Abstract
Background:
People experiencing psychosis are at greater risk of physical health conditions and premature mortality. It is likely that Indigenous Māori youth, who experience additional systemic inequities caused by settler-colonisation, face even greater physical health and mortality risks following a diagnosis of first-episode psychosis.
Objective:
Compare Māori and non-Māori for risk of hospitalisation and mortality for up to 15 years following first-episode psychosis diagnosis.
Methods:
A cohort (N = 14,122) of young people (16–24 years) with first-episode psychosis diagnosis between 2001 and 2019 were identified. Using crude Kaplan–Meier and adjusted Cox proportional hazards models, Māori (n = 5211) and non-Māori (n = 8911) were compared on hospitalisation and mortality outcomes for up to 15 years.
Results:
In the 15 years following first-episode psychosis diagnosis, Māori had higher adjusted risk of all-cause mortality (hazard ratio = 1.21, 95% confidence interval = [1.01, 1.45]), hospitalisation with diabetes (hazard ratio = 1.44, 95% confidence interval = [1.15, 1.79]), injury/poisoning (hazard ratio = 1.11, 95% confidence interval = [1.05, 1.16]), general physical health conditions (hazard ratio = 1.07, 95% confidence interval = [1.02, 1.13]) and also appeared to be at greater risk of cardiovascular hospitalisations (hazard ratio = 1.34, 95% confidence interval = [0.97, 1.86]). Kaplan–Meier plots show hospitalisation and mortality inequities emerging approximately 4–7 years following first-episode psychosis diagnosis.
Conclusions:
Māori are at greater risk for hospitalisation and premature mortality outcomes following first-episode psychosis. Early screening and intervention, facilitated by culturally safe health service delivery, is needed to target these inequities early.
Keywords
Background
Psychosis is a feature of some serious mental illness, such as schizophrenia and type I bipolar disorder (Firth et al., 2019). People with psychosis report a wide range of experiences, including symptoms related to impaired perception of reality. Some commonly reported psychosis symptoms include delusions, hallucinations, paranoia, grandiose ideation and loss of agency or control (Fusar-Poli et al., 2022). Both worldwide and in Aotearoa New Zealand (NZ), people experiencing psychosis have premature mortality, living, on average, approximately 20 years less than the general population (Cunningham et al., 2014; Laursen et al., 2014; Te Pou o Te Whakaaro Nui, 2014; Walker et al., 2015). Premature mortality for people experiencing psychosis is primarily driven by premature deaths from physical illness, injury/poisoning and suicide (Correll et al., 2017; Cunningham et al., 2014; Firth et al., 2019; Olfson et al., 2015).
There are many systemic contributors to physical health inequity for people experiencing psychosis. Relative to the general population, they are more often exposed to common risk factors for long-term physical health conditions (e.g. smoking, problematic alcohol use, poor access to nutrition and sedentary behaviour), such as cardiovascular disease (CVD) and diabetes (Di Florio et al., 2014; Jackson et al., 2015; Koskinen et al., 2009; Quigley and MacCabe, 2019; Stubbs et al., 2016; Teasdale et al., 2019; Vancampfort et al., 2017). Notably, risk of CVD and diabetes are increased by cardiometabolic side effects of commonly prescribed second-generation antipsychotic medications (Galletly et al., 2012, 2016; Mitchell et al., 2011). People experiencing psychosis also typically have reduced access to physical health care, and sub-optimal quality of care (Ayerbe et al., 2018; Cunningham et al., 2023a, 2023b; Firth et al., 2019).
It is also important to consider how disadvantages related to psychosis may be compounded for people also experiencing systemic racism. The experience of multiple systemic contributors to physical ill health (i.e. psychosis and racism, in this case) may produce a cumulative detriment to health for people in these communities (Denise, 2014; Dowd and Bengtson, 1978). When considering the intersection of psychosis and ethnicity, diagnoses of psychotic disorders have been reported as more common in non-dominant ethnicities across many settings (Gynther et al., 2019; Jongsma et al., 2018; Schofield et al., 2016; Termorshuizen et al., 2014; Veling, 2013). Empirical studies across these various settings have yielded mixed findings when comparing the physical health of dominant and non-dominant ethnic groups experiencing psychosis (e.g. Cunningham et al., 2020; Das-Munshi et al., 2017; Daumit et al., 2010; Musuuza et al., 2013).
In NZ, Indigenous Māori experience gross health inequity when compared to non-Māori, and particularly when compared to NZ Europeans (Ministry of Health, 2018, 2019). Māori health inequity is understood as an outcome of historical and contemporary settler-colonisation, which has entrenched systemic racism (Lacey et al., 2022; Moewaka Barnes and McCreanor, 2019; Reid et al., 2019). Due to systemic advantages for primarily NZ European people, Māori are more likely than non-Māori to experience determinants of poor health, such as socioeconomic deprivation and other forms of racism (Harris et al., 2018; Ministry of Health, 2019, 2023). Against this background, Māori experience higher rates of long-term physical health conditions, such as CVD, diabetes and cancer, and are also more likely to be diagnosed with psychotic disorders (Kake et al., 2008; Linscott et al., 2006; Ministry of Health, 2019, 2023; Petrović-van der Deen et al., 2020). Due to reported barriers, Māori have poorer access to health services, leading to worse outcomes from physical health conditions when compared to non-Māori (Espiner et al., 2021; Graham and Masters-Awatere, 2020; Jansen et al., 2009; Ministry of Health, 2019, 2023). Moreover, Māori-run service providers have been strategically underfunded by the Crown (Waitangi Tribunal, 2021). Consequently, Māori have a life expectancy approximately 7 years less than non-Māori (Ministry of Health, 2018).
First-episode psychosis (FEP) is the first clinical presentation of psychosis, and may be the onset of psychotic disorder, such as schizophrenia. Age of FEP onset is typically adolescence or early adulthood (Reed, 2008; Solmi et al., 2022a). Despite relatively early onset, several longitudinal studies have shown that the risk of premature mortality, from both physical illness and other causes, increases substantially following FEP diagnosis (Craig et al., 2006; Schoenbaum et al., 2017; Simon et al., 2018; Tanskanen et al., 2018; Yung et al., 2023). In NZ, rangatahi (young) Māori are approximately twice as likely as non-Māori youth to be diagnosed with FEP (Petrović-van der Deen et al., 2020). Given the increase in physical morbidity and mortality risk associated with psychosis, this is a considerable health equity concern.
Te Pūrākau o Te Pu Korokoro
This study is from Phase 1 of a larger research project, Te Pu Korokoro: Improving the physical health of Māori with psychosis. In May each year, the kuaka (godwit) birds migrate 12,000 km from Aotearoa (NZ) to Alaska. In September, the kuaka arrive back in Aotearoa from Alaska. They settle along the mudflats in Tāmaki (Auckland), and for over a 1000 km down the east coast to Ōtautahi (Christchurch). Te Pu Korokoro symbolises the air pocket formed during the kuaka’s flight; it is a place to rest and be protected from the winds. Tāngata whaiora (people seeking wellness) and their supporters describe falling through gaps and losing support, like falling from an air pocket into harsh winds. Like the migration cycle of the kuaka, Te Pu Korokoro has several phases, and all are essential. This paper is the first in a series that aims to generate knowledge to improve outcomes, and ultimately deliver recommendations based on the voices of Māori experiencing psychosis.
