Abstract
Objective:
Younger-onset dementia accounts for about 5–10% of all dementias in Australia. Little data is available on neuropsychiatric symptoms in people with younger-onset dementia compared to those with older-onset dementia. This study aims to compare the types of neuropsychiatric symptoms and their clinico-demographic characteristics of people with younger-onset dementia and older-onset dementia who are referred to a specific dementia support service.
Methods:
A 2-year retrospective observational cross-sectional analysis was undertaken on referrals with neuropsychiatric symptoms from Dementia Support Australia programmes. Neuropsychiatric symptoms were measured using the Neuropsychiatric Inventory total severity scores and distress scores. Contributing factors to neuropsychiatric symptoms for dementia groups were examined. Logistic regression was used to examine the relationship between individual neuropsychiatric symptoms and having older-onset dementia vs younger-onset dementia.
Results:
Of the 15,952 referrals, about 5% (
Conclusion:
Clinico-demographics of referrals with younger-onset dementia differ from those with older-onset dementia. There were some differences in the characteristics of neuropsychiatric symptoms between younger-onset dementia and older-onset dementia. Our findings have implications for service provision and support for people with dementia at different ages.
Keywords
Introduction
Younger-onset dementia (YOD) refers to a dementia where symptom onset occurs less than 65 years old (Rossor et al., 2010). YOD accounts for about 5–10% of all dementias in Australia with older-onset dementia (OOD; onset more than 65 years old), making up the vast majority of dementia cases (Brown et al., 2017; Draper and Withall, 2016). Neuropsychiatric symptoms (NPS), or the non-cognitive symptoms related to dementia including behaviours such as apathy, psychotic symptoms and agitation, occur in almost all people who have dementia (Lyketsos et al., 2000; Mulders et al., 2014) and can contribute to significant burden and adverse mental health in carers (Bakker et al., 2013b; Koyama et al., 2017), poor quality of life for people living with dementia (Millenaar et al., 2017) and are a predictor of entry into residential care (Bakker et al., 2013a; Balestreri et al., 2000).
Few studies have investigated differences in the frequency and the types of NPS according to age of onset. For people with dementia living in the community, it has been reported that there are fewer NPS in people with older-onset Alzheimer’s dementia (AD) compared to those with younger-onset AD, including fewer psychotic symptoms and less agitation, disinhibition and aberrant motor activity (Baillon et al., 2019; Van Vliet et al., 2012). A study of NPS in people with OOD and YOD living in residential aged care (RAC) found that people with YOD had higher levels of apathy whereas people with OOD had higher levels of agitation (Appelhof et al., 2019). A recent study reported that for both community-dwelling and those in RAC, people with YOD had higher frequencies of NPS, namely depression, anxiety and psychotic symptoms (Ryan et al., 2021). There is less information regarding the differential impact of NPS in OOD and YOD on carers. Carers who support people with YOD may have more distress and difficulties managing NPS compared to those who look after people with OOD (Arai et al., 2007; Baillon et al., 2019; Ryan et al., 2021).
NPS may reflect a response of a person to their unmet needs (e.g. thirst), an organic aetiology associated with their dementia (e.g. white matter lesions), or a combination of these factors (Macfarlane et al., 2021b). Identifying mechanisms of such NPS precipitants is vital in informing potential targets of intervention, especially as unmet needs (including pain, temperature sensitivity or psychological distress) are often highly modifiable through non-pharmacological strategies (Bakker et al., 2014; Cohen-Mansfield and Mintzer, 2005). In YOD, higher levels of unmet need may be associated with higher frequency of NPS (Bakker et al., 2014).
There is little Australian-based information comparing NPS in people with YOD and OOD and how carers respond to these. This study aims to report on NPS in people with YOD and OOD who have been referred to a specific dementia support service which provides individualised non-pharmacological strategies for NPS. The major objective of the study was to compare the different types of NPS between the two groups and their clinico-demographic characteristics over a 24-month period. To address the study objectives, we set the following hypotheses for the two groups: (1) there will be differences in demographic characteristics; (2) there will be differences in the frequency and severity of individual types of NPS; (3) there will be differences in the overall frequency and severity of NPS and (4) there will be differences in the overall distress reported by the carers. We set the exploratory hypothesis that (5) there will be differences in the rates of the most common contributing factors to NPS.
Materials and methods
Ethical considerations
The study was approved by the University of New South Wales (UNSW) Human Research Committee (HC190049). A waiver of informed consent was granted for the study as the data were analysed retrospectively and anonymously. The data were handled by a data custodian who complied with the Declaration of Helsinki of ethical principles, and the National Health and Medical Research Council’s National Statement on Ethical Conduct in Human Research (2018).
Study design, context and reporting
This study was a 2-year retrospective observational cross-sectional study from 1 January 2019 to 31 December 2020 with data extracted from the Dementia Support Australia (DSA) database. The DSA database contains demographic and clinical data such as age and dementia subtype for referrals with NPS seeking support from two national programmes, the Dementia Behaviour Management Advisory Service (DBMAS) for mild to moderate NPS and the Severe Behaviour Response Teams (SBRT) for severe NPS (Macfarlane et al., 2021a). These correspond to tiers 3-4 and tiers 5-6, respectively, of the Brodaty pyramid of service provision (Brodaty et al., 2003).
DSA has been responsible for the provision of DBMAS and SBRT programmes since 2016. Both programs are 24/7 national dementia-specific behaviour support services funded by the Australian Government to provide free multimodal person-centred psychosocial/non-pharmacological interventions for referrals with dementia-related NPS and their formal (staff at RAC) and informal (family) carers, in line with current best practice principles (Macfarlane et al., 2021a). The programmes are delivered by a large team of dementia consultants, geriatricians, psychogeriatricians and support staff. Dementia consultants are members of a multidisciplinary team including, but not limited to, nurses, occupational therapists, physiotherapists, social workers, psychologists, dieticians, speech pathologists and diversional therapists with accredited expertise and training in dementia and behaviour support (Macfarlane et al., 2021a).
