Abstract
Objective:
People living with severe and persistent mental illness experience poorer physical health, often due to medication and preventable lifestyle factors, and exacerbated by barriers to accessing healthcare services. Pharmacists are well-positioned to improve the physical and mental health of this population. However, little is known about pharmacists’ current practices when providing services to this population nor the impact of pharmacist-led interventions on consumer health outcomes. We undertook a systematic review to identify, describe and assess the effectiveness of pharmacist-led interventions for supporting people living with severe and persistent mental illness and the impact on consumer outcomes.
Methods:
MEDLINE, Embase, PsycINFO, CINAHL, Web of Science, Scopus, Cochrane Library, International Pharmaceutical Abstracts and ProQuest Dissertations and Theses were searched between January 1990 and April 2020. Full-text studies exploring pharmacist-led interventions in any setting for people living with severe and persistent mental illness were included. A risk of bias assessment was conducted.
Results:
A total of 37 studies were included. More than half of the pharmacist interventions were multifaceted. The most common components of pharmacist-led interventions included education and/or patient counselling, providing recommendations to healthcare professionals and conducting medication reviews. Multifaceted interventions demonstrated improvements in clinical outcomes, whereas single interventions focused mostly on consumer-reported outcomes. The methodological quality of included studies was moderate-to-high risk of bias and there was considerable heterogeneity in the study design, interventions described, and outcomes reported.
Conclusion:
There is evidence that pharmacist-led interventions improve consumer-reported and clinical outcomes for people living with severe and persistent mental illness. Pharmacists are capable and have a role in supporting people living with severe and persistent mental illness, either individually or as interprofessional collaborators with other healthcare professionals. Future research should attempt to better understand which particular intervention components have the greatest impact and also evaluate the implementation and long-term sustainability of such interventions.
Introduction
Severe and persistent mental illness (SPMI), including (but not limited to) schizophrenia and other psychotic disorders, bipolar disorder, and severe and recurrent major depression, refers to any mental illness that negatively impacts a person’s relationships, educational achievements and occupational performance (Carey and Carey, 1999). It also contributes to a significant burden of US$148 billion to the US economy annually (Carey and Carey, 1999). People living with SPMI have up to 25 years lower life expectancy than the general population (Royal Australian New Zealand College of Psychiatrists [RANZCP], 2016), mainly due to modifiable lifestyle factors such as smoking, physical inactivity, and unhealthy diet (World Health Organization, 2018), side effects of antipsychotic medications (World Health Organization, 2018), and compounded by stress intrinsic to living with a mental illness (Liu et al., 2017). As a result, this population experiences a significantly higher incidence of comorbid preventable chronic diseases including type 2 diabetes, cardiovascular and respiratory diseases (World Health Organization, 2014). There is evidence that inequality in healthcare access and provision contributes to these comorbid physical illnesses being under-recognised and sub-optimally treated among people living with SPMI, leading to poor overall physical and mental health (De Hert et al., 2011; Lawrence and Kisely, 2010).
Given the personal, social, and economic impacts of untreated and undertreated SPMI, involving primary healthcare professionals to ensure adequate support for this population is essential. In general, consumers are 1.5 to 10 times more likely to see their pharmacist than they see primary care physicians (Tsuyuki et al., 2018). Over the past decade, pharmacists have demonstrated their capability to improve health outcomes by educating consumers about psychotropic medication to improve adherence (Kyle, 2018), and providing medication information, either individually or collaboratively as integral members of the healthcare team (Davis et al., 2020).
Treatment for more severe mental health conditions, such as schizophrenia has historically relied on pharmacological approaches (Lally and MacCabe, 2015). However, recent studies have started to recognise the need for ongoing routine management and monitoring by healthcare professionals (Whiteford et al., 2017). Evidence exists around the role of the pharmacist to lead screening and risk assessment and conduct medication reviews to ensure quality use of medicines for a broad range of mental health conditions (Rubio-Valera et al., 2014). Mental health professionals including physicians and nurses have also been shown to have positive perceptions and expectations from pharmacists to help manage medication side effects and improve quality of care (Eltorki et al., 2019). However, there is still a lack of understanding as to how specific pharmacist-led interventions may impact health outcomes among people living with SPMI. A Cochrane review revealed that pharmacist-led services may be more cost-effective and can produce similar positive effects on patient health and physical function, in comparison to doctor-led services (de Barra et al., 2018). Another review conducted by Sud et al. explored pharmacists’ involvement as part of a multidisciplinary team in supporting people living with severe mental illness (Sud et al., 2021). It focused specifically on pharmacists’ involvement in the management of cardiometabolic risk and metabolic syndrome and showed that pharmacists could significantly improve cardiometabolic screening rates (Sud et al., 2021). To the best of our knowledge, no previous reviews have ever focused on exploring the overall evidence relating to pharmacist-led interventions for people living with SPMI. Therefore, this systematic review aimed to identify, describe and assess the effectiveness of pharmacist-led interventions for supporting people living with SPMI and all the associated impact on consumer and pharmacist-reported outcomes. Specifically, this review was guided by the following research questions:
How have pharmacists been involved in supporting people living with SPMI?
What is the nature, extent and outcome of pharmacist-led intervention for people living with SPMI?
What type of education and training is developed for and provided to pharmacists prior to their involvement in interventions for people living with SPMI?
What are the barriers to and facilitators of pharmacists’ involvement in supporting people living with SPMI?
Methods
This review was guided by the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analysis) guideline (Moher et al., 2009). The protocol was registered in PROSPERO, an international prospective register of systematic reviews (CRD42020170711) (El-Den et al., 2020a).
Search strategy and selection criteria
Potentially relevant publications published between 1 January 1990 and 1 April 2020 were identified by searching the following databases: MEDLINE, Embase (Ovid), PsycINFO, CINAHL, Web of Science, Scopus, Cochrane Library, International Pharmaceutical Abstracts and ProQuest Dissertations and Theses. In addition, reference lists of relevant reviews were screened for any potentially relevant individual studies. The year 1990 was chosen as it aligned with the introduction of second-generation antipsychotics (Tamminga, 2000), and a global shift towards community-based mental healthcare (Mechanic, 2007; Mental Health Council of Australia, 2005; Robson, 2008).
The following search terms or subject headings (and related concepts, depending on the database) were used.
Concept 1: pharmacist OR pharmacy; AND
Concept 2: severe and persistent mental disorders OR severe and persistent mental illnesses OR schizophrenia spectrum and other psychotic disorders OR bipolar and related disorders OR substance related disorders OR depressive disorders OR anxiety disorders.
