Abstract
Objective:
While diagnostically independent, autism and schizotypal disorders can co-occur. Their concurrent impact on outcomes and phenotypes has not been investigated. We investigated the impact of comorbid autism and schizotypal disorders in children on executive functioning and socio-pragmatic skills – core features of both disorders.
Method:
Executive functioning (assessed with the Cambridge Neuropsychological Test Automated Battery) and socio-pragmatic skills (assessed using the Melbourne Assessment of Schizotypy in Kids) were investigated in a total of 67 (6–12 year old) children with autism (n = 15; M/F = 10/5), schizotypal disorder (n = 8; M/F = 5/3) and comorbid autism and schizotypal disorder (n = 12; M/F = 5/7) and typically developing children (n = 32; M/F = 17/15).
Results:
Both the autism and schizotypal disorder groups performed more poorly than the typically developing group on socio-pragmatic skills and overall performance (i.e. number of stages completed) of the intra-/extra-dimensional set-shifting task (all ps < 0.001). Clear distinctions between the autism and schizotypal groups were present in the intra-/extra-dimensional task relative to the typically developing group – the autism group had difficulties with extra-dimensional shifts (p < 0.001), and the schizotypal disorder group with intra-dimensional shifts (p = 0.08). Interestingly, the overall performance of the comorbid group on the intra-/extra-dimensional task was not significantly different from the typically developing group, and they were superior to both the autism (p = 0.019) and schizotypal disorder (p = 0.042) groups on socio-pragmatic skills.
Conclusion:
The phenotypical overlap between autism and schizotypal disorders may be precipitated by different cognitive styles and/or mechanisms associated with attention and information processing. We propose that sustaining and switching attention represent two poles of irregularities across the autism and schizotypal spectra, which appear to converge in a compensatory manner in the comorbid group. Our findings highlight the importance of investigating children with a dual diagnosis of autism and schizotypal disorders, and raise intriguing questions about possible mechanisms to explain the attenuated impairment observed in the group of children with comorbid autism and schizotpyal disorders.
Introduction
Autism spectrum disorder (ASD) and schizotypal disorder (SD) are considered diagnostically independent (APA, 2013). Individuals with SD are characterized by cognitive and perceptual distortions, including odd behaviour, bizarre and fantastical ideation and preoccupation with self-referential thoughts and fantasy events (Asarnow, 2005; Roberts et al., 2001). On the other hand, individuals with ASD are characterized by pervasive social interaction difficulties, communication deficits and restricted behaviours and interests (APA, 2013). However, the nosologic separation between them is not clear (Lugnegard et al., 2012), particularly in light of accumulating evidence suggesting that SD and ASD can co-occur at both the diagnostic and trait levels. For example, reports show that 41% of adolescents with ASD (ages 11–18) met the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision (DSM-IV-TR) criteria for SD (Barneveld et al., 2011). Moreover, schizotypal symptoms are found at significant levels in children with ASD (ages 6–12) (Gadow, 2012), and vice versa (Esterberg et al., 2013). This raises important questions about the nature of their association and the effect of their co-occurrence on the individual’s phenotype and functional outcome.
The literature on children with SD is extremely limited, which is probably precipitated by the debate surrounding the diagnostic validity and stability of SD (Esterberg et al., 2010). There is only one study that has directly compared ASD and SD (Wolff and Barlow, 1979), which showed that the conditions were separable by the presence of fantasies and imaginations in the SD group. There is also one study that has examined differences between children with schizophrenia and children with comorbid schizophrenia and pervasive development disorders or autism (Sporn et al., 2004). This study reported that the comorbid group did not differ from the non-comorbid group at baseline or at 2- to 6-year follow-up on any of the clinical and neuropsychological measures, which included cognitive performance measures (IQ), the clinical global impression scale, scales for the assessment of positive and negative symptoms and the brief psychiatric rating scale.
Research comparing children with ASD and SD is important due to increased evidence linking them to schizophrenia spectrum disorders (SSD). SD, which has been identified in children as young as 6 years of age (Asarnow, 2005; Roberts et al., 2001), presents with symptoms that are similar to the schizophrenia prodrome and the ‘at risk mental state’ for psychosis (Yung et al., 1998). Longitudinal evidence suggests that youths with SD have a 17–25% chance of developing psychosis within a 1- to 3-year period (Esterberg et al., 2010). An independent line of research has identified important etiologic overlaps between ASD and SSD, leading to the suggestion that ASD may represent a gateway to SSD (Toal et al., 2009). Autism and autism traits during childhood have been associated with the presence of psychotic experiences in adolescence (Sullivan et al., 2013), and pervasive developmental disorders precede childhood-onset schizophrenia in 30–50% of children (Rapoport et al., 2009). Furthermore, there is increased risk of ASD in the offspring of parents with SSD, as well as increased risk for ASD in siblings of patients diagnosed with schizophrenia (Sullivan et al., 2012). Therefore, comparing ASD vis-à-vis children with SD will inform our understanding of the nature of their association, the impact of comorbidity on cognitive and behavioural phenotypes and inform research concerned with the neurodevelopmental origins of schizophrenia (Debbane and Barrantes-Vidal, 2015; Gogtay et al., 2011).
