Abstract
Objectives:
Survival information in dementia is important for future planning and service provision. There have been limited Australian data investigating survival duration and risk factors associated with mortality in younger-onset dementia.
Methods:
This was a cross-sectional retrospective study investigating survival in inpatients with a diagnosis of dementia admitted to a tertiary neuropsychiatry service from 1991 to 2014. The Australian Institute of Health and Welfare National Death Index was used to obtain mortality information.
Results:
A total of 468 inpatients were identified, of which 75% had symptom onset at ⩽65 years of age (defined as younger-onset dementia). Dementia was categorised into four subtypes, Alzheimer’s dementia, frontotemporal dementia, vascular dementia and other dementias; 72% of the patients had died. Overall median survival duration was 10.6 years with no significant differences in duration within the dementia subtypes (
Conclusion:
This is the largest Australian study to date investigating survival and risk factors to mortality in dementia. We report important clinical information to patients with dementia and their families about prognosis which will assist with future planning. Our findings suggest that for both older-onset dementia and younger-onset dementia, ‘new onset’ psychiatric symptoms precede the cognitive symptoms of a neurodegenerative process. This, and sex differences in survival depending on the age of onset of the dementia warrant further investigation.
Introduction
Dementia was the second leading cause of death in Australia in 2017 (Australian Institute of Health and Welfare [AIHW], 2019). Younger-onset dementia (YOD) is a dementia of which symptom onset is at 65 years or less (Rossor et al., 2010) and prevalence varies from 51.7 per 100,000 (Ikejima et al., 2009) to 68.2 per 100,000 (Withall et al., 2014). The commonest causes of YOD are Alzheimer’s dementia (AD) and frontotemporal lobar degeneration, including behavioural-variant frontotemporal dementia (bvFTD; Harvey et al., 2003; Loi et al., 2021). While survival and mortality risk factors have been described in patients with older-onset dementia (OOD; symptom onset at >65 years old; Brodaty et al., 2012; Guehne et al., 2005; Todd et al., 2013), survival and mortality in YOD is limited to a small number of studies. This information is crucial for future planning and service provision.
From age of onset, frontotemporal dementia (FTD) with motor neuron disease (MND) has the shortest survival of all YODs, at 2.4 years (Hodges et al., 2003), while the survival in FTD without MND has ranged from 8.7 (Roberson et al., 2005) to 16 years (Gerritsen et al., 2019). Survival for younger-onset AD has more consistently been reported between 12 and 13 years (Gerritsen et al., 2019; Go et al., 2013), whereas for younger-onset vascular dementia (VaD), a larger range of survival duration has been reported, with one study describing approximately 7 years from diagnosis (Kay et al., 2000), compared to a more recent study which found survival of 22.5 years from onset (Gerritsen et al., 2019). Survival duration in YOD compared to OOD, it remains unclear as to whether OOD has shorter survival times compared to YOD (Koedam et al., 2008) or whether survival times are comparable (Rhodius-Meester et al., 2019).
Risk factors contributing to mortality in YOD are similar to those found in OOD, including more severe cognition (Go et al., 2013; Koedam et al., 2008; Roberson et al., 2005), worse functioning such as in activities of daily living (Go et al., 2013; Koedam et al., 2008) and the presence of diabetes (Go et al., 2013). Few studies have examined whether comorbidities or the clinical presentation such as cognitive or psychiatric are risk factors, which is relevant to YOD, as YOD often presents with non-cognitive symptoms such as psychiatric presentation (Perry et al., 2019; Woolley et al., 2011). Depressive symptoms during the course of OOD (Ganguli et al., 2005; Larson et al., 2004) have been examined with Ganguli et al. (2005) identifying depressive symptoms as a significant risk factor. Australian data are limited to only one study reporting on younger-onset AD and FTD, and people with FTD had a shorter survival time from age at onset, with cardiovascular risk factors (CVRFs) possibly explaining these differences (Atkins et al., 2012) with no studies investigating initial presenting symptoms.
The objective of this study was to investigate survival in patients with YOD and OOD. First, we compared survival duration in YOD and OOD as well as in different dementia types. Second, we sought to identify risk factors associated with mortality, including comorbidities and initial presenting symptoms.
Methods
Setting
This was a retrospective cross-sectional study of patients of Neuropsychiatry, Royal Melbourne Hospital, located in metropolitan Victoria, Australia. Neuropsychiatry is a tertiary specialist referral centre and provides assessment, diagnosis and follow-up of people with a range of neuropsychiatric conditions, including dementia. Patients are admitted for a comprehensive multidisciplinary assessment that includes clinical assessments (neuropsychiatry, neurology, allied health and nursing) and investigations (structural and functional imaging, lumbar puncture and blood tests). Multidisciplinary reviews are held weekly, and diagnoses are made using consensus criteria where available for bvFTD (Rascovsky et al., 2011), AD (McKhann et al., 2011) and dementia (Diagnostic and Statistical Manual of Mental Disorders [DSM]-IV). A comprehensive summary is provided to referring clinicians following discharge which incorporates all clinical and investigational assessments.
Patient information
All Neuropsychiatry inpatient summaries for the period 1991–2014 were reviewed, and all patients with any dementia diagnosis were identified. Diagnoses were categorised as AD, FTD (including bvFTD and language-variant FTD and FTD-MND) and VaD (including mixed AD and VaD). We excluded people who had a known diagnosis of Huntington’s disease (HD) and analysed this group separately. All other dementia diagnoses were categorised as ‘Other dementia’ such as dementia with Lewy bodies (DLB) and progressive supranuclear palsy (PSP). This category also included patients who had had a primary psychiatric diagnosis with associated dementia and those with no specific aetiology found (‘dementia not otherwise specified’).
