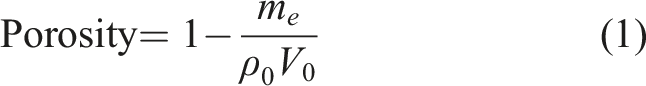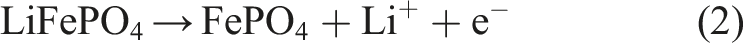Abstract
In this paper, an innovative approach was presented to manufacture woven carbon fabric cathodes via high-pressure spray to coat LiFePO4 (LFP), acetylene black (AB) and polyvinylidene fluoride (PVDF) onto carbon fibers. The fabrics were desized to improve conductivity prior to coating. Fourier-transform infrared spectroscopy (FTIR) and scanning electron microscopy (SEM) were adopted to ensure that the selected process parameters could achieve complete removal of the polymer sizing and dispersion of the LFP particles on both the spray surface and inner architecture of the fabrics. The subsequent electrochemical characterization showed that the fabric cathodes could, on average, produce excellent specific capacities of over 110 mAh/g and areal capacities of over 3.0 mAh/cm2 with good rate performance. With 98% efficiency and 0.97 retention after 100 cycles, the cathodes also depicted superb long-cycle performance, meaning that the coating with electrochemical activity could adhere firmly to the fiber surface.
Introduction
Over the last decade, the interest in developing electric vehicles (EVs) has increased rapidly to reduce greenhouse gas emissions and achieve a more sustainable society. Up-to-date, the most promising energy source for EVs is lithium-ion batteries, which possess superior properties, such as high energy density, exceptional performance, longevity and versatility. 1 Currently, the transportation sector is actively developing and promoting EVs with zero-emission to replace their fossil fuel-powered counterparts. However, the limited driving range of EVs has significantly restricts their application. To tackle this issue, it is essential to increase capacity or areal capacity of the batteries without expanding its physical size or weight.
A lithium-ion battery is made up with three major components: cathode/current collector, electrolyte/separator, and anode/current collector. Adopting novel electrodes/current collectors with a 3D configuration is a viable option for increasing the areal capacity. In this regard, carbon fibers have been proposed to serve as electrode materials in lithium-ion battery2,3 due to their high conductivity and capability to form fabrics with spatial structure. On the other hand, carbon fibers have great potential to be utilized for construction of lithium-ion structural batteries as they can be reliably embedded in resin matrix to form composites with high mechanical performance and excellent geometric stability.4,5
In the past few years, a large number of studies have been completed on directly using unidirectional (UD) carbon fibers as electrodes/current collectors.6–10 To further improve the electrochemical properties and promote the mechanical performance, some additional studies have been conducted to modify pure carbon fibers. Wang et al. 11 utilized cobalt fluoride and non-woven carbon nanotube fabrics to produce cathodes with stable cycling performance and benign mechanical properties, but the raw materials are very expensive. Johannisson et al. 12 introduced solid battery electrolyte to combine with UD carbon fibers to produce lithium-ion batteries, but the electrochemical performance is limited. Hagberg et al. 13 deposited LiFePO4 (LFP), carbon black and polyvinylidene fluoride (PVDF) onto UD carbon fibers via electrophoretic deposition (EPD) to produce structural cathodes. Although several studies have proved that UD carbon fibers can be integrated into lithium-ion batteries, some technical issues still need to be tackled. For example, UD carbon fibers are hard to maintain the original shape under external loading and have mechanical reinforcement along only one direction.
To overcome the limitations of UD carbon fibers, a growing number of researchers begin to study woven carbon fabrics recently due to their advantages in reinforcement along different directions, superb formability, containing of inner area, etc. Javaid and Ali 14 directly employed woven carbon fabrics as anode without coating any other materials and successfully bulid structure batteries. Moyer et al. 15 utilized blade-cast method to deposit LFP on woven carbon fabrics to manufacture cathodes, which were then fabricated into pouch-free structural batteries using the vacuum bag-assisted technique. The obtained batteries demonstrated satisfactory Young’s moduli and tensile strength and, but their electrochemical performance was unstable. Recently, Park et al. 16 investigated the characteristics of woven carbon fabrics as cathodes/current collectors, revealing their excellent electrochemical and mechanical properties, but also difficulty in accurate control in manufacturing of these electrodes, leading to low successful rate of the production. Besides the most popular lithium-ion batteries, woven carbon fabrics have also been employed to fabricate zinc-ion batteries17,18 by using conventional blade-cast or EPD method to coat the suitable active materials for energy storage.
According to the previous literature review, most existing manufacturing methods for woven carbon fiber electrodes to coat active electrode materials onto carbon fibers are of the contact type, but contactless coating should theoretically be more suitable for carbon fabrics with rough surfaces and internal structures. Some typical contactless techniques have been investigated to transfer active electrode particles onto surfaces of carbon fibers include pulsed laser deposition, 19 sputtering, 20 chemical vapor deposition 21 and atomic layer deposition. 22 These non-physical contactless deposition methods, however, are typically difficult to set up and require expensive facilities. As improvement, an innovative method, which is inspired by the spray pyrolysis technique to deposit TiO2, 23 CuO, 24 SnO2, 25 etc. on conducting substrates, has been developed by our team to enable contactless, easy-to-operate and controllable coating for production of carbon fabric cathodes with reliable performance. This approach can deposit electrode particles into woven carbon fabrics via a high-pressure spray. With optimal process parameters, active particles can be stored not only on surfaces but also inside the carbon fabrics with 3D inner architecture, leading to the achievement of higher areal capacity. In preparation of the raw fabric materials before actual spraying, thermal processing should be conducted to ensure close bonding between active particles and carbon fibers, maximizing conductivity of the produced electrodes.
After development of the novel high-pressure spray method, it was utilized to manufacture actual carbon fabric cathodes, the morphology of which was examined using a scanning electron microscope (SEM). Furthermore, the cathodes’ specific capacity, areal capacity, rate performance, cycle life, efficiency and retention were all tested experimentally. The results revealed that the obtained carbon fabric cathodes could achieve specific capacity of over 110 mAh/g, high areal capacity of over 3.0 mAh/cm2 and good rate performance. Cycling performance of the cathodes was also stable and excellent, with efficiency and retention remaining at 98% and 0.97, respectively, after 100 cycles. Setup and process of the high-pressure spray manufacturing approach, and experimental characterization procedure and results of the obtained carbon fabric cathodes are to be elaborated in the following sections.
