Abstract
Objective:
Whilst cannabis has been associated with an earlier age at onset in schizophrenia, the impact of amphetamine and/or cocaine plus cannabis consumption on age at onset remains unclear. The present study was designed to test the hypothesis that consumption of amphetamine and/or cocaine in addition to cannabis would lead to an earlier age at onset of schizophrenia than that seen for cannabis consumption alone. A secondary objective was to determine what kind of effect additional substance use exerted (e.g. additive, multiplicative).
Method:
Patients with a diagnosis of schizophrenia were recruited from consecutive admissions to the inpatient and outpatient services of a large psychiatric hospital in Perth, Australia and 167 participants were assessed using the Diagnostic Interview for Psychosis, which included detailed inquiry into illicit drug use in the 12 months prior to the onset of psychiatric symptoms. Participants were categorized into four groups: no illicit substance use (n = 65), cannabis use (n = 68), cannabis plus amphetamine use (n = 25), and cocaine plus cannabis/cocaine plus cannabis plus amphetamine use (n = 9). Analysis of variance was performed to detect trends, and linear regression used to analyze the consumption of each additional substance as a predictor of age at onset.
Results:
We observed a linear trend for mean age at onset: 23.34 (SD = 6.91) years for no illicit substance use, 22.51 (SD = 5.27) years for cannabis use, 20.84 (SD = 3.48) years for cannabis plus amphetamine use, and 19.56 (SD = 3.54) years for cocaine plus cannabis/cocaine plus cannabis plus amphetamine use; the variation in the means between groups was statistically significant: F(1,163) = 5.66, p = 0.008, Cohen’s d = 0.38. For the consumption of each additional substance, age at onset was earlier by 1.2 years: R2 = 0.034, F(1,165) = 5.72, p = 0.018.
Conclusions:
Whilst preliminary, these findings suggest that additional consumption of each substance predicted an earlier age at onset by approximately 1 additional year.
Introduction
Age at onset in schizophrenia: why does it matter?
Schizophrenia is a chronic and disabling disorder associated with significant health, social and financial burden. Estimates suggest psychosis costs Australia around $1.45 billion per annum, while societal costs (such as earnings forgone) approximate at $2.25 billion (Carr et al., 2003), or 0.23% and 0.36% of Australian gross domestic product (GDP), respectively. Earlier age at onset has been identified as a crucial factor and poor prognostic feature in schizophrenia (DeLisi, 1992; Veen et al., 2004) and has long been associated with non-modifiable risk factors, such as sex and genetics (DeLisi, 1992; Leung and Chue, 2000). Being able to identify and target modifiable risk factors, and at the very least delay the onset of schizophrenia in a proportion of people at risk, could allow for a number of years of psychosis-free functioning. As outlined by Large and colleagues (2011), one would anticipate that this psychosis-free window would be an opportunity for many at-risk individuals to achieve basic developmental milestones during late adolescence and early adulthood, and could have a significant impact not only on the distress and disability endured by patients, but also on the substantial financial costs of schizophrenia.
Illicit substances and age at onset
Illicit substance use has been associated with age at onset in schizophrenia, with a number of studies reporting that substance-using patients were younger than their non-using counterparts (Addington and Addington, 1998; Barnes et al., 2006; DeQuardo et al., 1994); however, other studies found no significant effect (Cantor-Graae et al., 2001; Sevy et al., 2001). More recently it has been demonstrated that there exists a stronger association between cannabis use, compared with other substances, and earlier onset of psychosis (Barnes et al., 2006; Gonzalez-Pinto et al., 2008; Veen et al., 2004). Several hypotheses have been proposed to explain this link, including that the illness is precipitated by cannabis (i.e. causality), possibly in those predisposed to develop psychosis (Henquet et al., 2006; Stefanis et al., 2004), or that early onset of symptoms is a risk factor for substance use (i.e. reverse causality), such that cannabis is used by patients to self-medicate, for instance (Addington and Addington, 1998). Others have suggested the association could be explained by confounding factors such as age (i.e. older patients are less likely to use cannabis) and sex (i.e. a larger proportion of male users) (Sevy et al., 2010).
