Abstract
Objectives:
Several components are known to underlie goal-directed pursuit, including executive, motivational and volitional functions. These were explored in schizophrenia spectrum disorders in order to identify subgroups with distinct profiles.
Methods:
Multiple executive, motivational and volitional tests were administered to a sample of outpatients with schizophrenia spectrum diagnoses (n = 59) and controls (n = 63). Research questions included whether distinct profiles exist and whether some functions are impacted disproportionately. These questions were addressed via cluster analysis and profile analysis, respectively.
Results:
Some such functions were significantly altered in schizophrenia while others were unaffected. Two distinct profiles emerged, one characterized by energizing deficits, reduced reward sensitivity and few subjective complaints; while another was characterized by markedly increased punishment sensitivity, intact reward sensitivity and substantial subjective reporting of avolitional symptoms and boredom susceptibility.
Conclusion:
These findings highlight the importance of considering distinct patterns of strengths and deficits in functions governing goal-directed pursuit in schizophrenia that demarcate identifiable subtypes. These distinctions have implications for treatment, assessment and research.
Introduction
Schizophrenia is a leading cause of worldwide disease burden (World Health Organization (WHO), 2006), and reduced goal pursuit is its most debilitating aspect, driving poor functional outcomes (Barch et al., 2014; Kiang et al., 2003). Reduced goal pursuit is the defining behavioural manifestation of negative symptoms including avolition and amotivation (American Psychiatric Association (APA), 2013).
Several mechanisms are known to underlie goal pursuit, both in the general population and in those with schizophrenia spectrum disorders (SSD). These (by definition) broadly consist of volitional and motivational functions, which are distinct but interconnected (e.g. Kornhuber et al., 1989; Kuhl, 2020), as well as the neurocognitive, personality and other mechanisms that subserve these functions. To clarify, the term ‘motivation’ is typically used to describe the quality and quantity of drives that give impetus to action and determine its goals, while ‘volition’ is used to describe the processes that govern its initiation, maintenance and completion towards goal achievement, as well as the overcoming of obstacles (e.g. changing external circumstances, or internal factors such as fatigue, conflicting goals or anxiety). This usage will be maintained throughout the current study.
The distinction between avolition and amotivation has not often been critically examined in the schizophrenia literature. Some have suggested that parsimony dictates that these be treated as identical constructs (e.g. Foussias and Remington, 2010). Furthermore, avolition is defined in the Diagnostic and Statistical Manual of Mental Disorders (5th ed.; DSM-5; APA, 2013) both as the ‘inability to initiate and persist in goal-directed activities’ (p. 818) and as ‘reduced drive to perform goal-directed behaviour’ (p. 100, italics added), a definition conflating volition and motivation in that it simultaneously refers to factors interfering with the execution of goal-directed pursuit (i.e. volitional factors) and a lack of any impetus to perform these actions in the first place (i.e. motivational factors).
In the broader literature, however, distinctions between volition and motivation are common. Kornhuber et al. (1989) define volitional functions as concerning the ‘what’, ‘how’ and ‘when’ of action, while defining motivation as the ‘input’ into the brain’s volitional action systems. Notably, the term ‘volition’ in contemporary psychology has largely been replaced by terms that describe its cognitive constituents, e.g. ‘executive functions’ (Zhu, 2004). In keeping with this, Kuhl (2009) suggests that ‘volition’ pertains to the planning, initiation and disengagement of action, as well as impulse control (i.e. factors currently considered to be under the rubric of executive function; Stuss and Alexander, 2007; Zhu, 2004). In the historic schizophrenia literature, Kraepelin (1919) highlighted the multifaceted nature of ‘volitional disorders’ in schizophrenia, and further noted that schizophrenia is a disorder not only of volition, but of its ‘emotional precursors’ (Kraepelin, 1920), which may be understood as motives using the current rubric. Current research (e.g. Barch and Dowd, 2010; Marder and Galderisi, 2017) likewise supports a multifaceted conception of motivational and volitional deficits in SSD.
An overlapping distinction between constructs relevant to goal direction, of interest in the current work, is reflected in the Research Domain Criteria (RDoC; Insel et al., 2010). RDoC distinguishes between behavioural inputs that are positively valenced vs negatively valenced, based on evidence of distinct neural systems and dynamics. Positive valence systems generally govern ‘approach’ behaviour, or positive motivation and action towards a goal state, whereas negative valence systems govern the inhibition of this, or behaviour that avoids undesired end states. In terms of the distinction between amotivation and avolition as seen in SSD, reductions in goal-directed behaviour often pertain to reduced approach-based or positively valenced motives (e.g. pursuing relationships, doing work for pay or pleasure, leisure activities and instrumental activities of daily living). Interference in such positively valenced activities, however, could be understood either in terms of a motivational failure per se (e.g. lack of affective impetus/positive motivation, reward sensitivity) or in terms of an inability to enact motives despite their presence (e.g. due to a hyperactive negative valence system, as reflected in anxiety, or due to executive dysfunction). These two hypotheses imply that clearly distinct underlying factors may be affected in SSD, and both may play a role in reduced goal direction (see Table 1).
Summary of related constructs underlying goal pursuit.
