Abstract
Objectives:
Tardive dyskinesia, psychotic relapse and treatment-refractory psychosis have long been associated. A common underlying mechanism involving antipsychotic-induced ‘supersensitivity’, albeit in different brain pathways, was proposed as early as 1978. This piece seeks to reappraise the concept and potential implications of antipsychotic-induced supersensitivity.
Conclusions:
Evidence increasingly suggests that chronic antipsychotic exposure induces neuroadaptive physiological changes in dopaminergic, and other, neurotransmitter systems that may render some individuals more vulnerable to psychotic relapse - including those receiving continuous antipsychotic treatment. It is possible that in treating every episode of psychosis with prolonged or indefinite antipsychotic therapy, we paradoxically increase the risk of psychotic relapse in a significant proportion of people. A greater appreciation of supersensitivity may allow us to optimise any potential benefits of antipsychotics while minimising the risk of inadvertent iatrogenic harms. More research is needed to improve our understanding of the underlying neurophysiology of supersensitivity and to better identify which individuals are most vulnerable to its development. It is time we paid more attention to the concept, emerging evidence and potential implications of antipsychotic-induced supersensitivity and, where appropriate, adjusted our practice accordingly.
Background
The recent meta-analysis published by Rubio et al (Lancet Psychiatry) demonstrated that patients with schizophrenia-spectrum disorders treated with continuous long-acting injectable (LAI) antipsychotics relapsed at a rate of 23 per hundred patient-years (Rubio et al., 2020). The strongest predictor of relapse was tardive dyskinesia (TD). The putative pathophysiology of TD involves upregulation and increased dopaminergic affinity of striatal dopamine-2 (D2) receptors (Teo et al., 2012). The process is one of basic synaptic physiology – chronic post-synaptic receptor blockade induces compensatory increases in both the density (Silvestri et al., 2000) and affinity (Seeman et al., 2005) of the receptors on the post-synaptic membrane. The response is hypothetically neuroadaptive, rooted in homeostatic mechanisms that allow neurons to maintain ‘synaptic equilibrium’ in the face of alterations to the neurophysiological environment (Hyman and Nestler, 1996). Antipsychotics create such an alteration through D2-receptor blockade, a mechanism thought crucial to their acute efficacy in reducing positive psychotic symptoms (Stahl, 2013).