Both DBMAS and SBRT utilise a case management framework (CMF) to ensure consistency and quality in the provision of service (Macfarlane et al., 2021a). This CMF includes a comprehensive onsite assessment with the client and carer/s to identify possible causes of NPS, the provision of detailed non-pharmacological support strategies and recommendations, and ongoing visits and/or correspondence with the referrer to assess the impacts of the interventions. Typically, DSA programmes receive referrals via a dedicated phone line, website or email from various care settings, including residential and community care (Macfarlane et al., 2021a).
Eligibility criteria for receiving support from DSA includes (1) having a confirmed or probable diagnosis of dementia; (2) exhibiting NPS related to the diagnosis of dementia; (3) exhibiting NPS that impact the care and well-being of individuals with living dementia and/or their carers; and (4) receiving consent from the person with dementia or their responsible carer (Macfarlane et al., 2021a).
For further details on DSA programmes and their impact, please see Macfarlane et al. (2021a).
International guidelines recommend that the first-line treatment of NPS should be non-pharmacological interventions (Ayalon et al., 2006; O’Connor et al., 2009). The clinical effectiveness of DSA programme-based interventions for NPS was recently demonstrated through improvement in NPS and associated carer distress in a large nationally representative sample (
We used the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement Checklist to guide our study reporting (Von Elm et al., 2007).
Study instruments
The Neuropsychiatric Inventory (NPI)
The Neuropsychiatric Inventory Nursing Home version (NPI-NH) (Wood et al., 2000) and the Neuropsychiatric Inventory Questionnaire (NPI-Q) (Kaufer et al., 2000) are valid, consistent and reliable tools which assess 12 domains of NPS such as delusions, agitation, depression and disinhibition (Kaufer et al., 2000; Wood et al., 2000). Each domain is rated on the presence, severity (ranging from ‘mild’ [0] to ‘severe’ [3]) and the distress caused to carers (ranging from ‘not at all’ [0] to ‘very severely’ [5]), whether formal or informal (Kaufer et al., 2000; Wood et al., 2000). The NPI-NH also measures the frequency of occurrence of each domain, ranging from ‘rarely’ (1) to ‘very often’ (4) (Wood et al., 2000). For each domain, the frequency scores and severity scores are multiplied (range, 0–12). These scores are cumulative so that a total frequency × severity (denoted ‘total score’) and total disruptive score (denoted ‘total disrupt’) can be calculated. Higher scores indicate more severe, more frequent and more disruptive and distressing behaviours. The NPI is administered by the DSA staff at each consultation. Administering which version of NPI to referrals depends on the DSA programme involved. DBMAS referrals receive the NPI-Q whereas SBRT referrals receive NPI-NH version (Macfarlane et al. 2021a). In general, if a person with dementia who is referred resides in RAC, a formal carer would complete the NPI-NH version and if a person with dementia who is referred resides in the community, an informal/family carer would complete the NPI-Q version.
Contributing factors
A fundamental component of the DSA model of care is the identification and modification of antecedents, or contributing factors, of NPS. Contributing factors are numerous and broadly include causes of NPS that are biological (e.g. untreated pain, constipation), environmental (e.g. temperature/light), social (e.g. carer approach) or psychological (e.g. relocation stress). Contributing factors are identified through a detailed and extensive onsite assessment with the person living with dementia (i.e. the referral) and their formal or informal carer/s, a review of medical and social history and the use of well-validated instruments (such as PainChek®) (Macfarlane et al., 2021a). These involve DSA consultants auditing medical records, conducting clinical observations and interviews, and collecting proxy assessments from people (e.g. nursing staff, family members) who had regular contact with the referral (Macfarlane et al., 2021a).
Pain was assessed using PainChek®, a valid and reliable regulatory cleared pain assessment medical device in the form of a point-of-care app for people living with cognitive impairment including those living with dementia (Atee et al., 2017a, 2018). The app has a 42-item pain scale that are distributed over six domains: Face (9 items), Voice, (9 items), Movement (7 items), Behaviour (7 items), Activity (4 items) and Body (6 items). PainChek® scores above 6 (i.e. ⩾7) indicate the presence of pain (Atee et al., 2017a, 2017b, 2018).
For the current study, differences in the prevalence of the most frequent contributing factors for each group were investigated.
Inclusion and exclusion criteria of participants for statistical analyses
Participants were eligible for statistical analyses if they met the following criteria: (1) referred to any of the DSA programmes (namely, DBMAS or SBRT) during the 2-year period of the study and (2) a referral was considered to be YOD if they were 65 or younger at the time of their first referral to DSA. This includes any referrals that occurred prior to the study period (i.e. if someone was 64 when they were referred in 2018, they were considered to be YOD if they were also referred at any time during the study period); and (3) when a referral was supported multiple times during the study period, only their earliest referral was used.
Referrals were excluded from the analyses if any of the above criteria were not met.
Statistical analyses
The demographic variables for sex, referral state, dementia subtype, referral location (the setting at which the person was residing at the time of referral to DSA programmes) and source of referral (the sector from which the referral was made to DSA programmes) were compared between OOD and YOD using Pearson’s chi-square test. Cramer’s
The difference in demographic characteristics of referrals with completed and non-completed NPI assessments for both groups was also reported (see Supplemental Materials Table S1).
The average total NPI severity, distress and number of domains were compared between groups using Welch’s
Statistical analyses were completed using R version 4.1.1 (R Core Team, 2021).