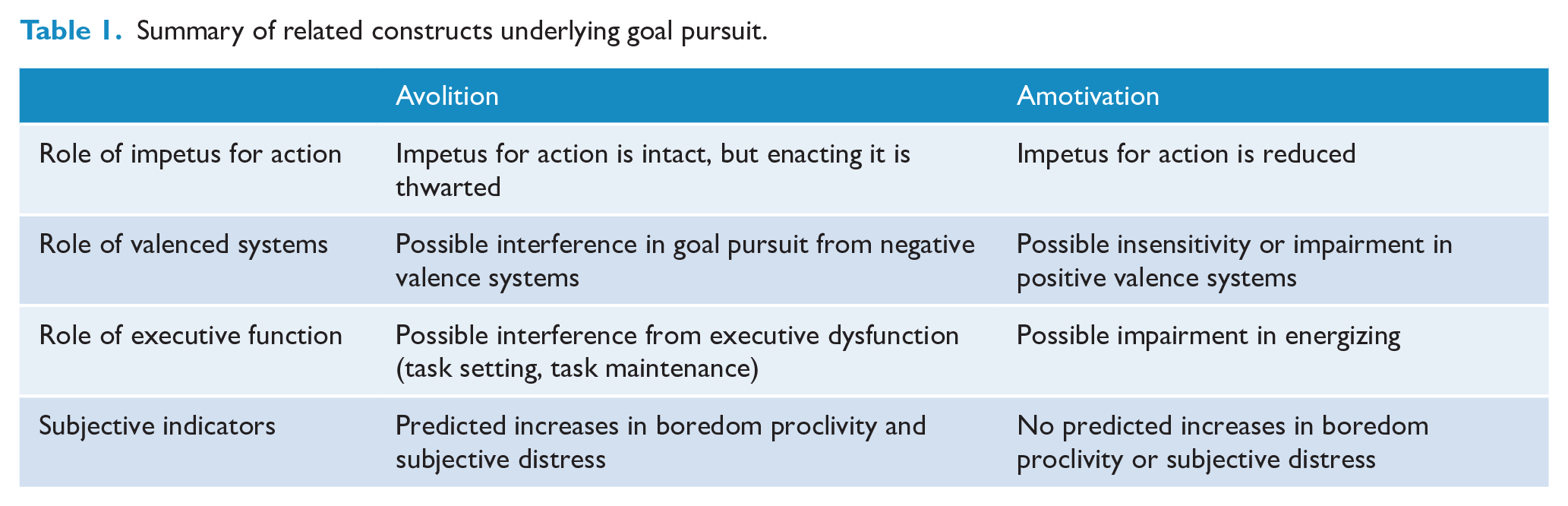
In summary, multiple systems are known to underlie goal pursuit, and these may be broadly classified under two categories (Table 1), each with the potential for further delineation. Problems in goal pursuit may be attributable to reduced impetus or positive emotional inputs for action, reflecting what has been termed ‘amotivation’ and driven by impairment in positively valenced systems; or they may exist in the context of intact positive motivation, e.g. if interference from impaired executive, volitional or negatively valenced systems is present. The latter is likely to produce feelings of subjective distress and increased experiences of related phenomena, such as complaints of difficulty initiating action and boredom (see the section below on boredom propensity) due to thwarted action; the former may not produce such emotional states given the lack of desire for such action in the first place (e.g. Tek et al., 2001).
The current study seeks to delineate which of a wide range of such mechanisms are altered within SSD, and whether subgroups with distinct profiles of such functions may exist, reflecting the binary between volitional disturbance and reduced motivation.
Given that symptoms such as amotivation and avolition are grounded only in observed behaviour and were not constructed with consideration of underlying processes, the current study does not seek to ground reduced goal-directed behaviour in existing symptom schemes, but rather seeks to examine underlying, cross-symptomatic and cross-diagnostic processes. This subtyping approach is crucial to the understanding of schizophrenia, especially given its implications for developing new, more specifically targeted interventions (Luther et al., 2015): It has long been acknowledged that schizophrenia is a heterogeneous disorder; however, subtyping schemes with implications for treatment have been elusive. The current scheme proposes to subtype according to a well-established distinction between factors underlying a single symptom type, with promise to refine treatment targets (i.e. by determining which factors to target in a given case; e.g. whether motivational or volitional in nature).
Constructs putatively underlying reduced goal pursuit in SSD for the current study have been selected from a broad cross-section of literature to include functions known to subserve action, rather than clinician-based ratings that reify existing, behaviour-based concepts of avolition and may conflate multiple underlying basic functions. Some of these are known to be impaired in SSD, and others were selected for breadth. These constructs are summarized below. Individual variables entered into analysis are bolded for ease of reference and are summarized in Table 3.
Volitional and executive constructs included in the current study
Frontal executive functions
The prefrontal cortex (PFC) governs the initiation, switching and termination of action. These ‘executive’ functions overlap definitionally with volition (Zhu, 2004) and are especially relevant to schizophrenia, in which avolition has been connected with prefrontal hypoperfusion (Liddle et al., 1992) and reduced resting-state connectivity (Woodward et al., 2012) in the PFC, and with dysexecutive symptoms (e.g. Fervaha et al., 2014). Stuss et al. (2005; Stuss and Alexander, 2007) have proposed at least three discrete PFC functions:
The above-noted functions may be measured objectively, as described in the ‘Methods’ section. Additional indices of
Delay discounting
Ainslie (1992) conceives of volitional action as the overriding of an irrationally steepened discounting curve: a function that relates the subjective value of a reward with the time that must elapse before it is given. It is operationalized using the
Maintenance of internal representations
The ability to maintain a representation of intended action and retrieve it in the appropriate context – a critical component of volition – is known as
Anticipation and the representation of reward
Several researchers have highlighted the importance of the mesolimbic dopaminergic system in evaluating anticipated reward (e.g. Treadway et al., 2012b), its role in anhedonia (Treadway et al., 2009, 2012a) and functioning in schizophrenia (Barch et al., 2014). It has been suggested that avolition may involve failure to translate intact hedonic capacity (Cohen and Minor, 2010; Kring and Moran, 2008; Strauss and Gold, 2012) into anticipated reward (Gold et al., 2013). Indeed, individuals with schizophrenia have shown greater
Self-directedness
This personality construct is defined by Cloninger (1987) as the ability to organize drives into a cohesive whole, as opposed to a set of disconnected impulses. It is reduced in schizophrenia (e.g. Hori et al., 2008).
State orientation
This refers to an avolitional tendency to fail to enact intended actions (Kuhl, 1981). It is divided into two distinct forms: ‘
Boredom propensity
This construct has a strong correlation with avolition (Blunt and Pychyl, 1998) and may be thought of as a desire to engage in meaningful action, coupled with the inability to do so (Eastwood et al., 2012). It is increased within schizophrenia and may be considered a marker of intact motivation in a context of reduced volition (Gerritsen et al., 2015).