Chronic D2-receptor blockade, however, has been demonstrated to cause 20–40% increases in the density of striatal D2-receptors, and 100–160% increases in the proportion of D2-receptors in the ‘high affinity’ state for dopamine (D2HIGH) (Samaha et al., 2007) – a process labelled ‘supersensitisation’. Admittedly, the assays that produced these figures did not allow for precise differentiation between pre- and post-synaptic changes; however, the authors drew on related evidence to suggest the clinically relevant changes are post-synaptic (see Figure 1) (Samaha et al., 2007).
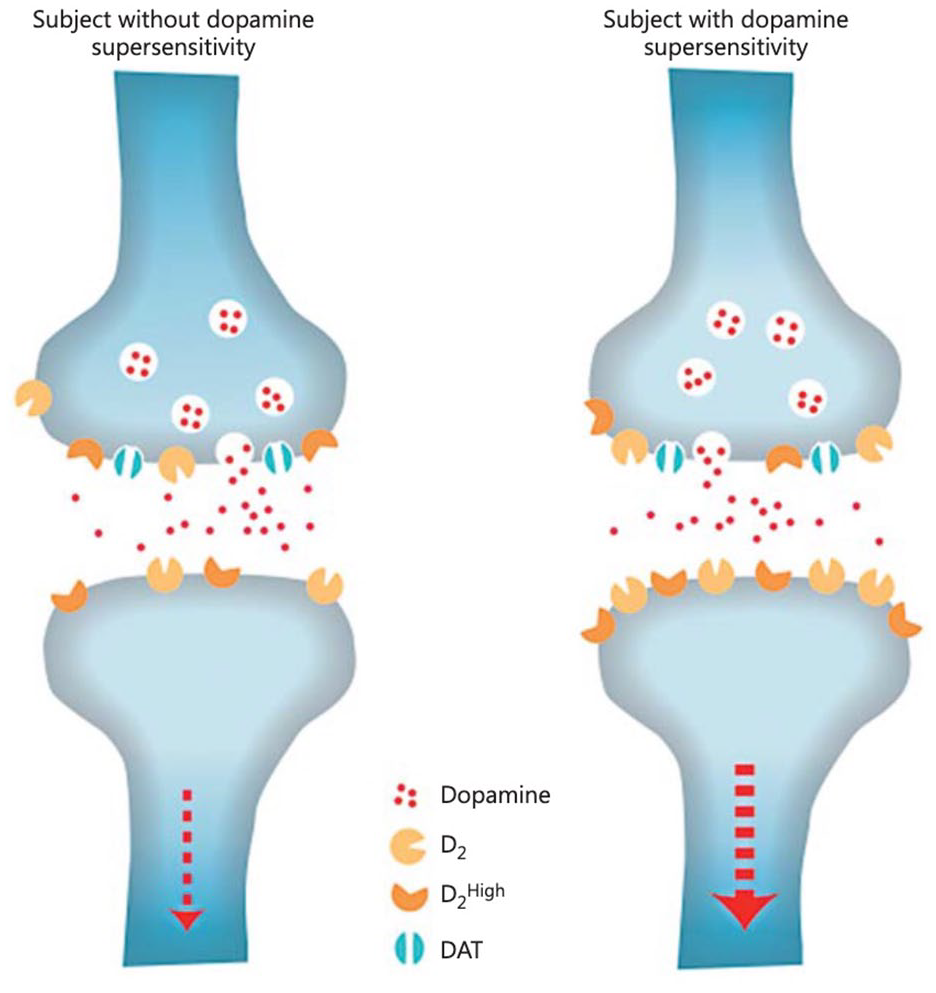
Diagram illustrating the neurophysiological changes that occur at a synaptic level underlying D2 supersensitivity. It is proposed that this occurs as a neuroadaptive homeostatic response to chronic D2 receptor antagonism caused by prolonged exposure to antipsychotics.
The pathophenotypic consequences of supersensitisation are hypothetically brain pathway-dependent – clinically, the evolving ‘tardive’ (i.e. late in onset) nomenclature reflects this. In the nigrostriatal pathway, we may see TD – that is, persistent, repetitive, abnormal involuntary movements, often of the lips and tongue (Teo et al., 2012). In the mesocortical pathway we may see ‘neuroleptic-induced deficit syndrome’ (NIDS) – a state of emotional indifference, apathy, amotivation anhedonia and blunted affect (Ueda et al., 2016). NIDS may be considered associated with, or analogous to, what others have termed ‘tardive dysmentia’ – a state of affective blunting, reduced activation and diminished interpersonal activity (Wilson et al., 1983). In the mesolimbic pathway, it is hypothesised we may see ‘neuroleptic-induced supersensitivity psychosis’ (Chouinard et al., 1978) or, where the psychosis is chronic and treatment-refractory, ‘tardive psychosis’ (Palmstierna and Wistedt, 1988). Indeed, worsening psychopathology in people with schizophrenia has been associated with worsening TD in a dose-dependent manner, suggesting ‘pandopaminergic’ dysfunction occurring concomitantly in both the mesolimbic and nigrostriatal pathways (Tenback et al., 2007). The development of TD has been associated with certain genetic vulnerabilities (Tiwari et al., 2005), it follows that the same may apply for supersensitivity psychosis (SP).
Neuroleptic-induced Supersensitivity Psychosis (NISP)
NISP was first described by Canadian psychiatrist Guy Chouinard in 1978. It describes a psychotic state hypothetically precipitated by stimulation of a ‘supersensitised’ dopaminergic post-synaptic membrane. It may occur during antipsychotic dose reduction or cessation, or during continuous treatment. Throughout this piece, SP refers to NISP.
Chouinard originally reported three key pieces of evidence supporting his theory of NISP (Chouinard et al., 1978):
People developed CNS tolerance to neuroleptics.
Psychosis following withdrawal of neuroleptics was correlated with signs of dopaminergic supersensitivity (e.g. TD).