Study selection and data extraction
Search results were downloaded into EndNote X9, duplicates were removed automatically and manually. One reviewer (R.N.) was involved in removing duplicates and screening titles. Two reviewers (R.N. and V.S.) independently screened studies by abstract and then by full-text for potentially eligible studies, in Covidence (Covidence Systematic Review Software, n.d.). Any discrepancies were discussed with COR and SE until consensus was reached. The following data were extracted where possible as per the terminology used in the study: author(s), year, country, study aim, setting, sample size, study population, study design, intervention(s), consumer- or pharmacist/clinician-reported outcomes or other outcomes related to pharmacists’ intervention(s). Due to a significant degree of heterogeneity between intervention and outcome measures, a meta-analysis was not deemed suitable.
Inclusion/exclusion criteria
The inclusion criteria were formulated using the PICOS (Participant, Intervention, Comparison, Outcome and Study) design (Moher et al., 2009). Studies were included if they met the following criteria:
Participants
The study population involved people living with SPMI. SPMI refers to any mental illness that has a continuous and significant lifelong impact on a person’s relationships, social functioning, education, and livelihood. It includes (but not limited to) schizophrenia, bipolar disorder, any psychotic illness (e.g. schizoaffective disorder, psychotic depression) and moderate-to-severe depression or anxiety.
For studies with a mixed population of participants, including people living with SPMI, the studies were included if the outcome(s) pertaining to pharmacist-led intervention(s) for people living with SPMI could be extracted;
For studies that did not report on a definitive diagnosis, the studies were included if it had a detailed description of illness severity and the authors were satisfied that it meets the definition of SPMI mentioned above. For example, a study that included participants in a psychiatric hospital receiving antipsychotic treatment would be eligible for inclusion;
For studies that included people living with SPMI also experiencing a comorbid substance-use disorder, the outcomes relating to both the SPMI and the substance-use disorder will be reported.
Intervention
‘Pharmacist-led intervention’ in this review refers to any service or contribution made by the pharmacist for people living with SPMI. For study inclusion, the intervention had to be delivered by pharmacists, working either individually or as part of a multidisciplinary healthcare team, in any healthcare setting, including but not limited to hospitals, community pharmacies and outpatient settings.
For studies with a mixed population of healthcare professionals, if the outcome(s) relating to the intervention(s) delivered by the pharmacist could be extracted then the study was included.
Comparator and outcome
As per the aforementioned definition of a pharmacist-led intervention, studies were included with or without a comparator group. Any outcomes attributable to pharmacist involvement such as consumer or pharmacist-reported outcomes were included.
Study design
All primary research published in English was considered for inclusion, regardless of study design.
The following types of studies were excluded:
Studies that only included people living with a substance-use disorder (e.g., opioid, tobacco, alcohol). Pharmacist-led intervention(s) (e.g., naloxone dispensing, smoking cessation therapy, alcohol misuse screening) for people living with substance-use disorders have been established and explored previously through published reviews of the literature (Greenhalgh et al., 2016; Mdege and Chindove, 2014; Nielsen and Van Hout, 2016; Saba et al., 2014; Thakur et al., 2020; Watson and Blenkinsopp, 2009);
Studies conducted in educational settings such as a university. For example, a study conducted in a university classroom setting was excluded as the aim of this review was to explore the role of pharmacists in practice healthcare settings;
Non-primary research publications, such as conference abstracts, case reports, editorials and letters.
Risk of bias
The risk of bias was assessed using the Cochrane risk-of-bias tool for randomised trials (RoB 2) (Higgins et al., 2016) and Risk Of Bias In Non-randomised Studies–of Interventions (ROBINS-I) (Sterne et al., 2016). The risk of bias assessment was conducted primarily by RN, in consultation with SE and COR. Randomised Controlled Trials (RCTs) were assessed according to the following RoB 2 domains: randomisation process, deviations from intended interventions, missing outcome data, measurement of the outcome and selection of reported result (Higgins et al., 2016). The ROBINS-I tool assesses the risk of bias in non-RCTs across seven domains: bias due to confounding, bias in the selection of participants, bias in the classification of the intervention, bias due to deviation from intended intervention(s), bias due to missing data, bias in measurement of outcome, and bias in selection of reported results (Sterne et al., 2016). Each risk of bias item was rated as either low, some concerns/moderate or serious/high risk of bias.
Results
The literature search yielded a total of 9264 potentially relevant studies. After the removal of 2222 duplicates, 7042 studies were considered for inclusion. Of which, 342 studies were identified as non-primary articles, and the remaining 6700 studies were screened by title (5287 removed based on titles) and then abstract. Thirty-three studies met the inclusion and exclusion criteria and were included in the review. Further screening for potentially relevant studies in the reference lists of literature reviews resulted in four additional studies, resulting in 37 included studies. Figure 1 outlines this process and the search results.