Age of illness onset was defined as the age at when the earliest symptom of the dementia occurred, obtained from the patient and caregiver. Initial presenting symptom information was categorised as cognitive (such as memory, spatial or language impairment or disorientation), psychiatric/behavioural (such as depressive or psychotic symptoms or personality change) and neurological (such as weakness or tremor).YOD was defined when symptom onset occurred at <65 years of age, while OOD was defined as symptom onset after 65 years of age.
The Neuropsychiatry Unit Cognitive Assessment Tool (NUCOG) was used to assess the cognitive domains of attention, visuospatial, memory, executive and language functions, with higher scores indicating better cognition (Walterfang et al., 2006). The Global Assessment of Functioning (GAF) rates function from 0 to 100, with higher scores indicating better function (Endicott et al., 1976). As well as with details of previous psychiatric history, we measured comorbidities including history of alcohol use; family history of dementia and neurological disorders; and the presence or absence of CVRFs. Demographics including sex, education level and ethnicity were included in the analyses.
This study was approved by the Melbourne Health (2016.038) and the AIHW human research ethics committees (EO2017/5/398), which approved a waiver of consent for this retrospective study.
Identification of death
All deaths in Australia are compiled by the AIHW via the Australian National Death Index (NDI). The NDI contains information relating to all deaths after 1980. The final day of follow-up for this linkage was 30 September 2019.
Statistical analysis
Event time was defined as the time of death. Survival time was defined as time from symptom onset until the date of death. The Kaplan–Meier method was used to estimate the mean and median survival from age of onset. Survival curves between YOD and OOD and the dementia types were compared using log rank test. Cox proportional hazards model was used to analyse independent predictors of survival – YOD/OOD, sex, dementia type, alcohol use, psychiatric history and presenting symptoms. The significance level was set at
Results
Demographics
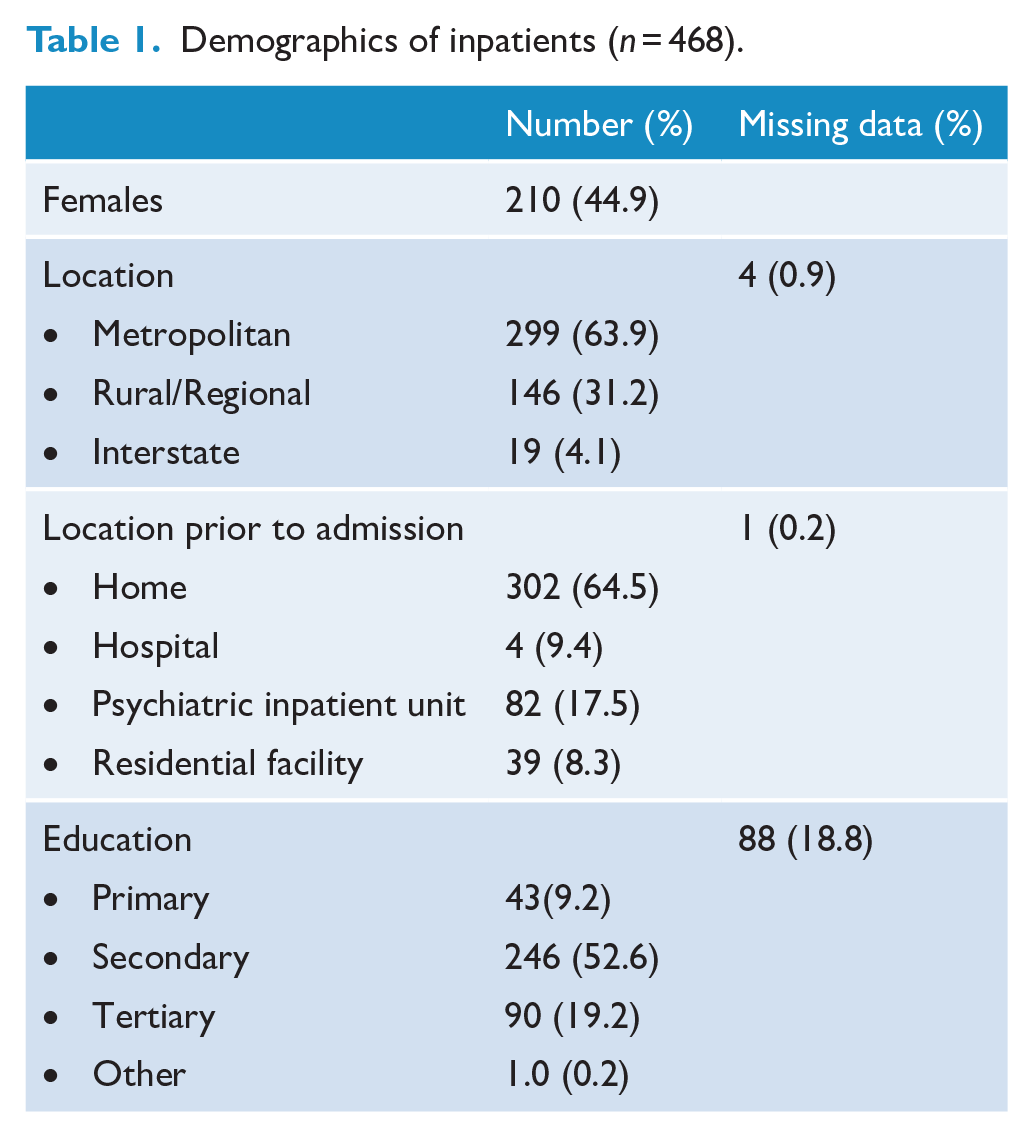
We identified 468 inpatients admitted to Neuropsychiatry from 1991 to 2014 who had a diagnosis of ‘dementia’ or ‘cognitive impairment’ due to any cause. There were more men than women (55% vs 45%), 64% lived in an urban area and 65% were living at home prior to their admission. A total of 93.0% were Caucasian. The average age of onset of symptoms was 57.9 years (SD = 11.8, range: 15–90) with mean admission age being 60.9 years (SD = 11.9, range: 15–91). The mean NUCOG score was 64.5 (SD = 18.4, range: 10–98.5), indicating moderate cognitive impairment. The most frequently occurring GAF categories were 31–40 (25.6%) and 41–50 (32.2%), suggesting moderate impairment in functioning (Table 1).
