Abstract
Objective:
The Royal Australian and New Zealand College of Psychiatrists recommends screening for a range of antibodies in first-episode psychosis, including anti-glutamic acid decarboxylase antibodies. Glutamic acid decarboxylase antibody–associated encephalitis occurs with high antibody titres and may cause cognitive dysfunction, seizures and psychiatric symptoms. However, glutamic acid decarboxylase antibodies are more frequently found in lower titre in association with other autoimmune disorders (such as diabetes mellitus type 1) and in healthy individuals. The utility of testing unselected populations of consumers with psychosis is unclear. The psychiatric manifestations of this disorder are also poorly described.
Methods:
First, systematic review of cohort and case–control studies that tested for IgG glutamic acid decarboxylase antibodies in psychiatric populations was conducted. Random-effects meta-analysis of odds ratio for antibody positivity in cases with psychosis and controls assessed prevalence. Second, literature review of all published cases and case series of glutamic acid decarboxylase antibody–associated limbic encephalitis was assessed for frequency and description of psychotic symptoms.
Results:
There were 17 studies, in which 2754 individuals with psychotic disorders were tested for glutamic acid decarboxylase IgG antibodies. Thirty-one consumers with psychosis (0.7%) had positive glutamic acid decarboxylase antibodies compared to 24 controls (1.0%), all at low titre and not fulfilling diagnostic criteria for autoimmune encephalitis. Meta-analysis found no significant difference in rates of glutamic acid decarboxylase antibody positivity (odds ratio = 1.8, 95% confidence interval: [0.90, 3.63]). Literature review found 321 cases of glutamic acid decarboxylase antibody–associated limbic encephalitis, with psychosis in 15 (4.3%) cases. Clinical screening would have identified all cases that presented to psychiatric services.
Conclusion:
Glutamic acid decarboxylase antibodies were uncommon in consumers with psychosis, with no significant difference in prevalence from controls and no cases of encephalitis identified. In cases with established glutamic acid decarboxylase antibody–associated limbic encephalitis, psychotic symptoms were uncommon and identifiable by clinical assessment. Targeted antibody testing guidelines should be further considered.
Introduction
There is an increasing awareness of the importance of testing for neuronal autoantibodies to cell surface and intracellular antigens in people with severe neuropsychiatric syndromes (Endres et al., 2020). Autoantibodies to neuronal cells are found in up to 5% of those experiencing their first-episode psychosis (FEP) (Colijn and Ismail, 2019; Lennox et al., 2017; Scott et al., 2018a; Zandi et al., 2011). There are multiple different autoantibodies identified in both serum and cerebrospinal fluid (CSF). When these are causative of illness, their early detection is important since rapid commencement of immunotherapy is associated with better clinical outcomes (Broadley et al., 2019). Currently, the Royal Australian and New Zealand College of Psychiatrists (RANZCP) guidelines recommend universal screening of all those with FEP for a range of antibodies, including those targeting anti-glutamic acid decarboxylase (GAD) (Galletly et al., 2016). However, the psychiatric syndromes associated with anti-GAD encephalitis are poorly defined (Vrillon et al., 2020).
GAD is an intracellular protein expressed in pre-synaptic gamma-amino-butyric-acid (GABA) neurons in the central nervous system (CNS) (Graus et al., 2020; Zhu et al., 2020). In the CNS, GAD is the rate-limiting enzyme that catalyses the decarboxylation of glutamate to GABA. Disruptions to GAD function are thought to result in neurotransmitter imbalance and neuronal hyperexcitability (Fenalti and Buckle, 2010; Zhu et al., 2020). GAD is also found in the pancreas, liver, kidney, adrenal glands and testes (McKeon and Tracy, 2017). Anti-GAD antibodies have been associated with a variety of neurological conditions, including stiff person syndrome, epilepsy and cerebellar ataxia (Muñoz-Lopetegi et al., 2020). Anti-GAD antibody–associated neurological syndromes are typically seen with very high (>1000 IU/mL) antibody titres (Graus et al., 2020; Muñoz-Lopetegi et al., 2020). At lower titres (<1000 IU/mL), anti-GAD antibodies are seen widely in extra-neurological disorders, including up to 80% of those with type 1 diabetes mellitus (Solimena and De Camilli, 1991). Low titre anti-GAD antibodies may also be found in those with autoimmune thyroid disease, thymus-related disorders, pernicious anaemia, psoriasis, coeliac disease, Addison’s disease, antiphospholipid syndrome and vitiligo (Saiz et al., 2008). Anti-GAD antibodies have also been shown to be present in healthy individuals, without any neurological or other clinical symptoms (Batstra et al., 1999).
