Abstract
Objective:
The Australian Genetics of Bipolar Disorder Study is a nation-wide cohort of adults living with bipolar disorder. The study aims to detect the relationships between genetic risk, symptom severity, and the lifetime prevalence of bipolar disorder, treatment response and medication side effects, and patterns and costs of health care usage.
Methods:
A total of 6682 participants (68.3% female; aged 44.8 ± 13.6 years [range = 18–90]) were recruited in three waves: a nation-wide media campaign, a mail-out based on prescriptions for lithium carbonate and through the Australian Genetics of Depression Study. Participants completed a self-report questionnaire. A total of 4706 (70%) participants provided a saliva sample and were genotyped and 5506 (82%) consented to record linkage of their Pharmaceutical and Medicare Benefits Schedule data.
Results:
Most participants were living with bipolar I disorder (n = 4068) while 1622 participants were living with bipolar II disorder and 992 with sub-threshold bipolar disorder. The mean age of bipolar disorder diagnosis was 32.7 ± 11.6 years but was younger in bipolar I (p = 2.0E-26) and females (p = 5.7E-23). Excluding depression with onset prior to bipolar disorder diagnosis, 64.5% of participants reported one or more co-occurring psychiatric disorders: most commonly generalised anxiety disorder (43.5%) and posttraumatic stress disorder (20.7%). Adverse drug reactions were common and resulted in discontinuation rates ranging from 33.4% for lithium to 63.0% for carbamazepine.
Conclusion:
Our findings highlight the high rate of comorbidities and adverse drug reactions among adults living with bipolar disorder in the general Australian population. Future genomic analyses focus on identifying genetic variants influencing pharmacotherapy treatment response and side effects.
Introduction
Bipolar disorder (BD) is a severe mental illness with a remitting/relapsing course that is classified as bipolar I disorder (BD-I) when it is characterised by episodes of mania (which are typically interspersed with major depression) and bipolar II disorder (BD-II) when characterised by episodes of hypomania that alternate with periods of major depression (American Psychiatric Association [APA], 2013).
In 2020–2021, there were approximately 744,800 Australians with lived experience of BD (Australian Bureau of Statistics [ABS], 2022c). The lifetime and 12-month prevalence rates for BD in the 2020–2021 Australian National Survey of Mental Health and Wellbeing were 3.8% and 2.2%, respectively (ABS, 2022c). However, the Australian prevalence rates are higher than those reported in the World Mental Health Survey Initiative, which reported lifetime prevalence rates of BD-I, BD-II and sub-threshold BD from the pooled sample of 11 countries were 0.6%, 0.4% and 1.4%, respectively (Merikangas et al., 2011).
Critically, BD is associated with significant economic burden, mortality and morbidity. In Australia, combined excess health sector and individual costs of BD were estimated to be $3.97–$4.95 billion using data from the 2004 South Australian Health Omnibus Survey, with mean annual costs per person estimated to be $9877 of which $8553 were costs paid by individuals and their families (Fisher et al., 2007).
A meta-analysis by Walker et al. (2015) found that all-cause mortality was elevated among people living with BD and estimated the relative risk of death was 2.00 (95% confidence interval, CI = [1.70, 2.34]). Furthermore, BD was associated with reduced life expectancy; a recent meta-analysis of years of potential life lost (YPLL) reported a weighted average of 12.89 years (95% CI = [12.72, 13.07]) (Chan et al., 2022). Of note, YPLL estimates owing to natural causes was higher in women than men (6.88 vs 5.14) while unnatural causes in men was almost double that of women (5.10 vs 2.68) (Chan et al., 2022). A substantial cause of unnatural deaths in BD was suicide; compared to the general population, the suicide rate is 20- to 30-fold higher among individuals living with BD (Plans et al., 2019).
BD is highly heritable with twin-based studies estimating that genetic factors explain 60–85% of the risk of developing BD (Johansson et al., 2019; Smoller and Finn, 2003). While identification of genetic variants influencing BD and lithium treatment response has progressed, relatively little is known about how genetic risk influences health care usage and response to other medications. To address these questions, we established a nation-wide cohort of 6682 adults living with BD in Australia. The Australian Genetics of Bipolar Disorder (GBP) Study aims to identify genetic risk factors influencing BD, medication treatment response and adverse drug reactions (ADRs); to characterise the patterns and costs of health care usage in BD; and to examine the relationship between genetic risk and symptom severity. This article describes our study design and sample characteristics, and summarises data collected on BD, psychiatric comorbidities and medication use.
Methods
Study design
Participants were recruited using three approaches. The first recruitment wave (n = 1524) was via a nation-wide media publicity campaign managed by a Sydney-based public relations company (VIVA! Communications). The campaign focused on recruiting volunteers from the public who had ever been diagnosed with or treated for BD and comprised a media launch on 20 November 2018 with follow-up via television, radio, print and online interviews and social media posts. Potential participants were directed to the study website (www.geneticsofbipolar.org.au).
The second recruitment wave (n = 3576) used the Services Australia mail-out service which identified prospective participants based on Pharmaceutical Benefits Scheme (PBS) prescription records using a query with the following selection criteria: individuals aged 18–65 years old at the time of mail-out who had two or more dispensed prescriptions for lithium carbonate (PBS Item Codes 3059B and 8290H) within the last 4.5 years. Exclusion criteria were New South Wales residents born before 1960 (due to another BD study being conducted within this State) and Medicare addresses corresponding to health care, correctional or aged-care facilities. Services Australia sent letters describing the study to 50,000 individuals on 29 March 2019. The letter explained that the recipient was being contacted on behalf of researchers at QIMR Berghofer Medical Research Institute (QIMR Berghofer) to invite them to participate in a study on the genetics of BD and that their personal details had not been released to the researchers. The letter also described the purpose of the study and asked willing individuals to visit the study website.
Individuals recruited in the first two waves were asked to confirm that they had read and understood the Participant Information Sheet and then asked to complete the online Consent Form to enrol in the study and to indicate whether they would provide a saliva sample so that DNA could be extracted and genotyped. Participants then entered their details onto an online form securely hosted on QIMR Berghofer servers and were assigned a unique link to an online questionnaire hosted on the Qualtrics website.
The third recruitment wave (n = 1582) started on 27 April 2020 as part of a follow-up survey of the Australian Genetics of Depression Study (AGDS; Byrne et al., 2020). The AGDS cohort (n = 23,814) was recruited between 2017 and 2020; a total of 2059 participants self-reported a BD diagnosis and 1457 reported that they had taken lithium carbonate in their lifetime. In the follow-up survey, AGDS participants were presented with the content from the core module of the GBP Study survey.
Ethics
Participants who completed the online survey (n = 6580) provided written informed consent for the study online. Participants who completed an abridged paper-based version of the survey (n = 102) completed a paper consent form. Ethics approval for all aspects of the project was obtained from the QIMR Berghofer Human Research Ethics Committee for the GBP (P3408) and AGDS (P2118) studies. The External Request Evaluation Committee of Services Australia approved the study mail-out and the consenting process for linkage to participant Medicare Benefits Schedule (MBS) and PBS data (EREC reference number MI10846).
Online survey
The online survey was designed to overlap with content of the AGDS baseline survey. A total of 6580 participants completed a core module which included demographic and medication questions, self-report mental health diagnostic history, BD diagnosis history, a BD screen using the Mood Disorder Questionnaire (MDQ; Hirschfeld et al., 2000) and additional items corresponding to Diagnostic and Statistical Manual of Mental Disorders BD diagnostic criteria (5th ed.; DSM-5; APA, 2013).