The current study is the first to investigate social and executive functioning in children with ASD, children with SD, children with comorbid ASD and SD and typically developing (TD) children. Socio-pragmatic skills were assessed using the Melbourne Assessment of Schizotypy in Kids (MASK; see Method for details), which reliably identifies children with SD and distinguishes these children from TD and from those with ASD (Jones et al., 2015). This measure shows that, while both ASD and SD have similar socio-pragmatic difficulties relative to TD children, children with SD exhibit significantly more positive symptoms (paranoid/magical thinking, bizarre fantasies and unusual perceptual experiences) than children with ASD. Executive functioning was assessed with the Cambridge Neuropsychological Test Automated Battery (CANTAB). We focused on the CANTAB tasks of executive function due to their sensitivity to performance deficits in schizophrenia (Pantelis et al., 1997, 1999) and in childhood disorders (ASD and children with attention deficit hyperactivity disorders [ADHD]) (Kempton et al., 1999; Ozonoff et al., 2004), as well as across normal development (De Luca et al., 2003).
We predicted that the clinical groups would perform more poorly than the TD group on both social and executive functioning. Moreover, given that children with either ASD or SD are known to have marked difficulties with social and executive functioning relative to healthy controls (Abu-Akel et al., 2000; Lenzenweger et al., 1991; Pilowsky et al., 2000), and evidence for shared etiological and risk factors between these conditions (Carroll and Owen, 2009; Chisholm et al., 2015; Rapoport et al., 2009; Sullivan et al., 2012), we hypothesized that (1) both the SD and ASD groups would exhibit similar levels of impairments on both social and executive functioning relative to the TD group and (2) the comorbid group would show poorer performance on both social and executive functioning compared with either the ASD or SD groups. The latter is based on the assumption that the combined presence of ASD and SD would cumulatively lead to further dysfunction in executive and social abilities. We also examined differences between the groups on the expression of positive symptoms as assessed by the MASK, and (3) predicted that the SD and comorbid groups would exhibit more positive symptoms than either the ASD or TD groups.
Method
Participants
In all, 67 children participated in the study (mean age = 9.64; SD = 2.06; range = 6–12; 37 male; 30 female). The clinical cases were recruited from two neurodevelopmental disorders clinics in Melbourne, and the TD children from the general community (e.g. sporting organizations, schools and recreational clubs). The children formed four groups: children with ASD (n = 15), children with SD (n = 8), children with comorbid ASD and SD (n = 12) and TD children (n = 32) (see Table 1 for further details). Allocation to these groups was made after each diagnosis was clearly confirmed by the research team (see clinical assessment below for diagnostic tools and process). Children were excluded if they had a head injury, a neurological condition or an intellectual disability defined as a full scale IQ (FSIQ) score <70 on the Wechsler Intelligence Scale for Children-Fourth Edition (WISC-IV) (Wechsler, 2003). Melbourne Health Research Ethics Committee and Monash University Research Ethics Committee approved the study. Written consent was obtained from the children’s parents or guardians.
Demographics, intellectual abilities, and medication information by group.
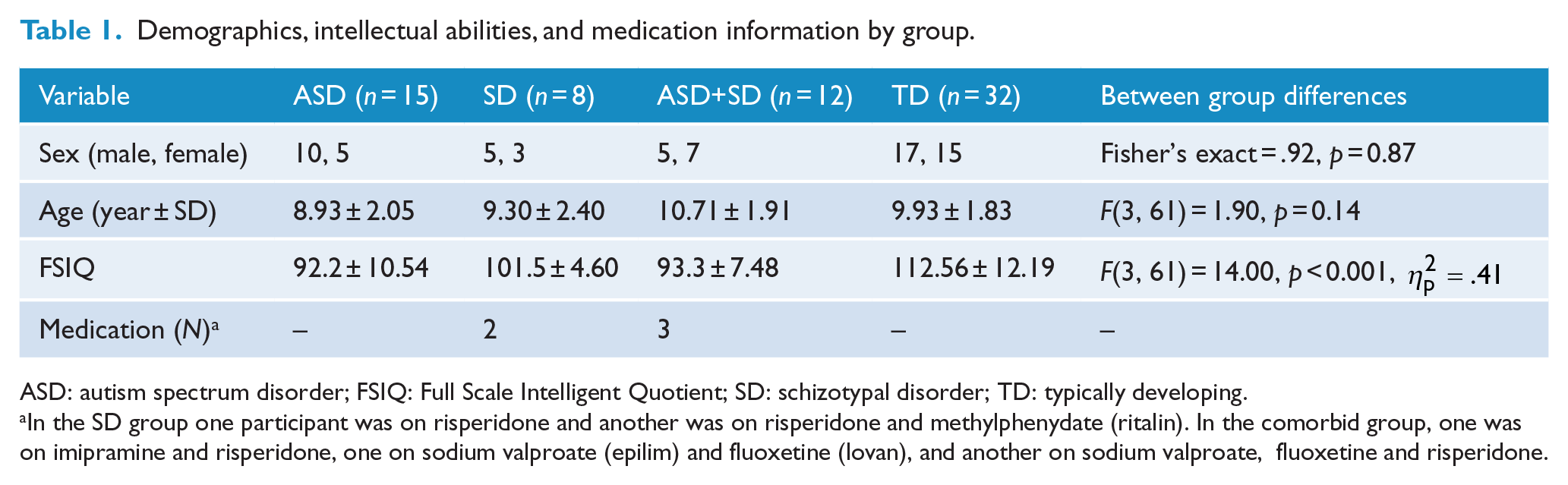
ASD: autism spectrum disorder; FSIQ: Full Scale Intelligent Quotient; SD: schizotypal disorder; TD: typically developing.