Psychosis was associated with a sudden drop in prolactin following cessation of the neuroleptic.
Chouinard hypothesised that TD and psychotic relapse were caused by the same mechanism, albeit in different brain pathways. He made several further observations that that supported this theory: (1) increasing the dose of neuroleptic decreased the severity of both TD and psychosis whereas, (2) decreasing the neuroleptic worsened them, (3) stress appeared to exacerbate both, as did and (4) amphetamines and L-dopa (dopamine agonists). He summed up his theory and implications as such: We suggest that neuroleptics can produce a dopamine supersensitivity that leads to both dyskinetic and psychotic symptoms. An implication is that the tendency toward psychotic relapse in a patient who has developed such a supersensitivity is determined by more than just the normal course of illness. This may explain why... [we are] unable to identify “good prognosis” patients who do not relapse when maintenance neuroleptics are discontinued. (Chouinard et al., 1978)
Forty-two years later, Rubio et al tell a familiar dyskinetic-psychotic story, pointing out that relapse seemed to occur in a ‘sizable proportion of cases’ even when the patient had attained full symptomatic remission with continuous antipsychotic treatment. In line with Chouinard, Rubio et al suggested that treatment responsiveness may be ‘dynamic for some individuals’ and acknowledge that TD, along with substance use, may be involved in the mechanism of relapse (Rubio et al., 2020). The authors hypothesised that the association between substance use and relapse may result from drug-induced ‘destabilisation’ of a dopaminergic system already ‘aberrantly regulated’ consequent to chronic dopaminergic blockade (Rubio et al., 2020). It is possible this ‘aberrant regulation’ is, in fact, dopamine supersensitivity.
An important feature of supersensitivity is the gradually reduced efficacy of continuous antipsychotic treatment. Increasing the dose may, at least transiently, improve efficacy (i.e. drug tolerance), but ‘treatment failure’ and ‘breakthrough psychosis’ appear inevitable in some (e.g. the 23% in the Rubio study). Continuous D2 occupancy at levels deemed at or above threshold for movement disorders (i.e. approx. 