Limbic encephalitis is significantly less common than other neurological or non-neurological syndromes associated with anti-GAD antibodies (Malter et al., 2010; Vrillon et al., 2020). Anti-GAD antibody–associated limbic encephalitis most commonly presents with seizures and/or cognitive deficits such as anterograde amnesia (Vrillon et al., 2020). Onset is typically sub-acute, although cases where delayed diagnosis occurs with gradual progression of symptoms have been described (Fauser et al., 2015). Psychiatric symptoms have been reported in up to two-thirds of cases during the course of the illness; most frequently this is with depressed mood, as well as, emotional instability and anxiety (Vrillon et al., 2020; Zhu et al., 2020). Behavioural changes, such as apathy, irritability and bizarre behaviours, have also been reported (Gagnon and Savard, 2016). Psychotic symptoms are infrequently described (Gagnon and Savard, 2016; Vrillon et al., 2020).
Widespread GAD antibody testing in FEP therefore has a low pre-test probability and may result in diagnostic uncertainty, invasive investigations and delay in traditional psychiatric care with additional health service financial implications (Chan et al., 2021; Ross et al., 2020). Guidelines do not specify whether antibody screening should be conducted in serum or CSF, although clinical practice dictates serum testing (Galletly et al., 2016). Currently, any positive anti-GAD antibody testing, irrespective of antibody titre or clinical presentation, results in the need for further neurological assessment. Typically, this includes testing for CSF antibodies, neuroimaging, electroencephalogram (EEG) and often neurology review (Graus et al., 2020).
This study aims to systematically review the utility of universal GAD antibody testing in FEP in two ways. First, cohort studies of consumers with psychotic disorders will be examined to determine the prevalence of GAD antibody positivity. It is appreciated that this is an emerging area of study and the numbers in the cohort studies may be insufficient to adequately examine for GAD antibody positivity. Therefore, a second aim is to review all cases of GAD antibody–associated limbic encephalitis that report psychotic symptoms. Existent clinical screening criteria for anti-N-methyl-
Methods
This study was pre-registered with PROSPERO (CRD 42021279274) (Booth et al., 2012). The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement recommendations were followed (Moher et al., 2015).
PubMed, Embase, PsycINFO, CINAHL and Google Scholar were searched from inception to September 2021 using the search terms: ‘psychosis’ OR ‘schizophrenia’ OR ‘psychiatric’ OR ‘mental illness’ AND ‘autoimmune encephalitis’ OR ‘limbic encephalitis’ OR ‘neuronal antibodies’ OR ‘glutamic acid decarboxylase’ OR ‘glutamate decarboxylase’. Search and screening at title, abstract and then full text was performed independently by two authors (N.W., K.F.) and references of selected articles were searched for additional studies. Any divergence was resolved between all authors.
Part 1: psychiatric cohorts tested for GAD antibodies
Studies that assessed for IgG GAD antibodies in cohorts of people with psychiatric disorders were included. Broader inclusion criteria that included FEP cohorts, as well as those with established diagnoses of psychosis and other psychiatric disorders, were employed as it was known that some studies of neuronal antibodies combine both. Studies that used controls, as well as those only assessing antibodies in psychiatric cases, were included. No age restrictions were applied. Studies assessing other anti-neuronal antibodies, not including GAD antibodies, or non-IgG GAD antibodies, were excluded, as were studies not reporting on individual antibody results. All languages were included.
Data extraction was completed independently by two authors (N.W., K.F.). Data extracted included demographics, psychiatric diagnoses, measures of psychiatric illness severity (admissions, duration of illness, standardised measures e.g. Brief Psychotic Rating Scale), medical comorbidity (especially diabetes and other causes of non-neurological GAD antibodies), GAD antibody testing method and normal values, GAD antibody positivity in both cases and controls, GAD antibody titre and reporting of GAD antibody–associated limbic encephalitis. High and low abnormal GAD antibody titres were determined by standardised cut off points for each method of antibody testing (Daif et al., 2018).
Descriptive statistics were employed to report on the cohort data. Meta-analysis of GAD antibody positivity prevalence in studies using controls compared to psychotic disorders was completed with Review Manager software package (RevMan, 2014: Version 5.4). Mantel–Haenszel method for meta-analytical estimation of odds ratios was employed, given the rarity of the outcomes, the number of zero events in the included studies and the differences in group sizes (Efthimiou, 2018). Between-study heterogeneity was assessed using the I2 statistic. A random-effects meta-analysis was chosen for all analyses due to presumed differences in patient groups and study methods.