In the SD group one participant was on risperidone and another was on risperidone and methylphenydate (ritalin). In the comorbid group, one was on imipramine and risperidone, one on sodium valproate (epilim) and fluoxetine (lovan), and another on sodium valproate, fluoxetine and risperidone.
Clinical assessment
Diagnoses of ASD, SD and comorbid ASD-SD were made prior to recruitment through the two multidisciplinary neurodevelopmental disorders assessment clinics in which either Tonge (child psychiatrist) or Testa (neuropsychologist) participated. The assessments used a structured in-depth psychiatric interview providing DSM-IV-TR criteria diagnoses (Einfeld and Tonge, 1991, 2002).
Diagnostic assessments, which contributed to a larger clinical research protocol (Jones et al., 2015) included (1) a neuropsychological assessment (the CANTAB) and the Behaviour Rating Inventory of Executive Function (BRIEF) (Gioia et al., 2000), (2) a speech and language assessment, (3) ASD assessment (the Autism Diagnostic Observational Schedule – 2nd Ed. [ADOS] (Lord et al., 2000) conducted by research standard trained clinicians [Testa and clinicians at the Centre for Developmental Psychiatry and Psychology]), and (4) behaviour assessment (the Behavioural Assessment System for Children-2nd Edition [BASC-II] (Reynolds and Kamphus, 2004), the Developmental Behaviour Checklist [DBC] which includes an autism screening algorithm and is completed by the parent/teacher (Einfeld and Tonge, 2002), and the Conner’s Rating Scale–Revised [CRS-R] (Conners, 1997) to assess ADHD symptoms). This assessment process provided a multiaxial DSM-IV-TR diagnostic formulation including any comorbid conditions, which relied on clinical expertise and judgment using the assessment tools as the gold standard. Children were then screened for exclusion criteria before being allocated to each group. One family with a child diagnosed with ASD declined to participate due to travel distance.
ASD group
Those in the ASD group received a DSM-IV-TR criteria diagnosis of either Asperger’s disorder (n = 8) or autistic disorder (n = 7) from the multidisciplinary assessment process described above, with a high inter-rater agreement between experienced independent clinicians (Cohen kappa = 0.98).
SD group
Subjects were allocated to the SD group if they met DSM-IV-TR criteria for SD (diagnosed by a multidisciplinary team described above). To establish diagnostic reliability, authors Tonge and Testa, both independently assessed eight children in this group (40%), achieving agreement on the diagnosis of SD for all of these children. Importantly, Tonge and Testa were blind to the assessment of the other clinician for the purposes of independent assessment. This step was considered necessary because there is no gold standard for the diagnosis of this disorder in children. Two children in the SD group had a comorbid diagnosis of ADHD, and one a diagnosis of reactive attachment disorder. Exclusion based on these preexisting diagnoses was not undertaken due the high comorbidity rate of these conditions.
ASD+SD group
Of those with SD, 12 children were confirmed by the multidisciplinary assessment described above to also meet the diagnostic criteria for ASD.
TD group
TD children completed the BASC (Reynolds and Kamphus, 2004), BRIEF (Gioia et al., 2000), DBC (Einfeld and Tonge, 2002), and CRS-R (Conners, 1997). In addition, school performance reports were provided to exclude the presence of a psychiatric disorder, learning or developmental delay. All children and their primary caregivers underwent an initial screening interview to assess exclusion criteria. These criteria included a history of abuse, neglect; intellectual disability (IQ < 70); neurological insults or head injury and other comorbid neurological conditions (APA, 2013).
Measures
Children’s overall intellectual abilities were measured using the WISC-IV (Wechsler, 2003), appropriate for children between 6 and 12 years of age. Socio-pragmatic skills were measured with the MASK (Jones et al., 2015). Executive functioning was assessed with the CANTAB. Inhibition was also measured by calculating the interference score on the Stroop test. Below is a brief description of the MASK and each of the cognitive assessments.
The MASK
The MASK is a semi-structured tool that measures SD features in children aged between 5 and 12 years (Jones et al., 2015). It consists of 57 items that are grouped into 9 domains: social anxiety, social skills, motor abilities, language/thought/ideation, fantasy/magical thinking, unusual perceptual experiences, behaviour, attention and affect. Each item is rated on a 4-point Likert scale (Never, Sometimes, Often, Always) by the child’s clinician only after obtaining information from both the child and parent. Scores on the MASK range from 57 to 228. The MASK has excellent internal consistency (Cronbach’s α = 0.98), high inter-rater reliability (.98; 95% confidence interval [CI] = [0.941, 0.993]), good convergent validity with both BASC-II (Reynolds and Kamphus, 2004) and the CRS-R (Conners, 1997). The MASK has excellent diagnostic utility differentiating ASD and SD (Jones et al., 2015). A receiver operating characteristic curve suggested that a cut-off score of 132 is a good indicator of SD (Jones et al., 2015). The MASK consists of two factors: factor 1 corresponds to socio-pragmatic skills and factor 2 corresponds to the child’s positive schizotypal symptoms (see Jones et al., 2015). Factor loadings determined by regression analysis of the positive symptoms and socio-pragmatic skills factors were used in subsequent analyses. Positive regression coefficients indicated poorer performance.