Results
Demographics
Table 1 shows comparisons between OOD and YOD on demographic characteristics. Over the study period, a sample of 15,952 referrals to DSA were eligible for the analysis. Of these referrals, approximately 5% (
Demographics of the study sample for OOD and YOD.
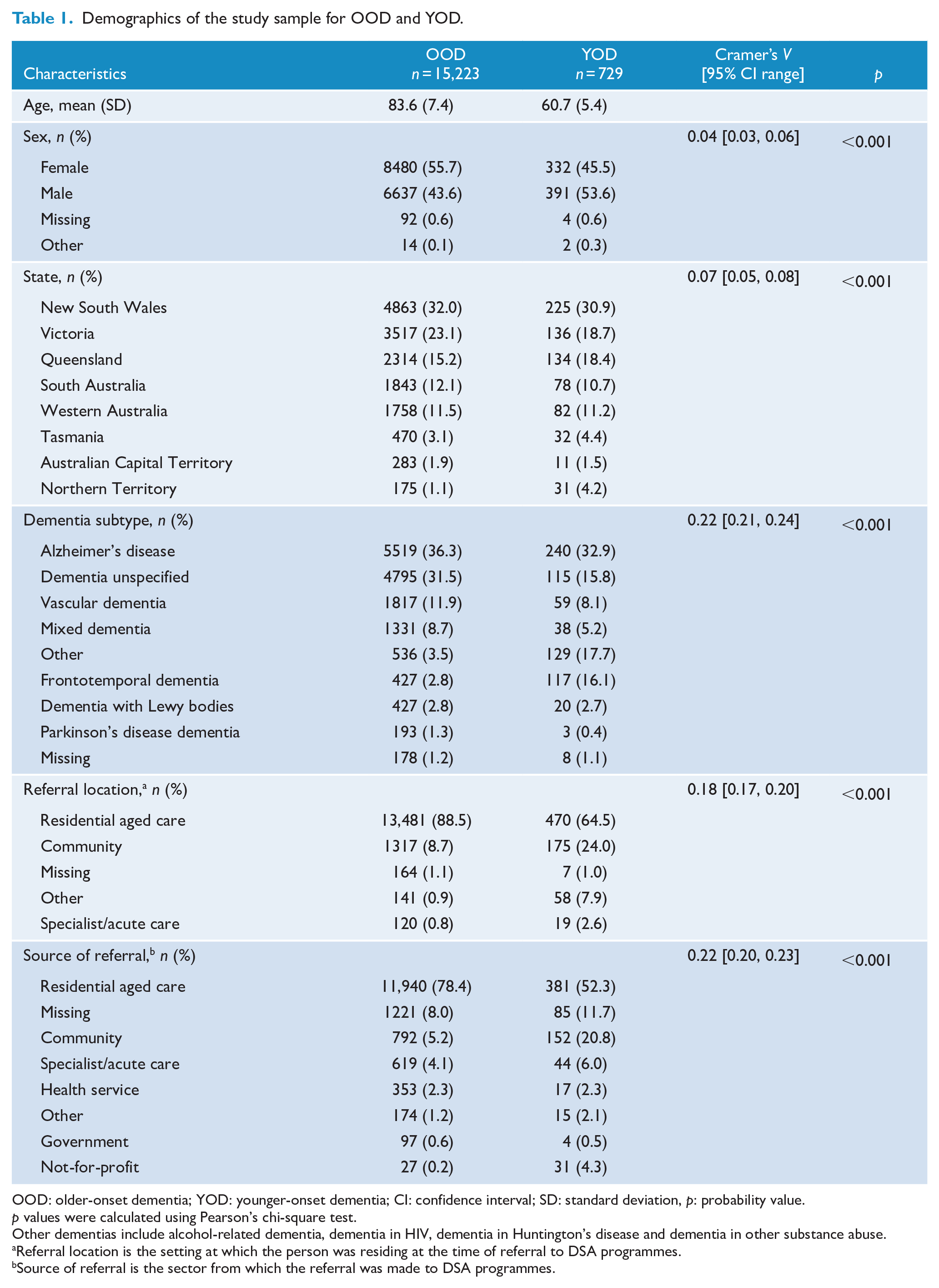
OOD: older-onset dementia; YOD: younger-onset dementia; CI: confidence interval; SD: standard deviation,
Other dementias include alcohol-related dementia, dementia in HIV, dementia in Huntington’s disease and dementia in other substance abuse.
Referral location is the setting at which the person was residing at the time of referral to DSA programmes.
Source of referral is the sector from which the referral was made to DSA programmes.
Neuropsychiatric symptoms
NPS demonstrated no differences in the number of NPI domains, frequency, total severity or total distress scores between those with YOD and OOD. Tables 2 and 3 and Figure 1 show details from NPI domain scores for YOD and OOD. Mean frequencies for the most common NPS for both groups (OOD vs YOD) were agitation/aggression (87.1% vs 88.0%), irritability/lability (62.9% vs 58.5%), anxiety (57.4% vs 60.5%) and depression/dysphoria (58.6% vs 56.3%). Please see Supplemental Materials (Table S1) for the differences in demographics for those with completed and non-completed NPI assessments for referrals with OOD and those with YOD.