80%) (Stahl, 2013) induces a compensatory increase in both D2 density and affinity for D2HIGH as a neuroadaptive response to persistently curtailed dopamine signalling. Consequently, the total proportion of D2 receptors occupied by antipsychotic decreases over time, potentially falling below the putative minimum required for antipsychotic efficacy (i.e. approx. 60–80%). Clinically, the result is the need for higher doses, or additional agents with higher D2-affinity, or both, to control symptoms. Hypothetically, the inadvertent consequence is further supersensitisation, movement disorders and ‘breakthrough’ psychoses (see Figure 2). Hence SP is not only confined to withdrawal states, but also instances of continuous antipsychotic exposure.
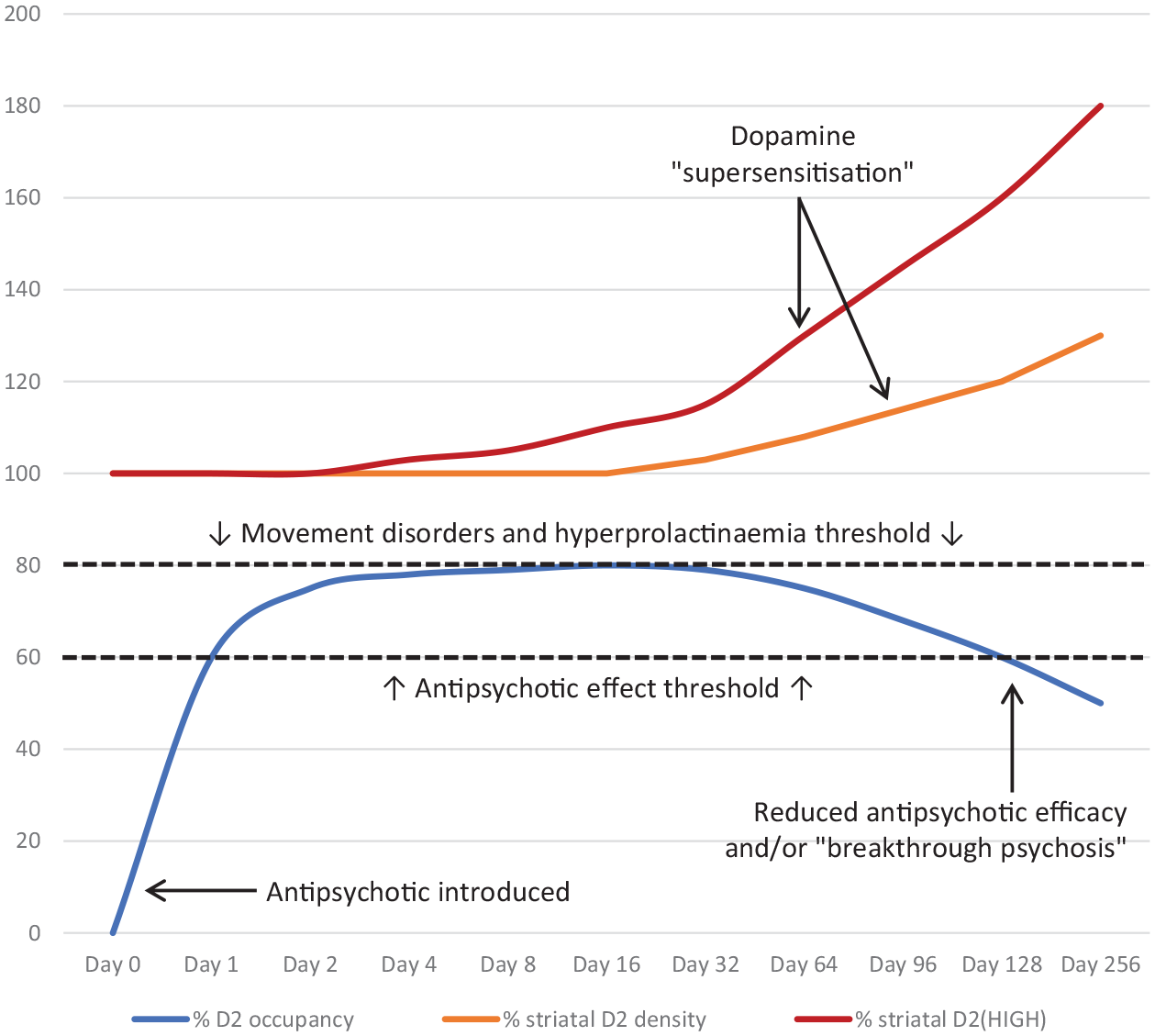
Hypothetical graphical representation of receptor-mediated dopamine supersensitisation over time in the setting of continuous antipsychotic exposure. Note the gradual reduction in antipsychotic efficacy when D2-receptor (antipsychotic) occupancy falls below 60–80% (Stahl, 2013). Putatively, this occurs consequent to increases in both the density of striatal D2-receptors and the proportion of D2-receptors in the high affinity state for dopamine (D2HIGH) (Samaha, 2007). Some evidence suggests clinically significant changes may only take 3 months (Chouinard, 2017). Increased striatal D2 density is believed to occur through either an increase in D2-receptor synthesis, a decrease in D2-receptor degradation, or both (Chouinard, 2017). It is important to note that not everyone will experience reduced antipsychotic efficacy and/or ‘breakthrough psychosis’ at, or beyond, 3 months cumulative antipsychotic exposure.
As evidence supporting the supersensitivity hypothesis continued to emerge, by 1991 diagnostic criteria for SP were proposed. In a cohort of 224 patients with chronic schizophrenia receiving continuous, long-term antipsychotic treatment, 45% developed TD and 22–43% developed SP (Chouinard, 1991). In an earlier paper, Chouinard et al. reported that in a sample of 300 patients with chronic schizophrenia treated continuously with first-generation antipsychotics (FGAs), 30% showed signs of SP (Chouinard and Jones, 1982). The relapse rates from these studies are notably similar to those demonstrated by Rubio et al in 2020.