Participants indicated which medications they had ever taken from a list of mood stabilisers, antipsychotics and antidepressants. These included mood stabilisers: lithium, carbamazepine, lamotrigine, valproate, topiramate; antipsychotics: asenapine, clozapine, lurasidone, ziprasidone, amisulpride, aripiprazole, olanzapine, paliperidone, quetiapine, risperidone, haloperidol, Fluanxol Depot [or Clopixol Depot], Largactil [or Modecate, or Stelazine or Neulactil]; and antidepressants: selective serotonin reuptake inhibitors (SSRIs; sertraline, citalopram, escitalopram, paroxitine, fluoxetine), serotonin-norepinephrine reuptake inhibitors (SNRIs; venlafaxine, desvenlafaxine, duloxetine) and two other antidepressant medications (amitriptyline, mirtazapine). Data on current use and the presence of ADRs associated with each mood stabiliser and four antipsychotics (asenapine, clozapine, lurasidone, ziprasidone) were collected.
The MDQ is a self-administered BD screening instrument comprising three parts; 13 yes/no response items reflecting Diagnostic and Statistical Manual of Mental Disorders (4th ed.; DSM-IV) criteria (APA, 1994) and clinical experiences to identify a lifetime history of hypomanic or manic episodes; a question assessing whether symptoms were experienced in the same period (yes, no); and a 4-point scale question (no problem, minor problem, moderate problem, serious problem) assessing the level of functional impairment from symptoms. A positive screen was defined as endorsement of seven or more items in part 1, that the symptoms occurred simultaneously and caused at least moderate impairment (Hirschfeld et al., 2000). A 2015 meta-analysis of 21 studies reported a screening sensitivity of 0.62 and specificity of 0.85 for the detection of BD (Wang et al., 2015).
Other items included in the online survey to determine BD diagnosis assessed: the duration of the longest manic or hypomanic episode (less than 4 days, less than a week [4–7 days], less than a fortnight [8–14 days], less than a month [15–30 days], more than a month [30+ days]); whether the respondent felt this way most, if not all, of the day (yes, no); whether the respondent was hospitalised during one of those periods (yes, no) and if they answered yes, the number of hospitalisations and longest time they were hospitalised; whether the respondent thought their episodes ever occurred as the result of physical causes (yes, no), and items corresponding to DSM-5 diagnostic criteria for major depressive disorder (MDD). The core module also included screens for suicidality, anxiety and phobias and a checklist of six psychotic-like experiences (PLEs).
Participants were also invited to complete two additional modules (completed by 3103 and 2553 participants, respectively) containing questions relating to general physical health and health care, work and sleep, life events, migraines, substance use and gambling. Figure 1 provides an overview of the GBP Study, and Supplemental Tables S1 and S2 summarise the instruments included and data collected in the survey.
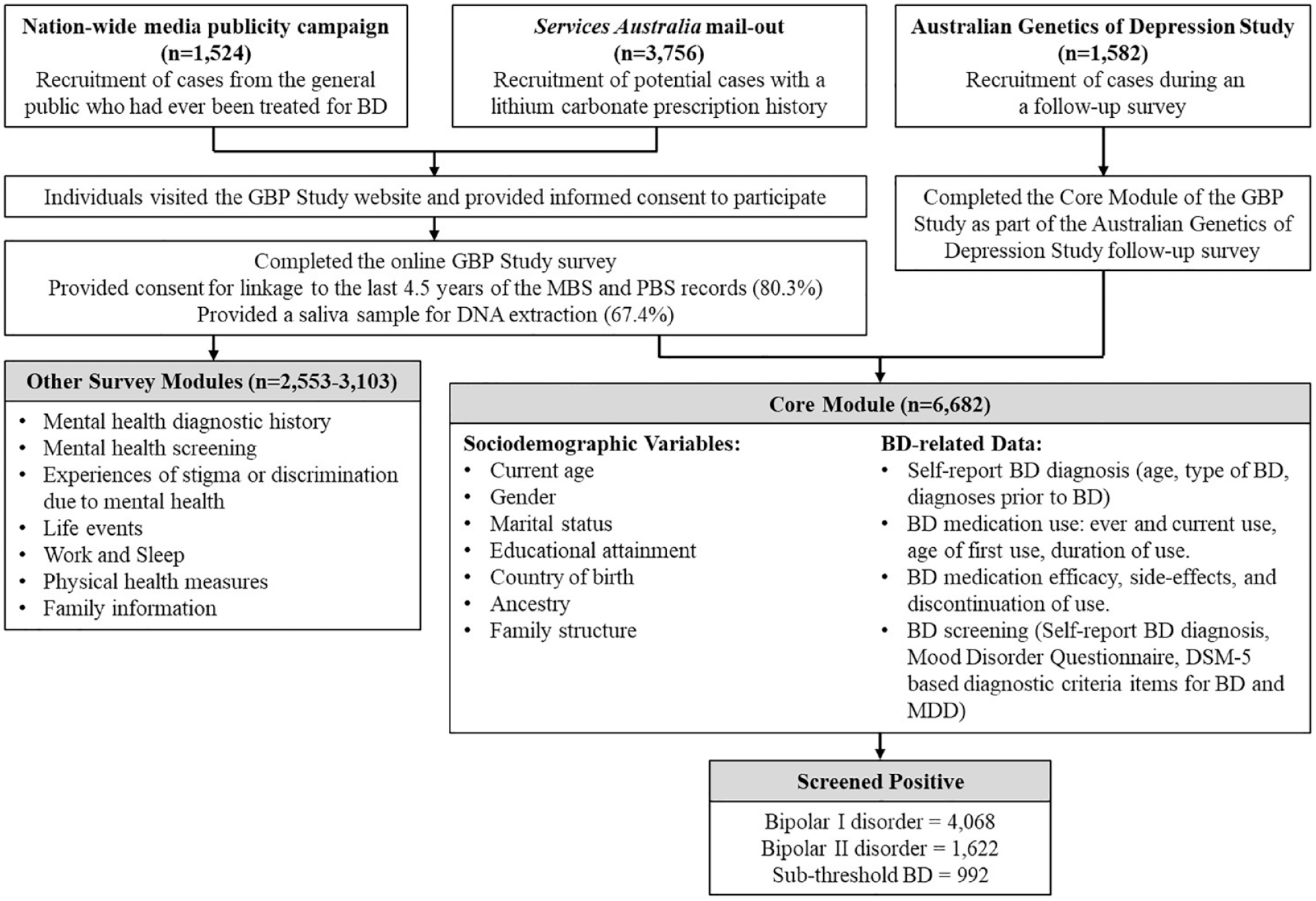
Overview of the Australian Genetics of Bipolar Disorder Study recruitment strategy and data collected.
Record linkage
Participants were asked to consent to allow the study to access 5 years (from 30 June 2014 to 30 June 2019) of their PBS and MBS records. PBS data contain records of pharmacy transactions for all drugs listed on the PBS schedule and dispensed to eligible Medicare card holders. MBS data contain records of health services, diagnostic procedures and tests provided outside of hospitals. Neither PBS nor MBS records contain diagnosis or indication information. Consent for PBS and MBS data linkage was provided by 80.3% of Wave 1 and 2 participants and previously for 89.3% of AGDS (Wave 3) participants. In January 2023, PBS and MBS data was obtained for 3978 Wave 1 and 2 participants.
