Abstract
Body dysmorphic disorder (BDD) is a persistently under-recognised psychiatric condition. Evidence suggests a degree of shared cognitive dysfunction and clinical presentation of BDD with autism spectrum disorder (ASD) and attention-deficit hyperactivity disorder (ADHD). The current study is the first to investigate the co-occurrence of BDD, ASD and ADHD in a large online community sample. Utilising data from an online survey, we investigated the comorbidity frequency of BDD, ASD and ADHD, as well as the presence of possible undiagnosed BDD in these neurodevelopmental populations (
Body dysmorphic disorder (BDD) is a psychiatric condition marked by a preoccupation with perceived appearance flaws that are minor or undetectable to others. People with BDD typically engage in repetitive behaviours (e.g. mirror checking and reassurance seeking) and/or mental acts (e.g. social comparison) in response to their appearance concerns (American Psychiatric Association [APA], 2013). The reported prevalence of BDD is estimated at 1.9% in community settings (McGrath et al., 2023); however, research has suggested rates of BDD in community samples are increasing (Gieler et al., 2016), with many affected individuals being undiagnosed (Veale et al., 2015). Under-recognition is exacerbated by a tendency to seek dermatological and cosmetic procedures rather than mental health treatment among a proportion of people with BDD (Mancuso et al., 2010). Individuals experiencing BDD often do not disclose appearance related concerns, instead reporting non-appearance related symptoms – e.g. low mood and social isolation – often resulting in a diagnosis of social anxiety or depression, and a failure to recognise either a primary diagnosis or comorbid BDD (Veale et al., 2015). Indeed, around 1 in 10 individuals with BDD will remain undiagnosed, instead receiving alternative diagnoses such as mood disorder or bipolar disorder (Conroy et al., 2008). Given the chronic under recognition of BDD and the high degrees of functional impairment associated with the condition, improving diagnostic accuracy in relation to BDD is imperative.
Further complicating diagnostic accuracy are the high rates of comorbidity in individuals with BDD. Research has demonstrated elevated rates of obsessive-compulsive, mood, anxiety and personality disorders (Frías et al., 2015; Phillips et al., 2005). BDD also shares high rates of comorbidity with anorexia nervosa (AN; Hartman et al., 2013), with both disorders sharing substantial body image dissatisfaction and obsessive preoccupations (Ruffolo et al., 2006). To further complicate matters, AN has been associated with high rates of comorbidity with autism spectrum disorder (ASD; Boltri and Sapuppo, 2021) and attention-deficit hyperactivity disorder (ADHD; Nazar et al., 2016), with emerging evidence suggesting shared underlying cognitive dysfunction across AN, ASD and ADHD (Inal-Kaleli et al., 2025; Levin and Rawana, 2016). As well as phenomenological similarities, BDD and AN share a number of cognitive features, for example facial emotion recognition difficulties and cognitive inflexibility (Hartmann et al., 2013; Holmes À Court et al., 2025; Miles et al., 2020), suggesting there may be similar overlap across BDD, ASD and ADHD. Despite this, investigation into neurodevelopmental comorbidity in BDD remains relatively unexplored.
Autism, ADHD and diagnostic overshadowing
Diagnostic overshadowing (DO) occurs when symptoms of a co-occurring illness are attributed to a pre-existing diagnosis, resulting in neglecting consideration of alternative pathogenetic mechanisms (Volkmar, 2020). This negative bias impacts clinical judgement and can lead to a failure to acknowledge comorbid conditions. DO has been demonstrated in individuals with ASD and ADHD (Hoover et al., 2020). For example, DO has contributed to under-recognition of the effects of trauma in individuals with ASD, and this has been attributed in part to overlapping symptomology such as temper tantrums, overactivity and sleep problems (Hoover et al., 2020). Moreover, there is also considerable overlap across ADHD and post-traumatic stress disorder (PTSD) symptoms (Hoover et al., 2020). Symptom overlap and heterogeneous presentations complicate diagnostic accuracy in first recognising, then diagnosing comorbid conditions in people with neurodevelopmental disorders (Rosen et al., 2018).
Given that similar clinical presentations across disorders may contribute to DO, clarifying symptom overlap between BDD and other disorders is critical to understanding whether DO contributes to the underdiagnosis of BDD. Moreover, the diagnostic criteria for BDD underwent significant changes in the Diagnostic and Statistical Manual of Mental Disorders (5th ed.; APA, 2013). For example, a new criterion was introduced specifying that repetitive behaviours and/or mental acts must be present. This inclusion may increase diagnostic accuracy, aiding in the differentiation between BDD and some disorders, such as depression and anxiety (Krebs et al., 2017). However, it may have inadvertently given rise to increased symptom overlap with other disorders such as ASD or ADHD, especially as a recent body of research indicated a significant relationship between neurodevelopmental disorders and body dissatisfaction (Baraskewich and Climie, 2022; Longhurst, 2023; Van Eck et al., 2018). This may lead clinicians to incorrectly attribute body image symptomology to ASD and ADHD without further investigation. Thus, while disorder-specific, differentiating characteristics can assist diagnostic differentiation, when neurodevelopmental conditions exist, overlapping symptomology may present a significant barrier to diagnosing comorbid BDD.
