Abstract

Drug-induced photosensitivity is a cutaneous reaction that occurs after the use of a photosensitizing medication with subsequent exposure to either ultraviolet or visible radiation. Although drug-induced photosensitivity is common, the diagnosis of this condition and the identification of the culprit drug can be challenging. 1
Herein, we report the first case of photosensitivity induced by perindopril/indapamide combination.
A 70-year-old male with a history of hypertension treated with perindopril/indapamide for 5 months and Parkinson’s disease treated with levodopa/benserazide for 4 months, was referred to our department with a 2-week history an erythematous rash. The patient acknowledged that he consulted a dermatologist who first diagnosed him with levodopa/benserazide-induced photosensitivity as it was the last introduced treatment. He interrupted levodopa-benserazide after the approval of his neurologist but the rash did not resolve. Therefore, he was referred to our department for investigation. On examination, the patient had an edematous, erythematous rash on the central face which might be due to sun exposure with skin burning sensation type IV according to the Fitzpatrick scale (Figure 1A). Mild rash was also noted on dorsal hands. The patient had no history of rosacea, psoriasis or seborrheic dermatitis prior to the skin reaction. The patient denied the use of sunscreen or other cream prior to rash. We considered the possibility of perindopril/indapamide-induced cutaneous reaction, thus, we recommended to discontinue this medicine and to reintroduce levodopa/benserazide. Laboratory tests were normal, in particular antinuclear antibodies. Local corticosteroids were applied. Within 3 weeks, the skin reaction and redness decreased significantly (Figure 1B). The patient had no further episodes of skin rash over a follow-up period of 4 months. The combination perindopril/indapamide was replaced by amlodipine, a calcium channel blocker.
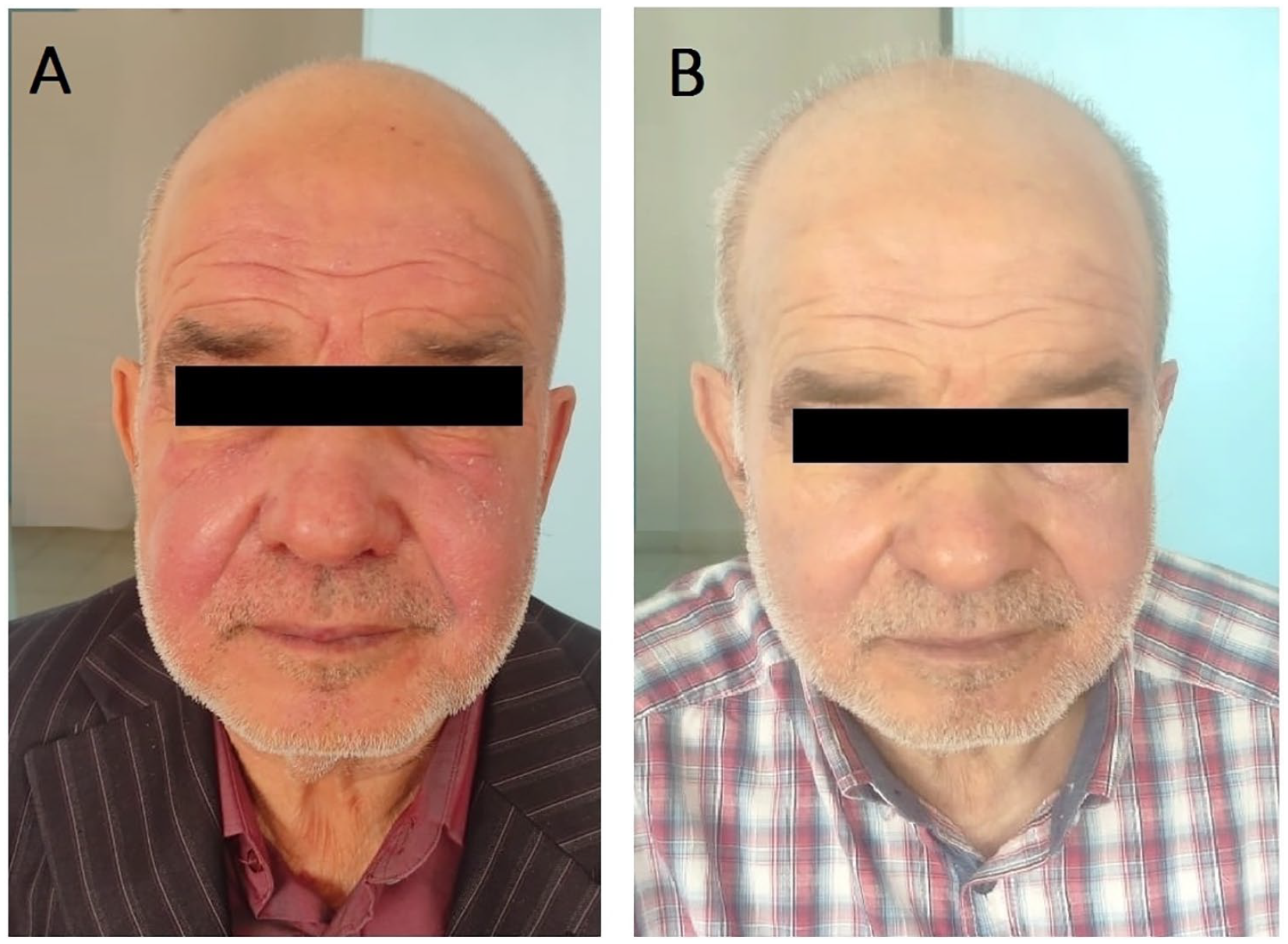
Edema and erythematous eruption on the face (A). Withdrawal of perindopril/indapamide combination for 3 weeks resulting in improvement of the face eruption (B).
Based on the history of drug intake, the clinical presentation and the resolution of symptoms on withdrawal of the perindopril/indapamide, the diagnosis of photosensitivity secondary to this medication in our patient was established. According to the objective causality assessment by the Naranjo probability scale, perindopril/indapamide-induced photosensitivity was probable.