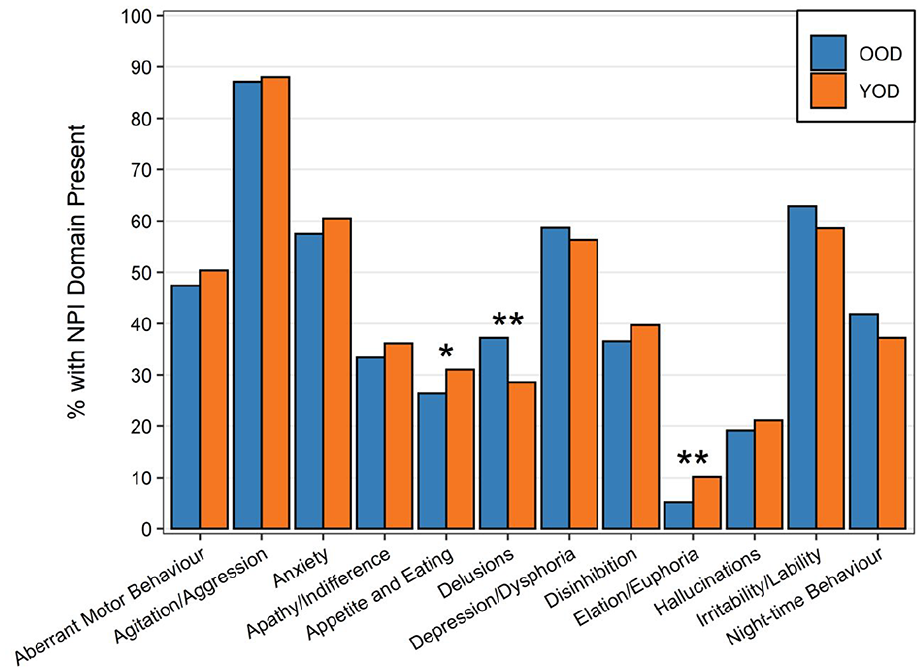
Frequency of NPI domains for YOD and OOD.
Differences in NPI metrics between OOD and YOD.
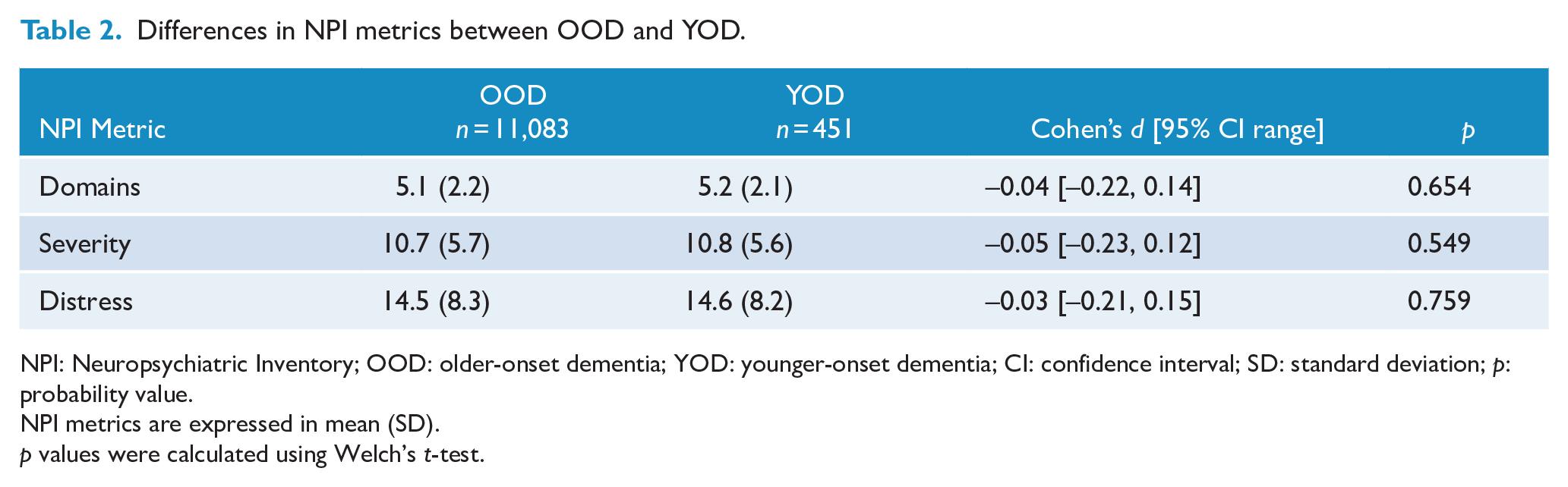
NPI: Neuropsychiatric Inventory; OOD: older-onset dementia; YOD: younger-onset dementia; CI: confidence interval; SD: standard deviation;
NPI metrics are expressed in mean (SD).
Descriptive statistics (mean and SD) for NPI domain severity scores for referrals with YOD and OOD.