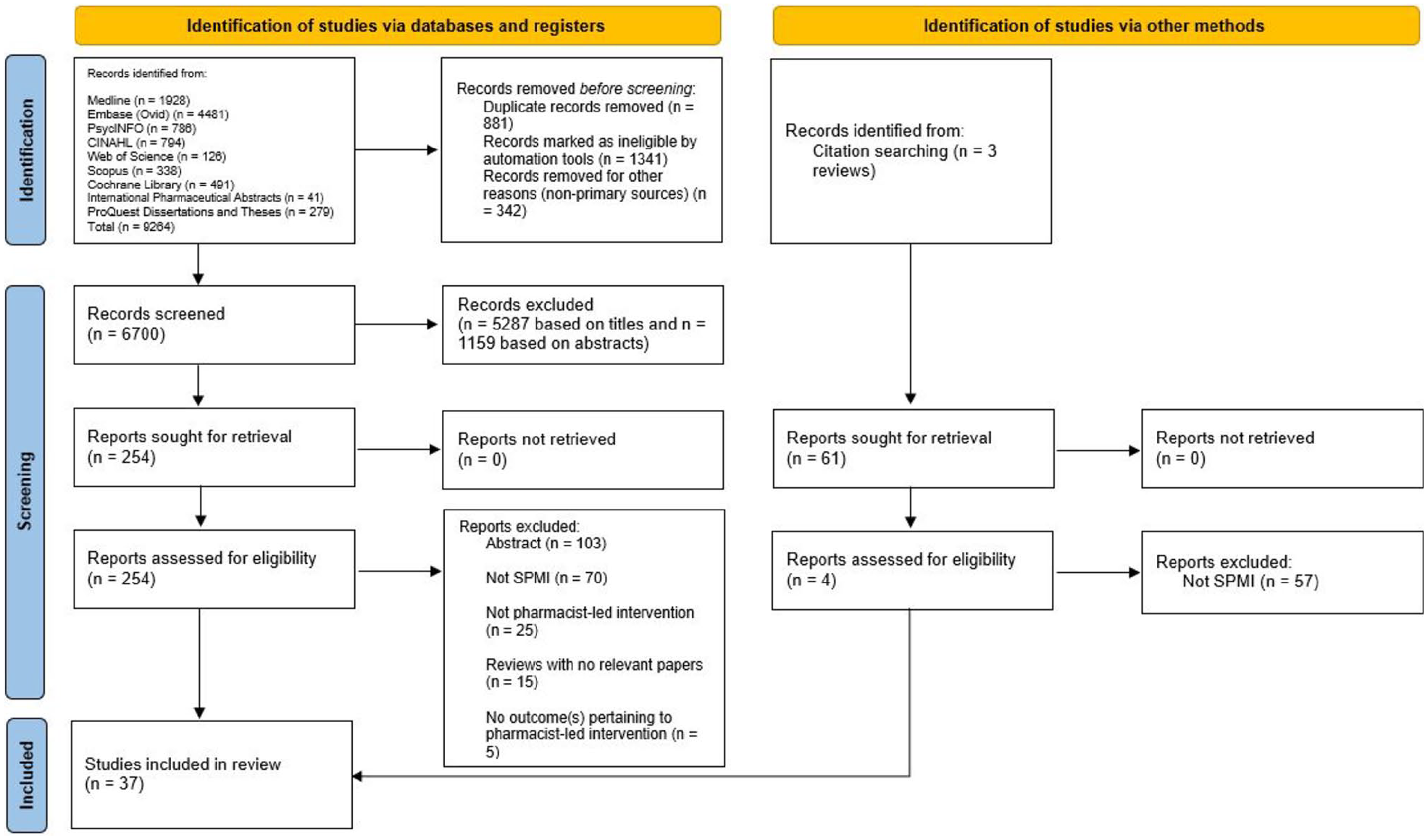
PRISMA flow diagram showing process of study selection for inclusion in systematic review.
Study characteristics
As described in Tables 1 and 2, studies were conducted in various countries worldwide, with the majority conducted in North America (n = 11) and Asia (n = 11). Ten of the 37 studies were RCTs and 27 were non-RCTs, of which 15 employed pre/post single (n = 8) or two-group (n = 7) study designs and 12 employed post-intervention single-group study designs.
Description of single pharmacist-led intervention studies.
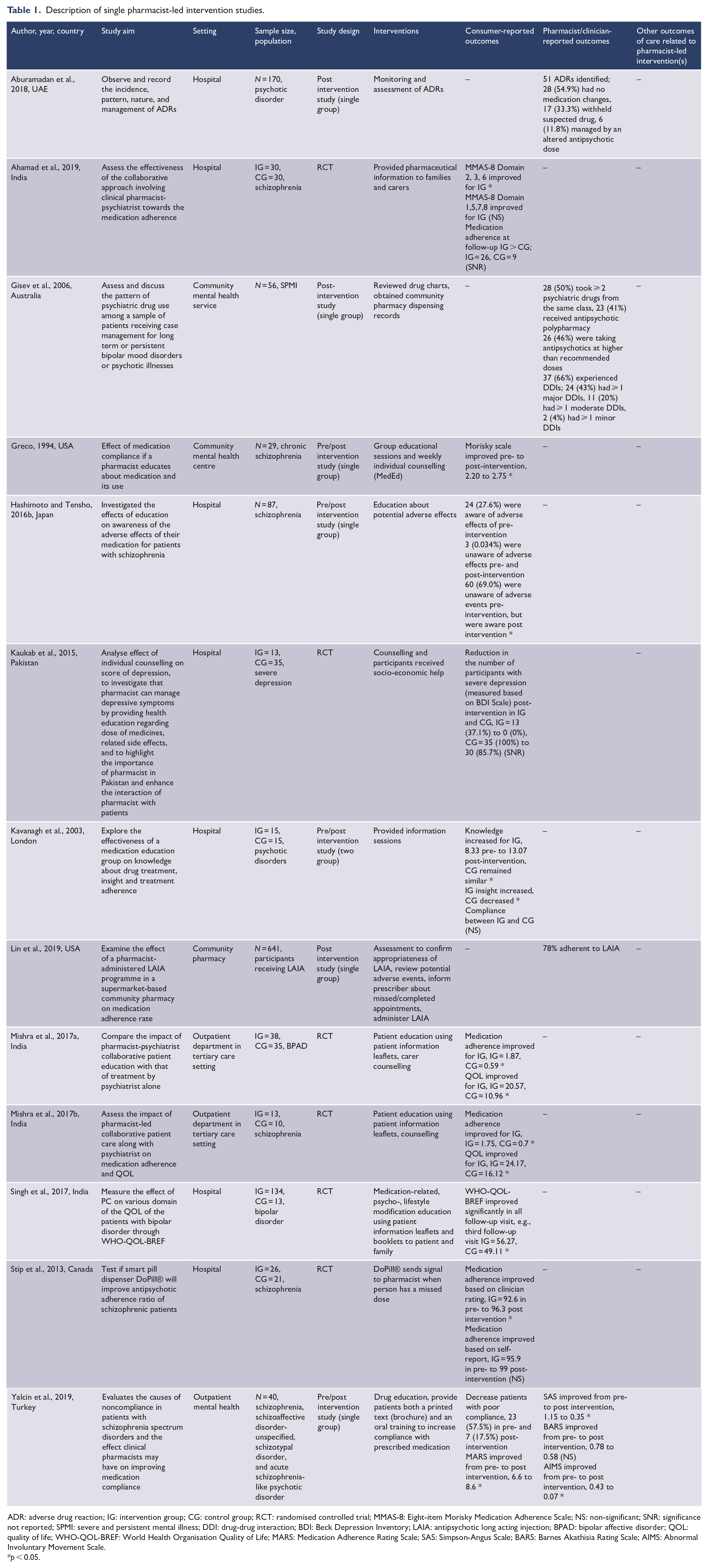
ADR: adverse drug reaction; IG: intervention group; CG: control group; RCT: randomised controlled trial; MMAS-8: Eight-item Morisky Medication Adherence Scale; NS: non-significant; SNR: significance not reported; SPMI: severe and persistent mental illness; DDI: drug-drug interaction; BDI: Beck Depression Inventory; LAIA: antipsychotic long acting injection; BPAD: bipolar affective disorder; QOL: quality of life; WHO-QOL-BREF: World Health Organisation Quality of Life; MARS: Medication Adherence Rating Scale; SAS: Simpson-Angus Scale; BARS: Barnes Akathisia Rating Scale; AIMS: Abnormal Involuntary Movement Scale.
p < 0.05.
