Yang et al. 2017), was decreased. No changes were found for Alpl, a marker of the SI (Wang et al. 2020), or for the Sonic Hedgehog pathway. At the protein level, NBCe1 immunolabeling was stronger in the SI and weaker in ameloblasts of KO pups compared with WT (Fig. 3B), whereas alkaline phosphatase (ALP) activity was similar (Appendix Fig. 9).
Enamel Characterization in Epithelium-Targeted Adult Inducible Cldn10 KO Mice
Since (1) the death of Cldn10 KO pups after a few hours prevents the exploration of the later stages of amelogenesis and enamel and (2) claudin-10 is expressed in the SI throughout all the stages of amelogenesis and in the papillary layer at the maturation stage, we generated inducible Krt14 Cre+/−-Cldn10 iKO (Cre+) mice. This Cre model is used to target epithelial cells (Vasioukhin et al. 1999), and Keratin 14 is expressed in the dental epithelium (Li et al. 2015) (Appendix Fig. 10). At 2 mo after tamoxifen or vehicle injection induction, we examined the enamel of the lower continuously growing incisor, which formed after Cldn10 deletion, and of molars, which had formed prior to Cldn10 deletion. Visual examination showed no striking difference in color or morphology of the incisor between tamoxifen- and vehicle-injected females. However, tamoxifen-injected males exhibited marked white discolored incisors (Fig. 4A). In males, sagittal reconstructions of micro–computed tomography revealed a delayed onset of enamel mineralization, resulting in a significantly lower enamel volume in iKO incisors (Fig. 4B). Consistently, enamel thickness was significantly lower in tamoxifen-induced mice under the distal aspect of the second molar compared with vehicle (Fig. 4D). In addition, enamel density was significantly lower in tamoxifen-induced males compared with vehicle (Fig. 4B). Scanning electron microscopy (SEM) analysis showed well-formed enamel rods in both groups (Fig. 4C and Appendix Fig. 11A). Using energy-dispersive X-ray spectroscopy (EDX), no significant difference was detected in the calcium-to-phosphorus ratio (Appendix Fig. 11B). Nanoindentation showed no difference in Young’s modulus or microhardness between animals (Appendix Fig. 11C). Similar investigations were carried out on molars, revealing a significantly lower enamel volume in tamoxifen-induced male mice compared with vehicle with no change in density (Appendix Fig. 12). In contrast, no differences were found between Cre+ female mice (Appendix Fig. 13) or similarly treated Cre− animals (Appendix Fig. 14–15). We next evaluated the pH of enamel matrix at the maturation stage. GBHA staining showed 2 red bands in vehicle-treated mice, reflecting the position of smooth-ended maturation ameloblasts (Takano et al. 1982). These bands appeared broader in the Cldn10 iKO mice, suggesting a more basic pH (Fig. 4E). Consistently, pH measurement across these bands using BCECF fluorescence (Damkier et al. 2014) showed that the pH was more basic in the Cldn10 iKO enamel at the proximal part of the maturation zone (proximal band) (Fig. 4E, F). We next investigated the expression of carbonic anhydrase 2 (CA2), the ATP6V1b subunit of the V-ATPase proton pump, and NBCe1, known to be involved in pH regulation during maturation (Lacruz et al. 2017). ATP6V1b and CA2 labeling were increased at the apical border of ameloblasts as well as NBCe1 labeling in the papillary layer in tamoxifen-induced Cldn10 iKO animals compared with vehicle (Fig. 4G; Appendix Fig. 16–17).
