Abstract
Contemporary cognitive models of attention deficit hyperactivity disorder, combined type (ADHD-CT) link the core features of the disorder with abnormalities of the executive functions of cognition [1–4]; Tannock [4] outlines that working memory and response inhibition are the most common executive functions of cognition systematically studied in ADHD-CT. Of the latter construct, the inhibition dysfunction hypotheses of ADHDCT have been espoused by a number of researchers and are a focus of current research [1–4]. Oosterlaan et al. [5] completed a meta-analysis of eight studies in which response inhibition was measured using the Stop Task and concluded that there is ‘consistent and robust’ evidence for a deficit in response inhibition in ADHD-CT. Douglas [6] contends that there are deficits in high level (executive) control processes of self-regulation and response inhibition rather than a failure in the ‘elementary’ tasks of perception and attention. Barkley [7] suggests that the core features of ADHD-CT represent a reduction in behavioural control by internally represented information subserved by ‘executive’ neuropsychological functions. Therefore improvement in these core symptoms of ADHD-CT require improvement in the hierarchy of ‘executive’ neuropsychological functions: working memory; speech internalization; selfregulation of affect; and reconstitution. Pennington and Ozonoff [8] reviewed 18 studies of executive functions in ADHD-CT. They reported that 15 found a significant difference between ADHD-CT and control subjects in one or more executive functions. From the total of 60 executive function measures used, 67% were performed significantly worse in the ADHD-CT group and none was significantly better in the ADHD-CT group. The studies by Chelune et al. [9] and Boucugagnani and Jones [10] compared the performance of ADHD-CT and control subjects on the Wisconsin Card Sorting Test (WCST). Both studies reported significant ADHDCT-normal control differences including categories achieved, perseverative errors, and the percentage of correct responses. Shue and Douglas [11] also found significantly more non-perseverative errors were made in an ADHD-CT-normal control comparison using the WCST, among other tests of executive function.
However, few studies have examined the relative contribution of various components of executive function in a systematic manner. This would help identify whether certain executive functions are specifically relevant to ADHD-CT, as has been suggested in other disorders [12–14]. In addition, deficits of executive function have been found in children with ADHD-CT despite the following methodological flaws: small sample sizes; inadequate matching of patient and control groups, particularly language-based learning abilities (reading, spelling, writing and arithmetic); differences in the tasks used to assess different executive functions; and the possibility of experimenter bias on ‘pencil and paper’ neuropsychological tasks. Preventing such flaws will allow more specific and systematic investigation of executive functions in ADHD-CT.
Short-term psychostimulant medication (singles dosage studies and 3–6 week controlled trials) has been consistently associated with improved performance on measures of executive function, particularly measures of verbal and visuospatial working memory and response inhibition [15–18]. To our knowledge, only two studies have investigated the effects of longer-term psychostimulant medication on these executive function measures, and both suggested a better executive function performance. Everitt et al. [19] reported that children with ADHD-CT (n = 7) treated with psychostimulant medication for 12 months had a better capacity to inhibit a perseverative tendancy as indicated by the WCST. Similarly, Kempton et al. [20] found that children with ADHD-CT (n = 15) treated with psychostimulant medication for a mean duration of 23 months had a better executive function performance compared to the unmedicated group of children with ADHD-CT (n = 15), on spatial span, spatial working memory, delayed matching to sample and attentional set-shifting tasks.
In contrast, there is evidence that the short-term improvements in the core symptoms of ADHD-CT are attenuated in a subgroup of children with ADHD-CT [21–23], while other children with ADHD-CT show ongoing improvement [24–26]. The reasons for this attenuation are unclear. Schachar et al. [23] suggest that it may be due to emergent affective adverse effects that are delayed in their onset. They published data on the 4 month follow-up of 91 children with ADHD-CT treated with methylphenidate or placebo in a controlled setting. Affective adverse effects such as mood lability, dysphoria, sudden severe sadness, sudden crying, social withdrawal and aggressive outbursts were delayed in their onset until after the titration with methylphenidate was complete. This may reflect longer-acting clinical effects that arise from a different mechanism of action to the short-acting clinical effects of 30 min to 3 h [27].