The intra-/extra-dimensional set-shifting task
The intra-/extra-dimensional set-shifting task (IDED) of the CANTAB measures discrimination learning, reversal learning and set-shifting ability. A detailed description of this task is available elsewhere (Pantelis et al., 1999). In short, the IDED task is comprised of nine stages and two stimulus dimensions (coloured shapes and white lines). Participants were presented with a forced choice between two coloured shapes (stages 1 and 2), two coloured shapes with white lines adjacently placed (stage 3) or two coloured shapes with overlying white lines (stages 4–9). Stages 1 through 5 ‘taught’ the examinee to respond to the shapes while ignoring the lines. In stage 6, children were presented with a new set of coloured shapes and white lines, but were still required to respond to the shapes while ignoring the lines. This shift is considered an intra-dimensional shift. In stage 8, the stimuli were similarly altered but now children were required to make an extra-dimensional shift by responding to the white lines instead of the coloured shapes. Stages 1, 3 and 4 capture discrimination learning. Stages 2, 5, 7 and 9 capture reversal learning that required the child to respond to the opposite stimulus within the same category. Participants progressed through the stages by responding correctly to six consecutive stimuli. The task was discontinued if they were unable to fulfill this criterion within a single stage after 50 attempts. Group differences were examined on the following five IDED variables: (1) the number of stages completed, (2) discrimination errors (stages 1, 3 and 4), (3) reversal errors (stages 2, 5, 7, and 9), (4) intra-dimensional errors (stage 6) and (5) extra-dimensional errors (stage 8).
The spatial working memory
The spatial working memory (SWM) task of the CANTAB assesses non-verbal working memory. Children were, by process of elimination, required to find blue tokens that were hidden within coloured boxes arranged on the touch-screen computer. The child was asked to move found tokens to a column on the right hand side of the screen and continue searching until they had found enough tokens to fill the column. Importantly, each child was informed that once a token was found, that particular box would remain empty for the remainder of that trial. Participants completed four trials within four difficulty levels: three boxes, four boxes, six boxes and eight boxes. Instances of the child returning to a previously emptied box within the same trial (between-search errors) were recorded, as were the number of times the child returned to an empty box during the same search (within-search errors). Between-search errors reflect lapses in working memory, while within-search errors reflect lapses in attentional focus. A strategy score was also generated by assessing how often a search within any particular trial commenced with a different box; a lower score indicates a more systematic search strategy, which helps to improve SWM performance. Group differences were examined on the following three SWM task variables: (1) between-search errors, (2) within-search errors and (3) search strategy scores.
Short term spatial span (SS)
The spatial span (SS) task of the CANTAB is a test of non-verbal short-term memory capacity. The child was presented with nine white squares on a black background. These squares individually changed colour, and the child was required to recall gradually increasing sequences. If the child made an error, the same number of squares was presented in a different sequence. The test was discontinued when the child failed three trials within a level.
The stroop colour-word test
The stroop colour-word test examines response inhibition on the assumption that reading is a more automatic response for humans than naming colours. Interference score for each participant was calculated as follows: Interference = C × W − (((216 − W) × C)/(216 − W) + C) (Chafetz and Matthews, 2004), where C denotes colour and W denotes word. Higher scores represent better performance.
Results
There were no differences among the groups in sex distribution or age. However, there were significant differences in FSIQ, with TD children scoring significantly higher than both the children with ASD (p < 0.001, Cohen’s d = 1.79) and the children with comorbid ASD+SD (p < 0.001, Cohen’s d = 1.90) (see Table 1). There were no other group differences in FSIQ. Moreover, age correlated with a number of measures as follows: positively with the number stages completed of the IDED task (r = 0.49, p < 0.001) and with better performance on the stroop task (r = 0.42, p < 0.001), and negatively with errors on the reversal and extra-dimensional trials of the IDED task as well as with within-, between- and strategy-search errors of the SWM task (−0.35 > rs > −0.69; all ps < 0.001).
Given the relatively large number of outcome measures, group differences on the CANTAB and MASK scores were first examined using a multivariate analysis of covariance (MANCOVA), controlling for age, sex and FSIQ, to protect against false positives (Snijders and Bosker, 2012). The MANCOVA indicated significant differences among the groups, Pillai’s Trace v = 1.78, F(36, 147) = 5.97, p < 0.001, ηp2 = .59, with statistical differences (see Table 2) discerned in the following: number of stages completed on the IDED task, the reversal trials of the IDED (stages 2, 5, 7 and 9), the intra-dimensional trials (stage 6) of the IDED and the extra-dimensional trials (stage 8) of the IDED. There were also statistically significant differences among the groups for positive symptoms and socio-pragmatics skills of the MASK. As can also be seen from Table 2, there were no significant differences between the groups on the following measures: discrimination errors (stages 1, 3 and 4) of the IDED, non-verbal short-term memory capacity (SS), response inhibition (stroop interference) and SWM measures for between errors (working memory load), within errors (reflecting attention) and strategy (strategic ability). In the following sections (‘Group differences on the IDED measures’ and ‘Group differences on the MASK’), we report group differences on the various measures using Tukey’s-corrected pairwise comparisons (six comparisons in all), to account for comparisons among groups of unequal sizes.
