Abstract
The efficacy of repetitive transcranial magnetic stimulation (rTMS) in the treatment of depression is disputed. This is partly because, to date, insufficient good-quality studies have been conducted, and a definitive clinical profile for which rTMS is best suited is yet to be determined. However, another equally important reason for questioning the efficacy of rTMS is that the clinical response to its application is thought to be the result of predominantly non-specific and placebo effects. Therefore, in this viewpoint we examine this possibility in detail and propose a meaningful way forward for rTMS research.
Keywords
The problem with rTMS
Repetitive transcranial magnetic stimulation (rTMS) is marketed as an effective therapy for depression, even though evidence for its efficacy remains incomplete and unconvincing. Therefore, it is important to ask how this is possible, given that, to be introduced into the market, every new therapy has to undergo scrutiny from regulatory bodies, such as the Food and Drugs Administration (FDA) in the United States (Malhi et al., 2021d) and the Therapeutic Goods Administration (TGA) in Australia, and also demonstrate efficacy in real-world practice so as to convince clinicians and patients?
Recently, we have pointed out that there is simply insufficient evidence to claim efficacy for rTMS in the treatment of depression – in particular, when pharmacotherapy has not been adequately trialled (Malhi et al., 2021b). And so once again, we ask, given that this is the case, why is it that rTMS is being increasingly regarded as an effective therapy for the treatment of depression? And why is it that some patients and clinicians firmly believe this to be so?
Our principal concern is that the clinical indication for rTMS is grossly out of line with the evidence that has been garnered to support its use, and we believe that this has come about because of a number of powerful psychological factors.
First, there is a dire need to address the enormous burden of depression, and naturally we all want better, faster and more affordable treatments that can be delivered with little or no side effects. This has created a sense of urgency in the field and it may explain why authorities have been less stringent when assessing rTMS research. For the same reason, governing bodies have perhaps been more inclined to look upon rTMS favourably, especially as it is seemingly side effect free and promises equal, if not better, outcomes as compared with alternatives.
Second, the intensity and close personal contact that the delivery of rTMS entails (almost daily sessions lasting considerable time and involving close physical contact) means that there are many powerful non-specific inter-personal psychological factors in play (see Box 1). This is borne out by the fact that studies that have controlled for these placebo effects by using sham therapy have found that sham rTMS, in and of itself, has considerable benefits (Burke et al., 2019; Razza et al., 2018). In other words, the majority of the ‘therapeutic’ response or clinical benefit is a consequence of factors that can be replicated using sham. This means that in practice the therapeutic effect of any magnetically induced stimulation (the ‘specific effect’ of TMS) appears to be minimal at best.
Factors thought to enhance the placebo effect of rTMS.
Treatment cues due to positioning of elaborate TMS device over patient’s head.
Effect of using hands-on procedures to set up and calibrate rTMS device.
Displaying motor-evoked potentials from electromyogram recording.
Lengthy interaction with person administering TMS (physician, nurse, technician).
Attending specialist centre for each treatment (e.g., specialist clinic, hospital).
Awareness of technical innovation of treatment via extensive media coverage.
Source: Adapted from Burke et al. (2019).
TMS: transcranial magnetic stimulation.
Third, the seemingly near absolute innocuousness of rTMS has meant that a careful and critical risk-benefit analysis has not been undertaken. And as a consequence, the efficacy of rTMS has not been evaluated properly. However, given the tremendous need for better treatments for depression, it is perhaps understandable that there may have been some leniencies afforded rTMS in terms of licencing. Nevertheless, some standards must be maintained, and these matters warrant critical scrutiny, and especially when new treatments are likely to draw heavily on the public purse (Medical Services Advisory Committee, 2018). The transition of rTMS into clinical practice has no doubt been facilitated by the safety of rTMS, but its long-term impact on brain function has not been tested and the possibility of any enduring side effects remains unknown. Among the many unknowns concerning rTMS, the key issue, that we wish to explore further in this Viewpoint is that of its clinical efficacy in the treatment of depression; in particular, to what extent is its clinical effect non-specific?
With this in mind, we argue that the supposed benefits of rTMS are largely the result of its placebo effect, and that this can be seen when rTMS is administered as a sham control. In other words, rTMS itself does not have a significant antidepressant effect, and its therapeutic benefit stems largely from placebo.