Motivational constructs included in the current study
Self-determination theory
Deci and Ryan’s (1980) Self-Determination Theory posits that as various goals and habits are ‘internalized’ they require less felt effort (e.g. Deci and Moller, 2005). Individual differences in the relative strength of intrinsic (vs extrinsic) goals exist (Kasser and Ryan, 1996), with more intrinsically motivated individuals characterized as more active and autonomous. Therefore,
Sensitivity to punishment and reward
In Gray’s (1982) ‘hypothetical nervous system’, two antagonistic systems known as the Behavioural Activation System (BAS) and Behavioural Inhibition System (BIS) regulate approach-related and freezing-related responses to conditioned stimuli. They comprise a well-validated parsing of personality structure (e.g. Carver and White, 1994; Cloninger, 1987; Torrubia and Tobeña, 1984). The emotional substrates underlying BAS and BIS are thought of as approach-related affect and anxiety, respectively (e.g. Corr, 2002; Harmon-Jones et al., 2002). Scholten et al. (2006) found elevations in BIS, but no difference in BAS, in schizophrenia, suggesting that avolition may represent an overabundance of anxiety rather than a lack of approach motivation.
The current study
Studies of goal pursuit in schizophrenia have typically examined only group-level effects and have assessed only a narrow range of underlying variables, which does not permit the observation of underlying heterogeneity. Some functions may be more impacted than others and identifiable subgroups may express different sources of reduced goal direction. The current study addresses these two issues using a novel approach: A range of such functions is examined simultaneously in a way that permits the observation of subgroup heterogeneity.
It is hypothesized that at least two clusters will emerge, reflecting the critical division between reductions in motivational inputs on one hand, and reduced volition, impaired executive functions and/or interference from negatively valenced factors on the other hand (Table 1). It is also hypothesized, given the above review, that specific functions within these broad categories will be impacted differently between the clusters.
Methods
Participants
Clinical sample
Outpatient participants with diagnoses of schizophrenia or schizoaffective disorder were recruited at the Centre for Addiction and Mental Health (CAMH) in Toronto, Ontario, and at St. Joseph’s Healthcare, Hamilton, Ontario (SJHH). Participants were recruited via registries and flyers. Participants were screened up to 2 weeks prior to participation to assess inclusion criteria. Clinical participants had to meet criteria for Diagnostic and Statistical Manual of Mental Disorders (4th ed., text rev.; DSM-IV-TR) diagnosis of schizophrenia or schizoaffective disorder, confirmed via the Mini International Neuropsychiatric Interview (MINI; LeCrubier et al., 1997), and be 18 or older. Exclusion criteria included any history of loss of consciousness longer than 5 minutes, neurological disorder, substance or alcohol-related disorder within 1 month of testing (as assessed via the MINI), current major depression or recent (past month) change in medication. A total of 63 outpatients were recruited (Table 2). Four were excluded prior to participation, two due to recent changes in medication, one because of diagnostic ambiguity and one because of head trauma. Six had a diagnosis of schizoaffective disorder while the remainder had a diagnosis of schizophrenia.
Clinical and demographic characteristics of clinical and control samples.
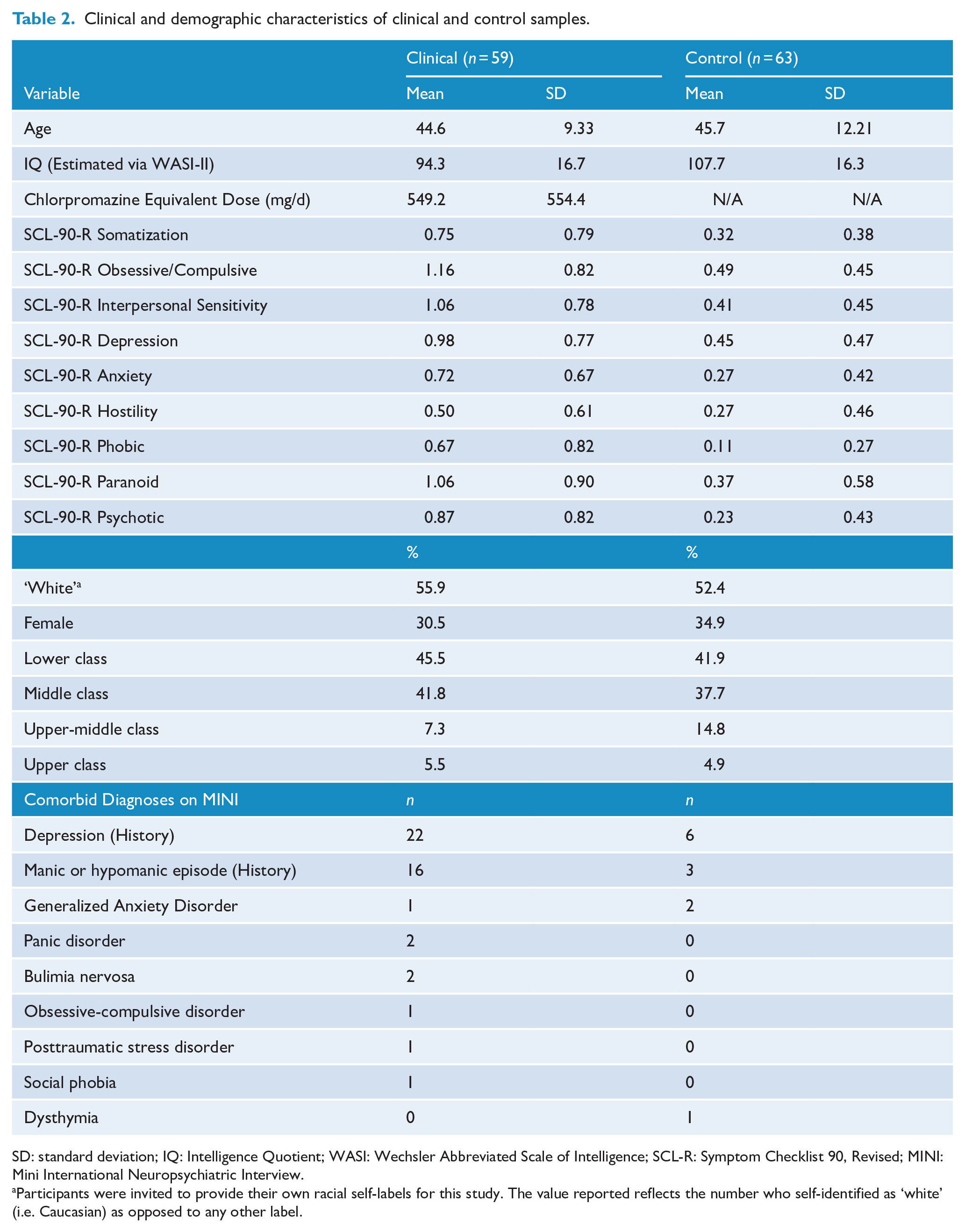
SD: standard deviation; IQ: Intelligence Quotient; WASI: Wechsler Abbreviated Scale of Intelligence; SCL-R: Symptom Checklist 90, Revised; MINI: Mini International Neuropsychiatric Interview.
