Abstract
Objective:
Alcohol use disorder may very well increase the likelihood of affective episodes in bipolar disorder, but prospective data on survival are inconsistent.
Method:
The authors examined the prevalence of alcohol use disorders and their impact on the risk of relapse. A total of 284 consecutively admitted International Classification of Diseases-10 bipolar I (n = 161) and II (n = 123) patients were followed up naturalistically over a period of 4 years.
Results:
The prevalence of alcohol use disorders was higher in bipolar II disorder than in bipolar I disorder (26.8% vs 14.9%; χ2 = 5.46, p = 0.019), with a global prevalence of alcohol use disorders of 20.1% in the whole sample. A total of 8.7% of bipolar I patients suffered from alcohol abuse and 6.2% from alcohol dependency, whereas 13% bipolar II patients had alcohol abuse and 13.8% alcohol dependency. Male bipolar subjects had a higher prevalence of alcohol use disorders than female patients (38.3% vs 12.8%; χ2 = 21.84, p-value < 0.001). The presence of alcohol use disorders was associated with an increased risk of depressive relapse in bipolar I patients (Cox regression analysis hazard ratio = 2.7, p = 0.005). The increased risk was not modulated by medication.
Conclusion:
Our data underline the negative long-term impact of alcohol use disorders on bipolar disorder with more depressive bipolar I episodes and the importance of its detection and treatment.
Introduction
The pooled lifetime prevalence of alcohol use disorder (AUD) in bipolar disorder is estimated to be around 35% (Di Florio et al., 2014). This represents one of the most common comorbid axis I disorder among bipolar patients and implies a greater risk of nonadherence to pharmacological treatment (Baldessarini et al., 2008; Keck et al., 1997), a higher risk of suicidality (Cardoso et al., 2008; Dalton et al., 2003; Oquendo et al., 2010) and violent behavior (Salloumet al., 2002). With respect to relapses, some studies found evidence for more affective episodes due to AUD comorbidity (DelBello et al., 2007; Goldstein and Levitt, 2006; Kessing, 1999; Strakowski et al., 1998; Tohen et al., 1990a, 1990b), whereas others did not reveal such a correlation (Keck et al., 1995; Strakowski et al., 2005; Van Zaane et al., 2010; Van Zaane et al., 2014; Winokur et al., 1994; Winokur et al., 1995). A recent review on that topic presented as most consistent finding that AUDs in bipolar disorder are associated with more rapid cycling and mood episode switching; however, conclusions with respect to AUD caused relapses were not conclusive due to inconsistent results across studies (Rakofsky and Dunlop, 2013). Furthermore, the authors found no negative effect of AUDs on time to remission and no consistent temporal pattern regarding whether excessive alcohol use preceded or followed the development of mood episodes.
To add further evidence to this debate, we tested the effect of AUDs in bipolar I and II patients in a large prospective study with a 4-year observational period. We hypothesized that both bipolar I and II patients with comorbid AUDs would suffer from more affective relapses than those without this comorbidity.
Material and methods
Study design
This inpatient trial was a prospective, naturalistic, observational study, conducted in the County Hospital Neunkirchen, Austria, from 2000 through 2008 (Simhandl et al., 2014a). All diagnoses were made by two independent psychiatrists following International Classification of Diseases-10 (ICD-10) criteria based on a detailed clinical interview and review of case-notes carried out by two independent psychiatrists. The clinical interview at admission included elements such as a cross-sectional, semi-standardized psychopathology description, detailed retrospective exploration of the course of the disease, life events, family history, physical comorbidities and prior medication record. A family member interview was conducted if available. For additional psychiatric comorbidities, the Mini-International Neuropsychiatric Interview (MINI) was used, which is validated for Diagnostic and Statistical Manual of Mental Disorders (4th ed.; DSM-IV) and ICD-10 (Sheehan et al., 1998). Furthermore, in all patients, a routine blood analysis was undertaken at admission. All information was gathered in the hospital records and in a web-based semistructured interview which was specifically designed for this prospective long-term observation (Simhandl et al., 2014a). Exclusion criteria of this analysis were limited to the presence of neurological diseases and abuse or dependence of substances other than alcohol and nicotine.