Thiazides are thought to be associated with drug photosensitivity. The potential of the thiazide group of drugs to cause photosensitivity was recognized soon after their introduction as early as 1959. However, to our knowledge there are no well published reports of cutaneous photosensitivity induced by indapamide a thiazide-like diuretic. Only one case of photo-onycholysis, a photosensitivity reaction limited to the nail due to indapamide, in a patient diagnosed with lupus erythematosus profundus has been described in the literature. 2 The possible mechanism of the indapamide photosensitizing potential may be related to the chlorine substituents it shares with thiazides. In fact, drugs with chlorine substituents in their chemical structure, such as indapamide, exhibit photochemical activity that is traced to the UV-induced dissociation of the chlorine substituent leading to free radical reactions with lipids, proteins and DNA. 3 Photosensitivity reactions have also been described for angiotensin converting enzyme inhibitors (ACEI). Actually, single case reports have been associated with captopril, ramipril, quinapril and enalapril. Perindopril has been implicated only in one case of lichenoid photodermatitis. 4 In a comprehensive and rigorous systematic review, associations between thiazides and phototoxicity are supported by low-quality evidence as determined with use of the GRADE system. 5 ACEI are not included in this review even in the section “association with very low evidence.” These findings may be explained in part that physicians are more likely to diagnose and subsequently report photosensitivity in newly marketed drugs, and less likely to report a case resulting from a known photosensitizer. 5 It is possible that cases of mild photosensitivity are benign and therefore unnoticed by physicians, and that only serious sensitive reactions merit reporting. Cases of mild photosensitivity may also not be recognized by physicians, pharmacists or patients and therefore are not reported. In addition, the lack of effective pharmacovigilance system and a publication bias toward publishing only extravagant cases in the medical literature may play a role. 4 Therefore, physicians and pharmacists should report photosensitivity reactions associated to drugs in order to better understand their association evidence and their true incidence. Considering the wide use of perindopril/indapamide combination in hypertensive population, clinicians and pharmacists should be aware of the possibility of adverse effects such as photosensitivity occurring during its use.
Footnotes
Author Contributions
CBS and DS participated in data collection and interpretation of results. All authors contributed to the manuscript writing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