Clearly, further systematic investigation of this subgroup of children with ADHD-CT is needed and should begin with executive functions of cognition that are clearly linked with the core features of the disorder [4, 7]. Specific executive functions delivered by computer with well-defined brain behaviour relationships in adequate sample sizes of carefully matched subjects with ADHDCT and controls are required. In this study we aim to compare primary school-age children with ADHD-CT who are: (i) psychostimulant medication-naïve; (ii) treated with longer-term psychostimulant medication but are again symptomatic for ADHD-CT; and (iii) healthy control children. We hypothesize that the two groups of children with ADHD-CT will have a similar pattern of executive function deficits compared to the control children and that these executive function deficits will be better in the longer-term psychostimulant medication group.
Method
Subjects
The total sample consisted of three groups of children: a control group of 26 normal children; a psychostimulant medication-naïve ADHD-CT group of 40 subjects; and a psychostimulant-medicated ADHD-CT group of 26 subjects who had received individually tailored doses of psychostimulant medication: methylphenidate (n = 13) or dexamphetamine (n = 13) (Table 1). The children in the two ADHD groups met the criteria for: (i) a DSM-IV diagnosis of ADHD-CT [28] on a semistructured clinical interview [29]; and (ii) a score greater than 1.5 standard deviations above the mean on the 10-item Abbreviated Conners Rating Scale [30]. The psychostimulantmedicated ADHD-CT group had all been identified clinically as having an initial response to psychostimulant medication but had subsequently become symptomatic again. The subjects were aged 6–12 years. The children in all three groups were carefully matched on age, gender, performance IQ, spelling, arithmetic and reading abilities to control for related differences in executive function performance.
Subject characteristics
The control group consisted of 26 children identified in local primary schools. No control subject met the clinical criteria for ADHD-CT or any other psychiatric condition.
Informed consent to participate in the study was obtained from each child's parent(s). All the children used in this study were living in a family home with at least one family member and were attending normal state schools. All subjects’ intelligence quotients were above 70 and there was no history of neurological impairment, closed head injury, prenatal or peri-natal birth trauma, chronic physical illness, sensory or motor impairment.
Measures
The measures used in this study were: the Child Behaviour Checklist (CBCL) [31]; Abbreviated Conners Rating Scale (ACRS) [30]; Anxiety Disorders Interview Schedule for Children (A-DISC) [29]; Wechsler Intelligence Scale for Children (WISC-3) [32]; Wide Range Achievement Test (WRAT-3) [33]; and the Neale Analysis of Reading Ability Revised [34]. The reader is referred to the extensive published literature for more detailed description of these tests. The main executive function testing procedures were taken from the Cambridge Neuropsychological Test Automated Battery (CANTAB) [35–37], a series of cognitive tests presented on a high-resolution IBM colour monitor with a touch-sensitive screen. Subjects were seated approximately 0.5 m from the monitor and it was explained that they would have to respond to stimuli by touching the screen. All children had reported previous experience with computers although none with a touch-sensitive screen. Five subtests from the battery measuring memory, planning, and attentional set shifting were administered to subjects in a pseudorandom order: spatial span, spatial working memory, attentional setshifting, simultaneous and delayed matching to sample (DMTS), and the pattern/spatial recognition tasks. The testing session lasted approximately 60 min. The tasks used in this study have been standardized in a large group of normal subjects [37] and in normal children [38]. Task administration in this study was according to standard protocols. The reader is referred to the extensive literature for detailed description of the CANTAB tasks.
Procedure
The children with ADHD-CT were tested at the Alfred Hospital and Maroondah Hospital Child and Adolescent Mental Health Services. The psychostimulant medication-treated group was tested from 90 to 120 min after ingesting their morning dose of medication to ensure peak psychostimulant medication clinical effect [39]. Testing took approximately 2 h and was conducted over two sessions: during the first session, a trained clinical psychologist administered the achievement tests to the child. The computerized neuropsychological tests were administered in a second session. The control children were assessed in their own homes and usually received both forms of testing on the same day. The CBCL parent form [31] was completed prior to the child's assessment. A senior registrar in child and adolescent psychiatry administered the A-DISC parent-form [29] while each child was tested on the Neale Analysis of Reading Ability Revised [34] to provide a measure of reading accuracy and comprehension, the WISC-3 [32] to provide verbal, performance and full-scale scores as a measure of intelligence and the third edition of the WRAT-3 [33] to provide a measure of spelling and arithmetic abilities.