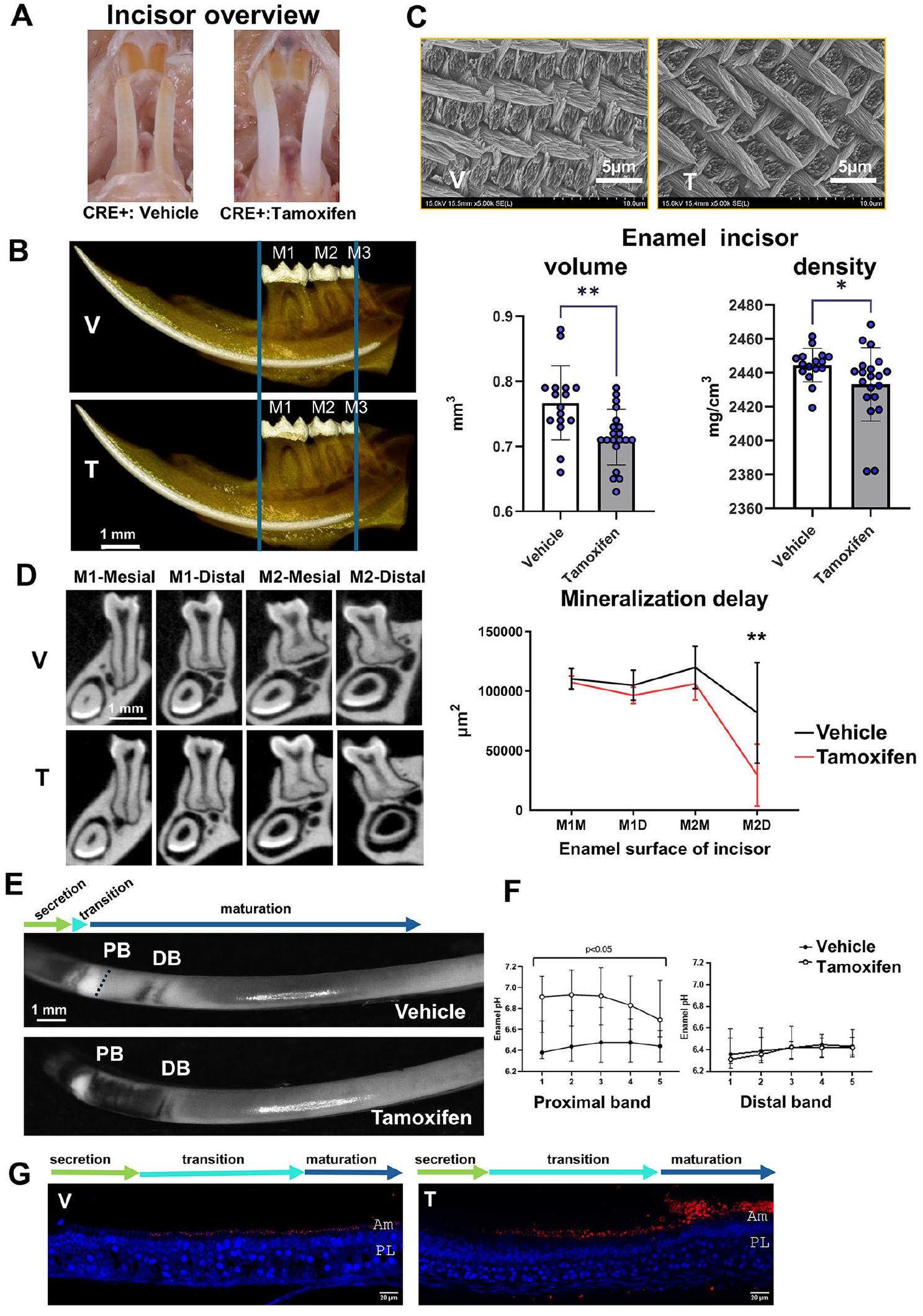
Dental phenotype of adult epithelium-specific inducible male Cldn10 KO mice. (
Enamel Characterization in Transplanted Cldn10 KO Molar Germs
We next transplanted molar germs of newborn Cldn10 KO pups into the kidney capsule of adult WT mice (Fig. 5A). In this model (Jimenez-Rojo and Mitsiadis 2023), the direct effect of the gene deletion on amelogenesis can be studied because the germs develop under normal mineral conditions. Three months upon implantation, a normal tooth with a periodontium formed in both genotypes (Fig. 5B). Enamel examination by SEM (Fig. 5C), EDX (Fig. 5D), and Raman (Fig. 5E) showed no difference between WT and KO germs, except for a slightly lower value of Cl content in the KO germs (Appendix Fig. 18). However, time-of-flight secondary ion mass spectrometry (TOF-SIMS), a highly sensitive method for chemical analysis and imaging of biological tissues (Van Nuffel and Brunelle 2022), revealed a higher level of aluminum species in KO germs compared with WT (×9.4). Ultimately, we assessed this finding in a HELIX tooth collected before tooth eruption (Obtel et al. 2022) and found a larger amount of this element in the enamel compared with a gender- and age-matched control tooth (×6.6) (Fig. 5F). Other elements, such as calcium, strontium, and magnesium, were increased in the murine KO germs compared with WT, but no difference was observed in the human enamel (Appendix Table 1).