Part 2: GAD antibody–associated limbic encephalitis
Case series/case studies or cohorts of GAD-associated limbic encephalitis were identified and examined for the presence of psychotic symptoms. Psychotic symptoms were defined as hallucinations, delusions, thought disorder or noting of ‘psychosis’ or ‘psychotic symptoms’. Studies without evidence of IgG GAD antibody positivity were excluded. Cases with mood or other psychiatric symptoms, without psychosis, were excluded. All ages and studies in all languages were included. Cases duplicated in other case reports or series were only included once, with the reference maintained for the case with the largest amount of clinical information.
Data extraction was completed independently by two authors (N.W., K.F.) and included demographics, clinical presentation, medical and psychiatric history, para-clinical investigations, GAD antibody testing and titre. Internationally recognised diagnostic criteria for ‘definitive’ and ‘probable’ autoimmune encephalitis were applied to ensure consistency of encephalitis diagnosis, as were established clinical screening criteria (Graus et al., 2016; Scott et al., 2018b). Descriptive statistics were utilised.
Quality assessment of all studies in both Part 1 and Part 2 was performed using Johanna Briggs Institute critical appraisal tools for cohort studies, case series and case reports (Munn et al., 2014).
Results
There were 2190 non-duplicated results following database searches with 122 studies identified after screening at title, abstract and full text (Figure 1). This includes 17 cohorts of psychiatric patients where GAD antibodies have been tested, as well as 105 studies, reporting on 346 cases of GAD antibody–associated limbic encephalitis. Quality assessment for the psychiatry cohort studies was moderate with an average JBI score of 7.5/10 (Supplementary Data). Quality assessment for the GAD encephalitis cases was moderate, with an average JBI score of 6.0/8 in the 74 cases and 6.5/10 in the 31 case series (Supplementary Data).