Saliva collection and genotyping
Isohelix GeneFix GFX-02 2 mL Saliva Collection Devices were mailed to Wave 1 and 2 participants who were willing to provide a saliva sample; saliva samples were returned to QIMR Berghofer via prepaid Australia Post. Following DNA extraction, genotyping was conducted using the Illumina Global Screening Array V.2.0 and is available for 3437 (67.4%) Wave 1 and 2 participants and 1269 (80.2%) Wave 3 (AGDS) participants. Overall, there are 3020 genotyped participants living with BD-I, 1069 with BD-II and 617 who did not meet the full criteria for BD (sub-threshold BD).
Statistical analyses
Statistical analyses were conducted using IBM SPSS Statistics for Windows, version 23.0 (IBM Corp., Armonk, NY, USA). Significance of differential responses between participants was assessed using chi-square tests, Student t-tests and one-way analysis of variance tests.
Results
Sample description
As of 11 February 2021, 6682 participants had completed the survey. Participants were aged 18–90 years old (on average 44.7 ± 13.7 years old) and predominantly female (68.2%). When answering the question ‘Are you male or female? Note: This question refers to biological sex, not gender’, 21 participants chose the option ‘Unspecified’ (n = 13) or ‘Prefer not to answer’ (n = 8). To reduce potential identifiability of these participants, their data were not aggregated in Table 1.
Basic demographics and bipolar disorder history of participants by gender.
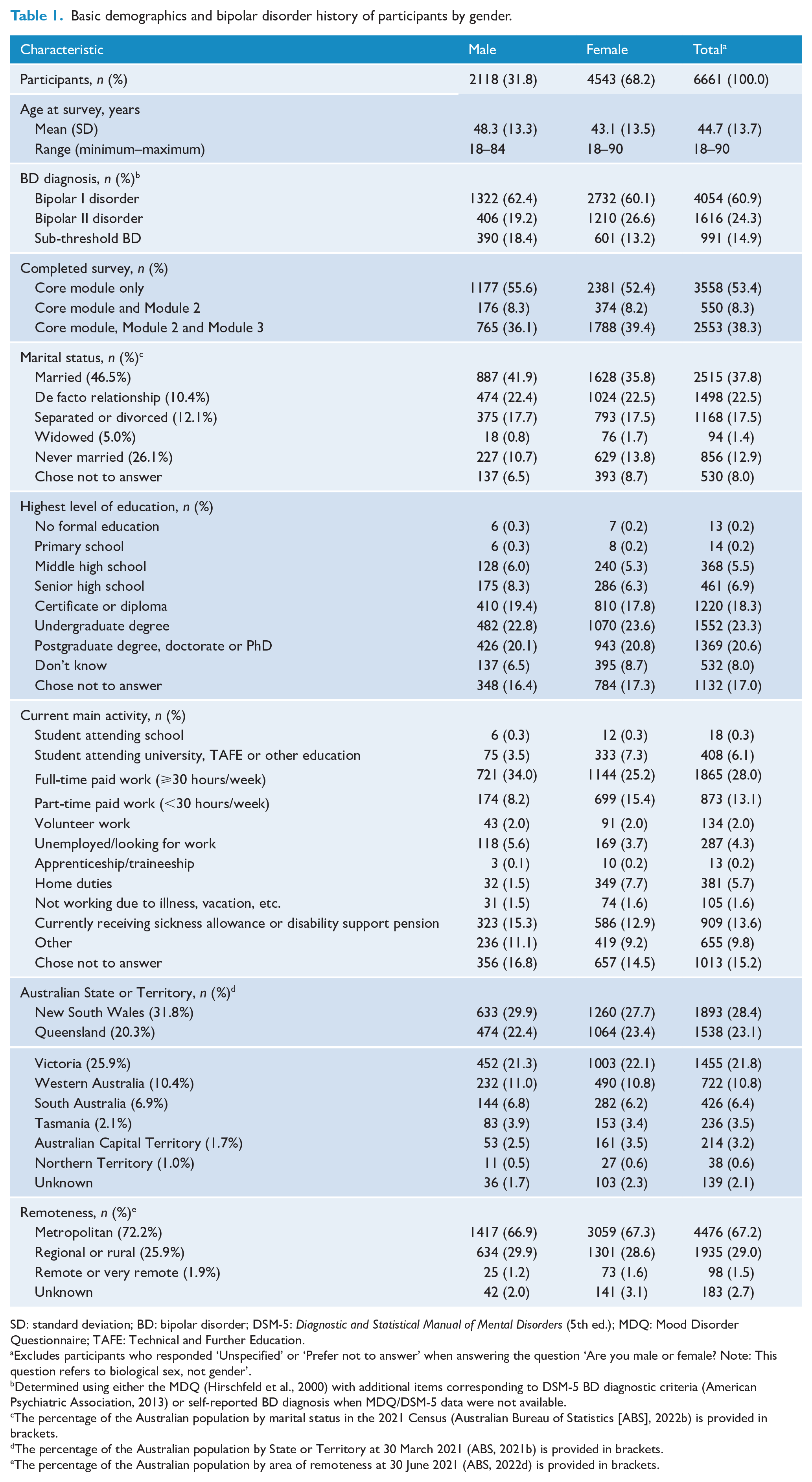
SD: standard deviation; BD: bipolar disorder; DSM-5: Diagnostic and Statistical Manual of Mental Disorders (5th ed.); MDQ: Mood Disorder Questionnaire; TAFE: Technical and Further Education.
Excludes participants who responded ‘Unspecified’ or ‘Prefer not to answer’ when answering the question ‘Are you male or female? Note: This question refers to biological sex, not gender’.
Determined using either the MDQ (Hirschfeld et al., 2000) with additional items corresponding to DSM-5 BD diagnostic criteria (American Psychiatric Association, 2013) or self-reported BD diagnosis when MDQ/DSM-5 data were not available.
The percentage of the Australian population by marital status in the 2021 Census (Australian Bureau of Statistics [ABS], 2022b) is provided in brackets.
The percentage of the Australian population by State or Territory at 30 March 2021 (ABS, 2021b) is provided in brackets.
The percentage of the Australian population by area of remoteness at 30 June 2021 (ABS, 2022d) is provided in brackets.
Sixty percent of participants were married or in a de facto relationship. More than 40% were in paid employment, 6.4% were studying, 13.6% were receiving sickness allowance or disability support pension, and 4.3% were unemployed or looking for work. Employment rates were lower compared to Australians aged 15–74 in May 2022 where 47.2% were employed full time, 22.3% part time and 1.6% as apprentices or trainees (ABS, 2021a). The ethnicity/ethnicities among 5851 participants who provided data were 93.0% Caucasian, 3.3% Australian Aboriginal and/or Torres Strait Islander peoples, 2.9% Eastern or South Eastern Asian, 1.6% Māori or Pacific Islander, 1.6% South Asian ethnicity, 1.4% Middle Eastern, 1.1% African and 0.5% American First Nations, Native American, Inuit or Métis descent. Compared to the Australian population (ABS, 2022a), participants were more likely to be of Caucasian ethnicity (93.0% vs 80.4%) and less likely to be of Asian ethnicity (4.5% vs 17.4%). At the time of survey, 29.0% of participants were living in regional or rural communities and 1.5% in remote or very remote communities based on the 2019 Modified Monash Model (Australian Government Department of Health, 2021). Importantly, the Services Australia mail-out enabled the recruitment of a higher proportion of participants from these communities (1.8%) than the media campaign (1.0%). Educational attainment levels among participants who responded (74.9% with a post-school qualification) were higher than that observed in May 2021 (62% of Australians aged 15-74 years) (ABS, 2021a), indicating that people with higher education levels were more likely to participate in the study.