Description of multifaceted pharmacist-led intervention studies.
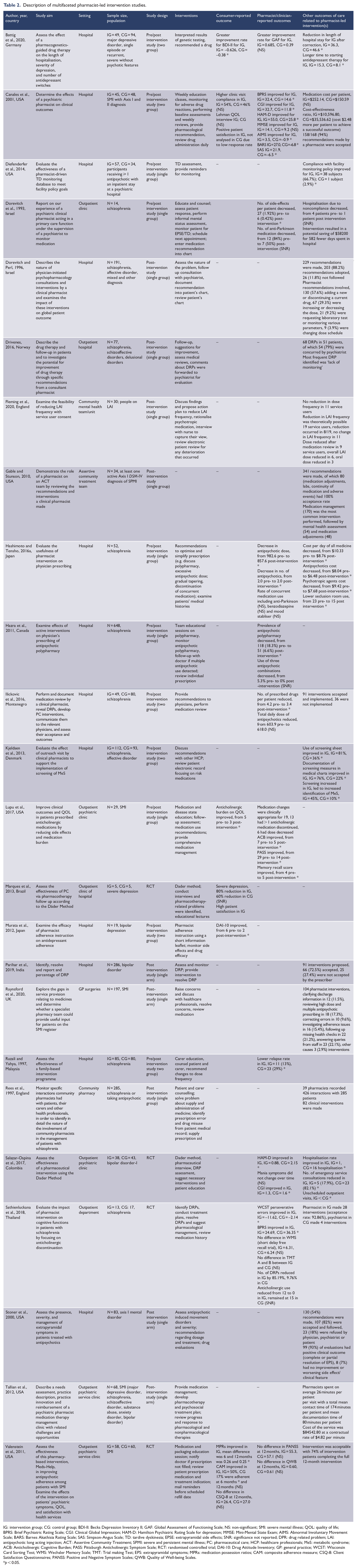
IG: intervention group; CG: control group; BDI-II: Becks Depression Inventory II; GAF: Global Assessment of Functioning Scale; NS: non-significant; SMI: severe mental illness; QOL: quality of life; BPRS: Brief Psychiatric Rating Scale; CGI: Clinical Global Impression; HAM-D: Hamilton Psychiatric Rating Scale for depression; MMSE: Mini-Mental State Exam; AIMS: Abnormal Involuntary Movement Scale; BARS: Barnes Akathisia Rating Scale; SAS: Simpson-Angus Scale; TD: tardive dyskinesia; EPSE: extrapyramidal side effects; SNR: significance not reported; DPR: drug related problem; LAI: antipsychotic long acting injection; ACT: Assertive Community Treatment; SPMI: severe and persistent mental illness; PC: pharmaceutical care; HCP: healthcare professionals; MeS: metabolic syndrome; ACB: Anticholinergic Cognitive Burden; PASS: Pittsburgh Anticholinergic Symptom Scale; RCT: randomised controlled trial; DAI-10: Drug Attitude Inventory; GP: general practice; WCST: Wisconsin Card Sorting Test; WMS: Wechsler Memory Scale; TMT: Trial making Test; EPS: extrapyramidal symptoms; MPRs: medication possession ratios; CAM: composite adherence measure; CSQ-8: Client Satisfaction Questionnaires; PANSS: Positive and Negative Symptom Scales; QWB: Quality of Well-being Scales.
p < 0.05.
A total of 20 studies were conducted in hospitals (n = 20), with only two in community pharmacies (n = 2). In relation to diagnosis, most studies included only people living with schizophrenia (n = 12), followed by bipolar disorder (n = 5) and severe depression (n = 3). Twelve studies included mixed SPMI populations, whereby participants had dual or multiple diagnoses.
The sample size of included studies ranged from 10 to 648 participants. Study duration was reported in 33 studies, ranging from 1 month to 10 years. Of these, 14 studies had a duration of 6 months or less. Twelve studies mentioned a follow-up period that ranged from 2 weeks to 12 months, of which eight studies had a follow-up period of 6 months or less.
Pharmacist-led intervention components
Pharmacist-led interventions were broadly classified into the following groups: education or patient counselling, recommendations to healthcare professionals, medication review, clinical assessments and monitoring and reminders. These are discussed further below.
Education and/or patient counselling
Seventeen studies involved interventions whereby pharmacists provided consumers education and/or patient counselling, either through face to face or telephone support. These interventions were delivered by the pharmacist individually or as a group and focused on medication and disease state information and lifestyle modification recommendations, discussing the importance of medication adherence, and providing advice on how to manage medication side effects. Educational and counselling materials were developed based on published information such as consumer information leaflets (Mishra et al., 2017a, 2017b) or information derived from the US pharmacopoeia (Kaukab et al., 2015) or guidelines from the American Society of Hospital Pharmacists (Greco, 1994).
Medication review
Eighteen studies involved a pharmacist-led medication review service as part of the intervention. Components of the medication review services included comprehensive medication history taking and medication regimen review. Half of the medication reviews (n = 9) were conducted in hospitals, and only four were conducted in community settings such as a community mental health team.
Clinical assessments and monitoring
Pharmacists conducted clinical assessments and monitoring for people living with SPMI in 15 studies. These assessments and monitoring helped identify adverse drug reactions (n = 5), medication-related problems such as reducing antipsychotic polypharmacy (n = 7), drug efficacy and appropriateness (n = 3), and symptom/illness related issues (n = 3).
Recommendations to healthcare professionals
Eighteen studies involved pharmacists providing recommendations or feedback to other healthcare professionals. All recommendations or feedback provided were after medication reviews and clinical assessments and monitoring conducted by pharmacists. Pharmacological recommendations provided to prescribers were all medication-related, whereby pharmacists made dose adjustments and suggested alternative medication recommendations to optimise treatment or reduce antipsychotic polypharmacy. Other recommendations involved requesting laboratory tests (Dorevitch and Perl, 1996), and monitoring for various parameters such as any changes in glucose and lipid profile (Diefenderfer et al., 2014).