Demographics of inpatients (
Dementia characteristics
Table 2 refers to YOD and OOD while Table 3 contains information pertaining to the different subtypes of dementia. There were 25 patients for whom age of onset was unclear (5.3%). Of the remaining 443 patients, 25.5% were designated as OOD (
Demographic characteristics of patients with older-onset dementia (OOD) and younger-onset dementia (YOD).
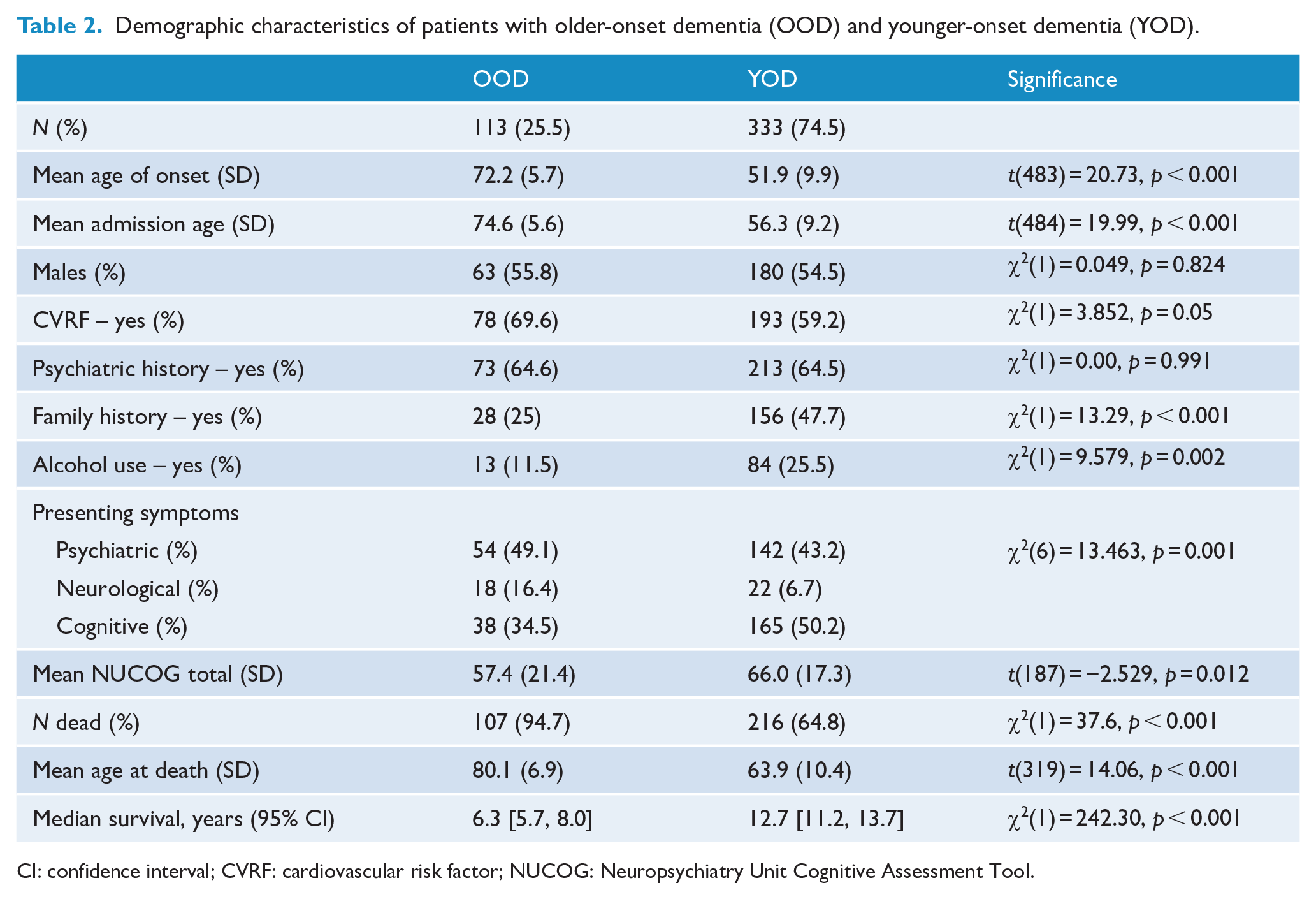
CI: confidence interval; CVRF: cardiovascular risk factor; NUCOG: Neuropsychiatry Unit Cognitive Assessment Tool.