BDD and neurodevelopmental disorders
Although there has been no direct investigation into neurodevelopmental comorbidity in BDD, incidental findings have emerged (Grant et al., 2019; Lee et al., 2023). For example, Enander et al. (2018) investigated the heritability of body dysmorphic symptoms in a large twin sample (
Cognitive and perceptual overlap
Furthermore, a range of overlapping features in certain cognitive and perceptual domains has been demonstrated across BDD, ASD and ADHD. For example, all three disorders have been associated with rigid thinking patterns and cognitive rigidity (Halleland et al., 2012; Holmes À Court et al., 2025; Lage et al., 2024; Malcolm et al., 2021; Rohlf et al., 2012; Westwood et al., 2016; Xie et al., 2020). Inattention and reduced working memory capacity have also been reported across all three disorders (Alderson et al., 2013; Demetriou et al., 2018; Malcolm et al., 2021; Townes et al., 2023; Wang et al., 2017; Willcutt et al., 2005). Similarly, comparable atypical visual processing (Cohen et al., 2019; Holmes À Court et al., 2025; Muth et al., 2014; Virgili et al., 2024) and facial emotion recognition challenges (Beilharz et al., 2017; Bora and Pantelis, 2016; Borhani and Nejati, 2018; Holmes À Court et al., 2025; Lozier et al., 2014; Uljarevic and Hamilton, 2013; Virgili et al., 2024; Yeung, 2022) have been demonstrated across all three disorders.
This cognitive and perceptual overlap may lead to similar clinical presentation across BDD, ASD and ADHD. Moreover, clinicians may attribute BDD-related symptoms to already diagnosed neurodevelopmental disorders, which are more likely be associated with ASD and ADHD than BDD based on clinician training and familiarity.
The present study
Given the lack of prior research investigating neurodevelopmental comorbidity in individuals with BDD, we aimed to investigate the frequency of ASD and ADHD comorbidity in BDD in a large population-based sample. In addition, in view of the cognitive and symptom overlap demonstrated across BDD, ASD and ADHD, we aimed to examine the likelihood that individuals with ASD and ADHD would have possible undiagnosed BDD. Specifically, we proposed the following hypotheses:
(H1) Individuals with BDD will have a higher frequency of neurodevelopmental diagnoses than individuals without BDD.
(H2) Individuals with the following diagnoses will be more likely to have possible undiagnosed BDD (as measured by the Dysmorphic Concern Questionnaire):
a.ASD
b.ADHD
c.ASD + ADHD
Method
Design
The study utilised an observational correlational design with data collected at a single time point using a self-report online survey.
Participants
Recruitment
Participants were recruited through a range of methods, including social media advertising (e.g. Facebook and Twitter), snowball recruitment, public noticeboard advertisement and word of mouth (
Inclusion/exclusion criteria
Eligibility criteria included being 18 years or older and residing in Australia.
Measures
National body image survey (NBIS)
The NBIS was developed by Professor Susan Rossell of Swinburne University of Technology, Centre for Mental Health and Brain Sciences. Data were collected for a range of variables, including demographic information, details on mental health diagnoses (as confirmed by a clinician), which included diagnosis of BDD, ASD and ADHD, current and past body image concerns (using the
DCQ
The DCQ is a 7-item self-report measure designed to assess appearance concerns and related distress, along with associated behaviours (Oosthuizen et al., 1998). Items are rated on a 4-point Likert-type scale ranging from 0 (not at all) to 3 (much more than most people). Total scores range from 0 to 21, with higher scores on the DCQ indicating higher levels of dysmorphic concern. The DCQ is considered a valid screening tool for BDD with good reliability (Cronbach’s α = 0.81 to 0.88; Oosthuizen et al., 1998; Schieber et al., 2018) and has been utilised in multiple studies as an indicator of BDD diagnosis (e.g. Enander et al., 2018; Gieler et al., 2016). The current study utilises a general population sample; thus, a DCQ cutoff value of ⩾ 11 was utilised as an indicator of
Procedure
The NBIS was available online through the Qualtrics platform from 2 August 2023 to 17 February 2024. The survey was undertaken at the participants’ convenience and took approximately 15 minutes to complete.
Statistical analyses
All data analyses were performed using IBM SPSS Statistics (Version 30, 2024). Descriptive statistics were reported for the sample demographics using frequencies and percentages. To examine H1, a Fisher’s Exact Test with BDD diagnosis as the predictor variable and diagnosis (ASD, ADHD or ASD + ADHD) as the outcome variable was performed.
To examine H2, we performed a binary logistic regression with diagnosis (ASD, ADHD or ASD + ADHD) as the predictor variable and possible BDD (as measured by a score of ⩾ 11 on the DCQ) as the outcome variable. Biological sex, age, education and socioeconomic status (SES) may differ across disorders, and were therefore entered as covariates in the regression analysis. All covariates were entered simultaneously into the model.
A receiver operating characteristic (ROC) analysis was conducted to evaluate the ability of the logistic regression model to discriminate between participants with and without possible undiagnosed BDD (DCQ ⩾ 11). The area under the curve (AUC) was calculated to assess overall model accuracy.
