Abstract
Definitions and main features
Bipolar disorder (BD) is for most people a recurrent, disabling illness. They experience periods of mania or hypomania, depression, and ‘mixed episodes’ or ‘dysphoric mania’ (both manic and depressive symptoms). Bipolar disorder is commonly subdivided into BD I (at least one manic episode) and BD II (hypomania and depression only).
Epidemiology
Bipolar disorder is more common than previously thought, with a lifetime prevalence of up to 1.6%.
Course and prognosis
Most people suffer many episodes (an average of 0.4–0.7 per year), each lasting 3–6 months [1], [2]. Four or more episodes in 12 months is termed ‘rapid cycling’.
Outcomes are poor. In a major US study of 75 patients who had recovered from an episode of mania [3], [4], only 28% remained in remission for the next 4 years. Twothirds were on lithium and one in five an anticonvulsant, but outcomes for them were no better. The mortality was 4%. Another study, Keck et al. [5] followed up 134 patients for 12 months following a manic or mixed episode. Only half the group made a syndromic recovery, and a quarter achieved symptomatic and functional recovery.
The suicide rate (10–19%) [6] is about 15 times that of the general population [7] and 80% of suicides occur during episodes of depression. Other risk factors are expressed intent, previous suicide attempts, hostility, poor social support, delusions and/or hallucinations, history or presence of mixed states, family history of suicide, personal and/or family history of violence, short latency from illness onset, poor medication response or tolerance and significant personal and interpersonal vulnerability (e.g. marital tension, poor self-esteem) [6].
Substance abuse comorbidity [8] is higher than in any other psychiatric condition [9]. The divorce rate is twice that of the general population. The likelihood of career dislocation is increased [10].
Despite the disabling symptoms, few people seek treatment. Kessler et al. [11] reported that although 93% of BD I patients had received some treatment, only 45% had been treated in the previous year. Of that 45%, only half were helped within the healthcare sector.
Method
The APA Practice Guideline Development Process (available from the American Psychiatric Association Office of Research) has been followed in developing these guidelines. The process involved: a comprehensive literature review (Index Medicus/MEDLINE/EMBASE, and reviews and articles known to the authors); drafting provisional guidelines for consultation and review by the expert committee; regular revision of the guidelines; and dissemination and review of drafts. Representatives of patients and carers were included.
Treatment guidelines developed in recent years include:
American Psychiatric Association practice guideline for the treatment of patients with bipolar disorder (Revision) [12] via literature review, clinical consensus and broad consultation.
The expert consensus guideline series medication treatment of bipolar disorder 2000 [13] via a survey of 65 US experts.
Clinical practice guidelines for bipolar disorder, Department of Veterans Affairs [14] via a multidisciplinary group of experts and clinicians.
The World Federation of Societies of Biological Psychiatry (WFSBP) guidelines for the biological treatment of bipolar disorders [15], [16] via expert comment on literature, conference proceedings and national and international guidelines.
In Australia, Therapeutic Guidelines: Psychotropic (5th and previous editions) [17] have served as de facto Australian guidelines, via consensus of a mental health group using evidence-based evaluation of the literature.
General issues in treatment
Bipolar disorder shares clinical features with major depressive disorder but its episodes of hypomania or mania are distinct. Since the latter may merge into psychosis, patients may remain undiagnosed for years or be incorrectly diagnosed as having schizophrenia or personality disorder. Thorough assessment is vital, with diagnostic monitoring when new information emerges, especially about comorbid substance abuse.
Treatment should aim to restore the person to full health and a meaningful life. Prevention of suicide must be a central goal. Integration of a range of health professionals, as well as family and friends, is required.
The varied nature of BD requires a broad range of interventions. While appropriate drug treatment is critical, attention must also be given to psychosocial contributors and consequences. Recent evidence suggests that psychological treatments are most effective in prophylaxis (in conjunction with drugs) and are best begun when the person is relatively well. They appear to be more effective in preventing depressive than manic episodes. The acute management of mania and depression is largely pharmacological.
Whereas some authorities view antimanic properties (with or without antidepressant ones) as sufficient to categorize a pharmacological agent as a ‘mood stabiliser’, others insist on a proven prophylactic action [18]. This is pertinent, since several new drugs, for example the atypical antipsychotics, are regarded by some as mood stabilisers. Arguably, lithium alone fulfils all three criteria [19]. Although lithium is still the most widely used BD drug, a shift to valproate and other new compounds is underway. Features that predict poor response to lithium are: dysphoric mania (mixed episodes); rapid cycling; depression–mania–interval pattern (depressed episodes preceding periods of mania followed by intervals of recovery); multiple episodes; no family history; and comorbid medical illness or substance abuse. Alternatively, lithium may well prevent suicide (an effect unrelated to its prophylactic role [20]) and it may be neuroprotective [21].
Dysfunction of various components of intracellular signalling has been proposed [21]. Although BD is strongly genetic, with heritable factors explaining 70–80% of the variance, no specific genes have been identified. This lack of knowledge limits the development of specifically tailored therapies.
Support groups play an important role in the management of BD. Wulsin et al. [22] found they had a positive effect on the spread of information, the management of lithium and other psychotropics, and the tackling of interpersonal difficulties. In a US survey [23], participation in support groups helped people to communicate with their doctor, follow medical recommendations, adhere to medication, clarify their treatment plan, and manage follow-up. In a retrospective study [24], support groups ‘contributed significantly to illness knowledge and attitude’.
Acute treatment of mania and mixed episodes
Initial assessment
For acutely manic patients, referral to a specialist psychiatric service for in- or out-patient care is necessary because aggression, excessive spending and disinhibited behaviour (e.g. sexual indiscretions) are likely to damage the person's reputation. Insight and judgement are usually impaired early, even in the absence of delusions. Outpatient treatment is preferable, and admission with consent is occasionally possible. However, involuntary hospitalization is frequently required to protect the patient and family from the effects of damage wrought by impaired judgement. The decision to admit is often traumatic for patient and family, all of whom will need support. A useful guiding rule is that manic patients are always worse than they appear. Corroborative information from family, friends or a general practitioner is invaluable in assessment (Fig. 1).
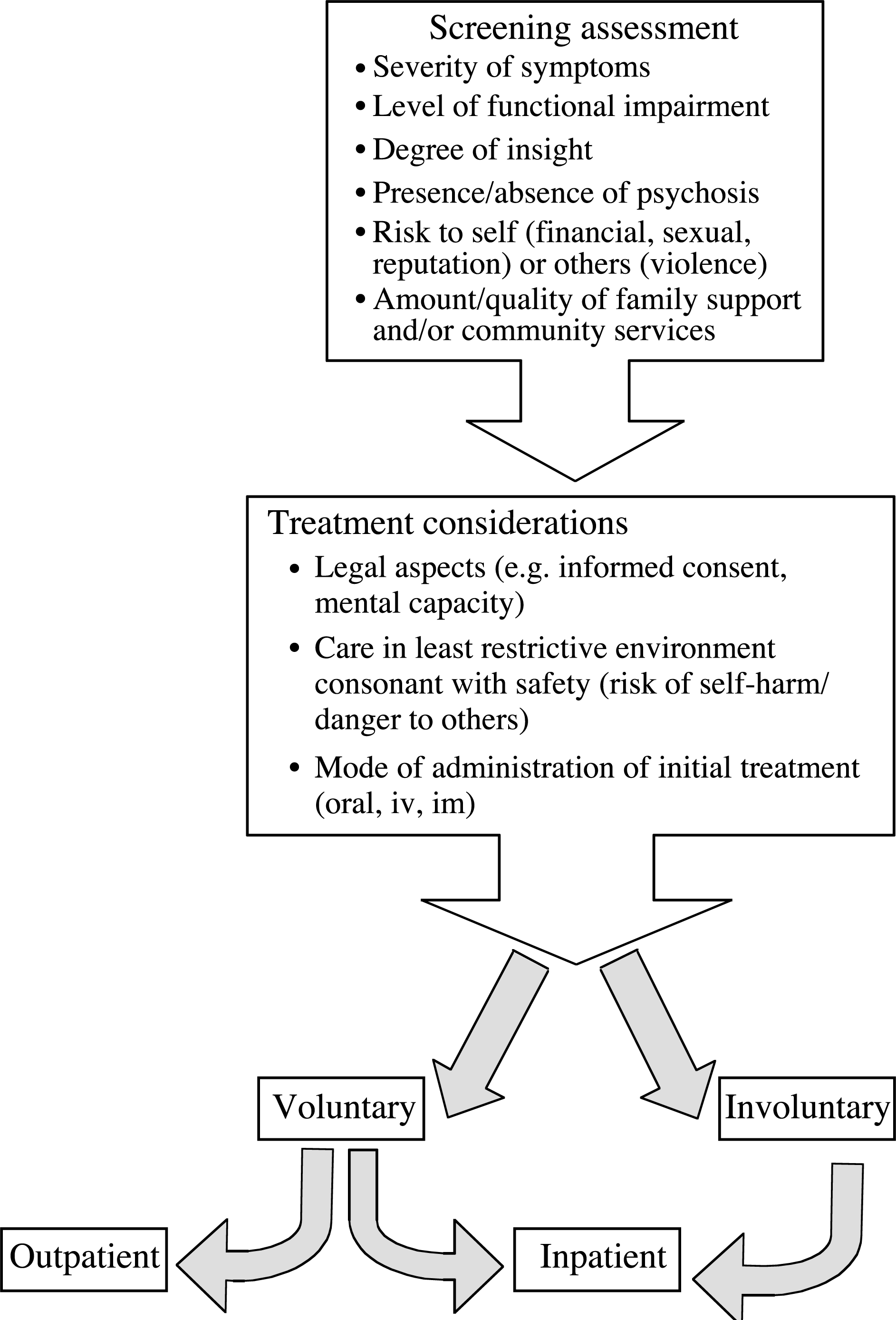
Initial clinical assessment of manic episode. The first presentation is often a non-specific psychotic episode where clear differentiation between mania and schizophrenia is problematic. This figure assumes that diagnosis has been established.