Aims
This study analyses the physical health of a cohort of young people for up to 15 years following FEP diagnosis. In particular, rates of hospitalisation and premature mortality are compared between Māori and non-Māori. Within hospitalisations, we focused particularly on CVD- and diabetes-related admissions, as major causes of morbidity associated with psychosis diagnoses. The aims of this study are:
Estimate and compare the risk of hospitalisation events (including CVD- and diabetes-specific admissions) in Māori and non-Māori for up to 15 years following FEP diagnosis.
Estimate and compare the risk of premature mortality (all-cause and cause-specific) in Māori and non-Māori for up to 15 years following FEP diagnosis.
Method
Ethics
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008. All procedures involving human subjects/patients were approved by University of Otago Ethics Committee, reference number HD22/089.
Participants
Two datasets from Te Whatu Ora/Health New Zealand National Collections were used to obtain a cohort of people with a diagnosis of FEP. The Programme for the Integration of Mental Health Data (PRIMHD) contains all contacts with secondary mental health and addiction services since July 2007 (Te Whatu Ora/Health New Zealand, 2023e). The National Minimum Dataset (NMDS) contains all public and many private hospitalisation data (Te Whatu Ora/Health New Zealand, 2023c). Encrypted National Health Index (NHI) identifiers were used to link patient records between all National Collections datasets used in this study. Participant consent is not required to analyse de-identified National Collections records (Te Whatu Ora/Health New Zealand, 2023a).
People with a Diagnostic and Statistical Manual of Mental Disorders (4th ed.; DSM-IV), International Classification of Diseases, Ninth Revision (ICD-9) or ICD-10 diagnostic code (principal, other relevant or provisional) recorded for a psychotic disorder between 2001 and 2019 in either PRIMHD or the NMDS were included, covering diagnoses associated with both inpatient and outpatient interactions. Diagnostic codes include schizophrenia, bipolar disorder, schizoaffective disorder, depressive disorder with psychotic symptoms, organic psychotic disorder, substance-induced psychotic disorder and psychosis not otherwise specified (NOS; see Supplementary Table S1 for codes).
Previous hospitalisation records were checked back to 1997 to rule out prior psychosis diagnosis. People aged 16–24 years at FEP diagnosis were retained in the cohort. For each person, a maximum of 15 years of follow-up data were extracted, meaning the oldest people (those aged 24 years at FEP diagnosis, with 15 years of follow-up data available) were 39 years old at the end of follow-up. The date of each person’s first recorded psychosis diagnosis (in either PRIMHD or NMDS) was taken as their index date for follow-up data extraction.
Measures
Ethnicity
Reported analyses compare rangatahi Māori to non-Māori youth (all aged 16–24 years at FEP diagnosis). Ethnicity is recorded in the Ministry of Health’s NHI dataset, and should be collected and checked at recommended intervals during healthcare interactions, with protocols for collection stipulating that this should be self-identified ethnicity (Health Information Standards Organisation, 2017). Māori were identified by prioritised ethnicity; people were included in the rangatahi Māori cohort if they self-identified as Māori, regardless of any other ethnicities. All other people were categorised as non-Māori.
Physical health measures
Records for hospitalisation (NMDS), pharmaceutical dispensing (Te Whatu Ora/Health New Zealand, 2023d) and mortality (Te Whatu Ora/Health New Zealand, 2023b) were linked to each person anonymously via encrypted NHI number.
Physical health status at FEP diagnosis (M3)
Physical health status at FEP diagnosis was assessed using a modified version of the M3 multimorbidity index (Stanley and Sarfati, 2017), which quantifies multimorbidity using ICD-10 diagnoses recorded in hospitalisation data over a 5-year lookback from index date (FEP diagnosis for the present cohort), and assigns weights from 55 medical conditions based on their predictiveness of mortality. For this study, five psychiatric categories were excluded from the M3 score calculation (major psychiatric disorder; mental and behavioural disorder; anxiety and behavioural disorder; substance use disorder) to focus on physical conditions. To describe physical health at FEP diagnosis, M3 scores were categorised into 0, > 0 to < 1, 1 to < 2, and ⩾ 2. A score of zero indicates that no relevant diagnostic codes were present in hospitalisation data; higher scores indicate presence of condition(s) associated with higher mortality.
Physical health following diagnosis
Following index date (FEP diagnosis), up to 15 years of follow-up health data were extracted for each person, up to 31 October 2021. Hospitalisation and mortality records were used to define the main outcomes following FEP diagnosis. All hospitalisations were filtered to only include medical and surgical hospital admissions (Health Specialty Codes beginning ‘M’ or ‘S’), hence excluding psychiatric unit admissions.
Physical health hospitalisations
Physical health hospitalisations were defined as hospitalisation events where there was a recorded diagnosis (primary or secondary) of a physical health condition (ICD-9 codes 001–289, 320–629, 680–739; ICD-10 codes A00–E89, G00–N99). Thus, hospitalisations with only psychiatric diagnoses were excluded. Admissions containing only diagnoses relating to pregnancy and childbirth (e.g. gestational diabetes) were also excluded to avoid bias, as Māori have a mean maternal age 3.5 years younger than non-Māori, and are more likely than non-Māori to have children before age 20 years (Marie et al., 2011; Rarere et al., 2023).
Injury/poisoning hospitalisations
Injury/poisoning hospitalisations were defined as hospitalisation events where any injury/poisoning (including burns) diagnosis (primary or secondary) was recorded (ICD-9 codes 800–999; ICD-10 codes S00–T88).
Diabetes hospitalisations
Diabetes hospitalisations included hospitalisations where any diagnosis (primary or secondary) of diabetes was recorded (ICD-9 codes 249 and 250; ICD-10 codes E08–E13), excluding gestational diabetes.
CVD hospitalisations
CVD hospitalisations included hospitalisations where any diagnosis (primary or secondary) of CVD was recorded. Categories included from both ICD-9 and ICD-10 codes were: myocardial infarction, unstable angina, ischaemic stroke, haemorrhagic stroke, transient ischaemic attack, other coronary heart disease, peripheral vascular disease, congestive heart failure, and other ischaemic CVD-related codes (see Supplementary Table S2 for codes).
All-cause mortality, non-medical mortality and medical mortality
Premature mortality was categorised into all-cause mortality (death from any cause), non-medical mortality (intentional and accidental events of ‘external cause’ resulting in injury, poisoning or burns; ICD-10 codes starting with V, W, X or Y as underlying cause of death) and medical mortality (any other ICD-10 code as underlying cause of death).
Covariates
Gender and date of birth were obtained from the Ministry of Health NHI dataset. Socioeconomic deprivation was assessed with NZDep2018 quintiles, which indicates the relative deprivation for small geographic areas of NZ based on the 2018 NZ census (Atkinson et al., 2019).
Non-psychotic psychiatric comorbidities were identified from the NMDS and PRIMHD, and were categorised into depression, anxiety, substance use disorders and personality disorders. In NZ, for inpatient admissions, diagnostic coding is performed by trained coders who code each diagnosis based on a standardised set of rules applied to the clinical file. In outpatient specialist care, a diagnosis must be entered by clinicians either at discharge or after 3 months of care.
