Abstract
In this study, effects of di- and trifunctional chain extenders including ethylene glycol (MEG), ethanolamine (MEA), diethanolamine (DEA), and glycerin (GLY) were investigated. 0.3, 0.6, and 0.9 php (parts per hundred parts of polyol mixture) of the chain extenders were added to the polyol formulation. Then, foam parts were produced by molding. The reaction profile, free rise density (FD), and mechanical properties of the specimens were tested regarding ASTM D7487 test methods. First of all, reaction profiles were affected by the chemical nature of the chain extenders. MEA and DEA which have a primary and secondary amine unit, respectively, accelerated both the polymerization and blow reactions and resulted in faster cream (CT), gel (GT), and end-of-rise time (RT) values. On the other hand, MEG also accelerated GT, but retarded CT. Finally, GLY retarded CT, GT, and RT of foam samples. FD of all samples were almost the same with a slight decrease by increasing the amount of chain extenders. Besides, force-to-crush (FTC) as a measure of closed- or open-cell structure, was investigated. Results showed that incorporating di- and trifunctional chain extenders led to a remarkable increase in FTC values. The influence was more profound with amine-based chain extenders. The effects of chain extenders on the mechanical properties were also studied according to the ASTM D3574. In the compression set test, using difunctional chain extenders up to 0.6 php, improved the performance. But higher amounts reversed the trend. On the other hand, trifunctional chain extenders declined the compression set properties of the foam. The addition of chain extenders dwindled the tensile and tear properties of the foams. However, the reduction was less with difunctional chain extenders.
Introduction
Polyurethane (PU) is a rapidly-growing class of polymers that is produced by the reaction between two components; namely the polyol and the isocyanate. The polyol component is usually a mixture of chemical substances; including multi-functional polyether or polyester polyols, surfactants, catalysts, chain extenders, and other additives. On the other hand, the isocyanate component is comprised of multi-functional isocyanate-containing molecules, prepolymers, and other additives. PU was first synthesized by Otto Bayer in 1937. 1 Since then, investigation on its utilization for industrial applications like elastomers, adhesives, coatings, foams, thermoplastic elastomers, etc. has been in progress.2–6 PUs are recognized to be mainly easy to produce and exhibit excellent properties in various situations. 7 Thus, PU is, nowadays, one of the most important classes of polymers being used for highly sophisticated products to everyday life applications. 8
PU foams constitute up to 67% of the total PU consumption in the globe which can be divided into two main groups: rigid and flexible. 4 High energy absorption capacity, dimensional stability, thermal and ageing resistance, and low density are the main characteristics that have made rigid PU foams an ideal candidate for insulation purposes in a variety of applications such as the building industry, refrigeration, and transportation.9–11 On the other hand, flexible PU foams are mainly recognized for their excellent reversible deformation and load-bearing capabilities. They are widely used in two main industries: first, the transportation industry, for applications like car, aircraft, and train seats, steering wheels, sound-insulation, etc., and second, the furnishing industry for applications like pillows, mattresses, furniture cushions, etc.12,13
PUs are segmented polymers, that comprise hard and soft segments. 7 Polyether or polyester polyols are the most common soft segments. On the other hand, hard segments are formed from the reaction between isocyanate and alcohol or amine moieties. These hard and soft segments are chemically linked in the backbone to form a PU chain. Hard segments are dispersed through soft segments and interact with each other to form strong hydrogen bonds, and finally phase separate. The amount, chemical nature, and state of phase-separated hard segments dominate the final properties of PUs.14–17 Sung studied the influence of isocyanate molecular structures on the sound absorption behavior of flexible PU foams. 18 It was reported that adding uretonimine-modified isocyanates results in higher microphase separation of hard domains and drainage flow in the cell wall, and consequently a higher fraction of interconnected pores. This transition in the pore structure affected the sound absorption and other properties of the flexible PU foam.
The impact of chemical parameters on the properties of PU foams has also been the focus of academic research in recent years.19–24 From the chemical point of view, there are several influential factors affecting a PU foam structure, including functionality and molecular weight of the base polyol, isocyanate, catalysts, blowing agents, silicone surfactants, and chain extenders. Many studies reveal the influence of these parameters on the properties of PU foams.25–30 Among them, chain extenders are used in minor amounts in the formulation, but can have a serious impact on the final properties of the PU foam.31–35 Chain extenders are low molecular weight molecules with two, three, four, or more functional groups. The functional group can be alcohol, amine, acid, or other active sites that can react with isocyanate groups. In the polymerization reaction, chain extenders go into reaction with isocyanate groups to form urethane, urea, or amide functional groups. Baghban et al. investigated the impact of MEG, MEA, and ethylenediamine as chain extenders on the micro-phase separation and properties of polyester-based flexible PU foams. 36 According to the results, by increasing the amine content of the chain extender, bidentate hydrogen bonds and consequently, the micro-phase separation degree increased. This way, the average cell sizes decreased and open-cell contents increased. Jiang and coworkers described the impact of a new biobased polyol and 1,4 butanediol as the chain extender on the properties of rigid PU foams. 35 In this article, the microphase separation behavior and thermodynamic properties of the foams with different chain extender contents were analyzed. According to the results, the addition of 5 wt% 1,4 butanediol increases the proportion of ordered hydrogen bonds and consequently, the degree of microphase separation which results in an increment in the compression strength of the foams. Li et al. studied the effect of four chain extenders on the morphology development of a PU foam. 37 It was reported that the addition of chain extenders affects the microphase-separated hard domains. Besides, the onset of microphase separation and foam modulus growth was delayed. Zou and coworkers investigated the impact of hard segments on the thermal and mechanical properties of semi-rigid PU foams. 38 In this study, MEG was used as a chain extender for the production of semi-rigid PU foams. It is evident that the tack-free time was reduced and the reaction was accelerated by the addition of MEG. Also, the apparent density of the foams decreased by increasing MEG in the formulation. The mechanical properties also differed by the chain extenders, which can be attributed to the change in the hard segment content and phase separation in the PU foam. Kim and coworkers described the impact of DEA loading on the sound absorption efficiency of flexible PU foams. 33 It is discussed that without using chain extenders, the PU matrix tend to microphase separate, due to the largely bidentate hydrogen interactions. On the other hand, using DEA leads to the formation of monodentate hydrogen interactions, which reduces cell opening by phase separation. Thus, by increasing the amount of DEA in the formulation, the number of open pores reduces. This change in the pore morphology impacted the sound absorption property of the final foam.
This work aims to study the impact of functionality and chemical nature of chain extenders on the mechanical properties and performance of cold-cure flexible PU foams. For this purpose, two difunctional chain extenders with amine or alcohol active sites including ethylene glycol (MEG) and ethanolamine (MEA) were chosen. On the other hand, diethanolamine (DEA) and glycerin (GLY) were chosen as trifunctional chain extenders with amine or alcohol active sites. Using a standard formulation for the polyol component comprising up to 0.9 php of the specific chain extender and an MDI-based isocyanate, cold-cure PU foams were produced by the molding process. Then the impact on the reaction profile and mechanical properties of the foams were tested according to the standard procedures.