Participants were invited to provide their own racial self-labels for this study. The value reported reflects the number who self-identified as ‘white’ (i.e. Caucasian) as opposed to any other label.
Control sample
Control participants were recruited via posters displayed on campus and in the surrounding community, Internet ads and a subject registry. Participants were screened up to 2 weeks prior to participation to confirm inclusion/exclusion criteria. The control sample was selected to match the clinical sample in age and gender. Control participants needed to demonstrate a lack of psychotic disorder as assessed by the MINI and be 18 or older. Exclusion criteria were otherwise identical to those applied to the clinical sample. Control participants were included if they had psychiatric morbidity (other than current depression) to reduce the likelihood that observed group differences were owing to co-morbidities common in schizophrenia, while balancing the need for ecological validity (i.e. without selecting out clinical participants with co-morbid conditions, which are common). Sixty-six individuals were recruited (Table 2). Three of these were ineligible due to histories of substance-related disorders and/or current major depressive episodes.
Procedures
After providing written informed consent, participants were assigned randomly to one of two conditions to counterbalance the Zeigarnik tasks and administered study measures in a fixed order. Testing lasted approximately 3 hours. Study measures included an interview to assess inclusion criteria and demographic data; the MINI (Lecrubier et al., 1997) to establish diagnosis; the Wechsler Abbreviated Scale of Intelligence, 2nd Edition (WASI-2; Wechsler, 1999) to estimate general cognitive functioning; the Symptom Checklist 90, Revised (SCL-90R; Derogatis and Savitz, 1999); and the measures listed in Table 3. Full detail of study procedures are specified in the online supplemental material. Data collection was approved by the Research Ethics Boards at CAMH, SJHH and York University.
Follow-up tests for profile analysis.
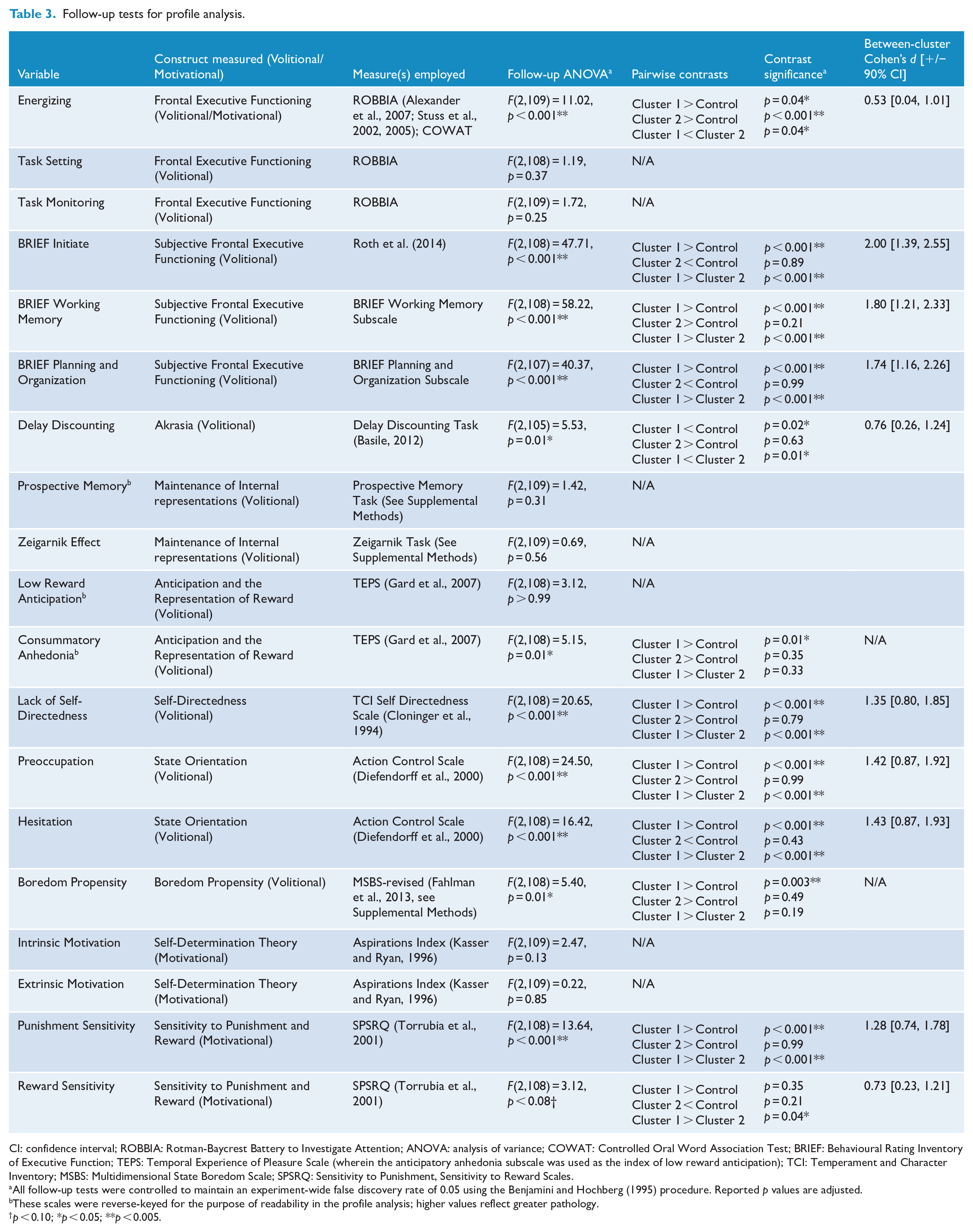
CI: confidence interval; ROBBIA: Rotman-Baycrest Battery to Investigate Attention; ANOVA: analysis of variance; COWAT: Controlled Oral Word Association Test; BRIEF: Behavioural Rating Inventory of Executive Function; TEPS: Temporal Experience of Pleasure Scale (wherein the anticipatory anhedonia subscale was used as the index of low reward anticipation); TCI: Temperament and Character Inventory; MSBS: Multidimensional State Boredom Scale; SPSRQ: Sensitivity to Punishment, Sensitivity to Reward Scales.