Reminders
Reminders were provided to both consumers and healthcare professionals. Reminders were given to consumers to improve medication adherence through mailing medication refill reminders and packing dose administration aids (Stip et al., 2013; Valenstein et al., 2011). However, doctors were provided reminders for metabolic monitoring and assessments of antipsychotic adverse effects by a pharmacist-driven antipsychotic monitoring database (Diefenderfer et al., 2014).
Intervention design
Single or multifaceted interventions
Twenty-four studies employed more than one intervention (multifaceted interventions), whereas 13 studies involved a single pharmacist-led intervention (shown in Tables 2 and 1, respectively). Of the 24 multifaceted intervention studies, two studies described the range and types of pharmacist-led interventions conducted (Gable and Stunson, 2010; Rees et al., 1997). Gable and Stunson (2010) reviewed all the interventions a clinical pharmacist made and showed that medication management was the most frequent intervention performed. Rees et al. (1997) documented the consumer-pharmacist interactions and found that almost half of the community pharmacist interactions with people with schizophrenia involved counselling about medication adverse effects.
Single intervention
Of the 13 studies that employed a single intervention, nine were education and/or patient counselling interventions, two involved clinical assessments and monitoring, one involved medication reviews and one study involved adherence reminder messages. The majority of studies (n = 9) relied solely on consumer-reported measures to evaluate outcomes such as medication adherence, consumer knowledge, and quality of life (QOL). Only one study evaluated outcomes such as medication adherence and adverse effect through scales such as the Medication Adherence Rating Scale, Abnormal Involuntary Movement Scale and Barnes Akathisia Rating Scale that were completed by both the consumer and clinician (Yalcin et al., 2019).
Multifaceted interventions
The 15 studies involving multifaceted pharmacist-led interventions included a combination of components such as providing recommendations to other healthcare professionals, medication reviews, and performing assessments and monitoring. Nine studies involved interventions focusing on education and/or patient counselling and other components such as performing clinical assessments and monitoring, providing feedback or conducting medication reviews. Details of the outcomes reported are presented in Tables 1 and 2. In terms of the effectiveness of the interventions, 10 studies showed significant improvement in medication adherence, QOL, antipsychotic polypharmacy and dose, disease or symptoms severity, adverse effects, hospitalisation and relapse rate.
Outcome measurement
Consumers and pharmacists/clinicians were involved in the evaluation of various outcomes, which included medication adherence, QOL, clinical, medication-related outcomes, medical practitioners’ acceptance rate of pharmacists’ recommendations and other healthcare outcomes such as healthcare service utilisation. Tables 1 and 2 provide detailed presentations of all study outcome(s) and their statistical significance. The outcome measures reported in more than five studies are reported below:
Consumer-reported outcomes
Medication adherence
A total of 10 studies measured medication adherence as an outcome. The tools used to assess adherence varied; eight studies relied solely on validated self-reported adherence tools, and two used objective adherence measures such as pharmacy records and drug concentration. Seven of the eight studies using self-reported measures showed significant improvement(s) in at least one measure or domain of adherence. Only two included studies involved multiple adherence measures (Stip et al., 2013; Valenstein et al., 2011). Stip et al. (2013) reported a lack of concordance between clinician-reported and self-reported adherence. Valenstein et al. (2011) assessed changes in adherence using objective and subjective measures, namely self-reported and pharmacy data, and found significant improvements in both.
Quality of life
Six studies investigated changes in consumers’ QOL. Different tools were used to assess QOL, but half of the studies (n = 3) used the WHO Quality of Life-BREF assessment and all showed significant QOL improvement (Mishra et al., 2017a, 2017b; Singh et al., 2017).
Clinical outcomes
A range of clinical outcome measures were reported in 11 studies. Most common reported outcomes included symptom/illness severity (n = 6), depression symptoms (n = 5), presence, number and severity of adverse effects (n = 4), and cognition (n = 3). All studies (n = 4) showed significant improvement in the number or severity of adverse effects (Canales et al., 2001; Dorevitch et al., 1993; Lupu et al., 2017; Yalcin et al., 2019). Various tools were used to assess changes in clinical outcomes, for example depression symptoms were commonly assessed by the Hamilton Depression Rating Scale or Beck Depression Inventory, and studies mostly used the Positive and Negative Syndrome Scale, Brief Psychiatric Rating Scale or Clinical Global Impression scale to assess symptom severity.
Medication-related outcomes
Of the eight studies that reported on a medication-related outcome, seven described reduction in the use of antipsychotics (n = 4), anti-Parkinson (n = 1), and anticholinergic (n = 2) medications. However, only three of the seven studies showed significant reductions in at least one type of medication use (Hashimoto and Tensho, 2016a; Hazra et al., 2011; Sathienluckana et al., 2018).
Medical practitioners’ acceptance rate of pharmacists’ recommendations
Seven studies reported on medical practitioners’ acceptance rate of pharmacists’ recommendations. All recommendations made were related to pharmacotherapy changes such as dose adjustments and changing or discontinuing inappropriate medications. Parihar et al. (2019) had the lowest acceptance rates of 52.7% whereas Canales et al. (2001) reported the highest (94%). Two studies demonstrated a 100% acceptance rate for specific recommendations relating to preventing unnecessary drug therapy among consumers living with schizophrenia (Sathienluckana et al., 2018), and medication adjustments, laboratory monitoring, continuity of medications, and adverse events among consumers living with a diagnosis of SPMI (Gable and Stunson, 2010).
Healthcare service utilisation
The use of healthcare services was reported in five studies. More than half (n = 3) were related to hospitalisation, of which two studies showed significant reduction in hospitalisation and length of stay (Battig et al., 2020; Salazar-Ospina et al., 2017).
Pharmacists’ qualifications and training provided for intervention delivery
The details of pharmacists’ qualifications and/or the training provided to pharmacists prior to their delivery of interventions were reported in eight studies. Two studies included board certified psychiatric pharmacists (Gable and Stunson, 2010; Tallian et al., 2012). Six provided descriptions of the intervention training provided, and all the intervention training content varied and the personnel delivering the intervention were different (Gisev et al., 2006; Ilickovic et al., 2016; Marques et al., 2013; Salazar-Ospina et al., 2017; Sathienluckana et al., 2018; Stoner et al., 2000). For example, some pharmacists received training by psychiatrists to prepare them to support people living with SPMI (Salazar-Ospina et al., 2017), whereas in the study by Gisev et al. (2006), training was delivered by a multidisciplinary team including specialist pharmacists, consumer educators, a psychologist, a mental health nurse and a psychiatrist. Training duration was reported in only two studies, which ranged from eight to 57 hours of training.