Materials and methods
Materials
4500 molecular weight polyether polyol (triol, OH value = 38 mg KOH/g) from Sanyo Chemical Industries, LTD, Kyoto, Japan was used as the base polyol. Bis(dimethylaminoethyl)ether (70:30 wt% in dipropylene glycol) and triethylenediamine (33:67 wt% in dipropylene glycol) from Jiangsu Maysta Chemical, Nanjing City, P.R.China, were used as blow and gel catalysts, respectively. Dabco DC2585 from Evonik was used as stabilizer in the formulations. Demineralized water was used as the chemical blowing agent. MEG, MEA, DEA, and GLY were purchased from Shazand Petrochemical Company, Arak, Iran. The isocyanate component was a polyol-modified MDI-based one with NCO% = 29.6 and a viscosity of 70 m Pa.s at 25°C. All materials were used as received.
Polyol component formulation
Polyol formulation for high resiliency PU foams.
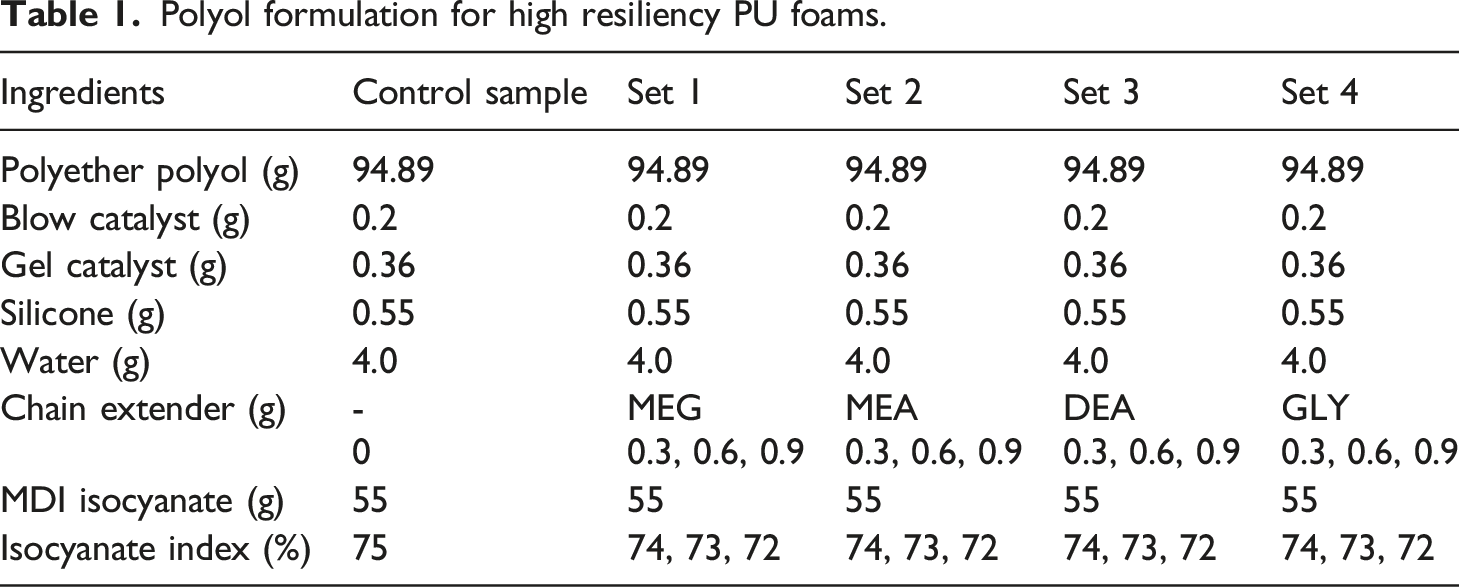
Chemical structure of the chain extenders used in this study.
Foam sample preparation
Foam samples were prepared by mixing the isocyanate and polyol components. The weight ratio (isocyanate/polyol) was 0.55. For cup tests, 30 g of polyol was poured into the cup. Then 16.5 g of the isocyanate was added. Subsequently, the mixture was mixed using a static mixer with a mixing rate of 3000 r/min. For mechanical tests, standard samples with dimensions of 300 × 300 × 100 mm3 were produced using a mold. The isocyanate and polyol components were mixed for 7 s with a mixing rate of 3000 r/min, and then the mixture was poured into the mold. The mold temperature was set to 45 ± 1°C and the demolding time was 5 min. After production, foam samples were allowed to post-cure for 24 h at room temperature and relative humidity of 50%. Mechanical tests were performed on crushed samples, 5 days after the production.
Test methods
Reaction profile and free rise density measurement
Briefly, 30 g of polyol was weighed in a cup. Then the desired amount of isocyanate was added to the cup and the materials were mixed using a 3000 r/min mixer. The reaction profile parameters and free rise density were then examined accurately according to the ASTM D7487 test method.
Force-to-crush test
FTC is the force required to press an uncrushed foam sample to 50% of its height in the first cycle and can be used as a simple measure of the overall cell structure in flexible PU foams.39,40 As the closed cell structure increases, air can flow through the foam with more difficulty. Thus, the FTC value increases. FTC of samples was tested immediately after removing the samples from the mold.
Scanning electron microscopy
The surface morphology of the foam samples was analyzed using the SEM method by a Hitachi scanning electron microscope operating at 20 kV. Specimens were cut from the center of the crushed foam samples. Specimens were coated with gold before the examination.
Compression force deflection and compression set tests
Compression force deflection and compression set tests were conducted according to the ASTM D3574 standard. Briefly, specimens with dimensions of 50 × 50 × 25 mm3 were cut from the core of the crushed samples, at least 20 mm away from the surfaces. For the CFD test, specimens were pressed to 75% of the initial height twice, at a rate of 250 mm/min, allowing the compression foot to fully clear the specimen after each compression. Then the samples were allowed to rest for 6 min. Afterward, the samples were pressed to 50% of the initial height three times at a rate of 50 mm/min and a 30 s rest between each cycle to make sure the foam is relaxed. Finally, the CFD in the last (fifth) cycle is calculated according to equation (1).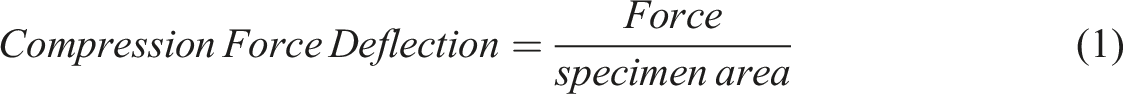

Mechanical tests
Mechanical properties of the samples, including tensile and tear tests were measured regarding the ASTM D3574 test method. For tensile tests, dumbbell-shaped specimens were cut from the crushed foam parts. Tests were conducted at the rate of 100 mm/min. For tear tests, trousers-shaped specimens were cut from the foam parts. The maximum force after 20 mm of torn in the sample was recorded and the tear strength value was reported using equation (3), where F represents force in N and T represents thickness in mm.