Demographic characteristics of the different dementia types,
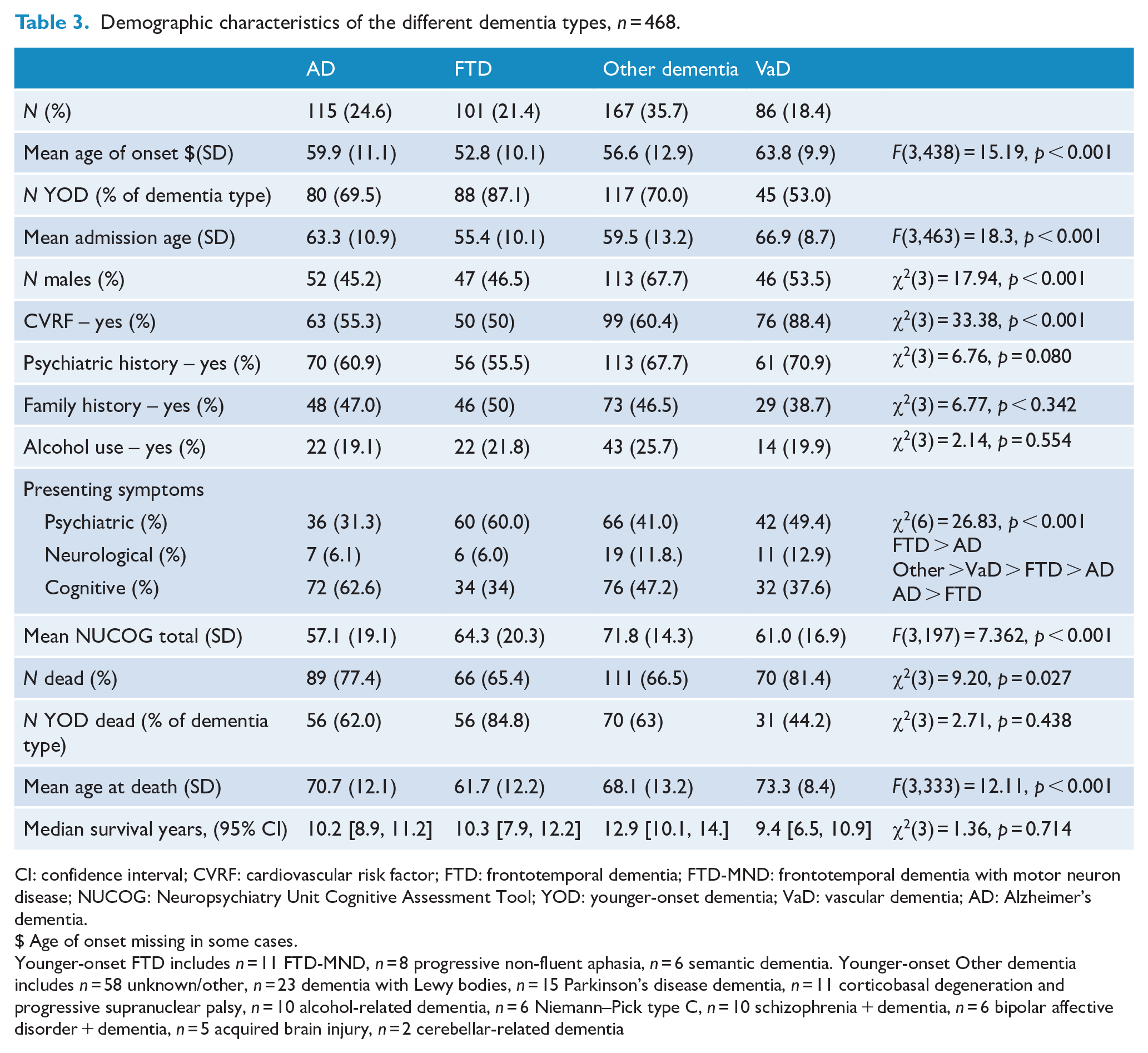
CI: confidence interval; CVRF: cardiovascular risk factor; FTD: frontotemporal dementia; FTD-MND: frontotemporal dementia with motor neuron disease; NUCOG: Neuropsychiatry Unit Cognitive Assessment Tool; YOD: younger-onset dementia; VaD: vascular dementia; AD: Alzheimer’s dementia.
$ Age of onset missing in some cases.
Younger-onset FTD includes
The most common dementia was ‘Other’ (35.7%), followed by AD (24.6%). There were significantly more men in the ‘Other’ dementia group, χ2(3) = 17.9,
Cognition and function
Presenting symptoms
Mortality
NDI data linkage revealed that 337/468 patients in this group had died (72.1%). The mean age at death for the entire group was 69.5 years (SD: 12.4, range: 18–96.3). Average follow-up time was 11.2 years (SD: 6.5, range: 10.5 months to 36.7 years). Significantly more people with OOD had died compared to YOD (94.7% vs 64.8%), χ2(1) = 37.6,