Dose–effect of cannabis and age at onset
An association between the dose–effect of cannabis exposure and schizophrenia risk has been reported (Andreasson et al., 1987), and several recent studies have focused on a possible association between dose–effect and age at onset. In a meta-analysis, Large and colleagues (2011) reported an earlier mean age at onset of psychosis in samples with more cannabis users, and found not only that this relationship was independent of age, but no evidence was present that the association could be explained by male sex. The meta-analysis also reported an association between the extent of cannabis use and the effect size (earlier age of onset by 2.72 years in the heavy or continuous users, compared with 2.07 years in those with lighter use or who had stopped using), suggesting a dose–response effect; however, this difference between the groups was not statistically significant. A dose–response effect has also been reported in a number of previous studies, where a significant gradual reduction of age at onset was found as dependence on cannabis increased (Fergusson et al., 2005; Gonzalez-Pinto et al., 2008).
Sensitization to illicit substance exposure?
These findings further strengthen the causality argument, and raise intriguing questions about the mechanism by which cannabis could potentially mediate the onset of psychotic disorders. Whilst it is possible that there is an interplay of a number of factors (such as family history, adverse childhood events, and socio-demographic factors), there is a large body of literature examining the possible abnormalities of brain structure and function. It has been suggested that perturbations in striatal biochemistry likely underpin this process and dopamine, which has long been considered in the aetiology of psychosis (Davis et al., 1991; Howes and Kapur, 2009; Van Rossum, 1966), has been suspected to be involved at some level (Luzi et al., 2008; Stefanis et al., 2004). Whilst the role of dopamine remains unclear and neuroimaging data of striatal dopaminergic activity following cannabis exposure, for instance, are conflicting (Bossong et al., 2009; Safont et al., 2011; Stokes et al., 2009; Voruganti et al., 2001), there is converging evidence that sensitization to repeated illicit substance exposure is important in the genesis of psychotic symptoms (Paparelli et al., 2011). This has led some authors to suggest a mechanism involving sensitization of the mesolimbic dopaminergic system, triggered by repeated stimulation with cannabis, to which genetically susceptible individuals may be especially sensitive (Kuepper et al., 2001; Stefanis et al., 2004; Van Os et al., 2010).
Study objective
In keeping with this hypothesis, one might postulate that if other substances with potential to sensitize brain circuitry in addition to cannabis were consumed (e.g. stimulants such as amphetamine or cocaine) (Paparelli et al., 2011), this might further lower age at onset, in essence compounding the effect seen for cannabis use. Studying the effects of amphetamine exposure on brain biochemistry has long illuminated many avenues for further schizophrenia research and, of note, underpins the dopamine sensitization hypothesis of schizophrenia. Amphetamine use increases the risk of psychosis, particularly if used earlier or in greater amounts (Chen et al., 2003; Farrell et al., 2002). Neuroimaging studies have reported approximately twice as much amphetamine-induced dopamine in the striatum of patients with schizophrenia compared to healthy controls (Laruelle et al., 1996), and increasing striatal dopamine release in response to repeated amphetamine exposure over time in healthy men (Boileau et al., 2006). Similarly, sensitization of striatal biochemistry has been observed following repeated cocaine administration, as has enhanced dopamine release in the striatum (Akimoto et al., 1989, 1990; Centonze et al., 2007). Further, it has been postulated that common mechanisms of sensitization are shared by stimulant-induced behavioral sensitization in rodents, stimulant-induced psychosis in humans and chronic schizophrenia (Ujike, 2002). Hence, evaluating the potential interaction of amphetamine and/or cocaine in addition to cannabis on age at onset is warranted.