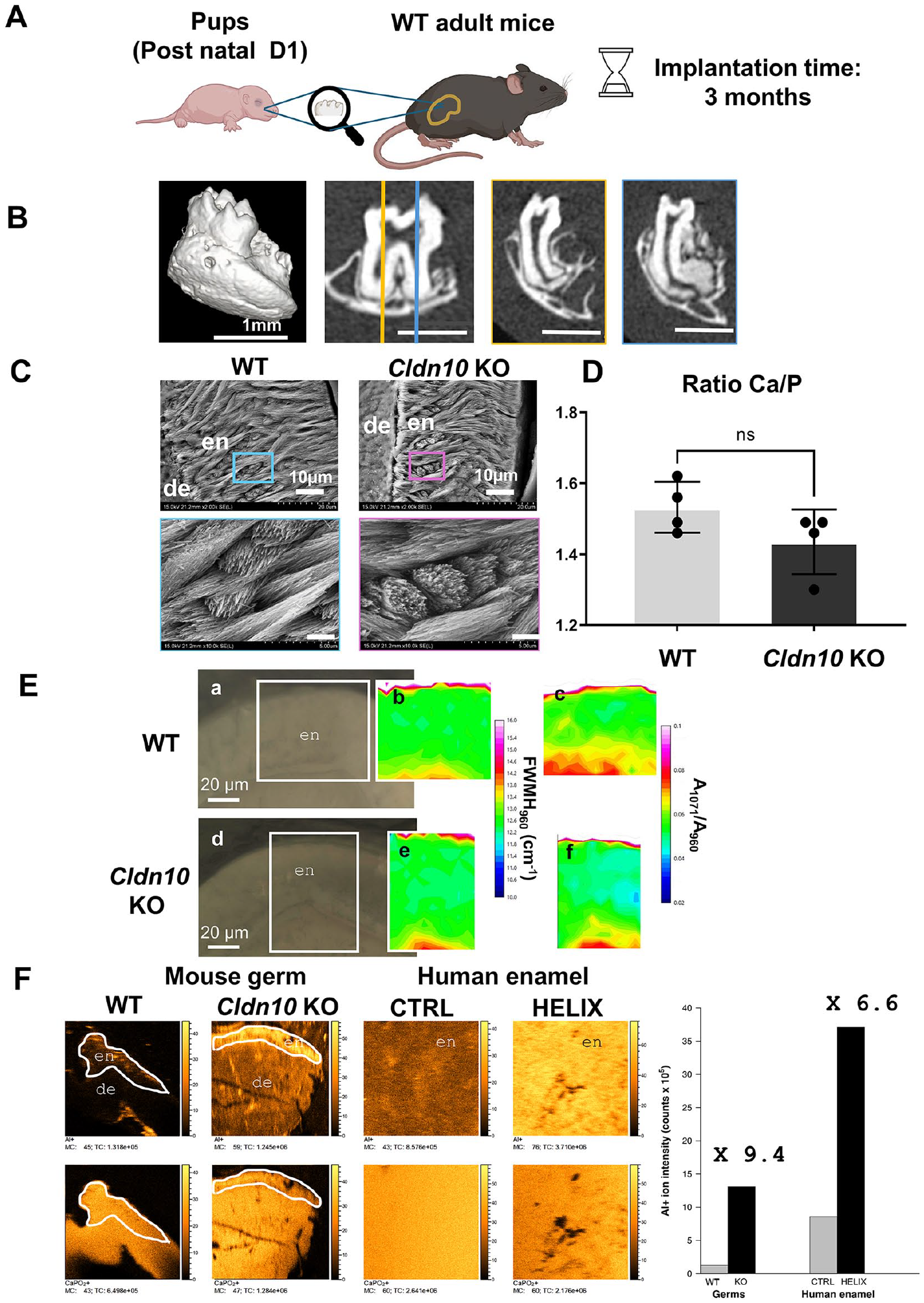
Analysis of the enamel structure of Cldn10 knockout (KO) molars after transplantation in the kidney capsule. (
Discussion
Accumulating data have shown that Cldn10/claudin-10 is expressed in the enamel organ since the early stages of amelogenesis, specifically in the SI, where it has been proposed to be a marker of dental epithelial cells (Ohazama and Sharpe 2007; Chiba et al. 2020; Wang et al. 2020). Our data demonstrate that claudin-10 is indeed located in the SI, in the cell membrane but not at the TJs. In the kidney, claudin-10 has been shown to be present in the TJs of a subset of medullary and cortical TAL cells, where it acts as a selective cation channel but also in other non–TJ-associated membrane domains (Prot-Bertoye et al. 2021; Meoli and Gunzel 2023). This latter observation is consistent with our findings in the SI; however, in both systems, the exact role of non–TJ-associated claudin-10 remains to be elucidated. Furthermore, our data show that claudin-10 is expressed in the papillary layer at the maturation stage.
At the secretion stage, the expression of several molecules involved in the ion transport and pH regulation of the enamel matrix was altered in both the Cldn10 KO SI cells and ameloblasts. Since claudin-10 is expressed in SI but not in ameloblasts, our data show that Cldn10 deletion has direct effects on the SI and indirect effects on ameloblasts. In addition, at the maturation stage, proteins involved in pH regulation were changed in ameloblasts and in the papillary layer, also indicating direct and indirect effects of Cldn10 deletion.
We found that the expression of Slc4a4, which encodes NBCe1, a bicarbonate sodium cotransporter, was significantly increased in the SI, whereas it was decreased in the ameloblasts, in KO mice, compared with WT. It was also increased in the papillary layer at the maturation stage in adult conditional mice. NBCe1 is important for enamel matrix pH regulation (Jalali et al. 2014), and pathogenic variants of SLC4A4 have been linked to