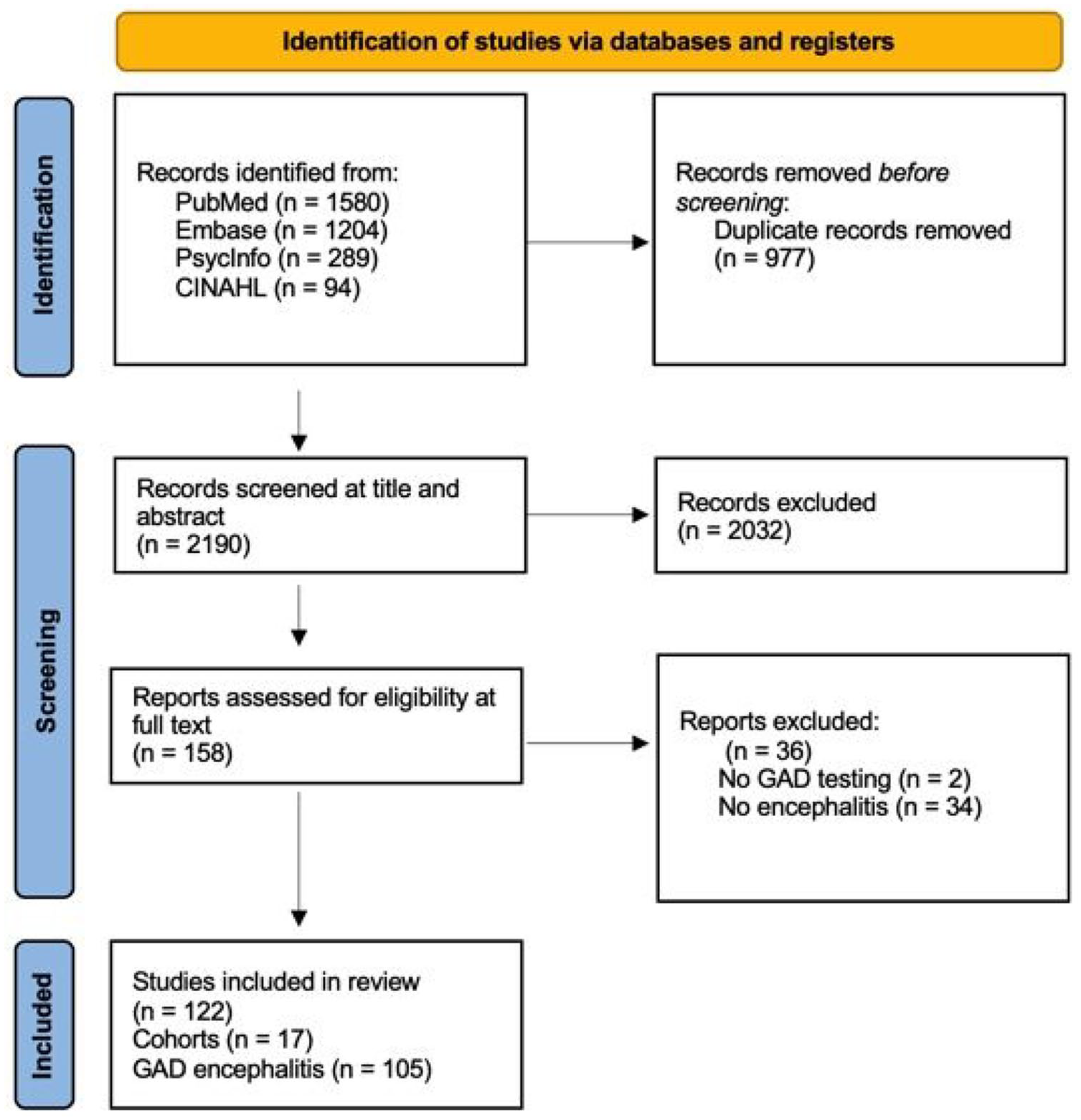
PRISMA 2020 flow diagram: GAD antibodies in psychosis.
Part 1: psychiatric cohorts tested for GAD antibodies
There were 17 psychiatric patient cohorts, in 13 different countries, which examined the prevalence of GAD antibodies published from 2004 to 2021 (Table 1). This includes a total of 4131 patients, with psychotic disorders (2754 patients, 66.7%), bipolar affective disorder (289 patients, 7.0%) or not specifically defined ‘psychiatric illness’ (1088 patients, 26.3%). There were 1670 patients (40.4%) who were inpatients at the time of the study, 685 (16.6%) were outpatients and 1776 did not have a clearly stated location. Age was reported in seven studies, with a weighted mean of 37.5 years. Gender was reported in eight studies with a 1:1.2 male to female gender ratio. Twelve of the studies include controls (total 2724 controls). Only three studies specifically excluded those with diabetes and known neurological disorders. Measures of psychiatric illness severity or symptoms were insufficiently documented.
Psychiatric cohorts tested for GAD antibodies.
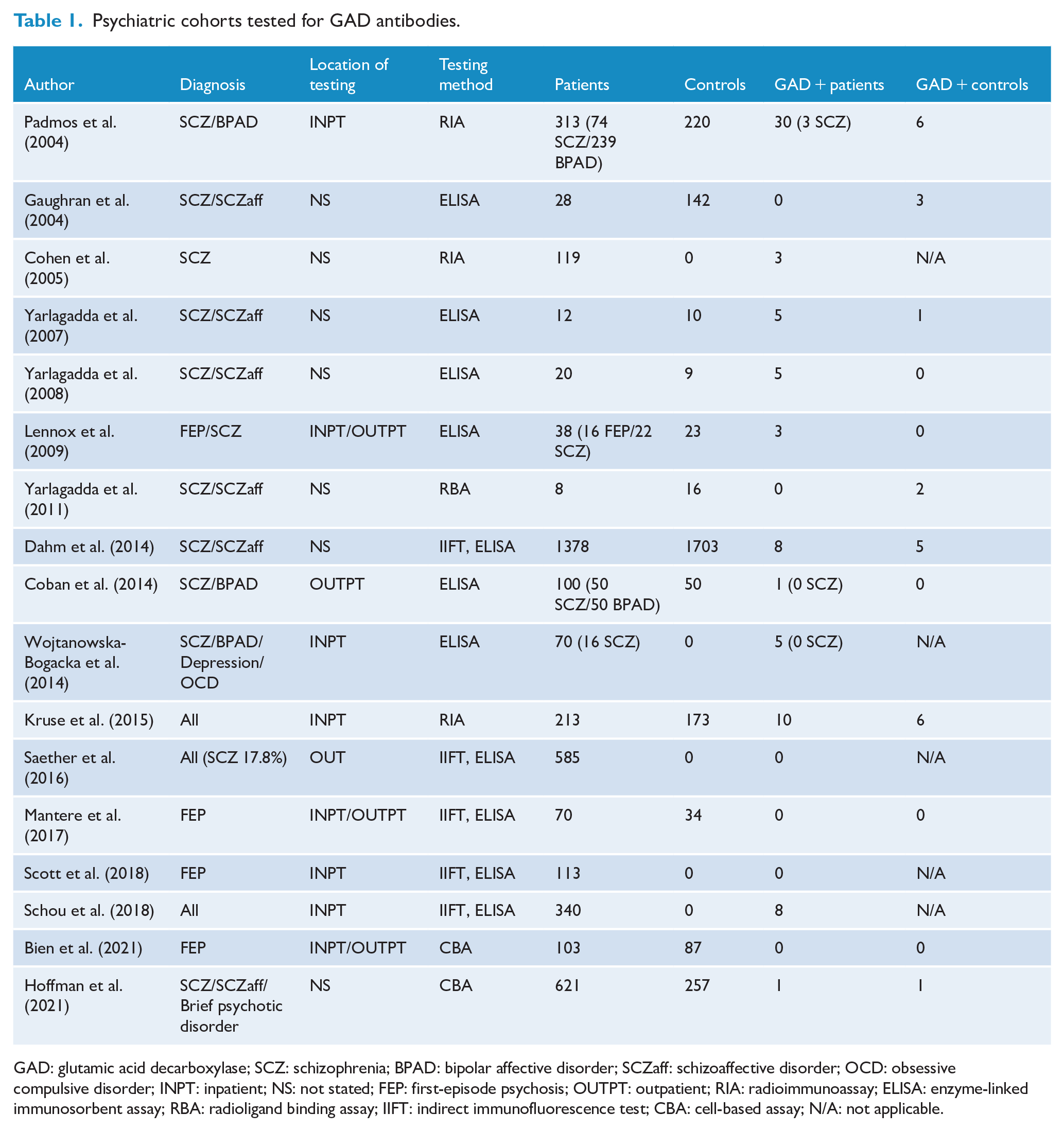
GAD: glutamic acid decarboxylase; SCZ: schizophrenia; BPAD: bipolar affective disorder; SCZaff: schizoaffective disorder; OCD: obsessive compulsive disorder; INPT: inpatient; NS: not stated; FEP: first-episode psychosis; OUTPT: outpatient; RIA: radioimmunoassay; ELISA: enzyme-linked immunosorbent assay; RBA: radioligand binding assay; IIFT: indirect immunofluorescence test; CBA: cell-based assay; N/A: not applicable.