Results and discussion
Foam samples preparation
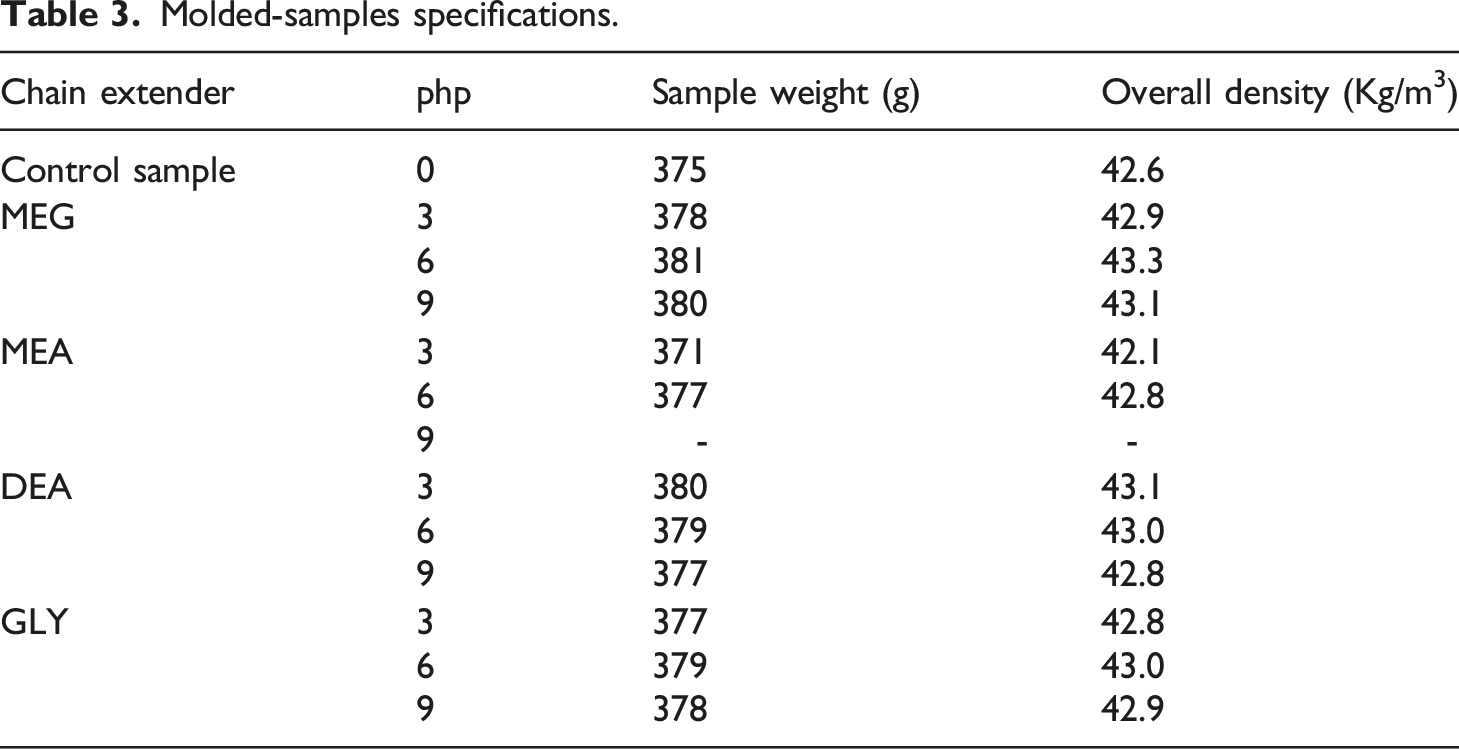
Polyol formulations were prepared using di- and trifunctional chain extenders with different chemical reactive sites. To study the reaction profile and free-rise density of the foams, cup samples were prepared as described earlier (Figure 1). For mechanical tests, standard molded samples were produced through the molding process. All samples could be made perfectly except for MEA samples. Using 0.9 php MEA, the reaction rate was so fast that it was impossible to have flawless molded samples for mechanical tests. Indeed, the cream time and gel time values were so low that proper mixing of the polyol and isocyanate components could not be achieved. Specifications of the molded samples are presented in Table 3. Cup sample foams with different chain extenders, (a) MEG, (b) MEA, (c) DEA, and (d) GLY. Molded-samples specifications.
Reaction profile and free rise density
The reaction profile and free rise density of the samples were measured and the results are presented in Figures 2 and 3, respectively. From Figure 2 it can be deduced that amine-based chain extenders accelerate the reaction and result in lower values for CT, GT, and RT. This can be attributed to the catalytic effect of amine moieties in PU reactions.
7
On the other hand, GLY which is an alcohol-based chain extender retarded the reaction and led to higher values of CT, GT, and RT. Using MEG as the chain extender also resulted in higher values of CT and RT, but lower values of GT. The lower reactivity of primary and secondary alcohols may lead to a portion of incompletely reacted alcohol-based chain extenders, during the microphase separation process.