More recently, one group reviewed the prevalence of SP in 3 studies examining people taking second-generation antipsychotics (SGAs) only. Hypothetically, owing to their relatively decreased affinity for D2, SGAs should be associated with a lower prevalence of SP. The review found otherwise. Similar to previous studies with FGAs, the authors reported a SP rate of 30% in those with schizophrenia, and up to 70% in those with so-called ‘treatment-resistant’ schizophrenia (Chouinard et al., 2017). In the same paper, refined diagnostic criteria for SP were proposed which more precisely captured psychotic symptoms co-occurring with movement disorders in the setting of discontinuation, dose reduction, switch, or in-between dose intervals of antipsychotics of 3 or more months cumulative exposure (Chouinard et al., 2017).
Recent longitudinal data on chronic antipsychotic exposure
In 2018, Tiihonen et al analysed the outcomes of 8378 patients diagnosed with first episode psychosis (FEP) over several years. The authors compared the relapse rates of those who continued antipsychotics versus those who discontinued (Tiihonen et al., 2018). The lowest overall risk of rehospitalisation or death was seen in the group who received ‘continuous antipsychotic treatment’ – made the reference group. More interestingly, however, the second lowest risk was seen in those who discontinued antipsychotic use immediately post discharge. Those who discontinued after 5 years (the longest group) had the highest risk of rehospitalisation or death (hazard ratio [HR] = 7.28) (Tiihonen et al., 2018). The study did not comment on the prevalence of TD or movement disorders, but the findings arguably provide evidence in support of the supersensitivity hypothesis in demonstrating that the longer the duration of antipsychotic treatment, the greater the risk therefore, of relapse upon discontinuation. The key counter-observation is that continuous antipsychotic treatment was protective in some–but this may be expected. As previously discussed, once supersensitivity is established, the continuous (or increased) presence of antipsychotics (D2 occupancy) may be protective as it counteracts the increased density and affinity of D2 receptors. This ‘protective’ effect is compromised, however, by any process that sufficiently stimulates the supersensitised dopaminergic post-synaptic membrane either via sudden reduction/cessation of the D2 antagonist or by significant provocation of mesolimbic dopaminergic neurotransmission – key drivers of which include psychological stress and substance use (Pruessner et al., 2004). Substance use was the second greatest predictor of relapse in the Rubio el al study.
Abrupt cessation of antipsychotics and relapse
In keeping with the supersenstivity hypothesis, abrupt cessation of antipsychotics frequently leads to relapse. One meta-analysis reported that relapse rates halved in those who tapered antipsychotics over the course of 3–9 months compared to abrupt cessation (Viguera et al., 1997). However, tapering over just 4 weeks made no difference to relapse rates (Leucht et al., 2012). Speculatively, the disparity is due to the same synaptic homeostatic mechanisms that underly supersensitivity – albeit in reverse. Over months of slow, symptom-tailored withdrawal, post-synaptic D2 receptors downregulate both the total number of receptors and the proportion in the ‘high affinity’ state. Thus, when a slow taper is applied there is a hypothetically reduced risk of abruptly exposing a supersensitised postsynaptic membrane, and therefore a reduced risk of SP. Furthermore, Horowitz et al recently highlighted the hyperbolic nature of D2 receptor occupancy with both increasing and decreasing doses of D2-antagonists (Horowitz et al., 2020); it is plausible that in order to further reduce the risk of SP on withdrawal, not only is a slow taper of antipsychotic required, but a logarithmic one.
Prevention of supersensitivity
Prevention of SP (and TD) involves prevention of supersensitisation in the first place – guidelines have been proposed. The key recommendations include detection and prevention of excessive D2 antagonism (as evidenced by signs of SP or TD) and the cautious and judicious use of particular SGAs at doses with the lowest association with SP or movement disorders (see Table 1.). Admittedly, this approach is not always possible, particularly where there is a complex or complicated history and/or a risk of harm to self or others necessitating alternative treatment pathways. This is a separate discussion beyond the scope of this piece.
Proposed guidelines for the prevention of supersensitivity psychosis (SP) and tardive dyskinesia (TD) (Adapted from Chouinard, 2017).

Source: Adapted from Chouinard (2017).