Whilst several studies have examined the association between the number of illicit substances consumed and age at onset, and reported no augmentation by the use of other drugs in addition to cannabis (Barnes et al., 2006; Gonzalez-Pinto et al., 2008), few if any studies have specifically evaluated the effect on age at onset of consumption of cannabis versus cannabis plus amphetamine, versus cocaine plus cannabis/cocaine plus cannabis plus amphetamine. The most likely reason for this are the small numbers of participants involved in these studies, and the significant polysubstance use, restricting subcategorization of samples. The present study was designed to test the hypothesis that consumption of amphetamine and/or cocaine in addition to cannabis would lead to an earlier age at onset of schizophrenia than that seen for cannabis consumption alone. A secondary objective was to determine the nature of the potential effect exerted by additional substances; for example, whether additional use exerted a multiplicative effect exerted a multiplicative or additive effect, for instance.
Method
The study has been approved by the North Metropolitan Area Mental Health Service Ethics Committee and by the Human Research Ethics Committee of the University of Western Australia; the project was conducted according to the principles of the Declaration of Helsinki. Written informed consent was obtained from all participants prior to interview.
Participants
A total of 167 patients were selected from the Western Australia Family Study of Schizophrenia (WAFSS), on the basis of available data regarding drug and alcohol use (see below). The WAFSS is comprised of a large cohort of patients aged 18–65 years and recruited from consecutive admissions to the inpatient and outpatient services of a large psychiatric hospital in Perth, Australia, with a diagnosis of schizophrenia or schizophrenia spectrum disorder made by a psychiatrist, and has been described elsewhere (Hallmayer et al., 2005; Jablensky, 2006). Exclusion criteria included a history of head injury (loss of consciousness > 10 minutes), a neurological disorder, and those who were not fluent in English, had not completed at least 8th grade of education, or were unable to give consent.
Measures
Participants were assessed using the Diagnostic Interview for Psychosis (DIP), a standardized semi-structured interview for psychosis designed to provide a diagnosis, assess symptom profiles, social functioning, disability and service utilization (Castle et al., 2006; Jablensky et al., 2000). The DIP is comprised of several modules (demography and social functioning, diagnostic, and service utilization). The diagnostic module (DIP-DM) follows the structure of the Operational Criteria for Psychosis (OPCRIT) (McGuffin et al., 1991), a 90-item checklist which allows the examiner to rate symptoms (present state, past year, and lifetime) in a number of domains. Performance on the DIP was scored using the OPCRIT diagnostic algorithm (McGuffin et al., 1991), and video-recorded interviews reviewed independently by two psychiatrists who assigned consensus research diagnoses. All participants met both International Classification of Diseases (ICD)-10 and Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV) criteria for a lifetime diagnosis of schizophrenia.
The DIP-DM also includes a detailed section for assessing current and past drug and alcohol use. The Substance use – Alcohol section includes questions on frequency and quantity of alcohol use in the 12 months before psychiatric symptoms first appeared. The Substance use – Drug section rates the frequency of drug use (cannabis, amphetamines, tranquillizers, heroin, cocaine, LSD/hallucinogens, ecstasy, inhalants/solvents) in the 12 months before psychiatric symptoms first appeared. Frequency is rated as not used, daily/almost daily, 1–2 days/week, two to four times/month, less than monthly, use not known.
Definition of age at onset
The age at onset was determined after interviewing the patient (i.e. ‘When did you first experience psychiatric problems?’; ‘When did others say that they thought you had a psychiatric problem?’; ‘How old were you when you first had contact with psychiatric services?’), interviewing available informants, and then reviewing the hospital file. Age at onset was recorded to the nearest year, and was defined as the earliest age at which medical advice was sought for psychiatric reasons, or at which symptoms began to cause subjective distress or impair functioning; the recorded age at first hospital admission either if there were no clear symptoms, or if it was the only available age (e.g. patient denies illness, no informant).