Bipolar patients entered the prospective follow-up in either a hypomanic or manic index episode, in a depressive index episode with or without psychotic symptoms or with ‘other syndromes’ as index episode. ‘Other syndromes’ were defined as an anxiety disorder, a mixed episode or rapid cycling following ICD-10 criteria. During their admissions, all patients received a psychoeducational program with respect to their diagnosis, possible risk factors and the importance of pharmacological treatment. After discharge, patients typically remitted clinically and were treated by their own psychiatrist. In case of a comorbid AUD diagnosis, patients were recommended to participate in specialized outpatient services available in the region in addition to their psychiatric treatment as they did not receive a specific AUD-orientated intervention during admission.
During the follow-up, they subsequently were evaluated in person at least once a year in the outpatient clinic of the hospital (78%), via telephone contact or personal contact with their own psychiatrist (22%). No affective rating scales were applied. At follow-up visits, the psychiatrists in charge of the study introduced their evaluation in the web-based database with regard to affective relapses; adherence to treatment; use of medication; drug, alcohol and nicotine misuse; and occurrence of life events and suicidality, among others. Relapses were defined as a deterioration or change of the affective state needing an explicit pharmacological intervention and/or rehospitalization. In case of a severe mood episode, patients continued the study and received additional follow-up. All data were always directly entered into the self-developed web-based database.
This study was approved by the ethics committee of the University of Vienna (Austria), and all patients provided written informed consent for participation in a long-term clinical study. The trial has been registered as www.clinicaltrials.gov with registration number: NCT01792128.
Subjects
In total, 515 bipolar I and II patients were consecutively admitted to the local psychiatric hospital in Neunkirchen (Austria) from 2000 through 2004. Of 515 admitted patients, 366 (71.1%) agreed to participate and signed the informed consent at discharge. Of those, 66 (18%) patients did not enter the follow-up period after discharge from the hospital due to the following reasons: 1 patient (0.3%) moved to another country, 23 (6.3%) received a different diagnosis when they were rediagnosed and 31 (8.4%) could not be contacted. Furthermore, five (1.4%) died from a natural death and six (1.6%) from suicide. A total of 15 other patients had to be discarded from this study because they had abuse or dependence of substances other than alcohol and nicotine: 2 (0.5%) had a cannabis use disorders, 1 (0.3%) had a cocaine abuse, 9 (2.5%) a sedative use disorders and 3 (0.8%) had a multiple substance use disorders. Follow-up time was missing for a bipolar II patient with alcohol abuse (AA). Thus, the final analysis included data from 284 bipolar patients, including 161 bipolar I and 123 bipolar II patients. They all received a 4-year follow-up, with at least two evaluation data. Mean duration of admission of included subjects was 23.7 days (range: 1–90 days).
All patients were prescribed typically used drugs in bipolar disorder, in monotherapy or in combination. Treatment was not influenced by the study protocol and included lithium, valproate, carbamazepine, lamotrigine or atypical antipsychotics, such as olanzapine, risperidone or quetiapine. Additionally, patients were also treated with antidepressants (mainly selective serotonin reuptake inhibitors [SSRIs]), benzodiazepines and medication for physical comorbidities (mainly for hypertension, diabetes and thyroid dysfunction).
Database
The web-based database was especially designed for this study with the objective of an uncomplicated data entry via Internet. All participating psychiatrists (n = 9) were instructed in the use of the database and personally entered corresponding data of their patients, thus avoiding errors that often occur during transcription of medical record to study protocol, and eventually data entry into the database. The principal investigator of the study, C.S., and the statistician, B.K., regularly checked the correctness of entered data.