All follow-up tests were controlled to maintain an experiment-wide false discovery rate of 0.05 using the Benjamini and Hochberg (1995) procedure. Reported p values are adjusted.
These scales were reverse-keyed for the purpose of readability in the profile analysis; higher values reflect greater pathology.
p < 0.10; *p < 0.05; **p < 0.005.
Data analysis
Cluster analysis
Each of the measures described above was entered into a hierarchical cluster analysis within the clinical sample. Missing data were handled via listwise deletion because imputation in cluster analysis may bias the formation of clusters (Everitt, 1993). Ward’s (1963) method was chosen given the continuous nature of all variables, and to generate clusters with minimal within-cluster variability. Squared Euclidian distance was used as a proximity measure given its satisfaction of the assumptions underlying Ward’s method and its performance in simulation studies (e.g. Everitt, 1993; Scheibler and Schneider, 1985). To determine the number of clusters that best fit, the differences between the within-cluster error values at which each cluster was formed, and then combined into a larger cluster, were examined. The cluster solution with the greatest difference was chosen per Romesburg (1990).
Profile analysis
Standardized mean scores on each variable were contrasted between the control and schizophrenia samples/clusters using multivariate analysis of variance (MANOVA) to assess profile parallelness and flatness, conducted with the SPSS general linear model (GLM) package. Flatness of profile plots is assessed to determine whether certain measures characterize reduced goal direction in schizophrenia more so than others at the population or subgroup level. Parallelness of profiles is contrasted between all subgroups to determine where significant differences lie. Follow-up tests including one-way analyses of variance (ANOVAs) and pairwise contrasts were conducted to determine significant between-group differences. Prior to profiling, data were re-standardized according to control sample means and standard deviations to improve interpretability of the profile plot.
Results
Cluster analysis
Nine clinical participants had missing data and were excluded. From the remaining data, a clear two-cluster solution emerged, containing 29 participants in cluster 1 and 21 in cluster 2. The within-cluster error coefficient at which the solution emerged was 775.0, and the two clusters merged at a coefficient of 931.0 (range = 156.0). The solution with the next highest range was a three-cluster solution (range = 68.3). The two-cluster solution was therefore chosen as the best fit (see Figure 1). Cluster means and standard deviations for each variable are provided in Table S1 in the supplemental material available online.
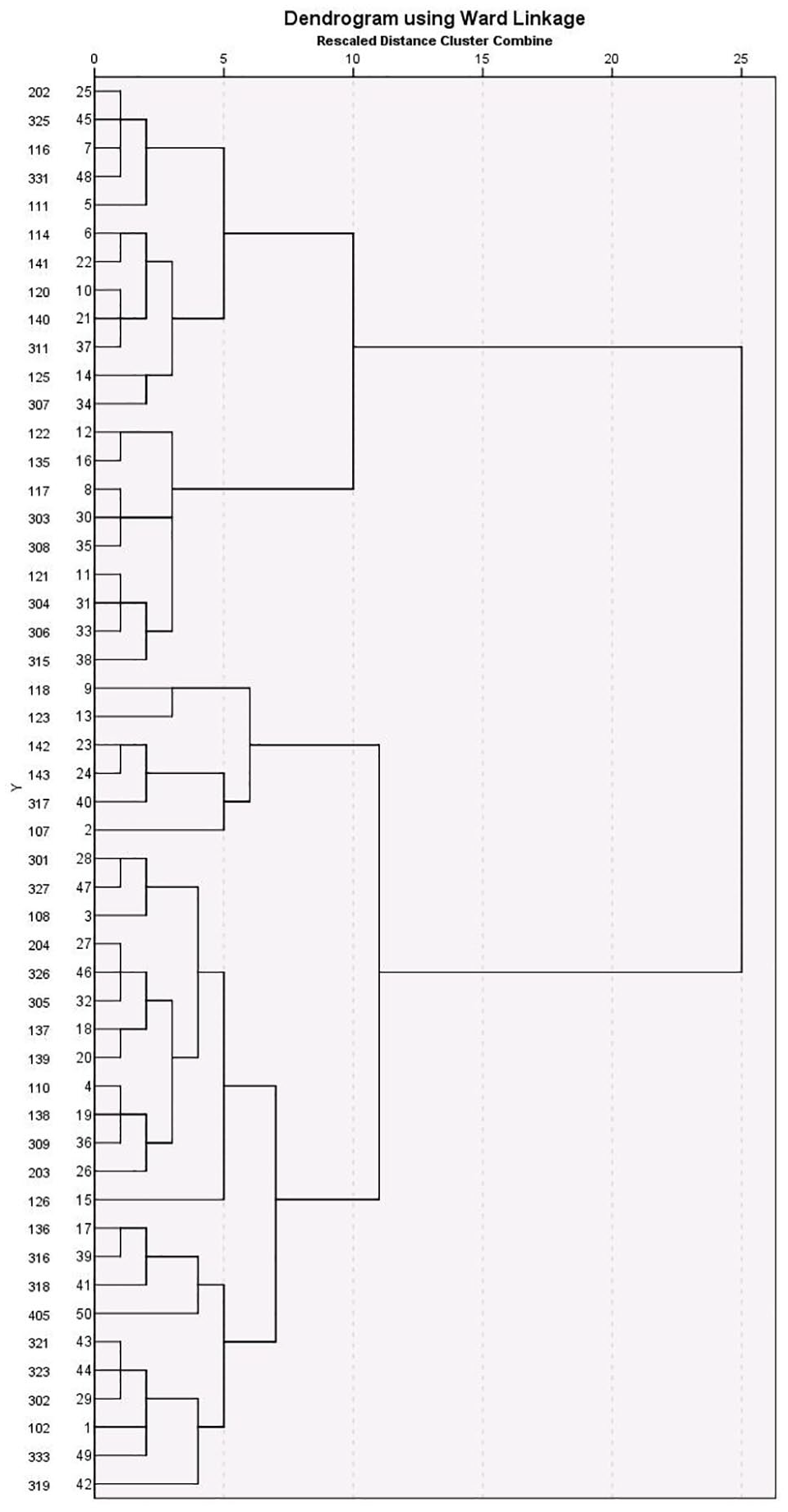
Dendogram depicting cluster analysis of clinical sample.