Data linkage and genotyping
Consenting to PBS and MBS record linkage was significantly associated with participant age (p = 4.3E-08), BD diagnosis (p = 2.7E-04) and age of diagnosis (p = 1.0E-06) whereby consenting participants were older on average (45.1 vs 42.7 years old), more likely to be diagnosed with BD-I and have later diagnoses (Supplemental Table S3). Consenting participants were also more likely to be male (p = 9.1E-08), married (p = 2.0E-05) and to have provided a saliva sample for genotyping (p = 2.0E-15). A similar pattern of association was also observed for participant consent for providing a saliva sample; however, no association with sex was evident (Supplemental Table S4).
Bipolar disorder diagnosis
MDQ and DSM-5 BD data were used to determine BD-I and BD-II diagnoses among 3747 and 976 participants, respectively (Table 2). For 967 participants with missing MDQ and DSM-5 BD data, their self-reported BD-I (n = 321) or BD-II (n = 646) diagnosis was used. Finally, almost 15% of participants (n = 992) self-reported a diagnosis of BD but did not meet the DSM-5 criteria for BD, and we refer to these participants as living with sub-threshold BD. Participants living with sub-threshold BD self-reported experiencing, on average, 1.3 ± 2.0 DSM-5 BD symptoms (range = 0–7). Overall, the GBP cohort includes 4068 participants living with BD-I, 1622 with BD-II and 992 with sub-threshold BD. BD diagnoses differed significantly between men and women (p = 7.2E-14), with women more likely (26.6% vs 19.2%) diagnosed with BD-II.
Participant diagnosis history and impact of bipolar disorder reported by type of bipolar disorder.
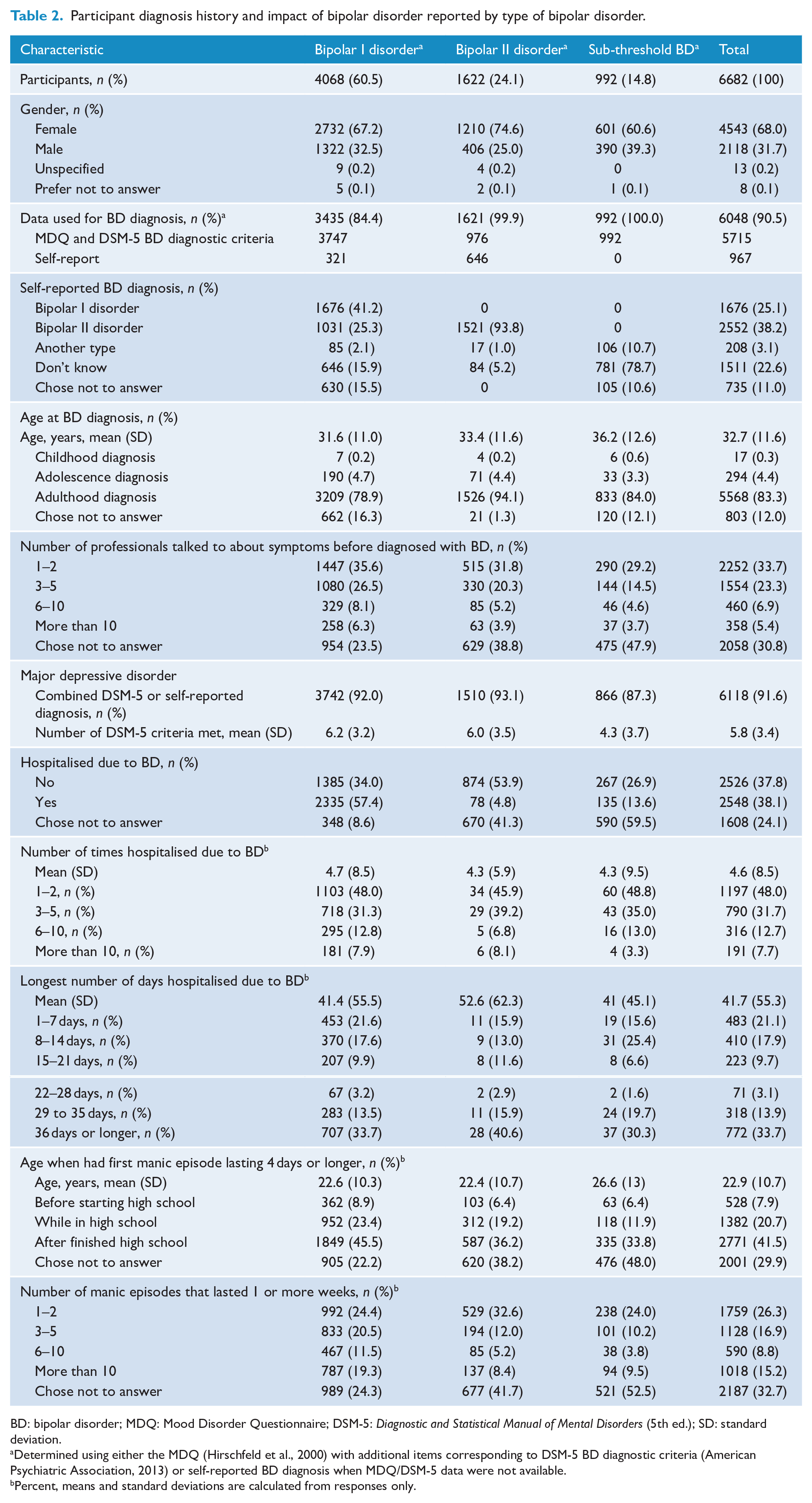
BD: bipolar disorder; MDQ: Mood Disorder Questionnaire; DSM-5: Diagnostic and Statistical Manual of Mental Disorders (5th ed.); SD: standard deviation.
Determined using either the MDQ (Hirschfeld et al., 2000) with additional items corresponding to DSM-5 BD diagnostic criteria (American Psychiatric Association, 2013) or self-reported BD diagnosis when MDQ/DSM-5 data were not available.
Percent, means and standard deviations are calculated from responses only.
Age of diagnosis
Self-reported age at BD diagnosis significantly differed between BD subtypes, F(2, 5861) = 59.8, p = 1.9E-26. Participants living with BD-I reported earlier diagnoses (31.6 ± 11.0 years old) compared to participants living with BD-II (33.4 ± 11.6) and sub-threshold BD (36.2 ± 12.6).
A bimodal distribution of age of BD diagnosis was observed particularly among male participants living with BD-I or BD-II (Figure 2). Compared to males living with BD-I or BD-II, females reported significantly lower, t(4976) = 8.668, p = 5.9E-18, age of diagnosis (31.3 vs 34.3 years old) and the odds that female participants were diagnosed with BD before the age of 20 were 1.6 times that of males (odds ratio [OR]= 1.64, 95% CI = [1.33, 2.02], p = 3.0E-06). More than one-third (34.3%) of participants living with BD-I or BD-II reported speaking to 3–5 professionals about their symptoms before being diagnosed with BD while 17.8% spoke to six or more.
