Abstract
Objective:
The current Guidelines aim to provide evidence-based management recommendations for treatment of people living with schizophrenia in Australia and Aotearoa New Zealand.
Methods:
The Australian and New Zealand Journal of Psychiatry (ANZJP) commissioned a panel of experts to establish these Guidelines. The existing literature was reviewed to address key health questions. The certainty of evidence was evaluated using the Grading of Recommendations, Assessment, Development and Evaluations (GRADE) approach, and the strength of the recommendation was determined by the panel.
Results:
The ANZJP GRADE Guidelines examined the current evidence base for a range of areas relevant to treatment for people with schizophrenia including: initial physical health assessment; pharmacological treatment; psychological and psychosocial interventions; family, whānau and carers; psychiatric comorbidities; physical health and lifestyle interventions; and special populations.
Conclusions:
It is hoped that the current Guidelines provide useful recommendations in important aspects of care for people living with schizophrenia and their family, whānau and carers in Australia and Aotearoa New Zealand, both at the individual and systemic levels.
Keywords
Introduction
The current Guidelines were commissioned by the Australian and New Zealand Journal of Psychiatry (ANZJP). These Guidelines are informed by the two previous Royal Australian and New Zealand College of Psychiatrists (RANZCP) clinical practice guidelines (Galletly et al., 2016; Royal Australian and New Zealand College of Psychiatrists Clinical Practice Guidelines Team for the Treatment of Schizophrenia and Related Disorders, 2005). In formulating the recommendations, we employed the Grading of Recommendations, Assessment, Development and Evaluations (GRADE) approach to develop and evaluate the key health questions (Guyatt et al., 2011a, 2011b; Lewin et al., 2015). In areas where there was an absence of quantitative research available, the GRADE-CERQual (Confidence in the Evidence from Reviews of Qualitative research) (Lewin et al., 2015) and the World Health Organization Qualitative Evidence Synthesis Framework (Downe et al., 2019) approaches were used to synthesise the evidence from qualitative studies. As such, the current Guidelines are more limited in scope than previous RANZCP clinical practice guidelines. It is important to emphasise that schizophrenia is a syndromal diagnosis with considerable heterogeneity in both the presentation and prognosis. While we hope that the current Guidelines help inform the delivery of care for people living with schizophrenia, each clinician needs to apply evidence appropriately and specifically to each person with schizophrenia within the context in which they work (Greenhalgh et al., 2014). The current Guidelines should be used in conjunction with an individualised, collaborative formulation to provide the optimal care for the person living with schizophrenia and their family, whānau and carers.
Schizophrenia
Schizophrenia is characterised by psychotic symptoms (i.e. delusions, hallucinations and disorganised thinking and speech), negative symptoms (including reduced emotional expression or motivation), cognitive impairment and abnormal motor behaviour (Jauhar et al., 2022; McCutcheon et al., 2020). The symptom profile and severity of symptoms can vary between individuals. The diagnostic criteria for the two main international classification systems (American Psychiatric Association, 2022; World Health Organization, 2022) are outlined in Table 1. Despite the ever-expanding body of work in the area (Jauhar et al., 2022), no objective measures, such as serum markers, neuroimaging and cognitive testing, are confirmatory in diagnosing schizophrenia. Thus, the diagnosis of schizophrenia remains clinical, based on comprehensive psychiatric assessment, collateral information and exclusion of other conditions that may imitate similar symptoms. The lifelong prevalence of schizophrenia is estimated to be around 0.7% globally (McGrath et al., 2008), with variations observed depending on study design, geographic region, time of assessment and study quality, as well as case identification methods. While these estimates remain relatively stable (Simeone et al., 2015), the raw global prevalence (14.2–23.6 million) and associated disability-adjusted life years (9.1–15.1 million) have increased significantly since 1990 (Solmi et al., 2023b). These findings indicate that although schizophrenia remains a low-prevalence disorder, its burden of disease globally is substantial and increasing (Charlson et al., 2018).
Diagnostic criteria for schizophrenia in DSM-5-TR and ICD-11.

DSM-5-TR = Diagnostic and Statistical Manual of Mental Disorders (5th ed., text rev.); ICD-11 = International Classification of Diseases, 11th Revision.
General principles in working with people with schizophrenia
Shared decision-making
A shared decision-making process of ensuring that the person with schizophrenia is the primary driver for the care should be implemented as much as possible. Whenever appropriate, their family, whānau and carers should be invited and involved in their care. Person-centred care is an important element in building strong and inclusive mental health systems and service cultures that value diversity and promote safety in service contact for all in the community. This also includes treating people from diverse backgrounds with appropriate cultural sensitivity within a culturally informed care model.
Working with family, whānau and carers
All mental health professionals, including psychiatrists and psychiatry trainees, should be mindful of how we work with family, whānau and carers to build our capability in family-focused care, and improve our confidence in managing confidentiality issues. We should also be proactive in recognising the emotional support needs of family, whānau and carers, while maintaining respect for the privacy and rights of the person receiving care. This is a crucial element in building strong and inclusive mental health systems and service cultures.
Adequate training of clinicians
Psychological, psychosocial and lifestyle interventions recommended in these Guidelines need to be provided by adequately trained and/or credentialed clinicians with appropriate supervision.
Medication considerations
When trialling a new medication, the dose should be reviewed proactively and adjusted according to its effectiveness and side effects in collaboration with the person with schizophrenia. The optimal dose should be the lowest effective dose with a tolerable adverse effect profile, taking into account the individual’s age, sex and ethnicity, as pharmacokinetic and pharmacodynamic differences exist among demographic groups (Halstead et al., 2025; McCutcheon et al., 2025). If an agent’s intolerability exceeds benefits, then switching to another medication should be considered. If another psychotropic agent is added for augmentation or combination, then both the effectiveness and tolerance should be monitored regularly. The additional agent should be withdrawn if no benefits are seen within 3 months to reduce the risks associated with polypharmacy. In accordance with existing national and international guidelines (Firth et al., 2019; Galletly et al., 2016; Halstead et al., 2025; Lambert et al., 2017; McCutcheon et al., 2025), people with schizophrenia should be screened and monitored for cardiometabolic abnormalities and modifiable lifestyle risk factors (e.g. substance use, physical inactivity, poor diet) regularly. This should begin from the outset of diagnosis and antipsychotic medication prescription, especially given the increased prevalence of multimorbidity observed among young adults with schizophrenia (Halstead et al., 2024a, 2024b). In addition, non-pharmacological and lifestyle interventions should also be offered at treatment commencement and all stages of treatment for people with schizophrenia to both enhance the effectiveness of interventions provided and mitigate the physical side effects of pharmacotherapy.
Methods
Guidelines panel
The authorship group (the Panel) was developed through the identification of clinical experts across Australia and Aotearoa New Zealand, including a psychiatry trainee and experts with lived experience. The Panel first met in January 2024 and subsequently met on a monthly to bi-monthly frequency, chaired by the senior author (D.S.). Representation across psychiatric career stages, including trainee representation, was considered in selecting the Panel. Potential conflicts of interest were documented, and management of such was discussed by consensus (Table 2).
Potential conflicts of interest.
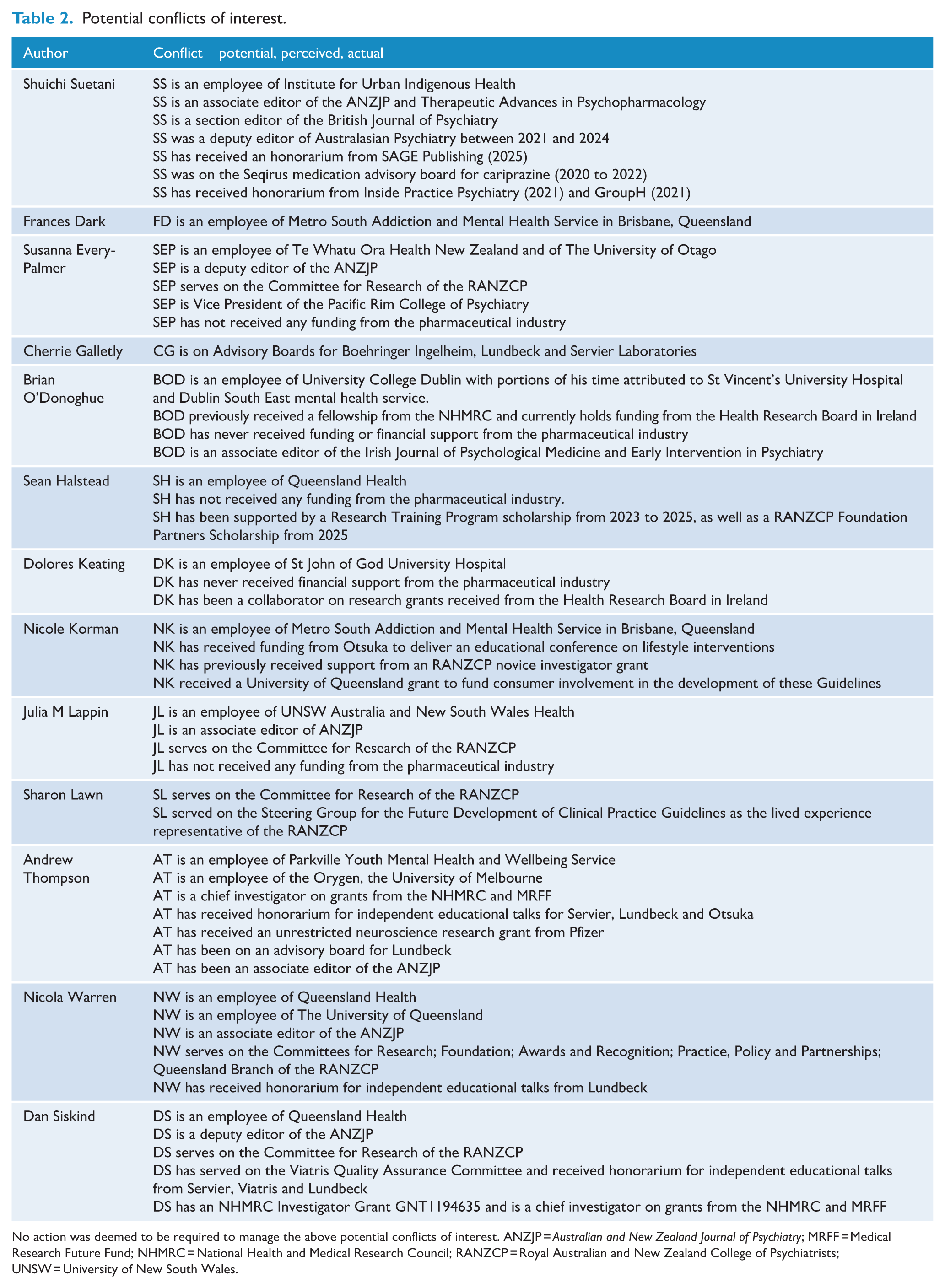
No action was deemed to be required to manage the above potential conflicts of interest. ANZJP = Australian and New Zealand Journal of Psychiatry; MRFF = Medical Research Future Fund; NHMRC = National Health and Medical Research Council; RANZCP = Royal Australian and New Zealand College of Psychiatrists; UNSW = University of New South Wales.