Definitions of substance using and non-using groups
Participants (n = 167) for whom information had been recorded in the DIP-DM regarding drug and alcohol use were included for analysis. These patients were categorized as either substance-users (if they reported daily/almost daily use, use 1–2 days/week, or two to four times/month, in the 12 months before psychiatric symptoms first appeared), or substance non-users (if they reported either no use, or used less frequently than once per month in the 12 months prior to the onset of psychiatric symptoms).
We attempted to determine if the consumption of amphetamine and/or cocaine in addition to cannabis would lead to an earlier age at onset of schizophrenia than that seen for cannabis consumption alone. Participants were divided into four groups depending on whether they had used no substances, or had used cannabis (but no cocaine, and no amphetamines), amphetamines (and used cannabis, but no cocaine) or used cocaine (plus cannabis/ plus cannabis plus amphetamine) in the 12 months prior to the onset of psychiatric symptoms. These groups were defined as follows: ‘no illicit substance use’ (n = 65), ‘cannabis use’ (n = 68), ‘cannabis plus amphetamine use’ (n = 25), ‘cocaine plus cannabis/cocaine plus cannabis plus amphetamine use’ (n = 9) (see Figure 1).
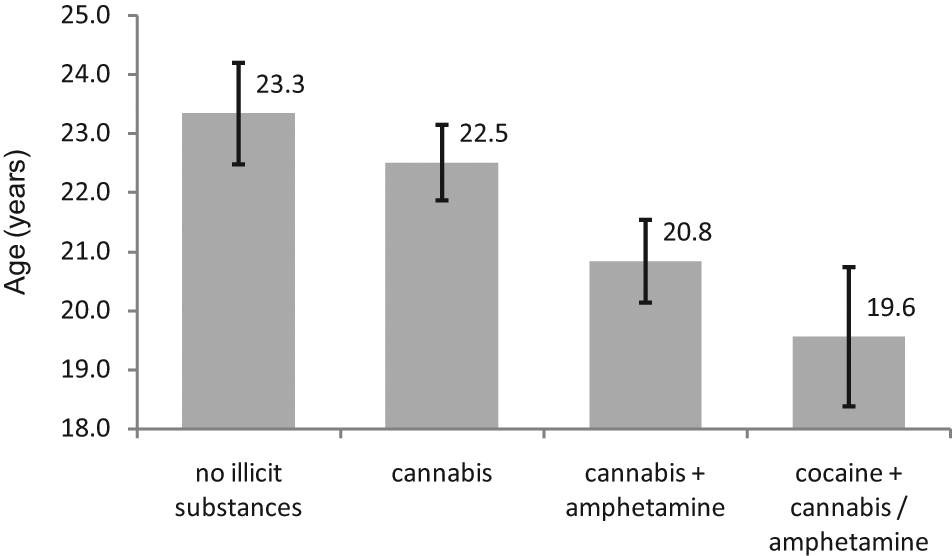
Figure showing the mean age at onset (with standard errors) of schizophrenia for groups according to illicit substance use. Groups included in the analysis: no illicit substance use (n = 65), cannabis use (n = 68), cannabis plus amphetamine use (n = 25), cocaine plus cannabis/cocaine plus cannabis plus amphetamine use (n = 9). The variation in the means between groups was statistically significant: F(1,163) = 5.66, p = 0.008, Cohen’s d = 0.38.
Within the three drug-using groups, a number of participants (n = 17) had used additional substances more frequently than once per month in the 12 months prior to the onset of psychiatric symptoms. Use of additional substances were distributed in the three drug-using groups as follows (bearing in mind that individual participants may have used a number of the following substances): in the cannabis group, four participants had used LSD, three used ecstasy and two used heroin; in the cannabis plus amphetamines group, three had used LSD, two used ecstasy and three used heroin; in the cocaine group, two had used LSD, two used ecstasy and two used heroin.