Dose-reduction / cessation of antipsychotics
In a randomised trial involving patients diagnosed with FEP (in remission), Wunderink et al compared the outcomes of two groups at 2 and 7 years – one received antipsychotic dose reduction/discontinuation and a second received ‘maintenance treatment’. At 7 years, the dose reduction/discontinuation group experienced twice the recovery rate (defined as symptomatic and functional remission) of those in the maintenance treatment group. Twenty-one percent of dose reduction/discontinuation group had ceased antipsychotics entirely. Symptom remission rates were similar in both groups, but the maintenance treatment group had higher overall relapse rates (did not differ significantly) (Wunderink et al., 2016). Prevalence of movement disorders or TD are not mentioned, and it is important to note this study has been criticised for its methodology.
A group led by Joanna Moncrieff at University College London are currently conducting a multi-centre randomised controlled trial of gradual antipsychotic reduction/discontinuation in people with schizophrenia and related disorders – the so-called ‘RADAR trial’. This study has randomised people to either antipsychotic dose reduction/discontinuation or maintenance treatment (Moncrieff et al., 2019). The results of this trial will provide relatively long-term outcome data on several important variables including symptomatology, relapse rates, social functioning and more – it may replicate the findings of the Wunderink study. It would be fascinating to analyse the two groups for signs of supersensitivity (as described by Chouinard et al.).
Criticism
Critics of the supersensitivity hypothesis often point out that even if the relapse rate for people on continuous antipsychotics is 23%, it is still better than that seen in those with FEP who discontinue antipsychotics – a recent meta-analysis provided figures of up to 77% and 90% at 1 and 2 years, respectively (Zipursky et al., 2014). But if one looks carefully at this data, yet again it is arguably consistent with the supersensitivity hypothesis. Individuals in the Zipursky study had taken antipsychotics for an average 15 months prior to discontinuation – evidence suggests ‘supersensitisation’ may only take some 3 months (Chouinard et al., 2017; Silvestri et al., 2000). Conceivably, therefore, those who relapsed at 1 and 2 years in the Zipursky study were already ‘supersensitised’, as they arguably were in the Tiihonen study. Other studies examining relapse rates in FEP suffer the same confounding because subjects are invariably exposed to prolonged (i.e. >3 months) antipsychotic therapy prior to cessation.
Another criticism is derived from the observation that intermittent and as-needed (symptomatic) antipsychotic treatment approaches have been studied and found to be inferior to continuous treatment. In 2002, Gaebel et al compared the relapse rates of people with first- or multi-episode (mean duration of illness, 9.7 years) schizophrenia over 2 years. One group was randomised to intermittent/symptom-responsive antipsychotic treatment, and another to ‘maintenance treatment’. The multi-episode group experienced more relapses overall than the first-episode group, with fewer relapses when treated with continuous antipsychotics (Gaebel et al., 2002). Those in the first-episode group, however, not only had fewer relapses overall but faired equally well when using either maintenance or intermittent/symptom-responsive treatment (Gaebel et al., 2002). A reasonable interpretation of these results is that the multi-episode group were more unwell, relapsed more, and therefore required continuous treatment to better manage their illness. An alternative view is that the multi-episode group were already supersensitised consequent to years of prior antipsychotic exposure (whereas the first-episode group were not) thus rendering them more susceptible to relapse, whatever the precipitant. In other words, the neuroadaptive horse had bolted. In this context, the increased relapse rates in the multi-episode group make sense, as do the better results they experienced with continuous antipsychotic treatment. It would have been intriguing to compare the relapse rates (and signs of supersensitivity) between these two groups over the 20 years since this study ended had they continued their antipsychotic regimens exactly as in the trial.