Statistical analysis
Data analysis was performed using the Statistical Package for the Social Sciences (SPSS/PC) [40]. Subject characteristics were compared across the three groups using one-way analysis of variance (ANOVA) (Table 1). Performance on the spatial working memory task and the DMTS task were compared with a repeated measures ANOVA using a two-factor design which included a between-subjects factor (group) and a within-subjects factor (difficulty/delay level). To protect against violation of the homogeneity of covariance assumption, analysis of within-group effects or interactions with degrees of freedom greater than one were examined using a repeated measures design within MANOVA (Wilk's Multivariate test of significance). Performance on the spatial span task, the spatial working memory strategy measure, the spatial and pattern recognition task, and the simultaneous and delayed matching to sample task were compared between groups using one-way ANOVA. Where the omnibus F was significant, the post-hoc Studentized Newman-Keuls (SNK) procedure (p < 0.05) was conducted to determine the source of this significance. Performance on the attentional set-shifting task was also examined by comparing the percentage of subjects who succeeded in reaching criterion at each stage of the task. The resulting contingency tables were analyzed using the likelihood ratio method, which allows for use where cell frequencies were small, with the resulting statistic 2i being distributed as χ 2 [41]. Where a significant difference was found between the groups, the estimated effect sizes were calculated using Cohen's d statistic.
Results
Table 2 presents the comparison of the group performance means for the executive function tasks.
Comparison of group performance means for executive function tasks