Consumer consultation group
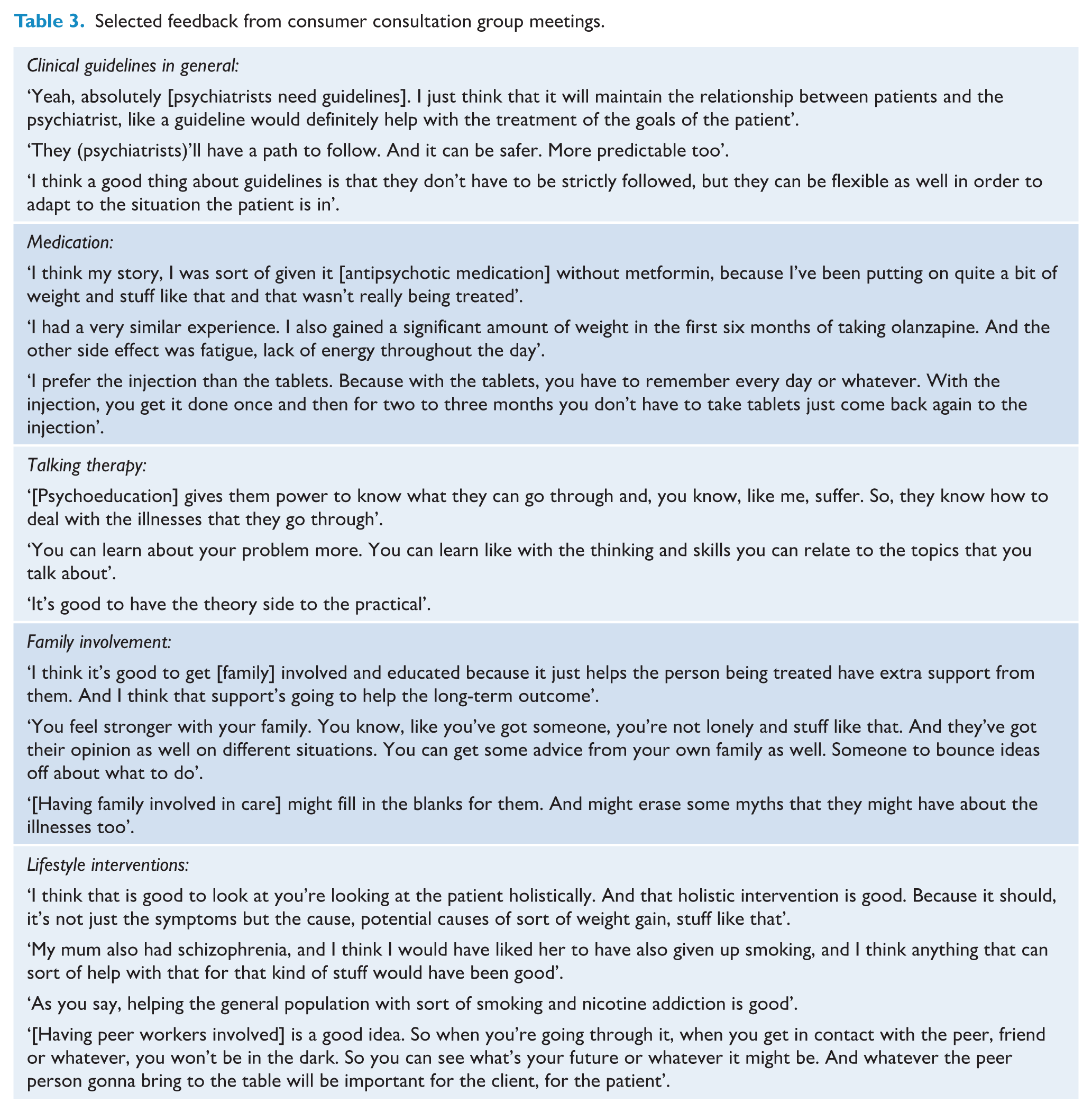
The consumer consultation group comprised seven individuals with lived experience of schizophrenia. Four meetings were held between August 2024 and August 2025, each attended by three to four group members alongside two Panel members (S.S. and N.K.). These meetings sought targeted input on the formulation of topics for inclusion in the Guidelines and the development of key health questions. Draft findings and recommendations were presented to the group for feedback and refinement. Table 3 summarises selected feedback provided during these consultations.
Selected feedback from consumer consultation group meetings.
GRADE methodology
In short, the GRADE approach provides a structured method to assess the level of certainty in the existing evidence base (very low, low, moderate or high) and the strength of the recommendation (weak or strong) (Guyatt et al., 2011a; Prasad, 2024) (Table 4). For section 4 (Family, whānau and carer involvement), the GRADE-CERQual (Lewin et al., 2015) and the World Health Organization Qualitative Evidence Synthesis Framework (Downe et al., 2019) approaches were used to synthesise the evidence from qualitative studies given the absence of randomised controlled trials (RCTs) specifically examining family, whānau and carer involvement. Judgements were based on the overall certainty of the evidence, the balance of benefit and harm and the inferred values and preferences of people with schizophrenia supported by the consumer consultation group (Andrews et al., 2013). The Panel decided on either a strong recommendation (i.e. most people with schizophrenia should be offered the recommended intervention), or a weak recommendation (i.e. different choices would be appropriate for different people, depending on their values and preferences) (Andrews et al., 2013). The GRADE evidence and recommendations were independently reviewed by two authors (C.G. and D.K.) who had not been involved in the original evidence review process.
Certainty of evidence and strength or recommendation definitions.

The topics for the relevant clinical sections were developed through consensus discussions and reviews of existing guidelines, as well as seeking additional input from the consumer consultation group. Following the GRADE framework, key health questions were developed by section leads, reviewed by consensus and discussed with a consumer consultation group on two separate occasions to establish relevance and priorities. The key health questions are presented in Table 5.
Key health questions.

GRADE = Grading of Recommendations, Assessment, Development and Evaluation.
To answer each of the key health questions, a database review was performed to identify the most recent and complete evidence. We searched databases, including PubMed, Google Scholar and Cochrane, from inception to March 2024, and the results were updated in June 2025. The evidence was selected based on the hierarchy of evidence levels (i.e. umbrella review > systematic review > RCTs). When multiple studies provided data for the same outcome, we selected the most comprehensive recent study. If identified evidence based on hierarchy was over 5 years old, more recent studies from the next level of evidence were also considered, unless a study older than 5 years was deemed to be more relevant than more recent studies. The Panel also decided to translate the confidence level directly from the findings from Cochrane reviews and Confidence in Network Meta-Analysis (CINeMA) ratings. Furthermore, the previous RANZCP clinical practice guidelines (Galletly et al., 2016) as well as other national/international guidelines and associated papers (Barnes et al., 2020; Buchanan et al., 2025; Castle et al., 2017; Correll et al., 2022; Falkai et al., 2025; Hasan et al., 2017, 2020; Keepers et al., 2020; McCutcheon et al., 2025; Remington et al., 2017) were consulted. The recommendations, along with their associated strengths and certainty of evidence, are summarised in Tables 6–10 with the overall summary of recommendation presented in Table 11. The GRADE summary of findings is presented in Supplemental Appendix Tables 1–19 with the definitions of estimate/magnitude of effect presented in Supplemental Appendix Table 20.
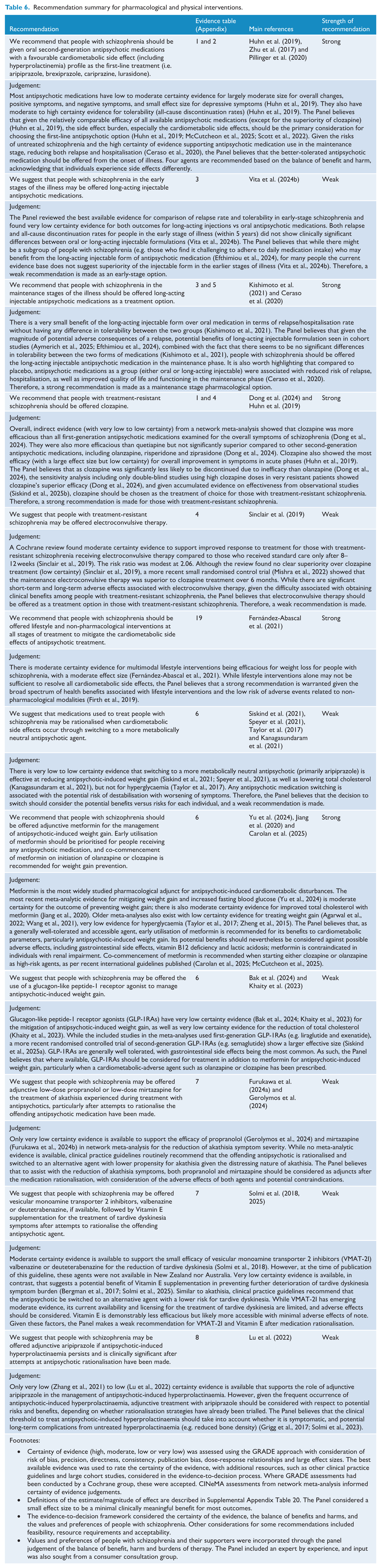
Recommendation summary for pharmacological and physical interventions.
Recommendation summary for psychological and psychosocial interventions.

Recommendation summary for family, whānau and carers.