Clinical and neuropsychological characteristics by group.
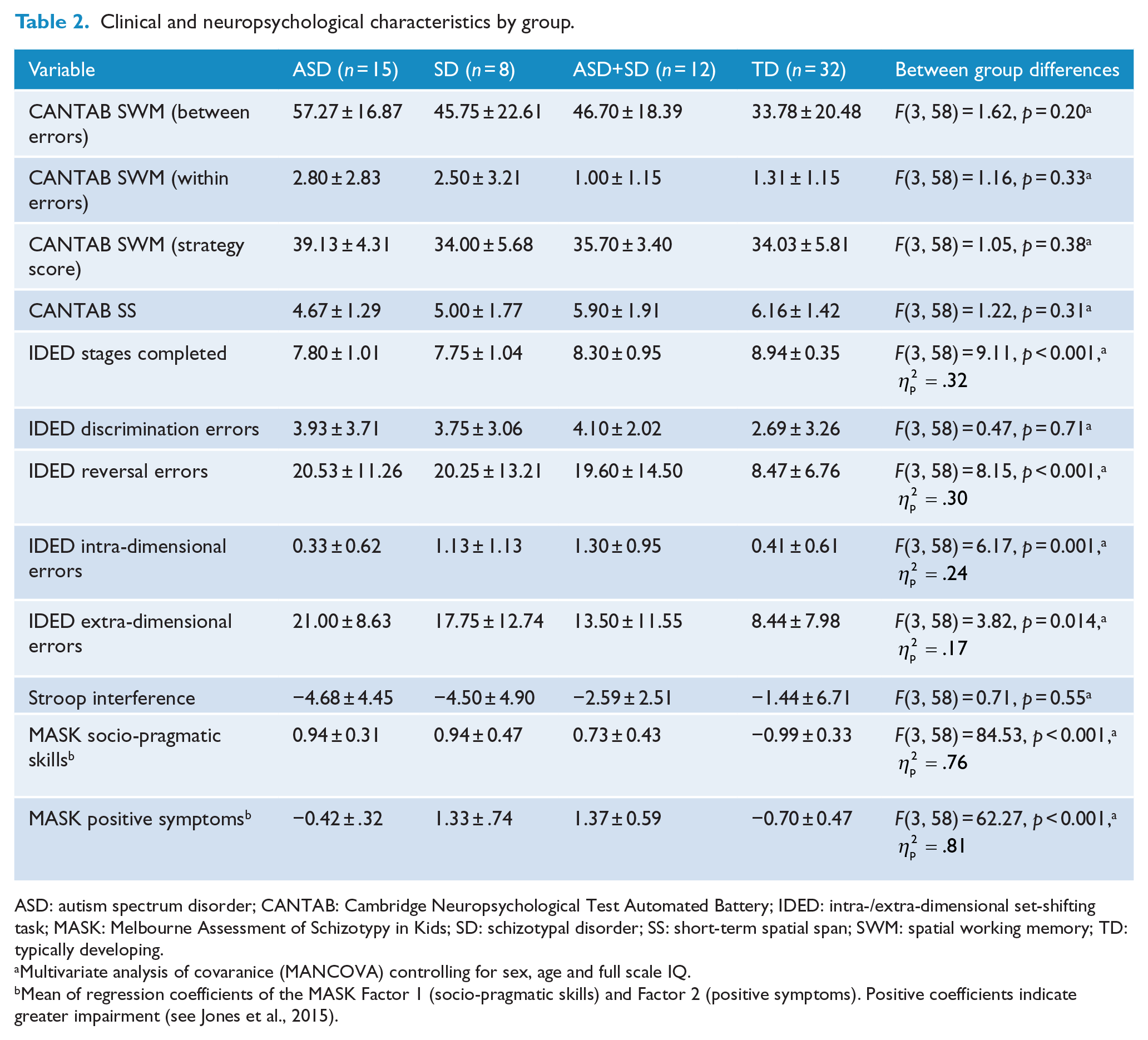
ASD: autism spectrum disorder; CANTAB: Cambridge Neuropsychological Test Automated Battery; IDED: intra-/extra-dimensional set-shifting task; MASK: Melbourne Assessment of Schizotypy in Kids; SD: schizotypal disorder; SS: short-term spatial span; SWM: spatial working memory; TD: typically developing.
Multivariate analysis of covaranice (MANCOVA) controlling for sex, age and full scale IQ.
Mean of regression coefficients of the MASK Factor 1 (socio-pragmatic skills) and Factor 2 (positive symptoms). Positive coefficients indicate greater impairment (see Jones et al., 2015).