The medication-naïve ADHD-CT group had significantly shorter spatial span than the control group (F [2,89] = 8.00, p < 0.01), while the medication-naïve ADHD-CT group had significantly more between search errors (defined by returning to an ‘empty’ box already targeted on a particular search) than the medicated ADHD-CT group and the control group (F [2,89] = 4.90, p < 0.01). There were no differences between groups for strategy score on the spatial working memory task. Figure 1 shows the mean between-search errors for each group according to the difficulty level of the task. There was a significant effect of group (F [2,89] = 4.28, p < 0.01) and task difficulty (Wilk's λ = 0.07, F [2,89] = 222.20, p < 0.001), and an interaction between the group and difficulty factors (Wilk's λ = 0.79, F [2,72] = 2.02, p < 0.05).
Mean number of ‘between search’ errors at each level of difficulty of the spatial working memory task. BSE2, between search errors 2 boxes; BSE3, between search errors 3 boxes; BSE4, between search errors 4 boxes; BSE6, between search errors 6 boxes; BSE8, between search errors 8 boxes.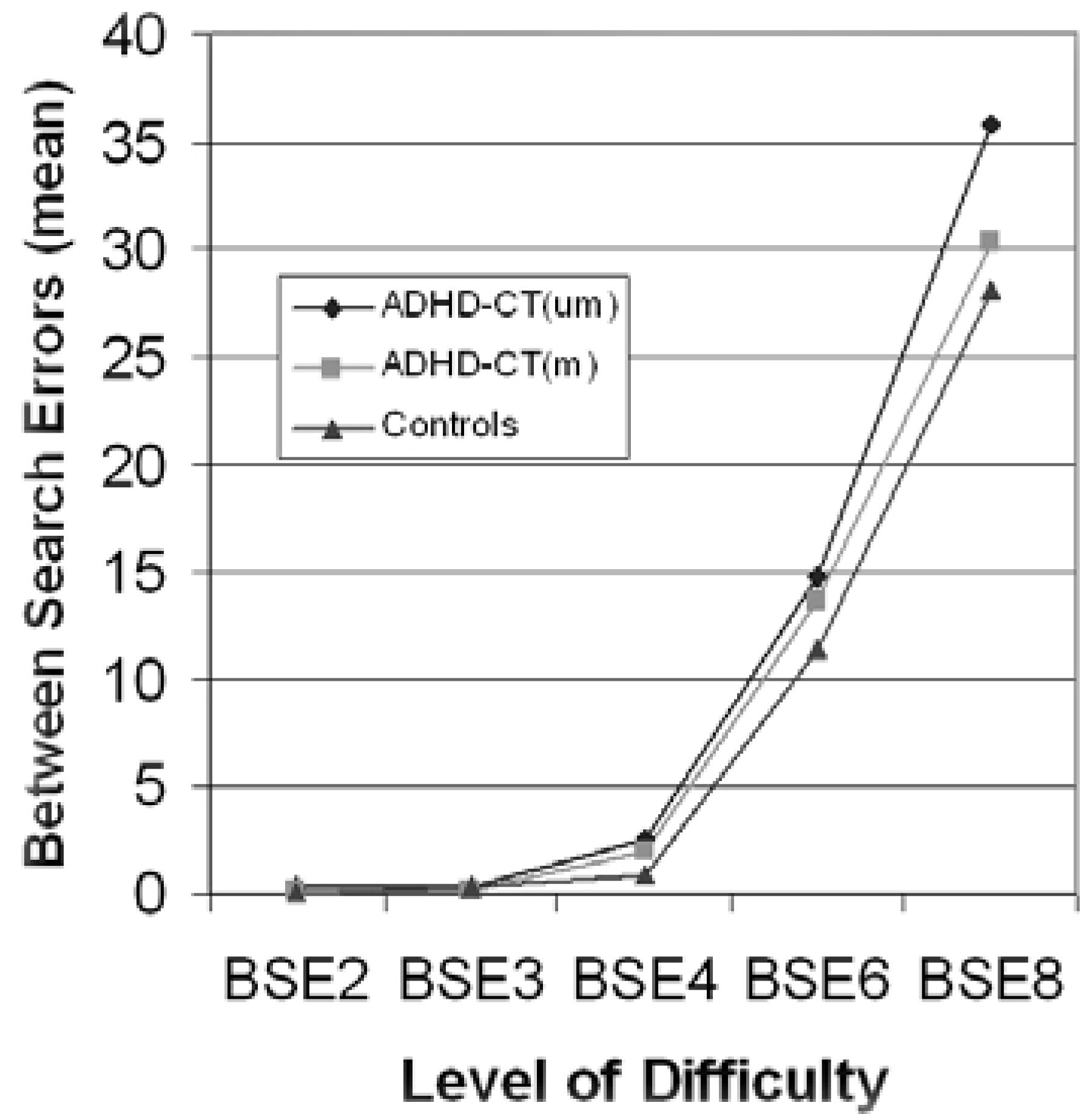
The medication-naïve ADHD-CT group made significantly more errors than the control group in the simultaneous (F [2,89] = 3.26, p < 0.05) and both the medicated ADHD-CT and the control groups in the three delay conditions (F [2,89] = 11.00, p < 0.01). Figure 2 shows the mean number of correct responses for the simultaneous and the three delay conditions of the DMTS task. There was a significant effect of group (F [2,89] = 6.80, p < 0.005) and task difficulty (Wilk's λ = 0.32, F [2,89] = 47.17, p < 0.001), but no interaction between the group and difficulty factors (Wilk's λ = 0.85, F [2,172] = 1.88, p = 0.09).