Given that a large proportion of all participants were reported as having used alcohol in the 12 months prior to the onset of psychiatric symptoms, excluding these participants would have prevented further analysis.
Statistical analysis
In the first instance, descriptive analyses were performed on the data set to ascertain demographics of the subset of 167 participants. To evaluate the effect of substance use on age at onset we performed analysis of variance (ANOVA) to ascertain trends between the groups; we tested for several common trends (linear, quadratic and cubic). Tests were one-tailed, given that we expected a trend for cannabis use and age at onset; we anticipated a similar trend for cannabis plus amphetamine use and, by analogy, for the addition of cocaine use). In order to ascertain whether the consumption of additional substances were mediating the observed effect, we repeated the analyses for the groups, in turn excluding those participants who had used heroin, LSD, or ecstasy in the 12 months prior to the onset of psychiatric symptoms. Finally, we used linear regression analysis to determine the effect (in years) on age at onset of schizophrenia with the consumption of each additional substance.
Results
The mean reported age of the sample of 167 participants was 33.77 (SD = 9.41) years, and the age range was 18–58 years. The mean age at onset was 22.42 (SD = 5.74) years, and the mean years of education was 11.14 (SD = 1.96). Males accounted for 81.0% of the sample, 24.4% had a family history of schizophrenia, and 32.1% had poor premorbid social adjustment. The sample of 167 patients included in this study were representative of the other 286 patients in the WAFSS cohort for whom drug and alcohol information was not recorded on the DIP (no statistically significant differences in demographic data were found between the groups; data not shown).
When we analyzed the data according to four categories (no illicit substance use, cannabis use, cannabis plus amphetamine use, cocaine plus cannabis/cocaine plus cannabis plus amphetamine use), we observed a linear trend (Figure 1). The mean age at onset for each group was as follows: 23.34 (SD = 6.91) years for those who did not use substances, 22.51 (SD = 5.27) years for those who used cannabis, 20.84 (SD = 3.48) years for those who used cannabis plus amphetamines, and 19.56 (SD = 3.54) years for those who used cocaine plus cannabis/cocaine plus cannabis plus amphetamine. The variation in the means between groups was statistically significant: F(1,163) = 5.660, p = 0.008, Cohen’s d = 0.381. Two other trends (cubic and quadratic) were not significant. This trend remained significant when excluding from the analysis, in turn, those participants who had used LSD, ecstasy or heroin in the 12 months prior to the onset of psychiatric symptoms. We also tested for the potential confounding effect of a family history of schizophrenia, male sex, mean length of illness, and mean years of education (Table 1). The distribution of family history of schizophrenia, male sex and mean years of education was similar across all groups; however, the variance in the means for length of illness was statistically significant. When we controlled for this potentially confounding variable, our findings remained significant.
Table showing descriptive data for each of the groups, and variance between groups.