Data cleaning
In total,
Results
Prior to analysis, data were examined to assess compliance with relevant statistical assumptions, with no violations identified. Sample 1 (utilised to test H1) consisted of
Sample 2 (utilised to test H2) consisted of
Sample demographics.
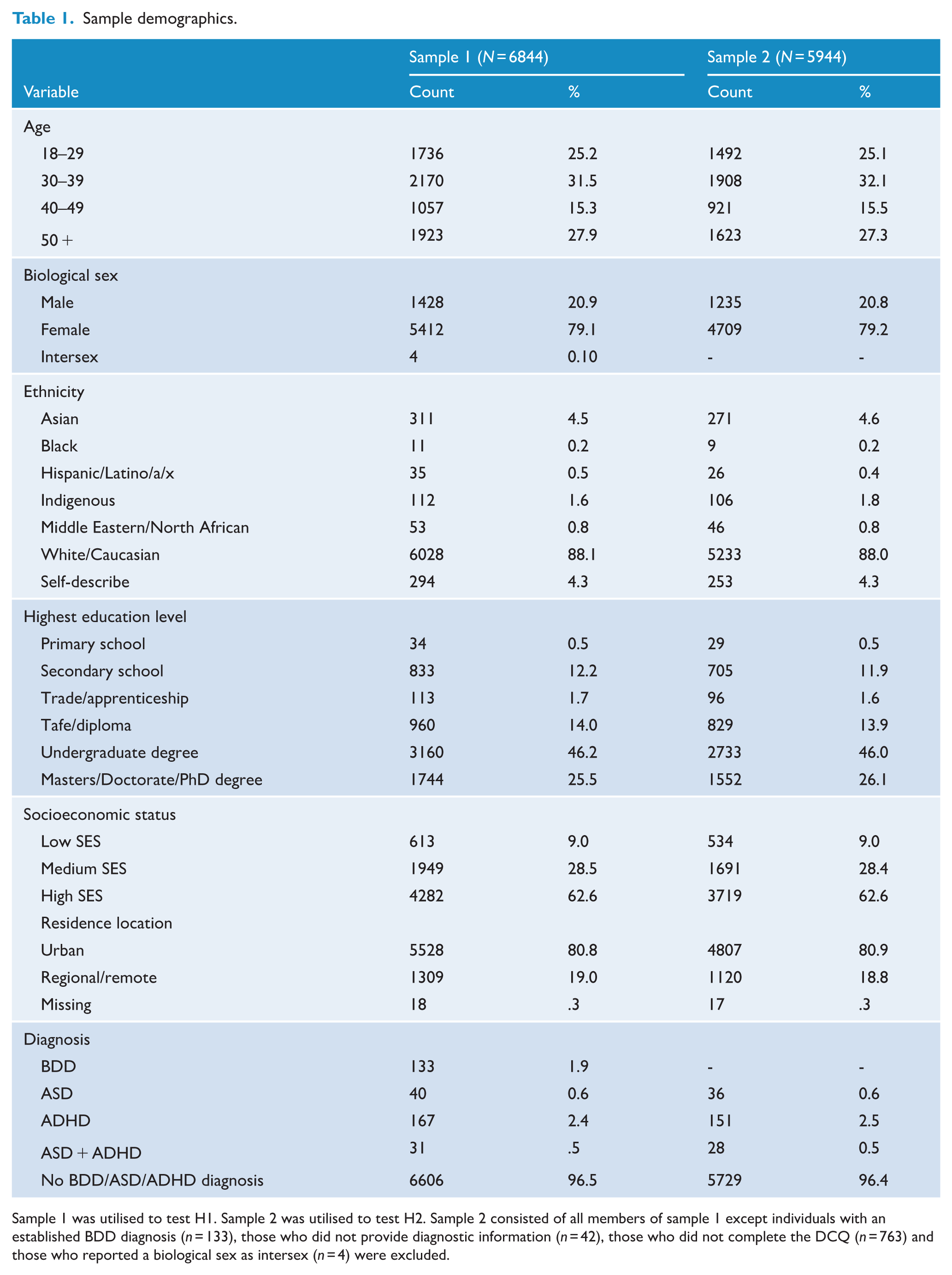
Sample 1 was utilised to test H1. Sample 2 was utilised to test H2. Sample 2 consisted of all members of sample 1 except individuals with an established BDD diagnosis (
Hypothesis testing
H1: association between BDD diagnosis and neurodevelopmental diagnoses
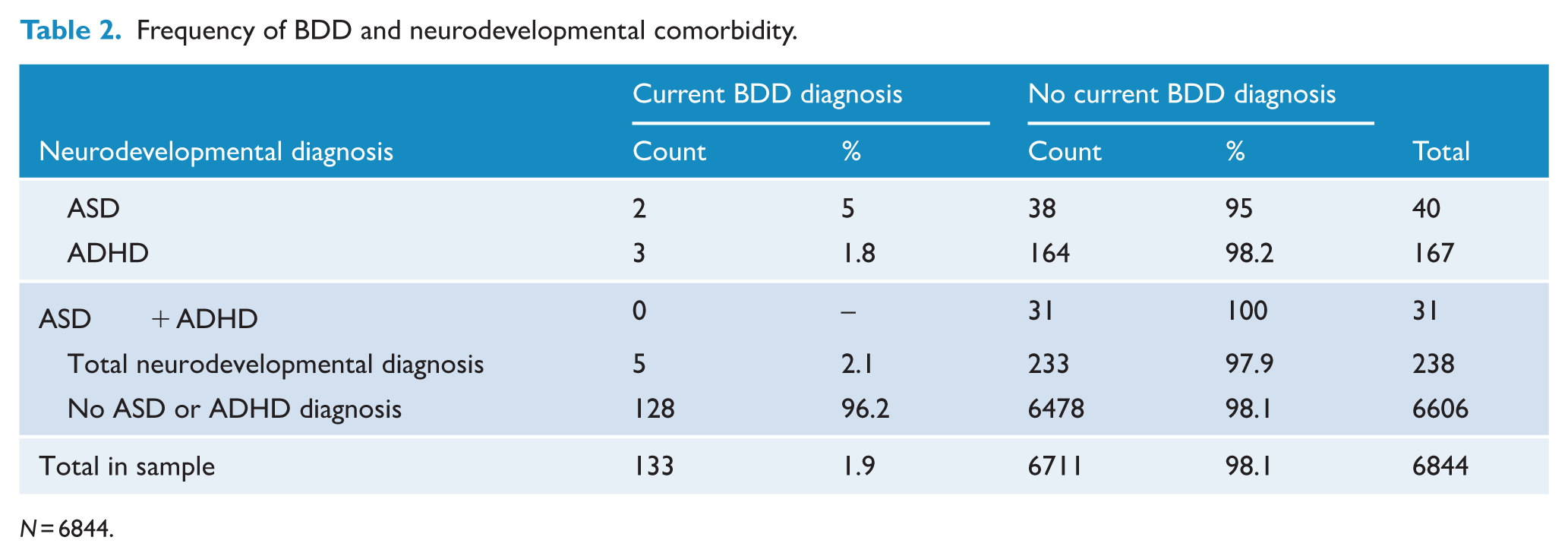
The frequencies for BDD and neurodevelopmental comorbidity are presented in Table 2. In the sample of 6844 participants, 133 (1.9%) reported a current BDD diagnosis, 40 (0.6%) reported an ASD diagnosis, 167 (2.4%) reported an ADHD diagnosis and 31 (0.5%) reported having been diagnosed with both ASD and ADHD. The Fisher’s Exact Test indicated no significant association between BDD diagnosis and ASD, ADHD or ASD + ADHD diagnosis,
Frequency of BDD and neurodevelopmental comorbidity.
H2: association between neurodevelopmental diagnosis and possible undiagnosed BDD in individuals without an established BDD diagnosis
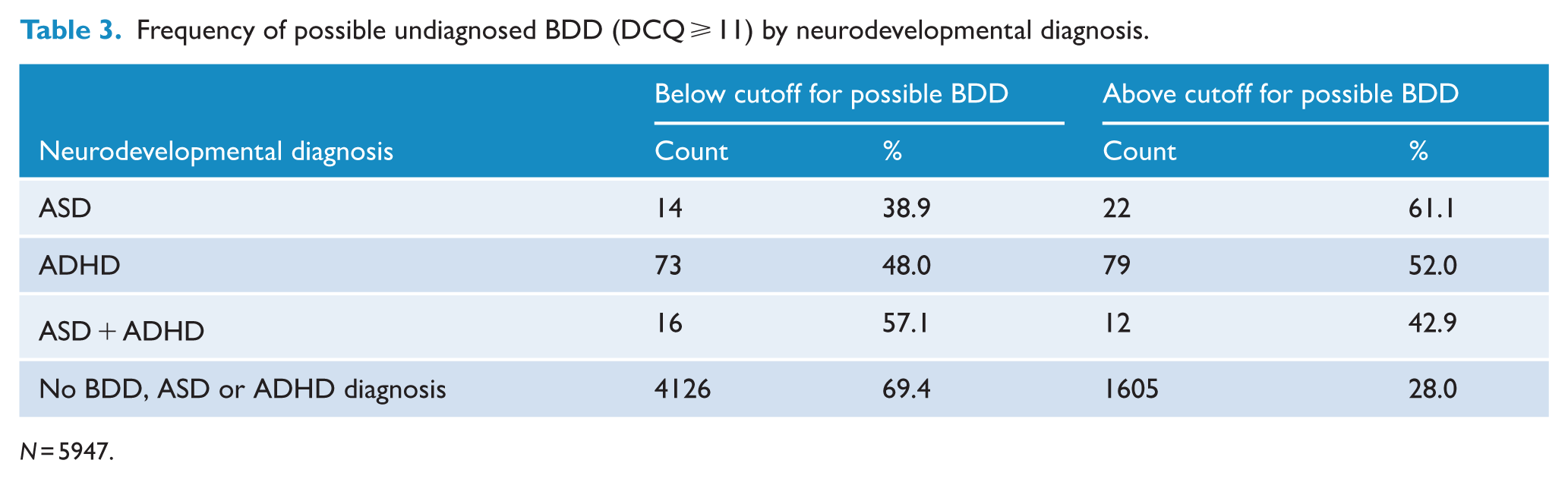
The frequency of possible undiagnosed BDD by neurodevelopmental diagnosis is presented in Table 3. The results of the binary logistic regression are presented in Table 4. The overall model was significant χ2(14,
Frequency of possible undiagnosed BDD (DCQ ⩾ 11) by neurodevelopmental diagnosis.