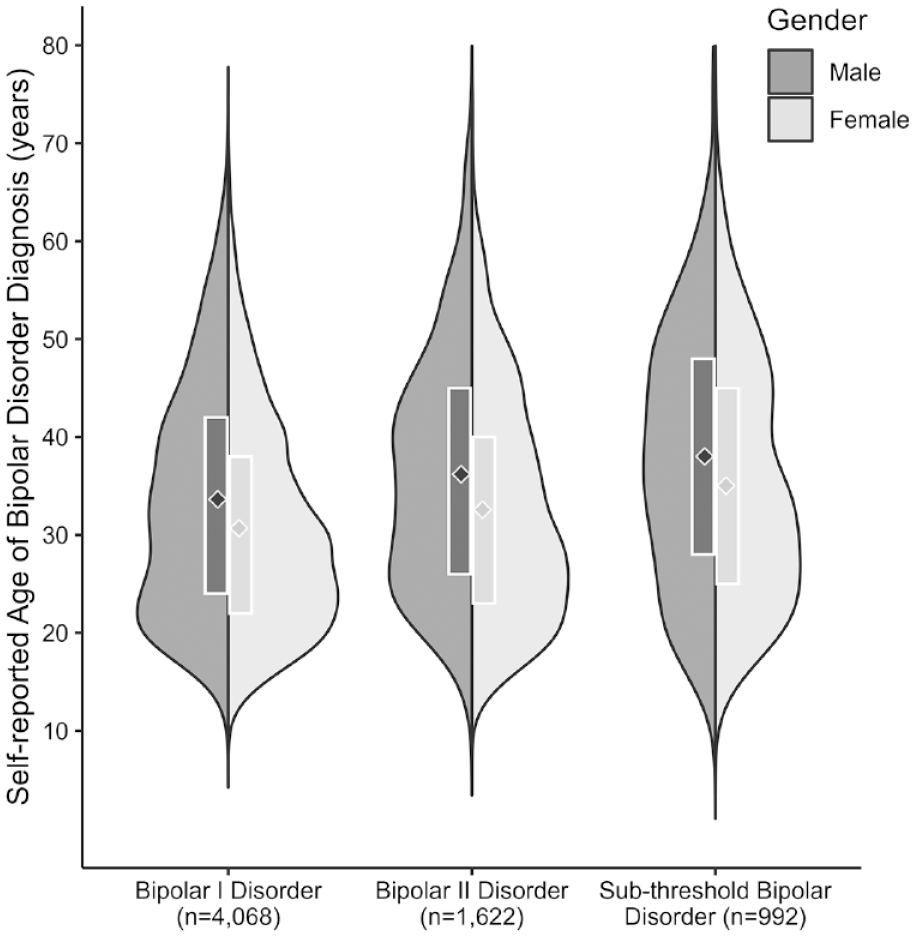
Split violin plots showing the distribution of self-reported age of diagnosis by sex and type of bipolar disorder. The diamonds depict the mean ages of bipolar disorder diagnosis and the white boxes represent the interquartile range.
Manic episodes, hospitalisations and depression
Among the 4681 participants who reported on their first manic episode lasting 4 days or longer, 11.3% had not started high school and 29.5% were in high school. The average age of first manic episode was 22 years old. The majority of the 2548 participants who reported being hospitalised due to BD were living with BD-I. The average number of hospitalisations was 4.6 ± 8.5; 48% of participants were hospitalised once or twice and 27.7% reported five or more hospitalisations. Most participants (91.6%) self-reported a diagnosis of depression or met DSM-5 criteria for MDD. Overall, the average number of diagnostic criteria met was significantly higher, F(1, 6680) = 246.9, p = 1.2E-54, among participants living with BD-I or BD-II (6.1 ± 3.3) than sub-threshold BD (4.3 ± 3.7).
Mental health comorbidity
Overall, 39.6% of participants reported a first-degree relative with any mood or anxiety disorder. Approximately 16% of participants reported a first-degree relative with a lifetime history of BD, 32.5% with depression and 17.7% with generalised anxiety disorder (GAD) (Table 3).
Medical and psychiatric comorbidities of participants reported by type of bipolar disorder.
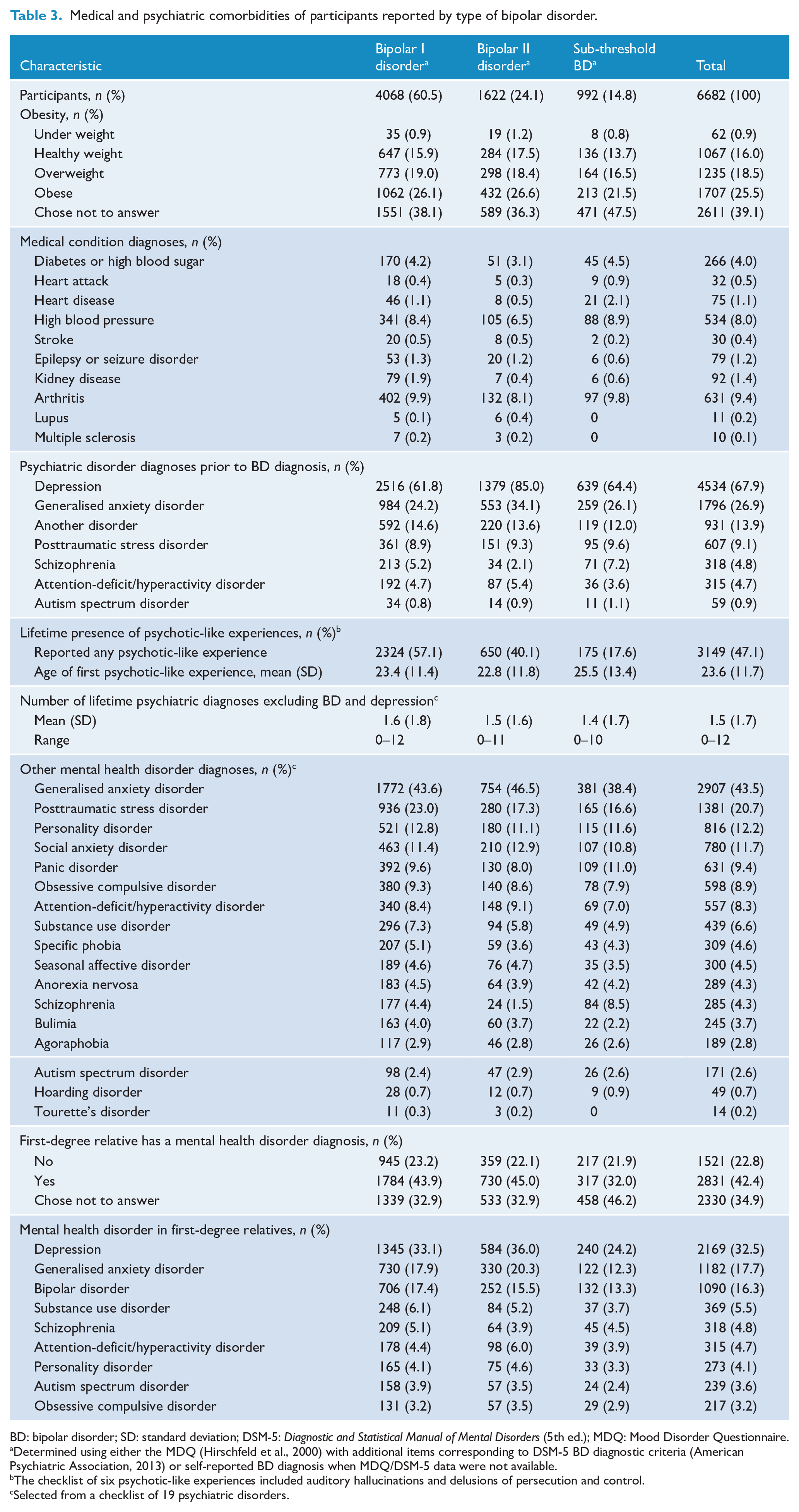
BD: bipolar disorder; SD: standard deviation; DSM-5: Diagnostic and Statistical Manual of Mental Disorders (5th ed.); MDQ: Mood Disorder Questionnaire.