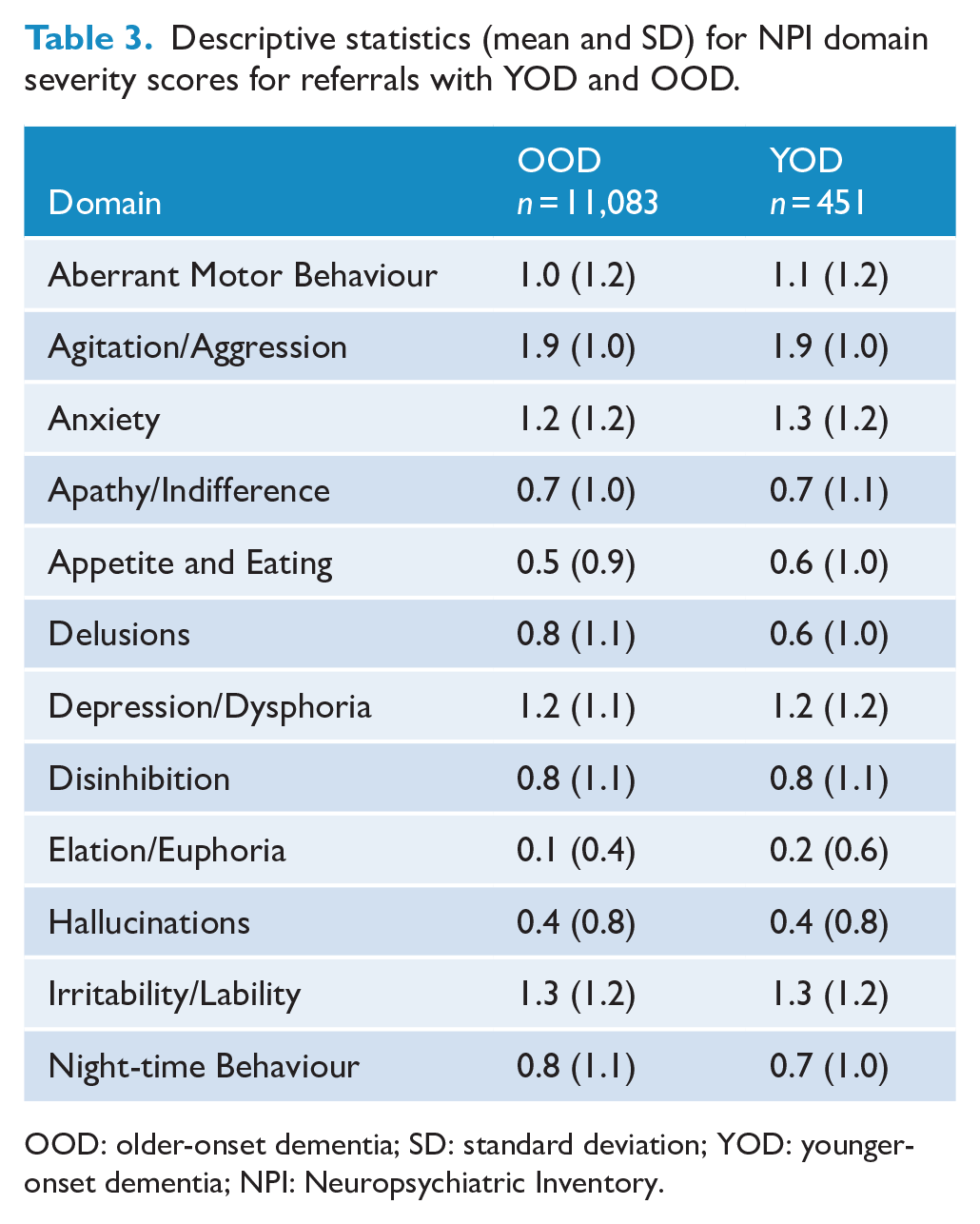
OOD: older-onset dementia; SD: standard deviation; YOD: younger-onset dementia; NPI: Neuropsychiatric Inventory.
The logistic regression examined the relationship between individual NPS and having OOD vs YOD (Table 4). There were significant effects for appetite and eating, irritability/lability, and night-time behaviour and larger effects for both delusions and elation/euphoria. For every 1-point increase in elation/euphoria severity, it was 1.49 times more likely that a referral was a person with YOD. Conversely, for every 1-point increase in delusions, it was 1.27 times more likely the person had OOD.
Coefficients for the logistic regression examining the association between NPI domain symptom severity and presence of OOD vs YOD.
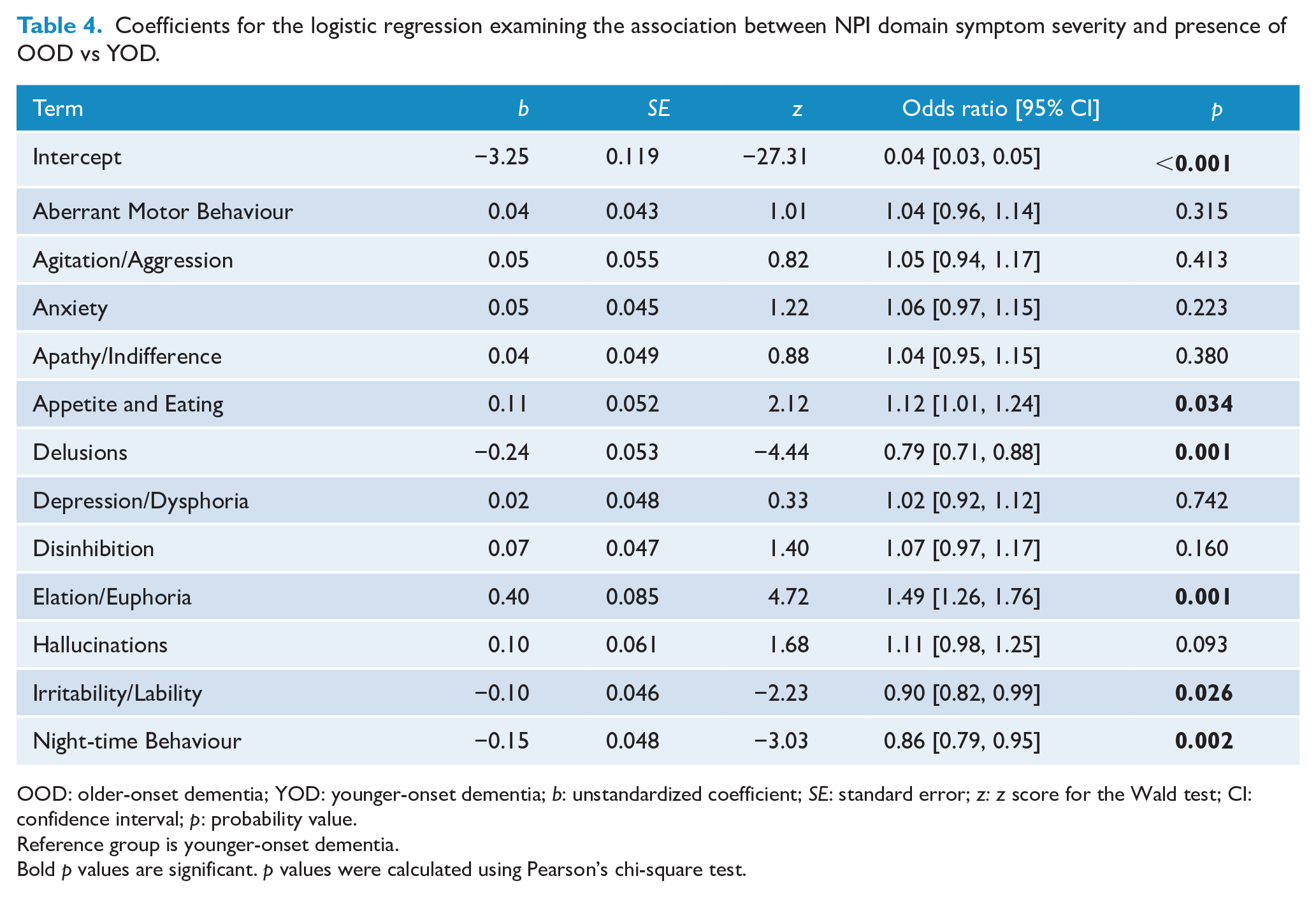
OOD: older-onset dementia; YOD: younger-onset dementia;
Reference group is younger-onset dementia.