Statistics
Relationships between the presence of AUDs and demographic (age, sex and civil status), clinical (age of onset, number of episodes, number of admissions, presence of somatic comorbidities, presence of comorbid anxiety disorders and presence of comorbid personality disorder [PD]), psychological (number of life events after the index episode), behavioral (good adherence to medication) and biological (family history of bipolar disorder and disorder type) variables were assessed using logistic regressions in case of continuous variables (with the continuous predictor, e.g. age, being the independent variable and the presence of an AUD being the dependent binary outcome) and chi-square tests in case of categorical variables.
Relationship between the presence of AUDs and the time to relapse was assessed using Cox proportional hazards regression models, in which the time to relapse was modeled as a function of the presence of an AUD. These analyses were also separately conducted for manic and depressive relapses and for patients with bipolar I and bipolar II disorders. All analyses were performed with the base and ‘survival’ (Therneau and Lumley, 2011) packages for R (http://cran.r-project.org/).
Results
Demographical and clinical data
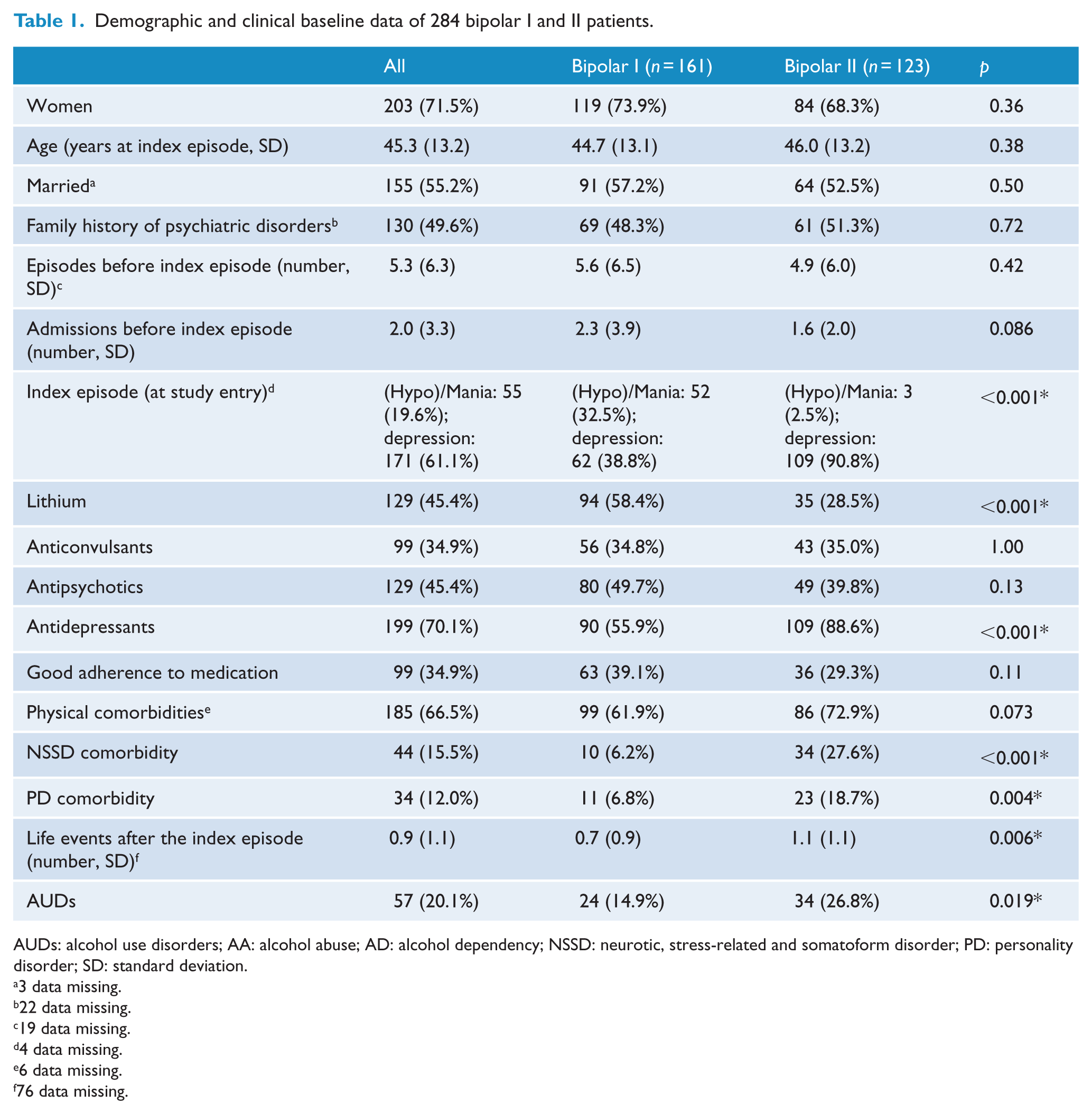
Baseline data are detailed in Table 1. Bipolar II patients showed more frequently depressive polarity in the index episode (90.8% vs 38.8%), a higher prevalence of comorbid neurotic, stress-related and somatoform disorders (NSSDs; 27.6% vs 6.2%) and PDs (18.7% vs 6.8%) and received more frequently antidepressants (88.6% vs 55.9%) and less frequently lithium (28.5% vs 58.4%). The number of life events during the follow-up was also higher in bipolar II patients (1.1 vs 0.7).