Analysis
All data cleaning and statistical analysis was performed in R (R 4.1, R Institute, Vienna, Austria).
Descriptive analyses present demographic characteristics, physical multimorbidity at index date and other clinical characteristics for Māori and non-Māori. These descriptive analyses are presented as profiles (counts and percentages): no formal statistical comparisons are made, as the objective of this table is to describe general differences by ethnicity that may be relevant for considering confounding (Vandenbroucke et al., 2007).
To handle variable follow-up periods, crude Kaplan–Meier analysis was performed to estimate the risk of outcome events occurring over a maximum of 15 years following FEP diagnosis. Separate Kaplan–Meier functions compared Māori and non-Māori for each outcome event: all-cause mortality, medical mortality, non-medical mortality, physical health hospitalisation, injury/poisoning hospitalisation, diabetes hospitalisation and CVD hospitalisation. In analyses of hospitalisation outcomes, records were censored at death if it occurred before hospitalisations were observed. Records were also censored at death in cause-specific mortality plots if the person died of another cause.
Further Kaplan–Meier analysis was performed to separate injury/poisoning hospitalisation and non-medical mortality categories into intentional (self-harm/suicide) and presumed accidental events. Injury/poisoning hospitalisations were coded as self-harm if any self-harm ICD-9 or ICD-10 code was recorded for the event; all other injury/poisoning hospitalisations were coded as accidental. Non-medical deaths were coded as suicide if any self-harm/suicide ICD-10 code was recorded for the death; all other non-medical deaths were coded as accidental.
Cox proportional hazards regressions were performed to adjust for confounding while allowing for censoring of follow-up time. Crude and adjusted (age, gender and socioeconomic deprivation) Cox regression models estimate the risk of follow-up events for Māori relative to non-Māori. Supplementary Cox regression analyses were performed to explore gender differences in Māori and non-Māori.
Results
A total of 14,122 people were diagnosed with FEP between 2001 and 2019 while aged 16–24 years. Of these, 9074 FEP diagnoses were recorded in hospital admissions data (NMDS), and an additional 5048 were identified from mental health service data (PRIMHD). 1 Of this total cohort, 5211 (36.9%) were rangatahi Māori. All analyses compare this rangatahi Māori cohort (n = 5211) to non-Māori youth (n = 8911).
Table 1 shows the demographic and clinical characteristics for rangatahi Māori and non-Māori youth. Māori were younger at FEP diagnosis, and had higher exposure to socioeconomic deprivation, with 46.7% of Māori living in areas in the most deprived quintile, and 73.5% in the two most deprived quintiles. Over the study (including FEP and follow-up diagnoses), Māori were more likely to receive diagnoses of schizophrenia, schizoaffective disorder, substance-induced psychosis, organic psychosis, other psychosis and psychosis NOS. Non-Māori were more likely to receive diagnoses of bipolar disorder and depression with psychosis, and were more likely to be solely diagnosed with psychosis NOS. In terms of non-psychotic psychiatric comorbidities, Māori were more likely to be diagnosed with a substance use disorder, and less likely than non-Māori to be diagnosed with depression or an anxiety disorder. Both Māori and non-Māori had very low physical multimorbidity (as measured by hospitalisation diagnoses) at the time of FEP diagnosis. Over 90% of both Māori (91.9%) and non-Māori (91.8%) did not receive any of the M3 index physical health diagnoses in an inpatient hospital setting in the 5 years prior to FEP diagnosis.
Cohort demographic and clinical characteristics compared between Māori (n = 5211) and non-Māori (n = 8911) who experienced FEP, at index date and in 15-year follow-up.
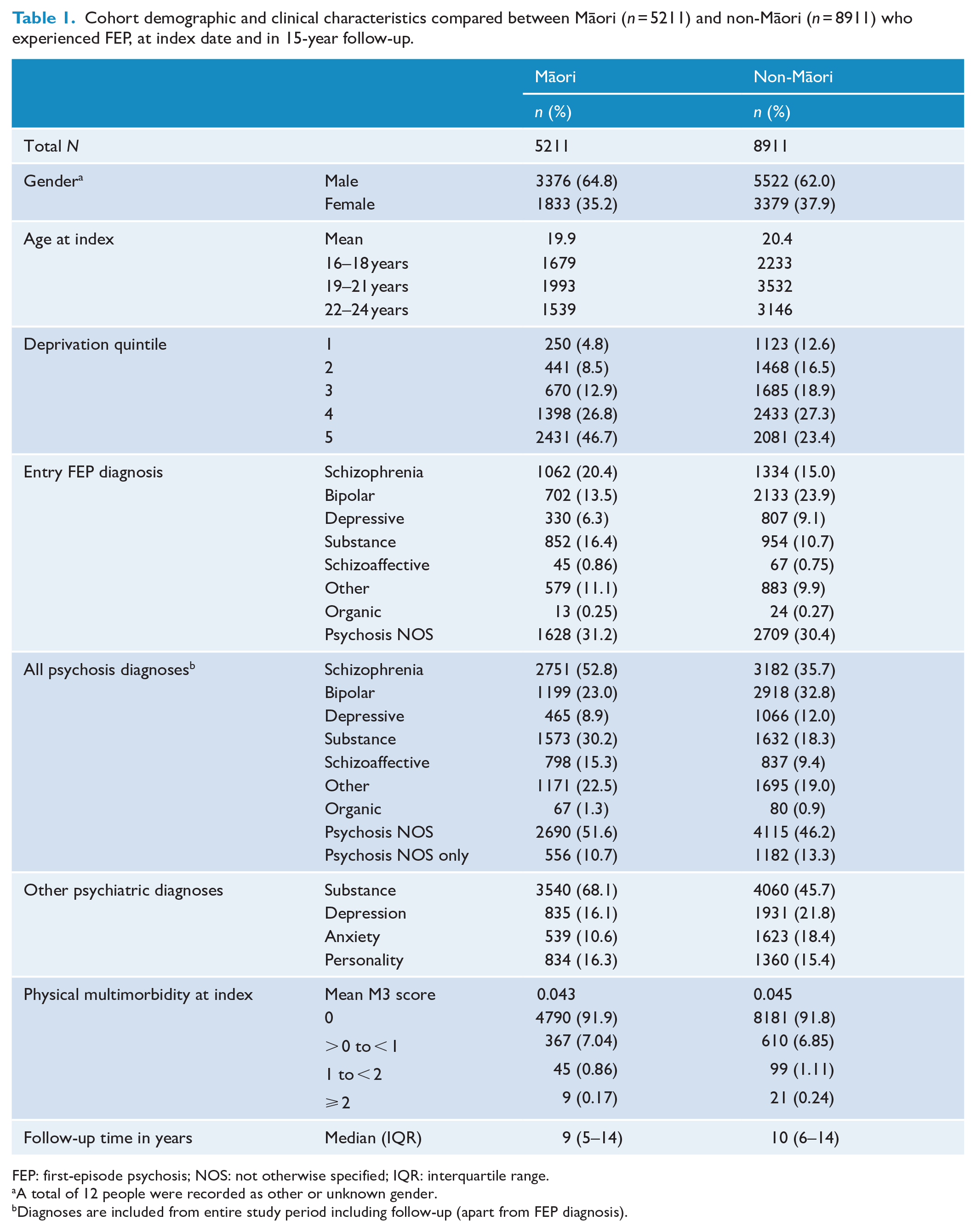
FEP: first-episode psychosis; NOS: not otherwise specified; IQR: interquartile range.