If treatment occurs in an outpatient setting, it is crucial to closely monitor risky behaviour, particularly of a financial nature or when of potential harm to others (e.g. hazardous driving). A financial power-of-attorney may be necessary, particularly if there is a history of excess spending. Since mania produces chaotic behaviour, outpatient attendance is often erratic. Legally enforced community treatment may be an option, using a ‘community treatment order’.
Comprehensive clinical assessment
A full psychiatric history, and mental state and physical examinations, are necessary to confirm the diagnosis, rule out organic causes (including prescription or illicit drugs), identify physical complications (e.g. dehydration) and ascertain level of risk to self or others. Since non-compliance with lithium frequently causes relapse, this should always be considered. Any concurrent antidepressant should be ceased (Table 1).
Comprehensive clinical assessment of manic episode
Pharmacological treatment
Lithium
Despite recent controversy about lithium's efficacy [25], [26], a series of meta-analyses (see following) confirms its value for both acute episodes and for prophylaxis. Indeed, lithium remains the benchmark when trialing new mood stabilisers.
Carbamazepine
With a few exceptions, the quality of the randomized controlled trials (RCTs) of carbamazepine in acute mania has been surprisingly poor. There have been only two double-blind placebo-controlled monotherapy trials [27], [28], involving only 31 patients; carbamazepine was effective in both. In another study [29], responders tended to be dysphoric or rapid-cyclers. Two trials found carbamazepine together with a typical antipsychotic more effective than an antipsychotic alone [30], [31].
In trials comparing the efficacy of carbamazepine and lithium [32], [33], no difference was found, although samples were small and no intention-to-treat analyses were undertaken.
Valproate
Valproate (valproic acid) is usually administered as a sodium salt; other forms are valpromide (the amide) and divalproex sodium (sodium valproate and valproic acid in a 1: 1 molar ratio). The term ‘valproate’ will cover all forms unless otherwise specified.
Three studies have compared valproate with placebo and/or lithium in treating mania. Pope et al. [34] found valproate significantly more effective than placebo. Freeman et al. [35] demonstrated no difference between valproate and lithium except in patients with mixed mania, where valproate was superior. Both studies suffered from small samples. Bowden et al. [36], in a large trial comparing valproate, lithium and placebo, found significantly greater (but equal) improvement in the active drugs compared to placebo. Lithium and valproate were also equally effective for rapid-cylers. Swann et al. [37] later reported that, in this sample, valproate was more effective than lithium for dysphoric mania (consistent with Freeman et al. [35]).
Mueller-Oerlinghausen et al. [38], in a randomised double-blind placebo-controlled comparison of sodium valproate as an adjunct to antipsychotic medication, found the combination superior to the antipsychotic alone.
‘Oral loading’ (initial high oral dose) of valproate may lead to a more rapid antimanic response. In an open random-assignment comparison with blind evaluators, McElroy et al. [39] found 20 mg/kg divalproex was as effective as haloperidol in patients with mania, with most improvement occurring within 3 days. In another study, oral loading (30 mg/kg for 2 days, followed by 20 mg/kg) resulted in a serum concentration of <50 μg/ mL in most patients by day 3 (without more side effects), whereas only a third of non-oral-loading patients achieved this level [40]. Although a prompt response with oral loading is likely, a confirmatory comparison of standard and oral-loading doses in an adequate sample has yet to be done.
The only comparison of valproate and carbamazepine in acute mania (albeit open-label), Vasudev et al. [41], found valproate more effective.
Lamotrigine
No placebo-controlled RCTs using lamotrigine in patients with mania or mixed episodes have been reported. In the one small comparison of lamotrigine and lithium in acute mania [42], no differences emerged.
Gabapentin
Only two RCTs of gabapentin have been conducted [43], [44]. In one [43], gabapentin was less effective than lamotrigine and no more useful than placebo in rapidcycling and unipolar patients. Another negative finding emerged in a large placebo-controlled trial [44]. Gabapentin does not therefore appear to have significant moodstabilizing properties.
Topiramate
No RCTs have been reported.
Clozapine
There have been no RCTs, but an open case series found clozapine to be useful in acute mania and in prophylaxis [e.g. 45].
Olanzapine
A mood-stabilizing property was first noted during trials for schizo-affective disorder. In two RCTs for mania [46], [47], olanzapine was more effective than placebo and from week 1. Two RCTs have compared olanzapine and valproate, with discrepant results. In one [48] comparing olanzapine and valproate in acute mania and mixed states, the former was more effective. The other [49] found no difference between olanzapine and valproate monotherapies. In a further study [50], the addition of olanzapine was more effective than placebo in manic patients who had partially responded to valproate or lithium monotherapy. Olanzapine has been found as effective as the typical antipsychotic haloperidol [51], but has not been compared against lithium.
Risperidone
No placebo-controlled studies of risperidone monotherapy in mania or mixed states have been reported. However, Sachs et al. [52] compared risperidone with haloperidol and placebo – with each as an adjunct to a mood stabiliser (lithium or valproate). Risperidone and a mood stabiliser was more effective than placebo and a mood stabiliser, but not more so than haloperidol and a mood stabiliser. In the only RCT (small) against haloperidol and lithium in mania [53], risperidone and lithium were equally effective.
Electroconvulsive therapy (ECT)
In the single randomised (but small) comparison of ECT and lithium in acute mania [54], ECT was more effective than lithium at weeks 6–8. Those patients who did not respond to unilateral ECT improved with bilateral ECT.
Adverse effects of pharmacological treatment
Lithium
At a supratherapeutic concentration, lithium toxicity may manifest through impaired consciousness (drowsiness to coma), apathy, hyper-reflexia, hypertonia, muscle fasciculations, dysarthria, ataxia, tremor, myoclonus, choreoathetoid movements, seizures, spasticity, cardiac dysrhythmia, acute renal failure and (rarely) death. Some symptoms (e.g. impaired consciousness) may persist beyond the elevated serum levels. A prolonged toxic level may lead to irreversible brain damage.
Carbamazepine
Less common but serious are:
Valproate
Rates of adverse effects in BD and epilepsy patients are similar. The prevalence of polycystic ovary syndrome is controversial [56]. Common adverse effects are concentration-dependent tremor, appetite stimulation with substantial weight gain, hair thinning, ankle swelling and cognitive impairment (less frequent than with carbamazepine).
Less common effects are thrombocytopaenia, hepatotoxicity (a fatality rate of 1 in 10 000 occurs in epileptic patients, particularly those aged under 3 years and taking more than one anticonvulsant), metabolic abnormalities (hyperammonaemia and hyperglycinaemia), encephalopathy and stupor (rare; occurs particularly during combination anticonvulsant therapy).