Determined using either the MDQ (Hirschfeld et al., 2000) with additional items corresponding to DSM-5 BD diagnostic criteria (American Psychiatric Association, 2013) or self-reported BD diagnosis when MDQ/DSM-5 data were not available.
The checklist of six psychotic-like experiences included auditory hallucinations and delusions of persecution and control.
Selected from a checklist of 19 psychiatric disorders.
Almost two-thirds of participants reported being diagnosed with depression prior to BD. The next most common diagnoses were GAD (26.9%) and posttraumatic stress disorder (PTSD; 9.1%). Excluding depression, 65.1% of participants reported one or more co-occurring psychiatric disorders.
Following multiple testing correction (p < 0.05/18), type of BD diagnosis was significantly associated with lifetime diagnoses of PTSD, χ2(2) =34.9, p = 2.6E-08, and GAD, χ2(2) = 16.4, p = 2.8E-04.
Compared to females, males were more likely to report diagnoses of schizophrenia (OR = 1.8, 95% CI = [1.4, 2.3], p = 2.0E-06) and attention-deficit/hyperactivity disorder (ADHD) (OR = 1.4, 95% CI = [1.2, 1.7], p = 4.6E-04). However, males were less likely to report diagnoses of anorexia (OR = 0.09, 95% CI = [0.05, 0.16], p = 2.1E-16), bulimia (OR = 0.11, 95% CI = [0.06, 0.19], p = 3.7E-14), personality disorders (OR = 0.43, 95% CI = [0.36, 0.52], p = 1.7E-18), agoraphobia (OR = 0.48, 95% CI = [0.33, 0.69], p = 9.9E-05), specific phobia (OR = 0.48, 95% CI = [0.36, 0.65], p = 9.4E-07), PTSD (OR = 0.49, 95% CI = [0.43, 0.57], p = 9.4E-23), GAD (OR = 0.53, 95% CI = [0.48, 0.59], p = 1.3E-30), panic disorder (OR = 0.60, 95% CI = [0.49, 0.73], p = 2.3E-07) and obsessive compulsive disorder (OCD; OR = 0.60, 95% CI = [0.50, 0.74], p = 7.2E-07).
Participants who received a BD diagnosis before the age of 20 were more likely to report a lifetime history of ADHD (OR = 2.1, 95% CI = [1.6, 2.7], p = 5.0E-09), autism spectrum disorder (ASD) (OR = 3.0, 95% CI = [2.0, 4.5], p = 1.4E-07), anorexia (OR = 2.1, 95% CI = [1.5, 3.0], p = 4.1E-05) and GAD (OR = 1.4, 95% CI = [1.1, 1.6], p = 0.001) compared to those diagnosed aged 20 years or older.
Finally, 3149 (47.1%) participants reported one or more PLE (Table 3). The prevalence of PLEs differed significantly between BD diagnoses, χ2(2) =234.7, p = 1.1E-51, with highest rates observed among participants living with BD-I. Of note, 3.5% of participants living with BD-I or BD-II also self-reported a diagnosis of schizophrenia, suggesting a diagnosis of schizoaffective disorder rather than BD. Compared to participants without a diagnosis of schizophrenia, these individuals were more likely to have reported PLEs (OR = 3.3, 95% CI = [2.3, 4.6], p = 3.9E-12).
Medication usage and ADRs
A total of 5774 (86.4%) participants reported ever taking mood stabilisers or antipsychotics (Table 4) and 4506 participants reported current use of mood stabilisers or antipsychotics. The most commonly prescribed class of listed medications was mood stabilisers, taken by 72.9% of participants. Approximately 54% of participants had ever taken a listed antipsychotic medication, with quetiapine (34.8%) and olanzapine (20.5%) taken most frequently. Of the 908 participants who reported never taking a listed mood stabiliser or antipsychotic, almost 90% had been prescribed one or more antidepressant medication, most commonly sertraline.
Medication use and adverse drug reactions reported by type of bipolar disorder.
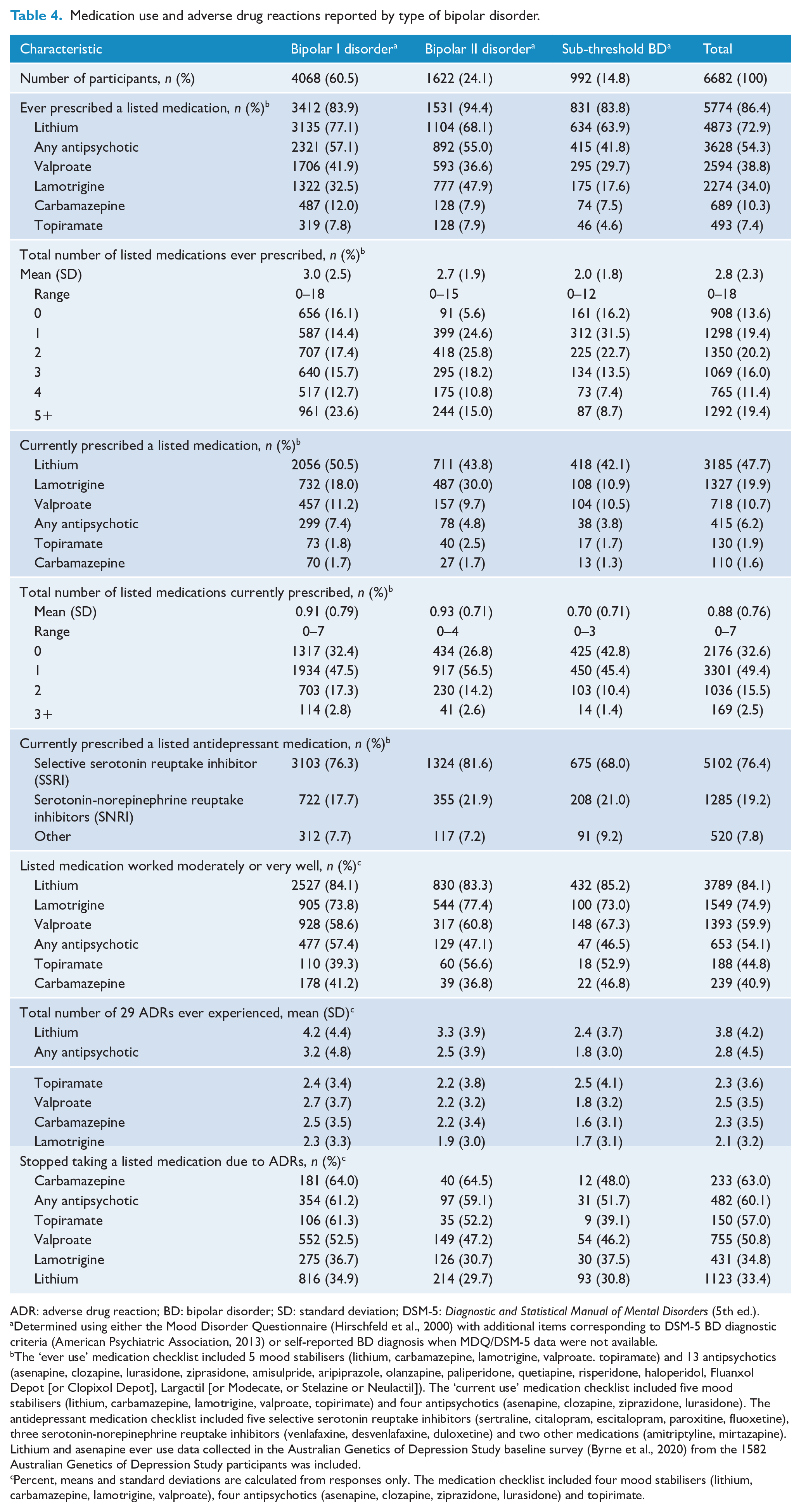
ADR: adverse drug reaction; BD: bipolar disorder; SD: standard deviation; DSM-5: Diagnostic and Statistical Manual of Mental Disorders (5th ed.).