Material and methods
The dry woven carbon fabric with 2 × 2 twill pattern demonstrated in Figure 1 was utilized in the research work presented in this paper. Details about preprocessing and electrode particle deposition of the raw fabric material are to be discussed below. Top surface of the selected dry woven carbon fabric with 2 × 2 twill pattern.
Desizing of the woven carbon fabric
Carbon fibers in the selected commercial-grade dry fabrics were initially coated with thin and homogeneous polymer sizing, which could significantly impair effective bonding between active particles and carbon fibers, and conductivity of fibers. To tackle this issue, thermal processing was conducted to desize the fabrics. Specifically, the selected fabrics were heated to 500°C for over 20 min in a muffle furnace. Afterwards, the processed fabrics were fully immersed in acetone and vibrated in an ultrasonic cleaner for over 1 h to completely wash away any residual of the decomposed sizing materials.
Production of the fabric electrodes
The electrode slurries to be coated into the desized fabrics contained LiFePO4 (LFP, MTI Corp.) powders as active cathode particles, acetylene black (AB, MTI Corp.) as conductive additives and polyvinylidene fluoride (PVDF, Dieckmann) as the binder. Weight ratio of the LFP/AB/PVDF was set to be 8:1:1, and they were mixed in N-Methyl-2-pyrrolidinone (NMP 99.5%, Aladdin) and ground in an agate mortar for 15 mins, then stirred in a magnetic stirrer for 24 h at 600 rpm to finish preparation of the electrode slurries. In this paper, the weight of the LFP/AB/PVDF was determined by a microbalance as 0.56 g, 0.07 g, 0.07 g respectively; and the applied NMP was 11 mL. The slurries should be of little viscosity to enhance their flow ability to make sure that they can be easily sprayed out. Next, the fresh slurries were immediately coated into the carbon fabrics to avoid recrystallization of the active materials after long time storage, which could block the spray nozzle. Top surface of the selected woven fabric depicted in Figure 1 is too rough for the conventional blade cast coating technique to deposit the active slurries. In addition, the blade cast coating cannot provide adequate driving force for the slurries to flow inside the fabrics. As alternative, high-pressure spray was applied to efficiently coat the slurries both onto surface and into inner structure of the woven carbon fabric. Figure 2(a) presents a schematic of the high-pressure spray setup and Figure 2(b) demonstrates a schematic of the ideal cross-section in the woven carbon fabric cathodes coated with LFP. The spray gun was purchased from SNDME with the model number of 134. The dry fabric with the size of 3 cm × 3 cm was mounted on a heated substrate to evaporate solvent of the slurries during coating so as to realize multiple coating. The spray gun that sent out the slurries was attached to a bottle of compressed Nitrogen for high pressure. The spray parameters were experimentally optimized in the aspects of substrate temperature (250°C), distance between substrate and spray nozzle (4.5 cm), nozzle diameter (0.3 mm) and pressure of the compressed Nitrogen (4.5 bar) to achieve homogeneous, uniform and reproducible coating. With the spray process, the dry fabric was coated with a LFP loading at 0.27 g (30 mg/cm2), that is, 48% of the total amount of the slurries were deposited on the carbon fabric. Afterwards, the electrodes were dried in a vacuum oven for over 24 h at 100°C. In addition, the porosity of the electrodes was calculated at around 37% based on equation (1) when they were moved out from the vacuum oven: Schematics of (a) setup of the innovative high-pressure spray process, and (b) ideal cross-section of the carbon fabric cathodes with LFP coating.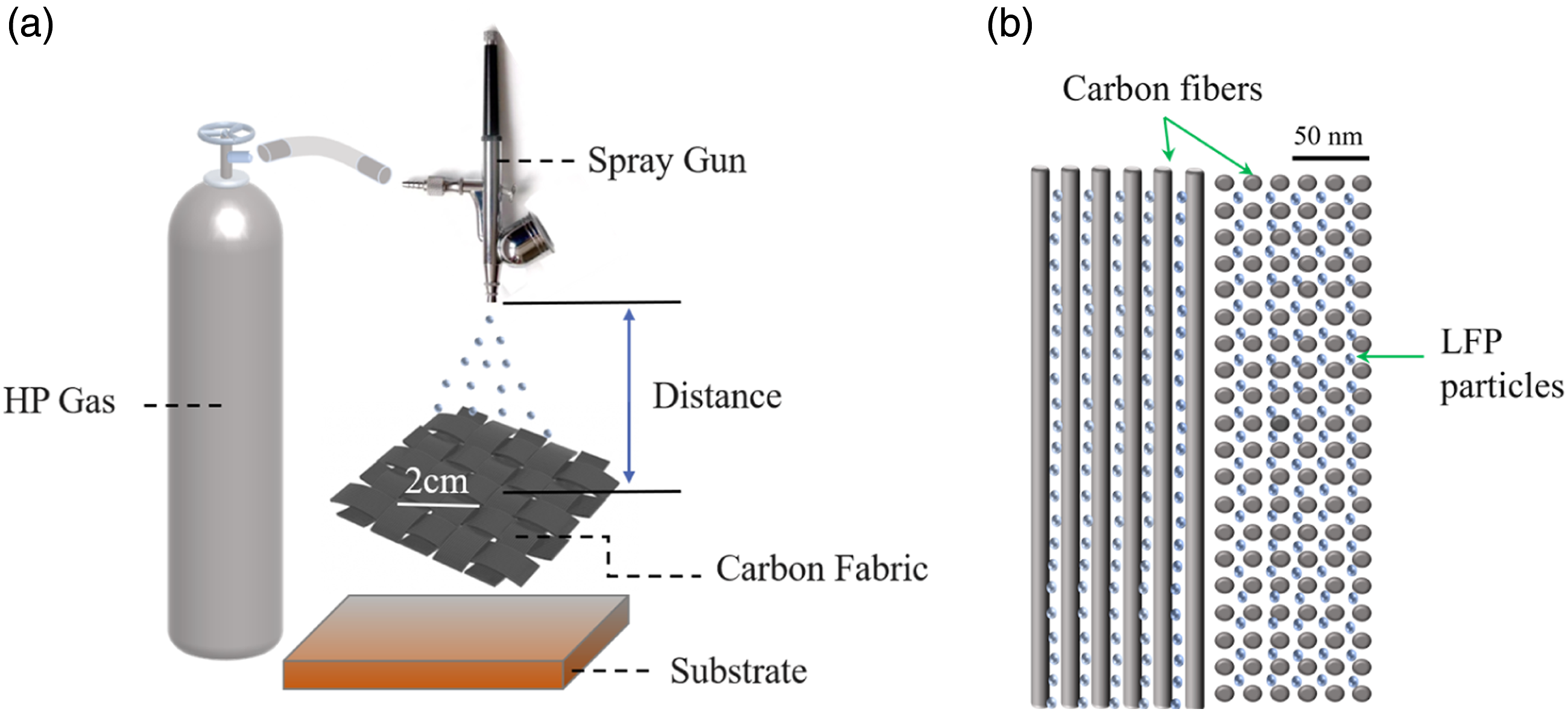
Preparation of half-cell batteries