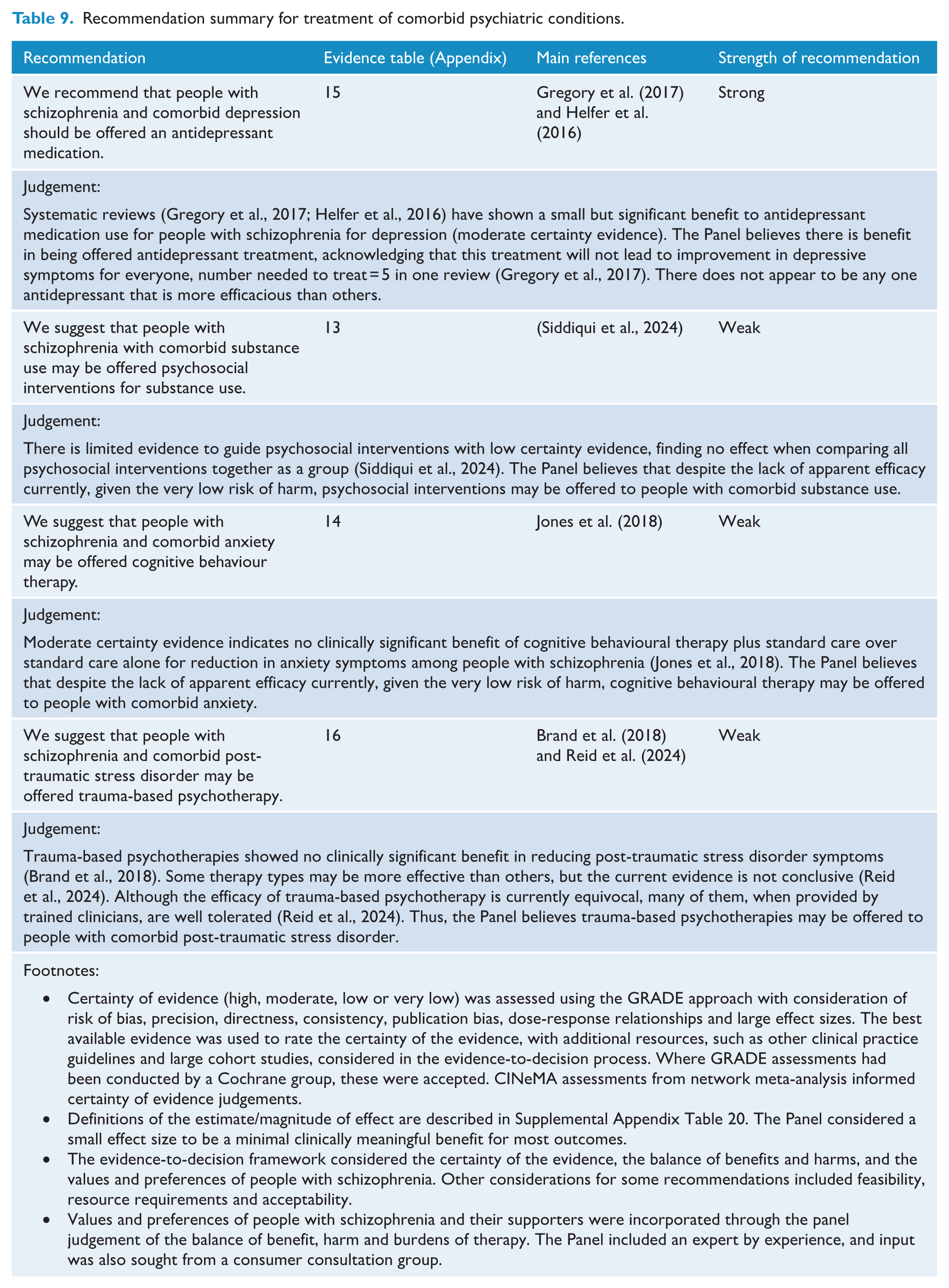
Recommendation summary for treatment of comorbid psychiatric conditions.
Recommendation summary for physical health and lifestyle interventions.

Overall summary of recommendations.

Section 1: physical health assessment
People with schizophrenia have poor physical health and premature mortality of approximately 15 to 20 years compared to the general population (Hjorthøj et al., 2017). The majority of years lost are due to cardiometabolic disease, which is associated with modifiable lifestyle risk factors (e.g. physical inactivity, poor diet, smoking) (Goldfarb et al., 2022; Firth et al., 2019). A comprehensive clinical assessment can help identify treatable physical health comorbidities early.
Secondary physical health, or organic, causes of psychosis have been identified in around 5% of individuals on first presentation (Blackman et al., 2025; Crede et al., 2011; Johnstone et al., 1987). The initial physical health assessment should also consider the higher prevalence of comorbid physical health conditions that present earlier in life for people with schizophrenia (Halstead et al., 2024a). A tailored physical health history and examination is key. Although clinically significant laboratory results are uncommon, it is reasonable to consider initial broad screening investigations of electrolytes, renal, liver and haematological functioning (Conigliaro et al., 2018; Eskelinen et al., 2020). Baseline and continued monitoring of metabolic parameters are also necessary (Castle and Li, 2023; Lambert et al., 2017), but the requirement for fasting samples may not be essential in the prediction of cardiovascular disease for most individuals (Nordestgaard et al., 2016; Selvin et al., 2010). Thyroid function tests are frequently indiscriminately ordered. Although psychotic symptoms have been shown in up to 15% of individuals with hypothyroidism, retrospective studies of both inpatient psychiatric admissions and community based populations demonstrate very low rates of clinically relevant abnormal thyroid results, many of which were already identified on physical assessment (Arce-Cordon et al., 2007; Garnier et al., 2016). Thus, thyroid function should only be requested when history and examination indicate clinical concern. Hyperprolactinaemia most commonly occurs as a consequence of antipsychotic use or pituitary prolactinoma (Cookson et al., 2012). Given the frequency and consequences of hyperprolactinaemia, expert consensus and more recent guidelines recommend prolactin testing at baseline, 3 months after initiation or changes to dose or medication, and when indicated by abnormal menstrual, sexual, or neurological assessment (Peveler et al., 2008; Grigg et al., 2017).
In the case of suspected intracranial pathology, with a history of headaches, nausea or vomiting, seizure-like activity, childhood (<12 years) or older age (>40 years) onset of symptoms, rapid onset or fluctuation of psychosis, significant neurocognitive deficits, catatonia and/or neurological examination findings, neuroimaging with magnetic resonance imaging (MRI) should be conducted. Clinical assessment should guide targeted serum anti-neuronal antibody testing and consideration of lumbar puncture for cerebrospinal fluid (CSF) analysis (Scott et al., 2018). For individuals with first-episode psychosis and no atypical features, neuroimaging and further investigations, such as an electroencephalogram (EEG), are less clearly indicated. They should be guided by collaborative discussions with the person and their family, whānau and carers, with further consideration given during future physical health assessments. While there may be a future role for advanced neuroimaging and other biomarkers to guide personalised treatment (Schmidt and Borgwardt, 2020; Warren et al., 2024), currently the frequency of neuroimaging abnormalities that have established causality with psychosis has been estimated at 0.4% (Forbes et al., 2019). The recommendations for the first-episode psychosis workup are summarised in Table 12.
First-episode psychosis workup.
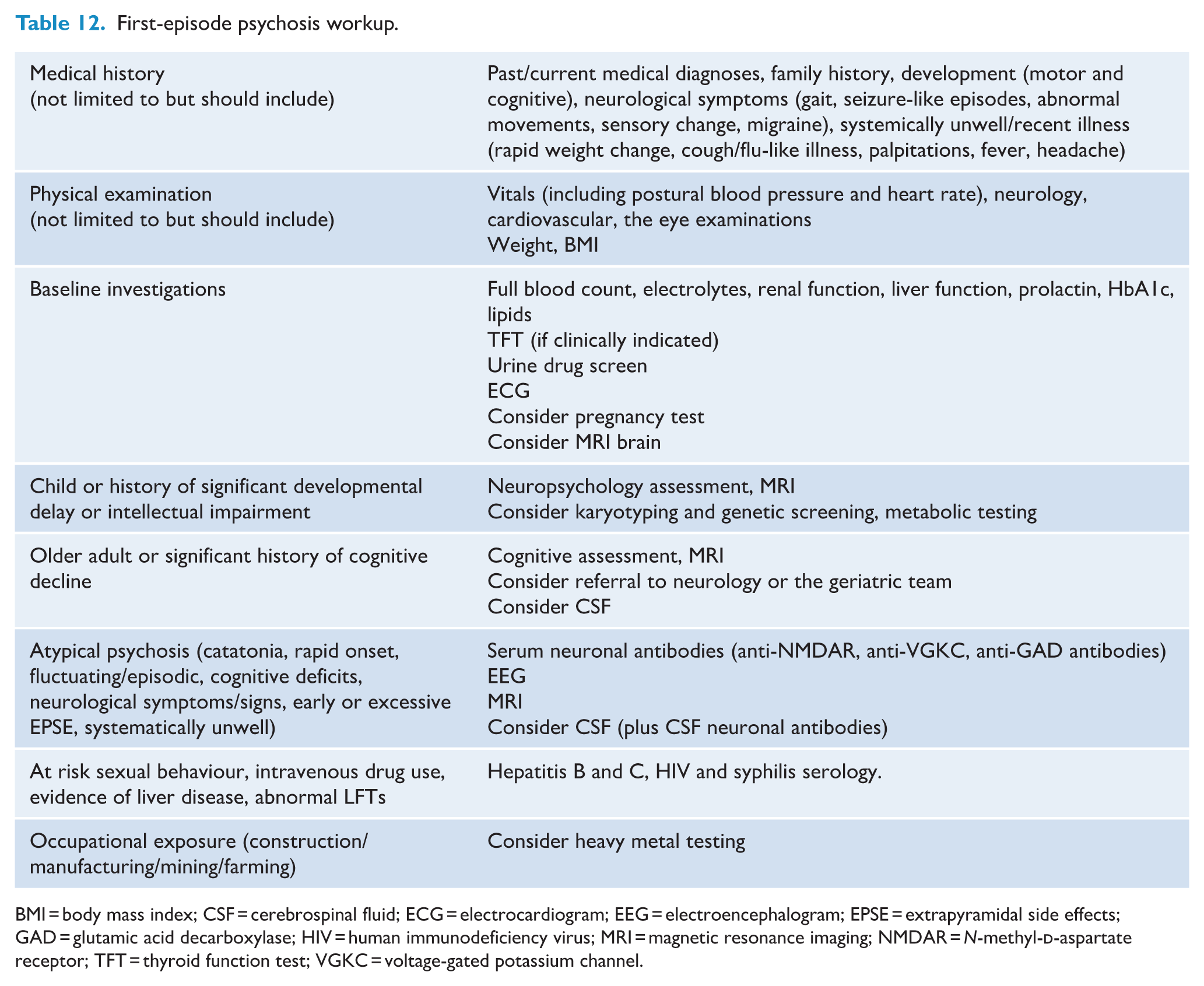
BMI = body mass index; CSF = cerebrospinal fluid; ECG = electrocardiogram; EEG = electroencephalogram; EPSE = extrapyramidal side effects; GAD = glutamic acid decarboxylase; HIV = human immunodeficiency virus; MRI = magnetic resonance imaging; NMDAR = N-methyl-
Section 2: pharmacological treatment
First-episode psychosis
Most previous international clinical guidelines have not been prescriptive as to which antipsychotics are preferred first-line agents for first-episode psychosis (Correll et al., 2022). Given that all antipsychotic medications demonstrate gradual rather than discrete differences in efficacy (Huhn et al., 2019; Oh et al., 2015; Zhu et al., 2017), the decisions on the first agent to trial should be individualised based on tolerability and side effect profile as well as individual preference (McCutcheon et al., 2025; Scott et al., 2022). In terms of long-acting injectable (LAI) formulations, an umbrella review (Etienne and Verdoux, 2025) suggested that LAIs may be useful in selected individuals with early psychosis; however, a meta-analysis found no clear benefit over oral formulations overall (Vita et al., 2024b). It has been noted that the benefits may be more likely to occur when the potential benefits and risks are discussed in a collaborative manner involving the individuals and families (Kane et al., 2021). For these reasons, we recommend initiating with one of the following antipsychotic medications for people with first-episode psychosis: D2 partial agonists (e.g. aripiprazole [oral rather than LAI formulation], brexpiprazole, or cariprazine), or lurasidone due to their broadly more tolerable adverse effect profiles, including lower metabolic risks (McCutcheon et al., 2025; Pillinger et al., 2023). Given the increased sensitivity of those with first-episode psychosis to antipsychotics compared to those with established courses of illness, lower doses may be more effective at eliciting clinical response (Zhu et al., 2017). Thus, we recommend starting low and going slow (e.g. titrating the dose up at 1–2 week intervals if inadequate response and agent well tolerated) for those with first-episode psychosis. Assertive mental state monitoring should be provided on the initiation of medication (e.g. weekly), with attention to adherence to medication in a non-judgement manner (non-adherence often refined as taking less than 80% of prescribed dose or absent plasma level; McCutcheon et al., 2025), and the dose should be reviewed based on effectiveness and side effects (McCutcheon et al., 2025; Scott et al., 2022).