In terms of motivational, volitional and executive factors, the clusters differed along several dimensions, as summarized in the ‘Profile analysis’ section below.
In terms of general psychiatric symptomatology, the clusters were contrasted on the General Symptom Index of the SCL-90-R. Cluster 1 endorsed significantly greater psychopathology on this measure (M = 1.11) than cluster 2 (M = 0.39), t(45.8) = 5.69, p < 0.001. The SCL-90-R subscale showing the greatest difference between the two clusters was the Obsessive-Compulsive subscale, with cluster 1 showing greater mean elevations on this subscale, cluster 1 = 1.40, cluster 2 = 0.58, t(48) = 4.93, p < 0.001.
Profile analysis
Mixed-model MANOVAs were run with individual measures as within-subjects variables and sample or cluster membership as the between-subjects factor. Four models are summarized below: an omnibus model, two models testing each cluster’s relationship with the control sample and a model contrasting the two clusters. Greenhouse-Geisser correction was applied to all MANOVAs given observed non-sphericity. The omnibus model showed significant non-flatness, F(11.8, 1213.2) = 10.80, p < 0.001, partial η2 = 0.095, and non-parallelness, F(23.6, 1213.2) = 9.73, p < 0.001, partial η2 = 0.159 (see Figure 2). These findings were examined in three follow-up analyses. Each cluster was first entered into a separate MANOVA along with the control sample, yielding significant non-parallelness for cluster 1, F(11.7, 970.8, 1213.2) = 15.12, p < 0.001, partial η2 = 0.154, and cluster 2, F(11.0, 834.9) = 3.76, p < 0.001, partial η2 = 0.047. Each cluster was furthermore shown to produce a non-parallel profile relative to the other, F(10.0, 468.9) = 8.68, p < 0.001, partial η2 = 0.156.
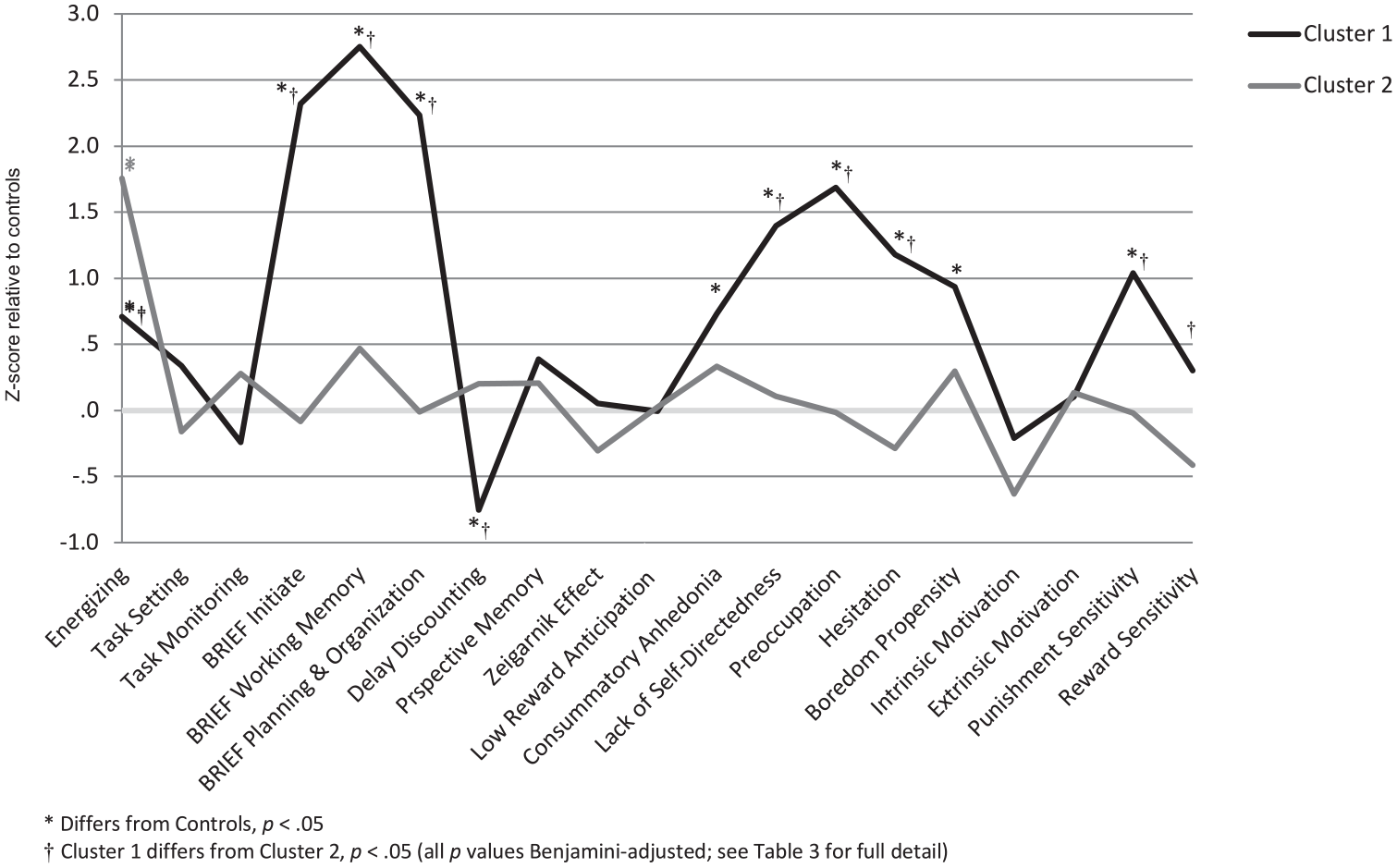
Profile plot for standardized study variables.