The Kaplan–Meier curves show survival of the patients with OOD and YOD (Figure 1(A)) and for the different dementia types (Figure 1(B); log-rank test:
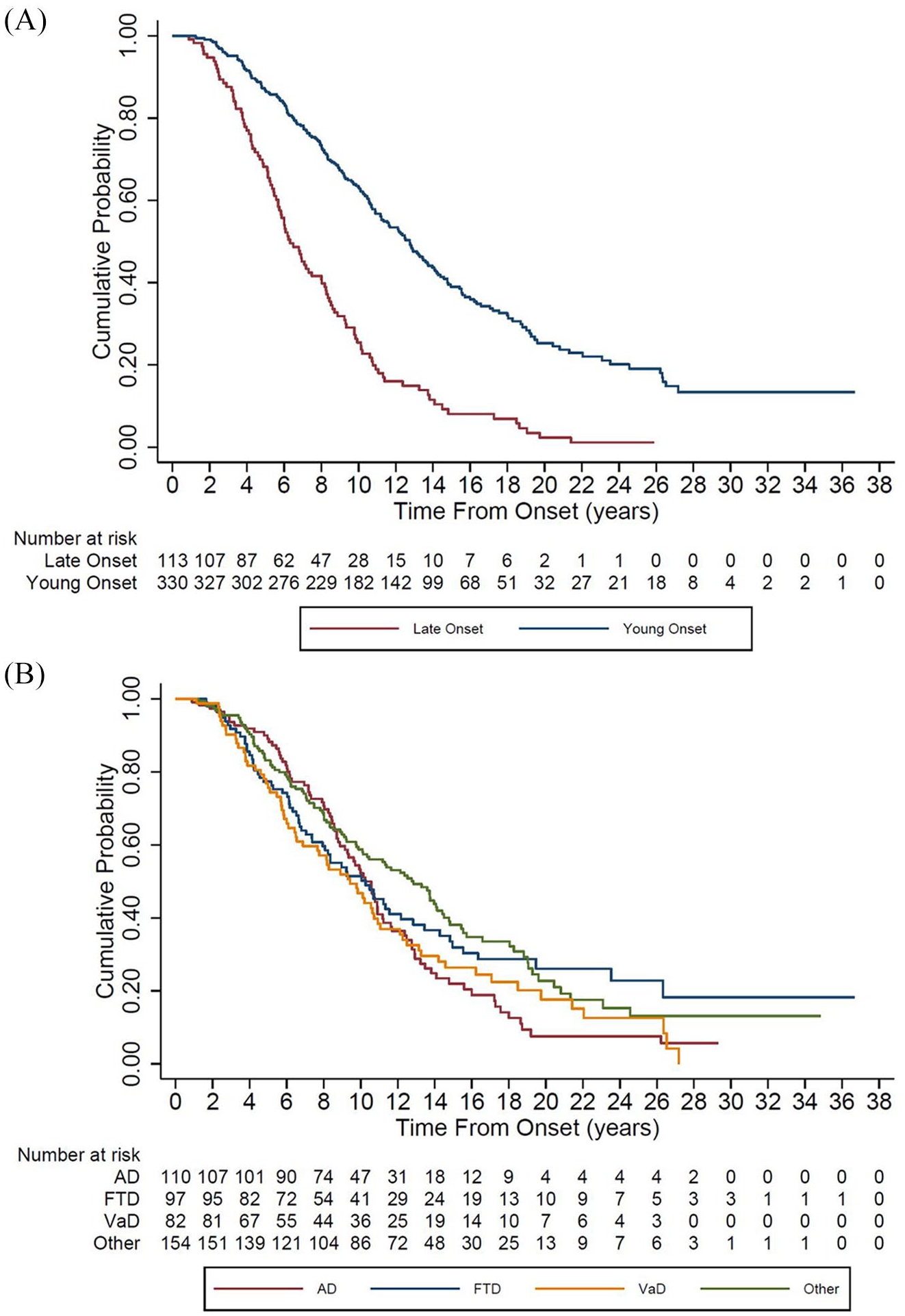
Kaplan–Meier survival curve (A) for older-onset dementia and younger-onset dementia and (B) for dementia subtypes.
Table 4a shows median survival for OOD, YOD, the dementia subtypes with comparisons by sex (Table 4b). Older-onset VaD has the shortest survival, 5.8 years (95% CI 4.6,9.3). For sex, females with OOD had longer survival compared to males (7.2 cf 5.7 years, respectively), but this was reversed for YOD, with males having longer survival (13.6 years cf 10.9 years, respectively), χ(1) = 10.49,
Median survival rates for OOD, YOD and dementia types.
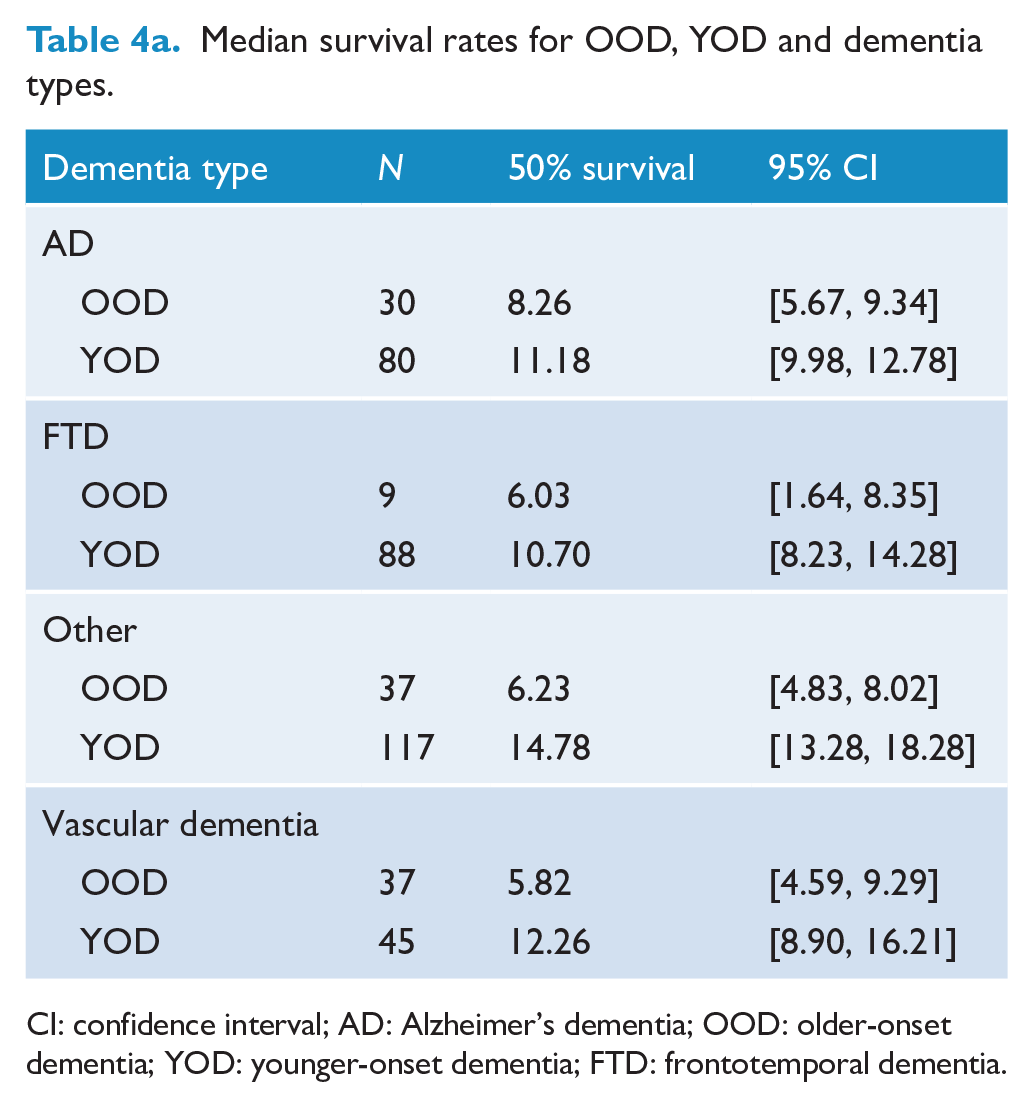
CI: confidence interval; AD: Alzheimer’s dementia; OOD: older-onset dementia; YOD: younger-onset dementia; FTD: frontotemporal dementia.