Optimisation of antipsychotic treatment
Adherence to antipsychotic medication as a group (oral or LAI) is associated with reduced risk of relapse and hospitalisation, as well as improved quality of life and functioning in the maintenance phase (Ceraso et al., 2020). It is important to discuss and, if necessary, address treatment adherence with the person with schizophrenia in an open and non-judgmental manner, particularly if there is a partial or no response to the initial medication. Where appropriate, consideration may be given to obtaining collateral reports from family, whānau and carers, as well as measuring serum concentration if feasible (McCutcheon et al., 2025). If there is no response within 2 weeks despite dose within target range and confirmed treatment adherence, it is unlikely that the person will respond to a more extended trial of the same medication (Samara et al., 2015c). At this stage, there is limited evidence on the optimal treatment strategy for those who do not respond to initial antipsychotic medication (Samara et al., 2018b). Thus, there remains no clear evidence to suggest whether it is better to switch antipsychotics or to continue the same antipsychotic in people with schizophrenia who did not respond to their initial treatment (Samara et al., 2025b). Therefore, we recommend changing the initial antipsychotic medication to another agent if (1) there is no response at all to the initial agent after 2 weeks at dose within target range with confirmed adequate adherence, or (2) there is a suboptimal response to the maximum recommended dose of the initial agent after 2–4 weeks with confirmed adequate adherence, or (3) the individual is not able to tolerate the adverse effects of the initial agent at any dose.
In choosing the second antipsychotic medication, there are several considerations. First, although a network meta-analysis of RCTs found no significant difference in efficacy in terms of relapse prevention between different non-clozapine antipsychotic medications either in oral or LAI formulation in the maintenance phase of schizophrenia (Schneider-Thoma et al., 2022), when observational studies have been included in analyses, individuals who are treated with LAIs displayed a lower risk of relapse and hospitalisation compared to those on oral antipsychotic medication (Kishimoto et al., 2021). Similar to the acute phase of treatment (Huhn et al., 2019), different antipsychotic medications exhibited varying tolerability profiles during the maintenance phase (Schneider-Thoma et al., 2022). Among different LAI antipsychotic medications, a network meta-analysis suggested that paliperidone (3-monthly) and aripiprazole may have greater efficacy for reducing relapse than placebo compared to others (Ostuzzi et al., 2021). Nevertheless, in head-to-head comparisons of LAIs, there was no difference in efficacy apart from haloperidol being inferior compared to aripiprazole, fluphenazine and paliperidone, and only aripiprazole showed superior acceptability compared to other LAIs (Ostuzzi et al., 2021). Moreover, a network meta-analysis using individual participant data from Swedish and Finnish national registries, as well as aggregate data from RCTs, also demonstrated that LAIs were more effective in the real world (i.e. national registry data) compared to the efficacy found in RCTs (Efthimiou et al., 2024). Furthermore, LAI antipsychotics are associated with a lower risk of all-cause and non-suicidal mortality in people with schizophrenia compared to oral antipsychotics (Aymerich et al., 2025). Combined, we recommend choosing the second antipsychotic medication from the remaining agents from the first-line treatment options (aripiprazole, brexpiprazole, cariprazine, or lurasidone) with additional consideration for agents with different receptor profiles such as amisulpride and risperidone (McCutcheon et al., 2023), as well as LAI formulation for aripiprazole and paliperidone. Similar to the initiation process, the choice of the second agent should be guided by the adverse effect profile and individual preference. In addition, we encourage prescribers to consider changing to a medication with a different pharmacodynamic profile (e.g. if you have already tried a dopamine partial agonist like aripiprazole, brexipiprazole or cariprazine, then try a dopamine antagonist like lurasidone, amisulpride, risperidone or paliperidone LAI) (McCutcheon et al., 2025).
Negative symptoms are common, disabling, and often show limited response to currently available antipsychotic medications (Correll and Schooler, 2020). It is therefore important to consider and address potential secondary causes, including suboptimally treated positive symptoms, depressive symptoms, substance use, untreated medical conditions and side effects of antipsychotic treatment (e.g. extrapyramidal symptoms). Certain medications, such as cariprazine, aripiprazole and low-dose amisulpride, may offer greater efficacy for negative symptoms and antidepressant augmentation may be helpful for some (McCutcheon et al., 2025). Similarly, cognitive symptoms associated with schizophrenia are frequently insufficiently addressed by antipsychotic medications. Furthermore, increased anticholinergic burden from some antipsychotic medications may worsen cognitive functioning. In such cases, dose reduction to minimise anticholinergic burden should be considered (McCutcheon et al., 2025). Non-pharmacological interventions targeting these domains are discussed in the coming sections.
Treatment-resistant schizophrenia
Treatment-resistant schizophrenia is defined as an inadequate response and persisting moderate to severe functional impairment following the use of two different antipsychotic medications, each taken with adequate dose, duration and confirmed adherence (Howes et al., 2017; Kane et al., 1988). The proportion of treatment resistance among people with schizophrenia is estimated to be around 30% (Howes et al., 2017; Kane et al., 2019; Siskind et al., 2022a). For those meeting the threshold for treatment-resistant schizophrenia, clozapine should be considered under the care of tertiary mental health services (Dong et al., 2024; Kane et al., 2019; Samara et al., 2025a). A Cochrane review (Sinclair et al., 2019) found that the addition of electroconvulsive therapy (ECT) may increase clinical response compared to standard care alone (various antipsychotics used as the standard care across different studies, including clozapine), at least in the short term (8–12 weeks). The benefit was not clear when ECT was directly compared to clozapine. It is also estimated that only around 40% of people with treatment-resistant schizophrenia would respond to clozapine (Siskind et al., 2017). Currently, there is limited evidence for treatment options for those who do not respond to clozapine. Some expert consensus statements (Wagner et al., 2019, 2020) recommend strategies such as augmentation with another antipsychotic medication (e.g. amisulpride and aripiprazole) or ECT for persistent positive symptoms, and combination with certain antidepressants (e.g. fluoxetine, duloxetine, citalopram) for negative symptoms. A recent cohort study found that clozapine combined with sertraline, duloxetine and escitalopram were associated with reduced risk of relapse (Taipale et al., 2025). For suicidality, augmentation with antidepressants or mood stabilisers, or ECT has been suggested, as well as augmentation with a mood-stabiliser or antipsychotic medication for aggression (Siskind et al., 2018; Wagner et al., 2019, 2020). A meta-analysis of antipsychotic augmentation strategies for clozapine among Swedish and Finnish nationwide cohorts demonstrated that only clozapine and medium-dose aripiprazole (9 to <16.5 mg/day) was associated with decreased hospitalisation rates for both psychosis and somatic causes compared to clozapine monotherapy. In contrast, high-dose augmentation with all other examined antipsychotics (olanzapine, quetiapine or risperidone) was associated with increased hospitalisation rates for psychosis (Tiihonen et al., 2025). Furthermore, supratherapeutic doses of antipsychotic medications do not appear to improve response rates (Samara et al., 2018a). The evidence for the effectiveness of antipsychotic polypharmacy is mixed, despite the frequency of its use in clinical practice. Although one systematic review found that antipsychotic polypharmacy was effective in individuals with more severe symptoms (Lochmann van Bennekom et al., 2024), another review found that those with antipsychotic polypharmacy had greater illness severity and poorer outcomes than those treated with antipsychotic monotherapy, and antipsychotic polypharmacy was associated with higher side effect burden, including all-cause mortality (Højlund et al., 2024). Thus, we recommend avoiding supratherapeutic doses or polypharmacy in general, unless clinically deemed necessary. In cases of polypharmacy or supratherapeutic doses, we suggest close monitoring of both the effectiveness and tolerance assertively. The additional agent should be withdrawn, or the dose should be reduced to the therapeutic dose, if no benefits are seen within 3 months. The summary of the recommended pharmacology algorithm is presented in Table 13.
Antipsychotic medication algorithm.