Group differences on the IDED measures
As indicated in Table 2, the groups differed on the mean number of stages completed. Pairwise comparisons revealed that the TD group significantly completed more stages than the SD (p = 0.001, Cohen’s d = 1.65) and ASD (p < 0.001, Cohen’s d = 1.54) groups. There were no other significant differences among the groups, and notably no difference between the TD and comorbid group (p = 0.096, Cohen’s d = .69). We note, however, that differences among the groups in the stages completed were due to children’s attrition at stages 8 and 9 of the task, corresponding to the extra-dimensional shift (eds) and reversal (edsr) stages (see Figure 1). To compare differences in the attrition rates across the groups, we conducted a binomial regression analysis. The overall model was significant, χ2(df = 3) = 25.41, p < 0.001, and accounted for 32.4% of the variance (Cox & Snell-R2 = 0.324). Pairwise comparisons, revealed that while both the SD (p = 0.012) and ASD (p = 0.006) groups were more likely to fail than the TD group, the comorbid group did not differ significantly from the TD group (p = 0.072). No other group differences were observed.
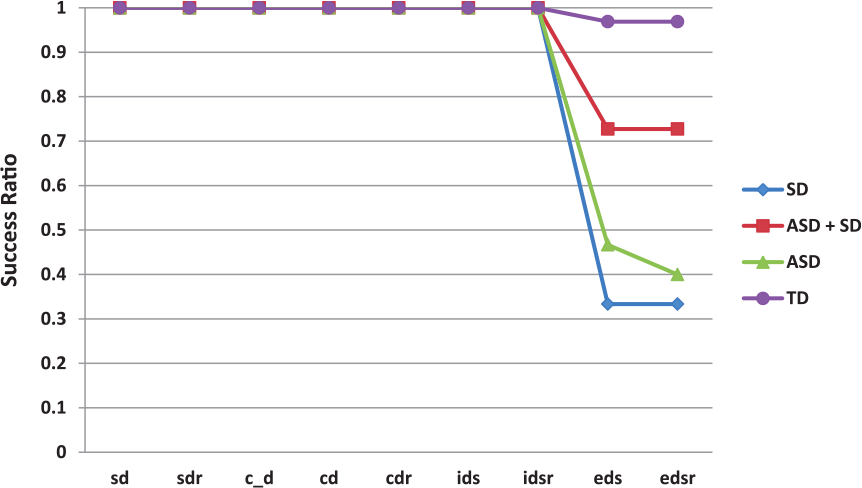
Children’s attrition rate (PASS/Fail) at each stage of the intra-/extra-dimensional attentional set-shifting task.
The groups also differed in the number of errors committed in the reversal trials, intra-dimensional trials, and the extra-dimensional trials. Pairwise comparisons of errors during the reversal trials (sdr+cdr+idsr+edsr) revealed that the TD group made significantly fewer errors compared to each of the clinical groups (all p-values<.028, Cohen’s d-values = 0.98–1.30).
During the ids trials, the comorbid group made significantly more errors than both the ASD (p = 0.014, Cohen’s d = 1.21) and TD (p = 0.01, Cohen’s d = 1.12) groups. There was also a non-significant trend for the SD group to make more errors than the ASD (p = 0.084, Cohen’s d = .87) and TD (p = 0.081, Cohen’s d = .79) groups. There were no other differences between the groups.
Conversely, when examining errors during the eds trials, the TD group made significantly fewer errors than the ASD group (p < 0.001, Cohen’s d = 1.51), and a similar but non-significant trend when compared with the SD group (p = 0.068, Cohen’s d = .88). Intriguingly, there was no difference between the comorbid and TD groups (p = 0.45, Cohen’s d = .51). Moreover, there were no statistically significant differences among the three clinical groups (all ps > 0.21), although it appears that the comorbid group committed fewer errors compared to the ASD (Cohen’s d = .74) and SD (Cohen’s d = .35) groups (see Figure 2).
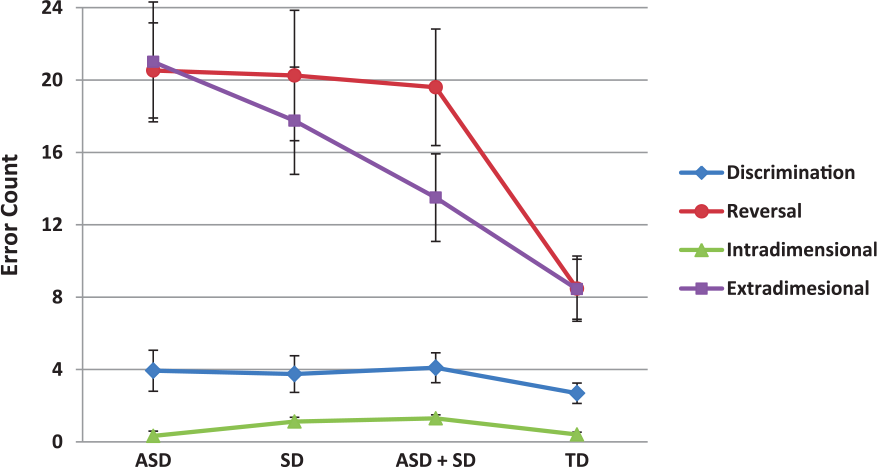
Error rate analysis in the discrimination, reversal, intra-dimensional (ids) and extra-dimensional (eds) trials. Bars represent standard errors.