37
Reaction profile values. Free rise density of the samples.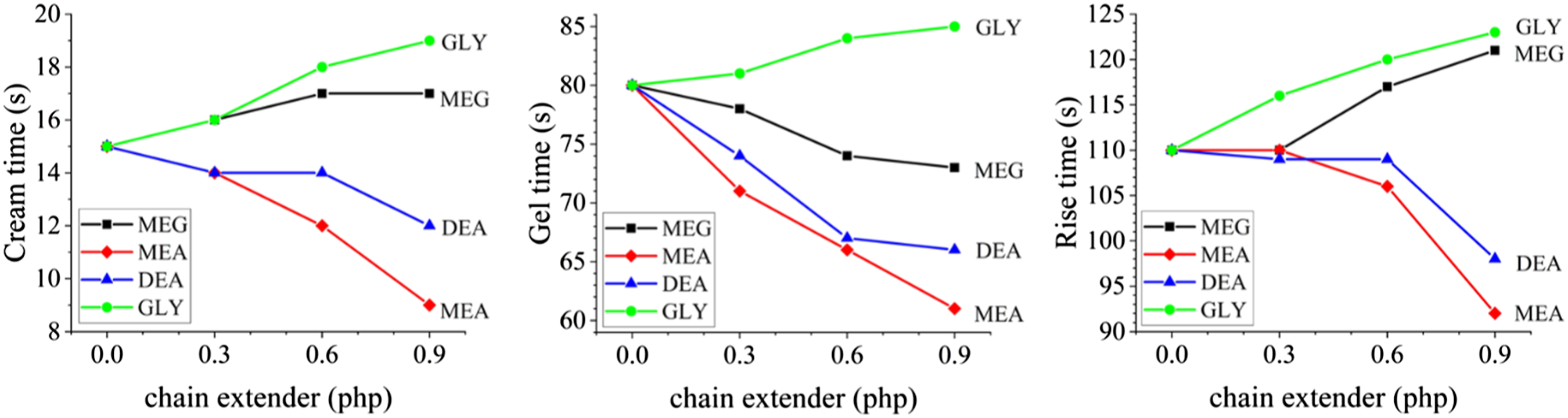
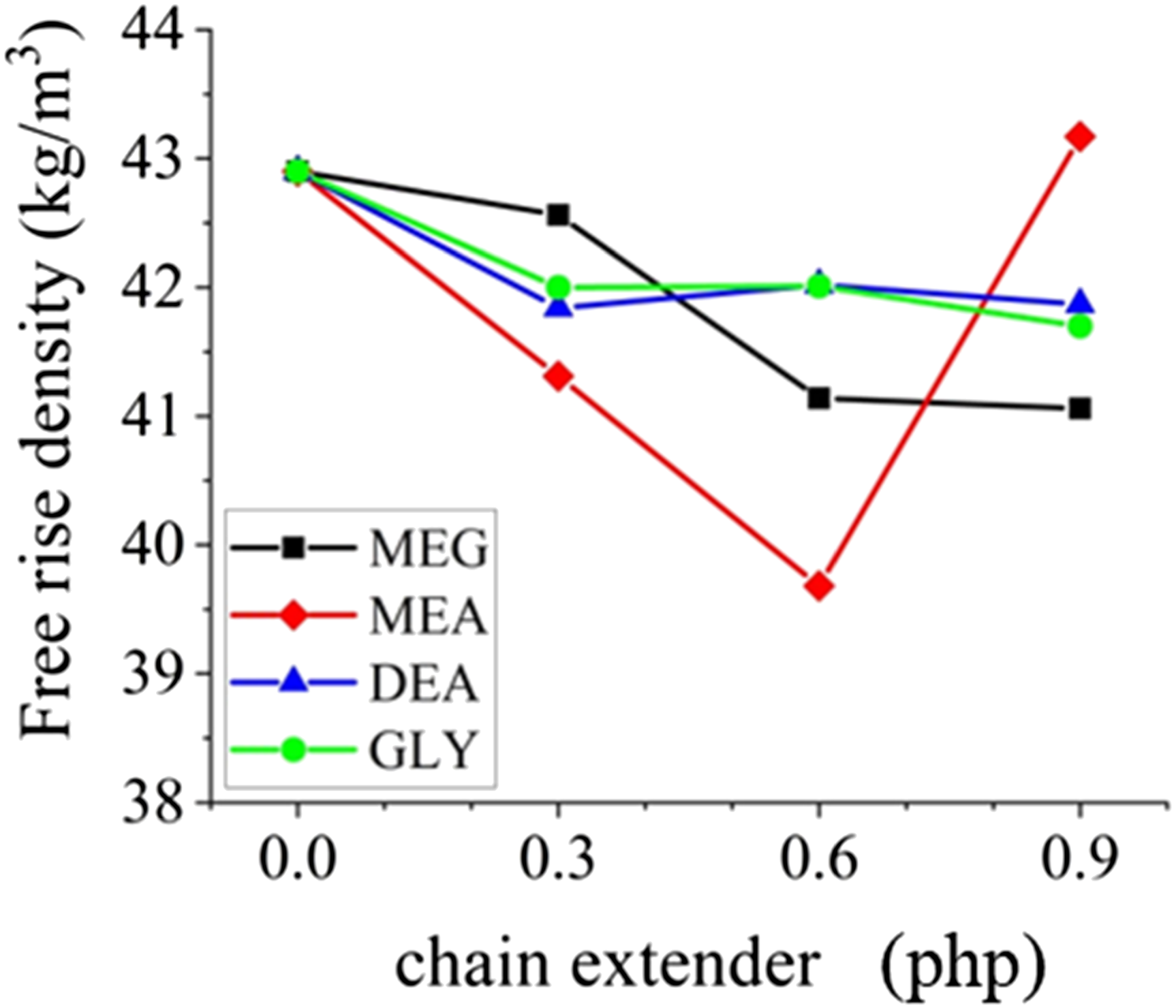
From Figure 3, it can be seen that the FD of all samples are almost the same, with a slight decrease by increasing the chain extender. In the case of MEA, increasing the amount of the chain extender up to 0.6 php resulted in a more rapid reduction of FD compared to other chain extenders. Finally, by 0.9 php of MEA, the trend reverses and the FD increases sharply. This can be due to the extremely fast reaction profile of this sample, which prevents the complete reaction of water with isocyanate groups. The trend is consistent with what was reported earlier by others. 38 This behavior also affected the sample preparation process, which will be discussed in the following section.
FTC results
To measure the FTC values and mechanical properties, standard foam samples were produced through the molding process. Except for 0.9 php MEA, flawless samples could be prepared from all formulations. The extremely fast reaction profile and the change in the FD of 0.9 php MEA formulation made it impossible to have normal samples for further studies. Thus, 12 standard samples were used to investigate the impact of chain extenders on the FTC and mechanical properties of the PU foams.
For FTC measurement, foam samples were used immediately after demolding. The results are presented in Figure 4. As it is evident, by increasing the amount of chain extenders the FTC values increase sharply, regardless of either functionality or chemical nature of the chain extender. This observation suggests that by the addition of each kind of chain extenders, the fraction of the closed-cells increases. In the PU foam formation process, chain extenders react with the isocyanate component and form urethane or urea groups. Thus, they promote the formation of hard segments. Hard segments form hydrogen bonds and crystallize to fabricate hard domains.
7
It is worth noting that PUs are segmented polymers with hard and soft domains. Hard domains are connected via strong hydrogen bonds and dispersed inside the soft domain matrix. The dispersion and morphology of these hard domains govern the final properties of PUs. Chain extenders disrupt the ordering of strong bidentate hydrogen bonds and facilitate the formation of monodentate hydrogen bonds between hard segments. This way, the microphase separation which finally leads to cell rupture is hindered and more closed cells are formed.33,41 Moreover, according to a previous study chain extenders delay the onset of microphase separation.