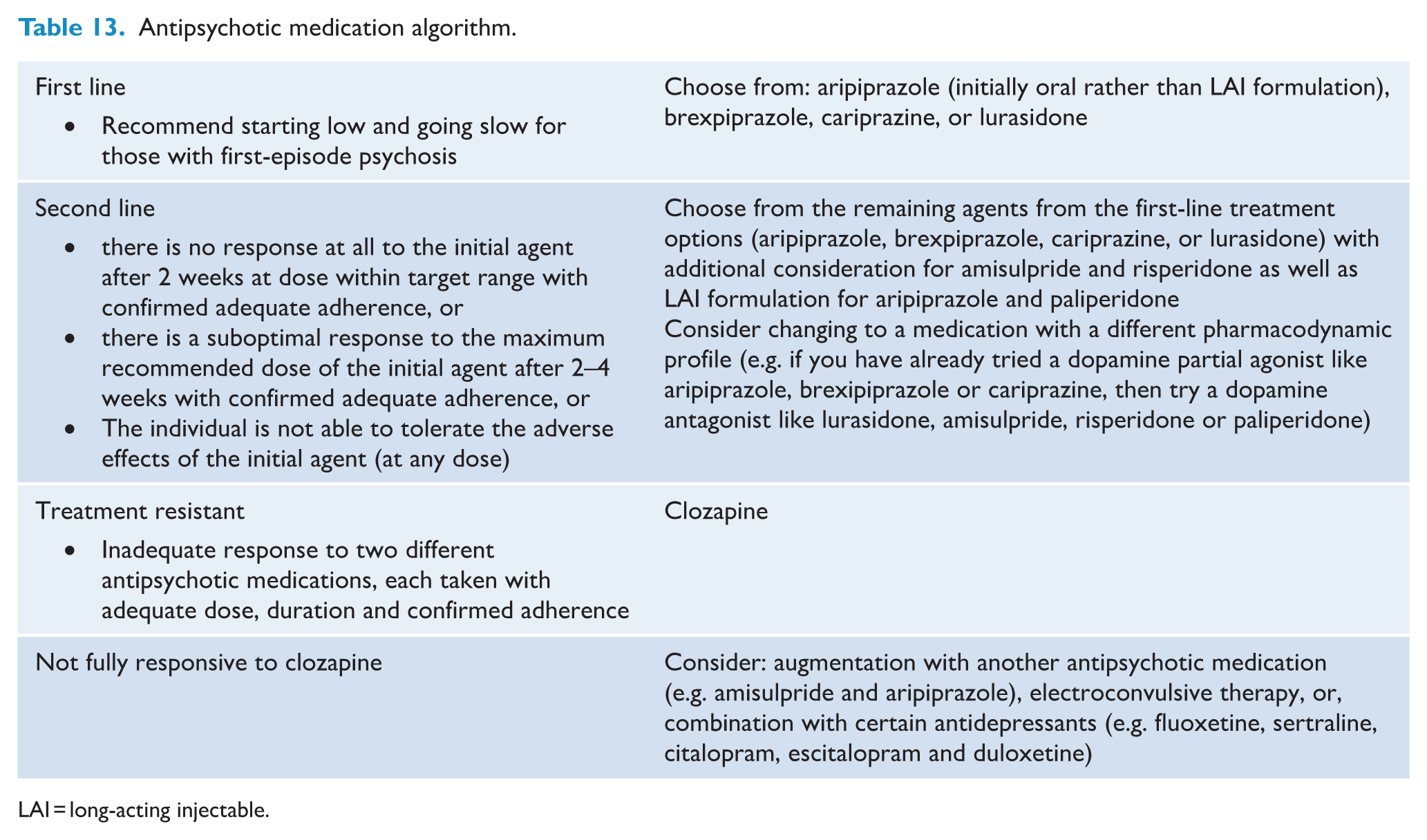
LAI = long-acting injectable.
Tolerability and adverse effect consideration
Side effects are common among people treated with antipsychotic medication, having been reported in over 75% of those treated (Iversen et al., 2018). It is also worth noting that some side effect burdens do not necessarily show a dose-response pattern. For instance, while most second-generation antipsychotic medications show an initial dose-related weight increase that reaches a plateau at a higher dose, some (e.g. aripiprazole, olanzapine and paliperidone) do not show such a plateau. Other agents, such as quetiapine and ziprasidone, on the other hand, exhibited bell-shaped curves, where the risk of weight gain appears to decrease at higher doses (Wu et al., 2022). Similarly, the discontinuation rates have diverse response patterns. Some antipsychotic medications (including amisulpride, cariprazine, olanzapine and quetiapine) show U-shaped curves, where increases in dosage lead to raised discontinuation rates, while others (including aripiprazole, asenapine, brexpiprazole, clozapine, paliperidone and risperidone) reach plateaus in discontinuation rates with increasing doses (Tian et al., 2025). A few antipsychotic medications, such as haloperidol, lurasidone and ziprasidone, do not reach a plateau within the examined doses (Tian et al., 2025). Combined, these findings underscore the importance of closely monitoring potential adverse effects and making dose adjustments accordingly, as the consequences of side effects are numerous, including negative impacts on physical health, quality of life, adherence and an increased risk of relapse (Pillinger et al., 2020).
While not based on evidence derived from RCTs (Tosh et al., 2014), due to the rapidity of metabolic disturbance, particularly in youth and the antipsychotic naïve (Curtis et al., 2024), numerous international and national expert consensus guidelines call for routine metabolic screening every 6–12 months integrated into multidisciplinary care at baseline and regular intervals from the outset of psychotropic medication (Hasan et al., 2017; Lambert et al., 2017; McCutcheon et al., 2025). GRADE evidence-based recommendations were formulated to guide the management of the following antipsychotic side effects where meta-analytic evidence was available: weight gain, dysglycaemia, dyslipidaemia, akathisia, tardive dyskinesia and hyperprolactinaemia. For antipsychotic-induced cardiometabolic disturbances, non-pharmacological strategies such as exercise and dietary interventions (detailed further in Section 6) and medication rationalisation steps should be offered for all individuals. Adjunctive metformin has the most evidence for both the prevention and treatment of antipsychotic-induced weight gain (Agarwal et al., 2022; Taylor et al., 2017; Wang et al., 2021; Yu et al., 2024; Zheng et al., 2015) and is generally well tolerated. Early utilisation of metformin is encouraged, and co-commencement of metformin with high-risk metabolic agents such as clozapine and olanzapine is recommended in alignment with other international guidelines (Carolan et al., 2025; McCutcheon et al., 2025). In addition, where available and accessible, glucagon-like peptide-1 receptor agonists (GLP-1RAs) may be considered in light of the emerging evidence of their efficacy in reducing antipsychotic-induced weight gain (Bak et al., 2024; Siskind et al., 2019, 2025; Khaity et al., 2023). Given its distressing nature, akathisia is typically managed through antipsychotic rationalisation in the first instance through attempts to reduce the medication dose and switch to an alternative antipsychotic medication (Furukawa et al., 2024; Gerolymos et al., 2024). When rationalisation attempts are insufficient to treat akathisia, adjunctive propranolol and mirtazapine have some evidence of being beneficial in symptom reduction, although the certainty of evidence is low (Furukawa et al., 2024; Gerolymos et al., 2024). The antipsychotic rationalisation is likewise the first step for the management of tardive dyskinesia. Beyond this, vesicular monoamine transporter 2 inhibitors (VMAT-2I), such as valbenazine and deuteterabenazine, have demonstrated evidence of benefits for tardive dyskinesia symptoms (Solmi et al., 2025). However, these compounds are not currently government subsidised in Australia and Aotearoa New Zealand and are not routinely accessible. Otherwise, vitamin E supplementation has low- certainty evidence to reduce further deterioration of tardive dyskinesia symptomatic burden (Bergman et al., 2017; Solmi et al., 2025).
After attempts at medication rationalisation, adjunctive aripiprazole can also be considered in the treatment of antipsychotic-induced hyperprolactinaemia (Lu et al., 2022; Zhang et al., 2021). The decision to treat antipsychotic-induced hyperprolactinaemia should consider symptom burden (e.g. gynaecomastia and sexual dysfunction), as well as risks associated with untreated hyperprolactinaemia (e.g. increased risk of breast cancer and osteoporosis) (Grigg et al., 2017; Solmi et al., 2023a, 2024; Taipale et al., 2021). The main side effects of antipsychotic medications and possible management options with empirical evidence base have been summarised in Table 14. The physical health monitoring schedule, adopted from the recent Lancet Psychiatry Physical Health Commission (Halstead et al., 2025), is presented in Table 15.
Management options for antipsychotic side effects.

GLP-1RA = glucagon-like-peptide-1 receptor agonist; VMAT-2 = vesicular monoamine transporter.
Physical health monitoring schedule.

Source: Adopted from Halstead et al. (2025).
BMI = body mass index; ECG = electrocardiogram; TFT = thyroid function test.
Duration of antipsychotic medication treatment
The risk of relapse in clinically stable people with schizophrenia is increased when undergoing dose reductions to subtherapeutic doses or complete discontinuation of antipsychotic agents (Højlund et al., 2021; Ostuzzi et al., 2022). Even among individuals with first-episode psychosis who have been clinically stable for 6 months to 2 years, antipsychotic medication discontinuation is associated with an increased risk of relapse (Kishi et al., 2019). However, it is important to acknowledge that many people, especially those with first-episode psychosis who have achieved remission, express their desire to discontinue antipsychotic medication. While the current evidence base (Højlund et al., 2021; Kishi et al., 2019; Ostuzzi et al., 2022) suggests that the risk of relapse and related complications is minimised if individuals continue antipsychotic medication within the recommended therapeutic range, the decision may be reached to trial medication discontinuation through a shared decision-making process after discussion with the person with schizophrenia and preferably with their family, whānau and carers. Although empirical evidence remains limited, we suggest that discontinuation of antipsychotic medication be approached through gradual dose reductions over a period of at least 3–6 months, accompanied by close monitoring for early signs and symptoms of relapse. Ideally, individuals should continue to receive support and monitoring for an additional 6–12 months following discontinuation, if not longer. It is essential that individuals, along with their family, whānau and carers, are well informed about potential indicators of relapse and have access to a clear and rapid pathway back to mental health services should the need arise. Further consideration should also be given to distress caused by residual symptoms and whether adjunctive psychological and psychosocial interventions may aid functional improvement in addition to symptom reduction (Cella et al., 2023).