A total of 12 people were recorded as other or unknown gender.
Diagnoses are included from entire study period including follow-up (apart from FEP diagnosis).
Table 2 shows the risk of follow-up events for Māori and non-Māori within 15 years of follow-up. The median follow-up time was 9 years (interquartile range [IQR] = 5–14) for Māori and 10 years (IQR = 6–14) for non-Māori. Visual inspection of Kaplan–Meier log–log transformation plots indicated that the proportional hazards assumption was acceptable for hospitalisation and mortality outcomes. Māori had higher risk of all measured outcomes compared to non-Māori at the end of follow-up. After adjusting for confounding (age, gender and socioeconomic deprivation), Māori had greater risk of all-cause mortality (hazard ratio [HR] = 1.21, 95% confidence interval [CI] = [1.01, 1.45]), physical health hospitalisation (HR = 1.07, 95% CI = [1.02, 1.13]), injury/poisoning hospitalisation (HR = 1.11, 95% CI = [1.05, 1.16]) and diabetes hospitalisation (HR = 1.44, 95% CI = [1.15, 1.79]). There was also some indication that Māori were at greater risk of CVD hospitalisation; however, this finding did not exclude the null (HR = 1.34, 95% CI = [0.97, 1.86]). Supplementary analysis showed that significant gender differences in outcome risk were the same for Māori and non-Māori: females were at greater risk of physical health, injury/poisoning and diabetes hospitalisations; males were at greater risk of all-cause mortality (see Supplementary Table S3).
Follow-up mortality and hospitalisation risk and Cox regression hazards for Māori (n = 5211) compared to non-Māori (n = 8911) experiencing psychosis.
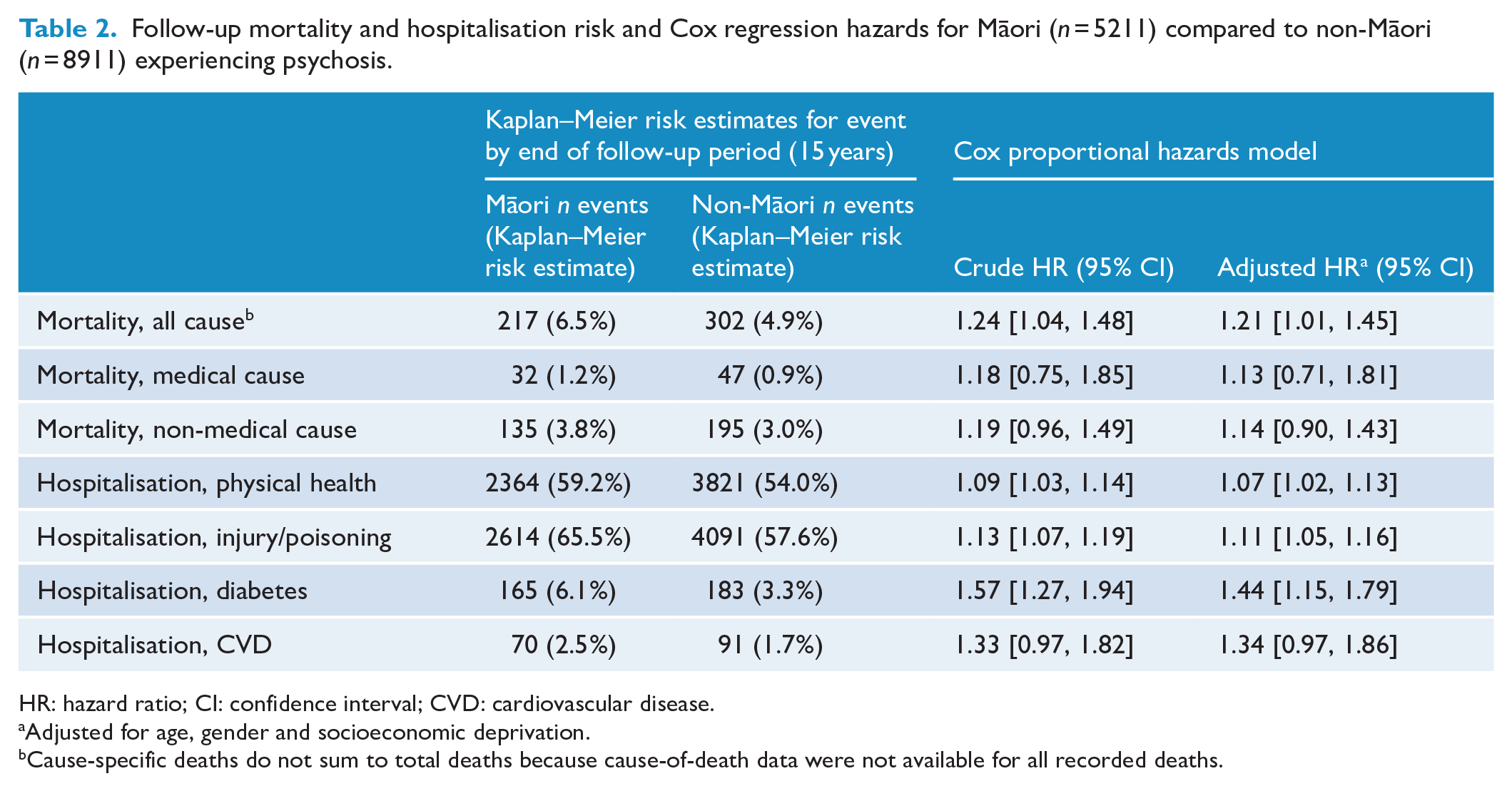
HR: hazard ratio; CI: confidence interval; CVD: cardiovascular disease.
Adjusted for age, gender and socioeconomic deprivation.
Cause-specific deaths do not sum to total deaths because cause-of-death data were not available for all recorded deaths.