Logistic regression results for predicting possible undiagnosed BDD (DCQ ⩾ 11) in those without a BDD diagnosis.
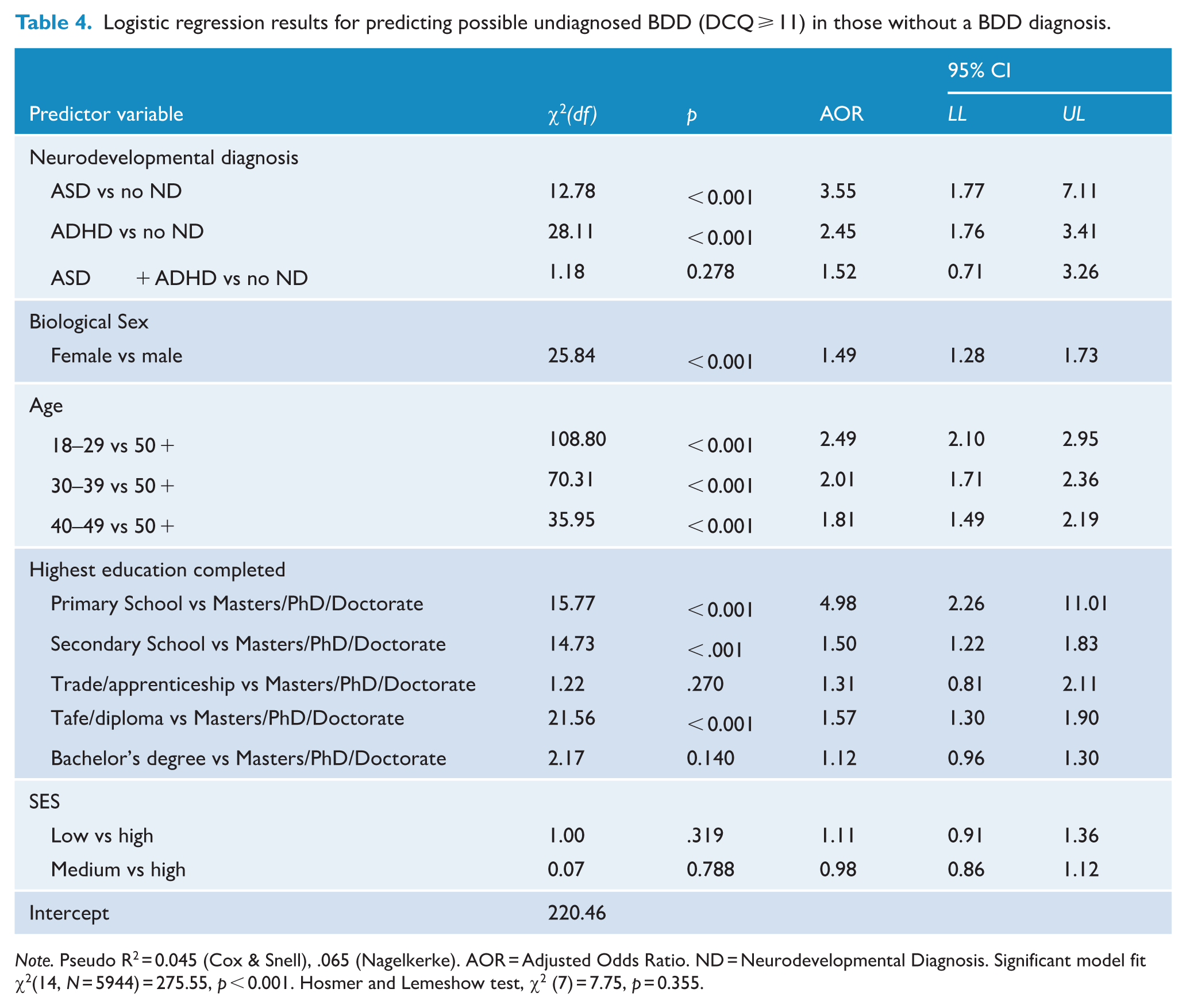
In the binary logistic regression model, individuals with a diagnosis of ASD were on average 3.55 times more likely to have possible undiagnosed BDD than those without a neurodevelopmental diagnosis (95% CI: 1.77, 7.11). Individuals with a diagnosis of ADHD also had significantly higher odds of having possible undiagnosed BDD than those without a neurodevelopmental diagnosis (AOR = 2.45, 95% CI: 1.76, 3.41). However, the odds of having possible undiagnosed BDD for individuals with ASD + ADHD were not significantly different from those without a neurodevelopmental diagnosis.