Demographic and clinical baseline data of 284 bipolar I and II patients.
AUDs: alcohol use disorders; AA: alcohol abuse; AD: alcohol dependency; NSSD: neurotic, stress-related and somatoform disorder; PD: personality disorder; SD: standard deviation.
3 data missing.
22 data missing.
19 data missing.
4 data missing.
6 data missing.
76 data missing.
Prevalence of AUDs
Global prevalence at baseline of AUDs was 20.1%, with 10.6% patients having AA and 9.5% patients having alcohol dependency (AD). Bipolar II patients had a statistically significant higher prevalence of AUDs than bipolar I patients (26.8% vs 14.9%, χ2 = 5.46 [AUD vs no AUD in bipolar II vs I], p = 0.019; Table 1). Differences were similar for AA and AD (AA: 13.0% vs 8.7%; AD: 13.8% vs 6.2%; χ2 = 0.22 [AD vs AA in bipolar II vs I], p = 0.641). This higher prevalence of AUDs in bipolar II patients than in bipolar I patients was also observed when male and female patients were analyzed separately (trend in male subjects: 43.6% vs 33.3%, χ2 = 0.52, p-value = 0.471; statistically significant in female subjects: 19.0% vs 8.4%, χ2 = 4.09, p-value = 0.043).
Male bipolar subjects also had a statistically significantly higher prevalence of AUDs than female patients (38.3% vs 12.8%; χ2 = 21.84 [AUD vs no AUD in male vs female patients], p-value < 0.001), with this difference being similar for AA and AD (AA: 21.0% vs 6.4%; AD: 17.3% vs 6.4%; χ2 = 0.01 [AD vs AA in male vs female patients], p = 0.641). This higher prevalence of AUDs in male patients than in female patients was also observed when bipolar I and bipolar II patients were analyzed separately (bipolar I: 33.3% vs 8.4%, χ2 = 13.31, p-value < 0.001; bipolar II: 43.6% vs 19.0%, χ2 = 6.97, p-value = 0.008).
Finally, the prevalence of AUDs was moderately higher in patients receiving anticonvulsants (28.3% vs 15.7%, χ2 = 5.63, p-value = 0.018). The trend was also observed when bipolar I and bipolar II (bipolar I: 17.9% vs 13.3%, χ2 = 0.29, p-value = 0.59; bipolar II: 41.9% vs 18.8%, χ2 = 6.48, p-value = 0.011) or male and female patients were analyzed separately (males: 50.0% vs 31.4%, χ2 = 2.04, p-value = 0.15; females: 18.8% vs 9.7%, χ2 = 2.64, p-value = 0.10), although statistical significance was only observed in bipolar II patients.