Risk of bias
RCTs
Figure 2 shows the results of the RoB 2 risk of bias assessments conducted for 10 RCTs. Eight had a high risk of bias, and the other two had some concerns. All RCTs had at least some concerns due to the nature of the intervention resulting in difficulty in blinding consumers and pharmacists. Six RCTs were assessed to have a high risk of bias relating to the ‘measurement of the outcome’ as they relied on self-report measures with non-blinded participants (Ahamad et al., 2019; Kaukab et al., 2015; Marques et al., 2013; Mishra et al., 2017a, 2017b; Singh et al., 2017). Although there were 10 RCTs included in this review, only two specified the randomisation technique, and were rated as having low risk of bias in the randomisation process (Ahamad et al., 2019; Singh et al., 2017). The randomisation process of the other eight RCTs was not reported, potentially introducing selection bias. One RCT had a high risk of bias in the randomisation process, as there were significant differences in baseline symptom severity (Kaukab et al., 2015).
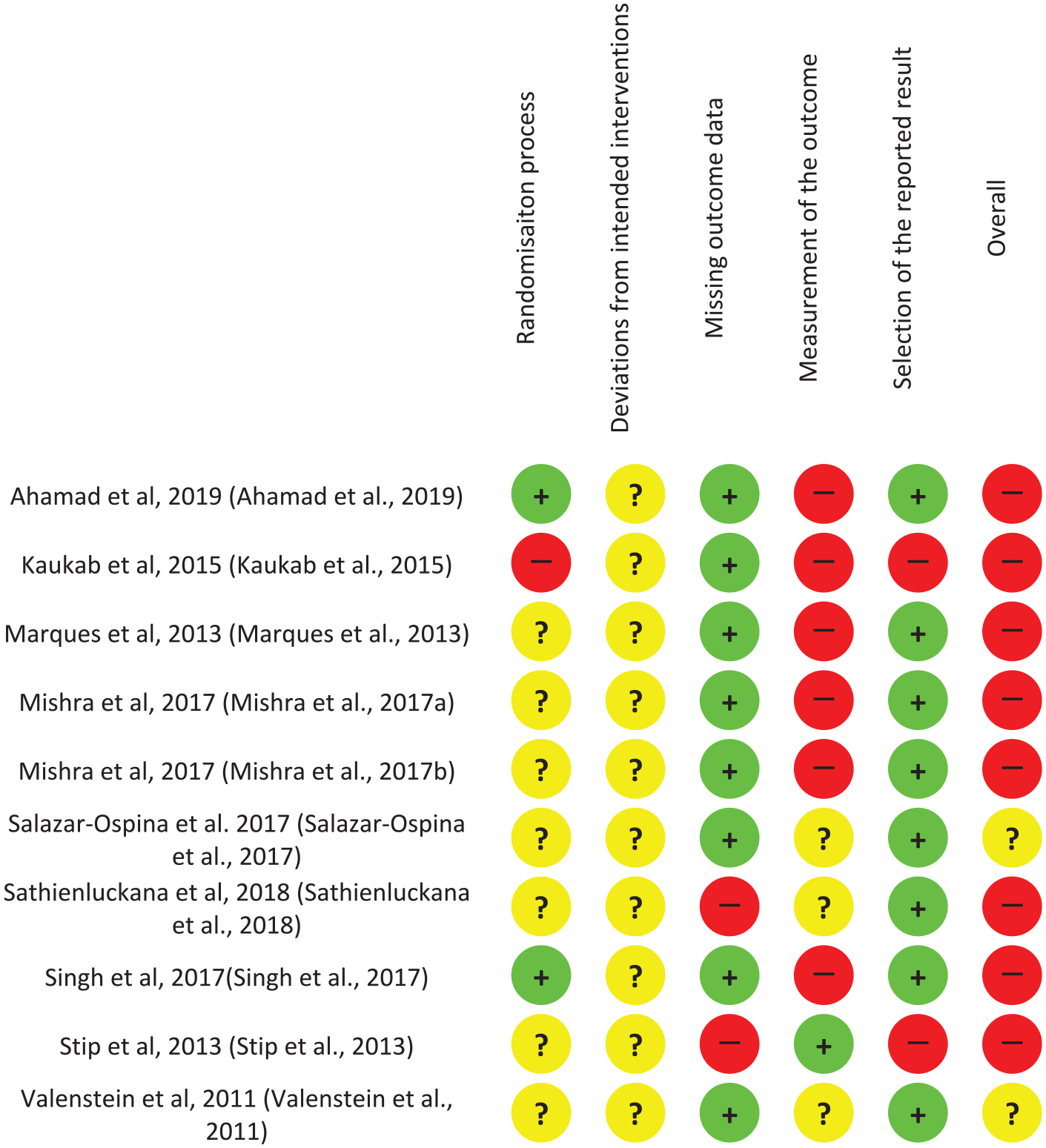
Risk of bias of randomised controlled trials using RoB 2 tool (Higgins et al., 2016).
Non-RCTs
Figure 3 shows the results of the ROBINS-I risk of bias assessment conducted for pre/post and post-intervention studies. Of the 27 non-RCT studies included in this systematic review, 20 had high risk, and 7 had moderate risk of bias. All post-intervention study designs were assessed as moderate-high in the ‘bias due to confounding’ category as they lacked a control group, thereby increasing the chance of confounding. All except one study had low risk of ‘bias due to deviations from intended interventions’ (Dorevitch et al., 1993). The study by Dorevitch et al. (1993) was assessed as high risk since the authors stated changes to hospitalisation criteria over the study period could have affected intervention delivery.