Lamotrigine
The major adverse effect in both epilepsy and BD patients is rash, particularly that related to the potentially fatal Stevens–Johnson syndrome. The risk is minimized by slow dose titration and by a lower dose if prescribed together with valproate. Other adverse effects are dizziness, insomnia, sedation, ataxia, headache, nausea and vomiting.
Therapeutic drug monitoring
Lithium
Given lithium's long half-life, steady state levels occur only after about 6 days. As the standardized concentration is determined for divided doses, a 10–25% increased level is likely with a change to once daily dosing (usually at night) [21].
Serum concentrations necessary for prophylaxis of BD are less than those required for acute treatment of mania. Once the dose is stabilized and the acute episode resolved, the frequency of monitoring depends on clinical stability but should be at least every 6 months [61].
This divergence of view reflects incomplete research. Only six studies have randomly assigned patients to either dose or serum concentration groups (see Gelenberg et al. [63]). In a study of acute mania, Stokes et al. [64] assigned patients to either placebo, high dose (0.72 mEq/kg/day), medium dose (0.5 mEq/kg/day) or low dose (0.24 mEq/kg/day) with serum levels of 0.62–2.00 mmol/L, 0.29–1.62 mmol/L, and 0.20–1.05 mmol/L, respectively. High and medium, but not low, doses were more effective than placebo.
Of the five studies of maintenance lithium, only two have found a difference between serum concentrations. Waters et al. [65] reported more relapses at low (0.3–0.8 mmol/L) than at high concentrations (0.8–1.4 mmol/L). Gelenberg et al. [66] randomly allocated BD patients to low (0.4–0.6 mmol/L) or standard (0.8–1.0 mmol/L) concentrations, and the low level group had fewer adverse effects but more episodes. Further analyses of these data have been reported, demonstrating more subsyndromal symptoms [67] and poorer psychosocial functioning [68] in low level patients. A criticism of the study, that some low level patients had a rapid dose reduction, suggests a relapse due to discontinuation.
By contrast, Jerram and McDonald [69] found no differences in recurrence between 0.49, 0.5 and 0.69 and 0.70 mmol/L. Neither did Coppen et al. [70] at levels of 0.8–1.2 and 0.45–0.79 mmol/L, or Vestergaard et al. [71] at levels of 0.8–1.0 or 0.5–0.8 mmol/L.
Other causes of increased concentrations are any cause of reduced glomerular filtration, and fluid and/or sodium depletion (the latter including vomiting, diarrhoea and excessive sweating).
Valproate
The ‘therapeutic’ range in epilepsy is 50–100 mg/mL (350–700 mmol/L), although the clinical value of this is controversial [76], [77]. Bowden et al. [78] have examined the valproate concentration and response in mania [36] and found that patients with concentrations of < 45 mg/ mL (315 mmol/L) were more likely than those with levels of < 45 mg/mL (315 mmol/L) to show improvement. There was no evidence of any more benefit from levels < 45 mg/mL (315 mmol/L), though toxicity was likely at < 125 mg/mL (885 mmol/L). There have been no studies of randomly allocated doses or concentration ranges for acute treatment of mania or prophylaxis.
Carbamazepine
No therapeutic serum concentration has been established in BD, although CSF levels of −10,11-epoxide metabolite did correlate with antidepressant efficacy in unipolar and bipolar disorders [79]. In an RCT, Simhandl et al. [80] found no difference in BD recurrence rates between low (15–25 mmol/L) and high (28–40 mmol/L) levels. As for valproate, concentration ranges established for epilepsy (6–12 mg/L, 20–50 mmol/L) are used as a guideline for BD patients [61].
Lamotrigine
There is no demonstrated utility in measuring serum concentrations of lamotrigine.
Meta-analyses
There are two published meta-analyses of the acute drug treatment of mania [81], [82], but no Cochrane reviews have been reported.
Poolsup et al. [82] identified 12 RCTs of lithium (658 patients) lasting 3–4 weeks. The response rate compared to placebo was 1.95 (95% CI = 1.17–3.23). The mean number needed to treat (NNT) was five (95% CI = 3–20). Lithium was more likely to achieve remission than chlorpromazine (rate ratio = 1.96, 95% CI = 1.02–3.77, with a mean NNT of four (95% CI = 3–9]), but was no more effective ‘globally’ than haloperidol. Carbamazepine and valproate were not more effective than lithium, with ratios of 1.01 (95% CI = 0.54–1.88) and 1.22 (95% CI = 0.91–1.64), respectively.
Emilien et al. [81] compared lithium, valproate and carbamazepine. Although valproate was more effective than placebo (OR = 0.16; 95% CI = 0.04–0.60; p ≤ 0.006), the three drugs did not differ in efficacy.
Acute treatment of bipolar depression
Initial clinical assessment
This is similar to the acute management of mania (Fig. 2).
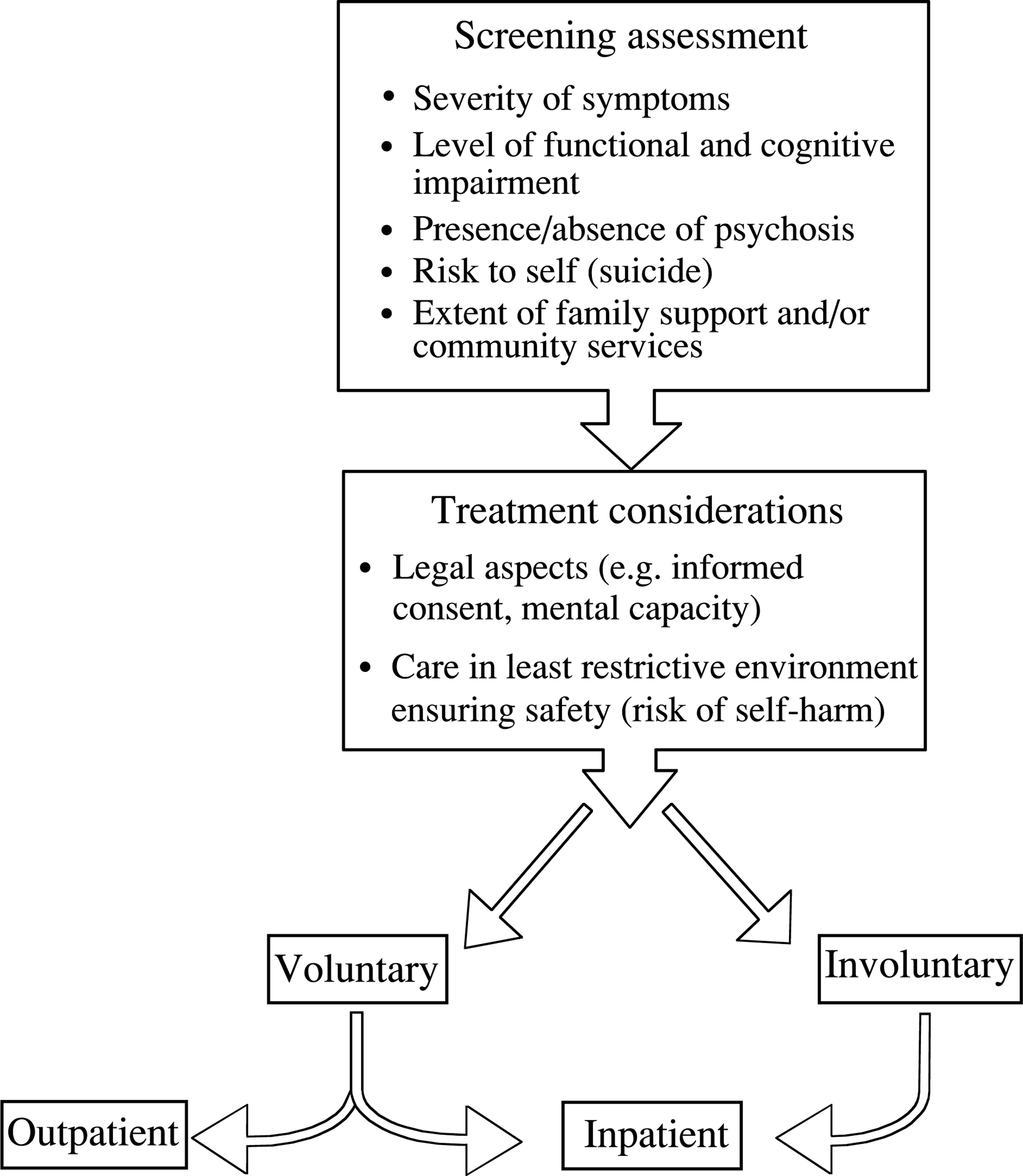
Initial clinical assessment of bipolar depressive episode. Differentiation between bipolar and unipolar depressive episodes is often difficult at first presentation. This figure assumes that diagnosis has been established.