A semblance of response
Recently, studies that have examined the effects of rTMS in the management of depression, and reviews and meta-analyses of research findings, have been carefully critiqued (Amad et al., 2019a, 2019b; Malhi et al., 2021b). The sum conclusion of these is that rTMS may have an effect in some patients who have failed to respond to several alternative therapies but that this effect is mild at best, and that clinically this patient group remains poorly defined. Hence the positioning of rTMS in the management of depression continues to be a matter of considerable debate, and many clinicians and academics agree that our current state of knowledge does not support the use of rTMS as a standard treatment for depression (Amad et al., 2021; Malhi et al., 2021a).
If rTMS lacks efficacy why do some people seem to benefit?
It is important to note that efficacy is not the same as effectiveness, and that the latter also takes into consideration tolerability. In the treatment of depression, rTMS appears to have few side effects and this contributes to its effectiveness, but as alluded to earlier, its purported efficacy has been called into question. So, why do some clinicians continue to believe rTMS should be positioned favourably in the treatment of depression (Fitzgerald et al., 2021; Malhi et al., 2021c)? And why is this belief persuasive? We believe there are two reasons for this. First, in clinical settings, rTMS appears to ‘work’, but its averred antidepressant properties are largely the result of a significant placebo effect. In other words, there is a discernible non-specific effect that does not arise because of rTMS per se, but more so as a consequence of the context and manner in which rTMS is administered (Burke et al., 2019). Second, the attribution of the placebo-driven response to rTMS is compelling because clinical improvement is a desirable outcome for everyone involved, and for which there is a desperate need. And therefore, we maintain that trust is one of the fundamental factors that drives the clinical response to rTMS.
Trust me I’m a doctor
For patients to engage in any treatment, it is necessary for them to trust the medical profession. Recently, we have seen this tested with respect to the uptake of vaccination. This is because patients have to believe that their doctor treats them with the sole intention of making them feel better, and they have to trust that their doctor knows the effects of any treatment they prescribe. In other words, the patient assumes that the doctor has the requisite knowledge, and that in their professional opinion the treatment they are prescribing is suited to them.
In a similar vein, doctors also have to believe that any treatment they are administering is going to benefit their patient. However, the difference in the investment of trust is that the doctor’s conviction is not based solely on the authority of an individual, or the professional body that they belong to. Instead, the doctor has to trust the evidence that supports the use of a particular treatment and this depends on the quality of the underlying research. In addition, they have to have confidence in the processes that govern the development, clinical testing and licencing of treatments. Put another way, just as society places trust in doctors to do no harm and always have the best interests of their patients in mind, so too, doctors have to trust the science that brings new treatments into practice. However, the development of new therapies is scientifically complex and not all doctors can be expected to know the detailed neuroscience that underpins the mechanisms of a therapy, or indeed be familiar with every aspect of its clinical effects. Therefore, as clinicians, they too need to trust the scientists and researchers in the field.
Thus, trust is an integral part of both the belief system that underpins the prescription of treatments, and the mechanism that drives their clinical effect; and while it is an essential ingredient of clinical response, it is also a major problem for rTMS. This is because the placebo response in rTMS depression trials is large, and sham rTMS produces significant effects (Burke et al., 2019; Razza et al., 2018). Part of the reason for this is that the placebo response is greater when clinical interactions are more intense, prolonged and of a higher frequency (Enck and Zipfel, 2019). Thus, the placebo effect may explain why some depressed patients who receive rTMS believe that they have benefitted. And of course, positive patient reports are likely to encourage doctors, who naturally then feel validated.
The power of suggestion
In the case of rTMS, the psychological forces in play are particularly pronounced, by virtue of its mode of administration and unique mechanism of action, which invariably invokes a degree of ‘neuroenchantment’ (Ali et al., 2014). In conjunction with the pressing need for advances in antidepressant therapy, factors such as these have perhaps exaggerated the belief in the therapeutic potential of rTMS and accelerated its introduction into clinical practice. However, to fully understand how, despite a paucity of evidence to substantiate the efficacy of rTMS in the management of depression, these influences have managed to both create the erroneous impression that it is effective in treating all kinds of depression, and simultaneously promote its clinical uptake earlier in the course of therapy, it is useful to consider the role of non-specific effects.