The carbon fabric cathodes produced using the approach presented in Figure 2 should be assembled into half-cell batteries for testing in order to characterize their electrochemical properties. Figure 3 displays a schematic of the battery’s assembly. The prepared cathodes were cut into circle disks and placed in coin cells (CR2025, Canrd) in an Argon atmosphere glove box (Lab 2000, Etelux) with both H2O and O2 concentration under 1 ppm. Metallic lithium foils were selected as the opposite electrodes and Celgard 2025 membrane made of polypropylene was utilized as the separator. The electrolyte was made of 1M LiPF6 conductive salt dissolved in a 1:1 vol% solution of ethylene carbonate (EC) and diethylene carbonate (DEC). The electrodes, lithium foil and separator with diameters of 15 mm, 19 mm and 12 mm, respectively, were all completely impregnated with the liquid electrolyte during the assembly. Schematic of the battery assembly.
Characterization of constituents and electrochemical property of the electrodes
SEM (JSM-7800F field emission scanning electron microscope, JEOL) and Fourier-transform infrared spectroscopy (FTIR) (NicoletiS50 R) were first employed to assess constituents attached to the desized carbon fibers after each manufacturing step and thoroughly investigate effectiveness of the steps in the newly established approach to produce woven carbon fabric cathodes. SEM could also capture fiber surface morphology in the fabric cathode samples. Afterward, cyclic voltammetry (CV) and electrochemical impedance spectroscopy (EIS) were carried out for the half-cell battery samples using an electrochemical workstation (CHI 760E). Finally, the half cells went through galvanostatic discharging/charging tests using a LAND CT3002A battery test system (Wuhan LAND electronic Co., Ltd). All these electrochemical experiments were conducted between 2.8 and 4.2 versus Li/Li + at room temperature.
Results and discussion
Characteristics of inner architecture of the woven carbon fabric
SEM images of inner architecture of the woven carbon fabrics are presented in Figure 4. Figure 4(a) includes a typical cross-section of the woven fabric containing both longitudinal and transverse fibers at ×250 magnification. Figure 4(b) and (c) are SEM images at regions A and B with higher magnification (×500) to provide clearer views of fiber alignment. The 3D structure within woven fabrics will enable storage of active cathode materials in heavy loading, significantly increasing areal capacity of the fabric cathodes. Furthermore, porous structure of the woven carbon fabrics can effectively mitigate the volume change of the electrodes caused by the insertion or extraction of Li + ions, improving the reversibility of the fabric cathodes during charging and discharging. However, as can be observed in Figure 1, top surface of this woven carbon fabric is rough and cannot be processed using the conventional blade cast coating method. To tackle this issue, contactless coating was achieved in the newly developed manufacturing approach using high-pressure gas. The high-pressure gas can bring additional benefit to drive LFP particles not only on top surface of but also inside the woven fabrics. SEM images of cross-section of the woven carbon fabric: (a) ×250 magnification of the cross-section, (b) ×500 magnification of the left region A, (c) ×500 magnification of the right region B.
Effectiveness of desizing under elevated temperature
As discussed previously, it is essential to thermally process the commercial-grade dry carbon fabrics to remove polymer sizing on carbon fibers before coating of the active particles. Effectiveness of this sizing removal process was verified using FTIR and SEM. The FTIR results plotted in Figure 5(a), including FTIR spectrums of the original and desized woven carbon fabrics, illustrate that FTIR spectrum peaks for O-H and carbonyl groups existing in the polymer sizing of the original fabrics vanish in the desized samples after the thermal process, indicating that all sizing had been eliminated completely. Moreover, SEM images of fibers in both original and desized carbon fabrics, as presented in Figure 5(b) and (c), also demonstrate that the sizing was successfully removed: the initial fibers in Figure 5(b) are surrounded by sizing, while the desized fibers shown in Figure 5(c) are clean. Both the FRIR and SEM results prove that the polymer sizing on the carbon fibers was successfully removed. (a) FTIR test results of both original and desized carbon fabrics, (b) SEM image of one fiber in the original carbon fabric, and (c) SEM image of fibers in the desized fabrics.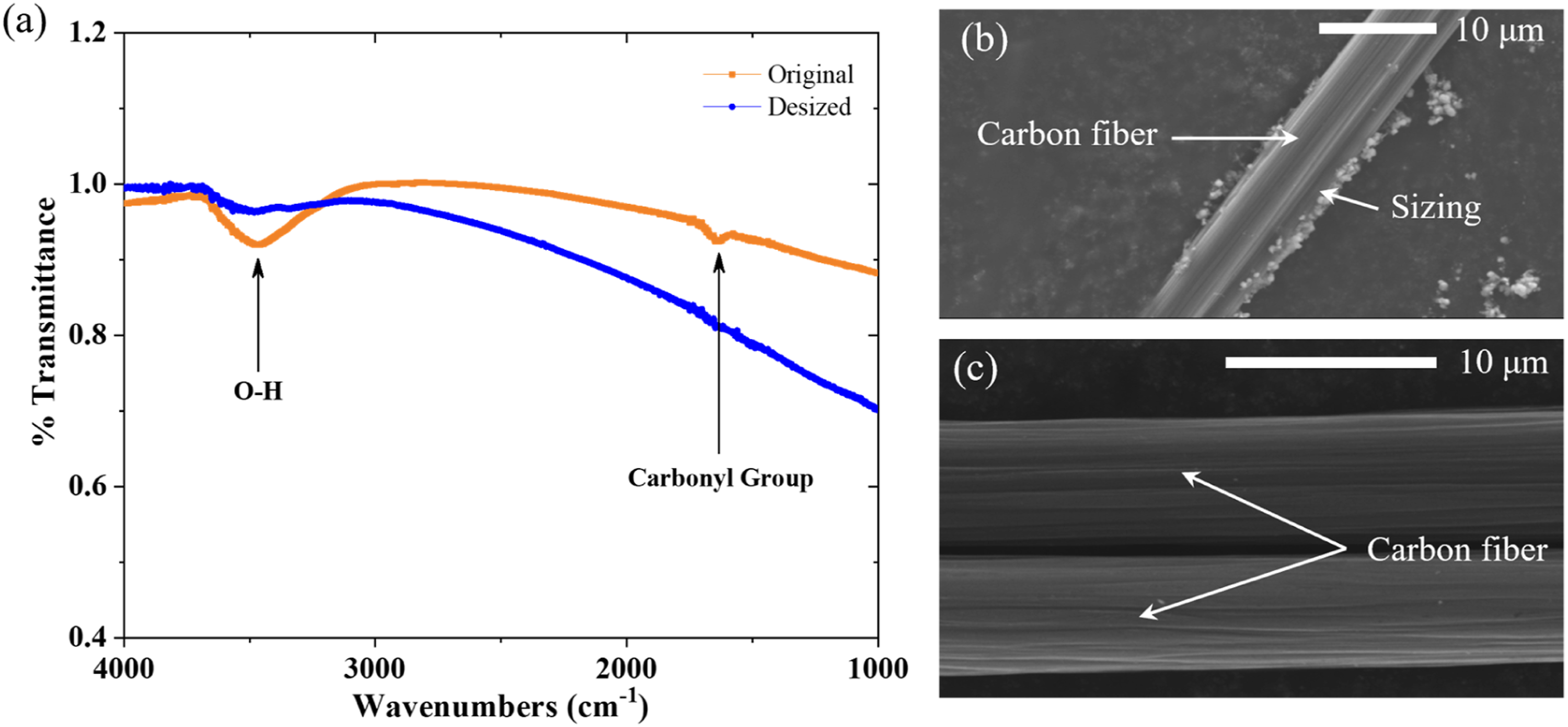