Determined using either the Mood Disorder Questionnaire (Hirschfeld et al., 2000) with additional items corresponding to DSM-5 BD diagnostic criteria (American Psychiatric Association, 2013) or self-reported BD diagnosis when MDQ/DSM-5 data were not available.
The ‘ever use’ medication checklist included 5 mood stabilisers (lithium, carbamazepine, lamotrigine, valproate. topiramate) and 13 antipsychotics (asenapine, clozapine, lurasidone, ziprasidone, amisulpride, aripiprazole, olanzapine, paliperidone, quetiapine, risperidone, haloperidol, Fluanxol Depot [or Clopixol Depot], Largactil [or Modecate, or Stelazine or Neulactil]). The ‘current use’ medication checklist included five mood stabilisers (lithium, carbamazepine, lamotrigine, valproate, topirimate) and four antipsychotics (asenapine, clozapine, ziprazidone, lurasidone). The antidepressant medication checklist included five selective serotonin reuptake inhibitors (sertraline, citalopram, escitalopram, paroxitine, fluoxetine), three serotonin-norepinephrine reuptake inhibitors (venlafaxine, desvenlafaxine, duloxetine) and two other medications (amitriptyline, mirtazapine). Lithium and asenapine ever use data collected in the Australian Genetics of Depression Study baseline survey (Byrne et al., 2020) from the 1582 Australian Genetics of Depression Study participants was included.
Percent, means and standard deviations are calculated from responses only. The medication checklist included four mood stabilisers (lithium, carbamazepine, lamotrigine, valproate), four antipsychotics (asenapine, clozapine, ziprazidone, lurasidone) and topirimate.
Participants reported experiences of 29 ADRs for each of the listed mood stabilisers and antipsychotics they had ever taken (Figure 3). Participants living with BD-I reported ever experiencing significantly, F(2, 5613) = 108.9, p = 4.0E-47, more ADRs (5.4 ± 5.3) than participants living with BD-II (3.9 ± 4.4) and sub-threshold BD (2.8 ± 4.2). Overall, the five most experienced ADRs were weight gain (42.1%), dry mouth (38.5%), shaking or tremor (36.6%), thirst (31.9%) and feeling mentally slow or fuzzy headed (31.4%). Critically, 5.4% of participants reported attempting suicide as an ADR (7.0% BD-I, 3.2% BD-II and 2.7% sub-threshold BD participants) while 10.9% of participants reported experiencing suicidal thoughts as an ADR (12.7% BD-I, 8.7% BD-II and 6.9% sub-threshold BD participants). Overall, 608 (14.9%) BD-I, 213 (13.1%) BD-II and 87 (8.8%) sub-threshold BD participants reported that the worst aspect of taking their mood stabilisers or antidepressants was increased suicidal thoughts or actions.
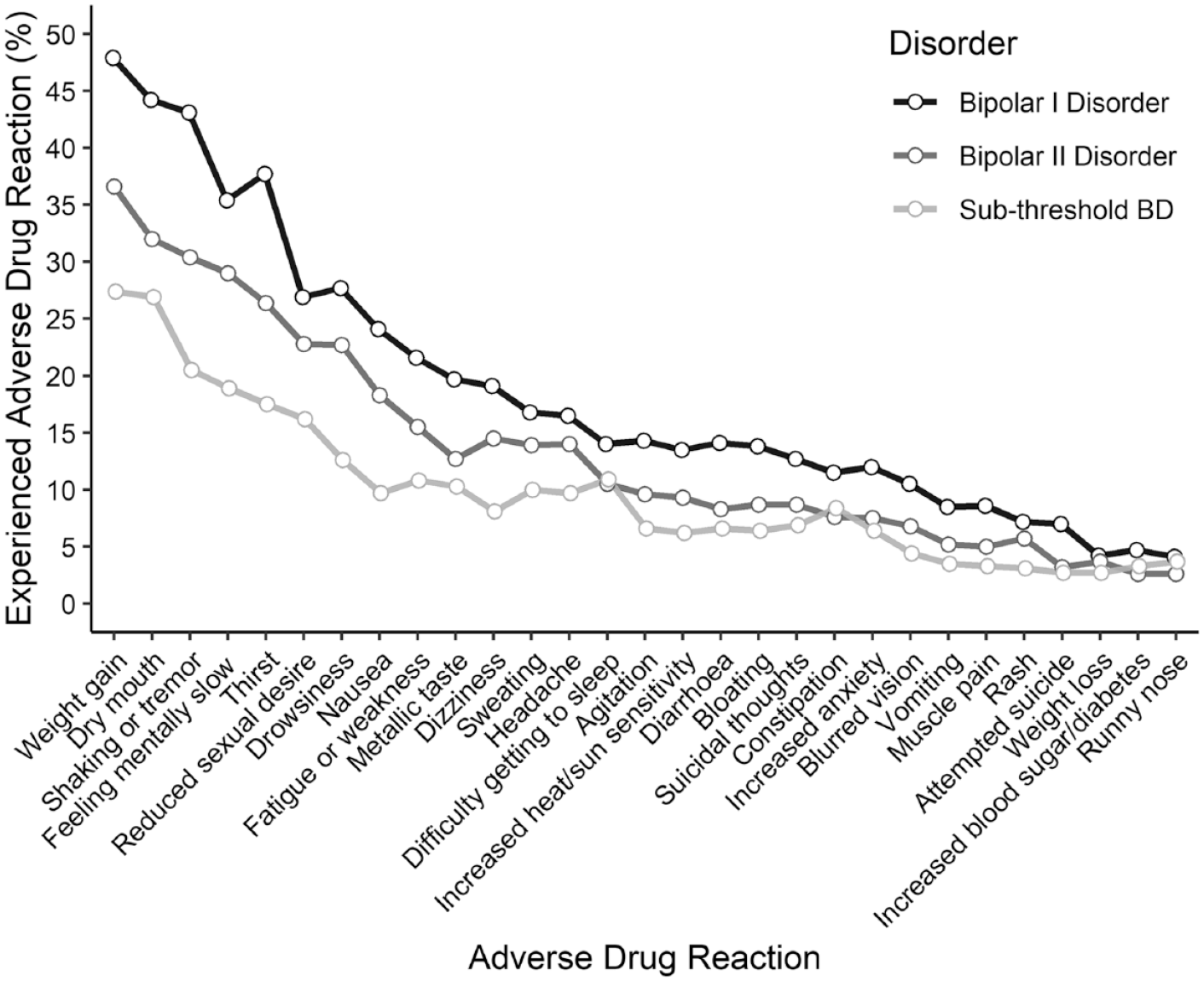
Frequency of reported adverse drug reactions (ADRs) by type of bipolar disorder.