Mean number of correct responses across simultaneous and three delay conditions on the delay matching to sample (DMTS) task. Sim, simultaneous matching to sample; DMTS–0, delayed matching to sample 0 s delay; DMTS–4, delayed matching to sample 4 s delay; DMTS–12, delayed matching to sample 12 s delay.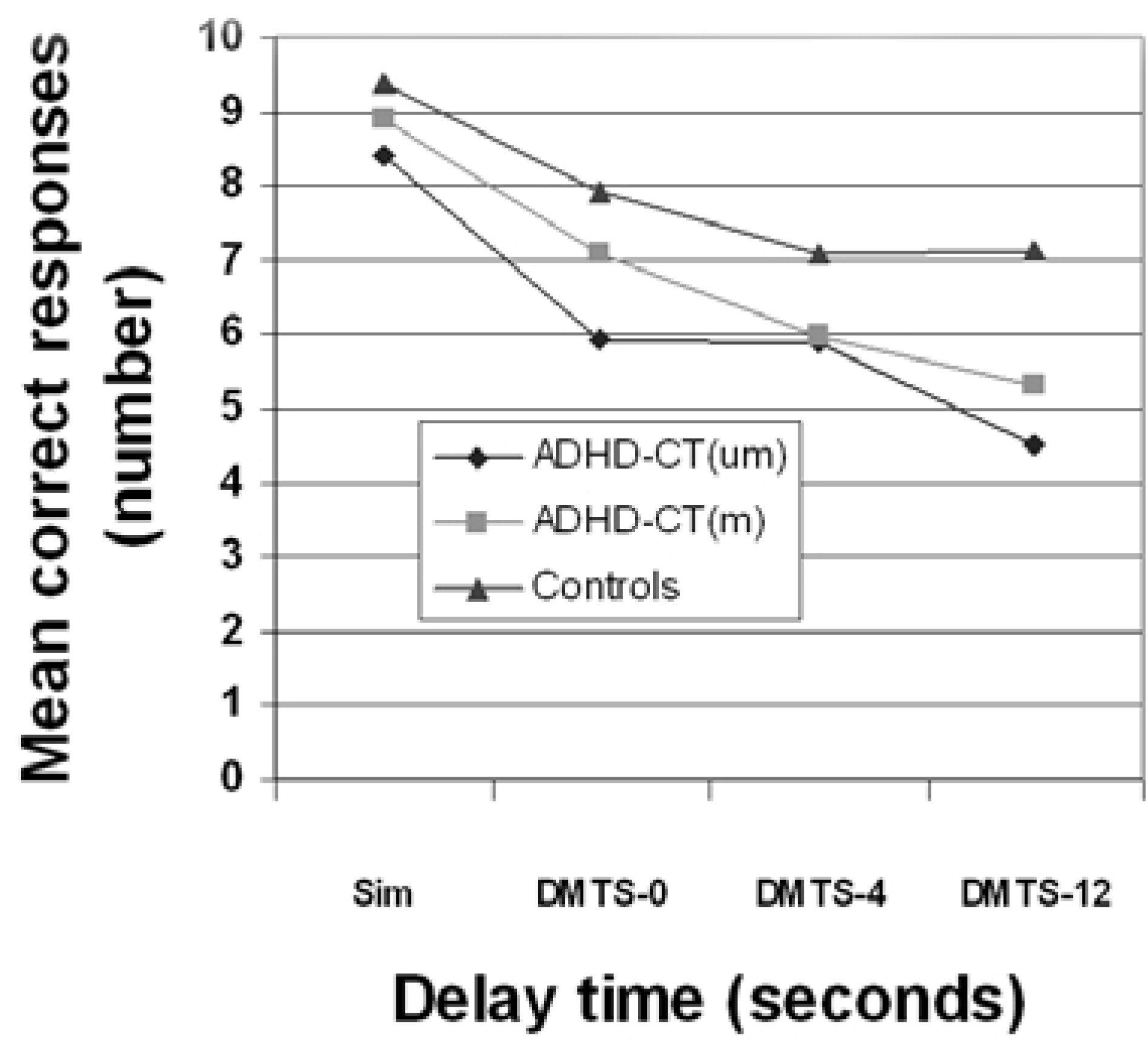
The medication-naïve ADHD-CT group recognized significantly fewer spatial locations correctly compared to the control group (F [2,89] = 7.08, p < 0.01).