The method for testing GAD antibodies included enzyme-linked immunosorbent assay – ELISA (six studies), combined ELISA and indirect immunofluorescence test (five studies), radioimmunoassay (three studies), cell-based assay (two studies) and radioligand assay (one study). Mean antibody titre was insufficiently reported to allow comparison. All studies assessed for GAD antibodies in serum except Bien et al. where both serum and CSF were tested. Additionally, in Kruse et al., the CSF of serum positive cases was assessed.
There were 79 cases of GAD-positive results in the psychiatric patient cohort (1.9%), of which 31 had psychosis (0.7%), compared with 24 GAD-positive cases in controls (1.0%) (Table 1). Four studies did not include controls, accounting for 12.8% of the total psychosis cohort. No GAD antibody positive result in cases or controls was documented to be clinically diagnosed with GAD antibody–associated limbic encephalitis, fulfilled proposed clinical diagnostic criteria for ‘possible autoimmune encephalitis’ or was in the ‘high abnormal value’ range (>1000 IU/mL). The studies that assessed for GAD antibodies in CSF did not find any positive results.
Meta-analysis of studies comparing GAD antibody–positive prevalence between psychosis cohorts (n = 2179) and controls (n = 2380) did not demonstrate a statistically significant result (odds ratio = 1.8; 95% confidence interval = [0.90, 3.63], p = 0.099), indicating there was no increased prevalence of GAD antibodies in those with psychosis compared to controls (Figure 2). There was no significant heterogeneity between studies (I2 = 0%). Visual inspection of the funnel plot did not demonstrate publication bias (Supplementary Data). Three studies had no events in both psychosis and control groups, accounting for 8.1% of the psychosis cohort.