No relationship of prevalence of AUDs were observed with age, civil status, family history, somatic comorbidity, NSSD comorbidity, age of onset, number of episodes, number of admissions, polarity of index episode, treatments other than anticonvulsants, number of life events after the index episode or adherence to medication (all p > 0.19). There was a weak trend to increased prevalence of AUDs in individuals with PDs (p = 0.093).
Course of the illness depending on presence of AUDs
The presence of AUDs was associated with an increased risk of a depressive relapse, although only in patients with bipolar I disorder (hazard ratio [HR] = 2.68, p = 0.005; Table 2 and Figure 1). The increase in depression relapse rate in bipolar I disorder was moderate but not statistically significant for AA (HR = 1.9, p = 0.19) and stronger and statistically significant for AD (HR = 3.2, p = 0.010). These increases in risk were not modulated by medication as none of the 12 potential interactions between the presence of AUDs and medication was statistically significant. Both the risk of a manic relapse in bipolar I disorder and the risk of a depressive episode in bipolar II disorder were not associated with the presence of AUDs.
Effects of the AUDs on the risk of manic and depressive relapse.
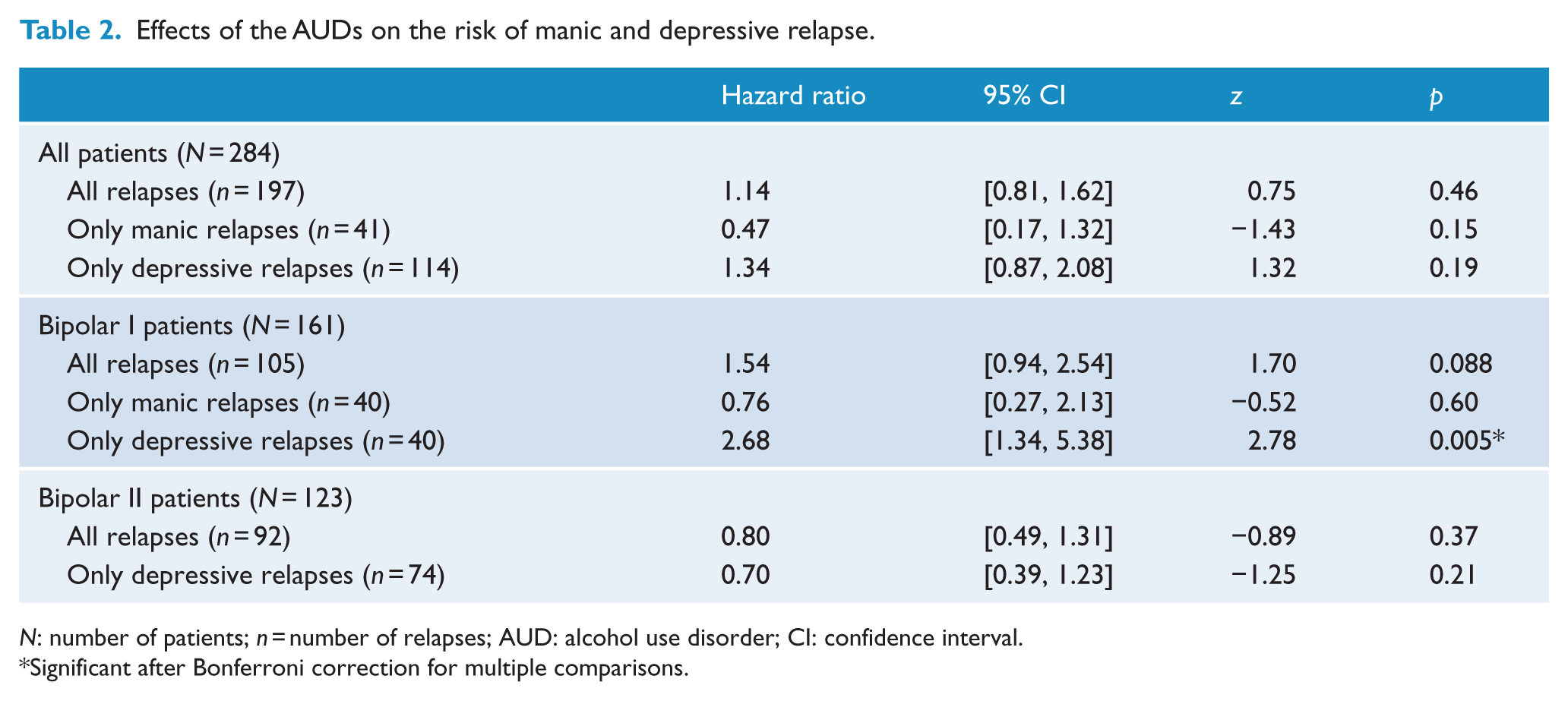
N: number of patients; n = number of relapses; AUD: alcohol use disorder; CI: confidence interval.