Bold
Contributing factors
Pain, carer approach, mood disorders and over/under stimulation were among the most common contributing factors identified for both groups (Table 5). In addition, for OOD, loneliness/boredom was a top five contributing factor, and for YOD, communication difficulties were in the top five. There were significant effects (two-sample
Common contributing factors to neuropsychiatric symptoms of YOD and OOD.
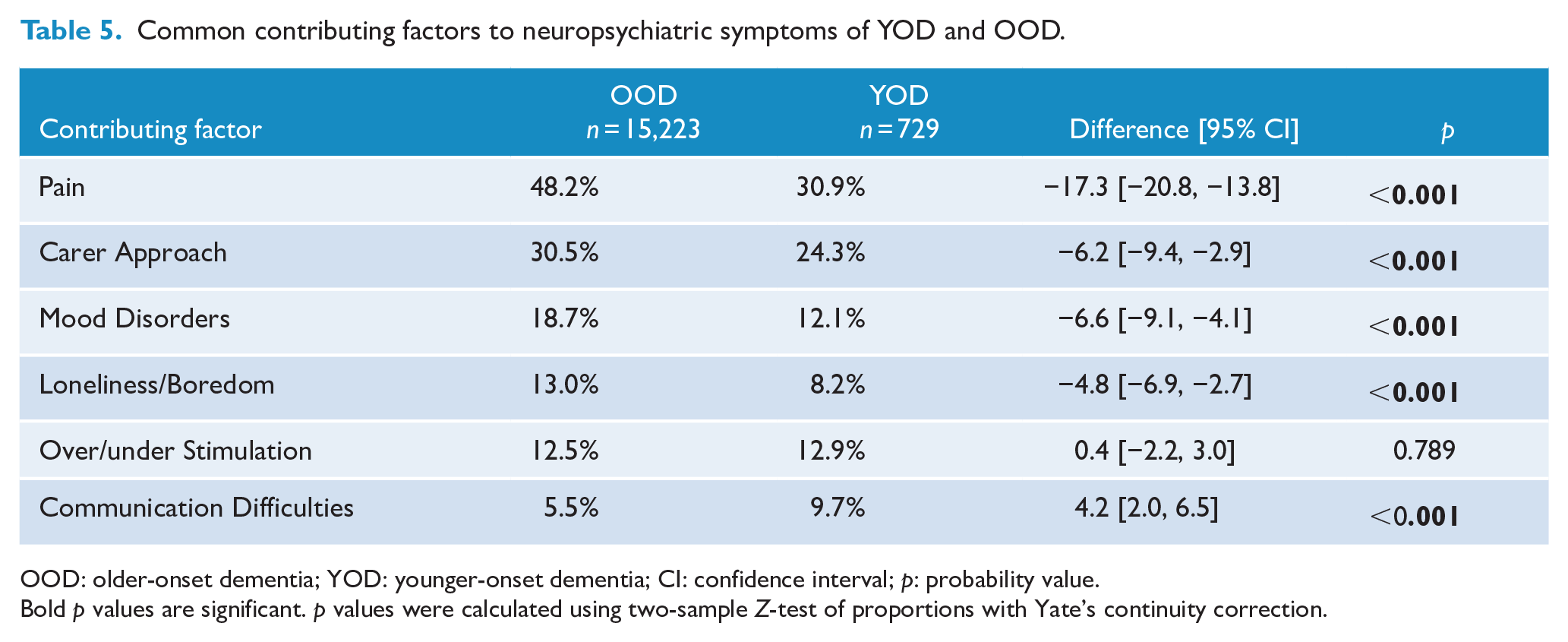
OOD: older-onset dementia; YOD: younger-onset dementia; CI: confidence interval;
Bold
Discussion
This study compared the demographic and clinical characteristics (e.g. types of NPS) in people with OOD and YOD who were referred to an Australian dementia support service. Our first hypothesis was confirmed in that there were differences in demographics between the two groups. We found no differences in the total severity and distress scores between the two groups, thus refuting our third and fourth hypotheses. However, partially confirming our second hypothesis, we observed that the severity of some individual domains (i.e. NPS) was related to whether the individual had YOD or OOD. Our fifth hypothesis, relating to differences in contributing factors relating to NPS being different in those with OOD and YOD, was also confirmed.