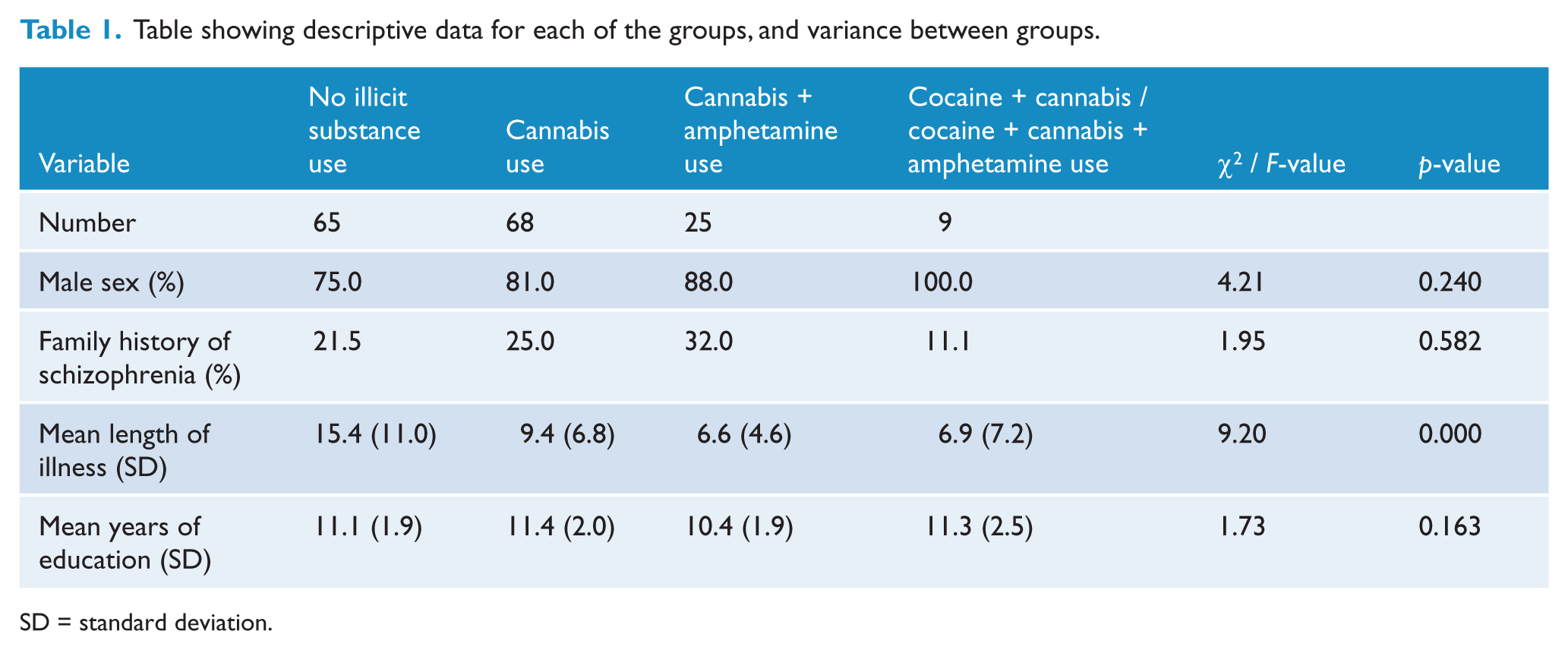
SD = standard deviation.
Given the linear trend observed, we used linear regression to consider the consumption of each additional substance as a predictor of age at onset. With the consumption of each additional substance (cannabis, amphetamine, cocaine), age at onset was earlier by 1.2 years: R2 = 0.034, F(1,165) = 5.722, p = 0.018.
Discussion
Findings
The results show that consumption of amphetamine and/or cocaine in addition to cannabis in the 12 months prior to the onset of psychiatric symptoms leads to an earlier age at onset in schizophrenia than that seen for cannabis consumption alone, and the trend observed was linear. We found that the additional consumption of each substance (cannabis, amphetamine, cocaine) predicted an earlier age at onset by approximately 1 additional year.
Implications of findings
If replicated, these findings will have important implications for those individuals using illicit substances who are susceptible to psychosis. Whilst preliminary, the potential public health message that emerges from these findings would be significant (e.g. one drug, 1 year; two drugs, 2 years). Further appropriately powered studies will be required to determine if this extends to illicit substances other than cannabis, amphetamine and cocaine. These findings add weight to the notion that there are modifiable factors that, if appropriately targeted, may increase the period of psychosis-free functioning for susceptible patients. The potential gains for patients one would anticipate might include further development of their sense of self, of their ability to relate to others and achieve intimacy, and of their role identity – all of which may have a future impact on the distress and disability endured from their disease, not to mention on financial costs involved in supporting such patients.