Section 3: psychological and psychosocial interventions
Currently, there is no definitive pharmacotherapy for the negative, neurocognitive and social cognitive symptom domains of schizophrenia. As such, alongside pharmacological interventions, psychological and psychosocial interventions are a critical part of care for people with schizophrenia, especially in the maintenance phase of the illness. Psychological and psychosocial interventions can be categorised into those that should be offered and provided to all individuals with schizophrenia, or those that should be offered and provided to people with specific needs. For example, relapse prevention interventions (including education for recognising early signs and symptoms of relapse, a system of symptom monitoring and a crisis plan) and psychoeducation should be provided to all individuals with schizophrenia as in addition to the need for individuals to know their condition, treatment and recovery, there is evidence that psychoeducation can reduce relapse (Bighelli et al., 2021). Cognitive behavioural therapy for psychosis (CBTp) is another psychological intervention that should be offered and provided to all individuals with schizophrenia, as there is evidence for a small effect in improving positive and negative symptoms and also overall psychiatric symptoms (Jauhar et al., 2014). There is also evidence that CBTp may improve long-term global mental state (Jones et al., 2018) and may reduce relapse (Bighelli et al., 2021). Furthermore, as relapse in schizophrenia can have a devastating impact on recovery for people with schizophrenia and their families, whānau and caregivers, we also recommend that relapse prevention interventions should be routinely offered to people living with schizophrenia (Bighelli et al., 2021).
For the therapies that should be offered and provided to those with specific needs, the choice of interventions should be guided by the symptom profile. Some of these therapies may also focus on functional outcome and self-management of the impact of residual symptoms as distinct from symptom remission. Neurocognitive impairment is common in people with schizophrenia, and cognitive remediation therapy should be offered to people with schizophrenia who present with cognitive deficits and associated poor functioning. While the overall effect of cognitive remediation therapy is very small to moderate for symptom domains such as positive and negative symptoms, the effect size is greater for global cognition and functioning (Vita et al., 2021, 2024a). Cognitive impairments can impact people with schizophrenia across different phases of illness, and pharmacotherapy has little established role in addressing them (Green et al., 2019). Deficits in social cognition are also common in people living with schizophrenia and social cognitive training has been developed to address these challenges. Although a meta-analysis found that social cognitive training was not effective for positive and negative symptoms, social functioning or cognition (Yeo et al., 2022), there is evidence that it may improve theory of mind (D’Arma et al., 2021) as well as emotion perception and social perception emotional recognition (Nijman et al., 2020). These symptom domains can limit social functioning even when neurocognitive symptoms have been addressed (Fett et al., 2011). For individuals who have persistent positive and negative symptoms, meta-cognitive therapy should be offered as it has a small to moderate impact in reducing the intensity of delusions, overall positive symptoms and negative symptoms, while improving insight and social cognition (Melville et al., 2024). There is also evidence that mindfulness-based interventions may have at least a small effect size on positive and negative symptoms as well as functional outcomes (Meinhart et al., 2025).
Overall, we recommend psychological and psychosocial interventions for people with schizophrenia from the onset of the illness. The type of intervention depends on several factors, including the phase of illness, personal preference and readiness, symptom profile and associated impact on function, as well as the availability of local resources. However, interventions such as psychoeducation, CBTp and relapse prevention should be available for all. Furthermore, cognitive remediation therapy, including social cognitive training, and metacognitive therapy interventions should be available for people with schizophrenia who have ongoing difficulties with specific symptom domains. It is worth emphasising that there is evidence showing enhanced effect when psychosocial interventions are delivered within a rehabilitation model of care (Vita et al., 2021; Wykes et al., 2011). Consideration needs to be given to the potential combined effects of these interventions and the settings in which they are delivered.
Section 4: family, wha-nau and carers
For the current Guidelines, we have utilised the term ‘family, whānau and carers’ to encapsulate those who provide unpaid care, support and assistance to a person with a diagnosed mental health condition or experiencing psychological distress. The terms ‘family’ and ‘whānau’ in particular are used broadly and inclusively within this document, encompassing kin and other informal supporters, such as friends. Living with schizophrenia takes courage and perseverance for the person and their family, whānau and carers. There can be significant challenges that have profound impacts on multiple aspects of life and relationships for the person and their family, whānau and carers, whether they are a parent, child, sibling, partner, other relative or close friend. ‘Carer’ is a label that family or friends of the person may or may not acknowledge or recognise, depending on circumstances and the relationship with the person. In Australia, family members who identify as primary mental health carers, on average, spend 40 hours per week providing care (Maybery et al., 2021), and the cost of informal support provided by family carers has been estimated to be approximately AU$8.4 billion (Diminic et al., 2021).
Family, whānau and carers are more than providers of support tasks, sources of information about the person, or conduits to assist mental health service priorities, including medication adherence, symptom management, and treatment (Cameron et al., 2023). Their priority is the person’s whole health and wellbeing. Family, whānau and carers seek collaboration with mental health services because they strive for health professionals to understand and view the person with a diagnosis of schizophrenia as an individual first (Forcheron et al., 2023; Huang et al., 2021). A Cochrane review of family-based interventions for family, whānau and carers of people with schizophrenia found that they may reduce the relapse of illness and carer burden but did not have a significant impact on hospital readmission (Chien et al., 2024). A systematic review of 24 studies (Iswanti et al., 2023) found support for a professionally engaged relationship between individuals with schizophrenia, families and mental health professionals to optimise care provision. When the illness called schizophrenia becomes part of their lives, many things they hope and expect for their futures change, and everyone must learn how to make sense of their changed lives. It can often be challenging to strike the right balance between involving family, whānau and carers while not overburdening them, as well as navigating systemic complexities and confidentiality issues as part of respecting the person’s privacy and autonomy (Jankovic et al., 2011; Landeweer et al., 2017; Vasava et al., 2023). Confidentiality, in particular, is challenging to navigate at times with subtle variations in different jurisdictions and services. Inconsistencies in family involvement often appear to stem from organisational culture, societal attitudes, providers’ neglect of family expertise and providers’ uncertainty regarding confidentiality policies and the absence of practice guidelines. In some situations, this may mean providing information and alleviating anxiety and tension for family, whānau and carers where the person with schizophrenia does not wish to have them involved or wishes to keep their mental health information confidential (i.e. a means of providing family, whānau and carers with support without breaching confidentiality in relation to the person) (Landeweer et al., 2017; Tham and Solomon, 2024). Psychoeducation and support, from the very onset of the illness, are therefore crucial not only for the person living with schizophrenia but also for their family, whānau and carers (Cameron et al., 2023; Iswanti et al., 2023). Developing strategies and supports within a ‘triangle of care’ that actively includes the person, their family, whānau and carer, and mental health professionals in the shared decision-making process can ensure that everyone is on the same page about what is helpful, needed and respectful to maximise the person’s autonomy, dignity, choice and agency (Cameron et al., 2023; Outram et al., 2015; Schuster et al., 2020).
Section 5: psychiatric comorbidities
Substance misuse and substance-induced psychosis
Comorbid substance use is common in people with schizophrenia, with an estimated one-third diagnosed with a lifetime substance use disorder (Toftdahl et al., 2016). In addition to precipitating psychosis, substance misuse can often develop for the first time after the diagnosis of schizophrenia (Lappin, 2019; Petersen et al., 2019). Indeed, people with schizophrenia have a more than threefold risk for developing substance misuse, with nicotine, cannabis, alcohol and stimulants the most implicated (Petersen et al., 2019). Given this, we urge clinicians to conduct regular enquiries regarding substance use, including type/s, pattern, effects on relationships and functioning, harms associated with use, and assess readiness for change.
Trauma
People with schizophrenia report higher rates of trauma and post-traumatic stress disorder (PTSD) than the general population, and people who have experienced trauma are more likely to develop psychotic symptoms (Bailey et al., 2018; Carr et al., 2018; De Bont et al., 2015; Trotta et al., 2015; Turner et al., 2019). Importantly, the experience of psychosis itself can give rise to trauma experiences, with the prevalence estimates of psychosis-related PTSD ranging from 14% to 47% (Berry et al., 2013; Buswell et al., 2021). Such trauma may arise both from distressing symptoms, such as derogatory auditory hallucinations and persecutory delusions, and from associated treatment, including restrictive/coercive practices and involuntary hospitalisation (Lu et al., 2017). While trauma-informed care is widely accepted as integral to reducing distress and supporting recovery, much work remains to be done to ensure its comprehensive implementation (Lommen and Restifo, 2009; Read et al., 2005).
Anxiety and depression
Anxiety and depressive symptoms are very common among people with schizophrenia; over 30% have a comorbid major depressive disorder (Etchecopar-Etchart et al., 2021), and nearly 40% have comorbid anxiety disorders, most commonly social phobia followed by PTSD and obsessive-compulsive disorder (OCD) (Achim et al., 2011; Braga et al., 2013). Of note, the rate of OCD increases with a longer duration of schizophrenia (Swets et al., 2014), and some antipsychotic medications may increase the risk of de novo OCD or symptoms (Burk et al., 2023). In clinical practice, these symptoms are often mistaken for symptoms of schizophrenia, such as negative symptoms or persecutory delusions. As such, it is crucial to take a thorough history of anxiety and depressive symptoms (including assessing for neurovegetative symptoms in detail and environmental contexts in which anxiety arises) to ensure that treatable symptoms are not missed due to diagnostic overshadowing.
Treatment for psychiatric comorbidities
There is a limited body of evidence related to treatment options specific to people with schizophrenia with a comorbid psychiatric condition. While several reviews have demonstrated possible benefits of antipsychotic medication (Krause et al., 2019), LAI treatment (Coles et al., 2021), repetitive transcranial magnetic stimulation (Johnstone et al., 2022) and psychological therapies (Bennett et al., 2017) in reducing substance use, and naltrexone in reducing alcohol use (Rosenstand et al., 2024), all were limited by the methodological limitations of the individual studies included. Some observational studies have shown potential beneficial effects of clozapine on reducing substance misuse; however, this finding is not yet sufficiently robust to recommend the use of clozapine for managing comorbid substance use (Rafizadeh et al., 2023). A systematic review (Siddiqui et al., 2024) found that psychosocial interventions did not show a significant benefit in reducing substance use for people with schizophrenia. Overall, despite being a common and important clinical issue, there remains very limited evidence to guide us in managing people with schizophrenia and comorbid substance use or, even more strikingly, those with substance-related psychosis (Fiorentini et al., 2021).