Median survival rates for OOD, YOD and sex.
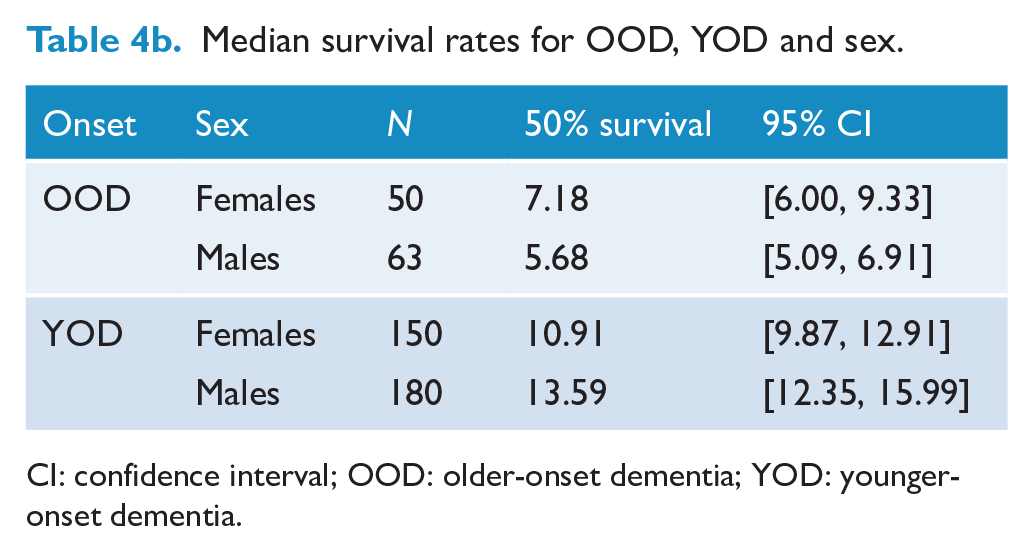
CI: confidence interval; OOD: older-onset dementia; YOD: younger-onset dementia.
Cox proportional hazard ratio (HR; Table 5) revealed two independent predictors of mortality – χ2(11) = 72.67,
Cox proportional regression.
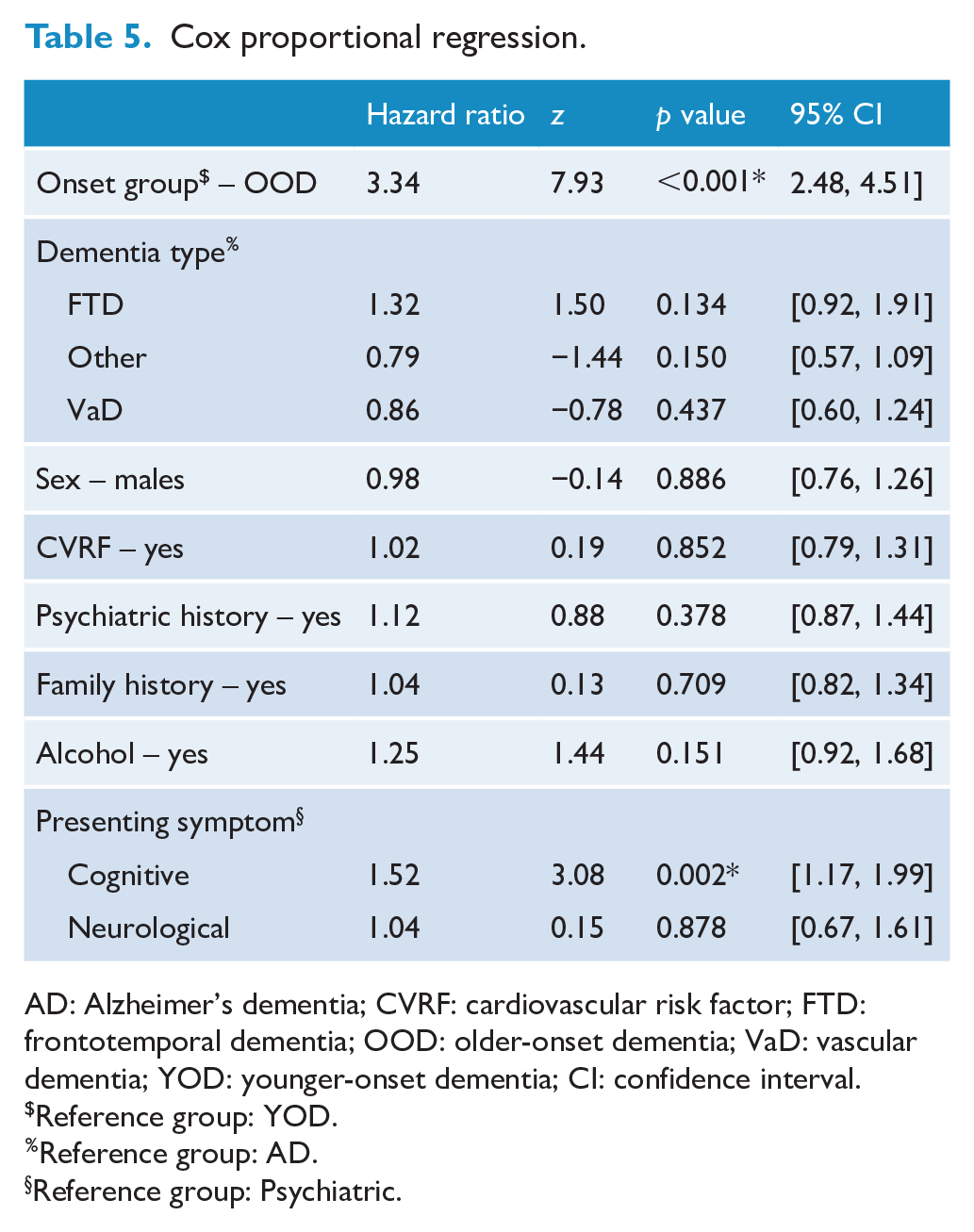
AD: Alzheimer’s dementia; CVRF: cardiovascular risk factor; FTD: frontotemporal dementia; OOD: older-onset dementia; VaD: vascular dementia; YOD: younger-onset dementia; CI: confidence interval.
Reference group: YOD.
Reference group: AD.
Reference group: Psychiatric.
Compared to the general Australian population, having an OOD had an SMR of 2.8 (95% CI = [2.3, 3.4]) and having a YOD had an SMR of 5.9 (95% CI = [5.2, 6.8]).
Discussion and conclusion
This was a retrospective study, with the objectives of investigating survival duration and risk factors of mortality in patients with dementia who attended a neuropsychiatry inpatient service over a 20-year period. This is the largest Australian study to date with the longest follow-up period and large numbers of patients in the major dementia subtypes.
In our study, median survival for those with OOD was shorter compared to YOD, which is consistent with some studies (Go et al., 2013; Guehne et al., 2005; Koedam et al., 2008). In contrast, Rhodius-Meester et al. (2019) reported survival duration from diagnosis of 6 years for OOD, YOD and the different dementia subtypes (AD, FTD and VaD). It has been suggested that updated treatment modalities such as use of cholinesterase inhibitors for AD and more aggressive management of CVRF may improve survival (Brodaty et al., 2012; Go et al., 2013; Rhodius-Meester et al., 2019); however, this may only be evident in OOD (Go et al., 2013; Rhodius-Meester et al., 2019).
A total of 75% of our patients had a YOD, and thus, comparisons to our study are made with other YOD-related literature. We found median survival to be similar across the dementia subtypes with no significant differences, between 9.4 (VaD) to 10 years (AD and FTD) to 12.9 years (Other).