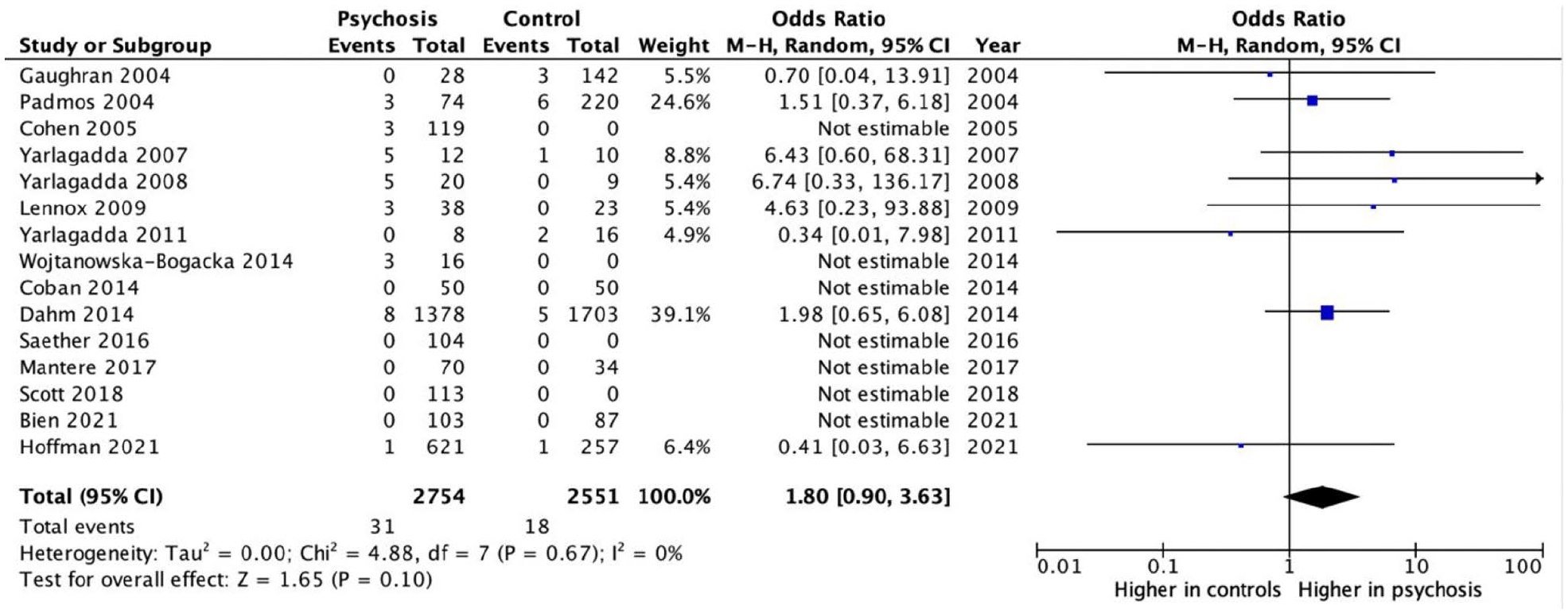
Forest plot for GAD antibody positivity in patients with psychosis compared to controls.
Part 2: GAD antibody–associated limbic encephalitis
There were 346 cases of GAD antibody–associated limbic encephalitis described in the literature from 2008 to 2021 (Supplementary Data). The most common symptoms of limbic encephalitis included seizures (198 cases, 57.2%), cognitive deficits (134 cases, 38.7%), behavioural or psychiatric symptoms (48 cases, 13.9%) and focal neurological deficits (35 cases, 10.1%). However, in 132 cases (38.2%), the presentation was only described as ‘limbic encephalitis’ or ‘encephalitis’.
Of the 48 cases reporting behavioural or psychiatric symptoms, the most common symptom reported was depression (28 cases), followed by anxiety (17 cases), psychosis (15 cases), behavioural or personality changes (11 cases) and emotional lability (2 cases) (Supplementary Data).
GAD antibody–associated limbic encephalitis with psychosis
The cases reporting psychotic symptoms are detailed in Table 2. The mean age of presentation was 31.4 years (SD = 18.2) with a female-to-male ratio of 10:4; one case not reporting on individual case demographics. Visual hallucinations were the most common phenomena reported (five cases), followed by auditory hallucinations (four cases) and delusions (four cases), although symptoms were described only as ‘psychosis’ in six cases. All cases that reported on para-clinical investigations (11/15, 73.3%) had abnormal findings such as neuroimaging, electroencephalography (EEG) or CSF abnormalities.
GAD antibody-associated limbic encephalitis cases with psychotic symptoms.