Approximately 5% of all referrals were for people with YOD. This is comparable to current prevalence data on OOD and YOD in Australia (Dementia Australia, 2018) and internationally (Hendriks et al., 2021). There were differences based on YOD/OOD in terms of demographics, with more females referred who had an OOD and the opposite found for YOD, where males were more frequently referred. Not surprisingly, there were many more referrals for people with OOD who were living in RAC, and more people with YOD referred who were living in the community. These findings are consistent with Ryan et al. (2021). Our study also reported four times as many people with FTD who had a YOD, compared to OOD, with FTD being a common cause of YOD (Loi et al., 2020).
Neuropsychiatric symptoms
Our findings contrast to previous findings which have reported differences in NPS in people with YOD and OOD. The literature reports a higher prevalence of NPS in YOD and different types of NPS being exhibited, depending on the age of onset of dementia. For example, community-dwelling individuals with younger-onset AD had a higher frequency of all NPS (using the NPI) except apathy compared to individuals with older-onset AD (Baillon et al., 2019). Another study based in RAC focused on all dementias, and found that people with YOD had higher levels of apathy, but people with OOD had higher levels of agitation (Appelhof et al., 2019). Appelhof et al. (2019) reasoned that residential care staff were more likely to rate apathy as a symptom requiring management when it arose in younger residents with dementia, as staff may perceive that apathy or inactivity is to be expected in older residents with dementia. They also hypothesised that a proportion of the younger-onset residents had FTD, where apathy may be more prominent. Distinguishing between depression and apathy can be clinically challenging, with hypoactive delirium also being a differential diagnosis (Gleason, 2003). Furthermore, it is worth noting that certain behaviours seen in FTD such as impulsivity and disinhibition, expressed by younger, more physically robust male residents may be perceived as more threatening and thus more likely to generate a referral to external NPS support services, such as DSA programmes.
Similar to Appelhof et al. (2019), Ryan et al. (2021) reported different frequencies and types of NPS between individuals with YOD compared with those OOD, both in the community and in RAC. Individuals with YOD had higher rates of depression and psychotic symptoms, compared to people with OOD, regardless of where they lived. While these studies reported a significant difference in NPS between OOD and YOD, our study did not. This is possibly due to methodological variations, such as study design (e.g. cross-sectional vs longitudinal study), study setting (e.g. community vs RAC), sample size, sample characteristics (e.g. gender distribution) and neuropsychiatric assessment used (e.g. NPI).
Contributing factors and the predictive value of NPS
We found differences in the frequency of contributing factors to NPS, as rated by the staff working at DSA. Pain was more commonly rated as a contributing factor in individuals with OOD, while communication difficulties were more frequent in those with a YOD. This might be related to the over-representation of FTD in the YOD sample. Pain has been reported as a precipitant to NPS (Husebo et al., 2011), particularly in older adults with dementia (Atee et al., 2021) but it is often under-assessed in this older group (Kaiser et al., 2014; Mulders et al., 2016). There is less reported on contributing factors for NPS in YOD, but communication difficulties, mobility issues and lack of socialisation have been reported (Bakker et al., 2014). Differences in contributing factors to NPS depending on YOD or OOD are expected due to age-related differences such as physical comorbidities and frailty.
While we reported no differences in the types of NPS depending on the age of onset of dementia, we were able to show that 5 out of 12 NPI domains (appetite and eating, delusions, elation/euphoria, irritability and lability and night-time behaviour) were significantly associated whether a person had an OOD or YOD. The effect was large for delusions and elation/euphoria (Table 4). Specifically, we found that an individual presenting with delusions was more likely to have an OOD, compared to elation/euphoria, which was more indicative of a YOD. Psychotic symptoms as part of NPS are common in the early and moderate stages of dementia, regardless of whether it is younger-onset or older-onset (Ryan et al., 2021) but over time, delusions may be more prevalent for OOD (Van Vliet et al., 2012). Elation and euphoria are less frequent NPS overall but possibly may be more common in younger patients and in dementias such as FTD (Mukherjee et al., 2017), consistent with our own finding.
Carer distress
We found no differences in carer distress for carers looking after individuals with YOD or OOD. Arai et al. (2007) compared 14 people with YOD and 54 people with OOD residing in the community. There was no difference in dementia subtype, hours of caring and duration of illness; however, carers of people with YOD had higher levels of perceived difficulties due to the behavioural changes. Baillon et al. (2019) investigated community-dwelling carers and their associated distress in looking after people with YOD and OOD. While their sample size was larger than Arai et al. (2007) (
Strengths and limitations
This was a retrospective, descriptive and observational study that applied a cross-sectional analysis. Thus, the limitations of observational studies such as the lack of causation are relevant. In addition, there were limitations in the dementia information. This included a lack of consensus diagnostic criteria used to diagnose dementia subtypes. The classification of referrals as YOD or OOD was based on the age cut-off of 65 years at the time of their first referral to DSA. As such, it is possible that some referrals received a diagnosis of dementia prior to the age of 65 but were classified as OOD at the time of referral. This may have biased the results in favour of one of the groups. We did not have clinical information pertaining to the dementia severity stage such as the Global Deterioration Scale, to indicate if a dementia was early/mild, moderate or advanced stage and therefore we were unable to make any inferences with our findings according to the stage of dementia. As DSA is supporting the individual with dementia, there is minimal demographic information collected about the formal or informal carer and we were unable to report carer differences in YOD and OOD.