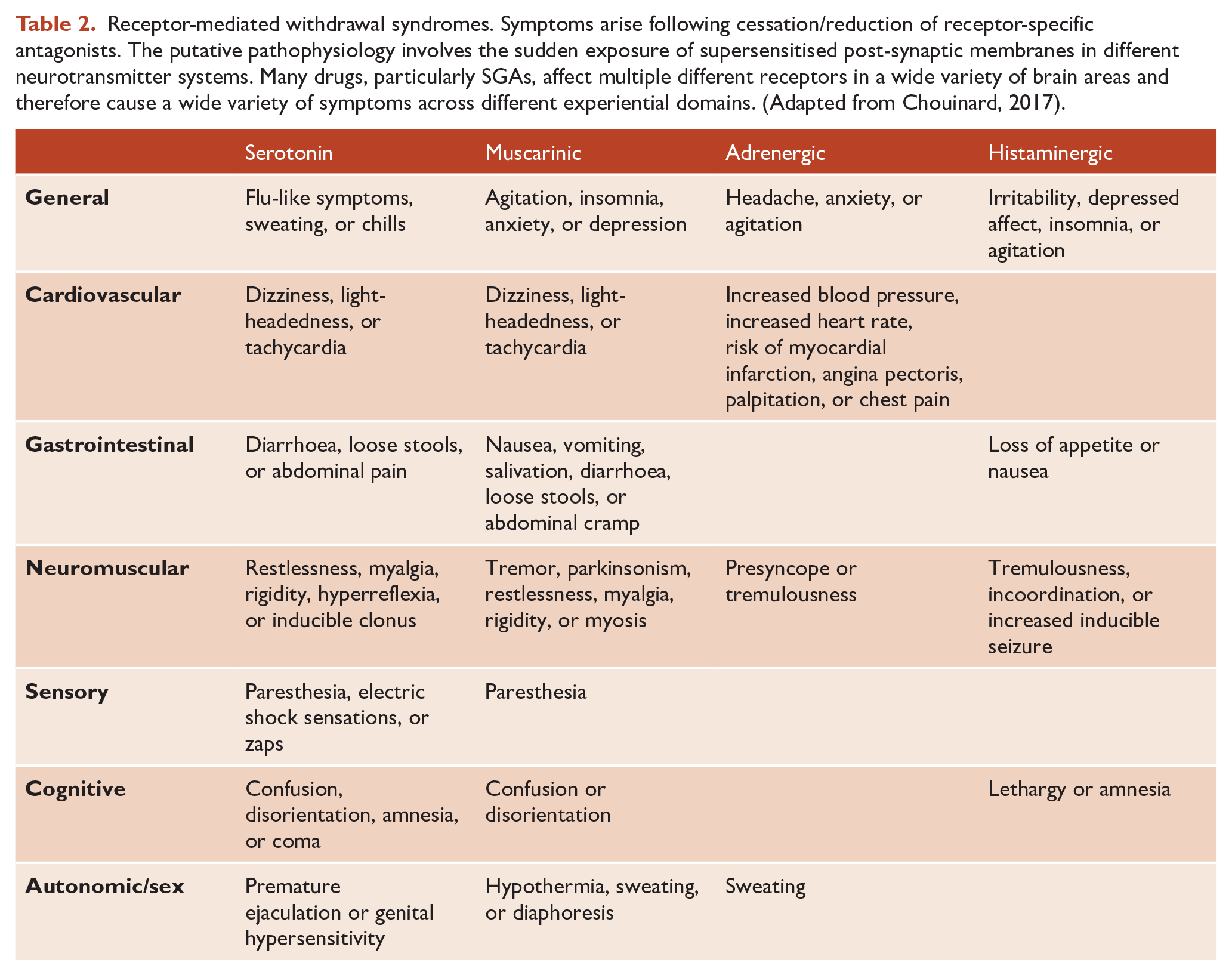
Another key counterargument pertains to D2 partial agonists (e.g. aripiprazole). Some evidence suggests (at least in animal models) that chronic treatment with partial agonists does not increase D2 receptor density nor cause overt signs of (dopamine) supersensitivity (Tadokoro et al., 2012). The partial agonists should therefore have lower (supersensitivity-related) relapse rates–yet this has not been consistently demonstrated. Psychosis is infinitely more complex than pure dopamine dysregulation. Myriad structural and functional systems are involved, dysdopaminergia is likely to be but one of them–albeit perhaps an important one. Supersensitisation of other neurotransmitter systems are likely contributary. For example, chronic 5HT2A blockade (e.g. consequent to chronic treatment with most SGAs) may induce comparable neuroadaptive changes to those seen at D2, and it is reasonable to consider ‘serotonin supersensitivity’. It follows that receptor-mediated ‘histamine supersensitivity’ and ‘acetylcholine supersensitivity’ (among others) exist and the clinical evidence for these are found in the characteristic ‘rebound syndromes’ seen upon abrupt cessation of drugs that interfere with specific neurotransmitter systems (see Table 2). Given clozapine’s relative D2 minimality, ‘serotonin/histamine supersensitivity’ may be a contributing mechanism to the so-called ‘clozapine rebound psychosis’ (a profoundly psychotic state seen on abrupt cessation of clozapine).