37
Thus, cell rupture is also delayed, more gas is entrapped inside the cells and the pressure of the cells increases. Consequently, FTC values of foams increase by increasing the amount of chain extenders. FTC values of foam samples.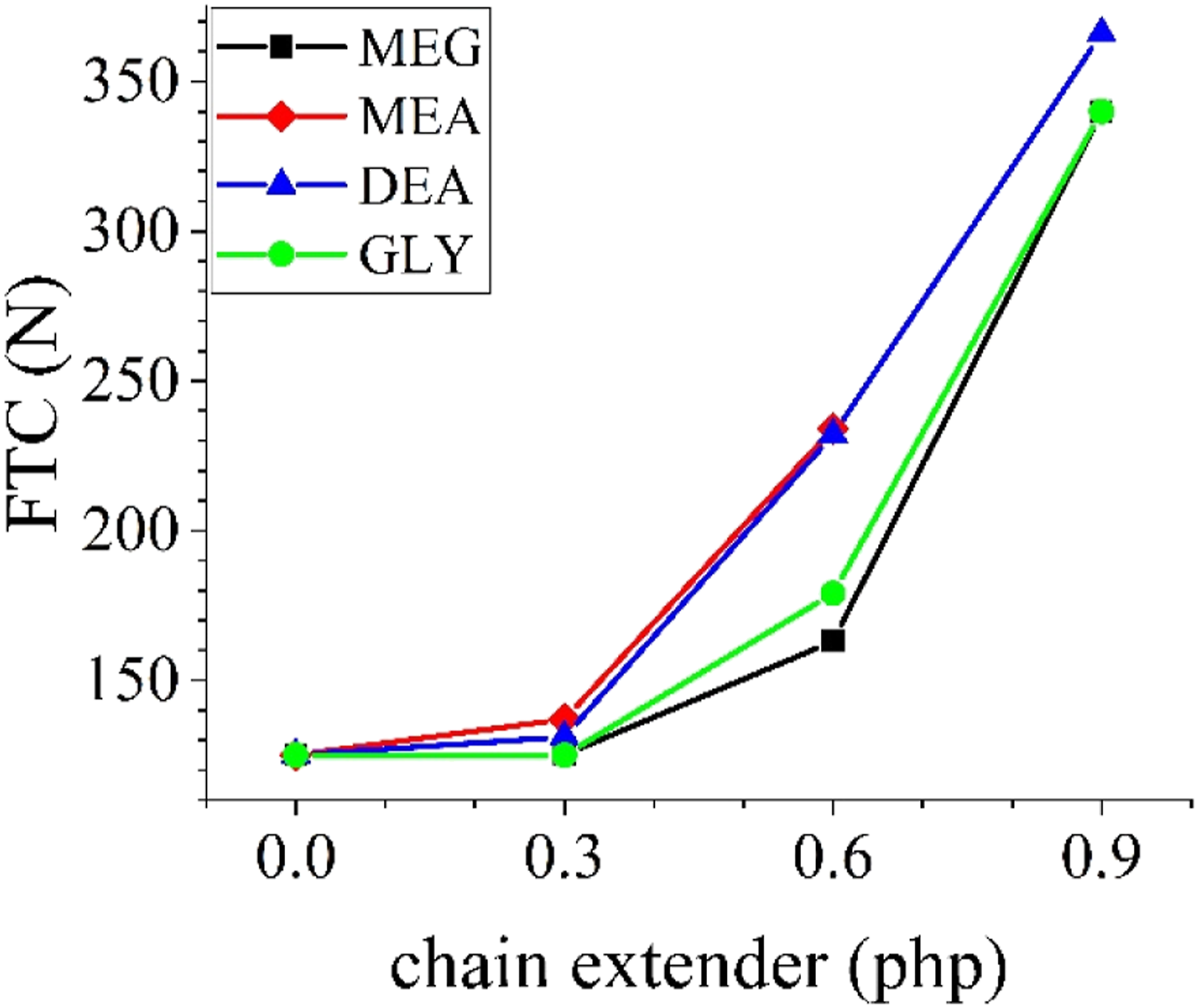
From Figure 4, it can be deduced that using amine-based chain extenders (MEA and DEA) results in higher values of FTC compared to alcohol-based ones. This can be assigned to two main factors; first, the higher activity of amine groups, and second, the formation of urea linkage from the reaction of amine groups and isocyanate groups. Urea groups form much stronger hydrogen bonds, compared to urethane groups, and facilitate the formation of interconnected domains inside PU chains.42,43
SEM analysis
SEM images of the produced foam samples after crushing are presented in Figure 5. As it is evident, there is a considerable number of open pores in all foam samples. Although the FTC values increase by the addition of chain extenders, the cell walls are still thin enough to be ruptured mechanically. The results suggest that regardless of the dominance of closed- or open-cell structure in the samples with different chain extenders, after crushing cell rupture occurs for all foams. SEM micrographs of the crushed foam samples.
CFD and compression set results
CFD values of the produced foams are presented in Figure 6. As it is evident, regardless of the chemical nature or functionality, by increasing the amount of chain extenders, CFD values increase. This increase can be assigned to the formation of hard segments and interconnected hard domains inside the PU chains, as discussed earlier. CFD values of the produced foams.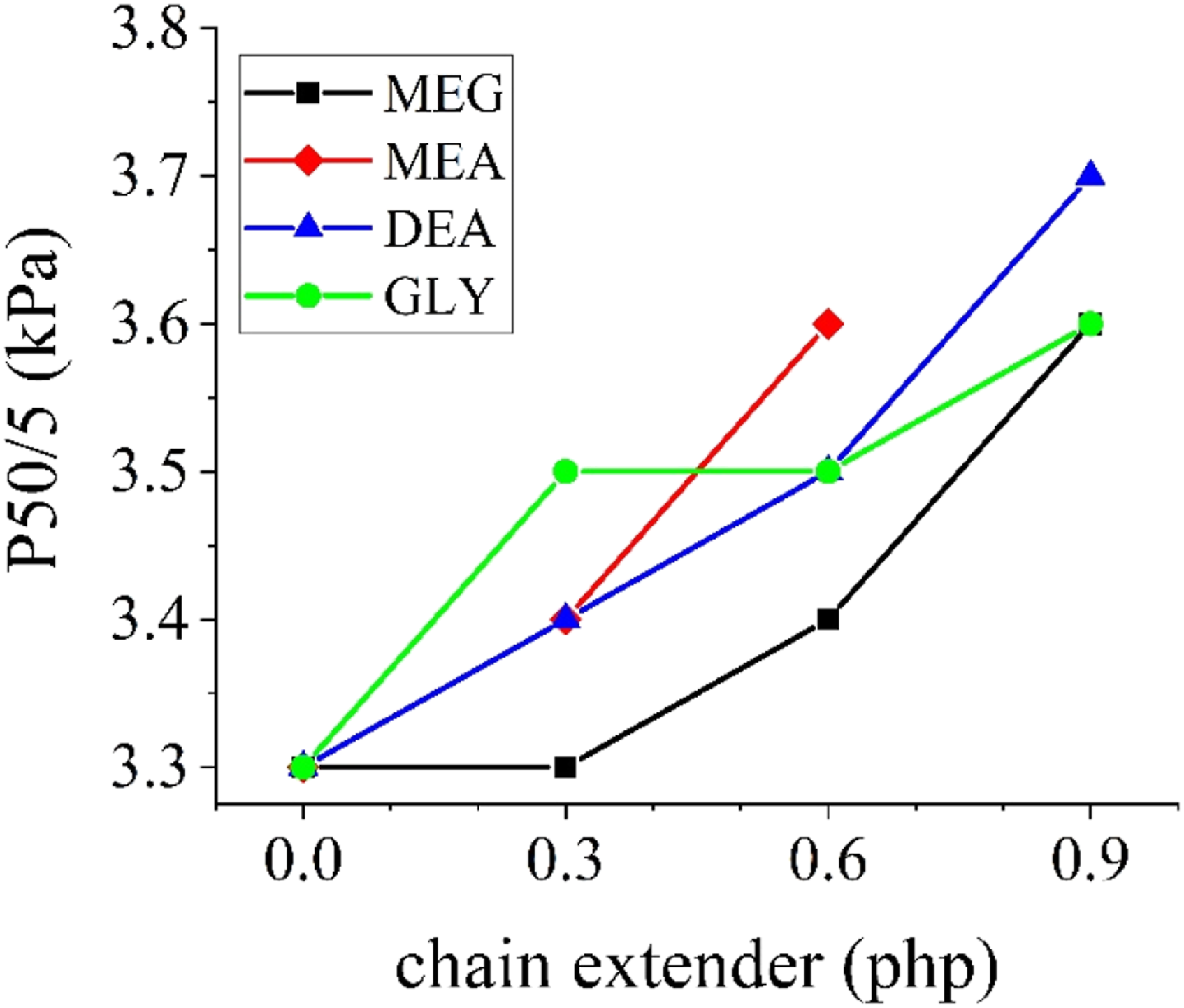