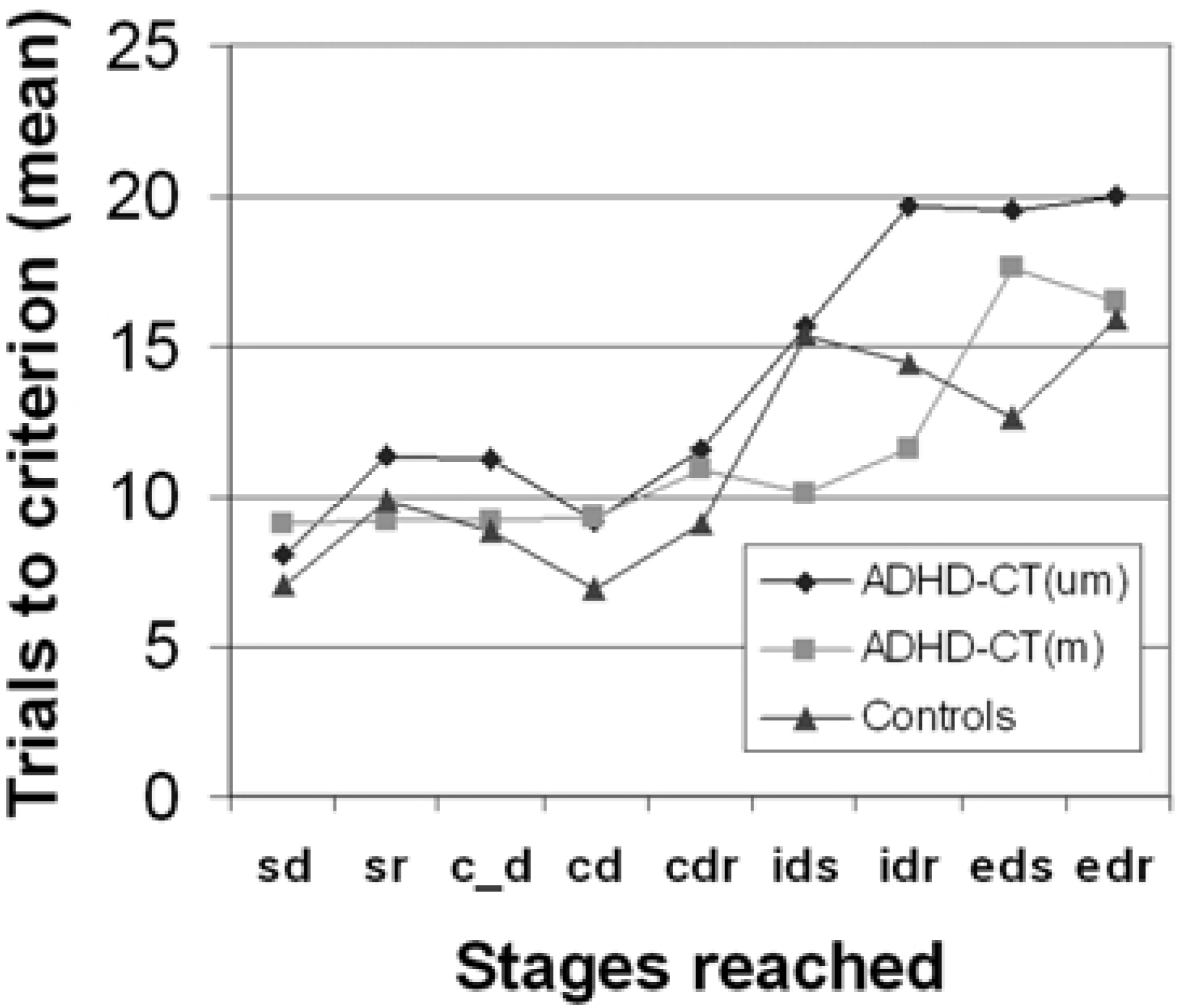
The medication-naïve ADHD-CT group had significantly fewer subjects complete all stages of the attentional set-shifting task successfully (2i = 10.12, p < 0.01). The proportion of subjects passing each stage of the task was also analyzed non-cumulatively by analyzing the data for those subjects actually attempting each stage (Fig. 3). More medicationnaïve ADHD-CT subjects failed the task at the extra-dimensional shift (EDS) (2i = 6.89, p < 0.01) and the extra-dimensional reversal (EDR) (2i = 10.12, p < 0.01) stages. The numbers of trials required to attain criterion at each stage were also analyzed non-cumulatively (Fig. 4). The medication-naïve ADHD-CT subjects required more trials to reach criterion than the controls and the medicated ADHD subjects, at the intra-dimensional reversal (IDR) (F [2,89] = 4.39, p = 0.01) stage, and more trials to reach criterion than the control group at the EDS stages (F [2,89] = 2.96, p = 0.05).