A ROC analysis with a DCQ score cut-off of 11 revealed an AUC of 0.63, 95% CI [0.62, 0.65],
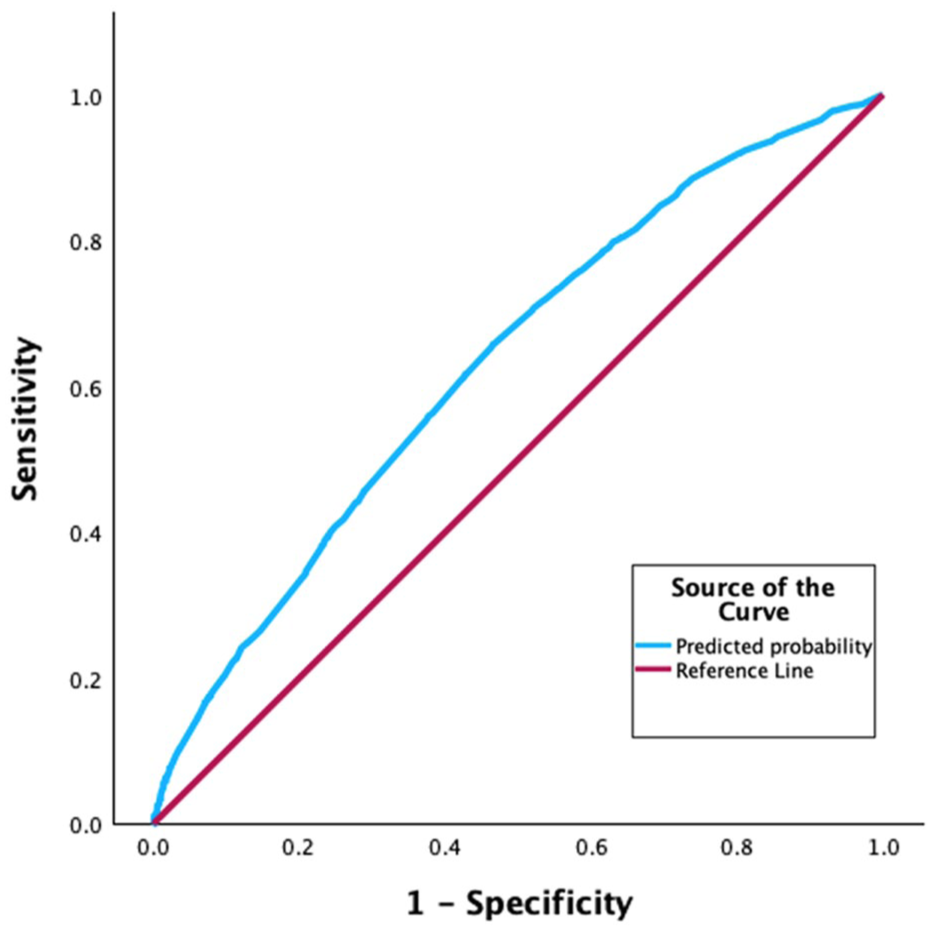
Reciever operating characteristic (ROC) showing model discrimination for those with and without possible undiagnosed BDD utilising DCQ ⩾ 11.
All the covariates except SES were significant in the binary logistic regression analysis, indicating that these variables (except perhaps SES) should be controlled for in future studies of this nature.
Given the novelty and exploratory nature of this work a-priori power calculations were not deemed appropriate. However, the following post hoc power analyses were conducted using G-Power 3.1.9.7 to provide evidence of the adequacy of the sample size. In the case of H1, the sample sizes for people with and without a BDD diagnosis were sufficient to detect, with 76% power, a 5% increase in the likelihood of neurodevelopmental disorders in people with a BDD diagnosis. Only an increase of 0.0029 was detected, hence the lack of a significant result.
In the case of H2, the sample sizes for people with and without a neurodevelopmental diagnosis were sufficient to detect:
a)with 78% power for ASD, a 21% increase in the likelihood of a DCQ score above the cutoff for possible BDD
b)with 79% power for ADHD, a 10% increase in the likelihood of a DCQ score above the cutoff for possible BDD
c)with 78% power for ASD+ADHD, a 24% increase in the likelihood of a DCQ score above the cutoff for possible BDD
In the case of (c), only an increase of 14.9% was detected, hence the lack of a significant result.
Discussion
The aim of the current study was to investigate the frequency of ASD and ADHD comorbidity in people who report a diagnosis of BDD in a large nationwide convenience sample. Our first hypothesis was not supported, with rates of BDD diagnoses not associated with rates of ASD, ADHD or ASD + ADHD diagnoses. A further aim was to examine the likelihood that individuals with ASD and ADHD could have possible undiagnosed BDD. This was tested with a second hypothesis – that individuals with ASD and/or ADHD would be more likely to have undiagnosed BDD (as measured by the DCQ) than individuals without a neurodevelopmental diagnosis. This hypothesis was partially supported. As predicted, individuals with ASD (AOR: 3.55) and ADHD (AOR: 2.45) were significantly more likely to have possible undiagnosed BDD than those without one of these neurodevelopmental disorders. However, contrary to expectations, the hypothesis that individuals with ASD + ADHD were not more likely to have possible undiagnosed BDD was not supported with the available data.