Comprehensive clinical assessment
A full psychiatric history, and mental state and physical examinations, are necessary to confirm the diagnosis, exclude underlying organicity (e.g. hypothyroidism), identify physical complications and ascertain the risk of self-harm. Since non-compliance is a frequent cause of relapse, it should always be suspected (Table 2).
Comprehensive clinical assessment of bipolar depressive episode
Pharmacological treatment
Lithium
In the nine RCTs of lithium, eight used a cross-over design [83–91]. Lithium was superior to placebo, with up to 80% of patients achieving at least a partial response [92]. However, abrupt cessation of lithium before the double-blind phase probably increased the likelihood of early relapse into mania or depression, and may have contributed to lithium's advantage [93]. When stringent criteria are applied to evaluate response [92], only a modest antidepressant effect emerges. This contrasts with the efficacy of lithium in augmenting antidepressants, for which there is robust evidence in both unipolar and bipolar depressed patients [93], [94].
An additional limitation of lithium is the 6–8 weeks delay in antidepressant effect [95]. This is much slower than its antimanic effect, which occurs within 6–10 days [92].
Carbamazepine
With no specific placebo-controlled study of carbamazepine in acute bipolar depression, we have to look at trials with mixed groups of bipolar and unipolar depressed patients [96], [97]. These suggested an antidepressant effect, which is significant given that most open and controlled studies have examined it in depressed patients who have not responded to lithium [98].
In one open study, carbamazepine led to remission in two-thirds of patients [99]. However, less than 100 patients have been studied, and carbamazepine's antidepressant effect is modest [97], [100], [101].
Valproate
There have been no controlled studies of valproate for bipolar depression, but of interest is a double-blind trial [102] which found that adding a second mood stabiliser (lithium or valproate) was as effective, though less welltolerated, than adding paroxetine.
Combining the results of several open studies, McElroy and Keck [103] found that only a third of bipolar depressed patients responded to valproate, but many other treatments had been used previously without success. A study of medication-naive bipolar II depressed patients [104] achieved a much better response (78%), suggesting that valproate may exert an antidepressant effect. This was also shown indirectly in the maintenance therapy study of Bowden et al. [105], in which combining valproate with an SSRI promoted adherence to the medications.
Lamotrigine
An RCT of 195 bipolar depressed patients found that lamotrigine 200 mg daily was superior to placebo [106]. It has also been found to be more effective than gabapentin [43]. Lamotrigine is therefore emerging as an effective agent with a distinct therapeutic profile, in that it works for depression but not for mania [107].
Antidepressants
Because often no distinction was drawn between bipolar depression and major depressive disorder, antidepressants have been ‘borrowed’ to treat bipolar depression. This is akin to the overspill of anticonvulsants from antiepileptic use. While antidepressants are clearly effective for major depression, the evidence is less convincing for bipolar disorder.
Antipsychotics
Antipsychotics are known to have independent antidepressant properties [122]. Their use to treat psychotic bipolar depression in combination with antidepressants is established. In a double-blind study of psychotic depression that included nine bipolar patients, the combination proved superior to either drug alone [123].
In a case series, olanzapine combined with an antidepressant was found to be useful [124]. Recently, a RCT has found olanzapine monotherapy, and combination with fluoxetine, to be more effective than placebo, with the combined therapy apparently being of greater value [125]. The long-term use of olanzapine is limited by sedation and weight gain [126–129].
Electroconvulsive therapy (ECT)
In a review of nine studies (700++ patients) on ECT's efficacy for depression [92], seven compared it with antidepressants, and five of these found ECT more effective. When unilateral and bilateral ECT in unipolar and bipolar depression are compared, the outcomes are similar, However, bipolar patients responded more rapidly, regardless of electrode placement and stimulus dose [130]. ECT is recommended for bipolar depression after an antidepressant trial has failed [131], [132].
Meta-analyses
No meta-analyses are available.
Prophylaxis
Most patients who experience mania will have further episodes, as well as depression [2]. The frequency is 0.37–0.66 episodes a year in those with established illness [2]. Selection of patients for long-term treatment is difficult. Using actuarial techniques, Grof and Angst [133] concluded that at least two episodes of mania or depression within 2 years was the best criterion, since this correctly classified 61% of patients. The National Institute of Mental Health (NIMH) Consensus Development Panel [132] recommends that having a single manic episode, or both hypomanic and depressed episodes, are grounds for prophylaxis, but factors such as suicide attempts in the past, psychotic symptoms and functional incapacity associated with episodes need to be considered. Goodwin and Jamison [6] recommended maintenance treatment after a second episode, regardless of frequency.
Assessment
As non-compliance is the most common factor in relapse, the clinician attempts to improve compliance by strategies such as educating patients and families about the disorder and its treatment as well as about sideeffects. Trying to minimize the latter is obviously a crucial task. Patients fear the potentially abrupt loss of control and its embarrassing consequences. They may resist accepting the diagnosis and need for treatment despite experiencing several episodes. Active BD support groups are widespread and contribute usefully to a treatment program. Written material about BD and its treatment (provided by health authorities and NGOs such as SANE) is helpful to enhance patient knowledge.
An under-acknowledged aspect of long-term care of BD is its continuity, relevant to both patient and clinician. Contact with the same clinician enhances early identification of recurrence and facilitates joint awareness of the continuing impact of the illness. Unfortunately, clinicians move about frequently (particularly in public systems). Bipolar disorder is also linked to a peripatetic lifestyle in some patients.
Pharmacological treatments
Lithium
Lithium remains the mainstay of prophylaxis [62]. Many placebo-controlled trials [6] demonstrate that it reduces both frequency and severity of manic and depressive episodes (lesser recurrence in 50% and no relapse at all in 20% [134]).
Unfortunately, non-compliance is common (18–53%) [6]. Patients cite four reasons: dislike of medication controlling mood; resistance to the idea of having a chronic illness; feeling depressed; and experience of side-effects, particularly lethargy, lack of coordination and ‘dulling’ of the senses. In the same study, clinicians list these reasons: feeling well and seeing no need to continue medication; missing the ‘highs’ of hypomania; and being bothered by the idea of having a chronic illness [135].
One consequence of non-compliance is that rapid discontinuation of lithium leads to a high rate of relapse, greater than the ‘natural’ pattern [93]; 50% of patients relapse within 5 months (mostly with mania).
Carbamazepine
The single placebo-controlled prophylactic study of carbamazepine [136], with 22 patients over 1 year, failed to demonstrate its superiority over placebo. Five randomised double-blind comparative studies against lithium have been reported [137–141], most of which found no difference between the two treatments. In a study by Denicoff et al. [141] 52 outpatients with BD were randomised in a double-blind fashion to 1 year of lithium or carbamazepine with a crossover in the second year and a combination in the third year. Lithium was more effective than carbamazepine in preventing mania but not depression, and the combination was better than monotherapy. Greil et al. [142], in an open randomised prospective study over 2.5 years, found lithium superior to carbamazepine in bipolar I but not bipolar II patients. No head-to-head prophylactic studies comparing lithium and carbamazepine have been done in rapid-cycling patients.
Valproate
In the only double-blind trial [105], 372 patients recently recovered from a manic episode were given valproate, lithium or placebo for 1 year. There was no difference between either anticolvulsant and placebo in time to first relapse, possibly due to the inclusion of milder cases. The largest open randomised study [143] compared valpromide and lithium in bipolar and recurrent unipolar patients over 18 months; patients experienced a reduction in depressed and manic episodes with both drugs. No details are offered in the Bowden et al. [105] maintenance trial for rapid-cycling illness; open studies [144] indicate prophylactic antimanic and antimixed, but not antidepressant, activity.
Lamotrigine
Recent trials by Bowden et al. [145] and Calabrese et al. [146] report that lamotrigine is more effective in preventing bipolar depressed episodes than lithium or placebo, but appears to be less effective than lithium in preventing manic relapse. Frye et al. [43] undertook a placebo-controlled randomised crossover trial of lamotrigine and gabapentin in patients, of whom most had refractory bipolar (mainly rapid-cycling) disorder. Lamotrigine was globally more effective than both gabapentin and placebo, but findings were not given for bipolar and unipolar groups.