The results of compression set experiments are presented in Figure 7. Using MEG and GLY as the chain extender in the formulation resulted in a better performance in the compression set test. However, there is a limit beyond which the trend reverses. From Figure 7, by increasing the amount of alcohol-based chain extenders up to 0.6 php, compression set values decrease. There are two main parameters affecting the compression set values of PU foams; the resilience of the PU chains and the cell structure. Less resilient PU segments result in more energy dissipation and lower recovery and consequently higher values of compression set. By the addition of alcohol-based chain extenders, and the formation of hard domains, PU segments resiliency increases. Thus, by using these chain extenders up to 0.6 php, the resiliency of PU foams increases which results in lower values of the compression set. But, higher loadings of alcohol-based chain extenders, the disordered hard domains show less resiliency and hinder the complete recovery of the foam which leads to higher values of compression set. Compression set values of the produced foams.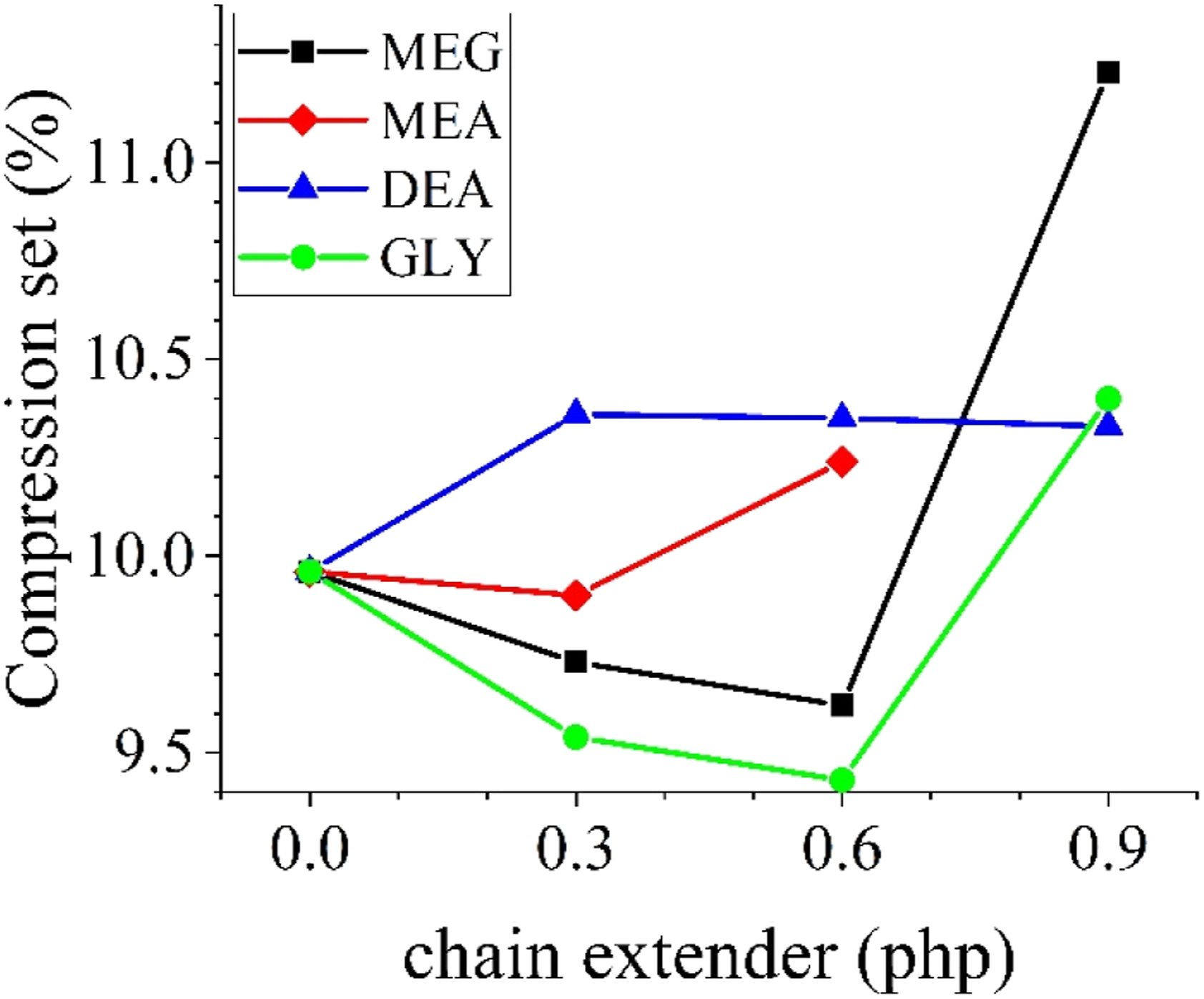
For amine-based chain extenders, however, the resiliency of the PU chains is limited due to the more disordered hard domains. Thus, a slight increase in compression set values can be seen even with minor amounts of the chain extenders.