To what extent is the effect of rTMS real?
To evaluate the effect of rTMS in the management of depression, it is useful to examine the clinical effect more closely. TMS devices, when used properly in clinical trials, induce electric currents in underlying neural tissue, and this neurostimulation of the brain is associated with a clinical response. However, the question remains – what proportion of the clinical response that is experienced by the patient is specifically a consequence of neural stimulation, as opposed to being a placebo effect? Put simply, what fraction of the response can be attributed to the direct effects of rTMS. This is the crucial point in question.
Clearly, the clinical response the patient experiences is genuine, as is its elicitation, but the fact that sham rTMS can produce a very similar clinical response suggests that the component of the overall response that occurs because of placebo and other factors is very significant. It is this ‘non-specific’ effect that is our concern. In Figure 1, we have illustrated the direct impact of rTMS as its specific effect and the remainder of the clinical response, which is the majority, as the result of placebo and other additional non-specific effects. In clinical trials, these non-specific effects drive the overall clinical response and obscure the specific response to rTMS. This is why the effects of sham rTMS are often indistinguishable from real rTMS. The component of the overall response that occurs directly because of neurostimulation is genuine; however, both in practice and in research, this nuance is often lost. Instead, it is presumed that the overall clinical response occurs largely because of neural stimulation that has been instigated by rTMS. This is incorrect, just as it would be incorrect to suggest that the whole clinical response occurs as a consequence of non-specific factors and placebo.
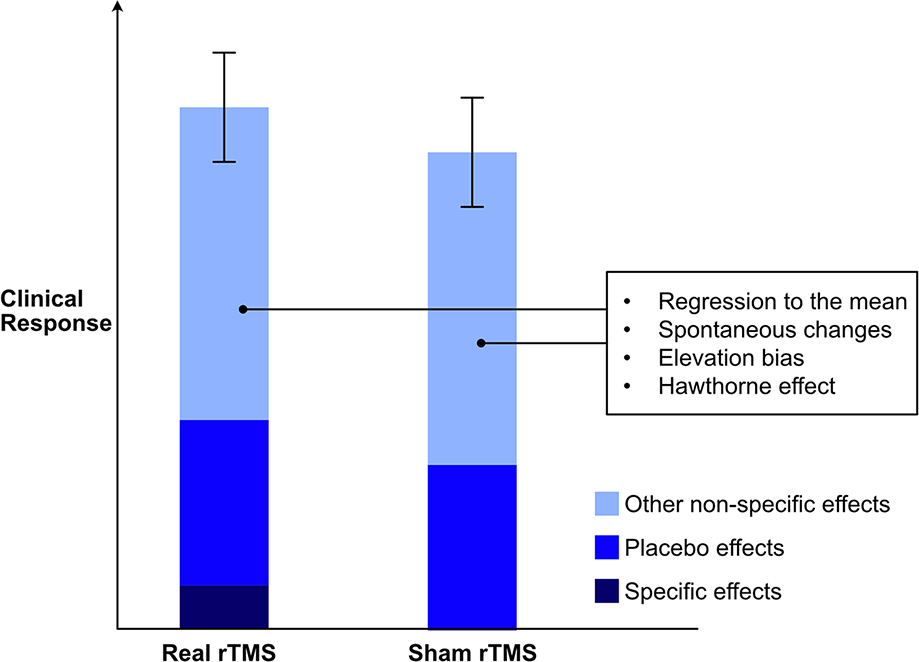
Schematic representation of differential contributions of specific, placebo and other effects to overall clinical response to rTMS.
What’s wrong with placebo?