Combination drug therapies
In practice, combinations of mood stabilisers are used frequently, particularly in prophylaxis. There is now growing, albeit still minimal, evidence from RCTs that combining lithium and carbamazepine and/or valproate enhances efficacy. Kramlinger and Post have reported, in both acute mania [147] and depression [100], that a substantial proportion of those who do not respond to carbamazepine will respond to the addition of lithium. Denicoff et al. [141] found a combination of lithium and carbamazepine more effective in prophylaxis than either alone. They also compared the effect of valproate plus lithium in 24 patients who had completed a 3-year trial comparing lithium, carbamazepine and their combination, and found that a third had a moderate or marked response [148].
Suicide prevention
Lithium may reduce the suicide rate, an effect seemingly unrelated to prophylaxis [20]. Discontinuing lithium can elevate the risk. Baldessarini et al. [149] reported a 6.5-fold reduction in suicide risk with treatment, and a 20-fold increase in the year after discontinuation.
Psychological treatments
Although drugs remain the primary treatment, associated psychological treatments are valuable [150]. The limitations of medication in alleviating symptoms and functional impairment highlight the need for psychosocial interventions which aim to reduce symptoms, prevent relapse and recurrence, restore social and psychological functioning and support patient and family. Specific goals include a therapeutic alliance, adherence to medication, regular cycles of activity and sleep, identification of prodromes, improved symptoms and coping, and amelioration of varied psychosocial effects.
Psychological adjustment covers stigma, fear of recurrence, interpersonal difficulties, marital, family and parenting issues, educational or occupational disruption and the negative consequences of acute episodes.
A range of treatments help, but the style, intensity and focus vary for each patient. The psychological treatments presented hereafter may be applied in combination in clinical practice.
Psychoanalysis
The use of psychoanalytically orientated psychotherapy to reduce relapse and improve interepisodic functioning has been described [151], but no controlled trials have been done.
Psycho-education
This offers patient and family a conceptual and practical approach to the illness and its treatment, and increases satisfaction with treatment and adherence [152–154]. Brief psychoeducation can enhance adherence and identify early warning signs [155], [156]. In an RCT, Perry et al. [156] found that teaching patients to recognize early symptoms of mania and to seek treatment increased the time to first relapse and improved social functioning and employment.
Cognitive therapy (CT)
The roles of cognitive vulnerabilities and stressful life events in unipolar depression have been well researched [157], [158]. The little work on BD suggests that negative coping style combined with stressful life events can likewise lead to episodes of depression and mania [159], [160]. Furthermore, patients tend to have more dysfunctional attitudes (even when euthymic) than the general population [157], [159]. Their self-esteem is more unstable; they have a greater need for social approval; they are perfectionistic and show poorer problemsolving skills [160].
Cognitive therapy aims to identify and manage stress, prodromes and symptoms, and to prevent relapse or recurrence through monitoring and challenging negative assumptions and thoughts [161]. Clinical advantages include brevity, a coherent theoretical framework and rationale, and enhancement of the sense of helping oneself [160].
Adaptation of cognitive behaviour therapy (CBT) to the particular needs of patients with BD has been described in both case reports [150],[162–164] and open trials [161],[164–167]. Controlled trials of individual CT conducted by Cochrane [155], Perry et al. [156], Zaretsky et al. [168], Lam et al. [169], [170] and Scott et al. [158], and of group CT by Hirschfeld et al. [171] show varying effectiveness. The principal targets of therapy have been adherence [155], recognition of prodromes [156] and management of symptoms [160]. Most studies show improved mood, coping and adherence and reduced admissions. Gains are greater for depressive than for manic symptoms.
Schema-focused CT has been recommended [172], [173] for delaying the onset of a new episode, reducing the duration and severity [172], and promoting adaptation to the illness [173]. Whereas CT seeks to alter distorted beliefs, schema-focused CT [174], [175] uses emotive, interpersonal, cognitive and behavioural interventions. Schemas are changed most effectively at times of high arousal. Use in personality disorders [176], [177] and BD [172] is encouraging.
Interpersonal and social rhythm therapy (IPSRT)
This therapy [178], [179] proposes that stressful life events influence the course of BD by disrupting daily routines, social patterns and sleep habits. Patients are guided to regulate their ‘social rhythms’ when stressed and to address interpersonal problems linked to the onset and persistence of bipolar episodes. Preliminary results of evaluation at the University of Pittsburgh show that after a year, patients treated with IPSRT are better than those in ‘intensive clinical management’.
Group therapy (GT)
This has been successfully applied as an adjunct to medication. The emphasis is on adherence and ‘here and now’ interpersonal issues [22], [180], [181], lithium monitoring and problem-solving [182]. In Bauer and McBride [183], psychoeducation and realistic goal setting were effective. A recent RCT by Colom et al. [184] found that group psychoeducation prevented recurrence of episodes in bipolar I and II patients.
Family-focused treatment (FFT)
This covers such aspects as communication, problem solving skills [185] and psychoeducation, which deals with multiple family stresses leading to high levels of expressed emotion [186].
In one random allocation controlled study comparing brief FFT (with medication) to standard hospital care [187], the family intervention yielded enduring benefits. A study of marital therapy in BD patients [188] found superior adherence and general functioning compared to a medication-only comparison group.
There have been two RCTs on patients who had experienced an acute bipolar episode [189], [190]. The comparison group in the first study received two sessions of family education followed by crisis management as required, and in the second, individual therapy concentrating on symptom management and problemsolving. Miklowitz et al. [189] found that patients receiving FFT had fewer depressive (but not manic) symptoms, especially in high EE families. Goldstein et al. [190] found positive effects only after 2 years.
Famil-focused treatment can improve quality of life and decrease the relapse rate in dysfunctional families [191]. Standard treatment (medication plus clinical management), standard treatment plus family therapy, and standard treatment plus multifamily group therapy have been compared [192]. The two family treatments proved superior, but in dysfunctional families only.
In summary, family or couple therapy, together with medication, is effective for patients recovering from an acute episode. Improving the family environment may protect the patient from relapse or residual symptoms.
The role of psychological treatments
Psychological treatments combined with medication yield the most effective and enduring outcome. Psychoeducation with a focus on adherence improves compliance and thus reduces relapse rates. Cognitive therapy alleviates symptoms (particularly depression) and improves social and psychological functioning. Initial data suggest that IPSRT improves functioning between episodes of illness. Family- or couple-focused psychoeducation improves the environmental milieu, decreases symptoms and prolongs the treatment benefit.
Meta-analyses
There are no meta-analyses of psychological treatments.
Of two Cochrane meta-analyses of the prophylactic value of lithium, the first [193] examined lithium as a maintenance treatment. Using relapse rate as the principal outcome measure, three studies included only BD patients, or had separately randomised BD groups [36], [194], [195] (212 patients). Lithium was more effective than placebo in preventing relapse (OR 0.28, 95% CI ++ 0.09–0.93); the NNT ranged from 4 (95% CI = 3–6) [192] to 14 (95% CI ≥ 4) [36]. When manic and depressive relapses were examined separately, results were similar but not significant.
In the other analysis [196] (of the Bowden et al. study [106]) lithium and valproate were not more effective than placebo in preventing relapse.
A meta-analysis by Dardennes et al. [197] of the role of carbamazepine and lithium in preventing relapse concluded that the efficacy of carbamazepine was still an open question.
Davis et al. [198] focused on the random-assignment, double-blind, placebo-controlled and case-control matched-subject studies of lithium in the prevention of recurrence (n = 514). They found that the drug reduced the rate by half (82% with placebo, 32% with lithium; χ2 = 126, p = 10 (–29), OR = 0.12, 95% CI = 0.09–0.18). Carbamazepine was only a little less effective than lithium (60% with lithium and 55% with carbamazepine).
Insufficient studies precluded determination of lithium's antisuicidal effect in both combined mood disorder and BD samples.
Treatment guidelines (by phase of illness)
As there are no specific treatment studies for BD II, these guidelines refer to BD generically (studies have either described a BD I sample or not differentiated between subgroups).