Mechanical properties
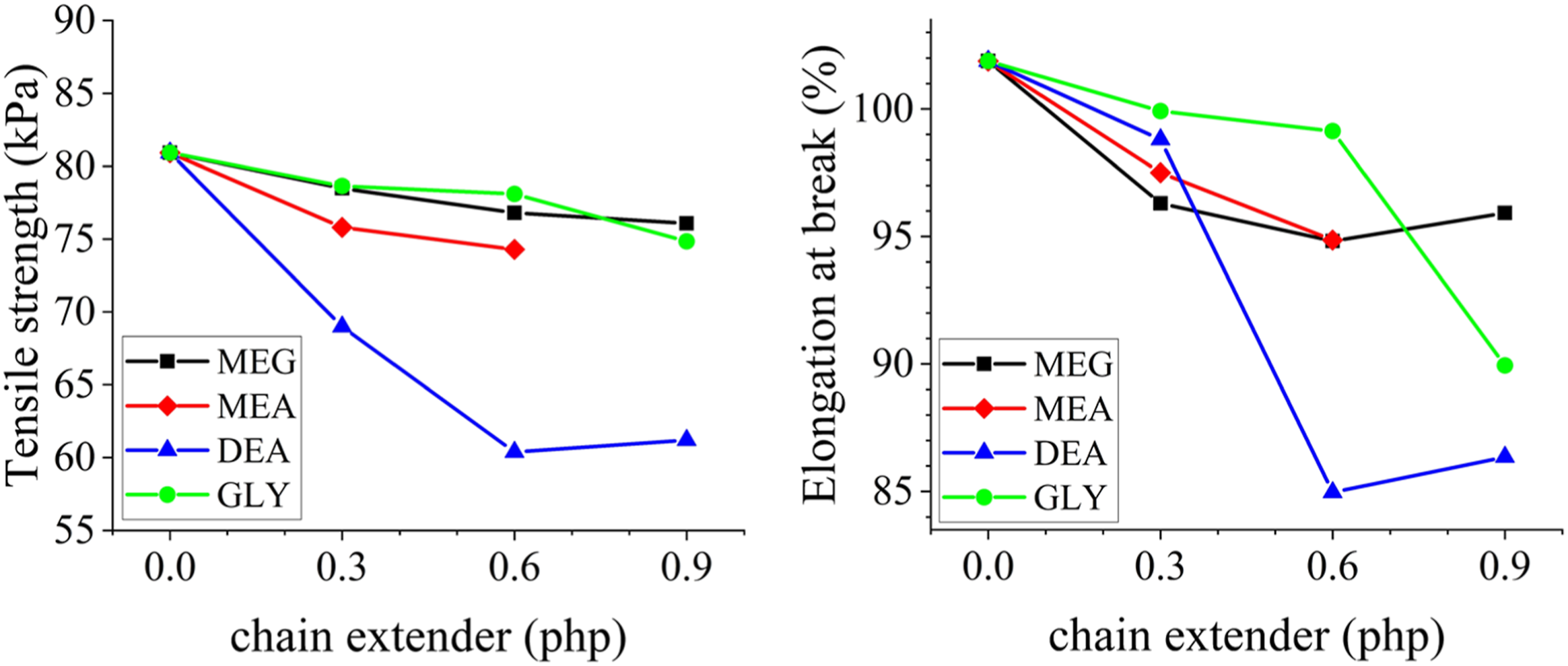
The mechanical properties of the produced foams are tested in the tensile mode. Stress-strain plots are presented in Figure 8 and the results are summarized in Figure 9. As it is evident, the tensile strength and elongation at break of the foams decline by the addition of chain extenders, due to the formation of disordered hard domains.
37
Elongation at break values decrease more sharply with trifunctional chain extenders, especially at higher loadings. This can be due to the formation of chemical crosslinks between hard segments. The trend is more pronounced for DEA. Forming crosslinks between hard segments, DEA promotes the formation of monodentate hydrogen interaction and disturbs the microphase separation of hard domains.41,44 Due to the lower activity of the secondary alcohol, GLY does not intensely affect the mechanical properties at low loadings. However, GLY-based formulations show almost the same behavior at higher loadings. In this regard, trifunctional chain extenders especially at higher loadings, reduce the mechanical properties more than difunctional chain extenders. Stress-strain plots of the produced foams using different chain extenders; (a) MEG, (b) MEA, (c) DEA, and (d) GLY. Mechanical properties of the produced foams.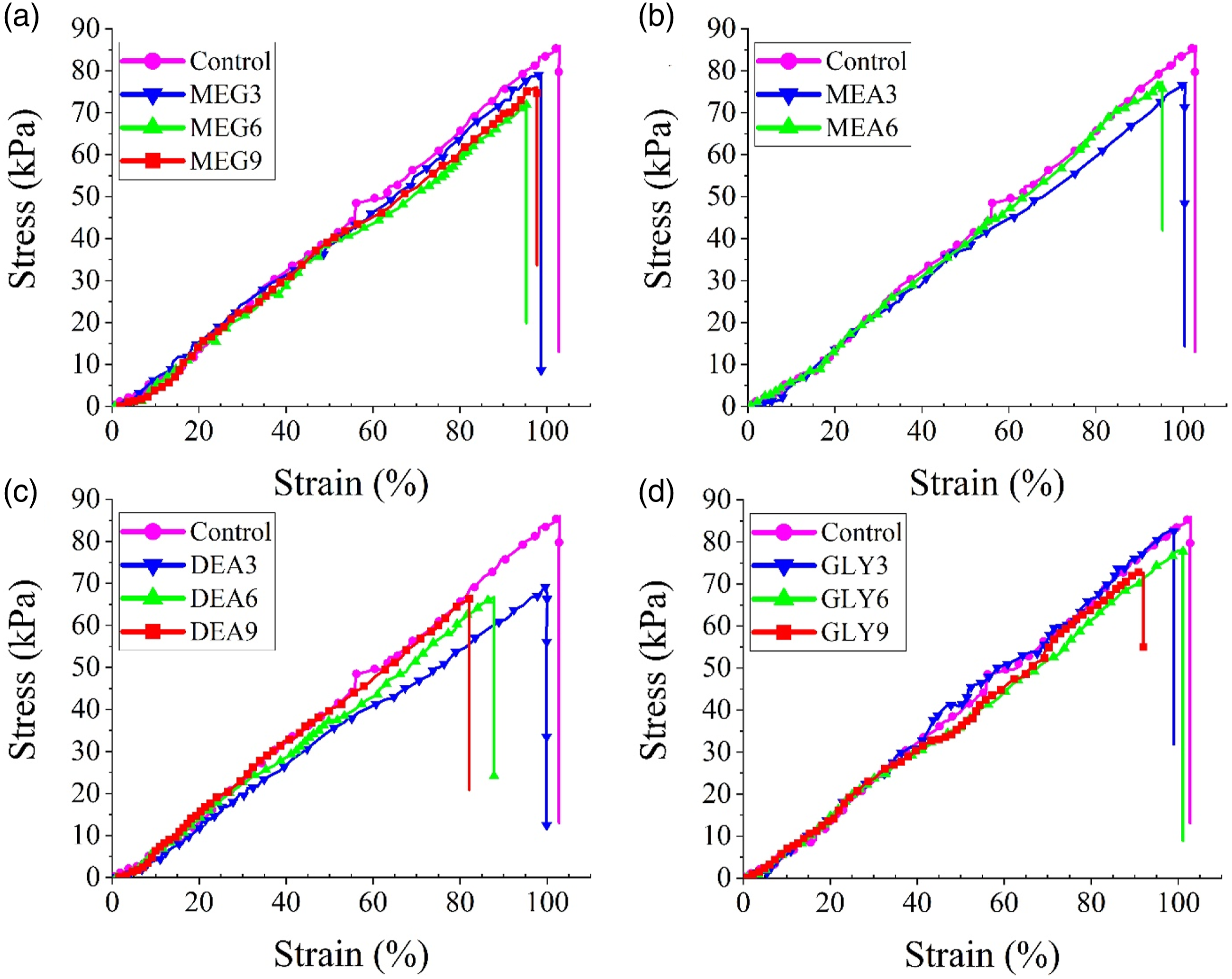
Another important mechanical property of PU foams is the tear strength. From Figure 10, it can be seen that the addition of chain extenders dwindles the tear strength of the foams. The impact is more intense with trifunctional chain extenders. Tear strength of the produced foams.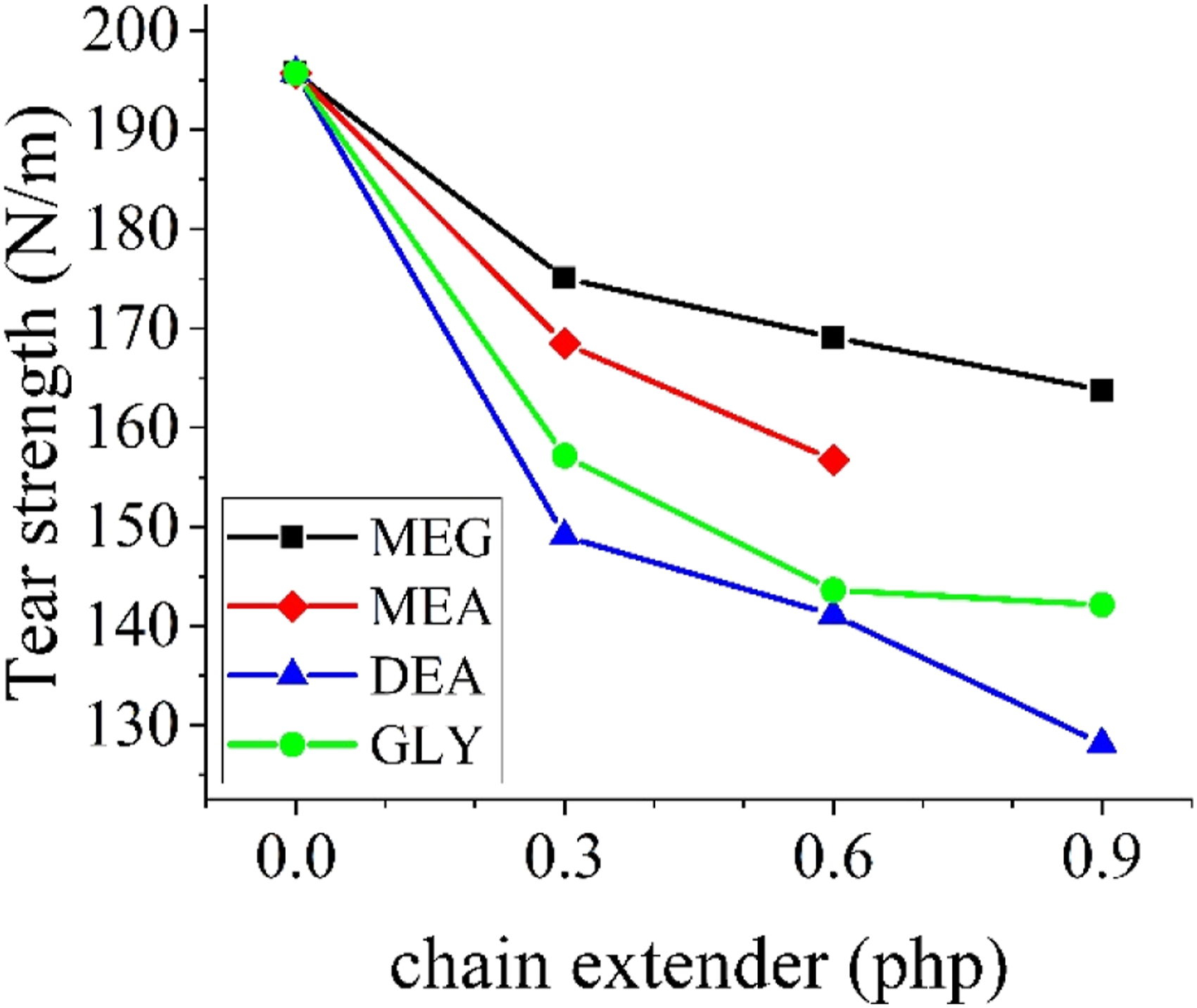
Conclusion