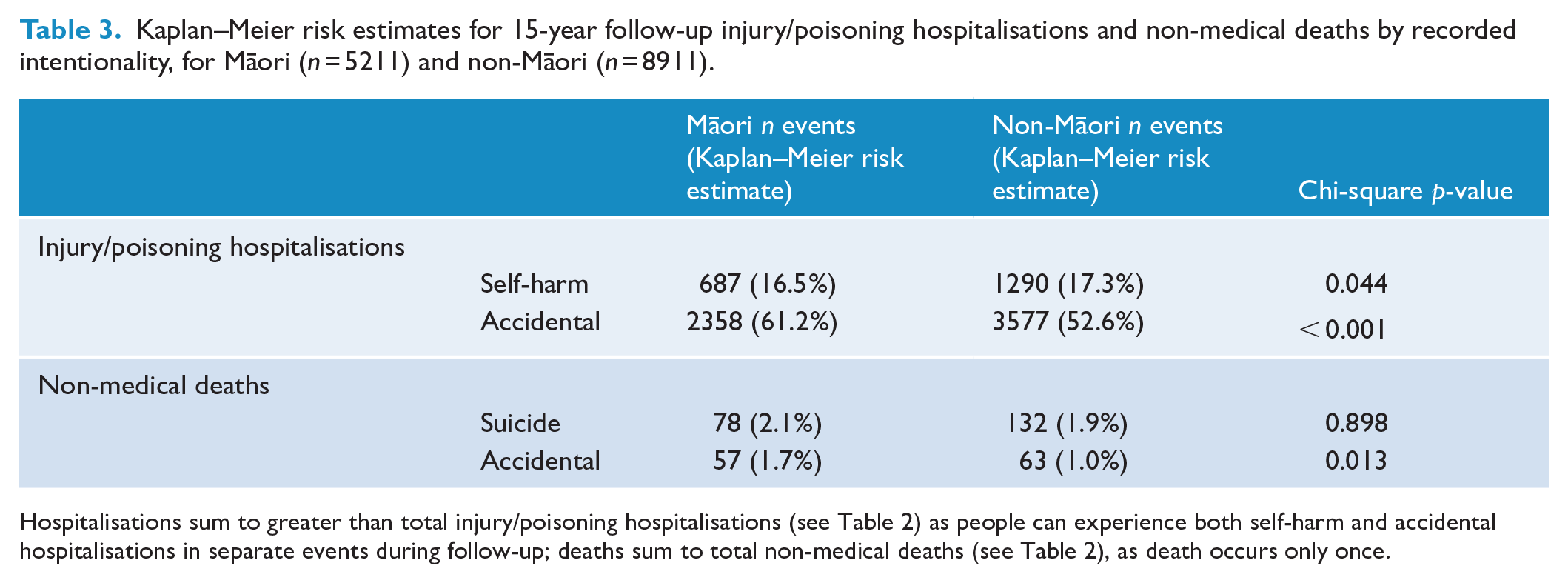
Further analysis was performed to separate injury/poisoning hospitalisation and non-medical mortality categories into self-harm/suicide and presumed accidental events. This analysis is reported in Table 3, with the risk of each event type for Māori and non-Māori. Māori had higher risk of accidental injury/poisoning hospitalisation (p < 0.001) and accidental death (p = 0.013) than non-Māori. However, suicide was the most common cause of death for both Māori and non-Māori (210/409 of all deaths with a recorded cause). Accidental poisoning (n = 44) and transport accidents (n = 37) accounted for 81/120 (67.5%) accidental deaths. Of the 44 accidental poisoning deaths, 22 were primarily due to medications and 22 were primarily due to narcotics, alcohol and other substances. 2
Kaplan–Meier risk estimates for 15-year follow-up injury/poisoning hospitalisations and non-medical deaths by recorded intentionality, for Māori (n = 5211) and non-Māori (n = 8911).
The Kaplan–Meier plots for all-cause mortality (Figure 1) diabetes hospitalisation (Figure 2) and CVD hospitalisation (Figure 3) are presented, comparing the crude Kaplan–Meier estimates for Māori and non-Māori over the follow-up period. Kaplan–Meier plots for other study outcomes are included in Supplementary materials (see Figures S1–S4). For outcomes with differences between Māori and non-Māori, the Kaplan–Meier functions for Māori and non-Māori appear to begin diverging approximately 4–7 years after FEP diagnosis, indicating physical health inequities for Māori experiencing psychosis begin to emerge in these data during this period.
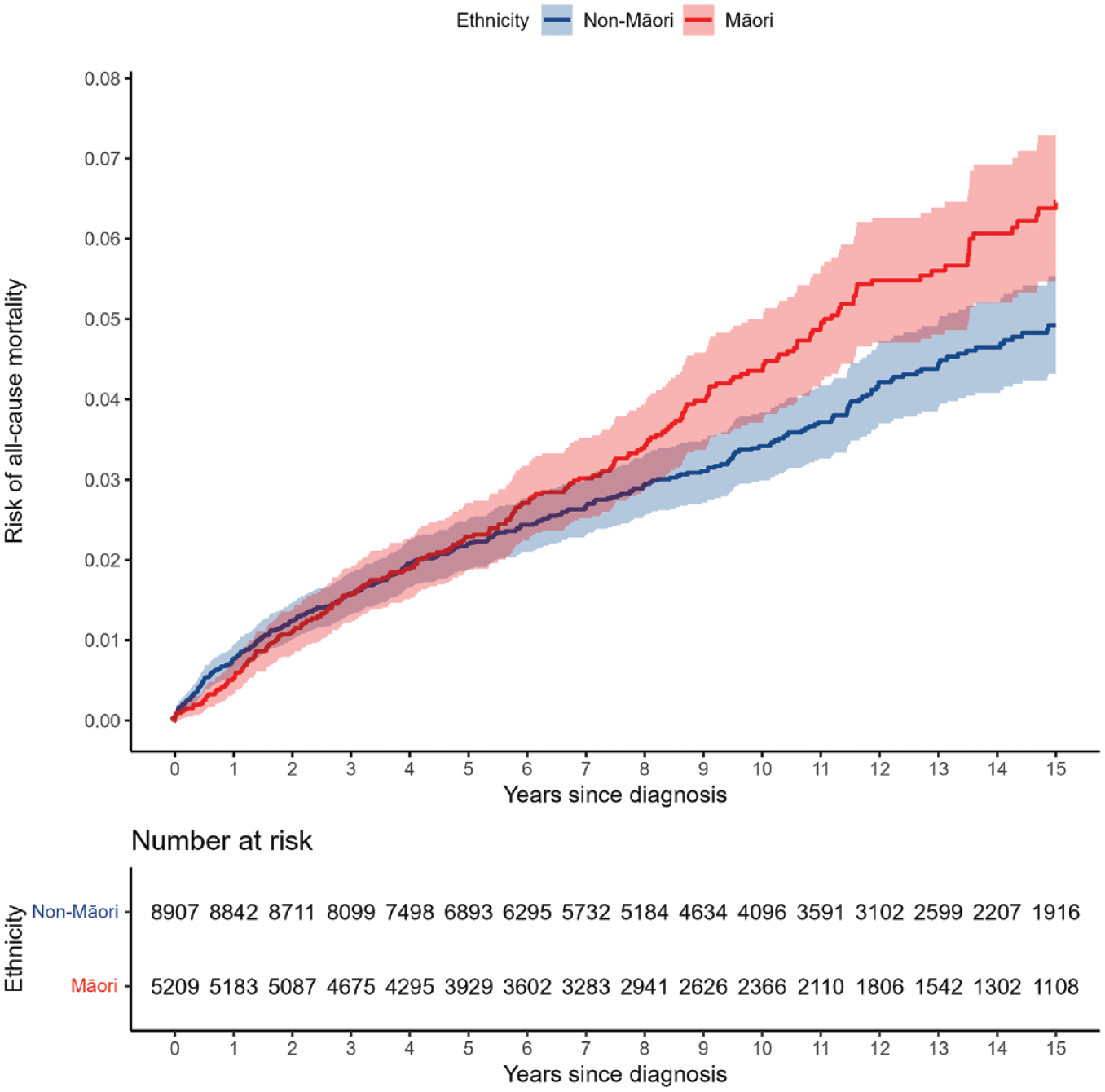
Māori and non-Māori Kaplan–Meier risk of all-cause mortality in 15-year follow-up from first-episode psychosis (shaded areas indicate 95% confidence intervals).