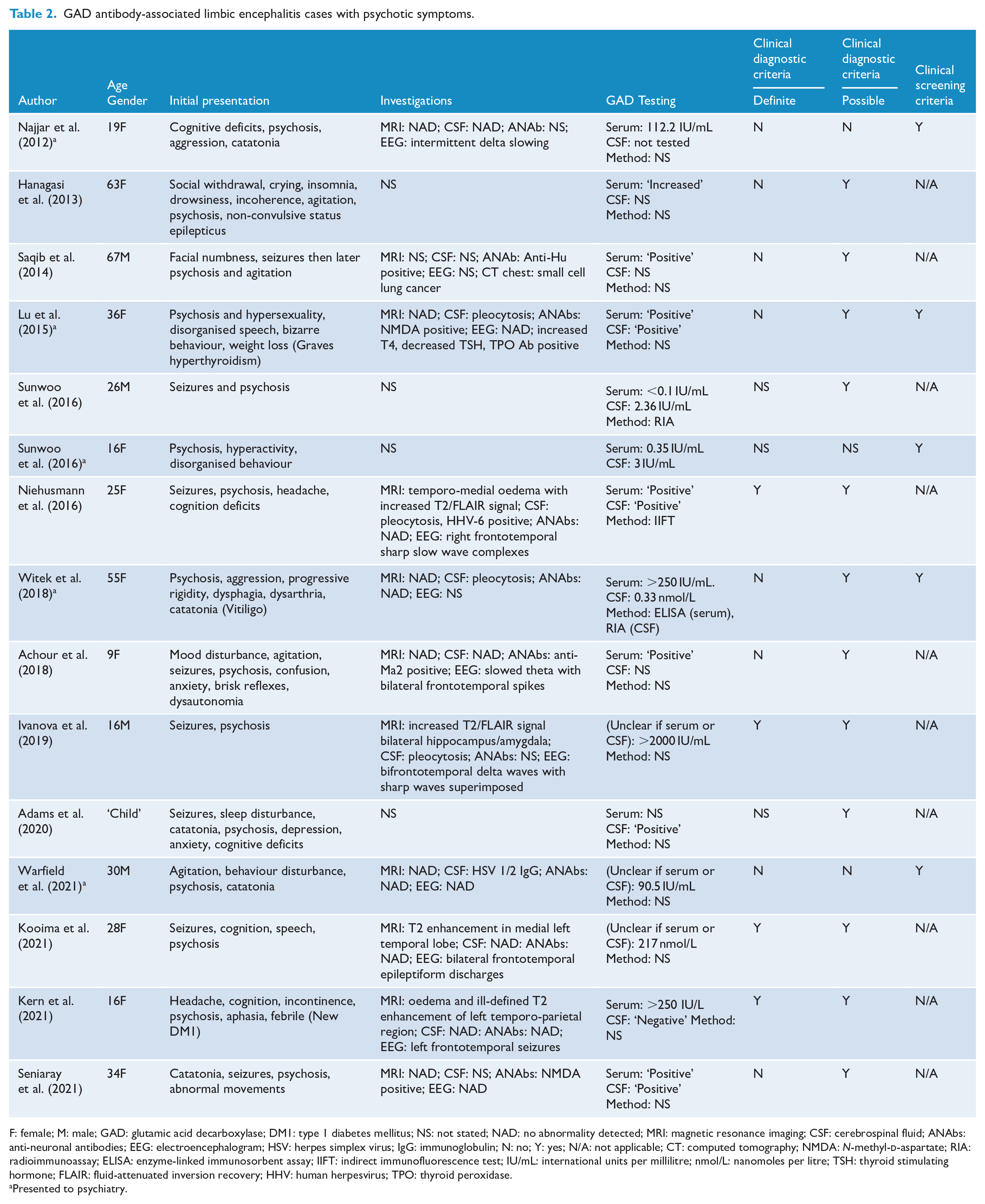
F: female; M: male; GAD: glutamic acid decarboxylase; DM1: type 1 diabetes mellitus; NS: not stated; NAD: no abnormality detected; MRI: magnetic resonance imaging; CSF: cerebrospinal fluid; ANAbs: anti-neuronal antibodies; EEG: electroencephalogram; HSV: herpes simplex virus; IgG: immunoglobulin; N: no; Y: yes; N/A: not applicable; CT: computed tomography; NMDA: N-methyl-
Presented to psychiatry.
The majority of cases (10/15, 66.7%) initially presented with neurological symptoms with psychotic phenomena occurring in the context of seizures. There were five cases that presented initially with psychotic symptoms. One case that presented with psychosis was also diagnosed with both anti-NMDA receptor encephalitis and Grave’s Disease, and one case followed a herpes simplex virus encephalitis. No case that presented to psychiatry had a GAD antibody titre considered in the ‘high abnormal value’ (>1000 IU/mL). All cases that presented with psychotic symptoms would have been screened as positive using the ‘Clinical Screening Criteria’.