There is emerging evidence that psychological interventions targeting trauma among people with schizophrenia are well tolerated and acceptable (Reid et al., 2024). To date, however, the evidence is inconclusive regarding whether trauma-focussed psychological therapies are effective in reducing PTSD and/or psychotic symptoms (Brand et al., 2018; Reid et al., 2024; Sin et al., 2017) with RCT-level evidence of benefit for eye movement desensitisation and reprocessing (EMDR) and prolonged exposure (PE) (De Bont et al., 2016; Van den Berg et al., 2018). A small number of studies have explored the efficacy of psychological therapies targeting anxiety symptoms among people with schizophrenia (Grover et al., 2019). Although one review concluded that there are benefits of CBT in reducing anxiety (and depressive) symptoms in people with schizophrenia (Opoka and Lincoln, 2017), a Cochrane review found no evidence of benefits with CBT for anxiety symptoms for people with schizophrenia compared to the standard care (Jones et al., 2018). Furthermore, an umbrella review found little evidence to support CBT for depressive or anxiety symptoms in people with schizophrenia (Berendsen et al., 2024). For the treatment of depression in schizophrenia, two meta-analyses showed that antidepressants may be efficacious, especially for those with more severe depressive symptoms, but the current evidence base was found to be limited (Gregory et al., 2017; Helfer et al., 2016). Neither review found a preference for any class or individual antidepressant medication.
Overall, despite high prevalences of comorbid psychiatric conditions among people with schizophrenia, there remains very limited evidence base to make strong recommendations at the moment. Therefore, we suggest: (1) conducting a thorough assessment of comorbid psychiatric symptoms and conditions in people with schizophrenia and (2) providing pharmacological and/or psychosocial interventions for comorbid conditions as indicated for the specific condition, acknowledging that there is an urgent need to build evidence base specifically for people with schizophrenia with a comorbid psychiatric condition.
Section 6: physical health and lifestyle interventions
Physical health
Preventive healthcare access is often inequitable: people with schizophrenia have lower rates of cancer screening (Solmi et al., 2020), immunisation/vaccination (Raffard et al., 2022), sexual health checks and blood-borne virus screening (Brown et al., 2025) compared to the general population. Oral health is frequently neglected (Mishu et al., 2024; Wey et al., 2016), despite significantly elevated rates of dental caries, tooth loss, periodontal disease and oral cancers (Kisely et al., 2015; Yang et al., 2018). Close collaboration and effective communication between mental health providers and primary care services are essential for addressing health inequities, particularly in coordinating preventive health checks and routine physical healthcare (Morden et al., 2009; Planner et al., 2014; Firth et al., 2019). This collaboration should extend to other allied health, oral health and specialist medical practitioners to ensure holistic recovery-oriented care that also prioritises shared decision-making with individuals, their family, whānau and carers (Halstead et al., 2024b).
Lifestyle interventions
Among lifestyle strategies, physical activity has shown the most consistent benefits for mental health, particularly in reducing negative symptoms (Kim et al., 2023; Maurus et al., 2024), which typically show limited response to pharmacological treatment (Correll and Schooler, 2020). There is moderate certainty evidence supporting the positive effects of physical activity on cognition, global functioning and quality of life (Rissmayer et al., 2024; Korman et al., 2023; Shimada et al., 2022), reinforcing its role as an important adjunctive treatment. Although a range of physical activities (including aerobic, resistance, mind-body) were included in the reviews examined, the majority of evidence is derived from aerobic studies, with greater improvements observed at moderate or greater intensity exercise (Korman et al., 2023; Rissmayer et al., 2024), and when supervised by exercise professionals (Rissmayer et al., 2024; Shimada et al., 2022). There is some evidence that yoga may benefit negative symptoms (Yin et al., 2024); however, the findings are inconsistent (Maurus et al., 2024), with most studies having been conducted outside of Australia and Aotearoa New Zealand, limiting the generalisability of the findings. Although currently based on a small amount of very low certainty evidence, peer-facilitated physical health interventions show future promise (Coles et al., 2022) and should be investigated further. Access to appropriate physical activity facilities and opportunities is vital, and interventions should incorporate strategies to promote autonomous motivation (Arnautovska et al., 2022; Vancampfort et al., 2025). Physical activity alone has not been found to impact weight loss consistently (Gallardo-Gomez et al., 2023; Maurus et al., 2024), despite often being one of the most commonly stated goals of people with schizophrenia (Firth et al., 2016). Multimodal lifestyle interventions (combinations of nutrition counselling, weight management programmes, physical activity, health education, motivational interviewing and CBT) have had the most significant effects on reducing weight and improving other cardiometabolic parameters (Fernández-Abascal et al., 2021). Although weight loss has been typically modest, we make a strong recommendation that lifestyle interventions should be integrated into usual psychiatric care from the earliest stages of illness and antipsychotic prescription, with prevention of weight gain an important target (Curtis et al., 2016). Factors associated with successful lifestyle interventions include; targeting multiple lifestyle components in one intervention, tailoring the intervention to the individual and their illness, delivery by qualified professionals (e.g. exercise physiologists, physiotherapists and dieticians), embedding lifestyle interventions into usual care and facilitating social networks to support engagement (Curtis et al., 2024; Lederman et al., 2017; Lee et al., 2022; Maurus et al., 2024; Teasdale et al., 2025; Vancampfort et al., 2025)
Smoking
People with schizophrenia continue to have disproportionately high rates of smoking despite declining rates in the general population (Cook et al., 2014). Although quit rates are lower than in the general population, multiple pharmacotherapies (varenicline, bupropion and nicotine replacement therapy) are more effective than placebo, with varenicline demonstrating the strongest evidence (Siskind et al., 2020). Furthermore, moderate certainty evidence supports tailored, behavioural interventions to support smoking cessation in people with schizophrenia (Spanakis et al., 2022).
Sleep
People with schizophrenia experience significant disturbance to both sleep continuity and architecture (Chan et al., 2017), with insomnia, nightmares and daytime hypersomnia commonly experienced (Chiu et al., 2018; Waite et al., 2020). A systematic review (Wilkinson et al., 2025) found that cognitive behavioural therapy for insomnia (CBT-i) was effective and associated with minimal harms; however, sleep restriction was excluded from the majority of studies to reduce the risk of exacerbating psychotic symptoms. Obstructive sleep apnoea (OSA) is highly prevalent among people with schizophrenia (Myles et al., 2016). Although there is no current RCT evidence, a large case-control study found continuous positive airway pressure (CPAP) therapy to be feasible and effective in people with schizophrenia (Giles et al., 2022). Given the potential for cognitive and cardiovascular health (McEvoy et al., 2016), those at risk of OSA should be proactively screened and supported to access OSA management.
Oral health
Currently, there is only very low certainty evidence for oral education interventions for people with schizophrenia (Macnamara et al., 2021). However, possible strategies for improving oral health may include: facilitating early oral screening; referring to affordable dental clinics; managing psychotropic-induced dry mouth (Kisely et al., 2015); and integrating oral concerns into psychiatric healthcare (Skallevold et al., 2023).
Section 7: special populations
Māori and Aboriginal and Torres Strait Islander peoples
While there is a limited body of research knowledge, especially in Australia, regarding the rates of schizophrenia among Indigenous peoples, it is generally reported that there is a higher prevalence of schizophrenia and related psychoses both in Aboriginal and Torres Strait Islander peoples (Black et al., 2015; Heffernan et al., 2012; Jewell and Mitchell, 2023; Ogilvie et al., 2021) and Māori populations (Kake et al., 2008; Tapsell et al., 2018). Māori and Aboriginal and Torres Strait Islander peoples also experience elevated rates of coercive care (Beaglehole et al., 2024; Kisely et al., 2020), higher social disadvantage (Badcock et al., 2018) and multimorbidity (Carman et al., 2022; Monk et al., 2024). It is important to recognise the possible roles of colonisation, social disadvantage and racism as factors in these mental health inequities.
In terms of effective clinical service models, although there are several important guiding principles such as those outlined in the Closing the Gap and the Gayaa Dhuwi (Proud Spirit) Declaration, the implementation in clinical practice has not been consistent (Grattan et al., 2025; Gupta et al., 2020). Nevertheless, it is essential to foster the development of a workforce that reflects the community it serves and ensures cultural safety in all clinical settings and research (Lacey et al., 2022). To this end, we advocate for universal cultural safety in mental health systems. While each individual determines cultural safety, the outcome of culturally safe health systems is to ensure that people with schizophrenia feel comfortable and secure accessing care. This is particularly important for Aboriginal and Torres Strait Islander peoples in Australia and Māori in Aotearoa New Zealand.
People in rural and regional areas
Rural communities have a significant unmet need for mental health care (Plana-Ripoll et al., 2021). These areas have fewer psychiatrists per population than urban regions, and distances from hospitals are greater (Amos et al., 2023; Darmawan et al., 2023). While there has been an increasing utilisation of telepsychiatry in Australia in recent years (Woon et al., 2024), general practitioners will often be the primary medical contact for people with schizophrenia rather than psychiatrists or mental health professionals in rural communities. To ensure the wellbeing of people with schizophrenia in rural areas, we need to continue to not only deliver service wherever possible, but also have an increased emphasis on upskilling general practitioners and providing support and supervision using appropriate technologies (Amos et al., 2022; Coleman et al., 2022).
Discussion
In the current Guidelines, we have utilised the GRADE approach to formulate recommendations related to different aspects of care provision for people with schizophrenia. At the individual level, we hope that these Guidelines are helpful for clinicians working with people with schizophrenia to provide the most optimal care in conjunction with an individualised, collaborative formulation. At the systematic level, we hope that the Guidelines provide a blueprint to aid clinical directors and policymakers in considering the breadth of workforce structures identified in the evidence presented, as well as the workforce and work environment required to care for people with schizophrenia.
Guideline utilisation and applicability
In considering potential barriers and enablers to implement the recommendations, the importance of systemic support cannot be overemphasised. It is essential, for example, that the quality of the therapists who provide psychological and psychosocial interventions, as well as lifestyle interventions, is adequately maintained through regular training and supervision. Given the strong interactions between mental health and physical health conditions, the link between mental health services and other medical services, including primary health care, should be further strengthened. In treating physical health comorbidities of people with schizophrenia, addressing and reducing organisational silos is critical. While there are examples of successful initiatives at a local level (e.g. Siskind et al., 2022b), there needs to be further efforts at the national and binational levels to improve service delivery. Silos in care may occur at various points, including within mental health services (e.g. from inpatient care to community care and vice versa), when interfacing with other medical services such as primary health care providers, and through non-health organisations, including non-governmental organisations. It is also important to acknowledge that most people with schizophrenia will not stay in the public mental health system indefinitely. Many people with schizophrenia are managed in the primary health sector with or without additional input from non-health organisations. People with schizophrenia and their family, whānau and carers must be supported in navigating often complex systems to ensure coordinated care at different stages of the illness.