Group differences on the MASK
With respect to the expression of positive symptoms, pairwise comparisons revealed no significant differences between the TD and ASD groups, or between the SD and comorbid groups. However, both the SD and comorbid groups scored significantly higher than both the ASD and TD groups (all p-values < 0.001, Cohen’s d-values = 3.07–3.88). Given these results, differences among the groups on the MASK’s socio-pragmatic skills factor were re-examined in a separate analysis while controlling for sex, FSIQ and the MASK positive symptom factor scores. The analysis showed significant differences among the groups, F(3, 58) = 54.16, p < 0.001, ηp2 = .75, (see Figure 3). Pairwise comparisons showed that the TD group outperformed all clinical groups (all p-values <0.001, Cohen’s d-values = 4.20–6.15). Moreover, there were significant differences among the clinical groups, such that the comorbid group scored better than the SD (p = 0.042, Cohen’s d = .62) and ASD (p = 0.019, Cohen’s d = .70) groups. There were no other group differences.
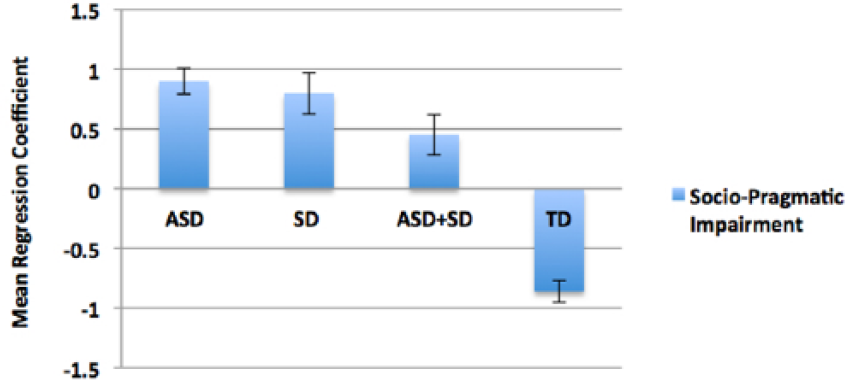
Socio-pragmatic impairment (Factor 1 of MASK). Positive coefficients represent poorer performance. Bars represent standard errors.
Discussion
In this study, we examined two core features of ASD and SD in children, namely difficulties with executive functioning and socio-pragmatic skills. Overall, we found that both the ASD and SD groups were significantly more impaired than the TD children on attentional and socio-pragmatic abilities. While these results appear consistent with the hypothesis positing that both conditions are part of a broad spectrum of overlapping neurodevelopmental disorders (Carroll and Owen, 2009), our findings revealed important differences between the groups. First, there was a clear distinction between the ASD and SD groups in terms of the relative expression of positive symptoms as measured by the MASK. Specifically, the SD group presented significantly more positive symptoms than both the ASD and TD groups, and there were no differences between the ASD and TD groups. Moreover, the ASD and SD groups differed in how they were distinguished from the TD group on the IDED task. Specifically, the ASD group (but not the SD group) committed more errors than the TD group on the extra-dimensional trials. Conversely, there was a higher, albeit non-significant, tendency for the SD group (but not for the ASD group) to make more errors on the intra-dimensional trials compared to the TD group. This suggests that the phenotypical overlap between these conditions may be precipitated by different cognitive styles and/or mechanisms associated with sustaining and switching attention.
Counter to our predictions, relative to the TD group, overall the comorbid group performed better than both the ASD and SD groups on the IDED task and presented better overall socio-pragmatic skills. One may suspect that this surprising result is due to attenuated clinical severity or better overall general cognitive abilities. However, the comorbid group presented similar levels of positive symptoms to the SD group, and had similar overall general cognitive abilities to both the ASD and SD groups. Our findings thus question current views opposing the addition of SD diagnosis to an ASD (Lugnegard et al., 2012), and suggest that a comorbid diagnosis may convey the possibility of attenuation in deficits manifest in pure forms of these disorders.
A close inspection of the performance on the IDED task provides insight into possible mechanisms. As presented in Figure 2, while the comorbid group committed more errors than the TD group during the intra-dimensional trials, they performed comparably to the TD group during the extra-dimensional trials. This suggests that the children with comorbid ASD and SD had difficulties in maintaining focus within the dimension, which in itself may be a facilitator during the extra-dimensional trials where one needs to switch across dimensions to successfully complete this stage. This also appeared to be true for the SD children, albeit to a lesser degree. Likewise, the difficulties of the children with ASD during the extra-dimensional trials and their success during the intra-dimensional trials may be, respectively, associated with their inability to switch across dimensions but to sustain attention over time. These data are consistent with the notion that sustaining and switching attention represent two poles of irregularities across the ASD (Goldstein et al., 2001; Reed and McCarthy, 2012) and SD (Yogev et al., 2004) spectra and which appear to converge in a compensatory manner in the comorbid group (see also Abu-Akel et al., 2016, 2017).