Acute treatment of mania and mixed episodes
Treatment of manic episodes
There are two components. The first is use of a mood stabiliser (lithium, valproate, carbamazepine or olanzapine) for the elevated mood. The second is concurrent use of an antipsychotic or benzodiazepine (or their combination) to calm or sedate until the mood stabiliser takes effect (approximately 1 week). If olanzapine is used as a mood stabiliser, no other antipsychotic is required. For doses of adjunctive antipsychotics and benzodiazepines, refer to Therapeutic Guidelines: Psychotropic [17] (Fig. 3)
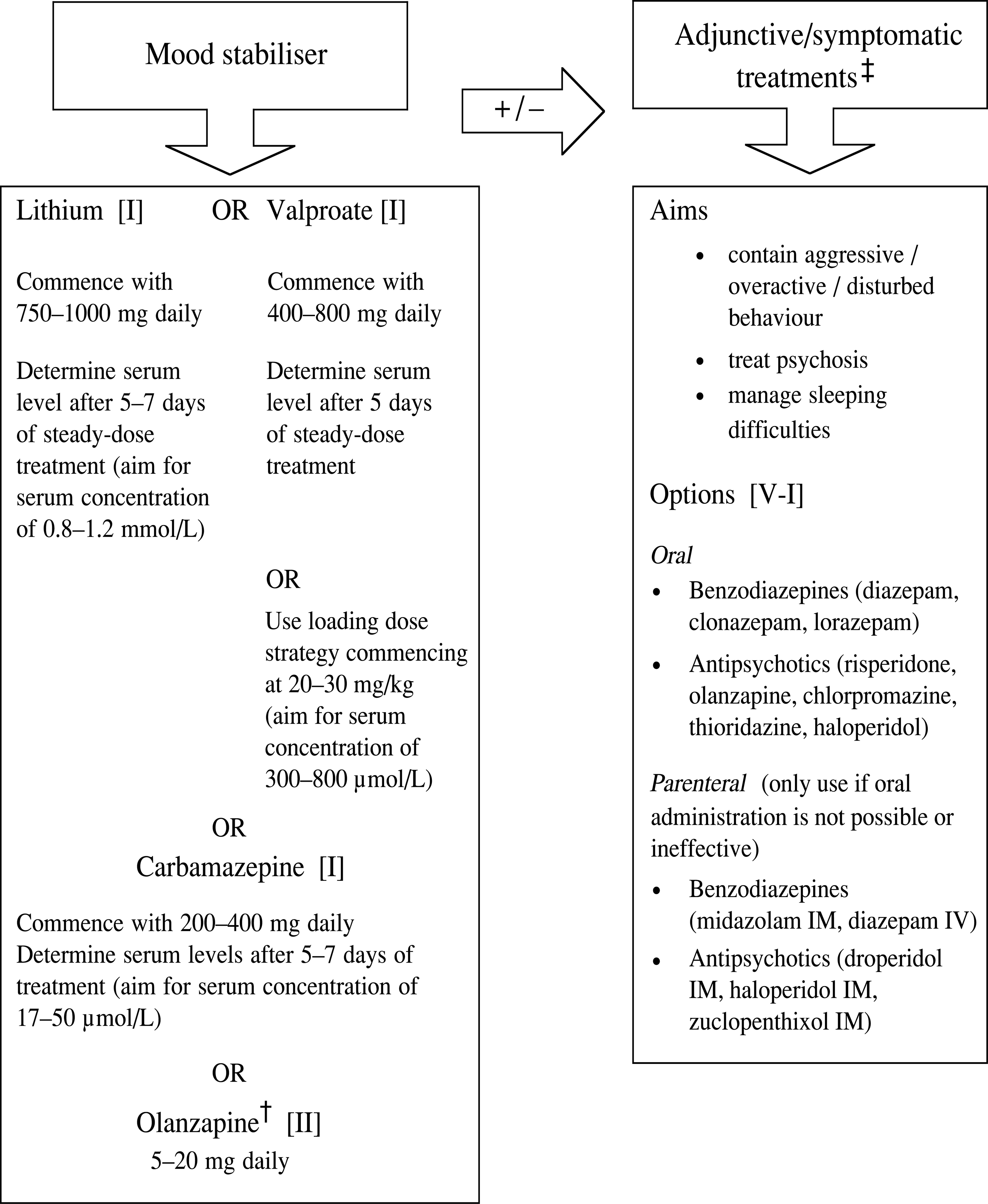
Treatment of manic episode. †If using olanzapine as a mood stabiliser, avoid adjunctive treatment with a antipsychotic. ‡Adjunctive treatments may be initiated when the patient is being assessed or their physical state corrected before or concurrently with the introduction of a mood stabiliser. May not be necessary if the manic episode can be contained by mood stabiliser alone.
For lithium and valproate, the therapeutic range to treat acute mania is well established (see section headed ‘Therapeutic drug monitoring’ earlier). For carbamazepine, the range for epilepsy is a guide, but dose is determined by monitoring clinical response.
Treatment of mixed episodes
The best evidence concerns valproate [37], though it is based on one post-hoc analysis [36]. The data for carbamazepine and lithium are limited, although the latter may be used if response to anticonvulsants is poor. Olanzapine is effective in samples of manic and mixed patients (Table 3).
Treatment of mixed episode
Failure to respond
When to change treatment depends on urgency and degree of response. Options include increasing the dose and/or blood levels of the mood stabiliser, switching mood stabilisers, combining mood stabilisers or adding an adjunctive antipsychotic like risperidone or haloperidol. The best evidence is for augmentation of the mood stabiliser with risperidone, olanzapine or haloperidol. Combining mood stabilisers is the next best option. ECT is considered if all other strategies fail (Fig. 4).
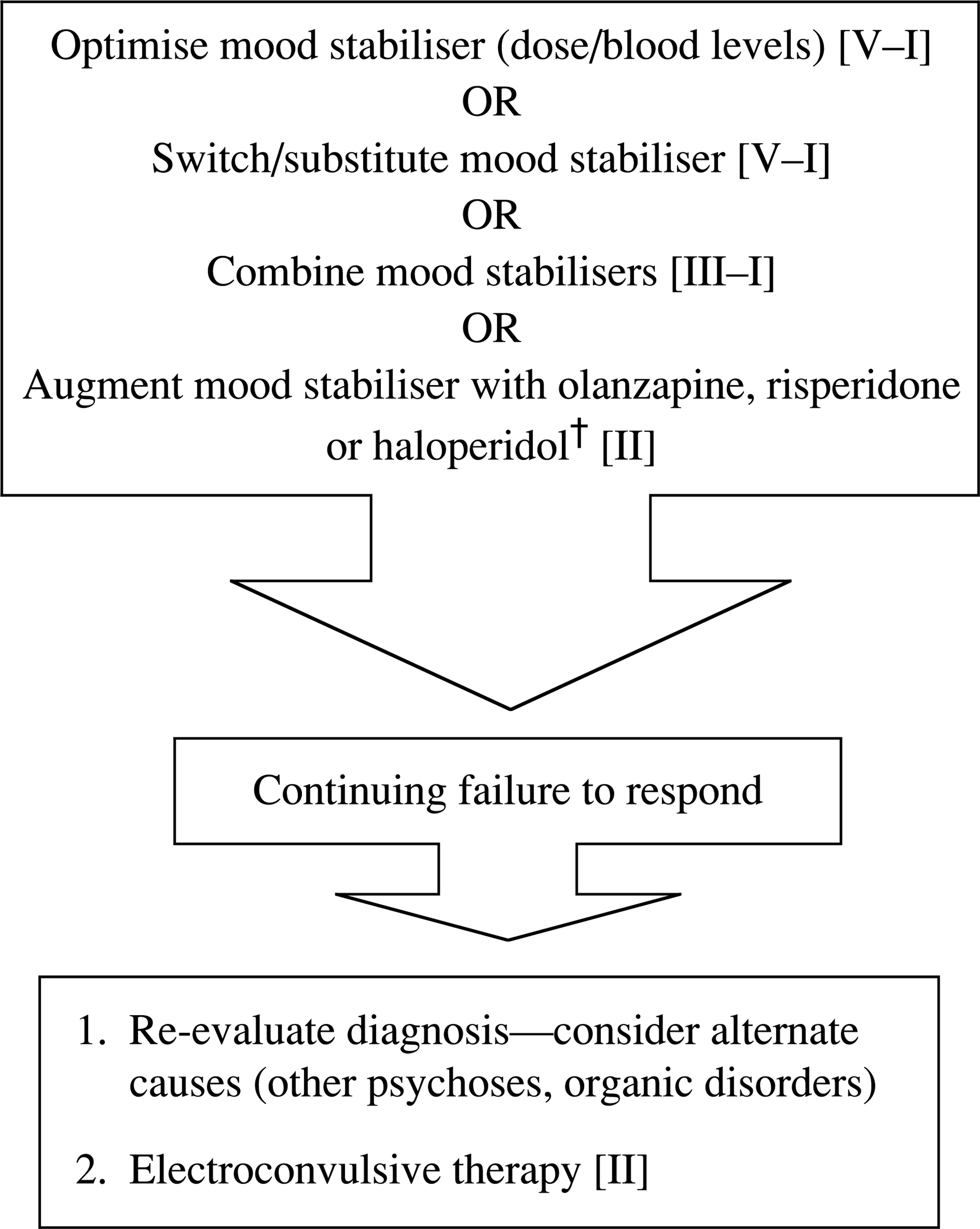
Failure to respond to treatment of mania and mixed episodes. †Do not use other adjunctive antipsychotics with this combination.