In the current Guidelines, we have presented algorithms to assist with antipsychotic medication choices and side effects monitoring for people with schizophrenia. In addition to publishing these Guidelines and their associated professional presentations, we plan to produce accompanying commentaries and companion documents to disseminate the recommendations widely. Nevertheless, these Guidelines can only be implemented effectively when they are utilised within an adequately resourced workforce with access to multidisciplinary health workers, including a lived experience workforce. High-level principles and models of care mentioned in these Guidelines (e.g. trauma-informed care, the Closing the Gap) can only be meaningfully implemented in real life if there is a stable and thriving mental health workforce and work environment. Related to this, we need to continue to evaluate and consider alternative ways to provide optimal care for people with schizophrenia. For instance, although the current evidence base is limited, peer-led interventions (Coles et al., 2022) and digital technologies (Sawyer et al., 2023) hold promise as complementary delivery modes for lifestyle interventions, particularly in areas where resources for qualified professionals are scarce.
Limitations
Although we tried to be as objective as possible by using the GRADE approach to evaluate the certainty of the existing empirical evidence relevant to clinical practice, the recommendations were based on subjective judgement based on the risks and benefits of each intervention by the Panel (Andrews et al., 2013; Guyatt et al., 2008). To mitigate the inherent biases associated with this process, we established a Panel consisting of psychiatrists with diverse areas of expertise who have worked in multiple settings, a trainee member, a representative with lived experience and a statistics expert. We also sought additional processes, such as consultations with a consumer consultation group and input from external reviewers, to reduce the risk of potential biases and groupthink when making the recommendations. Nevertheless, the values we hold would not be the same as the values of all the people with schizophrenia and those who care for them. These recommendations should be taken as guidelines only to help with the formulation and delivery of care that is most appropriate and optimal for the particular individual in the particular setting. Moreover, as these Guidelines were GRADE guidelines commissioned by the ANZJP, unlike the Clinical Practice Guidelines previously produced by the RANZCP, we have been limited to areas where there is empirical evidence. As such, we are unable to include expert opinion to cover areas where there is a lack of empirical evidence, limiting the breadth of these Guidelines. Previous RANZCP Clinical Practice Guidelines for schizophrenia (Galletly et al., 2016) should be considered for areas where we could not assess the certainty of empirical evidence.
Another limitation of these Guidelines was that most of the studies included in the reviews were not conducted in Australia or Aotearoa New Zealand. While some findings may be relevant even if based on studies in different settings (e.g. the efficacy of antipsychotic medications; Huhn et al., 2019), most topics examined in the current Guidelines would benefit from more relevant studies to determine their relevance in local settings. In particular, there was very little examination of the effectiveness of models of care for Indigenous peoples in either Australia or Aotearoa New Zealand, or those in rural areas. Given the uniqueness and disproportionate vulnerabilities facing these populations, further research is needed to advance the field. Related to this, we have limited exploration of issues relevant to other special subpopulations of people with schizophrenia in the current Guidelines. These include, but are not limited to, people from culturally and linguistically diverse groups (including refugee and migrant populations), people who are homeless, people who are incarcerated, women and people who identify as members of the lesbian, gay, bisexual, transgender and queer/questioning communities. As these sub-populations often have higher rates of mental health difficulties and unique sets of vulnerabilities and treatment needs, there is an ongoing need to improve the scientific evidence base specific to these cohorts. We plan to examine some of these issues in the upcoming accompanying commentaries.
Another topic with surprisingly little empirical evidence base was that of psychiatric comorbidities faced by people with schizophrenia. Given that comorbidities are common in psychiatry and their impact can be profound (Plana-Ripoll et al., 2020), furthering the understanding of the relationship between schizophrenia and comorbid psychiatric conditions would have significant clinical implications and enable the development of treatments tailored to address this complex comorbidity in people with schizophrenia. In particular, there is currently very little empirical research to guide us on the management of people with schizophrenia who have such comorbid conditions as neurodevelopmental disorders and substance use disorders. This is despite the fact that both attention deficit hyperactivity disorder (Cheng et al., 2025) and autism spectrum disorders (Zheng et al., 2018) are more prevalent among people with schizophrenia than the general population, and substance-induced psychosis is a very common clinical problem (Fiorentini et al., 2021).
Future directions
While we have attempted to capture what we considered to be the core aspects of care for people with schizophrenia, there remain many unanswered questions and much need for ongoing evaluation and research. Currently, many of our recommendations have been made with low to moderate levels of certainty, and some clinical questions require urgent investigation (e.g. what to do after clozapine therapy fails, and how best to manage comorbid psychiatric conditions, including substance misuse). We urgently need further research at both the basic science level examining the efficacy of different interventions, and the service/public health level evaluating the effectiveness of treatment implementation. For example, although we have made strong recommendations for a range of psychological and psychosocial interventions, implementing these in practice, particularly in rural and remote areas, remains a significant challenge. Novel and innovative models of care are needed to ensure that such interventions are delivered with sufficient quality and accessibility to those who need them most. Furthermore, to meet the GRADE guideline commission, we were unable to cover many important topics. They may include, but are not limited to, topics such as the use of the mental health legislation, pharmacological options for acute behaviour disturbances, medication adherence, substance-induced psychosis, care provision for other vulnerable subgroups, digital interventions, risk assessment and emerging interventions for people with schizophrenia and optimal service component structures. We hope to explore these topics in accompanying commentaries.
Conclusion
In conclusion, we echo the authors of the previous RANZCP clinical practice guidelines (Galletly et al., 2016) in acknowledging the extraordinary courage and perseverance shown by many people with schizophrenia and their families, whānau and carers. We hope that the current Guidelines provide useful recommendations based on the existing evidence base in important aspects of care for people with schizophrenia in Australia and Aotearoa New Zealand, both at the individual and systemic levels.
Supplemental Material
sj-docx-1-anp-10.1177_00048674251406058 – Supplemental material for Australian and New Zealand Journal of Psychiatry Grading of Recommendations, Assessment, Development and Evaluations (GRADE) guidelines for the management of schizophrenia
Supplemental material, sj-docx-1-anp-10.1177_00048674251406058 for Australian and New Zealand Journal of Psychiatry Grading of Recommendations, Assessment, Development and Evaluations (GRADE) guidelines for the management of schizophrenia by Shuichi Suetani, Frances Dark, Susanna Every-Palmer, Cherrie Galletly, Brian O’Donoghue, Sean Halstead, Dolores Keating, Nicole Korman, Julia Lappin, Sharon Lawn, Andrew Thompson, Nicola Warren and Dan Siskind in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
We thank the seven members of our consumer consultation group who provided invaluable insights into their lived experience throughout the process of the development of this guideline. They wish to remain anonymous. We also thank Rebecca Soole for helping us to summarise the consumer consultation group discussions.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: S.S. is an employee of Institute for Urban Indigenous Health. S.S. is an associate editor of the ANZJP and Therapeutic Advances in Psychopharmacology. S.S. is a section editor of the British Journal of Psychiatry. S.S. was a deputy editor of Australasian Psychiatry between 2021 and 2024. S.S. has received an honorarium from Sage Publishing (2025). S.S. was on the Seqirus medication advisory board for cariprazine (2020–2022). S.S. has also received honorarium from Inside Practice Psychiatry (2021) and GroupH (2021). F.D. is an employee of Metro South Addiction and Mental Health Service in Brisbane, Queensland. S.E.-P. is an employee of Te Whatu Ora Health New Zealand and of The University of Otago. S.E.-P. is a deputy editor of the ANZJP. S.E.-P. serves on the Committee for Research of the RANZCP. S.E.-P. is Vice President of the Pacific Rim College of Psychiatry. C.G. is on Advisory Boards for Boehringer Ingelheim, Lundbeck and Servier Laboratories. B.O. is an employee of University College Dublin with portions of his time attributed to St Vincent’s University Hospital and Dublin South East mental health service. B.O. previously received a fellowship from the NHMRC and currently holds funding from the Health Research Board in Ireland. B.O. is an associate editor of the Irish Journal of Psychological Medicine and Early Intervention in Psychiatry. S.H. is an employee of Queensland Health. S.H. has been supported by a Research Training Program scholarship from 2023 to 2025, as well as a RANZCP Foundation Partners Scholarship from 2025. D.K. is an employee of St John of God University Hospital. D.K. has never received financial support from the pharmaceutical industry. D.K. has been a collaborator on research grants received from the Health Research Board in Ireland. N.K. is an employee of Metro South Addiction and Mental Health Service in Brisbane, Queensland. N.K. has received funding from Otsuka to deliver an educational conference on lifestyle interventions. N.K. has previously received support from an RANZCP novice investigator grant. N.K. received a University of Queensland grant to fund consumer involvement in the development of these Guidelines. J.L. is an employee of UNSW Australia and New South Wales Health. J.L. is an Associate Editor of ANZJP. J.L. serves on the Committee for Research of the RANZCP. S.L. serves on the Committee for Research of the RANZCP. S.L. served on the Steering Group for the Future Development of Clinical Practice Guidelines as the lived experience representative of the RANZCP. A.T. is an employee of Parkville Youth Mental Health and Wellbeing Service. A.T. is an employee of the Orygen, the University of Melbourne. A.T. is a chief investigator on grants from the NHMRC and MRFF. A.T. has received honorarium for independent educational talks for Servier, Lundbeck and Otsuka. A.T. has received an unrestricted neuroscience research grant from Pfizer. A.T. has been on an advisory board for Lundbeck. A.T. has been an associate editor of the ANZJP. N.W. is an employee of Queensland Health. N.W. is an employee of The University of Queensland. N.W. is an associate editor of the ANZJP. N.W. serves on the Committees for Research; Foundation; Awards and Recognition; Practice, Policy and Partnerships; Queensland Branch of the RANZCP. N.W. has received honorarium for independent educational talks from Lundbeck. D.S. is an employee of Queensland Health. D.S. is a deputy editor of the ANZJP. D.S. serves on the Committee for Research of the RANZCP. D.S. has served on the Viatris Quality Assurance Committee and received honorarium for independent educational talks from Servier, Viatris and Lundbeck. D.S. has an NHMRC Investigator Grant GNT1194635 and is a chief investigator on grants from the NHMRC and MRFF.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Ethical approval and informed consent statements
Ethical approval was not required for this project.
ORCID iDs
Data availability statement
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