Arguably, the placebo response is an essential component of treatment response and so there is nothing wrong with having a placebo response per se, unless of course it is so significant that it makes the specific treatment effect redundant. Furthermore, the placebo response can be different depending on the circumstances in which a treatment is administered. Specifically, there is an ‘external context’ that comprises environmental cues and verbal and non-verbal suggestions and an ‘internal context’ that consists of the expectations of the patient, their memories and past treatment history (Burke et al., 2019). These contexts interact and the resulting combined effect varies considerably. Hence, the effects of placebos can differ in terms of size (differential placebo effects) and it is partly why some kinds of treatment produce a greater placebo response than others. For example, intravenous placebo is more effective than oral placebo, and similarly, physical placebo (such as sham surgery) is more effective than non-physical placebo. The placebo response associated with rTMS is thought to occur because of several factors (see Box 1), but in particular, the intense environment in which rTMS is administered, the strong expectations in patients that accompany the therapy, and the physiological response they experience because of direct stimulation of the scalp. These factors make the placebo response larger and hence why the response to rTMS is sometimes described as an ‘enhanced placebo’ (Burke et al., 2019). This kind of differential in placebo effects is a problem for rTMS research because it means that in clinical trials the effect of sham TMS is often the same as that produced by real rTMS.
What needs to be done and why?
Remarkably, there is little rTMS research examining the phenomenon of differential placebo effects. For example, the potential enhanced placebo effects of sham TMS compared with other forms of placebo have not been examined in a prospective study. One of the reasons why researchers may be reluctant is that perhaps they fear undermining the research that has already been conducted. After all, acknowledging that the bulk of the antidepressant effect of rTMS is a consequence of non-specific factors and placebo is likely to detract from current efforts for widespread acceptance of rTMS. However, not doing such research means that the field is resigning itself to working with a clinical effect – the precise composition of which remains unknown. In practice, it is doctors that have to prescribe any treatment and it is they who have to ultimately take responsibility for their patients’ welfare. One could argue that in the context of managing depression there is little or no harm in prescribing rTMS because it is well tolerated and has very few side effects; therefore, the only real disadvantage or risk is that it may not work (as an effective antidepressant). However, this argument is both flawed and morally questionable.
Prescribing a depressed patient a potentially ineffective treatment, and especially one that requires several weeks of intense administration, means that they are being denied the opportunity to receive an alternative potentially effective therapy. In the case of depression, this has two immediate consequences. First, the longer a person remains acutely depressed, the more their brain is subjected to the harmful effects of the illness. Hence why, once a diagnosis of depression is definitively made it is imperative to institute optimal management as soon as possible. Second, if the person has deep-rooted therapeutic expectations in relation to the treatment they are receiving, and eventually it is ineffective, they are prone to becoming more unwell and are at greater risk of suicide. Therefore, there are harms associated with administering rTMS, and especially when it is not clear to what extent the clinical response is directly attributable to neurostimulation. Again, one could argue that these concerns apply to all treatments, and indeed, these considerations are inevitably part and parcel of clinical management. However, in most cases, they are inconsequential, because doctors are generally prescribing treatments that have established efficacy, and a significant component of the clinical effect has been shown to be a consequence of the therapeutic agent. In the case of rTMS, we have argued that this is not so, and that in fact, the clinical situation is likely very different.
This then raises a moral issue. Is it ethical to prescribe a treatment without fully understanding its effects? We have established that there is considerable doubt as to how much of the clinical response observed in practice is accurately attributable to the real neurophysiological effects of neurostimulation, as opposed to being the result of placebo and nonspecific effects. This is simply not known. And so, how to proceed?
Further research
In TMS research, disentangling the effect of placebo from that of active treatment is especially difficult, because administering rTMS inevitably involves active engagement with the patient and frequent in-person contact that generates non-specific effects on patient outcomes (Burke et al., 2019). As outlined previously, these issues are not fully addressed by a sham condition, and thus placebo effects in rTMS studies are consistently strong and possibly increasing (Razza et al., 2018). However, to address the key issues discussed above concerning the rTMS literature, the effect of sham must not be ignored as ‘trivial’ or deemed inevitable due to the nature of the treatment (Burke et al., 2019). Even in instances where a sham procedure may seem impossible, studies can be devised to control for placebo effects. As an example, it was previously assumed that a successful and ethical sham for neurosurgical procedures was unattainable, until that is, researchers using technological ingenuity and elegant design developed a sham procedure to examine the efficacy of gamma knife anterior capsulotomy for the psychosurgical treatment of obsessive-compulsive disorder (Miguel et al., 2019). Thus, similarly, in the field of brain stimulation, improvements must be made in the methodology of studies examining the differential effects of sham and active treatment. And there is a ‘critical need for prospective, placebo vs. placebo studies (e.g. one kind of placebo versus another kind of placebo)’ (Burke et al., 2019) so as to elucidate the differential placebo effects of rTMS.