Our finding of no significant association between BDD diagnoses and neurodevelopmental diagnoses is in contrast with previous research that has found increased rates of ASD and ADHD comorbidity in individuals with BDD (Enander et al., 2018; Grant et al., 2019; Lee et al., 2023). Of note, Enander et al. (2018) and Grant et al. (2019) utilised BDD screening tools, such as the DCQ, where participants who scored over a threshold were indicated to have a BDD diagnosis, whereas the current study asked participants whether they had received a BDD diagnosis from a mental health professional. Our more stringent criterion for indicating a BDD diagnosis may partly explain the contrasting results. Studies utilising only screening tools as an indicator of BDD, without confirming diagnosis through clinical interviews, may overestimate comorbidity rates. As in the current study, Lee et al. (2023) classified participants using formal diagnoses. However, that study had an extremely large sample (
Despite the samples of the current study being overwhelmingly female, the rates of neurodevelopmental disorders across biological sexes were similar. For example, the rate of ASD was 0.8% for biological males and 0.6% for biological females. Similarly, the rate of ADHD was 2.1% for biological males and 2.8% for biological females. These figures are in line with research indicating sex ratios for ADHD adjust to around 1:1 in adulthood (da Silva et al., 2020). However, for ASD in adulthood, the male-to-female ratio is around 2.5:1 (Posserud et al., 2021). The overrepresentation of females in our sample likely contributed to the observed sex distribution for ASD and likely does not reflect ratios in the general population.
The finding of an elevated likelihood of undiagnosed BDD in individuals with ASD and ADHD is novel. No other study has investigated possible undiagnosed BDD in neurodevelopmental populations. Our findings lend support to previous research that has demonstrated elevated body dissatisfaction in people with neurodevelopmental disorders (Baraskewich and Climie, 2022; Bisset et al., 2019; Longhurst, 2023; Van Eck et al., 2018). Our findings further suggest that this elevated body image concern in ASD and ADHD is either being missed or attributed to neurodevelopmental diagnoses, as part of DO. Our non-significant finding for the ASD + ADHD group could be due to the small number of individuals with ASD + ADHD in the sample (
Taken together, our findings lend tentative support for DO occurring in ASD and ADHD; further investigation is warranted to clarify if DO is occurring in neurodevelopmental populations. The fact that we did not demonstrate heightened BDD and neurodevelopmental comorbidity (when it might be predicted based on other related-mental health conditions with extreme body dissatisfaction, such as in anorexia nervosa), coupled with the significantly increased likelihood of possible undiagnosed BDD in these populations, suggests that there is under-recognised and under-treated body image concern in individuals with ASD and ADHD. Moreover, the mean age of diagnosis for ASD is around 5 years (van’ t Hof et al., 2021) and for ADHD around 7 years (Caci et al., 2015). BDD symptoms do not usually begin until around 17 years (Bjornsson et al., 2013). Thus, it is likely that BDD symptoms emerge after initial neurodevelopmental diagnosis, potentially increasing the likelihood of DO and missed BDD diagnoses. Further, psychoeducation for health practitioners is arguably more robust for more well-known disorders such as ASD and ADHD, as evidenced by practitioner-related knowledge barriers reported by those experiencing BDD (Rossell et al., 2023). Collectively, these factors may contribute to BDD being missed as a co-occurring condition in neurodevelopmental populations.
Our findings also tentatively lend further support to BDD remaining a disorder that is underdiagnosed and under-recognised in clinical settings. Of participants who did not report a BDD diagnosis, around 29% scored above the validated cutoff for possible BDD. This suggests that there is a substantial number of individuals in the community with significantly elevated body image concern who may meet diagnostic criteria for BDD, yet do not have a BDD diagnosis.
Implications for clinical practice
The results of the current study provide preliminary evidence that the co-occurrence of BDD may be elevated in neurodevelopmental populations. If replicated, these results have significant implications for clinical practice, some of which are addressed below.
Training and awareness of BDD
A significant barrier to the recognition of BDD is the lack of awareness of the disorder among clinicians (Castle et al., 2020). BDD sufferers have described significant delays in receiving treatment due to clinicians not being aware of what BDD is or how to support those affected (Rossell et al., 2023). In addition, shame and embarrassment, exacerbated by concern about being judged, are common reasons why affected individuals choose not to disclose their BDD symptoms (Conroy et al., 2008). These concerns can be mitigated by effective and compassionate probing into body image concerns, fostered by improved clinician training in and increased awareness of BDD. Research has indicated that affected individuals are not likely to disclose body image concerns unless asked directly (Veale et al., 2015). Thus, clinicians probing with questions such as ‘Do you have any concerns about your appearance or how you look?’ may support disclosure (Rossell et al., 2023).
This lack of awareness among clinicians is mirrored in the general population (Rossell et al., 2023). Many of those experiencing BDD believe their condition is cosmetic rather than psychological in nature (Rossell et al., 2023), leading to a failure to seek mental health treatment. Thus, better education in the community about BDD and its symptoms is needed to support affected individuals in seeking psychological treatment. Initiatives such as public awareness campaigns through television and social media may support individuals in recognising they are experiencing a mental health condition and empower them to seek psychological support. This increased awareness may reduce some of the stigma and shame attached to BDD and body image concerns, enabling affected individuals to disclose their symptoms to those close to them as well as their treating clinicians.