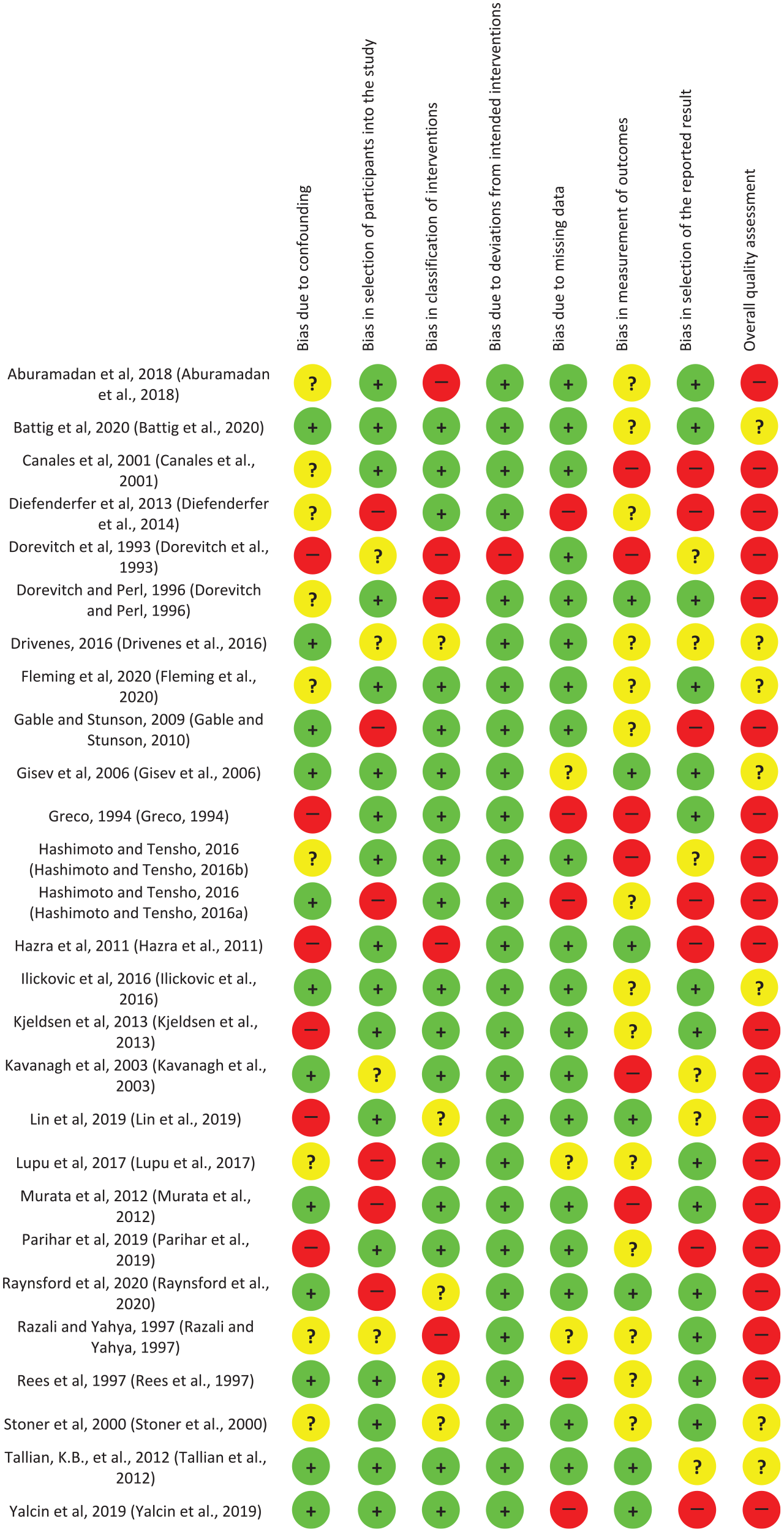
Risk of bias of nonrandomised controlled trials using ROBINS-I tool (Sterne et al., 2016).
Discussion
This review provides a comprehensive overview of the literature pertaining to the effectiveness and demonstrated the potential value of pharmacist-led interventions for people living with SPMI. This review aimed to explore pharmacists’ involvement, outcomes of pharmacist-led intervention(s) of supporting people living with SPMI, education and training for pharmacists prior to delivering the intervention(s) and barriers and facilitators for pharmacist-led intervention(s). The review’s research questions are explored in detail below.
Pharmacists’ involvement in supporting people living with SPMI
Pharmacists have demonstrated the ability to provide a wide range of services in a variety of healthcare settings to people living with SPMI. Findings from this review suggest most pharmacist-led interventions involve a component of consumer education and/or patient medication counselling, regardless of whether the intervention was single or multifaceted. While no included studies conducted a cost-effectiveness analysis of pharmacist-led educational intervention for SPMI, other studies have suggested that one reason for this may be the relatively easy implementation and cost-effectiveness of educational interventions (Milosavljevic et al., 2018). Educational interventions alone have been shown to be effective in the current literature, and the findings of this review align with this suggesting that pharmacist-led educational interventions improve medication adherence, levels of knowledge and QOL in people living with SPMI (Amer et al., 2018; Hätönen et al., 2008; Yeni et al., 2018). This systematic review found that multifaceted interventions with a larger number of elements were more successful in improving both medication adherence and clinical outcomes when compared to pharmacist-led single interventions. This may be due to several reasons, including reporting bias and the nature of multifaceted interventions. It is identified in this review that studies employing multifaceted interventions were more likely to report on both consumer- and clinician-reported outcomes, whereas single interventions focused on reporting consumer-reported outcomes. The lack of consistency in the intervention design, as well as outcome measures rendered it difficult for the authors to conduct comparisons of the effectiveness of the interventions. However, it is also arguable the multifaceted approach that involves multiple components is more likely to address the complex health issues that people living with SPMI experience (Ostler and Ackerson, 2008).
An example of this multifaceted approach was the Dader method, which was used in two studies (Marques et al., 2013; Salazar-Ospina et al., 2017). The pharmacists first conducted a pharmaceutical interview to assess consumers’ health problems. Next, consumers and prescribers were informed about any medication-related problems identified, and then education and recommendations were provided to consumers and prescribers, respectively. Another multifaceted approach is involving pharmacists in clozapine clinics to help monitoring and manage adverse events (Maryan et al., 2019). While none were included in this review as they did not meet the inclusion criteria, previous studies have explored how pharmacists could help overcome the barriers to clozapine use and the potential of pharmacists in clozapine monitoring and assessments was acknowledged (Kelly and Love, 2019; Maryan et al., 2019). Nevertheless, similar to previous studies exploring pharmacist-led multifaceted interventions, it is not known which components are specifically responsible for outcome improvements (Milosavljevic et al., 2018). There is a need to explore what components of multifaceted pharmacist-led interventions are most effective, feasible and acceptable. Further research is required to definitively determine if a multifaceted intervention is more effective than a single intervention, and which elements and combinations are most effective.