Cumulative failure at each stage of the visual discrimination learning/attentional set-shifting task. Stages: sd, simple discrimination; c_d, compound discrimination; cdr, superimposed compound discrimination reversal; idr, intradimensional reversal; edr, extradimensional reversal. Mean trials to criterion at each stage of the visual discrimination learning/attentional set-shifting task. Stages: sd, simple discrimination; sr, simple reversal; c_d, compound discrimination; cd, superimposed compound discrimination; cdr, superimposed compound discrimination reversal; ids, intradimensional shift; idr, intradimensional reversal; eds, extradimensional shift; edr, extradimensional reversal.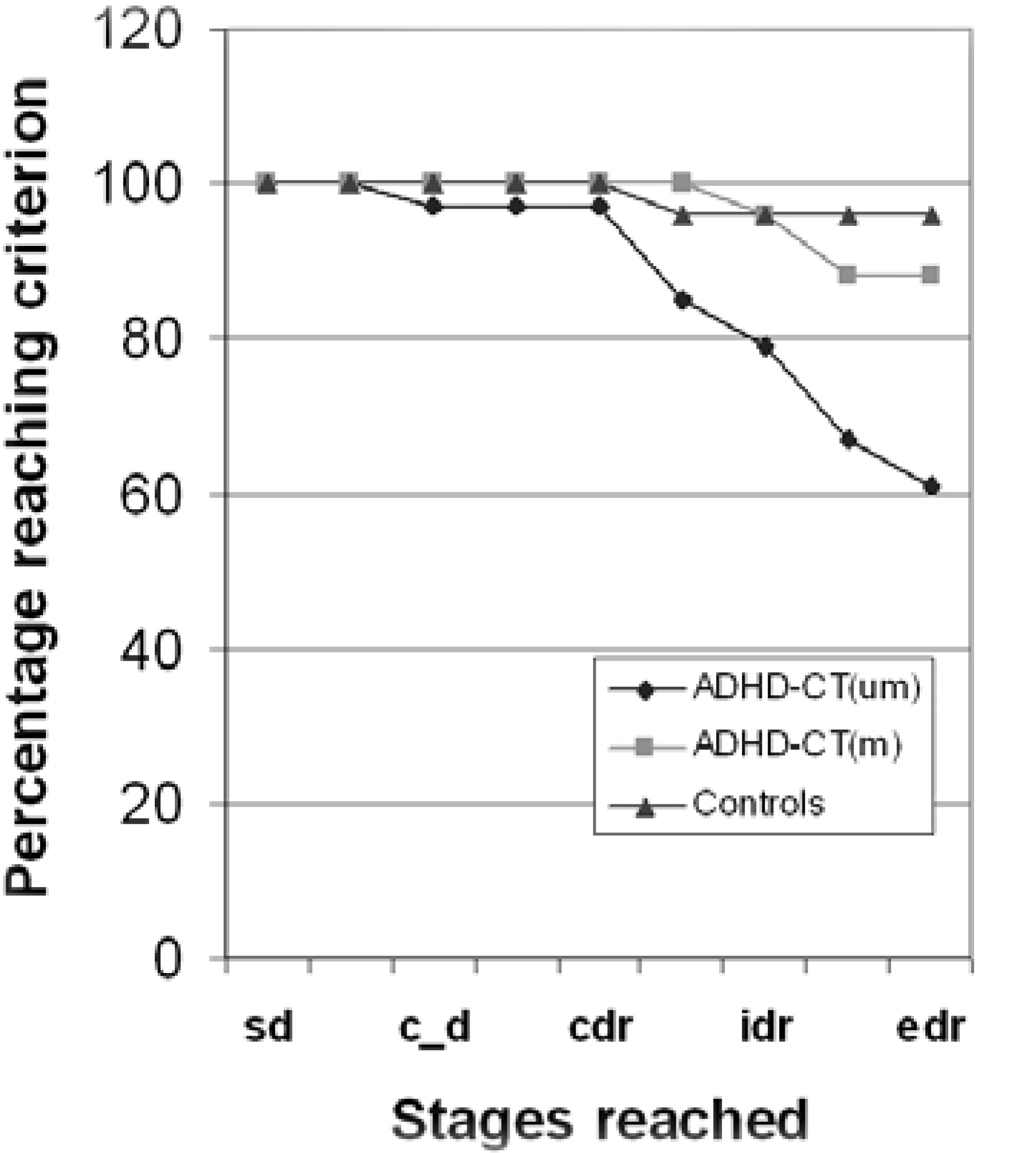
Discussion
The pattern of executive function deficits in primary school-age children with ADHD-CT is similar to that reported by Kempton et al. [20] except for the simultaneous matching to sample task where a significant increase in error rate was noted in the medication-naïve children with ADHD-CT compared to the control group in this sample. This may be associated with the better verbal and full scale IQ scores in the control group in this sample compared to the Kempton et al. [20] sample. Nevertheless, the replication of this pattern of executive function deficits reflects the careful definition of ADHDCT, using categorical and dimensional multi-informant measures, careful matching of the medication-naïve/ medicated ADHD-CT and control groups, and the use of computerized tasks of primarily non-verbal executive function in the three groups matched for performance IQ. This helps obtain a more accurate perspective of executive function in children with ADHD-CT, not obscured by comorbid language-based learning difficulties, even when such difficulties are controlled (Table 1).
In contrast, the effect of psychostimulant medication did differ in some tests of executive function: for example, spatial span was not significantly better in the medicated ADHD-CT group, while between-search error rates with increasing task difficulty of the spatial working memory task were significantly better in the medicated ADHD-CT group compared to the medicationnaïve group. These differences were not associated with age, gender, performance IQ, or language-based learning ability effects between the Kempton et al. [20] sample and this current sample, although the medicated ADHDCT group in the latter sample was