Individual variables were further examined via one-way ANOVAs followed by pairwise contrasts. Given the large number of contrasts performed, in order to limit type I error, the experiment-wide false discovery rate was held at alpha (0.05) using a Benjamini and Hochberg (1995) procedure. Contrasts revealed multiple significant differences (Table 3).
Discussion
Distinct profiles
The cluster and profile analyses revealed a robust two-cluster solution, with significant non-flatness and non-parallelness supporting both of the primary hypotheses. The clusters differed substantially along the lines hypothesized by a split between volitional and motivational dysfunction: Cluster 1 showed evidence of marked increases in self-reported avolition, elevated punishment sensitivity and intact reward sensitivity – factors that may interfere with goal-directed action. Cluster 2 showed a greater objectively assessed energizing dysfunction, a decrease in reward sensitivity and an overall lack of self-reported psychopathology – factors suggesting a lack of impetus for action. This distinction is also consistent with that drawn between hyperactive negative valence systems and hypoactive positive valence systems, respectively, as defined in the RDoC approach (Insel et al., 2010), as detailed below and in Table 1. In contrast to the hypotheses, however, task setting and task monitoring did not differ significantly between the clusters and were not impaired in the current sample.
The statistical and practical significance of these differences were supported by several findings. The two clusters emerged from smaller solutions with a small increase in within-cluster variance relative to the increase in variance accompanying the merging of the two clusters (Romesburg, 1990) and demonstrated several significant differences in the profile analysis, many with large effect sizes (Table 3). The observation of differences between the clusters in measures not entered into the cluster analysis (i.e. SCL-90-R) evidences their construct validity. It was hypothesized that any cluster structure would reflect the primary distinction between volition and motivation, and related constructs (Table 1). This hypothesis was supported by several observations summarized in each cluster description below.
Cluster 1: avolitional, or factors interfering with action
Cluster 1 may be understood as a subjectively distressed subgroup, given high self-report of pathology and impairment, without strong accompanying executive control deficits or deficient reward motivation, suggesting that other factors are representative of reduced goal direction in this group. Increased punishment sensitivity in this cluster stands out as a crucial factor: it is well established that factors associated with high punishment sensitivity can lead to a reduced subjective ability to exert effort. Such factors include obsessiveness, depression and neuroticism (e.g. Brinkmann and Gendolla, 2008; Fetterman et al., 2010). This interpretation accords with increased self-report of general psychopathology within this cluster. The finding of lower delay discounting in this cluster replicates the findings of Heerey et al. (2007) and also accords with this view. It is possible that these individuals take on a more prudent approach to risk given high punishment aversion. Interestingly, individuals with Obsessive-Compulsive Personality Disorder also demonstrate decreased delay discounting – a finding related to rigidity and perfectionism (Pinto et al., 2014). Obsessiveness, therefore, may contribute to the pattern of findings observed in this cluster, as supported by their relative elevation on the SCL-90-R Obsession subscale (Table 2). While the current findings do not support a direct relationship between punishment sensitivity per se and delay discounting, then, this somewhat counter-intuitive connection may be explained by the increased obsessiveness (or possibly other neurotic traits) observed in this cluster. This cluster’s elevation on the preoccupation subscale of the ACS (Action Control Scale) further supports this interpretation, as it indicates the interference of negative affect (i.e. hyperactive negatively valenced system). The finding of a significant elevation in boredom propensity in this cluster accords with recent findings regarding boredom (Eastwood et al., 2012; Mercer-Lynn et al., 2011), suggesting that it reflects a state of wanting, but being unable to engage in, meaningful activity. This cluster, in line with this view, evidences intact motivation, but conflicting factors contribute to impairments in engaging in goal pursuit. From an RDoC perspective, this cluster represents dysfunction of negative valence systems in that hypervaluation of anticipated threat (the ‘anxiety’ system) is a prominent marker of this subtype, while positive valence systems remain intact.
Cluster 2: amotivational, or lacking impetus for action
Cluster 2 closely matches traditional descriptions of ‘negative’ schizophrenia (e.g. Andreasen and Olsen, 1982). Individuals in cluster 2 were lower in reward sensitivity than those in cluster 1 and showed substantial energizing deficits. However, they did not self-report dysexecutive symptoms or avolition as currently defined. This pattern of observations would be expected in individuals low in BAS or reward sensitivity (Corr, 2002); they may exert less effort in goal pursuit and experience less subjective distress when thwarted. This finding is also congruent with the findings of Tek et al. (2001) of lower self-reported distress in patients with the deficit syndrome. Importantly, however, this group’s reduced reward sensitivity relative controls did not achieve significance, rather the deficit was observed relative to cluster 1; while this conception may help explain differences among those with schizophrenia, the status of cluster 2 as a deficit population (outside of energizing) is ambiguous and may require replication with greater power. In sum, this group lacks the reward-mediated motivation that serves as an impetus for action and therefore is not distressed by inactivity or the experience of failure, or does not attempt to initiate goal pursuit in the first place. The pattern of neurocognitive impairment in cluster 2 also accords with prior findings. For example, Walton et al. (2009) have shown decreased effort allocation among those with lesions of the anterior cingulate cortex (ACC), a primary substrate of energizing (Stuss and Alexander, 2007) with particular importance to schizophrenia (e.g. Fornito et al., 2009; Pantelis et al., 2003). The computation of effort expenditure is a complex process involving the ACC and subcortical structures (for a review, see Fervaha et al., 2013). Dopaminergic projections in schizophrenia in particular have been shown to play a role in the activation of the ACC (Dolan et al., 1995). This system’s disruption may therefore lead to both the amotivation and related energizing difficulties as observed currently. Per RDoC, this represents dysfunction of positive valence systems, while negative valence systems remain unaffected.