While lithium was associated with the highest number of ADRs (3.8 ± 4.2), 84.1% of users perceived that lithium worked moderately or very well and only one-third of users stopped taking lithium due to the ADRs. For the other medications, participants experienced an average of 2.1–2.8 ADRs. The highest rates of medication discontinuation due to ADRs were reported for carbamazepine and use of any antipsychotic.
Discussion
The mean age of BD diagnosis among GBP participants was 32.7 years, and 68.5% of participants living with BD-I and BD-II were diagnosed with depression prior to their first hypomanic or manic episode. More than half (51.3%) of the participants reported speaking to three or more professionals about their symptoms before diagnosis. This is consistent with research reporting a large time interval of 5–10 years between the onset of symptoms and the diagnosis and treatment of BD (Dagani et al., 2017; Drancourt et al., 2013; Keramatian et al., 2022; Medici et al., 2015).
Overall, 42% of participants reported a first-degree relative had been diagnosed with one or more mental health disorders. Approximately 16% of participants reported a family history of BD reflecting the moderate to high heritability of BD (Johansson et al., 2019; Smoller and Finn, 2003). Excluding depression, almost two-thirds of participants reported one or more diagnoses of another psychiatric disorder with GAD (20.5%) or PTSD (18.9%) the most common. Overall, 62.7% of participants reported a diagnosis of any anxiety disorder. This level of psychiatric comorbidity is consistent with that found in the World Mental Health Survey Initiative where three-quarters of participants living with BD met the criteria for one or more other disorders (Merikangas et al., 2011). While the rate of anxiety disorders in our cohort was similar to that observed among participants living with BD (52.0%) in the 1997 Australian National Survey of Mental Health and Well-Being (Mitchell et al., 2004), our rates of GAD and PTSD were approximately double those reported in that study.
Higher lifetime rates of PLEs among participants living with BD-I (57.1%) compared to those living with BD-II (40.1%) have been previously observed in the Jorvi Bipolar Study where almost half of the 191 BD patients reported psychotic symptoms and the rate was twice as common among patients living with BD-I (67.8%) than BD-II (33.7%) (Mantere et al., 2004). Participants self-reported being hospitalised due to their BD an average of 4.6 times in their lifetime. Mitchell et al. (2013) estimated that 11.5% and 7.6% of Australians living with BD and sub-threshold BD are hospitalised at least once per year, respectively. Recurrent hospitalisations (1.6 in an 8-year period with a mean duration of 19.5 days) were observed among BD patients in a nation-wide study of Portuguese public hospitals (Gonçalves-Pinho et al., 2022). Participants reported an average length of stay of 41.7 days for their longest hospitalisation which is similar to the average 41.2-day length of stay for admissions in 2006–2010 to English hospitals by patients with a primary diagnosis of BD or a manic episode (Jacobs et al., 2015).
Overall, 46.8% of participants reported having taken 3 or more of the 18 BD medications listed. While most participants endorsed experiencing considerable benefits from their medications, ADRs were common and resulted in medication discontinuation rates ranging from 33.4% for lithium to 63.0% for carbamazepine. Of note, ADRs are a frequent reason reported for non-adherence of mood stabilisers by 20–40% of BD patients (Mago et al., 2014). Medication discontinuation due to ADRs is an important clinical issue because it substantially contributes to poorer outcomes such as manic episode recurrences and increased risk of hospitalisations and suicide (Pompili et al., 2009).
While this study did not focus on genomic analyses, we plan to examine the genetic architecture underlying individual differences in pharmacotherapy treatment response and emergence of ADRs, and how these genetic risk factors may differ between BD subtypes and comorbidity profiles. Future analyses will also examine the relationship between genetic risk and symptom severity and health care utilisation.
A major strength of the study is its sample size; it is the largest population-based study of adults living with BD in Australia. Importantly, we were able to recruit participants from across Australia, including remote communities. Another strength is that 80.3% of Wave 1 and 2 participants consented to link the questionnaire data with their PBS and MBS records. For example, self-report medication use and discontinuation data during the 5-year window can be validated using PBS records.
Study limitations include the sampling frame for our medication-based recruitment strategy which only included adults with two or more dispensed prescriptions of lithium carbonate. This may have resulted in patterns of medication treatment that do not generalise to the majority of Australians living with BD. The relatively low participation rate (7.2%) from the 50,000 adults sent study invitation letters also means that selection bias cannot be excluded, and this is likely reflected in the higher levels of educational achievement in our cohort as compared to the general population.
Conclusion
Our study demonstrates that Australians with lived experience of BD are motivated to participate in genetically focused research studies if given the opportunity and provided with avenues to participate. Importantly, future analyses of PBS and MBS data will allow us to clarify the relationship between BD subtypes, pharmacotherapy treatment response, cost of illness and health service usage within an Australian context. Finally, by collecting phenotypically rich survey, genotypic and health care utilisation data, the GBP Study is an important resource that will facilitate future phenotypic and pharmacogenetic studies.
Supplemental Material
sj-docx-1-anp-10.1177_00048674231195571 – Supplemental material for Preliminary results from the Australian Genetics of Bipolar Disorder Study: A nation-wide cohort
Supplemental material, sj-docx-1-anp-10.1177_00048674231195571 for Preliminary results from the Australian Genetics of Bipolar Disorder Study: A nation-wide cohort by Penelope A Lind, Dan J Siskind, Ian B Hickie, Lucía Colodro-Conde, Simone Cross, Richard Parker, Nicholas G Martin and Sarah E Medland in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
We thank the participants for giving their time and support for this project. We acknowledge and thank M. Steffens for her generous donations and fundraising support.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: I.B.H. is the Co-Director of Health and Policy at the Brain and Mind Centre (BMC) University of Sydney. The BMC operates an early-intervention youth services at Camperdown under contract to Headspace. He is the Chief Scientific Advisor to, and a 3.2% equity shareholder in, InnoWell Pty Ltd. InnoWell was formed by the University of Sydney (45% equity) and PwC (Australia; 45% equity) to deliver the $30 M Australian Government-funded Project Synergy (2017–2020; a 3-year programme for the transformation of mental health services) and to lead transformation of mental health services internationally through the use of innovative technologies. The authors P.A.L., D.J.S., L.C.-C., S.C., R.P., N.G.M. and S.E.M. declare that there are no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Data collection was funded and data analysis was supported by the Australian National Health and Medical Research Council (No. APP1138514) to S.E.M. D.J.S. is supported by a National Health and Medical Research Council Investigator Grant (No. APP1194635). N.G.M. is supported by a National Health and Medical Research Council Investigator Grant (No. APP 1172990). S.E.M. is supported by a National Health and Medical Research Council Investigator Grant (No. APP1172917).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