Deposition of LFP particles in the woven carbon fabric
SEM images of the top surface of one piece of woven carbon fabric coated with LFP using the new high-pressure spray approach are presented in Figure 6. After dozens of times of repeated spray, a well-dispersed coating can be obtained, as illustrated in Figure 6(a). The higher magnification SEM image in Figure 6(b) and EDS maps in Figure 6(c)–(e) demonstrates the uniformly distributed LFP particles throughout the coating. It should be noted that the substrate was ought to be heated to over boiling point (202°C) of the NMP before starting of the spray, so the slurries could be baked immediately and would not flow away, resulting in continuous and high-quality coating. During the initial spray, the LFP particles could penetrate the fibrous structure through the gaps between individual fibers. As the particles gradually accumulated after repeated spray to fill the gaps, top surface of the fabric became relatively smooth as demonstrated in Figure 6(a) compared to that of initial fabric in Figure 1. Due to potential variation in parameters of the spray process, the coating thickness may not be non-uniform throughout one piece of woven with difference from one to several microns, which can lead to uneven current distribution, impairing electrochemical performance and causing capacity loss. Therefore, it is essential to compact the fabric electrodes before battery assembly, and the compaction pressure was experimentally optimized to be 2 MPa for the presented case. (a) SEM images of ×100 magnification of top surface of one woven fabric electrode; (b) ×10,000 magnification of the selected region in (a); EDS maps of SEM image (b) about the distribution of (c) Fe and (d) P related the presence of LFP; and (e) F related to the presence of PVDF.
Figure 7 includes SEM images and EDS maps of cross-section area of one piece of woven carbon fabric after the high-pressure spray coating. LFP particles inside the cross-section area in Figure 7(a) indicate that they could efficiently penetrate into the woven fabrics under high pressure, enabling complete utilization of the 3D architecture and significantly increasing areal capacity of the fabric electrodes. Figure 7(b)–(d) present the EDS maps of Figure 7(a), which can further verify successful penetration. Although Figure 7 also demonstrates that some carbon fibers may be over-coated, potentially resulting in loss of electrochemical contact and capacity, the electrochemical characterization results to be elaborated in following sections will prove that this effect is actually negligible. (a) Cross-section area of the fabric electrode under ×1000 magnification; EDS maps of the cross-section area in (a) about the distributions of (b) Fe and (c) P related to the presence of LFP; and (d) F related to the presence of PVDF.
Electrochemical characterization
Cyclic voltammetry
Cyclic voltammetry (CV) is a paramount electrochemical tool to investigate reduction and oxidation of molecular species.
26
Figure 8 presents typical CV traces of the obtained fabric cathodes. The x-axis represents the applied potential, while the y-axis means the resulted current. The traces contain an arrow in Figure 8 to indicate the start and sweep direction of the scanning potential applied. For the traces in Figure 8, the scanning rate (v) was set as 0.2 mV/s, meaning that the potential varied linearly at the speed of 0.2 mV per second. The two cycles plotted in Figure 8(a) and the corresponding faradaic redox couple remain stable over successive CV sweeps. Magnitude of the peak current can demonstrate the delamination of the electrode material LFP,
27
and the decline in peak current is also associated with decreased electrons in the redox processes happened in the electrode materials.