AI in patients with proximal renal tubular acidosis (Kantaputra et al. 2022). Follow-up of incisor growth in adult male mice with tamoxifen-induced Cldn10 silencing showed that enamel volume was reduced, concomitantly with an increase of pH in the enamel matrix at the early stages of maturation. This finding is in sharp contrast with our report on claudin-16, a cation-selective intercellular pore located in the apical TJs of secretory ameloblasts, the expression of which is required for high paracellular permeability to Ca2+ and Mg2+ and for which deletion is associated with an acidic pH of enamel matrix (Bardet et al. 2016). These data suggest that both claudin-16 and claudin-10 may contribute to pH regulation during amelogenesis with opposite roles. Noteworthy, it has been shown that, in the TAL, claudin-10 expression at TJs almost never overlapped with that of claudin-16 and claudin-19 (Brideau et al. 2024).
The exploration of amelogenesis in constitutive Cldn10 KO mice was partially hampered by the premature death of the pups a couple of hours after birth (Breiderhoff et al. 2012). In these pups, the enamel organ appeared disorganized but the enamel matrix was still forming, and secretory ameloblasts maintained their organization (cytoskeleton, polarity, junctions). Consistent with this observation, the exploration of enamel in the molar germ transplantation model and in the tamoxifen-induced Cldn10 KO adult male mice did not reveal significant disturbances, as evidenced by a well-distinguishable decussating prism pattern and normal Ca/P ratio. However, in tamoxifen-induced KO male mice, the enamel of the continuously growing incisors was discolored, suggesting a loss of iron uptake, which may result in an enamel less resistant to posteruptive acidic challenges (Srot et al. 2024). It is noteworthy that HELIX syndrome affects both genders in a similar manner (Hadj-Rabia et al. 2018), and the differences observed here between male and female Cre mice may be due to the construction of this CreERT model (Vasioukhin et al. 1999).