Not all referrals received an intake NPI assessment, with a higher rate of NPI completion recorded for OOD referrals (73%) compared to YOD referrals (62%). There were differences in completion rates of NPI depending on referral location and source of referral (Text S1, Table S1). The difference in the rates of NPI completion are partially due to the higher rates of YOD referrals living in the community, where both the carer and the person living with dementia are often present during the assessment and DSA staff may deem it inappropriate to complete the NPI.
Some of the contributing factors reported in the study were assessed using information obtained from clinical charts, medico-social and lifestyle history provided by proxies (i.e. aged care staff and/or family member). Therefore, identifying these factors was limited by the accuracy of documentation and proxy reporting/assessment; hence, the findings pertaining to such factors should be interpreted with caution. However, this was mitigated at least in part by the extensive training and significant experience of consultants in gleaning this information.
Both our study and the three studies which found certain types of NPS higher in YOD were of cross-sectional design, while longitudinal evaluations may be a more appropriate study design to provide a more accurate picture. Van Vliet et al. (2012) conducted a longitudinal study over 2 years and found higher levels of delusions, agitation, apathy and depression in those with OOD compared to those with YOD.
As well as methodological limitations, it is possible that the lack of differences in severity, frequency, distress and type of NPS seen in our study may be because the majority of the referrals of the younger-onset referrals were still living in the community. Agitation/aggression, irritability/lability, depression/dysphoria and anxiety were the most common NPS that were referred to DSA and these were of similar frequency across the YOD and OOD groups. These NPS are generally very common in the early and moderate stages of OOD (Lyketsos et al., 2000). For YOD, affective NPS and agitation may be seen in the early stages (Kaiser et al., 2014; Mulders et al., 2016) and aggression, agitation and apathy seen in the moderate and severe stages, in people with YOD living in RAC (Mulders et al., 2016). While our YOD sample contained four times the number of people with a FTD, the frequency of apathy was similar across OOD and YOD. Regardless of dementia subtype, apathy remains a significant cause of distress and burden for carers (Hiyoshi-Taniguchi et al., 2018; Zwijsen et al., 2014). Overall, these limitations may reduce the generalisability of the findings.
Our study has several significant strengths. This is one of the largest population-based studies that describes the clinico-demographic characteristics of a large YOD sample (
Conclusion
This large representative study investigated clinico-demographic differences between individuals who had OOD and YOD referred for support for NPS. There were more referrals for OOD who were females and from RAC, and more referrals for YOD in males and from the community. Within the YOD sample, there were many more referrals of people who had FTD. The type of NPS was similar between OOD and YOD, with similar levels of severity and similar levels of distress reported by the carers. Agitation, aggression, irritability, anxiety and depression were common NPS which warranted support from DSA. Contributing factors to NPS were different, with pain being more frequently endorsed for individuals with OOD and communication difficulties for those with YOD. Further investigation into the different types of NPS which required support according to gender and type of dementia would yield important Australian-based information which is currently lacking. Particularly, there is little known about the experiences of people in YOD who live in RAC, their quality of life and NPS. In addition, exploration of the pathways to assessment and care of NPS for people with YOD has also been minimally explored. This research is needed to better guide service provision for management of NPS, especially for people with YOD.
Supplemental Material
sj-docx-1-anp-10.1177_00048674221080709 – Supplemental material for Clinico-demographics of people with younger-onset dementia and neuropsychiatric symptoms referred to an Australian dementia support service: A comparison study with older-onset dementia
Supplemental material, sj-docx-1-anp-10.1177_00048674221080709 for Clinico-demographics of people with younger-onset dementia and neuropsychiatric symptoms referred to an Australian dementia support service: A comparison study with older-onset dementia by Samantha M Loi, Mustafa Atee, Thomas Morris, Daniel Whiting, Stephen Macfarlane, Colm Cunningham and Dennis Velakoulis in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
We would like to acknowledge the patients and families who have been referred to Dementia Support Australia (DSA) programmes.
Author Contributions
S.M.L., M.A., T.M. and D.W. drafted the first version of the manuscript. S.M.L. and M.A. conducted the literature review. D.W. processed the data and performed statistical analyses. S.M.L., M.A., T.M. and D.W. contributed to acquisition, analysis or interpretation of data. S.M.L., M.A., T.M., D.W. and S.M. contributed to preparation of manuscript. All authors contributed to study concept and design and critical revision of the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: M.A., T.M., D.W., S.M. and C.C. are staff members of The Dementia Centre; a research, education and consultancy arm of HammondCare, an independent Christian charity which auspices the DSA programmes – DBMAS and SBRT. M.A. is one of the originators of the PainChek instrument, which is marketed by PainChek Ltd. (ASX: PCK). He is also a shareholder of PainChek Ltd. He previously held the position of a Senior Research Scientist (October 2018–May 2020) at PainChek Ltd. and is currently serving the position of Research and Practice Lead at The Dementia Center. He had a granted patent titled ‘A pain assessment method and system; PCT/AU2015/000501’ in Australia, China, Japan and the United States, which was assigned to PainChek Ltd. S.M. is the Head of Clinical Services for The Dementia Centre, HammondCare. C.C. is the Director of The Dementia Centre, HammondCare. Other authors declare no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: S.M.L. is funded by a National Health and Medical Council Early Career Fellowship (GNT1138968) and the Yulgilbar Alzheimer’s Research Program. DSA programmes are funded by the Australian Government (Department of Health) under the Dementia Aged Care Services (DACS) Fund since 2016. The funders had no role in the design and conduct of the study; collection, management, analysis and interpretation of the data; preparation, review or approval of the manuscript; and decision to submit the manuscript for publication.
Data Availability Statement
All data used in this article will remain confidential to comply with the conditions of service provision of Dementia Support Australia (DSA).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