In this context, when designing TMS studies for the management of depression, it is also important to consider the patient population that is being administered the treatment. In depressed patients, the placebo response decreases with increasing treatment resistance as shown in Figure 2. This means that in the case of rTMS studies, separation of active versus sham is most likely to be found in those patients who have a severe depression and one that has failed to respond to several pharmacotherapeutic agents (Brunoni et al. 2009; Lam et al., 2008). Of note, this means that such studies need to be conducted in homogeneous patient populations, ideally with similar clinical and treatment histories, and further, findings cannot be automatically extrapolated from one group of patients (e.g. those with severe depression having undergone several treatments) to another (e.g. treatment naïve depression). Clearly, these considerations have significant implications for the positioning of rTMS in clinical practice guidelines.
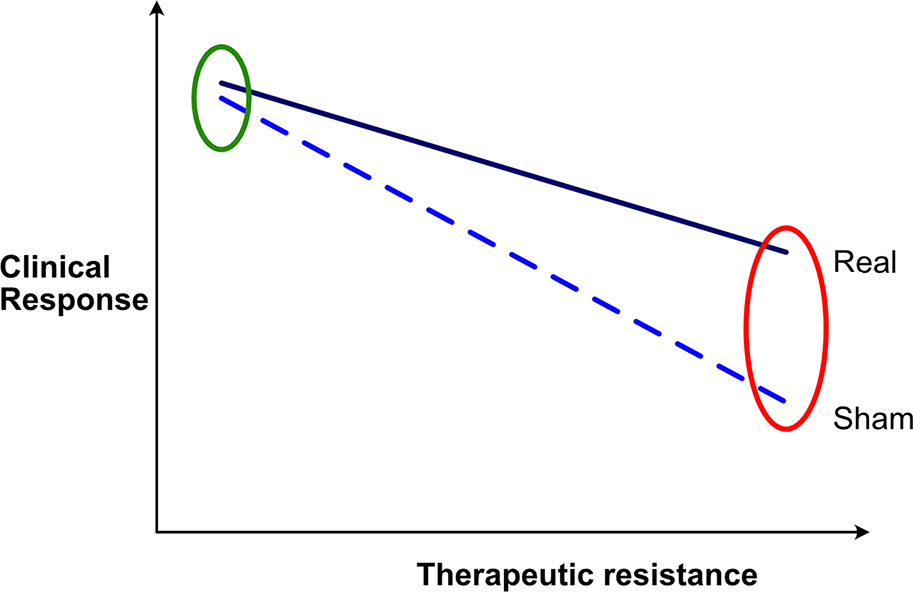
Differences in response to sham and active treatment as resistance to treatment varies.
Conclusion
The use of rTMS for the treatment of depression is based on research that has not fully evaluated the clinical response elicited by neurostimulation using this means. In many studies, the active therapy fails to separate from sham, and this is probably because the active treatment is also largely attributable to placebo and non-specific effects. Thus, the precise nature of the neurophysiological effect and clinical response of rTMS needs to be determined with much greater exactitude. In addition, the response to treatment with sham rTMS should be investigated in detail, to better understand the factors that enhance the placebo response. Clinically, the positioning of rTMS is dependent not only the matter of whether it works, but also for whom is it best suited (i.e. the kind of depression and the particular patient characteristics) and when it should be employed (i.e. as an adjunctive to medications or psychological treatment, as an alternative to Electroconvulsive therapy (ECT)). In other words, there is a significant amount yet to be determined about this new and evolving technology and a concerted effort is needed to advance our knowledge so that, sometime in the not-too-distant future, we can hope to employ this intervention meaningfully.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: G.S.M. has received grant or research support from National Health and Medical Research Council, Australian Rotary Health, NSW Health, American Foundation for Suicide Prevention, Ramsay Research and Teaching Fund, Elsevier, AstraZeneca, Janssen-Cilag, Lundbeck, Otsuka and Servier; and has been a consultant for AstraZeneca, Janssen-Cilag, Lundbeck, Otsuka and Servier. The author E.B. declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