Significant after Bonferroni correction for multiple comparisons.
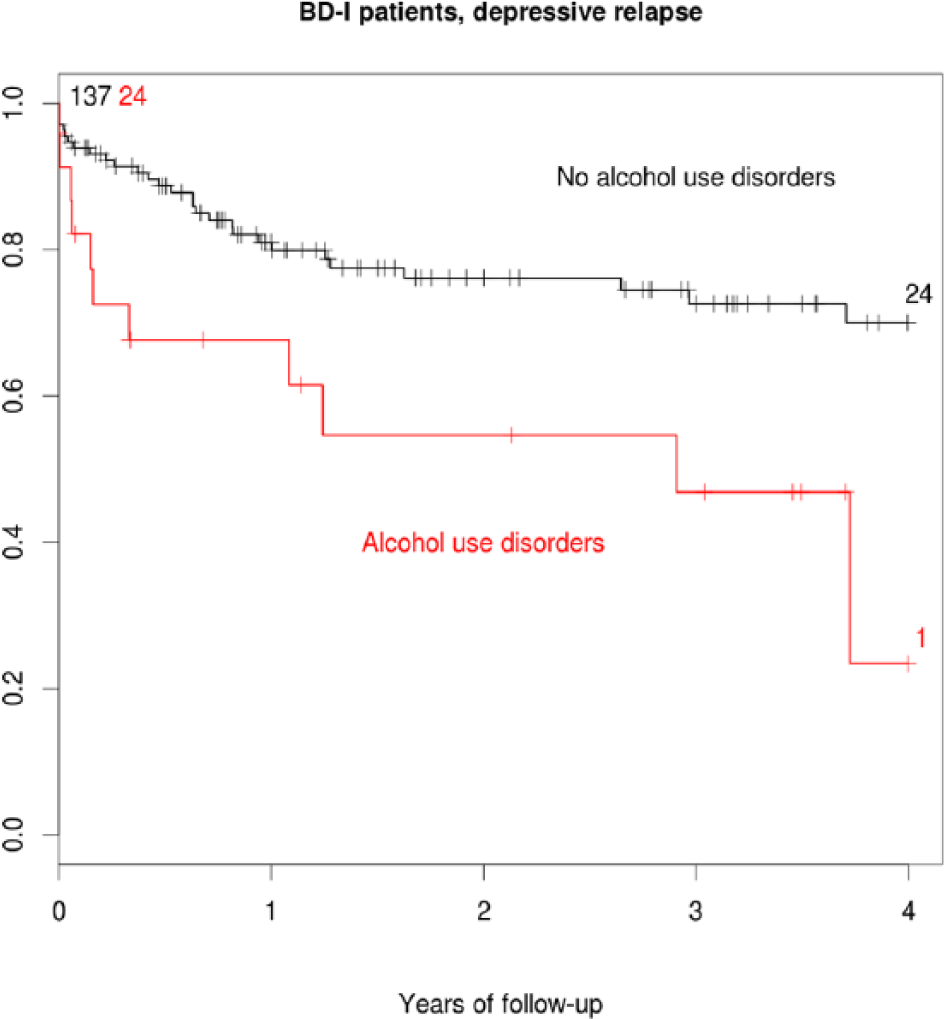
Effects of AUDs on the risk of depressive relapses in bipolar I disorder (n = 161).