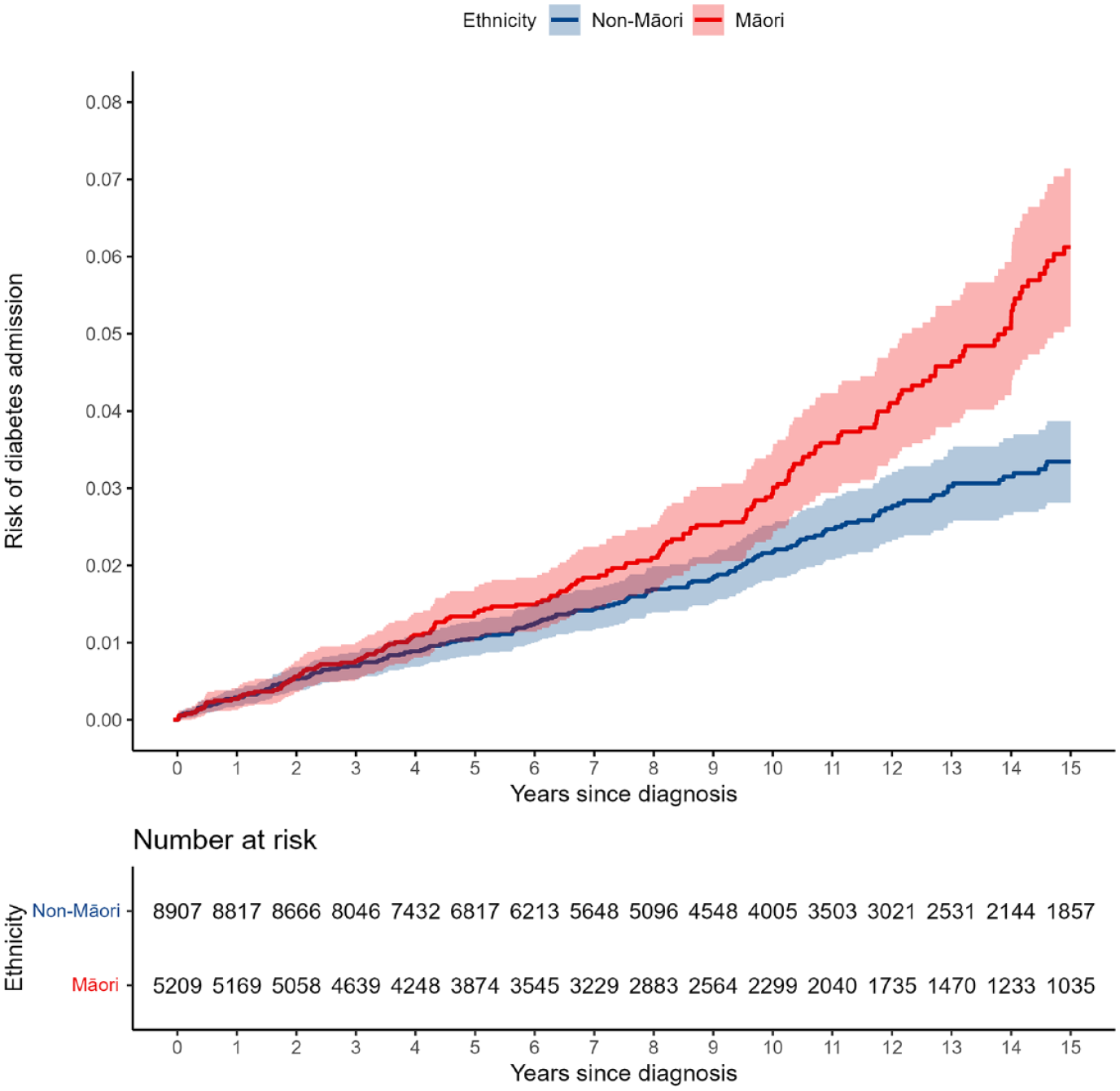
Māori and non-Māori Kaplan–Meier risk of diabetes hospitalisation in 15-year follow-up from first-episode psychosis (shaded areas indicate 95% confidence intervals).
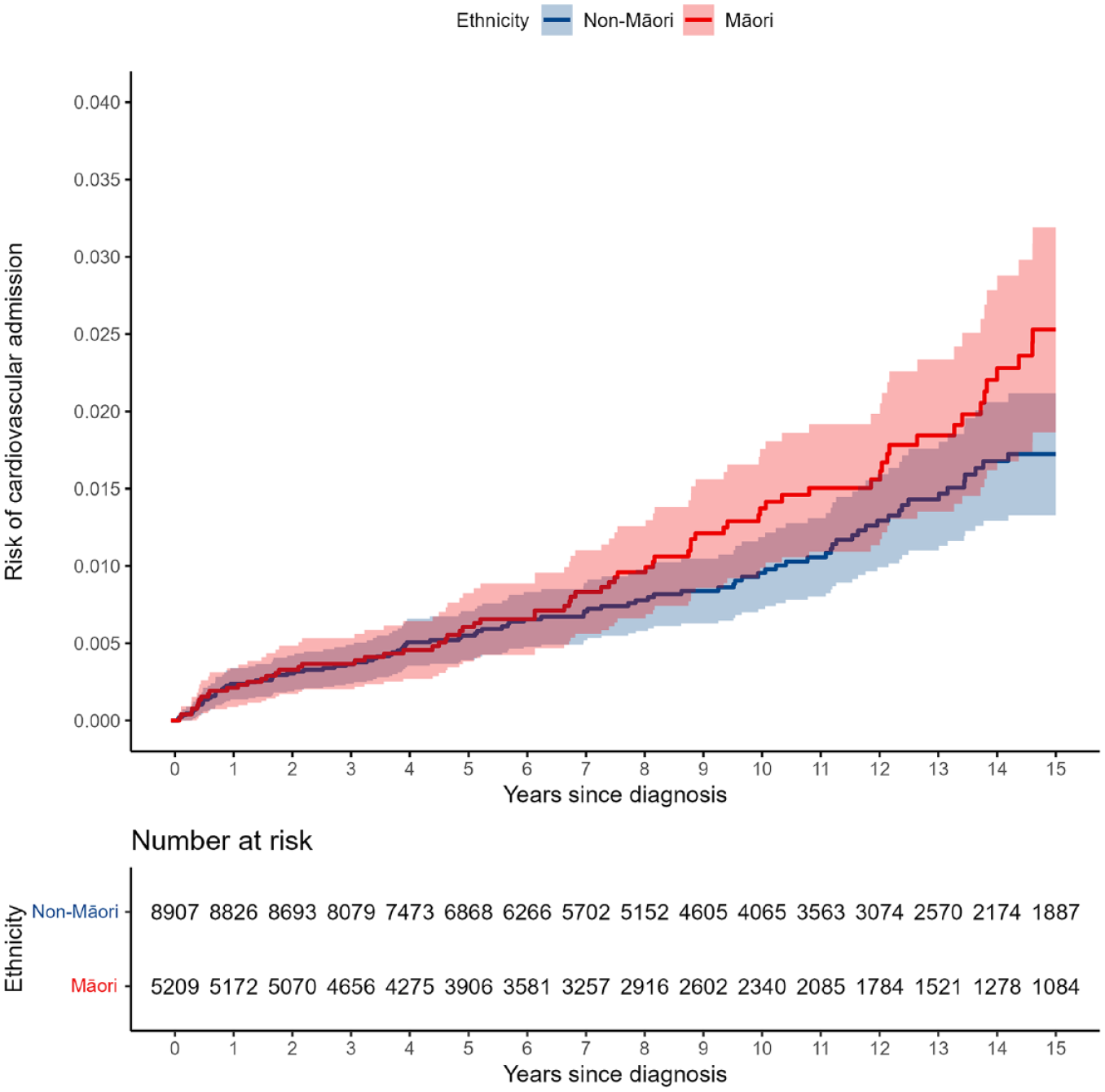
Māori and non-Māori Kaplan–Meier risk of cardiovascular diseases hospitalisation in 15-year follow-up from first-episode psychosis (shaded areas indicate 95% confidence intervals).