This survival duration has been reported previously (Atkins et al., 2012; Go et al., 2013; Pasquier et al., 2004; Roberson et al., 2005). Some studies calculated survival time from age at diagnosis (Koedam et al., 2008; Rhodius-Meester et al., 2019), which makes comparisons with our study difficult as it can be methodologically challenging to estimate age of onset (Gerritsen et al., 2019). Nevertheless, Go et al. (2013), who also included both younger- and older-onset AD, found median survival to be 12.6 years from age of onset. Kay et al. (2000) found similar survival duration in their younger-onset AD and VaD, approximately 6 years from age of diagnosis. Seven-year survival from diagnosis for younger-onset AD and FTD was found by Rhodius-Meester et al. (2019). In contrast, others have reported differences in survival length depending on the dementia subtype. Shorter survival in FTD compared to younger-onset AD has been described (Atkins et al., 2012; Roberson et al., 2005), and a recent Dutch study reported on very long survival in YOD, particularly for FTD and VaD, over 20 years (Gerritsen et al., 2019). These authors explained the extended survival due to improved ability to diagnose YOD with sophisticated neuroimaging and use of cerebrospinal fluid biomarkers. Their method of obtaining death information was using patient or family contact and had 21.7% attrition, which may have led to an overestimation of duration of survival.
We grouped together all patients diagnosed with FTD as we did not have sufficient numbers for analysis of the language- and behavioural-variants separately. The groups who have done this have found semantic dementia to have the longest survival, approximately 12 years (Roberson et al., 2005), with progressive non-fluent aphasia, approximately 10 years (Hodges et al., 2003; Roberson et al., 2005), and bvFTD having the shortest duration, 8.7 years (Roberson et al., 2005). Hodges et al. (2003) and Roberson et al. (2005) considered pathological-type, rather than phenotype, as a factor for survival and concluded that tau-positive pathology progressed slower than tau-negative pathology (10 years vs 5 years), suggesting that pathological process might be a factor for survival duration.
With regard to risk factors of mortality/survival, we found two significant predictors. Not unsurprisingly, having an OOD had 3 times risk compared to a YOD. This is comparable to Koedam et al. (2008). Increasing age of onset and OOD would infer increased mortality risk (and shorter survival) compared to YOD due OOD being closer to life expectancy (Brodaty et al., 2012; Gerritsen et al., 2019; Go et al., 2013).