Continuing treatment
Following remission of an initial episode of mania, the mood stabiliser is continued for at least 6 months. Benzodiazepines or antipsychotics are withdrawn once the episode has resolved. For those with a solid BD history, recommended guidelines are based on consensus and clinical wisdom, which consider illness frequency, severity and disability (Table 4).
Criteria for continuing treatment
Acute treatment of bipolar depression
In established BD, two clinical scenarios require consideration: bipolar depression that arises in the absence of medication (i.e. de novo depression) or when treatment is in place (i.e. breakthrough depression).
De novo depression
Treatment is determined by the nature and severity of the illness. The first step is to institute antidepressant treatment (Table 5)
De novo depressive episode†
The mood stabiliser's use is optimized by monitoring plasma levels periodically [129]. Lithium is generally preferred [13], since it is more effective than placebo for the depression and prevents both depressive and manic recurrences. However, evidence for an antidepressant effect is less impressive than that for an antimanic one and onset of action is slow. Lamotrigine is an effective antidepressant but its antimanic effect is unproven. Lithium or lamotrigine are first-line options. Valproate and carbamazepine are less effective in bipolar depression [13], [37], [199], although the former warrants consideration in rapid cycling [200].
Breakthrough depression
If a patient is maintained on a mood stabiliser, the first step is to optimize its effect by ensuring compliance and checking serum concentrations [120]. If this fails, one can add an antidepressant or a second mood stabiliser (Fig. 5)
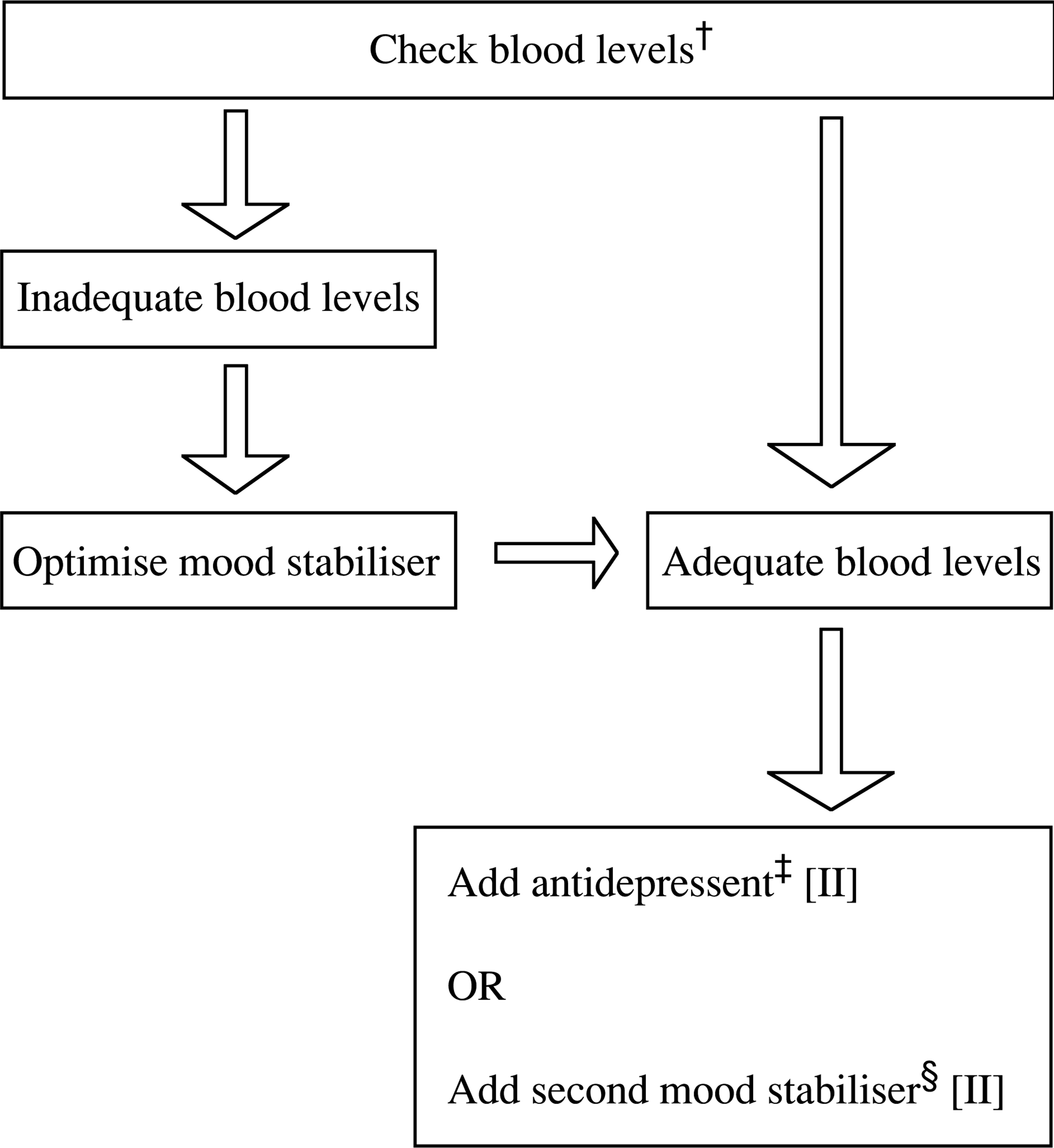
Breakthrough depressive episode on single mood stabiliser. †See ‘Therapeutic drug monitoring’ section. ‡Combining an antidepressant with a mood stabiliser may enhance and speed up antidepressant efficacy [102], [119]. §Data is only available for the combination of lithium and carbamazepine [141]. However, lamotrigine is another choice when considering the addition of a second mood stabiliser. The dose of lamotrigine should be halved when combined with valproate.
Once the bipolar depressive episode has resolved, antidepressants are to be tapered to minimize switching into mania, and the mood stabiliser continued. This is usually achieved between 2 weeks and a few months [204–206].
Overall, adding an antidepressant is probably the preferred strategy to treat breakthrough depression during monotherapy. Alternatively, a second mood stabiliser can be tried, particularly if combined therapy is likely to be long-term.
Choosing an antidepressant
SSRIs are the antidepressants of choice [102], [109], [119] because of their proven benefit and the low risk of switching. Venlafaxine is a suitable alternative. MAOIs and TCAs, which are effective for bipolar depression [108], [120], [194], [205], [207], are of less value in the long term because of side-effects and the switching risk. The threshold to prescribe MAOIs is less than that in major depressive disorder, especially when anergia and melancholic features are prominent (Table 6).