Discussion
Summary and interpretation of results
We analysed the hospitalisation and mortality records of 14,122 people for up to 15 years following FEP diagnosis at 16–24 years old. Māori had a higher adjusted risk of all-cause mortality, physical health hospitalisation, injury/poisoning hospitalisation and diabetes hospitalisation compared to non-Māori in the 15 years following FEP diagnosis. Māori also appeared to have a higher adjusted risk of CVD hospitalisation than non-Māori, although this result was less precisely estimated and data were compatible with no difference. Kaplan–Meier plots show that these inequities emerge approximately 4–7 years following FEP diagnosis. Comparable M3 scores for Māori and non-Māori at FEP diagnosis further indicate that physical health inequities for Māori and non-Māori emerge following the onset on psychosis. Gender differences in outcome risk were found in supplementary analyses, which were consistent between Māori and non-Māori: males had higher risk of all-cause mortality, while females were at greater risk of diabetes, injury/poisoning, and physical health hospitalisations. In this relatively young cohort, most recorded causes of death were non-medical (330/409 deaths with a recorded cause) and ethnic differences in premature mortality were thus driven mostly by these non-medical deaths. Nearly two-thirds (63.6%) of non-medical deaths were suicide, which was the most common cause of death for Māori and non-Māori. Risk of suicide during follow-up was similar for Māori and non-Māori; however, Māori had a higher risk of accidental death than non-Māori. The leading causes of accidental death were accidental poisoning and transport accident.
Overall, follow-up risk estimates for general hospitalisation events (physical health and injury/poisoning) were much higher than for condition-specific hospitalisations (diabetes and CVD). This is unsurprising, as the former are broad categories including many common health conditions, while the latter are specific long-term conditions. However, given the long-term morbidity and mortality associated with CVD and diabetes, it is concerning to find higher risk for hospitalisation with these particular conditions among Māori experiencing psychosis at this relatively young age (31–39 years at end of follow-up). This finding is consistent with higher rates of these conditions, and associated health markers, among Māori in the general population (Ministry of Health, 2019, 2023). Moreover, sub-optimal physical health care and management in primary care may mean these conditions are more likely to progress to a hospitalisation in Māori (Cunningham et al., 2023a, 2023b; Graham and Masters-Awatere, 2020; Ministry of Health, 2019).
Consistent with previous research with young FEP cohorts (Bromet et al., 2005; Simon et al., 2018; Wiersma et al., 1998; Yuen et al., 2014), we found that suicide and accidents (in particular, accidental poisonings) accounted for approximately 80% of mortality with a recorded cause in the 15 years following FEP diagnosis. One recent systematic review reported depression, suicidality and substance use as the strongest predictors of mortality in people with schizophrenia, and thus should be prioritised for screening, support and intervention (Correll et al., 2022). However, while poor physical health is not the most immediate cause of mortality in young people experiencing psychosis, it will increase mortality later in life. In particular, inequities in CVD and diabetes admissions reported in our young cohort should be expected to progress into substantial inequities in medical-cause mortality for Māori beyond our 15-year follow-up period.
Clinicians must consider both the mental and physical health of individuals experiencing psychosis. Antipsychotic medications produce strong cardiometabolic side effects (Firth et al., 2019; Galletly et al., 2012) compounded by complex factors such as problematic substance use, lack of access to good nutrition and lower physical activity (de Leon and Diaz, 2005; Di Florio et al., 2014; Firth et al., 2019; Jackson et al., 2015; Koskinen et al., 2009; Stubbs et al., 2016; Teasdale et al., 2019; Vancampfort et al., 2017). However, despite their established cardiometabolic side effects, recent meta-analyses have shown an overall reduction in all-cause mortality for people with schizophrenia taking antipsychotic medication (Correll et al., 2022; Jia et al., 2022). Reductions in mortality are most marked for people taking second-generation antipsychotics, especially clozapine and olanzapine, which are known to carry higher cardiometabolic risks (Correll et al., 2022). Thus, it appears that effective treatment of ongoing psychosis may reduce mortality from non-medical causes, despite elevating cardiometabolic risk. Antipsychotic use is also associated with greater adherence to cardiometabolic medications in people experiencing psychosis (Solmi et al., 2022b).
As a guideline for reducing physical health inequity related to psychosis and its treatment, the Healthy Active Lives (HeAL) consensus statement (Shiers and Curtis, 2014; International Physical Health in Youth (iphYs) working group, 2013) summarises the challenges, actions and targets for improving the physical health of young people experiencing psychosis, particularly those taking antipsychotic medication. In addition to pharmaceutical and clinical treatment, there is a need to further evaluate the use of integrated care models involving support from health improvement practitioners, health coaches and support workers, alongside clinicians. Integrated care programmes aim to improve outcomes by taking a broader ‘person-centred’ approach to treatment that places an emphasis on social need (Codyre et al., 2021). In NZ, pilot evaluations of one integrated care programme, Te Tumu Waiora, have indicated some promising physical, mental and social outcomes, including for Māori, but further rollout and evaluation is needed (Codyre et al., 2021).