28
As the peak current in Figure 8(a) reduces by less than 5% from the 1st to 2nd cycles, little materials have been delaminated, and the electrons in redox processes and the electroactive surface area maintain well during charging-discharging cycles of the fabric electrodes. Cyclic voltammograms of the produced fabric cathodes at the scanning rate of 2 mV/s: (a) the first and second cycles, (b) measurement of peak-heights for the cathodic (i
pc
) and anodic (i
pa
) currents.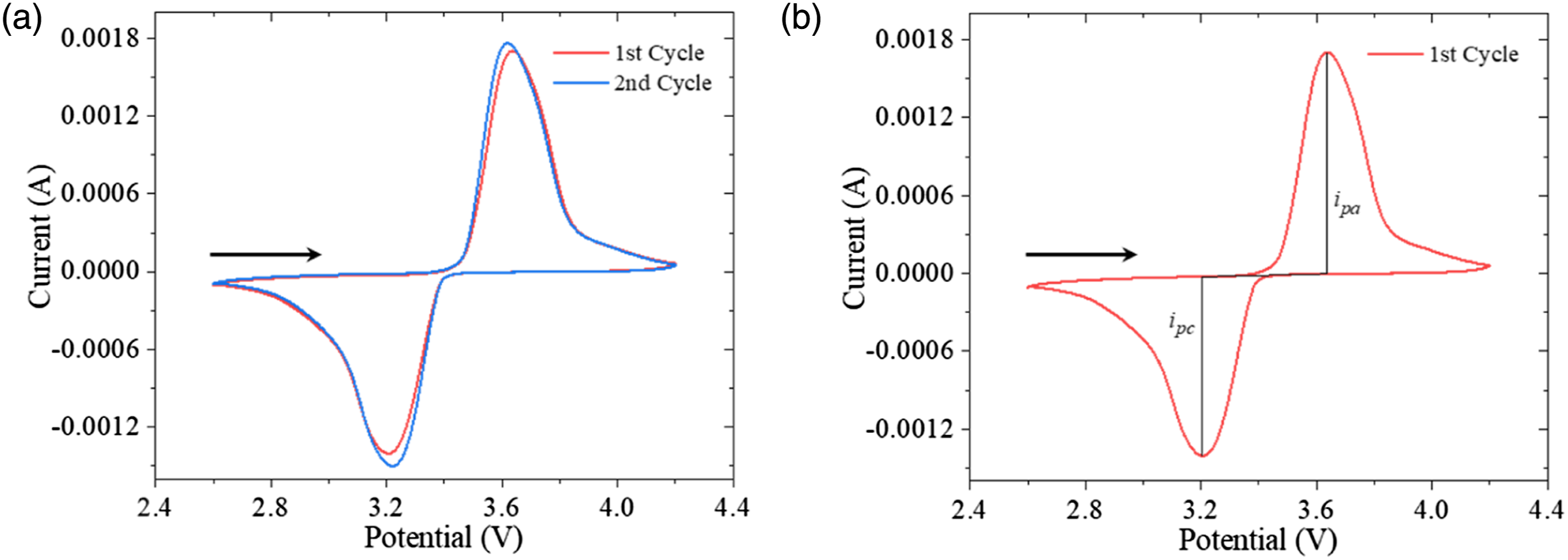
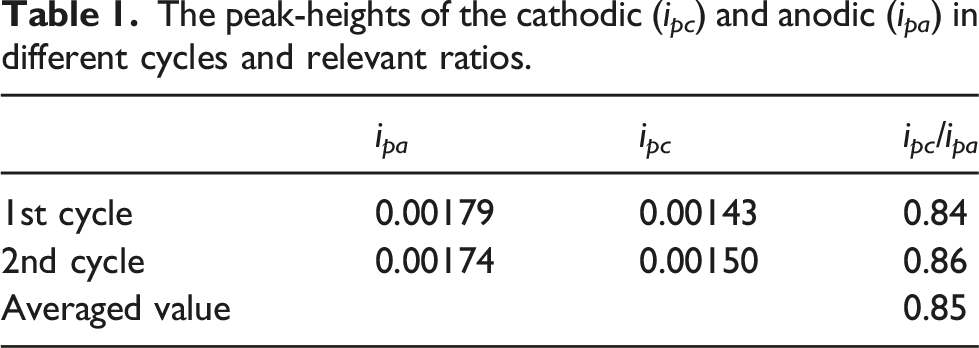
The peak-heights of the cathodic (i pc ) and anodic (i pa ) in different cycles and relevant ratios.
Electrochemical impedance spectroscopy
Electrochemical impedance spectroscopy (EIS) is a powerful and easy-to-operate tool to provide the information about the electrical resistance of the lithium-ion batteries.
29
The basic working principle of EIS is to measure relationship between the current and the applied potentials in a specific frequency domain.