It has been previously reported that Adam10 deletion in the dental epithelium induced downregulation of Notch1 expression, leading to loss of the SI layer and ALP activity and disorganization of ameloblasts (Mitsiadis et al. 2022). Here, our RT-qPCR analysis showed no change in the Notch pathway in the Cldn10 KO SI. In addition, ALP activity remained robust in Cldn10 KO SI. Therefore, in contrast to Adam10 (Mitsiadis et al. 2022), Cldn10 deletion does not significantly alter the Notch pathway, resulting in the maintenance of the SI layer. However, it seems relevant in the future to generate conditional models that target more specifically the various development stages of the dental epithelium (Yu and Klein 2020), for example, by generating a Notch1-CRE mouse for the SI (Mitsiadis et al. 2022).
We found a lower enamel volume in the molars of the activated Krt14-Cldn10 iKO male mice compared with the vehicle. Since Cldn10 deletion occurred long after molar eruption, this indicates exacerbated enamel wear due to salivary deficiency. The discoloration of the erupted enamel of the lower incisors in these mice may also result from hyposalivation. Our data are consistent with the decreased claudin-10 expression in NOD mice, a model of Sjögren’s syndrome, associated with hyposalivation (Scuron et al. 2019; He et al. 2024). Progressive enamel erosion has been shown in the molars of NOD mice (Tulek et al. 2020), which is consistent with our findings in the erupted enamel of the conditional KO mice and in HELIX patients (Hadj-Rabia et al. 2018).
Our recent investigation of a HELIX third molar, which had not yet erupted, revealed no significant abnormality in the enamel structure (Obtel et al. 2022), consistent with our present findings in the preeruptive incisor of the Cre-Lox model and in the transplanted germs. These data suggest that salivary dysfunction is the primary cause of the rapid enamel wear associated with HELIX.
TOF-SIMS imaging (Bich et al. 2013) revealed high aluminum content in the enamel of the HELIX tooth and Cldn10 KO germs that had mineralized upon implantation in WT mice. Although widely distributed in body fluids including blood, aluminum is considered an adventitious element present in food and water, with no known biological function (Exley and Mold 2015). In France, tap water contains about 16 µg/mL of aluminum (Le Bot et al. 2016), meaning that both mice and patients reported here were chronically exposed to aluminum during amelogenesis. Our data show that diffusion of aluminum through the enamel organ was greater in the Cldn10-deficient implanted germs and patient, suggesting a loss of enamel organ integrity in the absence of claudin-10.
Aluminum ions originating from the blood vessels adjacent to the outer layer of the enamel organ must go across its cell layers to reach the enamel matrix. Little is known about aluminum ion transport, which appears to occur by both transcellular and paracellular routes (Exley and Mold 2015). Enlargement of the basal TJs in Cldn10 KO ameloblasts together with the decreased expression of claudin-3, a sealing claudin, may be consistent with an increased paracellular transport of aluminum. Furthermore, the presence of aluminum in enamel, either integrated to the hydroxyapatite crystals or not (Chappard et al. 2016), may not increase its solubility (Kleber and Putt 1985) and is therefore unlikely to contribute to its fragility. It will be important to measure the levels of aluminum in other HELIX/Cldn10 KO tissues as, for example, the brain, where it has been shown to be neurotoxic (Kumar and Gill 2009).
In Cldn10 KO pups, we showed that the expression of NBCe1 was increased in the SI and decreased in the ameloblasts, suggesting a role of claudin-10 in acid and base transport. This finding is consistent with the basic pH measured in the proximal part (adjacent to secretion) of the tamoxifen-induced Cldn10 KO incisors at the maturation stage. However, we do not have an explanation for the overexpression of ATP6V1B1 at the apical membrane of iKO ameloblasts at the maturation stage. The exact mechanisms underlying this pH regulation need to be further investigated by functional studies in other organs that are easier to investigate than the enamel organ, such as the renal tissue, or by using acid and base transport inhibitors.