Our findings suggest that cocaine or amphetamine exert an additive effect. From a statistical viewpoint alone, the linear effect we observed on age at onset is suggestive of a unified process changing at a constant rate (Keppel, 1982). One plausible explanation for these findings is that there exists a mechanism common to cannabis, amphetamine and cocaine exposure that mediates the onset of psychosis in susceptible individuals. As postulated by Paparelli and colleagues (2011), it may be that within striatal circuitry an axis exists at a synaptic level involving dopamine receptors, endogenous cannabinoid transmitters and THC (CB1) receptors, and that cannabis, cocaine and amphetamine are able to modulate the activity of this axis, albeit at different points. An alternative explanation is that the use of an increasing number of substances is simply a marker for another factor that determines age at onset, such as childhood maltreatment or trauma that is associated with both substance misuse and risk for psychosis (Konings et al., 2012). Further studies are required to better elucidate potential mechanisms for the interaction of cannabis, cocaine and amphetamine and age at onset in schizophrenia.
Comparison with previous findings
The profile and prevalence of substance use in our sample are similar to those reported in previous samples of schizophrenia, which have reported high rates of polysubstance use in patients with schizophrenia (Barnes et al., 2006; DeQuardo et al., 1994), although direct comparison is difficult for a number of reasons. First, our study focused specifically on illicit substance use in the 12 months prior to the onset of psychiatric symptoms in patients with an established diagnosis of schizophrenia, compared with other studies that have considered current or lifetime use (Barnes et al., 2006), or patients admitted with first-episode psychosis (Gonzalez-Pinto et al., 2008). Second, many studies categorize patients according to cannabis use versus non-use (Veen et al., 2004), or cannabis use only versus cannabis use plus any other illicit substance (Gonzalez-Pinto et al., 2008).
The predominance of males in our sample (81%) was higher than reported in similar population-based samples (e.g. 66% in a Spanish sample (Gonzalez-Pinto et al., 2008), 72% in an English sample (Barnes et al., 2006) and 73% in a Dutch sample (Veen et al., 2004)). Although several previous authors have postulated that male sex may underpin the association between cannabis and age at onset (Sevy et al., 2010; Wade et al., 2006), this is refuted by recent studies that have found no evidence to substantiate this hypothesis (Gonzalez-Pinto et al., 2008; Large et al., 2011), hence making this an unlikely confounder in our study. Nonetheless, males are more likely to use illicit substances in cohorts of schizophrenia patients (Cantor-Graae et al., 2001; DeQuardo et al., 1994; Sevy et al., 2010), which might explain why we were able to form appropriately powered categories of substance users in this study. Furthermore, Western Australia’s annual rate of amphetamine use is the highest in Australia (at 4.2%), well above the national average of 2.3%, which in turn is greater than rates in other countries (Australian Institute of Health and Welfare, 2008).
Taken together, these above-mentioned factors are likely to explain why previous studies have not demonstrated an effect of amphetamine and/or cocaine in addition to cannabis on age at onset. Gonzalez-Pinto and colleagues (2008) reported that, using multivariate analysis, cannabis use in addition to another illicit substance (amphetamine, cocaine, ecstasy) did not mediate the association of cannabis and age at onset; it is quite possible that their findings were restricted by a small number of amphetamine or cocaine users (numbers not reported). Methodological differences also exist, as they defined cannabis use (and presumably use of other illicit substances) as having taken cannabis at least once in the last month, and at least four times in the last year, and data were obtained from hospitalized patients with first-episode psychosis. Nonetheless, their preliminary analysis lends some support to our own findings, as they demonstrated an increased hazard on age at onset of amphetamine use (HR = 1.94, p = 0.003), and a trend of increasing hazard ratios for age at onset with increasing number of substances consumed (one substance HR = 2.84, two substances HR = 2.62, three substances HR = 3.75). Given the challenges posed by poly-drug use in patients with schizophrenia, further studies with larger samples will be required in order to establish adequately powered subcategories, and better clarify not only the independent effects of individual substances on age at onset in schizophrenia, but the effects of combining different substances.