Receptor-mediated withdrawal syndromes. Symptoms arise following cessation/reduction of receptor-specific antagonists. The putative pathophysiology involves the sudden exposure of supersensitised post-synaptic membranes in different neurotransmitter systems. Many drugs, particularly SGAs, affect multiple different receptors in a wide variety of brain areas and therefore cause a wide variety of symptoms across different experiential domains. (Adapted from Chouinard, 2017).
Implications
Appropriate short-term use of antipsychotics has been demonstrated to attenuate distress, reduce positive symptoms and improve social functioning (Huhn et al., 2019). Antipsychotics may be appropriate as an acute treatment where a person is tormented by positive psychotic symptoms and overt emotional distress. The long-term effects (i.e. beyond 3 months) of continuous antipsychotic treatment, however, remain largely unknown and may be harmful. As discussed in this piece, evidence suggests between 20% and 70% of people treated with long-term antipsychotics may develop dopaminergic supersensitivity (Chouinard, 1991; Chouinard et al., 2017). If the supersensitivity hypothesis has validity, then prolonged/indefinite antipsychotic treatment in those with FEP may represent a hazardous self-fulfilling prophecy: the longer the patient is exposed to the drug, the greater the postsynaptic neuroadaptive changes, leading to a higher propensity to relapse either during withdrawal or continuous treatment. The consequent relapse is then interpreted as evidence of the need for even more antipsychotic treatment – and thus the cycle continues. Supersensitivity, in this context, could be considered an inadvertent iatrogenic harm.
Prominent British schizophrenia researcher Robin Murray recently recommended psychiatrists regularly review the need for ‘prophylactic’ (continuous) antipsychotic treatment given the risk of causing D2 supersensitivity. Murray suggested up to 40% of those with FEP in remission could achieve good long-term outcomes with either complete cessation of antipsychotic treatment, or very low doses, thus avoiding supersensitisation (Murray et al., 2016). The obvious question is, how do we determine who is in the 40%? It is interesting to note that the current Royal Australian and New Zealand College of Psychiatrists (RANZCP) clinical practice guidelines for schizophrenia and related disorders recommend all cases of FEP be treated with continuous antipsychotics for at least 2–5 years (Galletly et al., 2016). If the subpopulation vulnerable to developing antipsychotic-induced supersenstivity could reliably be identified, future RANZCP guidelines should change from a one-size-fits-all approach and spare this group from prolonged antipsychotic exposure. Alternatively, irrespective of this precise knowledge, future guidelines could change anyway, so as to allow for treatment of FEP to begin with symptom-responsive/intermittent dosing (in line with the Gaebel study, where the first-episode group faired equally well using symptom-responsive/intermittent dosing) with the option of continuous antipsychotic treatment where clinically indicated. Dose reduction strategies remain under investigation.
Conclusion
Since 1978, evidence has grown in support of the antipsychotic-induced supersensitivity hypothesis and the recent meta-analysis by Rubio et al arguably adds further weight. Importantly, some individuals appear more susceptible than others, and this remains a vulnerability hitherto poorly understood. More research is needed to explore the pharmacodynamic-related neurophysiology of supersensitivity and to understand which individuals are most vulnerable, when treated with which antipsychotics and for how long, so we may optimise any potential benefits of antipsychotics and avoid inflicting inadvertent iatrogenic harms. It is time we paid more attention to the concept and potential implications of supersensitivity and, where appropriate, adjusted our practice accordingly.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