Comparisons with previous findings
A clustered structure of disparate factors underlying goal-directed action in schizophrenia may account for inconsistencies in previous findings. For example, as reviewed by Cohen and Docherty (2004), correlations between negative symptoms in schizophrenia and neuropsychological impairment have been observed in some studies but not others. Kirkpatrick and Galderisi (2008) note several inconsistencies in the literature relating frontal dysexecutive symptoms to negative symptoms. It is possible that differing patterns of cognitive symptomatology between subgroups account for such inconsistencies. In addition, studies using classic neuropsychological tests may produce inconsistent findings given the multidetermined nature of many of these tests, whereas the ROBBIA (Rotman-Baycrest Battery to Investigate Attention) avoids many of these problems by examining homogeneous functions. The findings of Scholten et al. (2006), reviewed above, are also supported and further clarified: these authors’ observation of increased BIS activity, but no difference in BAS activity in schizophrenia, is surprising given models of schizophrenia that suggest decreased impetus for goal pursuit. In the current study, some participants showed a much increased level of punishment sensitivity (an indicator of BIS) without any decrease in BAS, while others showed the opposite pattern. Future research may avoid this confound using the grouping scheme proposed here.
Classical findings of increased anticipatory anhedonia in schizophrenia (Chan et al., 2010; Favrod et al., 2009; Gard et al., 2007) were not replicated in the current study, which instead reflected more recent findings (e.g. Gard et al., 2014) of increased consummatory anhedonia in schizophrenia and no difference in anticipatory anhedonia, adding to the growing evidence contradicting the model of anticipatory anhedonia being responsible for avolition.
Clinical implications
Assessment
Among the underlying factors examined, those showing the greatest elevations included subjective avolitional factors and punishment sensitivity (cluster 1) and energizing (cluster 2). This highlights the importance of attending to both objective testing and subjective report in clinical assessment. For example, a client denying any difficulties with goal pursuit or distress may in fact be amotivated, while a client who reports severe executive difficulties and problems of self-determination without accompanying signs of executive dysfunction may be hampered by personality factors that conflict with intrinsic drives. Attending to the possibility of amotivation, likely accompanied by more marked neuropsychological deficits, in those with limited subjective complaints is therefore a crucial consideration in an assessment context. The heterogeneity and variety of specific deficits shown here to be impacted in SSD, further, highlights the importance of broad assessment strategies to determine any avolitional factors that might underlie reduced goal pursuit, even in the context of apparently intact neuropsychological findings.
Intervention
Each of the two subtypes carries implications for treatment. The motivated but distressed, subjectively avolitional individual (cluster 1) may benefit from psychotherapy addressing the conflicting factors in accomplishing what they are motivated to do. If an individual is hampered by obsessiveness or anxious avoidance, e.g. exposure techniques may be of help. Individuals whose action is hampered by ruminative thought (as per the ‘preoccupation’ model) might best be helped by addressing these processes. Such an approach would be contra-indicated for those who lack strong motivation to begin with (cluster 2). Psychological intervention for this group may seek first to address concerns that these individuals do express subjectively, rather than inactivity per se; or it may seek to capitalize on any extant areas of reward motivation and generalize from these (as per the Premack principle). Pharmacotherapies targeting the distinct systems underlying these processes may also be informed by the current grouping scheme.
Limitations and future directions
This research examined only medicated outpatients with chronic schizophrenia or schizoaffective disorder. It is possible that any goal direction dysfunction in this population is distinct from that seen in first-episode schizophrenia, untreated schizophrenia or inpatients. Thus, the generalization of these findings should be made with caution, and any role of medication in inducing reductions in goal direction cannot be ruled out. An additional limitation relates to listwise deletion of missing data, which may have reduced the capacity to reveal the full heterogeneity of this population despite its necessity given the current statistical approach.
This classification scheme is not intended to replace distinctions among different negative symptoms. Rather, the current study examined goal pursuit as a superordinate concept that may help clarify discussions of its components in a data-driven manner. Further research is therefore required to describe more fully the relationship between the current scheme and existing negative symptom conceptions. While the current approach intentionally did not include existing negative symptom measures into cluster analysis, which would risk reifying pre-conceived theories about their underlying components, further study should attempt to determine what form of underlying dysfunction is reflected in a variety of negative symptom measures.
Future research in negative symptomatology may benefit from the distinctions drawn in the current research. Whereas research in this area has, to date, typically examined avolition and amotivation as synonymous, the application of the current cluster scheme may serve to address inconsistency in this field. Future studies may increase their power considerably by typologizing participants according to the most crucial factors underlying observed reductions in goal-directed action.
Supplemental Material
sj-docx-1-anp-10.1177_00048674221077031 – Supplemental material for Distinct profiles of psychological and neuropsychological functions underlying goal-directed pursuit in schizophrenia
Supplemental material, sj-docx-1-anp-10.1177_00048674221077031 for Distinct profiles of psychological and neuropsychological functions underlying goal-directed pursuit in schizophrenia by Cory J Gerritsen, Joel O Goldberg, Michael Kiang, Gary Remington, George Foussias and John D Eastwood in Australian & New Zealand Journal of Psychiatry
Footnotes
Acknowledgements
The authors wish to thank Dr Maggie Toplak for her input into the manuscript and Dr Donald Stuss for granting permission to use ROBBIA software.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Data Accessibility Statement
The study dataset is available upon reasonable request via email to the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