marginally older and had slightly higher performance IQ and reading accuracy and comprehension. Nor were they associated with a different timing of medication dose to test regimen. They may reflect the known heterogeneity of biological and psychosocial risk factors predisposing to ADHD-CT. However, a better performance in more than one type of executive function in primary school-age children with ADHD-CT who have been treated with longer-term psychostimulant medication has again been demonstrated in a larger sample. In addition, the medicated children were still symptomatic of ADHD-CT despite its use. However, these results have to be viewed with caution because the medicated ADHD-CT group in this study may have differed from the medication-naïve group before treatment began: for example, differences in parental perception of symptoms, possible differences in co-occurring disorders and the children with ADHDCT whose executive function may not improve with psychostimulant medication may have discontinued treatment, introducing a selection bias into the medicated group studied. A longitudinal study design is required for the two limitations above to be controlled. A longitudinal study design would also allow the important intra-individual along with the inter-individual variations in cognitive, emotional and behavioural development in childhood to be controlled, although the potential confounds of practice effects and maturational effects require a carefully matched control group.
From a clinical perspective, improved executive function as a marker of psychostimulant medication effect in the longer-term would allow the titration of this group of medications in a given child, the determination of the optimal individual dose and the relative effectiveness of psychostimulants to be more accurately determined. In addition, further clinical assessment of ongoing ADHDCT symptoms could be focused on a number of the following potential causative factors: co-occurring oppositional defiant disorder, conduct disorder, anxiety and depressive disorders, learning difficulties, medication adverse effects, parenting style, family systemic functioning, and factors in the school environment.
Footnotes
Acknowledgements
The 1997 Eli Lilly Psychiatry Fellowship awarded to Dr Vance supported this work. We acknowledge the support of Professor Bruce Tonge, Associate Professor Ernest Luk, Jan Costin and Marilyn Boots, Dr Liz Reed.