Conclusions
Our data demonstrate that claudin-10 is expressed in the cellular membrane of the SI where it contributes to the integrity of the enamel organ, directly in the SI and indirectly in ameloblasts. In its absence, enamel forms properly despite a basic pH of enamel matrix at early maturation stage, suggesting a role of claudin-10 in acid-base regulation of the enamel organ. Overall, xerostomia appears to be the main culprit for enamel wear observed in HELIX patients.
Author Contributions
T.N. Nguyen, C. Andrique, M. Requin, contributed to design, data acquisition, analysis, and interpretation, drafted and critically revised the manuscript; S. Ribes, N. Brouilly, A. Kiesow, A. Brunelle, A. Percot, S. Hadj-Rabia, C. Gaucher, F. Ramirez Rozzi, T. Coradin, contributed to design, data acquisition, analysis, and interpretation, critically revised the manuscript; J. Bouchet, contributed to design, data analysis and interpretation, drafted and critically revised the manuscript; N. Obtel, C. Torrens, A. Schmitt, T. Guilbert, contributed to design, data acquisition and analysis, critically revised the manuscript; L. Slimani, contributed to design, data acquisition, critically revised the manuscript; M. Morawietz, C. Bardet, D. Muller, contributed to design, data interpretation, critically revised the manuscript; A. Le Bivic, P. Houillier, contributed to conception, design, data acquisition, analysis, and interpretation, critically revised the manuscript; T. Breiderhoff, C. Chaussain, contributed to conception, design, data analysis and interpretation, drafted and critically revised the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work.
Supplemental Material
sj-docx-1-jdr-10.1177_00220345251349109 – Supplemental material for The Absence of Claudin-10 in the Enamel Organ Alters Its Integrity
Supplemental material, sj-docx-1-jdr-10.1177_00220345251349109 for The Absence of Claudin-10 in the Enamel Organ Alters Its Integrity by T.N. Nguyen, S. Ribes, C. Andrique, M. Requin, J. Bouchet, N. Obtel, L. Slimani, N. Brouilly, C. Torrens, A. Schmitt, T. Guilbert, M. Morawietz, A. Kiesow, A. Brunelle, A. Percot, S. Hadj-Rabia, C. Gaucher, A. Le Bivic, P. Houillier, C. Bardet, D. Muller, F. Ramirez Rozzi, T. Coradin, T. Breiderhoff and C. Chaussain in Journal of Dental Research
Footnotes
Acknowledgements
The authors thank Dr. Olivier Duverger (National Institutes of Health, Bethesda, MD, USA) for helpful discussion regarding the tooth germ transplantation model. We thank David Montero from the Institut des Matériaux de Paris Centre (IMPC FR2482) for servicing field emission gun SEM and EDX instrumentation and Sorbonne Université, CNRS, and C’Nano projects of the Région Ile-de-France for funding (Paris, France).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study received financial support from Université Paris Cité and from the T-JUST program (ANR-17CE14-0032-02). The Nghia Nguyen PhD was supported by the Ambassade de France in Vietnam. The IBDM imaging facility, member of the national infrastructure France-BioImaging is supported by the French National Research Agency (ANR-10-INBS-04 & ANR-10-INSB-04-01call “investissements d’Avenir”). MR and ALB salaries are supported by CNRS (Centre National de la Recherche Scientifique). IMAG’IC core facility (Institut Cochin) is part of the National Infrastructure France-BioImaging supported by the French National Research Agency (ANR-10-INBS-04). The electron microscopy experiments were performed on the PiCSL-FBI core facility (IBDM, AMU-Marseille), member of the France-BioImaging national research infrastructure (ANR-10-INBS-04) and member of the Marseille Imaging Institute, an Excellence Initiative of Aix Marseille University A*MIDEX, a French “Investissements d’Avenir” programme (AMX 19 IET 002).
ORCID iDs
Data Availability
The data that support the findings of this study are available on request from the corresponding author.
A supplemental appendix to this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