It is noteworthy that the differences among the groups were only discerned on the IDED task, and not when performing the other executive function tasks, namely the SWM and SS tasks. This suggests that better performance of the comorbid group is specific to attentional set-shifting. This distinction is important, as previous studies have shown that different anatomical substrates underlie these tasks, with the SWM task being associated with fronto-parietal structures (Kaufmann et al., 2013) and the IDED task with the fronto-striatal circuit (Levaux et al., 2007; Pantelis et al., 1997), particularly the orbito-frontal cortex (Dias et al., 1997). This suggests that the overall better performance of the comorbid group cannot be attributed to better overall executive functioning abilities, and that performance differences among the groups may be due to different profiles of fronto-striatal dysfunctions.
Moreover, the attenuated socio-pragmatic impairment of the comorbid group is consistent with recent findings showing that co-occurring autism and positive schizotypal traits in the general population interactively improved performance in perspective-taking during a referential communication task (Abu-Akel et al., 2015). Our finding is also consistent with the diametric model (Crespi and Badcock, 2008), positing that ASD and SD exert opposing effects on social cognition. Specifically, ASD, characterized with mechanistic cognition, is associated with hypomentalism (i.e. diminution in the ability to understand and attribute mental states) while SD, by contrast, is characterized by mentalistic cognition and is associated with hypermentalism (i.e. erroneously over-attributing mental states). While we did not test directly for hypo- versus hypermentalism when we assessed the children’s socio-pragmatic skills, there is evidence that ASD is associated with hypomentalism and SD is contrastingly associated with hypermentalism (Ciaramidaro et al., 2014). Thus, we speculate that the improved behaviour in the comorbid group may be the result of diametric modulation of co-occurring phenotypic traits that are disorder-specific. The anti-correlational nature of the default mode network (associated with mentalistic thinking) with the task positive network (associated with mechanistic thinking) (Jack et al., 2012) is a promising neural framework to investigating these contrasting cognitive styles in ASD and SD. Specifically, Jack et al. (2012) showed that social tasks (reasoning about the mental states of others) deactivated regions within task positive networks, while analytic tasks (reasoning the causal/mechanical properties of inanimate objects) deactivated regions within the default mode network, thus showing reciprocal suppression (see also Harrison et al., 2008).
We acknowledge a number of limitations of our study. First, findings of our study are limited by the small sample sizes of the clinical groups. Thus, future work with larger samples is needed in order to have a better understanding of the clinical phenotypes of children with SD or a comorbid ASD. Second, some expected differences among the groups in executive functioning were not observed. However, the results were in the expected direction, suggesting limited statistical power rather than poor sensitivity of the tasks used. Third, while sex did not appear to influence the obtained results, its potential effect should be examined in future studies with larger samples as there is evidence suggesting that females are less susceptible than males in developing neurodevelopmental disorders (Jacquemont et al., 2014), and may not be as impaired in some cognitive domains (Hull et al., 2016). This is particularly important in order to rule out the possibility that the attenuated impairment of the comorbid group is not due to the disproportionate representation of females in the group. Fourth, two children in the SD group had a comorbid diagnosis of ADHD, and one a diagnosis of Reactive Attachment Disorder. As such comorbidities are commonplace, their effect on outcome measures should be examined in larger samples. Fifth, a total of five children from the SD and comorbid groups received medications, of which four (two from each group) were on risperidone. While it is difficult to ascertain whether these children influenced the results, we think that this is unlikely given the reverse pattern of intra- and extra-dimensional errors, and the lack of difference in the presentation of positive symptoms in these two groups. Clearly, however, further work is needed to rule out the potential effect of medication in these complex disorders. Sixth, while the IDED task provides some clues about the mechanisms that might explain the attenuating effect we observed in the comorbid group, future research (both neural and behavioural) is needed to test the prediction that these mechanisms are highly interactive and of contrasting natures. In this regard, it is important to caution researchers and clinicians alike that apparent overlaps between ASD and SD might be precipitated by different cognitive styles or biases. Hence, assessments that combine contrasting abilities such as the IDED task as well as tasks that test for hypo- versus hypermentalism might be particularly beneficial in discriminating between the groups.
Our study is the first to observe that co-occurring ASD and SD can result in attenuation rather than exacerbation of deficits in these children. The findings indicate the necessity for clinics that assess children for ASD and neurodevelopmental disorders to also assess for the possibility of a SD. It is particularly important for future research into aetiology and phenomenology to differentiate the clinical and behavioural phenotypes of children with an ASD or a SD, but also recognize their potential comorbidity. This diagnostic differentiation is also likely to have implications for targeted pharmacological, behavioural, educational and social interventions. Specificity in diagnosis and recognition of dual diagnosis is also likely to have long-term implications. For example, those with a SD might be more at risk of developing early onset psychosis, and social communication might improve in those with an ASD as they mature or if they have received early psycho-educational intervention. Therefore, a longitudinal study of children with a SD or a comorbid ASD is imperative to better understanding of these significant neurodevelopmental disorders.
Footnotes
Acknowledgements
The authors thank Ian Harding for comments on an earlier version of the manuscript, and our participants and their families for their participation and enthusiasm for the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
Christos Pantelis was supported by a NHMRC Senior Principal Research Fellowship (IDs: 628386, 1105825). The work was supported by University of Melbourne – Research Grant Support Scheme (ID: 1452755) and Melbourne Neuropsychiatry Centre at The University of Melbourne. All authors declare that they have no competing interests relevant to this work.