30
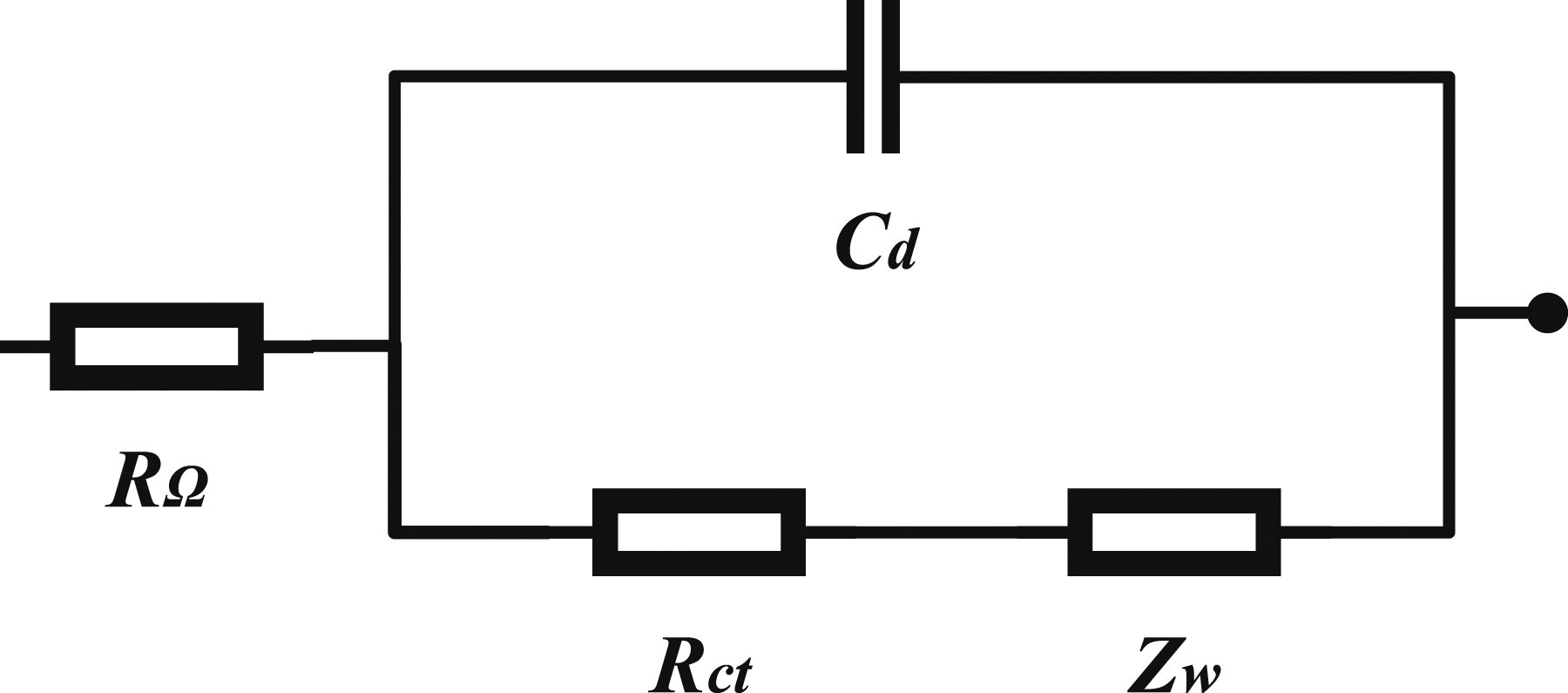
Then, the electrochemical reaction under different potential changing rates can be separately analyzed. For the fabric cathodes produced in the presented work, EIS could measure and analyze electrochemical properties of the half-cell batteries without damaging the cells, and the result is plotted in Figure 9. The Electrochemical impedance spectroscopy of the selected woven carbon fabric cathode. Equivalent circuit model for the EIS spectrum.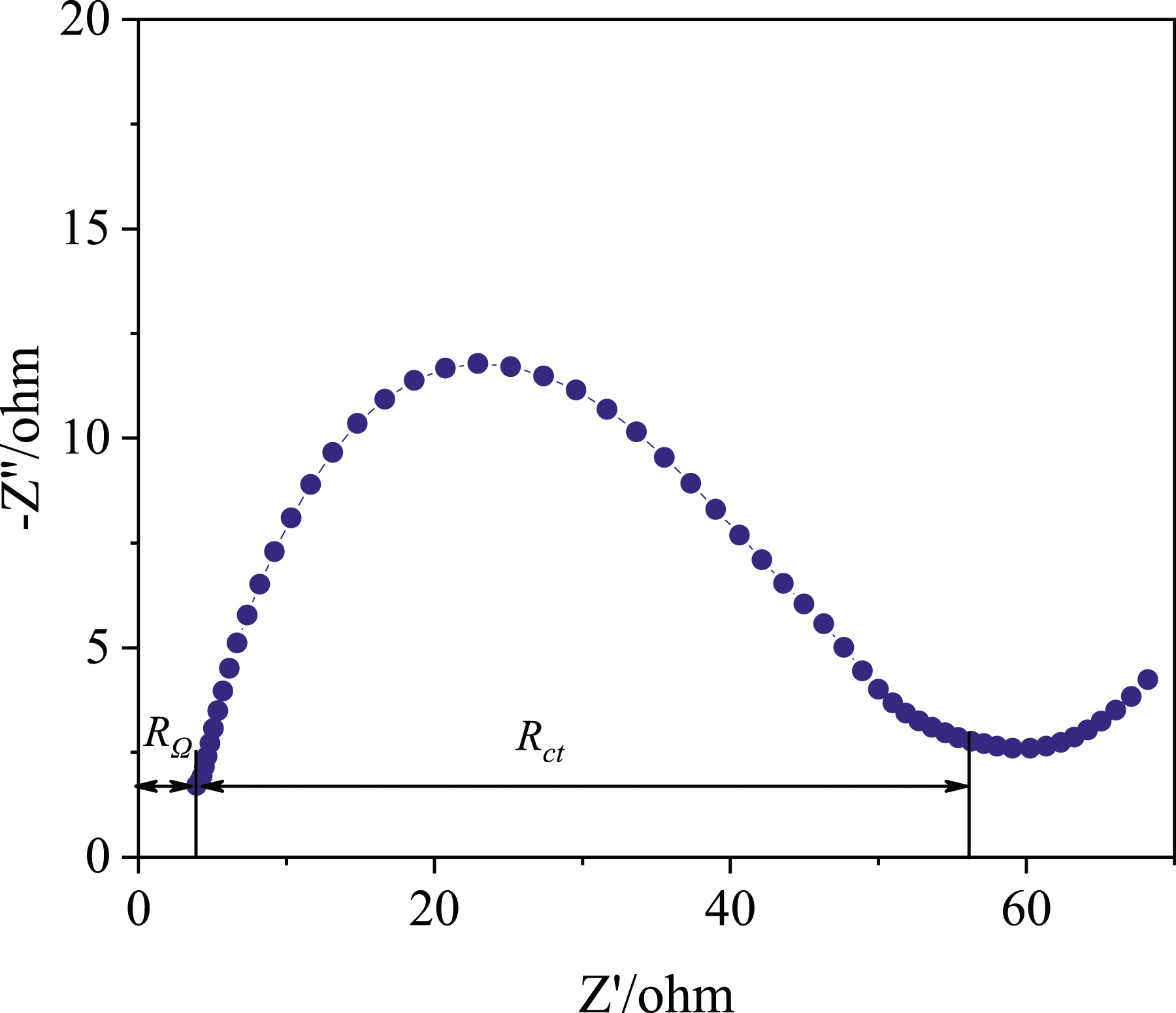
Galvanostatic discharging/charging tests
Examples of half-cell galvanostatic test results for the produced fabric cathodes are presented in Figure 11. The results of one sample are presented here for clear demonstration, but three other samples have also been tested and led to deviation of less than 5.3%. A representative charging/discharging voltage profile at 0.1 C-rate is plotted in Figure 11(a). The voltage profile demonstrates a typical plateau at around 3.4 versus Li/Li + for LFP. It is apparent that the charging and discharging processes are reversible in the presented 1st, 50th, and 100th cycles. In addition, differences between these three cycles are insignificant, indicating that electrochemical performance deterioration of the cathode from the first to the one hundredth cycle is inconspicuous. Furthermore, the difference between charging/discharging processes of the 1st and 50th cycles is smaller than that of the 50th and 100th cycles, with a difference at 0.83% and 3.01% respectively, meaning that the performance deterioration