Outcomes of pharmacist-led intervention(s)
Findings from this systematic review suggest that pharmacist-led interventions do lead to significant improvements across a broad range of health outcomes, including medication adherence, QOL, and reduce disease progression, symptom or illness severity, hospitalisations and antipsychotic polypharmacy. Pharmacists’ recommendations were generally well-accepted by medical practitioners, with up to 100% acceptance rate for medication-related or monitoring recommendations (Gable and Stunson, 2010; Sathienluckana et al., 2018). Despite these positive effects on consumer health outcomes, the heterogeneity in the outcomes reported did not allow for more targeted analyses to explore the pooled effects in terms of the benefit to consumers. While consumer-reported measures were the most common method used to evaluate outcomes, studies used a variety of measures including consumer, pharmacist or clinician-reported outcomes. It is interesting to note that the reliability of using consumer self-reported measures to assess QOL in people living with psychiatric disorders has been questioned previously (Revicki et al., 2014). Consumer-reported outcomes often poorly correlate with clinical or performance-related outcomes (Johnston et al., 2019), and the ability of people living with severe mental illness to self-assess their own health have been questioned (Milosavljevic et al., 2018). However, recent emerging evidence suggests the importance of consumer involvement in the assessment of treatment outcomes, particularly in the field of psychiatry (Deshpande et al., 2011; Sartorius, 2014). Specifically, it is important for consumers to assess their own QOL and the clinicians’ views should be used as a proxy to rate QOL (Sartorius, 2014). Therefore, it is recommended that future intervention studies include both consumer-reported and clinician/pharmacist-reported outcomes to assess the overall health and functioning of people living with SPMI (Sartorius, 2014).
Education and training provided prior to pharmacists’ intervention
This systematic review identified that standardised training was not provided to pharmacists in the studies included in this systematic review. The authors recommend the delivery of standardised training such as Mental Health First Aid training should be provided to pharmacists prior to the intervention delivery to increase their confidence in working with people living with SPMI (El-Den et al., 2020b; O’Reilly et al., 2011, 2019), and to allow for comparisons across studies.
Barriers and facilitators of pharmacist-led interventions
None of the studies included in this systematic review specifically reported on barriers or facilitators to implementing a pharmacist-led intervention for people living with SPMI. Other studies demonstrated that common barriers to implementing pharmacy mental health interventions included consumer- and pharmacist-related factors such as pharmacists’ attitudes and beliefs, and also social and healthcare system-related factors such as stigma and the lack of privacy in community pharmacy settings (Aaltonen et al., 2010). The majority of studies identified in this review were conducted in hospital settings and one important barrier to consider is the transferability of effective interventions identified in this review from inpatient hospital settings to community pharmacy settings. Community-based treatment has the potential to provide better clinical outcomes (Hoult et al., 1983), and have demonstrated higher consumer satisfaction compared to treatment in a psychiatric hospital (Hoult et al., 1983). The lack of access to clinical information in the community pharmacy may explain the limited community based interventions (Blalock et al., 2013). Another reason for the majority of studies occurring in hospital settings may be related to time constraints, in that the successful implementation of pharmacist-led services for people living with SPMI in community settings will require appointment-based consultation (Tallian et al., 2012). Community pharmacies traditionally do not have the capacity in terms of staffing, infrastructure and reimbursement to deliver appointment-based consultations; however, the introduction and expansion of pharmacist-led interventions such as depression screening clinics has shed light to the potential of pharmacist-led services in the area of mental health (O’Reilly et al., 2015). More pharmacies are starting to have private consult rooms, which will allow consumers to comfortably talk about their mental health. Nonetheless, the literature pertaining to community pharmacy-led interventions for people living with SPMI is largely lacking and there is a need for further research in this space and overcome other barriers such as time, stigma and the lack of awareness of the pharmacists’ role (O’Reilly et al., 2015). As suggested in previous research, there is a need for future research to evaluate the effectiveness of these interventions in community settings (Bell et al., 2005).
Strengths and limitations of the review and future directions
The current review was conducted with strong methodological rigour, guided by the PRISMA guidelines and a pre-registered protocol (PROSPERO) (El-Den et al., 2020a). A key strength of this review is the comprehensive search strategy that was developed, piloted and applied with the support of multiple medical librarians, to ensure all relevant literature was identified, reviewed and included where eligible. The screening process was undertaken by two independent reviewers who determined each study’s eligibility to reduce the potential for selection bias. Importantly, a quality assessment of the studies was also conducted using validated tools such as the Cochrane Risk of Bias assessments (RoB 2 and ROBINS-I) to explore risk of bias within included studies.
This systematic review highlights the heterogeneity of included studies in terms of participant groups, type of outcome(s) measured, length of intervention, intervention components, intervention delivery and pharmacists’ qualification and training prior to intervention delivery. The lack of well-designed RCTs and high degree of heterogeneity identified in this review suggests that there are potentially important differences that need to be accounted for and standardised across studies to allow for accurate comparisons. Most studies had a short follow-up period of less than 6 months and only a limited number of studies that have study durations of up to a year. Previous literature exploring pharmacist-led interventions to improve antidepressant adherence recommends that the duration of the intervention and follow-up period should be at least 2 years in order to assess intervention effectiveness (Al-Jumah and Qureshi, 2012). In this study, it is unclear whether reported improvements were sustained beyond the study duration. To demonstrate whether pharmacist-led interventions lead to significant, sustained effects on consumers’ health outcomes there is a need for future studies to conduct methodologically robust RCTs with large sample sizes, across multiple settings, for longer durations and longer follow-up periods to assess the long-term impacts.
Conclusion
Overall, this systematic review has shown the positive outcomes of pharmacist-led interventions for people living with SPMI and has highlighted a number of significant gaps in the current literature regarding pharmacist-led interventions for people living with SPMI. Future research should attempt to better understand which specific intervention components have the greatest impact and evaluate the long-term sustainability of such interventions.
Footnotes
Acknowledgements
The authors thank Ms Bonnie Dixon from Griffith University and Dr Yulia Ulyannikova from The University of Sydney for their help with the development and revision of the search strategy.
Author Contributions
R.N., S.E.-D., S.S.M., A.J.W. and C.O.R. designed the review. R.N. carried out the database search, collected and analysed the data and prepared the first draft of the manuscript. R.N. and V.S. conducted the screening. C.O.R., S.E.-D., S.S.M., A.J.W., V.S., J.C.C. and H.R. contributed to and reviewed the manuscript. R.N., C.O.R. and S.E.-D. worked to finalise the manuscript. All authors approved the final version of the manuscript and agreed to be accountable for all aspects of the work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: R.N. is supported by the Australian Government Research Training Programme (RTP) Scholarship. V.S. is supported by a Griffith University Postgraduate Association Scholarship.