Hypomanic episodes could not be associated with the presence of AUDs. This was due to the design of the original study protocol which did not consider hypomanic phases as relapses as they are often difficult to diagnose, and patients usually do not consult with their corresponding psychiatrist due to hypomanic symptoms (see definition of relapses in the ‘Study design’ section). Therefore, only depressive but not hypomanic episodes could be calculated in this sense in the bipolar II sample.
Discussion
To the best of our knowledge, this is one of the largest and longest studies investigating the effects of AUDs on the relapse rate of bipolar disorder, and one of the few studies including both bipolar I and II patients. We found that AUDs are a risk factor on the long-term course of the disorder, increasing the risk of depressive relapses in bipolar I disorder. This finding is in accordance with six previous studies (DelBello et al., 2007; Goldstein and Levitt, 2006; Kessing, 1999; Strakowski et al., 1998; Tohen et al., 1990a, 1990b). Five other studies did not find a significant relationship (Keck et al., 1995; Strakowski et al., 2005; Van Zaane et al., 2010; Van Zaane et al., 2014; Winokur et al., 1994; Winokur et al., 1995). However, the relatively low sample sizes (DelBello et al., 2007; Keck et al., 1995; Tohen et al., 1990a, 1990b; Winokur et al., 1994; Winokur et al., 1995) and the use of retrospective designs (Goldstein and Levitt, 2006) may have limited the statistical power of the majority of these studies. A case registry study of Denmark analyzed 2903 bipolar patients and found an increased rate of mood relapses in bipolar patients with AUD when compared to bipolar patients without AUD; limitation of this work was that data relate to readmissions rather than recurrence (Kessing, 1999).
Our results were not influenced by sociodemographical or clinical variables such as physical and psychiatric comorbidities, index episode, adherence or type of medication prescribed. The latter is of clinical interest as we could not detect a specific drug with a protective effect on relapses of bipolar patients with comorbid AUD. It is noteworthy that we had detected in a prior analysis that life events in our cohort were also associated with increased risk of depressive episodes in bipolar I patients (Simhandl et al., 2014b). However, we could not observe any relationship between prevalence of AUDs and the number of life events, and both were significantly associated with increased risk of relapse in a multiple Cox regression (p = 0.003 and 0.037) Therefore, their interaction was not significant, for what they should be considered independent risk factors. As far as we know, only one study so far studied the interaction between trauma, AUD and bipolar disorder (Nery et al., 2014). They found that comorbid AUD bipolar subjects were more likely to present with posttraumatic stress disorder than bipolar patients without AUD.
The global prevalence of AUDs in our bipolar sample was 20.1%, which is similar to the 23% prevalence rate of comorbid AUD reported in Brazil (Nery et al., 2014). These prevalence rates are lower than the pooled lifetime prevalence of 35% reported in a recent meta-analysis (Di Florio et al., 2014), but this is expectable given that lifetime prevalence should be superior to a transversal prevalence. We also found that bipolar male patients of our cohort had a more than three times higher risk of comorbid AUD than female patients. This finding highlights previous evidence of male bipolar patients being more prone to suffer from comorbid AUDs (Di Florio et al., 2014; Kessler et al., 1997; Nery et al., 2014). It must be noted, however, that two bipolar studies did not find significant gender differences on comorbidity with AUD (Chengappa et al., 2000; Maier et al., 1995), and mixed findings were reported in the retrospective study by Goldstein and Levit (2006).
A further finding of our study was that bipolar II patients had a higher risk of comorbid AUD than bipolar I patients, but no negative effect of this comorbidity was detected on relapse frequency. This is surprising bearing in mind that bipolar II patients suffer from more affective relapses than bipolar I patients (Amann and Radua, submitted). One reason for the absence of effects of AUDs on our bipolar II cohort might be that bipolar II disorder is considered as distinct categorical subtype within the bipolar spectrum (Parker, 2015), and bipolar II depressive episodes also differ from a phenomenological point of view from bipolar I depressive episodes (Frankland et al., 2015) and, therefore, are less prone to develop comorbid AUDs. Furthermore, it cannot be excluded that comorbid AUDs caused more hypomanic relapses in bipolar II subjects in our cohort, but, as stated before, we do not have data on hypomanic recurrences as they were not defined as relapses in the original study protocol.