The purpose of this study was to investigate the effects of different chain extenders on the properties of flexible cold-cure PU foam. For this purpose, four different chain extenders were chosen; MEG, MEA, DEA, and GLY. Among them, MEG and MEA are difunctional, and DEA and GLY are trifunctional. From the chemical aspect, MEG and GLY are alcohol-based chain extenders, while MEA and DEA consist of primary and secondary amine moieties, respectively. The chain extenders were added to the polyol formulation in 0.3, 0.6, and 0.9 php. Results showed that amine-based chain extenders caused a remarkable acceleration of the reaction. In the case of 0.9 php MEA, the extremely fast reaction rate prevented the formation of a flawless foam sample. FRD of all samples was almost the same and slightly decreased by increasing chain extenders. Regarding the FTC values as a measure of the openness of the cell structure of foams, it can be inferred that using chain extenders resulted in more closed-cell structure. The impact of amine-based chain extenders was more pronounced, which may be due to the faster reaction profile. The influence of the chain extenders on the mechanical properties was also examined. It was revealed that chain extenders deteriorated the mechanical performance of the foams, regardless of the either functionality or chemical nature, which can be attributed to the disordering of the hard domains. In comparison, difunctional chain extenders had better performances in compression set and elongation at break and tear strength values. It can be deduced that by forming chemical crosslinks between hard segments, the formation of ordered hydrogen interactions is more intensely suppressed. Consequently, microphase separation and mechanical properties decline.
Footnotes
Acknowledgments
The writer deeply appreciates Caspian Polyurethane Technologies Co. for their support in conducting mechanical tests.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