Improved screening and assessment
It is possible that the cognitive and perceptual overlap present across BDD, ASD and ADHD leaves individuals with neurodevelopmental disorders at increased risk of developing BDD. For example, the tendency to focus on specific details found in individuals with ASD and ADHD may make them more likely to fixate on minor appearance flaws. In addition, associated cognitive rigidity may lead to difficulty in shifting focus away from these perceived imperfections. Therefore, it is imperative to include measures like the DCQ in neurodevelopmental assessments to screen for body image concern. Moreover, given the early age at which neurodevelopmental disorders are usually diagnosed and the heterogeneous developmental trajectories of mental disorders in young people (Plana-Ripoll et al., 2019), it is important to screen individuals with ASD and ADHD for co-occurring disorders, including BDD, throughout their lifespan.
Considering the high rates of comorbidity found in individuals with neurodevelopmental disorders, improved training for clinicians is required to highlight the potential for DO in these populations. Clinicians need to be made aware of possible symptom overlap and analogous clinical presentation across disorders and be empowered to investigate potential co-occurring conditions. This includes being sensitive to what symptoms
Limitations and future directions
A limitation of the current study is the self-report nature of diagnosis information. Without clinical verification, it is possible diagnosis information was inaccurately reported. However, previous research has suggested self-reported psychiatric diagnoses are reasonably accurate (Sanchez-Villegas et al., 2008). A further limitation is potential sampling bias, consequent upon the use of social media and snowball recruitment strategies. Both recruitment channels might have led to self-selection and similarity bias, resulting in an unrepresentative sample. The sample was overwhelmingly female, Caucasian and highly educated; therefore, our results may not generalise to populations with differing demographics. Thus, the associations demonstrated in this study may not generalise to predominantly male, non-Caucasian individuals. In addition, the survey was advertised as being focused on body image, which could have selectively attracted individuals struggling with body image issues. This could mean the estimates of individuals with body image concerns in community samples are exaggerated. However, this potentially elevated body image concern would be equal across groups, and thus unlikely to account for the observed effect. Also, the survey was not advertised as investigating comorbidity patterns, neurodevelopmental diagnoses and body image issues. This is a strength of the current study as it avoided self-selection by participants with neurodevelopmental diagnoses. A further strength is that the results maintained significance with little change to odds ratios when a higher validated cutoff of ⩾ 14 on the DCQ was applied, suggesting the effect found is reliable and not an artefact of the chosen threshold for possible undiagnosed BDD.
The current study is a preliminary investigation into associations between BDD and neurodevelopmental disorders, and further investigation into these associations should be undertaken in larger, more representative samples. This could be achieved by adding the DCQ and diagnostic information to already established surveys to minimise self-selection bias. Moreover, future studies should investigate undiagnosed BDD in neurodevelopmental populations with clinical interviews as an adjunct to measures like the DCQ to ascertain actual undiagnosed BDD rates. Finally, future studies should include dimensional measures of autism and ADHD to verify self-reported diagnoses and explore potential associations with BDD. Given the cognitive and perceptual overlap present across BDD, ASD and ADHD, future research should further investigate this overlap to clarify phenomenological similarities and distinguishing features. This research will aid in refining nosology and improving diagnostic precision.
Conclusions
The current study explored the frequency of ASD and ADHD comorbidity in BDD in a large convenience sample. In addition, this study was the first to explore possible undiagnosed BDD in neurodevelopmental populations. The results cautiously suggest that individuals with ASD and ADHD are significantly more likely to have possible undiagnosed BDD, or at the very least elevated dysmorphic concern, than individuals without a neurodevelopmental diagnosis. Moreover, our results tentatively support the presence of DO in neurodevelopmental populations. BDD is a chronically under-recognised disorder, and uncovering potential barriers to BDD diagnosis is imperative to addressing these impediments and raising awareness of BDD. Future research should investigate undiagnosed BDD in neurodevelopmental populations, along with the similarities and differences in symptomology across disorders, with the aim of improving diagnostic accuracy.
Supplemental Material
sj-docx-1-anp-10.1177_00048674261418840 – Supplemental material for Body dysmorphic symptoms in autism and attention-deficit hyperactivity disorder: A comorbidity study
Supplemental material, sj-docx-1-anp-10.1177_00048674261418840 for Body dysmorphic symptoms in autism and attention-deficit hyperactivity disorder: A comorbidity study by Janelle Janus, Denny Meyer, Ella Byrne, Katrina Holmes à Court, David J Castle and Susan L Rossell in Australian & New Zealand Journal of Psychiatry
Footnotes
Author Contributions
SLR conceived the project and the design. All authors SLR, JJ, DM, EB, KAH and DJC finalised the design, constructed the survey, obtained ethics, engaged in data collection and interpretation of findings. JJ completed all the data analysis in consultation with other authors. All authors prepared the manuscript and agreed to its final form.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: Susan L Rossell reports that financial support was provided by NHMRC; she held an NHMRC Senior Research Fellowship during the conduct of this work. Other authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This research was funded by an NHMRC Senior Research Fellowship awarded to SLR (GNT1154651).
Ethical Considerations
The study was approved by the Swinburne Research Ethics Committee Ref. 2023720-16451.
Consent to Participate
Written consent was obtained from all participants, and study procedures were conducted in accordance with the Declaration of Helsinki.
Consent for Publication
Not applicable.
Data Sharing
This dataset is available on request by qualified researchers/scientists. Requests require a concept proposal describing the purpose of data access, appropriate ethical approval and provision for data security. All data analysis scripts and results files are available for review.
Supplemental Material
Supplemental material for this article is available online.