rate will increase as the cathodes age. Galvanostatic half-cell test results for one example of the carbon fabric cathodes: (a) charging and discharging curves of specific capacity versus voltage during the 1st, 50th and 100th cycle at 0.1 C, (b) specific capacity from the 1st to 100th cycle at 0.1 C, (c) efficiency and retention from the 1st to 100th cycle at 0.1 C, and (d) specific capacity at various C-rates.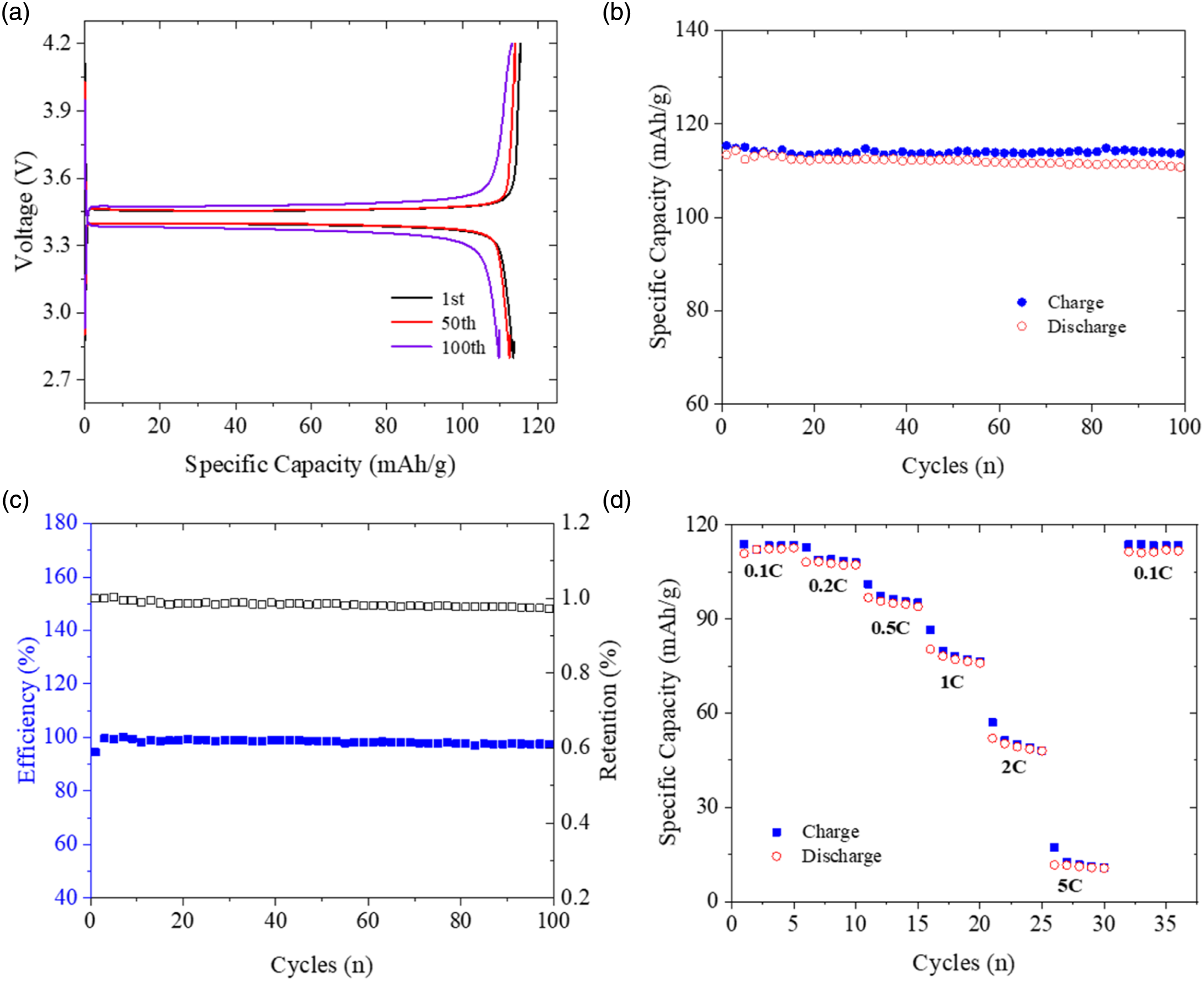
Figure 11(b) depicts curves of specific capacity versus cycle number during both charging and discharging processes. Specific capacity of the fabric cathodes can achieve greater than 110 mAh/g, which is comparable to the specific capacity of pure LFP published in. 14 Figure 11(c) provides the results of efficiency and retention for one example of the fabric cathodes, and it can be noticed that these two parameters could maintain a high level of 98% and 0.97, respectively, from the 1st to 100th cycles, indicating that the produced fabric cathodes have achieved excellent long-cycle performance, which is also supported by results in Figure 11(b) where the specific capacity during charging and discharging are close to each other in every cycle. Moreover, the well-matched charging/discharging points in Figure 11(b) and the high efficiency in Figure 11(c) imply that (1) the insertion and extraction of lithium ions and (2) lithium ions transporting between cathodes and anodes could achieve equilibrium, demonstrating reliability of the proposed high-pressure spray method to produce carbon fabric cathodes coated with LFP.