Limitations and validity
The study is limited in its scope as it addresses the consumption of illicit substances in the 12 months prior to the onset of psychiatric symptoms, and does not consider the age of first use, pattern or frequency of lifetime use, or current use. Whilst enabling us to consider the association of illicit substance exposure and age at onset, we are unable to address the issue of reverse causality, for instance. Another consideration that should be mentioned is the manner in which we defined illicit substance non-use (i.e. no use, or used less frequently than once per month). Whilst studies have varied in their definitions of substance use, we have used a similar approach to other authors (Large et al., 2011; Veen et al., 2004).
It is possible that our results have been confounded by the use of other substances by participants, including alcohol, ecstasy, heroin and LSD. Additional consumption of alcohol has not been associated with a significantly earlier age at onset (Barnes et al., 2006; Gonzalez-Pinto et al., 2008; Large et al., 2011), and hence it is unlikely to have confounded our findings. We attempted to account for the possible confounding by the consumption of additional illicit substances (LSD, ecstasy, heroin), by excluding, in turn, each additional substance, and our results remained unchanged. Nevertheless, we cannot be entirely certain that additional substances or certain combinations thereof do not mediate the effects seen on age at onset in our study, and it is likely that larger samples will be required to elucidate the independent effects of individual substances on age at onset in schizophrenia.
Recall bias is a further limitation of the study, like many similar studies. We relied on retrospective data on substance use (i.e. in the 12 months prior to the onset of psychiatric symptoms) that were obtained by self-report, and this was not corroborated by an informant. Although Barnes and colleagues (2006) make an argument for the reliability and validity of substance use data by self-report, the majority of participants in our study had to recall their substance use many years prior to the DIP-DM interview. However, when we controlled for length of illness our findings remained significant, suggesting that time since onset was not a mediating factor in self-reported substance use.
We anticipate addressing several of the above-mentioned limitations in a subsequent study, by analyzing data available from the SHIP (Survey of High Impact Psychosis) study. In particular, we will have access to data from a significantly larger number of participants (and hence have a larger number of participants in categorized substance-using groups) and will have additional data on the age of first use, pattern or frequency of lifetime use, and current use (i.e. to ascertain whether polysubstance use might have a longer trajectory than single substance use, for instance, and evaluate if there is a differential effect between recent and more distant exposure to drugs). Furthermore, we will have available data on additional factors to include in our analysis, such as socio-demographic status and adverse childhood events, for instance.
Notwithstanding, the present study has a number of strengths that are worthy of mention. First, the study was population-based and recruited patients from admissions to local psychiatric services (both inpatient and outpatient). Second, establishing the diagnosis of schizophrenia was comprehensive, with performance on the DIP-DM scored using the OPCRIT diagnostic algorithm, and video-recorded interviews reviewed independently by two psychiatrists who assigned consensus diagnoses. Finally, we collected data on a range of substances used, together with their frequency of use in the 12 months prior to the onset of psychiatric symptoms, and this allowed us to evaluate not only the association between the exposure and outcome of interest, but to investigate the possible interaction of various illicit substances on age at onset in schizophrenia.
Conclusion
Whilst preliminary, the findings of our study indicate that amphetamine and/or cocaine consumption in addition to cannabis in the 12 months prior to the onset of psychiatric symptoms leads to an earlier age at onset in schizophrenia than that seen for cannabis consumption alone. If replicated they will carry an important public health message to individuals susceptible to psychosis, and raise intriguing questions about how amphetamine and/or cocaine may exert an additive effect on brain biochemistry.
Footnotes
Acknowledgements
The authors would like to thank the patients, family members and other volunteers who participated, and the staff at Graylands Hospital and mental health clinics within the North Metropolitan Health Services, Perth, Australia.
Funding
The study was supported by grants from the National Health and Medical Research Council of Australia (grants 37580400, 37580900).
Declaration of interest
The authors declare that there is no conflict of interest. The authors alone are responsible for the content and writing of the paper.