Choosing an antidepressant
Choosing a mood stabiliser
Choice is dictated as much by patient features and clinician preference as by clinical indication. The evidence favouring lithium and lamotrigine is noteworthy [43], [106]. Combining olanzapine and fluoxetine is another option [125] (Table 7)
Choosing a mood stabiliser
Failure to respond
Compliance and optimizing serum concentrations and dose should be carefully considered. Later prescribing is more complex, as less is known about the efficacy of drug combinations. Mood stabilisers and antidepressants can be substituted, or another mood stabiliser added. Lithium should have been trialled by this stage (Fig. 6)
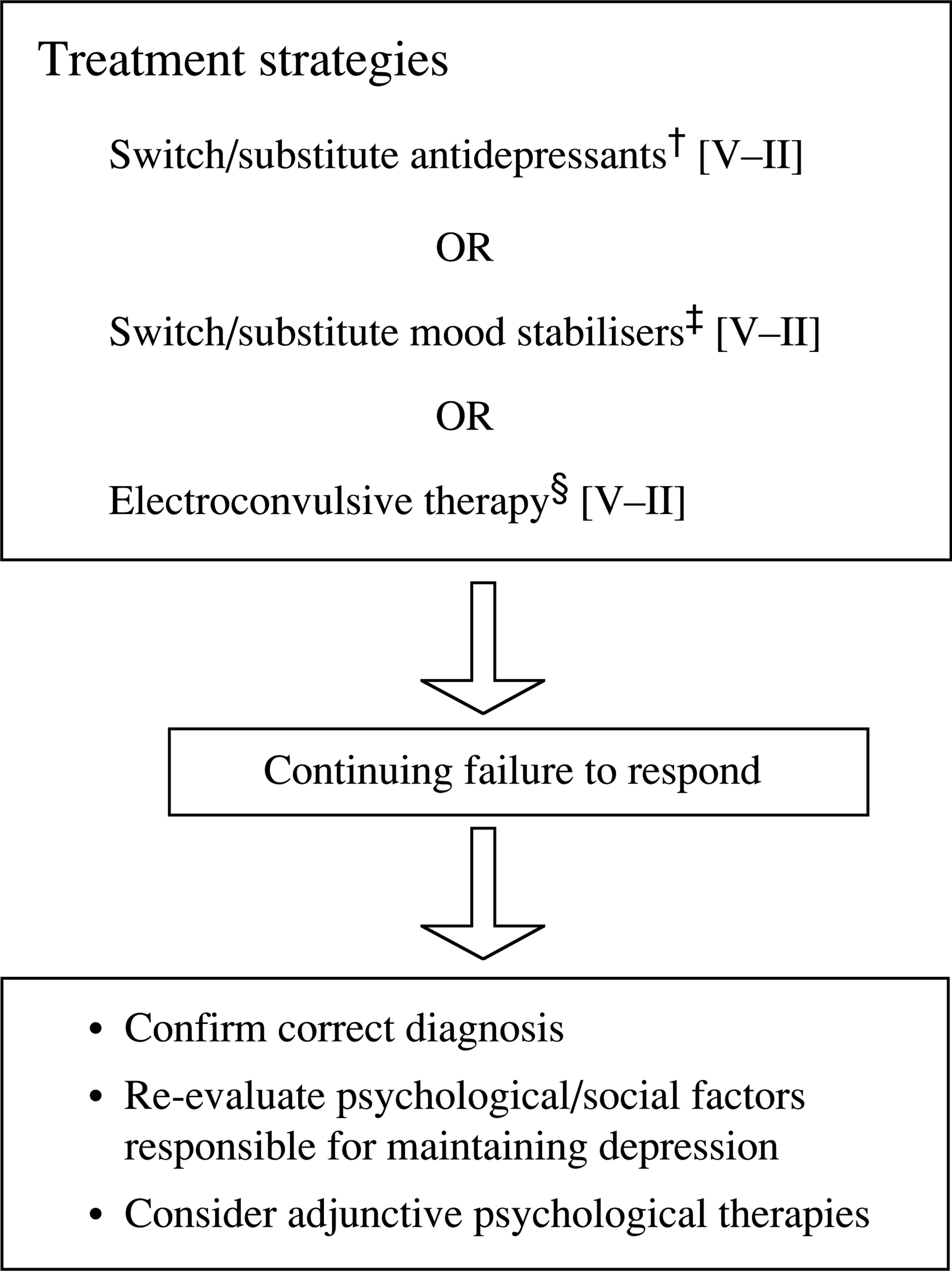
Failure to respond to treatment of bipolar depression. †No evidence for the treatment of bipolar depression. Switch between classes of antidepressants to enhance effectiveness and within class to reduce side-effects and intolerance. ‡Administration of mood stabilisers may overlap. See Table 7. §ECT should not be delayed, especially if previously effective or with psychotic symptoms [208].
Various mood stabiliser pairs can be tried and to these a range of antidepressants may be added, one at a time. However, choice of the latter is guided by the differences between antidepressant classes. If the patient remains depressed or responds only partially, the depression can legitimately be described as treatment-resistant.
With continuing failure, diagnosis and all past treatments are reviewed. Organicity may be overlooked and must be confidently excluded. Furthermore, the impact of a comorbid medical or psychiatric condition is thoroughly assessed. Consideration is also given to psychosocial factors that may be hindering recovery.
ECT is arguably the most effective treatment for both unipolar and bipolar depression. It should be tried in resistant cases, particularly if it has been effective in the past or psychotic features are evident [208]. Bipolar patients respond sooner and require a briefer course than unipolar patients [130].
Continuing treatment
Following remission of the depressive episode, it is appropriate to withdraw antidepressant treatment after 2–3 months to avoid precipitating mania or rapidcycling. However, it is necessary always to balance the need to treat bipolar depression against these two risks (Table 8).
Continuing treatment after bipolar depression
Prophylaxis of BD
Criteria for launching long-term treatment
These are specified in Table 4.
Long-term treatment: non-rapid cycling
For patients on lithium, renal function (creatinine and electrolytes) should be monitored every 3–6 months, and thyroid function (including TSH) every 6–12 months, in addition to clinical assessment. Abrupt cessation of lithium leads to relapse of mania (or, less likely, depression), often within months. Lithium should therefore be withdrawn gradually over 1–2 months.
For carbamazepine and valproate, haematological and hepatic function should be monitored every 3–6 months.
Long-term treatment: rapid-cycling
Psychological interventions
Psychological treatments should be considered, in conjunction with medications. Long-term psychological difficulties in BD patients include not wishing to accept the diagnosis, negative effects on self-concept and selfesteem, and stigma. Random controlled trials of psychoeducation therapies (individual, family and group) show their utility in reducing relapse (particularly depression). Cognitive behaviour therapy is also effective. Interpersonal and social rhythm therapy (IPSRT) has been recommended and the outcome of controlled trials is awaited (Table 11).
Psychological interventions in bipolar disorder
Failure to respond
Failure to respond to prophylaxis in bipolar disorder: non-rapid cycling
Failure to respond to prophylaxis in bipolar disorder: rapid cycling
Special groups
In pregnancy and breastfeeding
Pregnancy
The ‘floppy baby’ syndrome (hypotonia, cyanosis and poor suckling) may result from lithium toxicity [213]. Lithium has also been reported to affect neonatal thyroid function. A 5-year follow-up of 60 children exposed to lithium in the second and third trimesters found no increase in developmental anomalies [214]. This is supported by a follow-up of 21 infants, in whom developmental milestones were normal [215].
Breastfeeding
The post-partum period is emotionally challenging for any woman, but for those with BD the risk of mania, depression and psychosis is particularly high. Reich and Winokur [221] examined the frequency of post-partum episodes in 20 bipolar mothers and 29 first-degree women relatives, and found the very high rates of 30% in the patients and 20% in the relatives. Piontek et al. [222] reported that episodes of depression in BD women can reach 70% post-partum.
Conclusions
Substantial evidence from RCTs and/or meta-analyses are available for a range of treatments for BD. Acceleration of progress since the early 1990s has been marked. The advent of these therapies, both drug and psychological, means that the majority of patients with this recurrent and disabling condition may be effectively treated.
Footnotes
Acknowledgements
This team included clinicians and researchers from psychiatry, clinical psychology and general practice. We also drew upon the experience of patients and their families. Philip Mitchell, Gin Malhi, Bernette Redwood and Jillian Ball (School of Psychiatry, Mood Disorders Unit, Prince of Wales Hospital, Sydney).
Meg Smith (School of Applied Social and Human Sciences, University of Western Sydney); Bronwyn Gould (GP); Peter Joyce (Department of Psychological Medicine, University of Otago, Christchurch); Ken Kirkby (Department of Psychiatry, University of Tasmania, Hobart).
We thank Jean Dunn and Sidney Bloch for their editorial comments.
Statement of competing interests
Philip Mitchell has received no financial support from the pharmaceutical industry for the last three years, and has not served on any pharmaceutical industry advisory board over that period of time. Gin Malhi has served on advisory boards for GSK, Wyeth, and Eli Lilly, and received honoraria from Pfizer, Astra Zeneca, Oganon and Lundbeck in the last three years. (GM has also been the recipient of an Eli Lilly Young Investigator Bipolar Research Award). Jillian Ball and Bernette Redwood have never received any financial support from the pharmaceutical industry.