Specific capacity of the produced carbon fabric cathodes at different C-rates (0.1 C, 0.2 C, 0.5-C, 1 C, 2 C, 5 C and again 0.1 C) was determined using the half-cell experiments and the results are plotted in Figure 11(d). It can be noticed that throughout the five testing cycles under each C-rate, the specific capacity remains stable, and the charging/discharging points are highly repeatable, all with difference of less than 8.9%. Moreover, Figure 11 illustrates that specific capacity of the fabric cathodes at 0.1C can revert to its initial value after going through different C-rate states, suggesting that cathodes have benign C-rate performance. In conclusion, the specific capacity performance measured via standard electrochemical tests can completely demonstrate viability of the new high-pressure spray coating method to manufacture fabric cathodes.
Figure 12 displays capacity and areal capacity of the carbon fabric cathodes. It is clear that large areal capacity, which is more than the 3.0 mAh/cm2 industry requirement,
33
could be attained by using the high-pressure spray method and woven carbon fabrics. In this way, electrodes with limited volume and lightweight is capable of providing adequate capacity, alleviating current challenges in pack-level energy density and packaging of the batteries. Figure 12(a) also verifies reliability of the produced cathodes by illustrating that their capacity and areal capacity can remain above 3.5 mAh and 3.0 mAh/cm2,with difference of 4.85% and 4.51%, respectively, from the first to the one hundredth cycles. On the other hand, Figure 12(b) demonstrates that the manufacturing approach is applicable to produce cathodes with variable loading of active materials. Galvanostatic test results for the carbon fabric cathodes: (a) capacity and areal capacity from 1st to 100th charging/discharging cycle, (b) capacity and areal capacity with various loadings of active materials.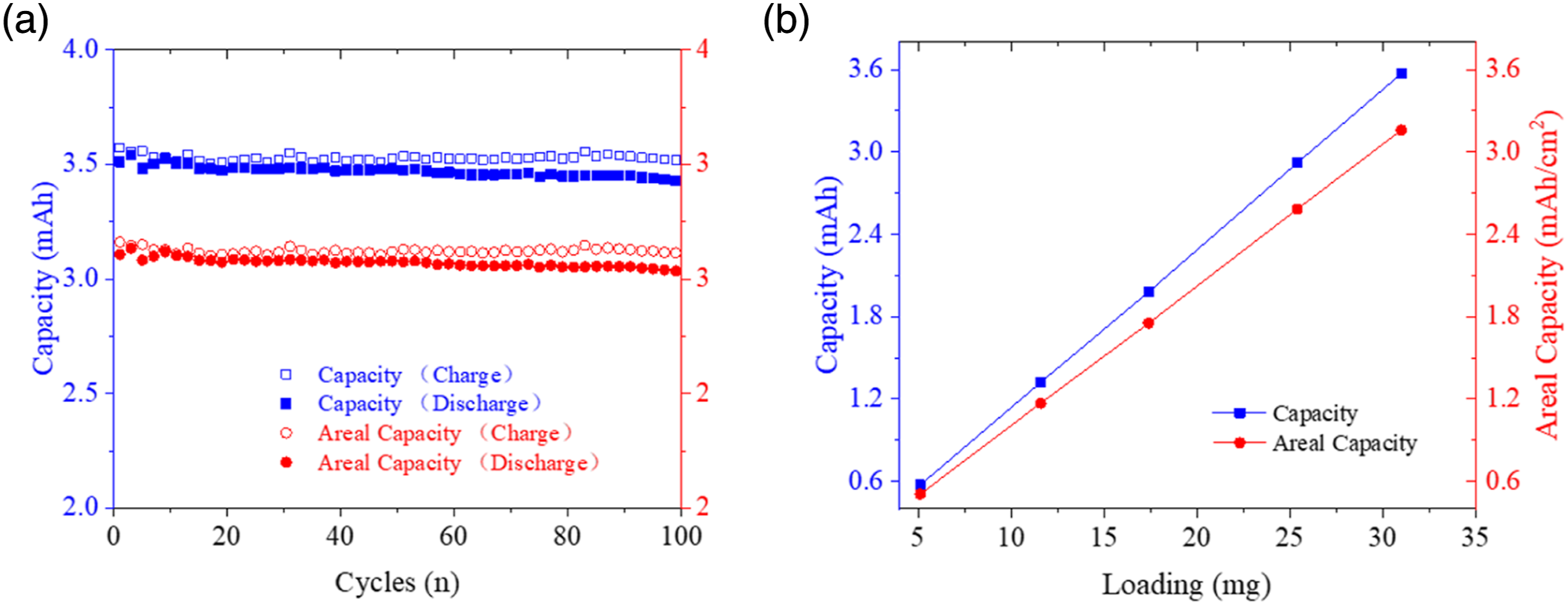
Conclusions
Woven carbon fabrics have great potential to serve as current collectors of lithium-ion batteries because of their high conductivity and 3D inner structure. Nevertheless, due to their rough surfaces and the requirement to deposit active material particles inside, woven carbon fabrics cannot be processed using the conventional blade cast coating technique. This paper presents a new pathway to manufacture carbon fabric cathodes by coating active materials (LFP/AB/PVDF) onto fibers via high-pressure spray. This approach is contactless and can deposit active particles not only on the surface of but also inside the woven carbon fabrics, resulting in heavy loading and a high areal capacity.
Electrochemical characterization experiments demonstrate that the produced carbon fabric cathodes can achieve (1) specific capacity of above 110 mAh/g, (2) areal capacity of over 3.0 mAh/cm2, (3) outstanding rate performance from 0.1 C to 5 C, and (4) excellent long-term performance with high efficiency of 98% and high retention of 0.97 after 100 cycles, indicating great application potential of this new electrode material. The following research will focus on assembly of these cathodes with other components to construct structural batteries with both energy storage and loading bearing capability. Specifically, the next step will be manufacturing full-cells using modified woven carbon fabrics as both cathodes and anodes and carbon fabric prepregs instead of metal as the packaging material.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work described in this paper was supported by grant from the Research Grants Council of the Hong Kong Special Administrative Region, China (Project No. CUHK 24208921).
Data availability statement
Data will be made available on request.