Inequities in physical health and predictors of health (e.g. socioeconomic deprivation and substance use disorders) found in this FEP cohort reflect Māori health inequities in the general population (Ministry of Health, 2018, 2019, 2023). Adverse health and social exposures for Māori are a consequence of multi-generational harm caused by settler-colonisation; this history must be navigated in pursuit of health equity (Reid et al., 2014; Waitangi Tribunal, 2021; Waitoki et al., 2014). Importantly, the NZ health system sits within this history. While most Māori report receiving good healthcare, the current health system does not meet practice guidelines for many Māori, particularly Māori experiencing psychosis (Cunningham et al., 2023a, 2023b; Espiner et al., 2021; Haitana et al., 2022a; Jansen et al., 2009). Māori experiencing psychosis may experience culturally unsafe care in the health system, including both structural and interpersonal racism (Haitana et al., 2022b, 2023; Manuel et al., 2023), and diagnostic overshadowing (Cunningham et al., 2023a, 2023b). These experiences may be improved by providing clear pathways through care, improving the clinical culture and the enhancement of whānau (family) involvement and support (Haitana et al., 2022a). Cultural safety for Māori is needed to improve engagement with the health system; particularly for Māori who may not view their psychosis through a colonial lens or feel comfortable with staff unable to relate to their experience (Taitimu et al., 2018). To this end, Kaupapa Māori healthcare and support grounded in Kaupapa Māori research should be available for ranagtahi Māori experiencing psychosis (e.g. Meihana model, Hui process and Whānau Ora; Haitana et al., 2020, 2022b; Huria et al., 2019; Pitama et al., 2014; Te Rau Matatini, 2015). Given these findings, effective physical and mental health care and support, facilitated by culturally safe delivery, should be prioritised for Māori immediately following FEP diagnosis.
Future research directions
It is likely that the risks of hospitalisation and mortality reported here represent large risk increases from the same-age general population (Craig et al., 2006; Simon et al., 2018; Smith et al., 2013; Tanskanen et al., 2018; Yuen et al., 2014). People experiencing psychosis are an especially disadvantaged group with regard to physical health, and ethnic inequities we have identified for Māori sit atop this baseline inequity. However, as we did not compare the FEP cohort to the general population, we do not make these inferences on the present data. Moreover, it is difficult to compare estimated risks of diabetes and CVD hospitalisations to the general population as general population hospitalisation data for these conditions are not readily available. As a soft point of reference, the estimated rate of diabetes among 35–39-year-olds in the general population is 3.8% (Te Whatu Ora/Health New Zealand, 2024). The 15-year estimated diabetes hospitalisation risks in this study, which corresponds to approximately this age band (31–39 years old), were 6.1% for Māori and 3.3% for non-Māori. However, as these risk estimates only capture hospitalisation events, they will under-represent the actual rate of diabetes in the study cohort. It would be worthwhile for future research to compare Māori and non-Māori with FEP to the age-equivalent general population to better understand the compounding effect that psychosis and Māori ethnicity may have on physical health.
Inequities in risk of hospitalisation with diabetes and CVD at this relatively young age (at the end of follow-up, the oldest people were 39 years old) will likely contribute to mortality inequities later in life; future research is needed to analyse later mortality and health inequities. Moreover, the hospitalisation measures used in this study will not detect manageable risk factors for cardiometabolic disease (e.g. elevated lipids, HbA1c and blood pressure), which typically occur prior to hospitalisation and are also higher among Māori in the general population (Ministry of Health, 2023). There is a need to measure engagement between the health system and Māori with a diagnosis of psychosis. In particular, it would be useful to examine: primary care access, receipt of routine blood screening (lipids and HbA1c), CVD risk assessment completion, management of pre-diabetes, completion of diabetes annual reviews (for those with diabetes diagnosis), interventions made on abnormal screening results (e.g. medications such as metformin, statins) and monitoring and follow-up of abnormal screening results (e.g. follow-up screening and service contact). Considering these findings and barriers to care reported by Māori experiencing psychosis, this is an urgent research area.
Strengths and limitations
Both PRIMHD and NMDS data were used identify a national cohort from recorded psychosis diagnoses based on standard diagnostic code sets (ICD and DSM). Using two identification sources reduces the probability of missing individuals meeting our inclusion criteria. Furthermore, using these data to rule out previous psychosis admissions increases our confidence that the selected index date for each cohort member is a good approximation of FEP date. It should be noted that National Collections may be subject to biases. For instance, the NHI dataset we used to code ethnicity has been estimated to undercount Māori by 16% (Harris et al., 2022), meaning some Māori will be erroneously classified as non-Māori, which may lead to underestimates of differences. We also recognise that different non-Māori ethnic groups experience varying deprivation/privilege with regard to health, healthcare access and health determinants. This study used Tiriti o Waitangi-aligned comparisons (Māori vs non-Māori); however, it is noted that health inequities for Māori experiencing psychosis would likely be higher if considered against only the dominant NZ European ethnic group.
We recognise that hospital admissions are a reasonably extreme indicator of physical ill health, and it is likely that most ill health in this age group does not result in hospitalisation. Hospitalisation outcomes (e.g. diabetes and CVD admissions) represent a conjunction of physical health needs, access to care and quality of care, rather than representing the prevalence or incidence of these health conditions in the cohort. For instance, some Māori may be considerably debilitated by a physical health condition, but not go to hospital due to barriers (Espiner et al., 2021; Graham and Masters-Awatere, 2020). It is also plausible that Māori may be more likely to present to hospital for a health condition, due to unmet need in primary health care (Harris et al., 2019; Ministry of Health, 2018, 2023). To soundly interpret these findings, it is important to recognise that the additional factors influencing hospitalisation risk (beyond the health condition itself) do systematically vary in these ways between Māori and non-Māori.
Conclusion
Māori have higher risk of hospitalisation and mortality in the 15 years following FEP diagnosis compared to non-Māori. Suicide and accidental injuries/poisonings together caused approximately 80% of deaths with a recorded cause in the 15-year period following FEP. However, diabetes and CVD inequities found at this early life stage will lead to worse physical health and mortality outcomes later in life for Māori experiencing psychosis. Future research is needed to understand the physical health and mortality of Māori experiencing psychosis into middle and later adulthood. These findings warrant consideration of how the health system is working with Māori experiencing psychosis to prevent premature mortality and to prevent, monitor and treat cardiometabolic health conditions.
Supplemental Material
sj-docx-1-anp-10.1177_00048674241270981 – Supplemental material for The physical health and premature mortality of Indigenous Māori following first-episode psychosis diagnosis: A 15-year follow-up study
Supplemental material, sj-docx-1-anp-10.1177_00048674241270981 for The physical health and premature mortality of Indigenous Māori following first-episode psychosis diagnosis: A 15-year follow-up study by Nathan J Monk, Ruth Cunningham, James Stanley, Sue Crengle, Julie Fitzjohn, Melissa Kerdemelidis, Helen Lockett, Andre D McLachlan, Waikaremoana Waitoki and Cameron Lacey in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
We acknowledge the 519 people in the study cohort who passed. We acknowledge the strength of rangtahi Māori and young Pākehā living with psychosis.
Author Contributions
C.L., R.C., J.S. and N.J.M. contributed to the concept and design. N.J.M., R.C. and J.S. obtained and analysed the data. All authors contributed to the interpretation of results. N.J.M. drafted the manuscript; R.C., J.S., C.L., S.C., J.F., M.K., H.L., A.D.M. and M.W. critically reviewed the first draft. N.J.M. revised the manuscript. All authors approved the final submission.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is from Phase 1 of Te Pu Korokoro, funded by the Health Research Council (HRC) of New Zealand (HRC; grant no. 21/558). The HRC had no influence on the study.
Data availability
The data are not publicly available due to ethical compliance regarding de-identified National Health Service records. The data will be destroyed upon publication.
Supplemental material
Supplemental material for this article is available online.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