Discussion
This is the first systematic review exploring the role of GAD antibodies in psychosis. GAD antibodies were uncommonly found (less than 2%) in cohorts with psychiatric disorders and did not differ in prevalence between cohort and control groups. No cases of encephalitis, CSF antibody positive or high antibody titre was found in >2700 individuals with psychosis. Similarly, psychotic symptoms appear to be uncommon in cases of GAD antibody–associated encephalitis, occurring in less than 5%. When present, psychotic symptoms were the presenting feature in a third of cases, but all had non-psychiatric features that would be identified by clinical examination or established clinical screening criteria.
The current approach to testing for neuronal antibodies in psychiatry has been informed by the characteristics of anti-N-methyl-
Alternatively, GAD antibodies may serve a role as a biomarker for CNS inflammation or are present due to systemic autoimmunity, in particular Type 1 diabetes (Graus et al., 2020). A history or testing for diabetes and other systemic autoimmune disorders were frequently not documented in the psychosis cohorts. This is an important consideration, given the prevalence of these disorders has been shown to be up to 50% higher in those with a known psychotic disorder and may explain the trend seen in the meta-analysis (Benros et al., 2011; Eaton et al., 2006). Interestingly, GAD antibody titres typically decrease over time when associated with diabetes, whereas they remain elevated in neurological disorders, indicating utility of repeated GAD antibody testing to assist in differentiating complex cases (Nakajima et al., 2018).
There are inherent limitations that are important to consider when interpreting the results of this review. In the psychiatric cohorts tested for GAD antibodies the method of testing has varied over time, resulting in heterogeneous measurement techniques. The cell-based assay and immunohistochemistry are thought to better detect GAD antibodies of higher titres and are more relevant for autoimmune-related neurological disease, whereas positive results from radioimmunoassay (RIA) or ELISA testing are more likely to be associated with diabetes and systemic autoimmunity (Hoffmann et al., 2021). Studies using cell-based assay demonstrate very low rates of GAD antibody positivity (0.2%), lower proportion in psychosis cases (1/724) than controls (1/338). There was only one study which assessed CSF antibodies in all participants. This likely reflects what is feasible to achieve in research studies on clinical populations.
The cases of GAD antibody–associated limbic encephalitis were retrospectively identified with publication bias potentially increasing the number of more clinically challenging cases. Additionally, although duplicates were removed, there may be an over-estimation of case numbers if case reports were included in case series, which is relevant for such a rare disorder. Data from cases could only be gathered from what was reported and it was felt this may particularly impact the details of psychiatric symptoms as many of the cases were published by neurology or immunology specialities. There has recently been a recommendation that cases without CSF antibody confirmation should not be considered an autoimmune GAD antibody–related disorder, which may impact the five cases in which CSF antibodies were either not tested for or demonstrated in Graus et al. (2020).
GAD antibody–associated encephalitis appears to be rare in cohorts of people with psychosis, and screening all individuals with first episode of psychosis for GAD antibodies does not seem justified. This aligns with research supporting the recommendation for targeted rather than routine screening in FEP (Cohn et al., 2021; Theorell et al., 2021). We propose adapting existing screening guidelines to include modified clinical screening criteria and removing routine serological testing for GAD antibodies. Where encephalitis is suspected, thorough evaluation including neurology review, CSF analysis and imaging should be considered. Further prospective studies may be beneficial to comment on the utility and cost–benefit analysis of clinical screening to identify high pre-test probability cases.
Supplemental Material
sj-docx-1-anp-10.1177_00048674221089560 – Supplemental material for Anti-glutamic acid decarboxylase antibody screening in first-episode psychosis
Supplemental material, sj-docx-1-anp-10.1177_00048674221089560 for Anti-glutamic acid decarboxylase antibody screening in first-episode psychosis by Nicola Warren, Karen Freier, Dan Siskind, Cullen O’Gorman, Stefan Blum, David Gillis and James G Scott in Australian & New Zealand Journal of Psychiatry
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: N.W. receives speaker fees from Otsuka, Lundbeck and Janssen. D.S. is funded in part by NHMRC Emerging Leadership Fellowship GNT1194635. Employers of JGS (QIMR Berghofer Medical Research Institute and The University of Queensland) have received honoraria from Lundbeck, Janssen Cilag and Otsuka for his speaking at educational events and/or advisory board participation. For the remaining authors, no conflicts of interest were declared.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