However, a unique finding in our study was that cognitive presenting symptoms had a 1.5 risk of mortality compared to when psychiatric symptoms were the sentinel symptoms. To our knowledge, this is the first time this has been investigated in the literature and is particularly relevant in YOD. In OOD, Ganguli et al. (2005) found an increased risk of mortality with depressive symptoms, whereas Saz et al. (1999) reported that depression was ‘protective’ against mortality with lower odds ratio (OR; OR 1.8 depression + dementia compared to OR 5.0 dementia only). These studies only considered depression as a comorbid condition, rather than as presenting symptoms. There are a number of explanations for our finding. For someone presenting with non-cognitive symptom who receives a dementia diagnosis, receiving a diagnosis earlier would infer a decreased risk of mortality (conversely, the onset of cognitive symptoms may mean that the dementia has further progressed, indicating increasing severity). Our exploratory data found that patients presenting with psychiatric symptoms had 2-year longer duration of illness compared to those who presented with cognitive symptoms. Therefore, these findings suggest for many dementias, regardless of older- or younger-onset, psychiatric symptoms precede the onset of cognitive symptoms. As a Neuropsychiatry service, we are often referred people who have a primary psychiatric diagnosis who are suspected of having a YOD – it could be that some of these people actually have a dementia associated with their psychiatric disorder, rather than a dementia per se such as AD or FTD. Although, this cohort of people may fulfil the criteria for a dementia (e.g. significant cognitive impairment, imaging changes, functional decline) but not have a specific dementia such as AD or FTD. We hypothesise that while having a dementia/neurodegenerative disorder is a risk factor for death, perhaps a dementia related to psychiatric disorders confers less risk. Recent literature reports of increased risk of dementia in people with late-onset psychosis/schizophrenia with increased rate of ageing and rate of death (Kodesh et al., 2020). There is likely a complex interaction of mechanism of action, which warrants further exploration. Further investigation into symptom onset is recommended as these might have an association with rate of progression of the dementia.
We did not find CVRFs or alcohol associated with increased mortality risk, consistent with Gerritsen et al. (2019). Possibly, specific CVRFs are more relevant such as with diabetes, which was a significant predictor in younger-onset AD (Go et al., 2013). This should be further investigated as possible preventive factors, as we were unable to examine these effects. Comorbidity, whether this is measured via CVRFs, falls or gait changes have had inconsistent findings as predictors of mortality. People with YOD compared to OOD are often more physically robust which might suggest that if comorbidity is present, this may have an important role for mortality.
Our study also did not find sex to be independently associated with mortality; however, we did describe sex differences in survival depending on onset of the dementia. Females who had an OOD had longer survival compared to males with an OOD, with the reverse for YOD, where males with YOD had longer survival. While sex is genetically unmodifiable, its contributions to mortality and survival remain disparate. A review by Brodaty et al. (2012) stated there was no difference, whereas others (Guehne et al., 2005; Todd et al., 2013) reported males had increased mortality. These differences could be due to psychosocial factors, with males seeking care later than females and hence are affected more severely, from a cognitive and functional perspective, and thus closer to death. Alternatively, this could be partially related to greater longevity in females (Brodaty et al., 2012; Buckley et al., 2019). Authors have postulated that older women are ‘ protected’, i.e. they have more cognitive reserve, progress slower and survive longer (Davis et al., 2020; Go et al., 2013), whereas others have shown that women deteriorate cognitively faster (Lin et al., 2015). In AD, neuroimaging studies have demonstrated increased uptake of only tau in women with younger-onset AD (Edwards et al. 2021, preprint) but with similar performance with men on cognitive tests, suggesting better cognitive reserve in women. The effect of sex on dementia is clearly very complex with pathology, comorbidity, resilience and other psychosocial factors interacting with each other and more longitudinal studies are required. Nevertheless, regardless of sex, our findings are that having an OOD or YOD confers 3 times and 6 times risk of death, compared to the general population.
In conclusion, the important clinical messages from this study are that presenting with cognitive symptoms and an OOD are risk factors for increased mortality. We are also able to provide a reasonable ‘ballpark’ figure for average survival for the various subtypes of dementia (approximately 10 years from age of onset). Our study was limited, in that it was retrospective and from a tertiary public hospital service, with the usual challenges in interpreting the data. We also lack pathological confirmation of dementia subtype. The ‘Other dementias’ group was broad and included diagnoses such as corticobasal degeneration and DLB, but also dementias related to psychiatric disorders. It is possible that this heterogeneous group may have affected results in terms of survival and risk factors. However, the strength of this study was that the sample was from a single centre with high internal consistency of diagnoses and our summaries are comprehensive, taking into consideration collateral history from the patient, carer and previous clinicians when available. In addition, a large proportion of our sample had died, and we used an NDI so we were able to accurately estimate survival and cause of death with no attrition.
There are many unanswered questions in this area of dementia. Clearly, there is a complex interaction of risk factors relating to survival, as some individuals with severe dementia can live for a very long time, whereas some with less severe dementia die sooner. Having strategies to improve and mitigate survival is important, but it is also crucial to ensure a good quality of life while living with dementia.
Footnotes
Acknowledgements
The authors would like to thank all the current and previous Neuropsychiatry staff as well as the patients and families who attended.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: S.M.L. is funded by the Yulgilbar Alzheimer’s Research Program and a National Health and Medical Research Council (NHMRC) Early Career Fellowship (GNT1138968). NorthWestern Mental Health seed grant also provided funding for this project. All the other authors report no relevant disclosures.