Bipolar II disorder has been understudied in general, and the evidence of comorbid AUD in bipolar II disorder is, thus, scarce. The majority of previous studies did not differentiate the type of affective relapse or only included bipolar I patients. Of interest, Goldstein et al. (2006) evaluated both bipolar I and II patients in their retrospective study and found more hypomanic relapses in bipolar II patients, especially in female subjects (Goldstein and Levitt, 2006). However, these results have to be considered with caution as hypomanic episodes are difficult to evaluate, especially in a retrospective design. Another longitudinal study included also 47 bipolar II patients (Van Zaane et al., 2010, 2014) and did not find any effect of comorbid AUD on the course of the illness of bipolar I or II patients.
The strengths of our work include the large sample size of the cohort, the long duration of the follow-up and the inclusion of both bipolar I and II subjects. Furthermore, all subjects were diagnosed by two psychiatrists (who also re-evaluated the patients throughout all the follow-up) and were recruited in a community psychiatric hospital setting rather than in a specialized bipolar clinic, for what they can be considered to be more representative of the population of individuals with bipolar disorder.
Methodological caveats of our study must be also taken into account when translating results to clinical work. First, it has to be noted that we did not measure weekly alcohol use patterns, and we did not use standardized instruments such as the alcohol use identification test. We could, thus, not differentiate the impacts of current versus past AUDs on the long-term course of the disease. Previous studies suggest that current and ongoing AUDs influence more negatively the course of the illness than past AUDs (Hirschfeld et al., 1989; Mueller et al., 1994). Second, we did not evaluate time to remission, episode switching and rapid cycling. Third, and as stated before, we cannot draw conclusions on the effect of AUDs on hypomanic episodes in our bipolar II sample as hypomanic episodes were not defined as relapses in the original study protocol. Finally, some strict methodological issues of randomized controlled trials could not be integrated in the design of a naturalistic, observational study, such as defined dosages of drugs or blood sampling during all visits.
Despite these limitations, findings from our study suggest that comorbid AUD has a negative impact on bipolar I subjects, increasing depressive episodes. This finding highlights the importance of a continuous detection and treatment of AUDs in bipolar patients and is of special relevance in primary care as subjects might present in a depressive state associated with recent alcohol use. Furthermore, the identification of a history of manic symptoms in depressed subjects is recommended to avoid a worse outcome with an acceleration of depressive episodes in a possible bipolar I disorder. Clinicians should be especially careful with male bipolar subjects as they seem more prone to develop comorbid AUD during the course of the disease.
Footnotes
Acknowledgements
We acknowledge support by the Research Group Industrial Software Engineering of the Technical University of Vienna and their department head Prof. DI Dr Thomas Grechenig. Furthermore, we thank Drs Jutta Bilonoha, Elisabeth Denk, Birgit Gasselseder, Jens Mersch, Klaudia Mitterwachauer, Stephanie Schiebel, Christian Wunsch, Ali Zoghlami and Daniela Renhofer for data entry. The authors acknowledge support from Thomas Simhandl, BSc, for the first version of the web-based interview and database.
Declaration of interest
The authors do not have any conflicts of interest related to this manuscript.
Funding
Development of the web-based interview and database was funded by the Österreichische Forschungsförderungsgesellschaft mbH (FFG), which is the Austrian Federal Department for Transport, Innovation, and Technology, Department of Innovations and Telecommunications. Additional support came from the Centro de Investigación Biomédica en Red de Salud Mental (CIBERSAM) and by the Instituto de Salud Carlos III, Spain, with grants (PI07/1278 and PI10/02622), a Stabilization Contract grant (CES 12/024) to B.L. Amann and a Miguel Servet research contract (CP14/00041) to J. Radua.
